Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
of the valence electrons by putting them lower in energy<br />
wells. The filled shells of the noble gases (helium,<br />
neon, argon, etc.) are particularly low energy arrangements<br />
of the electrons. These elements do not react<br />
with other elements because the reaction does not provide<br />
a means to an overall lowering of the electron energies.<br />
The effective oxidation state of the noble gases is<br />
identically zero. Many ions achieve this “noble gas<br />
electron configuration,” but of course no change has<br />
been made to the nucleus.<br />
In general, metallic elements near the left of the<br />
Periodic Table assume positive oxidation states in<br />
chemical reactions. Nonmetallic elements near the right<br />
side of the Periodic Table assume negative oxidation<br />
states. Near the middle of the Periodic Table things are<br />
somewhat ambiguous. Will an element like carbon<br />
(which has four valence electrons) lose its four valence<br />
electrons (like a metal), or gain four to fill its shell (like<br />
a nonmetal)? The answer depends on the specific compound<br />
that is being formed. An element in the middle,<br />
such as carbon, will do whichever best lowers its electron<br />
energy. Thus, for elements near the middle of the<br />
chart there is a tendency for atoms to assume different<br />
oxidation states in different chemical reactions, depending<br />
on the particular compound which is being formed.<br />
Although many elements can have more than one<br />
oxidation state (depending on the compound), the primary<br />
(likely) oxidation state is related to the atom’s position<br />
in the Periodic Table. All of the elements in column<br />
1 (labeled IA in the Periodic Table) will have an oxidation<br />
state of +1 in ionic compounds. Elements in IIA<br />
have +2 oxidation states. What would you predict for<br />
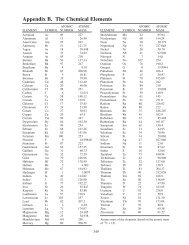
column IIIA? Check your answer in Figure <strong>20</strong>.4.<br />
Elements in columns VIA <strong>and</strong> VIIA of the Periodic<br />
Table tend to gain electrons to fill their valence shells<br />
<strong>and</strong> can be predicted with confidence to have primary<br />
oxidation states of –2 <strong>and</strong> –1, respectively. Elements in<br />
these columns sometimes have additional possible oxidation<br />
states in compounds. See sulfur <strong>and</strong> chlorine in<br />
Figure <strong>20</strong>.4, for example.<br />
Elements in the B columns of the Periodic Table are<br />
also prone to having more than one possible oxidation<br />
state. However, we can use the additional information<br />
that these elements are all metals (<strong>and</strong>, therefore, tend to<br />
be electron donors) to predict that at least in some compounds<br />
these elements will have positive oxidation<br />
states that correspond directly to the number of valence<br />
electrons indicated by their column number. For example,<br />
chromium ( 24 Cr) in column VIB will in at least<br />
some cases have an oxidation state of +6. The rule is<br />
even less precise for the elements in the centermost<br />
columns of the Periodic Table (labeled VIII [neither A<br />
nor B]). Here we can only predict that, as metals, the<br />
elements will have positive oxidation states, but we<br />
must rely on experimental analysis of the individual<br />
compounds to determine the precise oxidation states of<br />
the constituent elements.<br />
Why do the regularities exist among the elements?<br />
They are based on ionization energies whose precise values<br />
can be traced back to the Wave Model <strong>and</strong> the<br />
Exclusion Principle. If only one electron in an atom has<br />
a low ionization energy, the oxidation state will be +1. If<br />
two electrons have low ionization energies, the oxidation<br />
state will be +2, <strong>and</strong> so on. If there are no electrons with<br />
low ionization energies, but there is a vacancy in an<br />
orbital deep in the well, the atom will try to fill the vacancy<br />
with an electron. This results in a –1 oxidation state.<br />
<strong>Compounds</strong> Between <strong>Metals</strong> <strong>and</strong> Nonmetals<br />
Imagine a metallic sodium ion, Na + , <strong>and</strong> a nonmetallic<br />
chloride ion, Cl – , that could form an ionic bond. But<br />
why should the pairing stop there? Could the Na + attract<br />
another negative ion <strong>and</strong> the Cl – attract another positive<br />
ion? Yes. Long chains of alternating Na + <strong>and</strong> Cl – ions<br />
form a sheet like a checkerboard (Fig. <strong>20</strong>.5), <strong>and</strong> finally<br />
the checkerboards stack so that positive ions are always<br />
above negative ions <strong>and</strong> vice versa. The result is a crystal<br />
of salt based on the electrical attraction of the ions.<br />
Figure <strong>20</strong>.5. Drawing of a small part of a single layer<br />
of a NaCl (sodium chloride) crystal.<br />
A salt is much different than a metal. Ionic bonding<br />
accounts for the characteristics of salts discussed<br />
below.<br />
1. Electrically nonconducting. The electrons in<br />
both ions of a salt are all in stable, closed<br />
shells, <strong>and</strong> are difficult to remove from their<br />
respective ions. Because there are no mobile<br />
electrons, solid salts have no way to conduct<br />
electricity. However, if the salt is melted,<br />
whole ions may move about <strong>and</strong> conduct electricity,<br />
but they are so big <strong>and</strong> awkward they<br />
are not nearly as conductive as the electrons in<br />
a metal. Also, if the salt is dissolved in a liquid<br />
like water, the ions separate <strong>and</strong> are able to<br />
186