DICOM Conformance Statement
DICOM Conformance Statement
DICOM Conformance Statement
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>DICOM</strong> <strong>Conformance</strong> <strong>Statement</strong> for Cerner ProVision TM PACS Release 4.5<br />
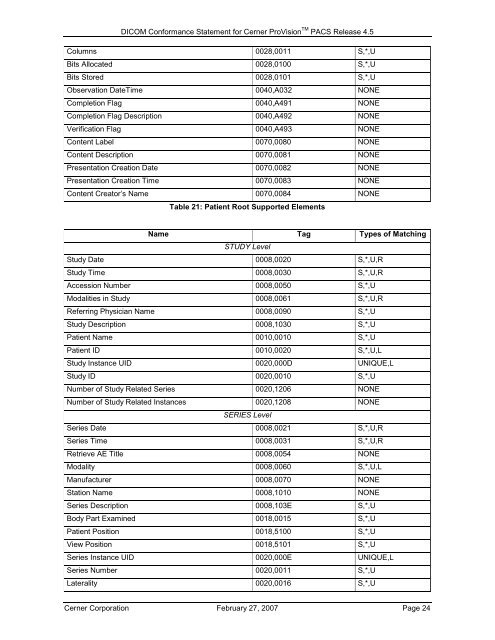
Columns 0028,0011 S,*,U<br />
Bits Allocated 0028,0100 S,*,U<br />
Bits Stored 0028,0101 S,*,U<br />
Observation DateTime 0040,A032 NONE<br />
Completion Flag 0040,A491 NONE<br />
Completion Flag Description 0040,A492 NONE<br />
Verification Flag 0040,A493 NONE<br />
Content Label 0070,0080 NONE<br />
Content Description 0070,0081 NONE<br />
Presentation Creation Date 0070,0082 NONE<br />
Presentation Creation Time 0070,0083 NONE<br />
Content Creator’s Name 0070,0084 NONE<br />
Table 21: Patient Root Supported Elements<br />
Name Tag Types of Matching<br />
STUDY Level<br />
Study Date 0008,0020 S,*,U,R<br />
Study Time 0008,0030 S,*,U,R<br />
Accession Number 0008,0050 S,*,U<br />
Modalities in Study 0008,0061 S,*,U,R<br />
Referring Physician Name 0008,0090 S,*,U<br />
Study Description 0008,1030 S,*,U<br />
Patient Name 0010,0010 S,*,U<br />
Patient ID 0010,0020 S,*,U,L<br />
Study Instance UID 0020,000D UNIQUE,L<br />
Study ID 0020,0010 S,*,U<br />
Number of Study Related Series 0020,1206 NONE<br />
Number of Study Related Instances 0020,1208 NONE<br />
SERIES Level<br />
Series Date 0008,0021 S,*,U,R<br />
Series Time 0008,0031 S,*,U,R<br />
Retrieve AE Title 0008,0054 NONE<br />
Modality 0008,0060 S,*,U,L<br />
Manufacturer 0008,0070 NONE<br />
Station Name 0008,1010 NONE<br />
Series Description 0008,103E S,*,U<br />
Body Part Examined 0018,0015 S,*,U<br />
Patient Position 0018,5100 S,*,U<br />
View Position 0018,5101 S,*,U<br />
Series Instance UID 0020,000E UNIQUE,L<br />
Series Number 0020,0011 S,*,U<br />
Laterality 0020,0016 S,*,U<br />
Cerner Corporation February 27, 2007 Page 24