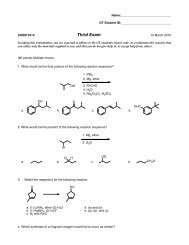
Diels-Alder Reaction (pericyclic reaction)
Diels-Alder Reaction (pericyclic reaction)
Diels-Alder Reaction (pericyclic reaction)
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Diels</strong>-<strong>Alder</strong> <strong>Reaction</strong><br />
(<strong>pericyclic</strong> <strong>reaction</strong>)<br />
H<br />
H<br />
C<br />
C<br />
CH 2<br />
CHCH<br />
CH 2<br />
CH 2<br />
O<br />
100 o<br />
O<br />
CH<br />
1,3-butadiene<br />
the diene<br />
propenal<br />
the dienophile<br />
3-cyclohexene-1-carboxaldehyde<br />
(100 %)
Additional unsaturation enhances the reactivity of<br />
the dienophile’s C=C by electron-withdrawal<br />
O<br />
+<br />
CN<br />
heat<br />
O<br />
CN<br />
(39 %)<br />
O<br />
O<br />
+ O<br />
heat<br />
O<br />
O<br />
(100 %)<br />
O
Stereochemistry<br />
OHC<br />
OHC<br />
H<br />
H<br />
heat<br />
H<br />
H<br />
CHO<br />
CHO<br />
a cis-dienophile<br />
a cis-product
cis or syn-addition<br />
bridge<br />
endo (favored)<br />
OHC<br />
H<br />
H<br />
H<br />
heat<br />
OHC<br />
H<br />
H<br />
H<br />
trans to bridge<br />
exo<br />
H<br />
H<br />
CHO<br />
H<br />
heat<br />
H<br />
H<br />
H<br />
CHO<br />
cis to bridge
O<br />
+ CH 2 =CHCOCH 3<br />
cyclopentadiene<br />
methyl acrylate<br />
O<br />
C<br />
H<br />
OCH 3<br />
+<br />
H<br />
C<br />
O<br />
OCH 3<br />
endo isomer<br />
(75 %)<br />
exo isomer<br />
( 25 %)<br />
1) both isomers are of almost equal stability<br />
2) kinetically controlled preference for<br />
formation of endo product<br />
<strong>Alder</strong> rule - accumulation of maximum unsaturation
O<br />
furan<br />
O<br />
NPh<br />
NPh<br />
+ +<br />
H<br />
O<br />
O<br />
H<br />
O<br />
exo<br />
O<br />
O<br />
endo<br />
3<br />
H<br />
O<br />
O<br />
H<br />
NPh<br />
N-phenylmaleimide<br />
7-Oxabicyclo[2.2.1]hept-5-ene-<br />
2,3-dicarboxy-N-phenylimide<br />
1) Synthesis<br />
Separate endo & exo isomers<br />
using column chromatography<br />
TLC to monitor separation<br />
Obtain 1 H NMR spectra of endo & exo isomers<br />
2) Follow <strong>reaction</strong> by obtaining 1 H NMR spectra<br />
of <strong>reaction</strong> mixture over a period of 4 weeks<br />
at either room temperature or 0 o C
7<br />
Hb<br />
3<br />
2<br />
1<br />
4<br />
6<br />
5<br />
Hx<br />
Hx<br />
norbornene<br />
bicycle[2.2.l]heptene)<br />
Hb<br />
Hn<br />
Hn<br />
stereochemistry can be deduced experimentally<br />
the coupling constants between the bridgehead<br />
protons on C-1 and C-4 (H b )<br />
and the exo (H x ) protons or endo (H n ) protons<br />
on C-5 and C-6
H<br />
O<br />
7-oxabicyclo[2.2.l]heptene ring system<br />
H<br />
H<br />
H<br />
O<br />
O<br />
NPh<br />
H<br />
O<br />
H<br />
O<br />
H<br />
H<br />
O<br />
NPh<br />
Karplus equation: J = 8.5cos 2 θ -0.5cosθ -0.028<br />
θ = the dihedral angle
J (Hz)<br />
10<br />
9<br />
8<br />
7<br />
6<br />
5<br />
4<br />
3<br />
2<br />
1<br />
0<br />
0 30 60 90 120 150 180<br />
Dihedral angle (θ)