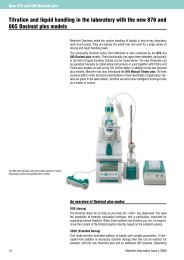
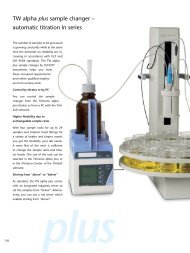
857 Titrando The intelligent all-rounder with ... - Lambda-Med Kft.
857 Titrando The intelligent all-rounder with ... - Lambda-Med Kft.
857 Titrando The intelligent all-rounder with ... - Lambda-Med Kft.
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Quality and compatibility<br />
Compatibility and traceability<br />
GLP, GMP, 21 CFR Part 11 are becoming increasingly important in day-to-day laboratory life. <strong>The</strong><br />
<strong>Titrando</strong> System is entirely oriented towards quality management in the laboratory and offers the<br />
following possibilities:<br />
• Each time that it is switched on the <strong>Titrando</strong> System carries out a self-diagnosis.<br />
• If it has been programmed accordingly then the instrument will remind you about any validation<br />
or service work that is due.<br />
• You can enter limits for results; their observance will be checked for each determination.<br />
• <strong>The</strong> titer of the titrant can be monitored as a function of time in both tabular form or as a graph,<br />
similar to a control card.<br />
• <strong>The</strong> calibration history of the sensors can be c<strong>all</strong>ed up. This means, for example, that alterations<br />
to the sensor caused by aging can be recognized before they affect the results.<br />
• All changes to the methods are documented; traceability is guaranteed.<br />
<strong>The</strong> <strong>Titrando</strong> <strong>all</strong>ows strict control of rights of access using login and password. <strong>The</strong> requirements<br />
defined in FDA Regulation 21 CFR Part 11 regarding «electronic signature» and «electronic record»<br />
are fulfilled in the stand-alone system using Touch Control as well as in the version controlled via PC.<br />
This also applies to the other points contained in this regulation, namely the protection of electronic<br />
records against accidental or intentional alteration and the complete traceability. <strong>The</strong> <strong>Titrando</strong> <strong>with</strong><br />
Touch Control is the only stand-alone titration system <strong>with</strong> Audit Trail.<br />
Quality management at Metrohm<br />
Quality has always been extremely important for Metrohm. On 5 November 1993 our company<br />
achieved the ISO 9001 certificate of quality. <strong>The</strong> Metrohm quality management system is continu<strong>all</strong>y<br />
being perfected and checked by both internal and external audits.<br />
<strong>The</strong> <strong>Titrando</strong> System is modern. This also applies to the object-oriented development system used in<br />
its creation. <strong>The</strong> Touch Control as a color display and operational element is unique, as are the many<br />
other innovations of the <strong>Titrando</strong> System.<br />
16