Endoproteinase Asp-N from Pseudomonas fragi ... - Sigma-Aldrich
Endoproteinase Asp-N from Pseudomonas fragi ... - Sigma-Aldrich
Endoproteinase Asp-N from Pseudomonas fragi ... - Sigma-Aldrich
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
ENDOPROTEINASE ASP-N<br />
Suitable for Protein Sequencing<br />
and Peptide Mapping<br />
Product Code P 3303<br />
Storage Temperature 2-8 °C<br />
TECHNICAL BULLETIN<br />
CAS# 9001-92-7<br />
EC: 3.4.24.33<br />
Product Description<br />
<strong>Endoproteinase</strong> <strong>Asp</strong>-N is a metallo endoprotease,<br />
isolated <strong>from</strong> a mutant strain of <strong>Pseudomonas</strong> <strong>fragi</strong>,<br />
which hydrolyzes peptide bonds on the N-terminal side<br />
of aspartic and cysteic acid residues. 1-3<br />
<strong>Endoproteinase</strong> <strong>Asp</strong>-N is HPLC purified, resulting in a<br />
product that is suitable for proteomic work. In 100 mM<br />
NH 4 HCO 3 , pH 8.5, or 100 mM Tris HCl, pH 8.5, <strong>Asp</strong>-N<br />
specifically cleaves peptide bonds on the N-terminal<br />
side of aspartic and cysteic acid residues. 1-3 <strong>Asp</strong>-N is<br />
used in proteomics for peptide mapping and protein<br />
sequence work due to its highly specific cleavage of<br />
peptides resulting in a limited number of peptide<br />
fragments. 1-6<br />
<strong>Endoproteinase</strong> <strong>Asp</strong>-N has an average molecular<br />
weight of 24.5 kDa and a pH optimum between pH 6.0<br />
and 8.5. 1<br />
Vial content: 2 µg of lyophilized <strong>Asp</strong>-N containing Tris-<br />
HCl.<br />
Precautions and Disclaimer<br />
Self digestion may occur if temperatures above 37 °C<br />
are used. <strong>Asp</strong>-N retains most of its activity in 2.0 M<br />
urea, 1.0 M guanidine HCl or 0.1% SDS. 1,6 A known<br />
peptide such as glucagon should be run as a control for<br />
all experiments.<br />
Preparation Instructions<br />
Reconstitute the lyophilized product in 50 µl water. The<br />
protease will be in a solution containing 10 mM Tris-<br />
HCl, pH 8.0.<br />
Storage/Stability<br />
The lyophilized powder is stable for at least one year if<br />
stored desiccated at 2-8 °C. After reconstitution in<br />
water, frozen aliquots can be stored for several<br />
weeks. 1,3<br />
Procedure<br />
For peptide or protein digestion, a ratio between 1/50<br />
and 1/200 (w/w) of enzyme to substrate is<br />
recommended. Dissolve the peptide or protein to be<br />
digested in 100 mM NH 4 HCO 3 , pH 8.5, or 100 mM Tris<br />
HCl, pH 8.5. Recommended incubation time is<br />
between 2 and 18 hours at 37 °C depending on the<br />
enzyme to substrate ratio. <strong>Endoproteinase</strong> <strong>Asp</strong>-N may<br />
also be used for in gel digestions of proteins. 7-10<br />
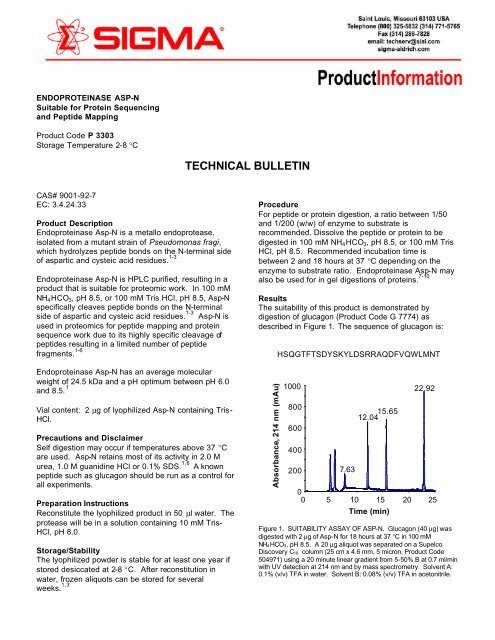
Results<br />
The suitability of this product is demonstrated by<br />
digestion of glucagon (Product Code G 7774) as<br />
described in Figure 1. The sequence of glucagon is:<br />
Absorbance, 214 nm (mAu)<br />
HSQGTFTSDYSKYLDSRRAQDFVQWLMNT<br />
1000<br />
800<br />
600<br />
400<br />
200<br />
7.63<br />
12.04 15.65 22.92<br />
0<br />
0 5 10 15 20 25<br />
Time (min)<br />
Figure 1. SUITABILITY ASSAY OF ASP-N. Glucagon (40 µg) was<br />
digested with 2 µg of <strong>Asp</strong>-N for 18 hours at 37 °C in 100 mM<br />
NH 4HCO 3, pH 8.5. A 20 µg aliquot was separated on a Supelco<br />
Discovery C 18 column (25 cm x 4.6 mm, 5 micron, Product Code<br />
504971) using a 20 minute linear gradient <strong>from</strong> 5-50% B at 0.7 ml/min<br />
with UV detection at 214 nm and by mass spectrometry. Solvent A:<br />
0.1% (v/v) TFA in water. Solvent B: 0.08% (v/v) TFA in acetonitrile.
The <strong>Asp</strong>-N proteolytic fragments were identified as<br />
follows:<br />
Retention Time (min) Mass (Da) Fragment<br />
7.63 731.3 <strong>Asp</strong>(15)-Gln(20)<br />
12.04 863.3 His(1)-Ser(8)<br />
15.65 787.3 <strong>Asp</strong>(9)-Leu(14)<br />
22.92 1152.3 <strong>Asp</strong>(21)-Thr(29)<br />
During the 18 hour digestion only the expected peptides<br />
were generated with no indication of other major<br />
proteolytic activity.<br />
References<br />
1. Drapeau, G.R., J. Biol. Chem., 255, 839-40, (1980).<br />
2. Wilson, K.J. et al., Methods in Protein Sequence<br />
Analysis: Proceedings of the 7th International<br />
Conference, Berlin, July 3-8, 1988, Wittmann-<br />
Liebold, B. (ed.), Springer-Verlag, (New York,<br />
1989), p 310.<br />
3. Ponstingl, H. et al., Advanced Methods in Protein<br />
Microsequencing Analysis, Wittmann-Liebold, B. et<br />
al., (eds.), Springer-Verlag, (New York, 1986)<br />
p 316.<br />
4. Cooper, J.A. et al., J. Biol. Chem., 259, 7835-41,<br />
(1984).<br />
5. Guild, B.C. and Strominger, J.L., J. Biol. Chem.,<br />
259, 9235-40, (1984).<br />
6. Aitken, A. et al., Protein Sequencing: A Practical<br />
Approach, Findlay, J.B.C. and Geisow, M.J. (eds.),<br />
IRL Press, (Oxford, 1989), p 43.<br />
7. Eckerskorn, C. and Grimm, R., Electrophoresis, 17,<br />
899-906, (1996).<br />
8. Scheler, C. et al., Electrophoresis, 19, 918-27,<br />
(1998).<br />
9. Jimenez-Asensio, J., et al., J. Biol. Chem., 274,<br />
32287-94, (1999).<br />
10. Jeno, P., et al., Anal. Biochem., 224, 75-82, (1995).<br />
SIGMA HPLC Purified Products:<br />
Product Package Product<br />
Name Size Code<br />
Carboxypeptidase Y 25 µg C 4046<br />
α-Chymotrypsin 25, 100 µg C 6423<br />
<strong>Endoproteinase</strong> Arg-C 5 µg P 6056<br />
<strong>Endoproteinase</strong> <strong>Asp</strong>-N 2 µg P 3303<br />
<strong>Endoproteinase</strong> Glu-C 25 µg P 6181<br />
<strong>Endoproteinase</strong> Lys-C 5 µg P 3428<br />
Leucine aminopeptidase 50 µg L 9776<br />
Trypsin 100 µg T 8658<br />
Insulin Chain B, Oxidized 100 µg I 1764<br />
Melittin 100 µg M 1407<br />
Glucagon 100 µg G 7774<br />
α-Melanocyte 1, 5 mg M 4135<br />
Stimulating Hormone<br />
Titles of Related Books (Product Code):<br />
MASS SPECTROMETRY OF PROTEINS AND<br />
PEPTIDES: Methods in Molecular Biology (M 7059)<br />
MICROCHARACTERIZATION OF PROTEINS<br />
(M 9807)<br />
NEW METHODS IN PEPTIDE MAPPING FOR THE<br />
CHARACTERIZATION OF PROTEINS (Z36,931-4)<br />
A PRACTICAL GUIDE TO PROTEIN AND PEPTIDE<br />
PURIFICATION FOR MICROSEQUENCING<br />
(Z35,034-6)<br />
SEQUENCING OF PROTEINS AND PEPTIDES:<br />
Laboratory Techniques in Biochemistry and<br />
Molecular Biology, 2nd Ed. (S 2521)<br />
PROTEOLYTIC ENZYMES: A Practical Approach<br />
(P 4926)<br />
PROTEIN STRUCTURE: A Practical Approach<br />
(Z37,548-9)<br />
HIGH PERFORMANCE LIQUID CHROMATOGRAPHY<br />
OF PEPTIDES AND PROTEINS: Separation,<br />
Analysis and Conformation (H 8147)<br />
HPLC OF MACROMOLECULES: A Practical Approach<br />
(H 3409)<br />
PROTEIN SEQUENCING PROTOCOLS: Methods in<br />
Molecular Biology (Z37,395-8)<br />
LKB 6/01<br />
<strong>Sigma</strong> brand products are sold through <strong>Sigma</strong>-<strong>Aldrich</strong>, Inc.<br />
<strong>Sigma</strong>-<strong>Aldrich</strong>, Inc. warrants that its products conform to the information contained in this and other <strong>Sigma</strong>-Aldric h publications. Purchaser<br />
must determine the suitability of the product(s) for their particular use. Additional terms and conditions may apply. Please see reverse side of<br />
the invoice or packing slip.