- Page 1 and 2: .. '7" ! A ~S1fj[ OJ :,J.Ll'i,j ~8r
- Page 3 and 4: • _ ~ I I • : I e i • O(U , -
- Page 5 and 6: .e ABSTRk(;'r OF r.U;ui.T.'!:IiLY R
- Page 7 and 8: 6 ~ Abstract ot ~uarterly Re~ort (C
- Page 9 and 10: e 8 1. REACTOR :rtJEIS Inorganic, P
- Page 11 and 12: 10 e The pH meueuremento were carri
- Page 13 and 14: 2 - I!Illd where R is a constant at
- Page 15 and 16: we 14 suggests that a particular ag
- Page 17 and 18: e 7 changes aan be tolmd. extends a
- Page 19 and 20: - 19 The sucoess ot such studies ap
- Page 21 and 22: e 21 were Ao aDd Fo M'e the initial
- Page 23 and 24: e 23 Por constant b and h e~at10n 1
- Page 25 and 26: e .. ~ 25 / Process Chemistry s A o
- Page 27 and 28: e 27 Pre-equilibration of pbaaes be
- Page 29 and 30: e 100 e -- P- - . - -- 29 DrawlOi #
- Page 31 and 32: 0 C\J
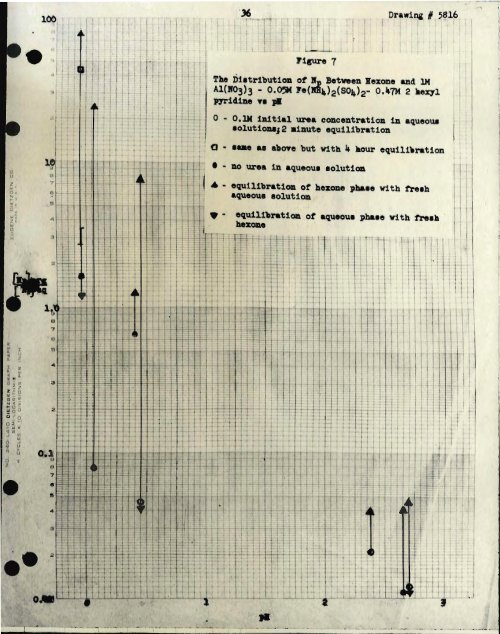
- Page 33 and 34: e 33' It vaa found that the data fo
- Page 35: , e e e " e - • 180 J'1pre 6 TI
- Page 39 and 40: e aqueous phase pH IE 2.70 39 hexon
- Page 41 and 42: e 41 present as the base itself' bu
- Page 43 and 44: e 43 TABLR I The Dete1'lB1Daticm of
- Page 45 and 46: e e 45 'i. stock fJolution of tet:t
- Page 47 and 48: e 47 TABLE III (CCilt. ) Vol: Bunl
- Page 49 and 50: e 49 .I'our coutant. rele.t1ftg SuA
- Page 51 and 52: - Hexane Phase e Time between equil
- Page 53 and 54: e 53 F1gul'e 16 Proposed E:z;plazaa
- Page 55 and 56: 55 -. . Xenon Cross Sections as a F
- Page 57 and 58: 5'-' A number of extractionrs of Xe
- Page 59 and 60: .. 59 Calibr~t.ion ~f Assal Equipme
- Page 61 and 62: 61 e Since the amount of cont;minat
- Page 63 and 64: e In the experimontH.l work d. seri
- Page 65 and 66: -. 65 Tah1e III H~lf tire of Jodine
- Page 67 and 68: . >- 12 I- > I U « « 10 I w c
- Page 69 and 70: e e e 5.0. Ore. w.i'J II 5801 FiG.
- Page 71 and 72: .. 71 Program t E-xpor1J'lental inv
- Page 73 and 74: • RecQverY or Ru 106 (rom Met!!l
- Page 75 and 76: - 75 }'rum the first portion of Tab
- Page 77 and 78: 77 - No explanation is offered for
- Page 79 and 80: e 79 Table I Inacti ve Carbon in 0w
- Page 81 and 82: - 81 An additional :.200 ml. of 18
- Page 83 and 84: e 83 Experimental Use of C14 (C" J.
- Page 85 and 86: e e 85 t',llquots of the aqueous so
- Page 87 and 88:
e e e 87 with stirring.' Two mL of
- Page 89 and 90:
e J .. StJPPQRTING CHH1ICAL RFEFAJl
- Page 91 and 92:
e be accurst ae follows }~uby ar.al
- Page 93 and 94:
e r,. ~88 V(Jlt~3 wse obl~1n(td f'o
- Page 95 and 96:
e 95 ' In these separi'ltiofls yttr
- Page 97 and 98:
e 97 At best the separation is poor
- Page 99 and 100:
99 - Lanthanum Studies The value qu
- Page 101 and 102:
· 101 - Hard Gamma R8)ys from ySS
- Page 103 and 104:
• e e Solid Chemistry ot' Rutheni
- Page 105 and 106:
• 105 Separation of the Long-Live
- Page 107 and 108:
300. .. ~ FIRST CYCLE SHOWING CONCE
- Page 109 and 110:
• 110 151 B- ~ e- (""""50 Kev) Pr
- Page 111 and 112:
FIG. IV RADIATIONS OF LONG-LIVED FI
- Page 113 and 114:
I - . ! ~ rf " ~ , 1 I ~ I , ~ I ..
- Page 115 and 116:
116 - D .. Xc> 1. Study of Baok Rea
- Page 117 and 118:
.- F1(0) Table 1 • (Continued) 11
- Page 119 and 120:
I • c::: w f-- -.J .......... ~ 2
- Page 121 and 122:
FIG. 3 . INITIAL RATE OF PEROXIDE R
- Page 123 and 124:
~ -~ I 124 -- Table 3 0 DecQnPo~it1
- Page 125 and 126:
~ - e • 100 i 126 ______ _ _ _ _
- Page 127 and 128:
128 -- Tab~e 5. Amount of n.p reaot
- Page 129 and 130:
130 • preBsur~ drop here cannot b
- Page 131 and 132:
flit 132 A S1g>lltled. Feuation of
- Page 133 and 134:
• 134 The System Uranyl Sulfate -
- Page 135 and 136:
-. e • Tc - - - -- - - - --------
- Page 137 and 138:
138 '" Cloud Chamber Studiea (Ro Li
- Page 139 and 140:
IIIIIIIIIIIIIII e 140 The disintegr
- Page 141 and 142:
142 e experiments BO that a SUII ot
- Page 143 and 144:
144 PolarogrE!Ph1c Ana.l;Ysis of Ra
- Page 145 and 146:
e 146 7 Te. 10 I \: .&oj""" 'r' .Jr
- Page 147 and 148:
148 e The effect of temperature, ti
- Page 149 and 150:
150 e in the pipette have been only
- Page 151 and 152:
152· - e The llama preparatiOD use
- Page 153 and 154:
e e • Figure 2. Kurie plot of Cb
- Page 155 and 156:
e e e Figure 4. Compton electrons a
- Page 157 and 158:
e e e Counts per minute per unit mo
- Page 159 and 160:
e 160 where the..B and L nter to th
- Page 161 and 162:
e Electron and 0 TL:--~Newman, 162
- Page 163 and 164:
e e ~. 164 In a continuation of the
- Page 165 and 166:
to •
- Page 167 and 168:
-166" e Zircon1um - neY U. S. Burea
- Page 169 and 170:
SEPARATION OF LONG-LIVED Sm AND EuA
- Page 171 and 172:
e e -- , • Film D-43 . Black prod
- Page 173 and 174:
• X-R!l Diffraction ()t. A. Bredi
- Page 175 and 176:
e. e 175 Another observation may tu
- Page 177 and 178:
~ 177 The results obtained thus tar
- Page 179 and 180:
e e 179 o . RU0 2 (Strukturber1oht)
- Page 181 and 182:
e 181 Instruments and PAlste&! Meas
- Page 183 and 184:
- f 183 Allot these circuits operat
- Page 185 and 186:
185 e 4- ANALYTICAL SERVICE Lab. an
- Page 187:
, , ...J o x ~ C) %- Z ~ ~