2009 Recommended Adult Immunization Schedule - Omnia Education
2009 Recommended Adult Immunization Schedule - Omnia Education
2009 Recommended Adult Immunization Schedule - Omnia Education
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
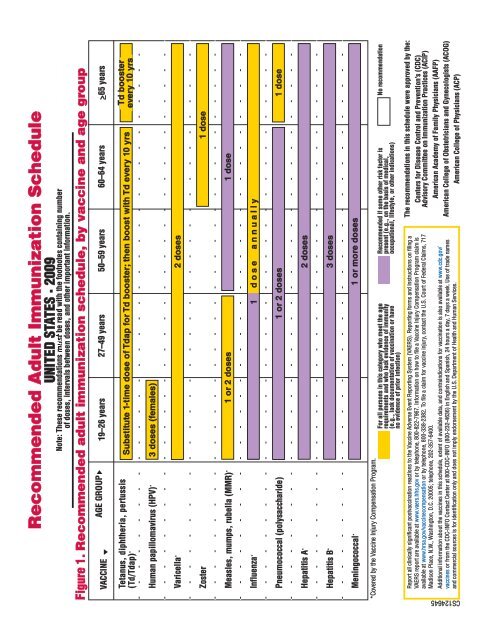
λ½±³³»²¼»¼ ß¼«´¬ ׳³«²·¦¿¬·±² ͽ¸»¼«´»<br />
ËÒ×ÌÛÜ ÍÌßÌÛÍ i îððç<br />
Ò±¬»æ ̸»» ®»½±³³»²¼¿¬·±² ¾» ®»¿¼ ©·¬¸ ¬¸» º±±¬²±¬» ½±²¬¿·²·²¹ ²«³¾»®<br />
±º ¼±»ô ·²¬»®ª¿´ ¾»¬©»»² ¼±»ô ¿²¼ ±¬¸»® ·³°±®¬¿²¬ ·²º±®³¿¬·±²ò<br />
Ú·¹«®» ïò λ½±³³»²¼»¼ ¿¼«´¬ ·³³«²·¦¿¬·±² ½¸»¼«´»ô ¾§ ª¿½½·²» ¿²¼ ¿¹» ¹®±«°<br />
ÊßÝÝ×ÒÛ<br />
ßÙÛ ÙÎÑËÐ ïçŠîê §»¿® îéŠìç §»¿® ëðŠëç §»¿® êðŠêì §»¿® âêë §»¿®<br />
Ì» ¬ ¿ ² « ô ¼ · ° ¸ ¬ ¸ » ® · ¿ ô ° » ® ¬ « · <br />
ø Ì ¼ ñ Ì ¼ ¿ ° ÷ ö<br />
Í « ¾ ¬ · ¬« ¬» ïó ¬ · ³ » ¼ ± » ± º Ì ¼ ¿ ° º ± ® Ì ¼ ¾ ± ± ¬ » ® å ¬ ¸ » ² ¾ ± ± ¬ © · ¬ ¸ Ì ¼ » ª» ® § ïð § ®<br />
Ì ¼ ¾ ± ± ¬ » ®<br />
» ª» ® § ïð § ®<br />
Ø « ³¿ ² ° ¿ ° ·´´±³ ¿ ª·® « øØ Ð Ê÷ ö<br />
í ¼ ± » ø º » ³ ¿ ´ » ÷<br />
Ê¿ ®· ½»´ ´¿ ö î ¼ ± » <br />
Æ ± ¬» ®<br />
ï ¼ ± »<br />
Ó» ¿ ´» ô ³«³° ô ® « ¾ » ´ ´¿ ø ÓÓÎ ÷ ö<br />
ï ± ® î ¼ ± » ï ¼ ± »<br />
ײº ´« »² ¦¿ ö<br />
ï ¼ ± » ¿ ² ² « ¿ ´ ´ §<br />
Ð ²» «³±½ ±½½ ¿´ ø ° ± ´§ ¿ ½ ½¸ ¿ ® ·¼ » ÷<br />
Ø » °¿¬· ¬· ß ö<br />
Ø » °¿¬· ¬· Þ ö<br />
ï ± ® î ¼ ± » î ¼ ± » <br />
í ¼ ± » <br />
ï ¼ ± »<br />
Ó» ²· ² ¹±½± ½ ½ ¿ ´ ö<br />
ï ± ® ³ ± ®» ¼ ± » <br />
ö Ý ± ª » ® » ¼ ¾ § ¬¸ » Ê ¿ ½ ½ ·² » ײ « ® § Ý ± ³ ° » ² ¿ ¬·± ² Ð ® ± ¹ ® ¿ ³ò<br />
Ú ± ® ¿ ´´ ° » ® ± ² ·² ¬¸ · ½ ¿ ¬» ¹ ± ® § © ¸ ± ³ » » ¬ ¬¸ » ¿ ¹ »<br />
® » ¯ « ·® » ³ » ² ¬ ¿ ² ¼ © ¸ ± ´¿ ½ µ » ª ·¼ » ² ½ » ± º ·³ ³ « ² ·¬§<br />
ø » ò ¹ ò ô ´¿ ½ µ ¼ ± ½ « ³ » ² ¬¿ ¬·± ² ± º ª ¿ ½ ½ ·² ¿ ¬·± ² ± ® ¸ ¿ ª »<br />
² ± » ª ·¼ » ² ½ » ± º ° ® ·± ® ·² º» ½ ¬·± ² ÷<br />
Î » ° ± ®¬ ¿ ´ ½ ´·² ·½ ¿ ´´§ ·¹ ² ·º·½ ¿ ² ¬ ° ± ¬ª ¿ ½ ½ ·² ¿ ¬·± ² ®» ¿ ½ ¬·± ² ¬± ¬¸ »Ê ¿ ½ ½ ·² »ß ¼ ª » ® » Û ª » ² ¬Î » ° ± ®¬·² ¹Í § ¬» ³øÊ ß Û Î Í ÷ò Î » ° ± ®¬·² ¹ º± ®³ ¿ ² ¼ ·² ¬®« ½ ¬·± ² ± ² º·´·² ¹ ¿<br />
Ê ß Û Î Í ®» ° ± ®¬ ¿ ®» ¿ ª ¿ ·´¿ ¾ ´» ¿ ¬ © © © òª ¿ » ® ò¸ ¸ ò¹ ± ª ± ®¾ § ¬» ´» ° ¸ ± ² » ôè ð ð óè î î óé ç ê é ò ײ º± ®³ ¿ ¬·± ² ± ²¸ ± © ¬± º·´» ¿ Ê ¿ ½ ½ ·² » ײ « ®§ Ý ± ³ ° » ² ¿ ¬·± ²Ð ®± ¹ ®¿ ³½ ´¿ ·³ ·<br />
¿ ª ¿ ·´¿ ¾ ´» ¿ ¬ © © © ò¸ ® ¿ ò¹ ± ª ñª ¿ ½ ½ ·² » ½ ± ³ ° » ² ¿ ¬·± ² ± ®¾ § ¬» ´» ° ¸ ± ² » ôè ð ð óí í è óî í è î ò̱ º·´» ¿ ½ ´¿ ·³ º± ® ª ¿ ½ ½ ·² » ·² « ® § ô ½ ± ² ¬¿ ½ ¬ ¬¸ »Ë òÍ òÝ ± « ®¬ ± ºÚ » ¼ » ®¿ ´Ý ´¿ ·³ ô é ï é<br />
Ó ¿ ¼ · ± ²Ð ´¿ ½ » ô Ò òÉòô É ¿ ¸ ·² ¹ ¬± ² ô Ü òÝ òî ð ð ð ë å ¬» ´» ° ¸ ± ² » ôî ð î óí ë é óê ì ð ð ò<br />
ß ¼ ¼ ·¬·± ² ¿ ´·² º± ®³ ¿ ¬·± ² ¿ ¾ ± « ¬ ¬¸ » ª ¿ ½ ½ ·² » ·² ¬¸ · ½ ¸ » ¼ « ´» ô » ¨ ¬» ² ¬ ± º ¿ ª ¿ ·´¿ ¾ ´»¼ ¿ ¬¿ ô ¿ ² ¼½ ± ² ¬®¿ ·² ¼ ·½ ¿ ¬·± ² º± ® ª ¿ ½ ½ ·² ¿ ¬·± ² · ¿ ´ ± ¿ ª ¿ ·´¿ ¾ ´» ¿ ¬ © © © ò½ ¼ ½ ò¹ ± ª ñ<br />
ª ¿ ½ ½ ·² » ± ® º®± ³ ¬¸ »Ý Ü Ý ó×Ò Ú ÑÝ ± ² ¬¿ ½ ¬Ý » ² ¬» ® ¿ ¬è ð ð óÝ Ü Ý ó×Ò Ú Ñ øè ð ð óî í î óì ê í ê ÷ ·²Û ² ¹ ´· ¸ ¿ ² ¼ Í ° ¿ ² · ¸ ô î ì¸ ± « ® ¿¼ ¿ § ô é¼ ¿ § ¿ © » » µ ò Ë » ± º ¬®¿ ¼ » ² ¿ ³ » <br />
¿ ² ¼½ ± ³ ³ » ®½ ·¿ ´ ± « ®½ » · º± ® ·¼ » ² ¬·º·½ ¿ ¬·± ² ± ² ´§ ¿ ² ¼¼ ± » ² ± ¬ ·³ ° ´§ » ² ¼ ± ® » ³ » ² ¬ ¾ § ¬¸ »Ë òÍ ò Ü » ° ¿ ®¬³ » ² ¬ ± ºØ » ¿ ´¬¸ ¿ ² ¼ Ø « ³ ¿ ² Í » ®ª ·½ » ò<br />
Î » ½ ± ³ ³ » ² ¼ » ¼ ·º ± ³ » ± ¬¸ » ® ® · µ º¿ ½ ¬± ® ·<br />
° ® » » ² ¬ ø » ò ¹ ò ô ± ² ¬¸ » ¾ ¿ · ± º ³ » ¼ ·½ ¿ ´ô<br />
± ½ ½ « ° ¿ ¬·± ² ¿ ´ô ´·º» ¬§ ´» ô ± ® ± ¬¸ » ® ·² ¼ ·½ ¿ ¬·± ² ÷<br />
Ò ± ® » ½ ± ³ ³ » ² ¼ ¿ ¬·± ²<br />
Ì ¸ » ® » ½ ± ³ ³ » ² ¼ ¿ ¬·± ² ·² ¬¸ · ½ ¸ » ¼ « ´» © » ® » ¿ ° ° ® ± ª » ¼ ¾ § ¬¸ » æ<br />
Ý » ² ¬» ® º± ® Ü · » ¿ » Ý ± ² ¬® ± ´ ¿ ² ¼ Ð ® » ª » ² ¬·± ² Ž ø Ý Ü Ý ÷<br />
ß ¼ ª · ± ® § Ý ± ³ ³ ·¬¬» » ± ² ׳ ³ « ² ·¦ ¿ ¬·± ² Ð ® ¿ ½ ¬·½ » ø ß Ý ×Ð ÷<br />
ß ³ » ® ·½ ¿ ² ß ½ ¿ ¼ » ³ § ± º Ú ¿ ³ ·´§ Ð ¸ § ·½ ·¿ ² ø ß ß Ú Ð ÷<br />
ß ³ » ® ·½ ¿ ² Ý ± ´´» ¹ » ± º Ñ ¾ ¬» ¬® ·½ ·¿ ² ¿ ² ¼ Ù § ² » ½ ± ´± ¹ · ¬ ø ß Ý Ñ Ù ÷<br />
ß ³ » ® ·½ ¿ ² Ý ± ´´» ¹ » ± º Ð ¸ § ·½ ·¿ ² ø ß Ý Ð ÷<br />
Ý Í ï î ì ê ì ë
Ú·¹«®» îò Ê¿½½·²» ¬¸¿¬ ³·¹¸¬ ¾» ·²¼·½¿¬»¼ º±® ¿¼«´¬ ¾¿»¼ ±² ³»¼·½¿´ ¿²¼<br />
±¬¸»® ·²¼·½¿¬·±²<br />
ÊßÝÝ×ÒÛ<br />
×ÒÜ×ÝßÌ×ÑÒ<br />
Ю»¹²¿²½§<br />
׳³«²±ó<br />
½±³°®±³··²¹<br />
½±²¼·¬·±²ø»¨½´«¼·²¹<br />
¸«³¿²<br />
·³³«²±¼»º·½·»²½§<br />
ª·®« ÅØ×ÊÃ÷<br />
Ø×Ê ·²º»½¬·±²<br />
ÝÜìõ Ì ´§³°¸±ó<br />
½§¬» ½±«²¬<br />
äîðð<br />
½»´´ñkÔ<br />
âîðð<br />
½»´´ñkÔ<br />
Ü·¿¾»¬»ô<br />
¸»¿®¬ ¼·»¿»ô<br />
½¸®±²·½<br />
´«²¹ ¼·»¿»ô<br />
½¸®±²·½<br />
¿´½±¸±´·³<br />
ß°´»²·¿<br />
ø·²½´«¼·²¹<br />
»´»½¬·ª»<br />
°´»²»½¬±³§<br />
¿²¼ ¬»®³·²¿´<br />
½±³°´»³»²¬<br />
½±³°±²»²¬<br />
¼»º·½·»²½·»÷<br />
ݸ®±²·½ ´·ª»®<br />
¼·»¿»<br />
Õ·¼²»§ º¿·´«®»ô<br />
»²¼ó¬¿¹» ®»²¿´<br />
¼·»¿»ô<br />
®»½»·°¬ ±º<br />
¸»³±¼·¿´§·<br />
Ø»¿´¬¸ó½¿®»<br />
°»®±²²»´<br />
Ì»¬¿²«ô ¼·°¸¬¸»®·¿ô °»®¬«·<br />
ø̼ñ̼¿°÷ ö<br />
̼<br />
Í«¾¬·¬«¬» ïó¬·³» ¼±» ±º ̼¿° º±® ̼ ¾±±¬»®å ¬¸»² ¾±±¬ ©·¬¸ ̼ »ª»®§ ï𠧮<br />
Ø«³¿² °¿°·´´±³¿ª·®« øØÐÊ÷ ö<br />
í ¼±» º±® º»³¿´» ¬¸®±«¹¸ ¿¹» îê §®<br />
Ê¿®·½»´´¿ ö Ʊ¬»®<br />
ݱ²¬®¿·²¼·½¿¬»¼ ݱ²¬®¿·²¼·½¿¬»¼<br />
î ¼±»<br />
ï ¼±»<br />
Ó»¿´»ô ³«³°ô ®«¾»´´¿ øÓÓÎ÷ ö ݱ²¬®¿·²¼·½¿¬»¼ ï ±® î ¼±»<br />
ײº´«»²¦¿ ö<br />
в»«³±½±½½¿´ ø°±´§¿½½¸¿®·¼»÷<br />
Ø»°¿¬·¬· ß ö<br />
Ø»°¿¬·¬· Þ ö<br />
Ó»²·²¹±½±½½¿´ö<br />
ï ¼±» Ì×Ê ¿²²«¿´´§<br />
ï ±® î ¼±»<br />
î ¼±»<br />
í ¼±»<br />
ï ±® ³±®» ¼±»<br />
Ú±® ¿´´ °»®±² ·² ¬¸· ½¿¬»¹±®§ ©¸± ³»»¬ ¬¸» ¿¹»<br />
öݱª»®»¼ ¾§ ¬¸» Ê¿½½·²» ײ«®§ ݱ³°»²¿¬·±² Ю±¹®¿³ò<br />
®»¯«·®»³»²¬ ¿²¼ ©¸± ´¿½µ »ª·¼»²½» ±º ·³³«²·¬§<br />
ø»ò¹òô ´¿½µ ¼±½«³»²¬¿¬·±² ±º ª¿½½·²¿¬·±² ±® ¸¿ª»<br />
²± »ª·¼»²½» ±º °®·±® ·²º»½¬·±²÷<br />
λ½±³³»²¼»¼ ·º ±³» ±¬¸»® ®·µ º¿½¬±® ·<br />
°®»»²¬ ø»ò¹òô ±² ¬¸» ¾¿· ±º ³»¼·½¿´ô<br />
±½½«°¿¬·±²¿´ô ´·º»¬§´»ô ±® ±¬¸»® ·²¼·½¿¬·±²÷<br />
ï ¼±» Ì×Ê<br />
±® Ôß×Ê<br />
¿²²«¿´´§<br />
Ò± ®»½±³³»²¼¿¬·±²<br />
̸»» ½¸»¼«´» ·²¼·½¿¬» ¬¸» ®»½±³³»²¼»¼ ¿¹» ¹®±«° ¿²¼ ³»¼·½¿´ ·²¼·½¿¬·±² º±® ©¸·½¸ ¿¼³·²·¬®¿¬·±² ±º ½«®®»²¬´§<br />
´·½»²»¼ ª¿½½·²» · ½±³³±²´§ ·²¼·½¿¬»¼ º±® ¿¼«´¬ ¿¹» ïç §»¿® ¿²¼ ±´¼»®ô ¿ ±º Ö¿²«¿®§ ïô îððçò Ô·½»²»¼ ½±³¾·²¿¬·±²<br />
ª¿½½·²» ³¿§ ¾» «»¼ ©¸»²»ª»® ¿²§ ½±³°±²»²¬ ±º ¬¸» ½±³¾·²¿¬·±² ¿®» ·²¼·½¿¬»¼ ¿²¼ ©¸»² ¬¸» ª¿½½·²»Ž ±¬¸»® ½±³°±²»²¬<br />
¿®» ²±¬ ½±²¬®¿·²¼·½¿¬»¼ò Ú±® ¼»¬¿·´»¼ ®»½±³³»²¼¿¬·±² ±² ¿´´ ª¿½½·²»ô ·²½´«¼·²¹ ¬¸±» «»¼ °®·³¿®·´§ º±® ¬®¿ª»´»® ±® ¬¸¿¬<br />
¿®» ·«»¼ ¼«®·²¹ ¬¸» §»¿®ô ½±²«´¬ ¬¸» ³¿²«º¿½¬«®»®Ž °¿½µ¿¹» ·²»®¬ ¿²¼ ¬¸» ½±³°´»¬» ¬¿¬»³»²¬ º®±³ ¬¸» ß¼ª·±®§<br />
ݱ³³·¬¬»» ±² ׳³«²·¦¿¬·±² Ю¿½¬·½» ø©©©ò½¼½ò¹±ªñª¿½½·²»ñ°«¾ñ¿½·°ó´·¬ò¸¬³÷ò<br />
Ü»°¿®¬³»²¬ ±º Ø»¿´¬¸ ¿²¼ Ø«³¿² Í»®ª·½»<br />
Ý»²¬»® º±® Ü·»¿» ݱ²¬®±´ ¿²¼ Ю»ª»²¬·±²
Footnotes<br />
<strong>Recommended</strong> <strong>Adult</strong> <strong>Immunization</strong> <strong>Schedule</strong> – United States • <strong>2009</strong><br />
For complete statements by the Advisory Committee on <strong>Immunization</strong> Practices (ACIP), visit www.cdc.gov/vaccines/pubs/ACIP-list.htm.<br />
1. Tetanus, diphtheria, and acellular pertussis (Td/Tdap) vaccination<br />
Tdap should replace a single dose of Td for adults aged 19 through 64 years who have not received a dose of Tdap previously<br />
<strong>Adult</strong>s with uncertain or incomplete history of primary vaccination series with tetanus and diphtheria toxoid--containing vaccines should begin or complete a primary vaccination series. A<br />
primary series for adults is 3 doses of tetanus and diphtheria toxoid--containing vaccines; administer the first 2 doses at least 4 weeks apart and the third dose 6--12 months after the<br />
second. However, Tdap can substitute for any one of the doses of Td in the 3-dose primary series. The booster dose of tetanus and diphtheria toxoid--containing vaccine should be<br />
administered to adults who have completed a primary series and if the last vaccination was received 10 or more years previously. Tdap or Td vaccine may be used, as indicated.<br />
If a woman is pregnant and received the last Td vaccination 10 or more years previously, administer Td during the second or third trimester. If the woman received the last Td<br />
vaccination less than 10 years previously, administer Tdap during the immediate postpartum period. A dose of Tdap is recommended for postpartum women, close contacts of infants<br />
aged less than 12 months, and all health-care personnel with direct patient contact if they have not previously received Tdap. An interval as short as 2 years from the last Td is<br />
suggested; shorter intervals can be used. Td may be deferred during pregnancy and Tdap substituted in the immediate postpartum period, or Tdap may be administered instead of Td to<br />
a pregnant woman after an informed discussion with the woman.<br />
Consult the ACIP statement for recommendations for administering Td as prophylaxis in wound management.<br />
2. Human papillomavirus (HPV) vaccination<br />
HPV vaccination is recommended for all females aged 11 through 26 years (and may begin at age 9 years) who have not completed the vaccine series. History of genital warts,<br />
abnormal Papanicolaou test, or positive HPV DNA test is not evidence of prior infection with all vaccine HPV types; HPV vaccination is recommended for persons with such histories.<br />
Ideally, vaccine should be administered before potential exposure to HPV through sexual activity; however, females who are sexually active should still be vaccinated consistent with<br />
age-based recommendations. Sexually active females who have not been infected with any of the four HPV vaccine types receive the full benefit of the vaccination. Vaccination is less<br />
beneficial for females who have already been infected with one or more of the HPV vaccine types.<br />
A complete series consists of 3 doses. The second dose should be administered 2 months after the first dose; the third dose should be administered 6 months after the first dose.<br />
HPV vaccination is not specifically recommended for females with the medical indications described in Figure 2, "Vaccines that might be indicated for adults based on medical and other<br />
indications." Because HPV vaccine is not a live-virus vaccine, it may be administered to persons with the medical indications described in Figure 2. However, the immune response and<br />
vaccine efficacy might be less for persons with the medical indications described in Figure 2 than in persons who do not have the medical indications described or who are<br />
immunocompetent. Health-care personnel are not at increased risk because of occupational exposure, and should be vaccinated consistent with age-based recommendations.<br />
3. Varicella vaccination<br />
All adults without evidence of immunity to varicella should receive 2 doses of single-antigen varicella vaccine if not previously vaccinated or the second dose if they have received only<br />
one dose, unless they have a medical contraindication. Special consideration should be given to those who 1) have close contact with persons at high risk for severe disease (e.g.,<br />
health-care personnel and family contacts of persons with immunocompromising conditions) or 2) are at high risk for exposure or transmission (e.g., teachers; child care employees;<br />
residents and staff members of institutional settings, including correctional institutions; college students; military personnel; adolescents and adults living in households with children;<br />
nonpregnant women of childbearing age; and international travelers).<br />
Evidence of immunity to varicella in adults includes any of the following: 1) documentation of 2 doses of varicella vaccine at least 4 weeks apart; 2) U.S.-born before 1980 (although for<br />
health-care personnel and pregnant women, birth before 1980 should not be considered evidence of immunity); 3) history of varicella based on diagnosis or verification of varicella by a<br />
health-care provider (for a patient reporting a history of or presenting with an atypical case, a mild case, or both, health-care providers should seek either an epidemiologic link to a<br />
typical varicella case or to a laboratory-confirmed case or evidence of laboratory confirmation, if it was performed at the time of acute disease); 4) history of herpes zoster based on<br />
health-care provider diagnosis or verification of herpes zoster by a health-care provider; or 5) laboratory evidence of immunity or laboratory confirmation of disease.<br />
Pregnant women should be assessed for evidence of varicella immunity. Women who do not have evidence of immunity should receive the first dose of varicella vaccine upon<br />
completion or termination of pregnancy and before discharge from the health-care facility. The second dose should be administered 4--8 weeks after the first dose.<br />
4. Herpes zoster vaccination<br />
A single dose of zoster vaccine is recommended for adults aged 60 years and older regardless of whether they report a prior episode of herpes zoster. Persons with chronic medical<br />
conditions may be vaccinated unless their condition constitutes a contraindication.<br />
5. Measles, mumps, rubella (MMR) vaccination<br />
Measles component: <strong>Adult</strong>s born before 1957 generally are considered immune to measles. <strong>Adult</strong>s born during or after 1957 should receive 1 or more doses of MMR unless they have a<br />
medical contraindication, documentation of 1 or more doses, history of measles based on health-care provider diagnosis, or laboratory evidence of immunity.<br />
A second dose of MMR is recommended for adults who 1) have been recently exposed to measles or are in an outbreak setting; 2) have been vaccinated previously with killed measles<br />
vaccine; 3) have been vaccinated with an unknown type of measles vaccine during 1963--1967; 4) are students in postsecondary educational institutions; 5) work in a health-care facility;
or 6) plan to travel internationally.<br />
Mumps component: <strong>Adult</strong>s born before 1957 generally are considered immune to mumps. <strong>Adult</strong>s born during or after 1957 should receive 1 dose of MMR unless they have a medical<br />
contraindication, history of mumps based on health-care provider diagnosis, or laboratory evidence of immunity.<br />
A second dose of MMR is recommended for adults who 1) live in a community experiencing a mumps outbreak and are in an affected age group; 2) are students in postsecondary<br />
educational institutions; 3) work in a health-care facility; or 4) plan to travel internationally. For unvaccinated health-care personnel born before 1957 who do not have other evidence of<br />
mumps immunity, administering 1 dose on a routine basis should be considered and administering a second dose during an outbreak should be strongly considered.<br />
Rubella component: 1 dose of MMR vaccine is recommended for women whose rubella vaccination history is unreliable or who lack laboratory evidence of immunity. For women of<br />
childbearing age, regardless of birth year, rubella immunity should be determined and women should be counseled regarding congenital rubella syndrome. Women who do not have<br />
evidence of immunity should receive MMR vaccine upon completion or termination of pregnancy and before discharge from the health-care facility.<br />
6. Influenza vaccination<br />
Medical indications: Chronic disorders of the cardiovascular or pulmonary systems, including asthma; chronic metabolic diseases, including diabetes mellitus, renal or hepatic<br />
dysfunction, hemoglobinopathies, or immunocompromising conditions (including immunocompromising conditions caused by medications or human immunodeficiency virus [HIV]); any<br />
condition that compromises respiratory function or the handling of respiratory secretions or that can increase the risk of aspiration (e.g., cognitive dysfunction, spinal cord injury, or<br />
seizure disorder or other neuromuscular disorder); and pregnancy during the influenza season. No data exist on the risk for severe or complicated influenza disease among persons with<br />
asplenia; however, influenza is a risk factor for secondary bacterial infections that can cause severe disease among persons with asplenia.<br />
Occupational indications: All health-care personnel, including those employed by long-term care and assisted-living facilities, and caregivers of children less than 5 years old.<br />
Other indications: Residents of nursing homes and other long-term care and assisted-living facilities; persons likely to transmit influenza to persons at high risk (e.g., in-home household<br />
contacts and caregivers of children aged less than 5 years old, persons 65 years old and older and persons of all ages with high-risk condition[s]); and anyone who would like to<br />
decrease their risk of getting influenza. Healthy, nonpregnant adults aged less than 50 years without high-risk medical conditions who are not contacts of severely immunocompromised<br />
persons in special care units can receive either intranasally administered live, attenuated influenza vaccine (FluMist ® ) or inactivated vaccine. Other persons should receive the<br />
inactivated vaccine.<br />
7. Pneumococcal polysaccharide (PPSV) vaccination<br />
Medical indications: Chronic lung disease (including asthma); chronic cardiovascular diseases; diabetes mellitus; chronic liver diseases, cirrhosis; chronic alcoholism, chronic renal<br />
failure or nephrotic syndrome; functional or anatomic asplenia (e.g., sickle cell disease or splenectomy [if elective splenectomy is planned, vaccinate at least 2 weeks before surgery]);<br />
immunocompromising conditions; and cochlear implants and cerebrospinal fluid leaks. Vaccinate as close to HIV diagnosis as possible.<br />
Other indications: Residents of nursing homes or other long-term care facilities and persons who smoke cigarettes. Routine use of PPSV is not recommended for Alaska Native or<br />
American Indian persons younger than 65 years unless they have underlying medical conditions that are PPSV indications. However, public health authorities may consider<br />
recommending PPSV for Alaska Natives and American Indians aged 50 through 64 years who are living in areas in which the risk of invasive pneumococcal disease is increased.<br />
8. Revaccination with PPSV<br />
One-time revaccination after 5 years is recommended for persons with chronic renal failure or nephrotic syndrome; functional or anatomic asplenia (e.g., sickle cell disease or<br />
splenectomy); and for persons with immunocompromising conditions. For persons aged 65 years and older, one-time revaccination if they were vaccinated 5 or more years previously<br />
and were aged less than 65 years at the time of primary vaccination.<br />
9. Hepatitis A vaccination<br />
Medical indications: Persons with chronic liver disease and persons who receive clotting factor concentrates.<br />
Behavioral indications: Men who have sex with men and persons who use illegal drugs.<br />
Occupational indications: Persons working with hepatitis A virus (HAV)--infected primates or with HAV in a research laboratory setting.<br />
Other indications: Persons traveling to or working in countries that have high or intermediate endemicity of hepatitis A (a list of countries is available at<br />
http://wwwn.cdc.gov/travel/contentdiseases.aspx) and any person seeking protection from HAV infection.<br />
Single-antigen vaccine formulations should be administered in a 2-dose schedule at either 0 and 6--12 months (Havrix ® ), or 0 and 6--18 months (Vaqta ® ). If the combined hepatitis A and<br />
hepatitis B vaccine (Twinrix ® ) is used, administer 3 doses at 0, 1, and 6 months; alternatively, a 4-dose schedule, administered on days 0, 7, and 21 to 30 followed by a booster dose at<br />
month 12 may be used.<br />
10. Hepatitis B vaccination<br />
Medical indications: Persons with end-stage renal disease, including patients receiving hemodialysis; persons with HIV infection; and persons with chronic liver disease.<br />
Occupational indications: Health-care personnel and public-safety workers who are exposed to blood or other potentially infectious body fluids.<br />
Behavioral indications: Sexually active persons who are not in a long-term, mutually monogamous relationship (e.g., persons with more than 1 sex partner during the previous 6 months);<br />
persons seeking evaluation or treatment for a sexually transmitted disease (STD);current or recent injection-drug users; and men who have sex with men.<br />
Other indications: Household contacts and sex partners of persons with chronic hepatitis B virus (HBV) infection; clients and staff members of institutions for persons with developmental
disabilities; international travelers to countries with high or intermediate prevalence of chronic HBV infection (a list of countries is available at<br />
http://wwwn.cdc.gov/travel/contentdiseases.aspx); and any adult seeking protection from HBV infection.<br />
Hepatitis B vaccination is recommended for all adults in the following settings: STD treatment facilities; HIV testing and treatment facilities; facilities providing drug-abuse treatment and<br />
prevention services; health-care settings targeting services to injection-drug users or men who have sex with men; correctional facilities; end-stage renal disease programs and facilities<br />
for chronic hemodialysis patients; and institutions and nonresidential daycare facilities for persons with developmental disabilities.<br />
If the combined hepatitis A and hepatitis B vaccine (Twinrix ® ) is used, administer 3 doses at 0, 1, and 6 months; alternatively, a 4-dose schedule, administered on days 0, 7, and 21 to 30<br />
followed by a booster dose at month 12 may be used.<br />
Special formulation indications: For adult patients receiving hemodialysis or with other immunocompromising conditions, 1 dose of 40 µg/mL (Recombivax HB ® ) administered on a 3-<br />
dose schedule or 2 doses of 20 µg/mL (Engerix-B ® ) administered simultaneously on a 4-dose schedule at 0,1, 2 and 6 months.<br />
11. Meningococcal vaccination<br />
Medical indications: <strong>Adult</strong>s with anatomic or functional asplenia, or terminal complement component deficiencies.<br />
Other indications: First-year college students living in dormitories; microbiologists routinely exposed to isolates of Neisseria meningitidis; military recruits; and persons who travel to or<br />
live in countries in which meningococcal disease is hyperendemic or epidemic (e.g., the "meningitis belt" of sub-Saharan Africa during the dry season [December--June]), particularly if<br />
their contact with local populations will be prolonged. Vaccination is required by the government of Saudi Arabia for all travelers to Mecca during the annual Hajj.<br />
Meningococcal conjugate vaccine (MCV) is preferred for adults with any of the preceding indications who are aged 55 years or younger, although meningococcal polysaccharide vaccine<br />
(MPSV) is an acceptable alternative. Revaccination with MCV after 5 years might be indicated for adults previously vaccinated with MPSV who remain at increased risk for infection<br />
(e.g., persons residing in areas in which disease is epidemic).<br />
12. Selected conditions for which Haemophilus influenzae type b (Hib) vaccine may be used<br />
Hib vaccine generally is not recommended for persons aged 5 years and older. No efficacy data are available on which to base a recommendation concerning use of Hib vaccine for<br />
older children and adults. However, studies suggest good immunogenicity in patients who have sickle cell disease, leukemia, or HIV infection or who have had a splenectomy;<br />
administering 1 dose of vaccine to these patients is not contraindicated.<br />
13. Immunocompromising conditions<br />
Inactivated vaccines generally are acceptable (e.g., pneumococcal, meningococcal, and influenza [trivalent inactivated influenza vaccine]) and live vaccines generally are avoided in<br />
persons with immune deficiencies or immunocompromising conditions. Information on specific conditions is available at http://www.cdc.gov/vaccines/pubs/acip-list.htm.<br />
These schedules indicate the recommended age groups and medical indications for which administration of currently licensed vaccines is commonly indicated for adults ages 19 years<br />
and older, as of January 1, <strong>2009</strong>. Licensed combination vaccines may be used whenever any components of the combination are indicated and when the vaccine's other components<br />
are not contraindicated. For detailed recommendations on all vaccines, including those used primarily for travelers or that are issued during the year, consult the manufacturers' package<br />
inserts and the complete statements from the Advisory Committee on <strong>Immunization</strong> Practices (http://www.cdc.gov/vaccines/pubs/acip-list.htm).<br />
Report all clinically significant postvaccination reactions to the Vaccine Adverse Event Reporting System (VAERS). Reporting forms and instructions on filing a VAERS report are<br />
available at http://www.vaers.hhs.gov or by telephone, 800-822-7967.<br />
Information on how to file a Vaccine Injury Compensation Program claim is available at http://www.hrsa.gov/vaccinecompensation or by telephone, 800-338-2382. To file a claim for<br />
vaccine injury, contact the U.S. Court of Federal Claims, 717 Madison Place, N.W., Washington, D.C. 20005; telephone, 202-357-6400.<br />
Additional information about the vaccines in this schedule, extent of available data, and contraindications for vaccination is also available at http://www.cdc.gov/vaccines or from the<br />
CDC-INFO Contact Center at 800-CDC-INFO (800-232-4636) in English and Spanish, 24 hours a day, 7 days a week.<br />
Use of trade names and commercial sources is for identification only and does not imply endorsement by the U.S. Department of Health and Human Services.