NHS Ayrshire & Arran Formulary Bulletin - NHS Ayrshire and Arran.
NHS Ayrshire & Arran Formulary Bulletin - NHS Ayrshire and Arran.
NHS Ayrshire & Arran Formulary Bulletin - NHS Ayrshire and Arran.
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
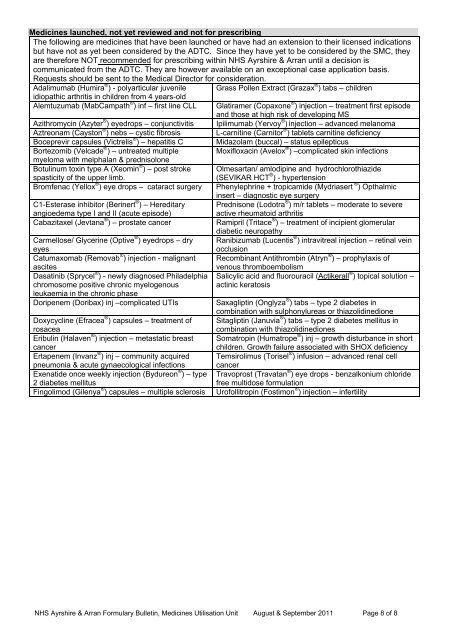
Medicines launched, not yet reviewed <strong>and</strong> not for prescribing<br />
The following are medicines that have been launched or have had an extension to their licensed indications<br />
but have not as yet been considered by the ADTC. Since they have yet to be considered by the SMC, they<br />
are therefore NOT recommended for prescribing within <strong>NHS</strong> <strong>Ayrshire</strong> & <strong>Arran</strong> until a decision is<br />
communicated from the ADTC. They are however available on an exceptional case application basis.<br />
Requests should be sent to the Medical Director for consideration.<br />
Adalimumab (Humira ® ) - polyarticular juvenile Grass Pollen Extract (Grazax ® ) tabs – children<br />
idiopathic arthritis in children from 4 years-old<br />
Alemtuzumab (MabCampath ® ) inf – first line CLL Glatiramer (Copaxone ® ) injection – treatment first episode<br />
<strong>and</strong> those at high risk of developing MS<br />
Azithromycin (Azyter ® ) eyedrops – conjunctivitis Ipilimumab (Yervoy ® ) injection – advanced melanoma<br />
Aztreonam (Cayston ® ) nebs – cystic fibrosis L-carnitine (Carnitor ® ) tablets carnitine deficiency<br />
Boceprevir capsules (Victrelis ® ) – hepatitis C Midazolam (buccal) – status epilepticus<br />
Bortezomib (Velcade ® ) – untreated multiple Moxifloxacin (Avelox ® ) –complicated skin infections<br />
myeloma with melphalan & prednisolone<br />
Botulinum toxin type A (Xeomin ® ) – post stroke Olmesartan/ amlodipine <strong>and</strong> hydrochlorothiazide<br />
spasticity of the upper limb.<br />
(SEVIKAR HCT ® ) - hypertension<br />
Bromfenac (Yellox ® ) eye drops – cataract surgery Phenylephrine + tropicamide (Mydriasert ® ) Opthalmic<br />
insert – diagnostic eye surgery<br />
C1-Esterase inhibitor (Berinert ® ) – Hereditary Prednisone (Lodotra ® ) m/r tablets – moderate to severe<br />
angioedema type I <strong>and</strong> II (acute episode)<br />
active rheumatoid arthritis<br />
Cabazitaxel (Jevtana ® ) – prostate cancer<br />
Ramipril (Tritace ® ) – treatment of incipient glomerular<br />
diabetic neuropathy<br />
Carmellose/ Glycerine (Optive ® ) eyedrops – dry Ranibizumab (Lucentis ® ) intravitreal injection – retinal vein<br />
eyes<br />
occlusion<br />
Catumaxomab (Removab ® ) injection - malignant Recombinant Antithrombin (Atryn ® ) – prophylaxis of<br />
ascites<br />
Dasatinib (Sprycel ® ) - newly diagnosed Philadelphia<br />
chromosome positive chronic myelogenous<br />
leukaemia in the chronic phase<br />
Doripenem (Doribax) inj –complicated UTIs<br />
Doxycycline (Efracea ® ) capsules – treatment of<br />
rosacea<br />
Eribulin (Halaven ® ) injection – metastatic breast<br />
cancer<br />
Ertapenem (Invanz ® ) inj – community acquired<br />
pneumonia & acute gynaecological infections<br />
Exenatide once weekly injection (Bydureon ® ) – type<br />
2 diabetes mellitus<br />
Fingolimod (Gilenya ® ) capsules – multiple sclerosis<br />
venous thromboembolism<br />
Salicylic acid <strong>and</strong> fluorouracil (Actikerall ® ) topical solution –<br />
actinic keratosis<br />
Saxagliptin (Onglyza ® ) tabs – type 2 diabetes in<br />
combination with sulphonylureas or thiazolidinedione<br />
Sitagliptin (Januvia ® ) tabs – type 2 diabetes mellitus in<br />
combination with thiazolidinediones<br />
Somatropin (Humatrope ® ) inj – growth disturbance in short<br />
children. Growth failure associated with SHOX deficiency<br />
Temsirolimus (Torisel ® ) infusion – advanced renal cell<br />
cancer<br />
Travoprost (Travatan ® ) eye drops - benzalkonium chloride<br />
free multidose formulation<br />
Urofollitropin (Fostimon ® ) injection – infertility<br />
<strong>NHS</strong> <strong>Ayrshire</strong> & <strong>Arran</strong> <strong>Formulary</strong> <strong>Bulletin</strong>, Medicines Utilisation Unit August & September 2011 Page 8 of 8