07 ED Magna EOAI AR02199_A - Edwards Lifesciences
07 ED Magna EOAI AR02199_A - Edwards Lifesciences
07 ED Magna EOAI AR02199_A - Edwards Lifesciences
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
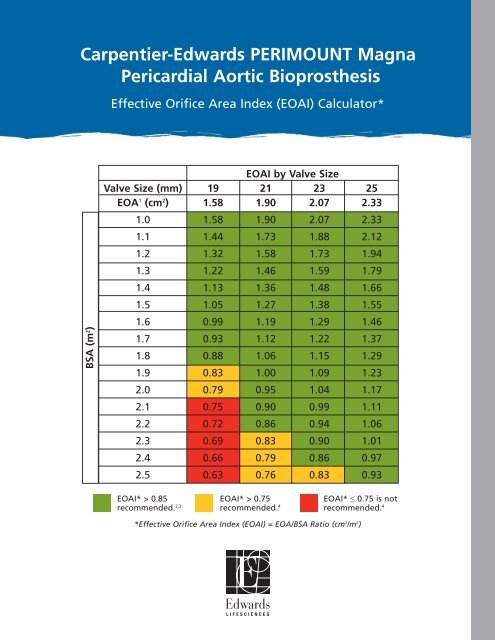
Carpentier-<strong>Edwards</strong> PERIMOUNT <strong>Magna</strong><br />
Pericardial Aortic Bioprosthesis<br />
Effective Orifice Area Index (<strong>EOAI</strong>) Calculator*<br />
BSA (m 2 )<br />
<strong>EOAI</strong> by Valve Size<br />
Valve Size (mm) 19 21 23 25<br />
EOA 1 (cm 2 ) 1.58 1.90 2.<strong>07</strong> 2.33<br />
1.0 1.58 1.90 2.<strong>07</strong> 2.33<br />
1.1 1.44 1.73 1.88 2.12<br />
1.2 1.32 1.58 1.73 1.94<br />
1.3 1.22 1.46 1.59 1.79<br />
1.4 1.13 1.36 1.48 1.66<br />
1.5 1.05 1.27 1.38 1.55<br />
1.6 0.99 1.19 1.29 1.46<br />
1.7 0.93 1.12 1.22 1.37<br />
1.8 0.88 1.06 1.15 1.29<br />
1.9 0.83 1.00 1.09 1.23<br />
2.0 0.79 0.95 1.04 1.17<br />
2.1 0.75 0.90 0.99 1.11<br />
2.2 0.72 0.86 0.94 1.06<br />
2.3 0.69 0.83 0.90 1.01<br />
2.4 0.66 0.79 0.86 0.97<br />
2.5 0.63 0.76 0.83 0.93<br />
<strong>EOAI</strong>* > 0.85<br />
<strong>EOAI</strong>* > 0.75<br />
recommended. 2,3 recommended. 4<br />
<strong>EOAI</strong>* ≤ 0.75 is not<br />
recommended. 4<br />
*Effective Orifice Area Index (<strong>EOAI</strong>) = EOA/BSA Ratio (cm 2 /m 2 )
Rx only. See instructions for use for full prescribing information.<br />
REFERENCES<br />
1. Dalmau MJ, et al. The Carpentier-<strong>Edwards</strong> Perimount <strong>Magna</strong> aortic xenograft: a new design<br />
with an improved hemodynamic performance. Interact Cardio Vasc Thorac Surg 2006;5:263-7.<br />
2. Frapier JM, et al. Influence of patient-prosthesis mismatch on long-term results after aortic<br />
valve replacement with a stented bioprosthesis. J Heart Valve Dis 2002;11(4)543-51.<br />
3. Pibarot P, et al. Patient-prosthesis mismatch can be predicted at the time of operation.<br />
Ann Thorac Surg 2001;71(5 Suppl):S265-8.<br />
4. Rao V, et al. Prosthesis-patient mismatch affects survival after aortic valve replacement.<br />
Circulation 2000;102(19 Suppl 3):III5-9.<br />
<strong>Edwards</strong> <strong>Lifesciences</strong> devices placed on the European market<br />
meeting the essential requirements referred to in Article 3 of<br />
the Medical Device Directive 93/42/EEC bear the CE marking of<br />
conformity.<br />
<strong>Edwards</strong> is a trademark of <strong>Edwards</strong> <strong>Lifesciences</strong> Corporation.<br />
<strong>Edwards</strong> <strong>Lifesciences</strong>, the stylized<br />
E logo, Carpentier-<strong>Edwards</strong> and PERIMOUNT <strong>Magna</strong> are trademarks<br />
of <strong>Edwards</strong> <strong>Lifesciences</strong> Corporation and are registered in<br />
the United States Patent and Trademark Office.<br />
© 20<strong>07</strong> <strong>Edwards</strong> <strong>Lifesciences</strong> LLC<br />
All rights reserved. <strong>AR02199</strong>_A<br />
<strong>Edwards</strong> <strong>Lifesciences</strong> LLC · One <strong>Edwards</strong> Way · Irvine, CA 92614 USA · 949.250.2500 · 800.424.3278 · www.edwards.com<br />
<strong>Edwards</strong> <strong>Lifesciences</strong> (Canada) Inc. · 1290 Central Pkwy West, Suite 300 · Mississauga, Ontario · Canada L5C 4R3 · Phone 905.566.4220 · 800.268.3993<br />
<strong>Edwards</strong> <strong>Lifesciences</strong> S.A. · Ch. du Glapin 6 · 1162 Saint-Prex · Switzerland · Phone 41.21.823.4300