CMP Mycophenolate - BDNG
CMP Mycophenolate - BDNG
CMP Mycophenolate - BDNG
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
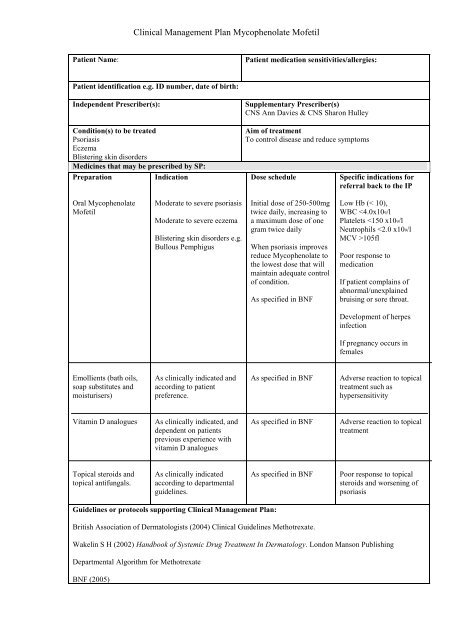
Clinical Management Plan <strong>Mycophenolate</strong> Mofetil<br />
Patient Name:<br />
Patient medication sensitivities/allergies:<br />
Patient identification e.g. ID number, date of birth:<br />
Independent Prescriber(s):<br />
Condition(s) to be treated<br />
Psoriasis<br />
Eczema<br />
Blistering skin disorders<br />
Medicines that may be prescribed by SP:<br />
Preparation<br />
Indication<br />
Supplementary Prescriber(s)<br />
CNS Ann Davies & CNS Sharon Hulley<br />
Aim of treatment<br />
To control disease and reduce symptoms<br />
Dose schedule<br />
Specific indications for<br />
referral back to the IP<br />
Oral <strong>Mycophenolate</strong><br />
Mofetil<br />
Moderate to severe psoriasis<br />
Moderate to severe eczema<br />
Blistering skin disorders e.g.<br />
Bullous Pemphigus<br />
Initial dose of 250-500mg<br />
twice daily, increasing to<br />
a maximum dose of one<br />
gram twice daily<br />
When psoriasis improves<br />
reduce <strong>Mycophenolate</strong> to<br />
the lowest dose that will<br />
maintain adequate control<br />
of condition.<br />
As specified in BNF<br />
Low Hb (< 10),<br />
WBC
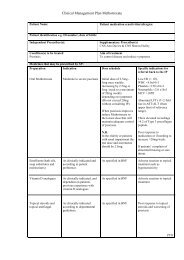
Clinical Management Plan <strong>Mycophenolate</strong> Mofetil<br />
Frequency of review and monitoring by:<br />
Supplementary prescriber<br />
As per departmental algorithm<br />
Supplementary prescriber and independent prescriber<br />
12 months or sooner if there are complications<br />
Process for reporting ADRs:<br />
Write to GP<br />
Report to independent prescriber.<br />
Yellow card if severe drug reaction<br />
Shared record to be used by IP and SP:<br />
Patients medical notes<br />
Agreed by independent<br />
prescriber(s)<br />
Date<br />
Agreed by supplementary<br />
prescriber(s)<br />
Date<br />
Date agreed with<br />
patient/carer