HEALTH AND HUMAN SERVICES COMMISSION TEXAS ...
HEALTH AND HUMAN SERVICES COMMISSION TEXAS ...
HEALTH AND HUMAN SERVICES COMMISSION TEXAS ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
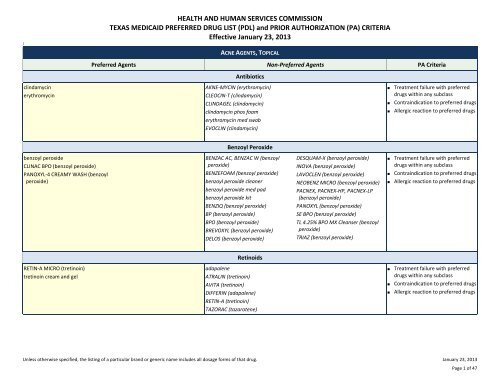
ACNE AGENTS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Antibiotics<br />
clindamycin<br />
erythromycin<br />
AKNE‐MYCIN (erythromycin)<br />
CLEOCIN‐T (clindamycin)<br />
CLINDAGEL (clindamycin)<br />
clindamycin phos foam<br />
erythromycin med swab<br />
EVOCLIN (clindamycin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Benzoyl Peroxide<br />
benzoyl peroxide<br />
CLINAC BPO (benzoyl peroxide)<br />
PANOXYL‐4 CREAMY WASH (benzoyl<br />
peroxide)<br />
BENZAC AC, BENZAC W (benzoyl<br />
peroxide)<br />
BENZEFOAM (benzoyl peroxide)<br />
benzoyl peroxide cleaner<br />
benzoyl peroxide med pad<br />
benzoyl peroxide kit<br />
BENZIQ (benzoyl peroxide)<br />
BP (benzoyl peroxide)<br />
BPO (benzoyl peroxide)<br />
BREVOXYL (benzoyl peroxide)<br />
DELOS (benzoyl peroxide)<br />
DESQUAM‐X (benzoyl peroxide)<br />
INOVA (benzoyl peroxide)<br />
LAVOCLEN (benzoyl peroxide)<br />
NEOBENZ MICRO (benzoyl peroxide)<br />
PACNEX, PACNEX‐HP, PACNEX‐LP<br />
(benzoyl peroxide)<br />
PANOXYL (benzoyl peroxide)<br />
SE BPO (benzoyl peroxide)<br />
TL 4.25% BPO MX Cleanser (benzoyl<br />
peroxide)<br />
TRIAZ (benzoyl peroxide)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Retinoids<br />
RETIN‐A MICRO (tretinoin)<br />
tretinoin cream and gel<br />
adapalene<br />
ATRALIN (tretinoin)<br />
AVITA (tretinoin)<br />
DIFFERIN (adapalene)<br />
RETIN‐A (tretinoin)<br />
TAZORAC (tazarotene)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 1 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ACNE AGENTS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Combination and Other Agents<br />
CERISA (sulfacetamide/sulfur)<br />
CLENIA (sulfacetamide/sulfur)<br />
clindamycin/benzoyl peroxide<br />
CLINAC BPO (clindamycin/benzoyl<br />
peroxide)<br />
SASTID (salicylic acid/sulfur)<br />
SULPHO‐LAC (sulfur)<br />
SULFOAM (sulfur)<br />
TOPISULF (sulfacetamide/sulfur)<br />
ACANYA (benzoyl<br />
peroxide/clindamycin)<br />
ACZONE (dapsone)<br />
AVAR, AVAR‐E, AVAR‐E LS, AVAR‐LS<br />
(sulfacetamide/sulfur)<br />
AZELEX (azelaic acid)<br />
BENZACLIN (benzoyl<br />
peroxide/clindamycin)<br />
BENZAMYCIN (benzoyl<br />
peroxide/erythromycin)<br />
CLARIFOAM EF (sulfacetamide/sulfur)<br />
CLINDACIN PAC KIT (clindamycin)<br />
DUAC (benzoyl peroxide/clindamycin)<br />
EPIDUO (benzoyl<br />
peroxide/adapalene)<br />
erythromycin/benzoyl peroxide<br />
GARIMIDE (sulfacetamide/sulfur)<br />
KLARON (sodium sulfacetamide)<br />
NUOX (benzoyl peroxide/sulfur)<br />
OVACE PLUS (sulfacetamide)<br />
PLEXION (sulfacetamide/sulfur)<br />
PRASCION (sulfacetamide/sulfur)<br />
ROSANIL (sulfacetamide/sulfur)<br />
SEB‐PREV (sulfacetamide)<br />
SE 10‐5 SS (sulfacetamide/sulfur)<br />
SSS 10‐4 (sulfacetamide/sulfur)<br />
sulfacetamide<br />
sulfacetamide/sulfur<br />
sulfacetamide/sulfur cleaner<br />
sulfacetamide/sulfur TS<br />
sulfacetamide/sulfur/urea<br />
SUMAXIN (sulfacetamide/sulfur)<br />
SUMADAN (sulfacetamide/sulfur)<br />
SUPHERA (sulfacetamide/sulfur)<br />
VELTIN (clindamycin/tretinoin)<br />
ZIANA (clindamycin/tretinoin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ALZHEIMER’S AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Cholinesterase Inhibitors<br />
donepezil 5 and 10 mg<br />
donepezil ODT<br />
EXELON Transdermal (rivastigmine)<br />
galantamine<br />
galantamine ER<br />
ARICEPT 23 mg (donepezil)<br />
ARICEPT 5 <strong>AND</strong> 10 MG (donepezil)<br />
ARICEPT ODT (donepezil ODT)<br />
EXELON caps & solution<br />
(rivastigmine)<br />
Galantamine solution<br />
RAZADYNE (galantamine)<br />
RAZADYNE ER (galantamine)<br />
rivastigmine<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 2 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ALZHEIMER’S AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
NMDA Receptor Antagonist<br />
NAMENDA (memantine) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANALGESICS, NARCOTIC – LONG ACTING<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
BUTRANS (buprenorphine)<br />
DURAGESIC (fentanyl) PPG<br />
morphine ER PPG (generic MS Contin)<br />
tramadol ER<br />
AVINZA (morphine)<br />
CONZIP (tramadol)<br />
EMBEDA (morphine/naltrexone)<br />
EXALGO (hydromorphone)<br />
fentanyl patch PPG<br />
KADIAN (morphine)<br />
morphine ER (generic Kadian)<br />
MS CONTIN (morphine)<br />
NUCYNTA ER (tapentadol)<br />
OPANA ER (oxymorphone)<br />
oxycodone ER<br />
OXYCONTIN (oxycodone)<br />
oxymorphone ER<br />
RYZOLT (tramadol)<br />
ULTRAM ER (tramadol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 3 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANALGESICS, NARCOTIC – SHORT ACTING (NON‐PARENTERAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
APAP/codeine PPG<br />
butalbital/APAP/caffeine/codeine<br />
butalbital/ASA/caffeine/codeine<br />
codeine<br />
hydrocodone/APAP PPG<br />
hydrocodone/ibuprofen<br />
hydromorphone PPG<br />
morphine PPG<br />
oxycodone PPG<br />
pentazocine/APAP<br />
pentazocine/naloxone<br />
ROXICET SOLUTION<br />
(oxycodone/APAP)<br />
tramadol<br />
tramadol/APAP<br />
TREZIX<br />
(dihydrocodeine/APAP/caffeine)<br />
ABSTRAL (fentanyl)<br />
ACTIQ (fentanyl)<br />
Carisoprodol compound/codeine<br />
(carisoprodol, aspirin, codeine)<br />
COCET PLUS (codeine/APAP)<br />
DEMEROL (meperidine)<br />
dihydrocodeine/APAP/caffeine<br />
DILAUDID (hydromorphone)<br />
fentanyl<br />
FENTORA (fentanyl)<br />
FIORICET W/CODEINE<br />
(butalbital/APAP/caffeine/ codeine)<br />
FIORINAL W/CODEINE<br />
(butalbital/ASA/caffeine/codeine)<br />
HYCET (hydrocodone/APAP)<br />
hydromorphone suppositories<br />
IBUDONE (hydrocodone/ibuprofen)<br />
levorphanol<br />
LORCET (hydrocodone/APAP)<br />
LORTAB (hydrocodone/APAP)<br />
meperidine<br />
NORCO (hydrocodone/APAP)<br />
NUCYNTA (tapentadol)<br />
ONSOLIS (fentanyl)<br />
OPANA (oxymorphone)<br />
OXECTA (oxycodone)<br />
oxycodone/APAP<br />
oxycodone/ASA<br />
oxycodone concentrate<br />
oxycodone/ibuprofen<br />
OXYFAST (oxycodone)<br />
oxymorphone<br />
PERCOCET (oxycodone/APAP)<br />
PRIMLEV (oxycodone/APAP)<br />
REPREXAIN (hydrocodone/ibuprofen)<br />
ROXICODONE (oxycodone)<br />
RYBIX ODT (tramadol)<br />
SUBSYS (fentanyl sublingual spray)<br />
SYNALGOS‐DC<br />
(dihydrocodeine/ASA/caffeine)<br />
TALACEN (pentazocine/APAP)<br />
TALWIN NX (pentazocine/naloxone)<br />
TYLENOL W/CODEINE<br />
(APAP/codeine)<br />
ULTRACET (tramadol/APAP)<br />
ULTRAM (tramadol)<br />
VICODIN (hydrocodone/APAP)<br />
VICOPROFEN<br />
(hydrocodone/ibuprofen)<br />
XODOL (hydrocodone/APAP)<br />
ZAMICET (hydrocodone/APAP)<br />
ZOLVIT (hydrocodone/APAP)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 4 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
<strong>AND</strong>ROGENIC AGENTS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
<strong>AND</strong>ROGEL (testosterone) <strong>AND</strong>RODERM (testosterone)<br />
AXIRON (testosterone)<br />
FORTESTA (testosterone)<br />
TESTIM (testosterone)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANGIOTENSIN MODULATORS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Ace Inhibitors<br />
benazepril<br />
captopril<br />
enalapril<br />
fosinopril<br />
lisinopril<br />
quinapril<br />
ramipril<br />
ACCUPRIL (quinapril)<br />
ACEON (perindopril)<br />
ALTACE (ramipril)<br />
CAPOTEN (captopril)<br />
LOTENSIN (benazepril)<br />
MAVIK (trandolapril)<br />
moexepril<br />
MONOPRIL (fosinopril)<br />
perindopril<br />
PRINIVIL (lisinopril)<br />
trandolapril<br />
UNIVASC (moexepril)<br />
VASOTEC (enalapril)<br />
ZESTRIL (lisinopril)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 5 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANGIOTENSIN MODULATORS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ACE Inhibitor/Diuretic Combinations<br />
benazepril/HCTZ<br />
captopril/HCTZ<br />
enalapril/HCTZ<br />
fosinopril/HCTZ<br />
lisinopril/HCTZ<br />
quinapril/HCTZ<br />
BENICAR (olmesartan)<br />
DIOVAN (valsartan)<br />
losartan<br />
BENICAR‐HCT (olmesartan/HCTZ)<br />
DIOVAN‐HCT (valsartan/HCTZ)<br />
losartan/HCTZ<br />
ACCURETIC (quinapril/HCTZ)<br />
CAPOZIDE (captopril/HCTZ)<br />
LOTENSIN HCT (benazepril/HCTZ)<br />
moxepril/HCTZ<br />
MONOPRIL HCT (fosinopril/HCTZ)<br />
PRINZIDE (lisinopril/HCTZ)<br />
UNIRETIC (moexepril/HCTZ)<br />
VASERETIC (enalapril/HCTZ)<br />
ZESTORETIC (lisinopril/HCTZ)<br />
Angiotensin II Receptor Blockers (ARBs)<br />
ATAC<strong>AND</strong> (candesartan)<br />
AVAPRO (irbesartan)<br />
COZAAR (losartan)<br />
EDARBI (azilsartan)<br />
MICARDIS (telmisartan)<br />
TEVETEN (eprosartan)<br />
ARB/Diuretic Combinations<br />
ATAC<strong>AND</strong>‐HCT (candesartan/HCTZ)<br />
AVALIDE (irbesartan/HCTZ)<br />
EDARBYCLOR<br />
(azilsartan/chlorthalidone)<br />
HYZAAR (losartan/HCTZ)<br />
MICARDIS‐HCT (telmisartan/HCTZ)<br />
TEVETEN‐HCT (eprosartan/HCTZ)<br />
Direct Renin Inhibitors<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
TEKTURNA (aliskerin) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 6 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANGIOTENSIN MODULATORS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Direct Renin Inhibitor/Diuretic Combinations<br />
TEKTURNA HCT (aliskerin/HCTZ) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANGIOTENSIN MODULATOR COMBINATIONS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
EXFORGE (valsartan/amlodipine)<br />
EXFORGE HCT<br />
(valsartan/amlodipine/HCTZ)<br />
TARKA (trandolapril/verapamil)<br />
AZOR (olmesartan/amlodipine)<br />
benazepril/amlodipine<br />
LOTREL (benazepril/amlodipine)<br />
TEKAMLO (aliskerin/amlodipine)<br />
trandolapril/verapamil<br />
TRIBENZOR<br />
(olmesartan/amlodipine/HCTZ)<br />
TWYNSTA (telmisartan/amlodipine)<br />
VALTURNA (valsartan/aliskerin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIMICROBIALS, GASTROINTESTINAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
metronidazole tablet<br />
TINDAMAX (tinidazole)<br />
vancomycin<br />
ALINIA(nitazoxanide)<br />
DIFICID (fidaxomicin)<br />
FLAGYLl, FLAGYL ER(metronidazone)<br />
metronidazole capsule<br />
NEO‐FRADIN (neomycin)<br />
neomycin<br />
tinidazole<br />
VANCOCIN (vancomycin)<br />
XIFAXIN (rifaximin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 7 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIBIOTICS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
bacitracin<br />
bacitracin/polymyxin<br />
DOUBLE ANTIBIOTIC<br />
(bacitracin/polymyxin b)<br />
gentamicin<br />
mupirocin<br />
neomycin/polymyxin/pramoxine<br />
triple antibiotic<br />
ALTABAX (retapamulin)<br />
BACTROBAN (mupirocin)<br />
CENTANY (mupirocin)<br />
NEOSPORIN(bacitracin/neomycin/<br />
polymyxin b)<br />
NEOSPORIN PLUS PAIN RELIEF<br />
(bacitracin/neomycin/polyxyxin<br />
b/pramoxine)<br />
POLYSPORIN (bacitracin/polymyxin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIBIOTICS, VAGINAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
clindamycin<br />
metronidazole<br />
CLEOCIN (clindamycin)<br />
CLINDESSE (clindamycin)<br />
V<strong>AND</strong>AZOLE (metronidazole)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTICOAGULANTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ARIXTRA (fondaparinux)<br />
LOVENOX (enoxaparin)<br />
PRADAXA (dabigatran)<br />
warfarin<br />
XARELTO (rivaroxaban)<br />
COUMADIN (warfarin)<br />
Enoxaparin<br />
fondaparinux<br />
FRAGMIN (dalteparin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 8 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIDEPRESSANTS, OTHER<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
bupropion<br />
MARPLAN (isocarboxazid)<br />
mirtazapine<br />
NARDIL (phenelzine)<br />
PRISTIQ (desvenlafaxine)<br />
trazodone<br />
Venlafaxine ER capsules<br />
APLENZIN (bupropion)<br />
EFFEXOR (venlafaxine)<br />
EFFEXOR XR (venlafaxine)<br />
EMSAM (selegiline)<br />
nefazodone<br />
OLEPTRO ER (trazodone)<br />
PARNATE (tranylcypromine)<br />
phenelzin<br />
REMERON (mirtazapine)<br />
tranylcypromine<br />
venlafaxine IR<br />
venlafaxine ER tablets<br />
VIIBRYD (vilazodone)<br />
WELLBUTRIN (bupropion)<br />
ZYBAN SR (bupropion)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIDEPRESSANTS, SSRIS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
citalopram<br />
fluoxetine<br />
fluvoxamine<br />
LEXAPRO (escitalopram)<br />
paroxetine<br />
sertraline<br />
CELEXA (citalopram)<br />
escitalopram<br />
fluoxetine capsule DR<br />
LUVOX CR (fluvoxamine)<br />
paroxetine CR<br />
PAXIL (paroxetine)<br />
PAXIL CR (paroxetine)<br />
PEXEVA (paroxetine)<br />
PROZAC (fluoxetine)<br />
SARAFEM (fluoxetine)<br />
ZOLOFT (sertraline)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 9 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIEMETIC‐ANTIVERTIGO AGENTS<br />
(EXCLUDES INJECTABLES)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Anticholinergics, Antihistamines, Dopamine Antagonists<br />
dimenhydrinate<br />
meclizine RX & OTC<br />
metoclopramide<br />
prochlorperazine<br />
promethazine<br />
trimethobenzamide<br />
TRANSDERM‐SCOP (scopolamine)<br />
METOZOLV ODT (metoclopramide)<br />
REGLAN (metoclopramide)<br />
Cannabinoids<br />
CESAMET (nabilone)<br />
dronabinol<br />
MARINOL (dronabinol)<br />
5‐HT3 Receptor Antagonists<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ondansetron ANZEMET (dolasetron)<br />
Granisetron<br />
KYTRIL (granisetron)<br />
SANCUSO TRANSDERMAL<br />
(granisetron)<br />
ZOFRAN (ondansetron)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Substance P Antagonists<br />
EMEND (aprepitant) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 10 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIFUNGALS, ORAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
clotrimazole<br />
fluconazole<br />
griseofulvin<br />
GRIS‐PEG (griseofulvin)<br />
ketoconazole<br />
nystatin tabs and suspension<br />
terbinafine<br />
ANCOBON (flucytosine)<br />
DIFLUCAN (fluconazole)<br />
flucytosine<br />
GRIFULVIN V (griseofulvin)<br />
itraconazole<br />
LAMISIL (terbinafine)<br />
NOXAFIL (posaconazole)<br />
ORAVIG (miconazole)<br />
SPORANOX (itraconazole)<br />
TERBINEX (terbinafine)<br />
VFEND (voriconazole)<br />
voriconazole<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIFUNGALS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Antifungals<br />
clotrimazole OTC<br />
ketoconazole shampoo<br />
miconazole<br />
nystatin<br />
terbinafine<br />
tolnaftate<br />
AZOLEN TINCTURE (miconazole)<br />
BENSAL HP (benzoic acid/salicylic<br />
acid)<br />
CICLODAN (ciclopirox)<br />
ciclopirox<br />
clotrimazole Rx<br />
CNL 8 (ciclopirox)<br />
DESENEX AERO POWDER<br />
(miconazole)<br />
econazole<br />
ERTACZO (sertaconazole)<br />
EXELDERM (sulconazole)<br />
EXTINA (ketoconazole)<br />
FUNGOID (miconazole)<br />
ketoconazole cream/ointment<br />
KETODAN (ketoconazole)<br />
KURIC (ketoconazole)<br />
LAMISIL (terbinafine)<br />
LOPROX (ciclopirox)<br />
LOTRIMIN (clotrimazole)<br />
MENTAX (butenafine)<br />
MONISTAT (miconazole)<br />
NAFTIN (naftifine)<br />
NIZORAL (ketoconazole)<br />
OXISTAT (oxiconazole)<br />
PERIADERM AF (nystatin)<br />
PEDIPIROX‐4 (ciclopirox)<br />
PENLAC (ciclopirox)<br />
TINACTIN (tolnaftate)<br />
VUSION<br />
(miconazole/zinc/petrolatum)<br />
XOLEGEL (miconazole)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 11 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIFUNGALS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Antifungal/Steroid Combinations<br />
nystatin/triamcinolone clotrimazole/betamethasone<br />
LOTRISONE<br />
(clotrimazole/betamethasone)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIHISTAMINES, MINIMALLY SEDATING<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Antihistamines<br />
cetirizine<br />
loratadine<br />
cetirizine/pseudoephedrine<br />
loratadine/pseudoephedrine<br />
ALLEGRA (fexofenadine)<br />
cetirizine chewable<br />
cetirizine solution 5mg/5ml<br />
CLARINEX (desloratadine)<br />
CLARITIN (loratadine)<br />
fexofenadine<br />
levocetirizine<br />
XYZAL (levocetirizine)<br />
Zyrtec (cetirizine)<br />
Antihistamine/Decongestant Combinations<br />
ALLEGRA‐D<br />
(fexofenadine/pseudoephedrine)<br />
CLARINEX‐D<br />
(desloratadine/pseudoephedrine)<br />
CLARITIN‐D<br />
(loratadine/pseudoephedrine)<br />
fexofenadine/pseudoephedrine<br />
SEMPREX‐D<br />
(acrivastine/pseudoephedrine)<br />
■ Treatment failure after no less than<br />
a 30‐day trial of preferred drugs<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure after no less than<br />
a 30‐day trial of preferred drugs<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 12 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIHYPERURICEMICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
allopurinol<br />
probenecid<br />
probenecid/colchicine<br />
COLCRYS (colchicine)<br />
ULORIC (febuxostat)<br />
ZYLOPRIM (allopurinol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIMIGRAINE AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
MAXALT (rizatriptan)<br />
RELPAX (eletriptan)<br />
sumatriptan tablets, nasal and<br />
injection (kit and vial)<br />
AMERGE (naratriptan)<br />
AXERT (almotriptan)<br />
CAMBIA (diclofenac)<br />
FROVA (frovatriptan)<br />
IMITREX injection, nasal, tablets<br />
(sumatriptan)<br />
naratriptan<br />
sumatriptan injection (disposable<br />
syringe and pen injector)<br />
SUMAVEL (sumatriptan)<br />
TREXIMET (sumatriptan/naproxen)<br />
ZOMIG (zolmitriptan)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIPARASITICS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
EURAX Cream (crotamiton)<br />
permethrin OTC<br />
permethrin RX<br />
piperonyl butoxide/pyrethrins<br />
ULESFIA (benzyl alcohol)<br />
ACTICIN (permethrin)<br />
ELIMITE (permethrin)<br />
EURAX Lotion (crotamiton)<br />
lindane<br />
malathion<br />
NATROBA (spinosad)<br />
OVIDE (malathion)<br />
SKLICE (ivermectin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 13 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIPARKINSON’S AGENTS (ORAL/TRANSDERMAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Anticholinergics<br />
benztropine<br />
trihexyphenidyl<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
COMT Inhibitors<br />
COMTAN (entacapone)<br />
TASMAR (tolcapone)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
bromocriptine<br />
pramipexole<br />
ropinirole<br />
trihexyphenidyl<br />
carbidopa/levodopa tablets<br />
STALEVO (levodopa/carbidopa/<br />
entacapone)<br />
Dopamine Agonists<br />
MIRAPEX (pramipexole)<br />
MIRAPEX ER (pramipexole)<br />
NEUPRO Transdermal (rotigotine)<br />
PARLODEL (bromocriptine)<br />
REQUIP (ropinirole)<br />
REQUIP XL (ropinirole)<br />
MAO‐B Inhibitors<br />
AZILECT (rasagiline)<br />
ELDEPRYL (selegiline)<br />
selegiline<br />
ZELAPAR (selegiline)<br />
Others<br />
carbidopa/levodopa ODT<br />
COMTAN (entacapone)<br />
PARCOPA (levodopa/carbidopa)<br />
SINEMET (levodopa/carbidopa)<br />
TASMAR (tolcapone)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 14 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIPSYCHOTICS (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Antipsychotics<br />
ABILIFY solution, tablets<br />
(aripiprazole)<br />
clozapine<br />
chlorpromazine<br />
FANAPT (iloperidone)<br />
fluphenazine<br />
haloperidol<br />
INVEGA (paliperidone)<br />
LATUDA (lurasidone)<br />
olanzapine<br />
perphenazine<br />
risperidone<br />
SAPHRIS (asenapine)<br />
quetiapine<br />
SEROQUEL XR (quetiapine)<br />
thioridazine<br />
thiothixene<br />
trifluoperazine<br />
ziprasidone<br />
ZYPREXA ZYDIS<br />
ABILIFY DISCMELT (aripiprazole)<br />
clozapineODT<br />
CLOZARIL (clozapine)<br />
FAZACLO (clozapine)<br />
GEODON (ziprasidone)<br />
loxapine<br />
NAVANE (thiothixene)<br />
olanzapine ODT<br />
ORAP (pimozide)<br />
risperidone ODT<br />
RISPERDAL (risperidone)<br />
SEROQUEL (quetiapine)<br />
ZYPREXA (olanzapine)<br />
Antipsychotic/SSRI Combinations<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
amitriptyline/perphenazine<br />
SYMBYAX (olanzapine/fluoxetine)<br />
olanzapine/fluoxetine ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 15 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
ANTIVIRALS, ANTIHERPETIC (ORAL/NASAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
amantadine<br />
acyclovir<br />
famciclovir<br />
RELENZA (zanamivir)<br />
rimantadine<br />
TAMIFLU (oseltamivir)<br />
VALTREX (valacyclovir)<br />
FAMVIR (famciclovir)<br />
valacyclovir<br />
ZOVIRAX (acyclovir)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ANTIVIRALS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ABREVA (docosanol)<br />
DENAVIR (penciclovir)<br />
ZOVIRAX OINTMENT (acyclovir)<br />
XERESE (acyclovir/hydrocortisone)<br />
ZOVIRAX CREAM (acyclovir)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
BETA BLOCKERS (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Beta Blockers<br />
atenolol<br />
betaxolol<br />
INNOPRAN XL (propranolol)<br />
LEVATOL (penbutolol)<br />
metoprolol IR<br />
metoprolol XL<br />
propranolol IR<br />
sotalol<br />
timolol<br />
TOPROL XL (metoprolol succinate)<br />
acebutolol<br />
BETAPACE (sotalol)<br />
bisoprolol<br />
BYSTOLIC (nebivolol)<br />
CORGARD (nadolol)<br />
INDERAL LA (propranolol)<br />
KERLONE (betaxolol)<br />
LOPRESSOR (metoprolol)<br />
nadolol<br />
pindolol<br />
propranolol ER<br />
SECTRAL (acebutolol)<br />
TENORMIN (atenolol)<br />
ZEBETA (bisoprolol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 16 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
BETA BLOCKERS (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Beta Blocker Combinations<br />
atenolol/chlorthalidone<br />
bisoprolol/HCTZ<br />
metoprolol/HCTZ<br />
nadolol/bendroflumethiazide<br />
propranolol/HCTZ<br />
Carvedilol<br />
labetalol<br />
CORZIDE<br />
(nadolol/bendroflumethiazide)<br />
DUTOPROL (metoprolol succinate<br />
ER/HCTZ)<br />
INDERIDE (propranolol/HCTZ)<br />
LOPRESSOR HCT (metoprolol/HCTZ)<br />
TENORETIC (atenolol/HCTZ)<br />
ZIAC (bisoprolol/HCTZ)<br />
Beta‐ and Alpha‐Blockers<br />
COREG (carvedilol)<br />
COREG CR (carvedilol)<br />
TR<strong>AND</strong>ATE (labetalol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
BILE SALTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ursodiol ACTIGALL (ursodiol)<br />
CHENODAL (chenodiol)<br />
URSO (ursodiol)<br />
URSO FORTE (urosodiol)<br />
■ Treatment failure with preferred<br />
drug<br />
■ Contraindication to preferred drug<br />
■ Allergic reaction to preferred drug<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 17 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
BLADDER RELAXANT PREPARATIONS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
oxybutynin IR<br />
oxybutynin ER (oxybutynin)<br />
TOVIAZ (fesoterodine)<br />
VESICARE (solifenacin)<br />
DETROL (tolterodine)<br />
DETROL LA (tolterodine)<br />
DITROPAN XL (oxybutynin)<br />
ENABLEX (darifenacin)<br />
GELNIQUE (oxybutynin)<br />
OXYTROL (oxybutynin)<br />
SANCTURA (trospium)<br />
SANCTURA XR (trospium)<br />
trospium<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
BONE RESORPTION SUPPRESSION <strong>AND</strong> RELATED AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Bisphosphonates<br />
alendronate PPG<br />
FORTICAL (calcitonin)<br />
MIACALCIN (calcitonin)<br />
ACTONEL (risedronate)<br />
ATELVIA (risedronate)<br />
BONIVA (ibandronate)<br />
DIDRONEL (etidronate)<br />
FOSAMAX (alendronate)<br />
FOSAMAX PLUS D<br />
(alendronate/vitamin D)<br />
ibandronate<br />
Other Bone Resorption Suppression and Related Agents<br />
calcitonin nasal<br />
EVISTA (raloxifene)<br />
FORTEO (teriparatide)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 18 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
BPH AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Alpha Blockers<br />
Doxazosin<br />
tamsulosin<br />
terazosin<br />
UROXATRAL (alfuzosin)<br />
alfuzosin<br />
CARDURA (doxazosin)<br />
CARDURA XL (doxazosin)<br />
Flomax (tamsulosin)<br />
RAPAFLO (silodosin)<br />
tamsulosin<br />
5‐Alpha‐Reductase (5AR) Inhibitors<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
finasteride AVODART (dutasteride)<br />
PROSCAR (finasteride)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Alpha Blocker/5AR Inhibitor Combinations<br />
JALYN (dutasteride/tamsulosin) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
BRONCHODILATORS, BETA AGONIST<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Inhalers, Short‐Acting<br />
PROVENTIL HFA (albuterol)<br />
XOPENEX HFA (levalbuterol)<br />
MAXAIR (pirbuterol)<br />
PROAIR HFA (albuterol)<br />
VENTOLIN HFA (albuterol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ History of intolerable side effects<br />
to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 19 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
BRONCHODILATORS, BETA AGONIST<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Inhalers, Long‐Acting<br />
ARCAPTA (indacaterol)<br />
FORADIL (formoterol)<br />
SEREVENT (salmeterol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ History of intolerable side effects<br />
to preferred drugs<br />
Inhalation Solution<br />
albuterol ACCUNEB (albuterol)<br />
BROVANA (arformoterol)<br />
levalbuterol<br />
PERFOROMIST (formoterol)<br />
XOPENEX (levalbuterol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ History of intolerable side effects<br />
to preferred drugs<br />
Oral<br />
albuterol IR albuterol ER<br />
BRETHINE (terbutaline)<br />
Metaproterenol<br />
terbutaline<br />
VOSPIRE ER (albuterol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ History of intolerable side effects<br />
to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 20 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
CALCIUM CHANNEL BLOCKERS (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Short‐Acting<br />
diltiazem<br />
nicardipine<br />
nifedipine<br />
verapamil<br />
amlodipine<br />
diltiazem ER<br />
felodipine ER<br />
nifedipine ER<br />
verapamil ER<br />
CALAN (verapamil)<br />
CARDENE (nicardipine)<br />
CARDIZEM (diltiazem)<br />
DYNACIRC (isradipine)<br />
isradipine<br />
nimodipine<br />
NIMOTOP (nimodipine)<br />
PROCARDIA (nifedipine)<br />
VERELAN (verapamil)<br />
Long‐Acting<br />
ADALAT CC (nifedipine)<br />
CALAN SR (verapamil)<br />
CARDENE SR (nicardipine)<br />
CARDIZEM CD (diltiazem)<br />
CARDIZEM LA (diltiazem)<br />
COVERA‐HS (verapamil)<br />
DILACOR XR (diltiazem)<br />
diltiazem LA<br />
DYNACIRC CR (isradipine)<br />
MATZIM LA (diltiazem)<br />
nisoldipine<br />
NORVASC (amlodipine)<br />
PROCARDIA XL (nifedipine)<br />
SULAR (nisoldipine)<br />
TIAZAC (diltiazem)<br />
verapamil ER PM<br />
verapamil 360 mg caps<br />
VERELAN ER PM (verapamil)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 21 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
CEPHALOSPORINS <strong>AND</strong> RELATED ANTIBIOTICS (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Beta Lactam/Beta‐Lactamase Inhibitor Combinations<br />
amoxicillin/clavulanate suspension<br />
amoxicillin/clavulanate IR tablets<br />
cefadroxil<br />
cephalexin<br />
cefaclor IR capsules, suspension<br />
cefprozil<br />
cefuroxime tablets<br />
Cefdinir<br />
SUPRAX (cefixime)<br />
amoxicillin/clavulanate XR<br />
amoxicillin/clavulanate chew tabs<br />
AUGMENTIN Suspension<br />
(amoxicillin/clavulanate)<br />
AUGMENTIN<br />
(amoxicillin/clavulanate) tablets<br />
AUGMENTIN XR<br />
(amoxicillin/clavulanate)<br />
Cephalosporins – First Generation<br />
KEFLEX (cephalexin)<br />
PANIXINE (cephalexin)<br />
Cephalosporins – Second Generation<br />
cefaclor ER<br />
CEFTIN tablets (cefuroxime)<br />
CEFTIN suspension (cefuroxime)<br />
cefuroxime suspension<br />
Cephalosporins – Third Generation<br />
CEDAX (ceftibuten)<br />
cefditoren<br />
cefpodoxime<br />
SPECTRACEF (cefditoren)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
COLONY STIMULATING FACTORS<br />
LEUKINE (sargramostim, GM‐CSF)<br />
NEULASTA (pegfilgrastim)<br />
NEUPOGEN (filgrastim, G‐CSF)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 22 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
COPD AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Anticholinergics<br />
ATROVENT HFA (ipratropium)<br />
ipratropium inhalation solution<br />
SPIRIVA (tiotropium)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Anticholinergic‐Beta Agonist Combinations<br />
COMBIVENT (albuterol/ipratropium) albuterol/ipratropium<br />
DUONEB (albuterol/ipratropium)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Phosphodiesterase Inhibitors<br />
DALIRESP (roflumilast) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
COUGH <strong>AND</strong> COLD AGENTS<br />
See Separate Preferred Cough and Cold Agent Listing.<br />
CYTOKINE <strong>AND</strong> CAM ANTAGONISTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
CIMZIA (certolizumab)<br />
ENBREL (etanercept)<br />
HUMIRA (adalimumab)<br />
ACTEMRA (tocilizumab)<br />
KINERET (anakinra)<br />
ORENCIA SC (abatacept)<br />
SIMPONI (golimumab)<br />
STELARA (ustekinumab)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 23 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
EPINEPHRINE, SELF‐INJECTED<br />
Preferred Agents Non‐Preferred Agents<br />
EPIPEN, EPIPEN JR ■ Treatment failure with preferred<br />
products<br />
■ Contraindication to preferred<br />
products<br />
■ Allergic reaction to preferred<br />
products<br />
ERYTHROPOIESIS STIMULATING PROTEINS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
EPOGEN (RhUEPO)<br />
PROCRIT (RhUEPO)<br />
ARANESP (darbepoetin) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
CIPRO suspension (ciprofloxacin)<br />
ciprofloxacin IR<br />
levofloxacin tablets<br />
AVELOX (moxifloxacin)<br />
CIPRO (ciprofloxacin)<br />
ciprofloxacin ER<br />
FACTIVE (gemifloxacin)<br />
LEVAQUIN tablets and solution<br />
(levofloxacin)<br />
levofloxacin solution<br />
NOROXIN (norfloxacin)<br />
ofloxacin<br />
PROQUIN XR (ciprofloxacin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 24 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
GLUCOCORTICOIDS, INHALED<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Glucocorticoids<br />
ASMANEX (mometasone)<br />
FLOVENT (fluticasone)<br />
PULMICORT Flexihaler (budesonide)<br />
PULMICORT Respules<br />
QVAR (beclomethasone)<br />
ADVAIR (fluticasone/salmeterol)<br />
DULERA (mometasone/formoterol)<br />
SYMBICORT (budesonide/formoterol)<br />
AEROBID (flunisolide)<br />
AEROBID‐M (flunisolide)<br />
ALVESCO (ciclesonide)<br />
AZMACORT (triamcinolone)<br />
Budesonide<br />
Glucocorticoid/Bronchodilator Combinations<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
GROWTH HORMONE<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
GENOTROPIN (somatropin)<br />
NUTROPIN AQ (somatropin)<br />
SAIZEN (somatropin)<br />
HUMATROPE (somatropin)<br />
NORDITROPIN (somatropin)<br />
NUTROPIN (somatropin)<br />
OMNITROPE (somatropin)<br />
SEROSTIM (somatropin)<br />
TEV‐TROPIN (somatropin)<br />
ZORBTIVE (somatropin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
HEPATITIS C AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Pegylated Interferons<br />
PEGASYS (pegylated IFN alfa‐2a)<br />
PEG‐INTRON (pegylated IFN alfa‐2b)<br />
INFERGEN (consensus IFN) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 25 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
HEPATITIS C AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Protease Inhibitors<br />
INCIVEK (telaprevir)<br />
VICTRELIS (boceprevir)<br />
REBETOL solution<br />
ribavirin<br />
Ribavirin<br />
COPEGUS<br />
RIBAPAK<br />
RIBASPHERE 400, 600 mg<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
HYPOGLYCEMICS, INCRETIN MIMETICS/ENHANCERS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Amylin Analogs<br />
SYMLIN (pramlintide) Patient must meet all of the following<br />
criteria:<br />
Incretin Enhancers<br />
■ Diagnosis of diabetes mellitus<br />
■ Age >18 years<br />
■ HbA1C in past 6 months<br />
■ No history of gastroparesis,<br />
neurologic manifestations of<br />
diabetes or recent treatment of<br />
hypoglycemia<br />
JENTADUETO (linagliptin/metformin)<br />
KOMBIGLYZE XR<br />
(saxagliptin/metformin)<br />
ONGLYZA (saxagliptin)<br />
TRADJENTA (linagliptin)<br />
JANUMET (sitagliptin/metformin)<br />
JANUMET XR (sitagliptin/metformin)<br />
JANUVIA (sitagliptin)<br />
JUVISYNC (sitagliptin/simvastatin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 26 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
HYPOGLYCEMICS, INCRETIN MIMETICS/ENHANCERS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Incretin Mimetics<br />
BYETTA (exenatide)<br />
SYMLIN (pramlintide)<br />
BYDUREON (exenatide ER)<br />
VICTOZA (liraglutide)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
HYPOGLYCEMICS, INSULIN<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
HUMALOG vials (insulin lispro)<br />
HUMALOG MIX vials (insulin<br />
lispro/lispro protamine)<br />
LANTUS (insulin glargine)<br />
NOVOLIN (insulin)<br />
NOVOLOG (insulin aspart)<br />
NOVOLOG MIX (insulin aspart/aspart<br />
protamine)<br />
APIDRA (insulin glulisine)<br />
HUMALOG pens (insulin lispro)<br />
HUMALOG MIX pens (insulin<br />
lispro/lispro protamine)<br />
HUMULIN (insulin)<br />
LEVEMIR (insulin detemir)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
HYPOGLYCEMICS, MEGLITINIDES<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
nateglinide<br />
PR<strong>AND</strong>IN (repaglinide)<br />
PR<strong>AND</strong>IMET (repaglinide/metformin)<br />
STARLIX (nateglinide)<br />
■ Separate prescriptions for the<br />
individual components should be<br />
used instead of the combination<br />
drug.<br />
HYPOGLYCEMICS, TZD<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Thiazolinediones<br />
ACTOS (pioglitazone)<br />
AV<strong>AND</strong>IA (rosiglitazone)<br />
pioglitazone<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 27 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
HYPOGLYCEMICS, TZD<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
TZD Combinations<br />
ACTOPLUS MET<br />
(pioglitazone/metformin)<br />
AV<strong>AND</strong>AMET<br />
(rosiglitazone/metformin)<br />
AV<strong>AND</strong>ARYL<br />
(rosiglitazone/glimepiride)<br />
DUETACT (pioglitazone/glimepiride)<br />
pioglitazone/metformin<br />
■ Separate prescriptions for the<br />
individual components should be<br />
used instead of the combination<br />
drug.<br />
IMMUNOMODULATORS, ATOPIC DERMATITIS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ELIDEL (pimecrolimus) PROTOPIC (tacrolimus)<br />
INTRANASAL RHINITIS AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Glucocorticoids<br />
flunisolide<br />
fluticasone<br />
NASONEX (mometasone)<br />
BECONASE AQ (beclomethasone)<br />
FLONASE (fluticasone propionate)<br />
NASACORT AQ (triamcinolone)<br />
NASAREL (flunisolide)<br />
OMNARIS (ciclesonide)<br />
QNASL (beclomethasone<br />
dipropionate)<br />
RHINOCORT AQUA (budesonide)<br />
triamcinolone<br />
VERAMYST (fluticasone<br />
furoate)isolide)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 28 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
INTRANASAL RHINITIS AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Others<br />
ASTEPRO (azelastine)<br />
PATANASE (olopatadine)<br />
ASTELIN (azelastine)<br />
ATROVENT nasal spray (ipratropium)<br />
Azelastine<br />
DYMISTA (azelastine/fluticasone)<br />
ipratropium nasal spray<br />
ZETONNA (ciclesonide)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
See Separate Listing Of Preferred Oral Iron Drugs.<br />
IRON, ORAL<br />
LEUKOTRIENE MODIFIERS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ACCOLATE (zafirlukast)<br />
montelukast tab and chew tab<br />
SINGULAIR GRANULES (montelukast)<br />
SINGULAIR tab and chew tab<br />
(montelukast)<br />
zafirlukast<br />
ZYFLO, ZYFLO CR (zileuton)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
LIPOTROPICS, OTHER<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Bile Acid Sequestrants<br />
cholestyramine<br />
Colestipol tablet<br />
COLESTID granules (colestipol)<br />
COLESTID tablets (colestipol)<br />
Colestipol granules<br />
QUESTRAN (cholestyramine)<br />
WELCHOL (colesevalam)<br />
Cholesterol Absorption Inhibitors<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ZETIA (ezetimibe) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 29 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
LIPOTROPICS, OTHER<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Fibric Acid Derivatives<br />
gemfibrozil<br />
TRICOR (fenofibrate)<br />
TRILIPIX (fenofibric acid)<br />
niacin<br />
NIACOR (niacin)<br />
NIASPAN (niacin)<br />
ANTARA (fenofibrate)<br />
fenofibrate<br />
fenofibric acid<br />
FENOGLIDE (fenofibrate)<br />
FIBRICOR (fenofibric acid)<br />
LIPOFEN (fenofibrate)<br />
LOFIBRA (fenofibrate)<br />
LOPID (gemfibrozil)<br />
TRIGLIDE (fenofibrate)<br />
Niacin<br />
Omega‐3 Fatty Acids<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
LOVAZA (omega‐3 fatty acids) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 30 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
LIPOTROPICS, STATINS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Statins<br />
atorvastatin<br />
LESCOL (fluvastatin)<br />
LESCOL XL (fluvastatin)<br />
pravastatin<br />
simvastatin<br />
ALTOPREV (lovastatin)<br />
CRESTOR (rosuvastatin)<br />
fluvastatin<br />
LIPITOR (atorvastatin)<br />
LIVALO (pitavastatin)<br />
lovastatin<br />
MEVACOR (lovastatin)<br />
PRAVACHOL (pravastatin)<br />
ZOCOR (simvastatin)<br />
Statin Combinations<br />
■ Treatment failure with at least two<br />
preferred drugs accounting for no<br />
less than 120 days of therapy<br />
combined<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
SIMCOR (simvastatin/niacin) ADVICOR (lovastatin/niacin)<br />
CADUET (atorvastatin/amlodipine)<br />
VYTORIN (simvastatin/ezetimibe)<br />
■ Treatment failure with at least two<br />
preferred drugs accounting for no<br />
less than 120 days of therapy<br />
combined<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
MACROLIDES/KETOLIDES (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Ketolides<br />
KETEK (telithromycin) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 31 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
MACROLIDES/KETOLIDES (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Macrolides<br />
azithromycin<br />
BIAXIN suspension (clarithromycin)<br />
clarithromycin IR<br />
clarithromycin suspension<br />
EES (erythromycin)<br />
ERY‐TAB (erythromycin)<br />
ERYTHROCIN (erythromycin)<br />
erythromycin<br />
PCE (erythromycin)<br />
Z‐MAX (azithromycin)<br />
BIAXIN tablets (clarithromycin)<br />
BIAXIN XL (clarithromycin)<br />
clarithromycin ER<br />
ERYPED (erythromycin)<br />
ZITHROMAX (azithromycin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
NEUROPATHIC PAIN<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Oral Agents<br />
CYMBALTA (duloxetine)<br />
LYRICA (pregabalin)<br />
SAVELLA (milnacipran)<br />
HORIZANT (gabapentin enacarbil ER) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
capsaicin OTC<br />
LIDODERM (lidocaine)<br />
Topical Agents<br />
QUTENZA (casaicin)<br />
ZOSTRIX DIABETIC CREAM (capsacin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 32 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
NSAIDS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Nonspecific<br />
diclofenac<br />
etodolac<br />
flurbiprofen<br />
ibuprofen (except Rx suspension)<br />
INDOCIN suspension (indomethacin)<br />
Indomethacin<br />
ketoprofen<br />
ketorolac<br />
naproxen<br />
piroxicam<br />
sulindac<br />
ANAPROX (naproxen)<br />
CATAFLAM (diclofenac)<br />
CLINORIL (sulindac)<br />
DAYPRO (oxaprozin)<br />
diclofenac sodium<br />
diclofenac SR<br />
diflunisal<br />
etodolac SR<br />
FELDENE (piroxicam)<br />
fenoprofen<br />
ibuprofen Rx suspension<br />
INDOCIN (indomethacin) (excluding<br />
suspension<br />
Indomethacin ER<br />
ketoprofen ER<br />
meclofenamate<br />
mefenamic acid<br />
MOTRIN (ibuprofen)<br />
nabumetone<br />
NALFON (fenoprofen)<br />
NAPRELAN (naproxen)<br />
NAPROSYN (naproxen)<br />
naproxen EC<br />
oxaprozin<br />
PONSTEL (meclofenamate)<br />
SPRIX (ketorolac)<br />
tolmetin<br />
VOLTAREN (diclofenac)<br />
VOLTAREN XR (diclofenac)<br />
ZIPSOR (diclofenac)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
NSAID/GI Protectant Combinations<br />
ARTHROTEC (diclofenac/misoprostol)<br />
DUEXIS (ibuprofen/famotidine)<br />
VIMOVO (naproxen/ esomeprazole)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
COX‐II Selective<br />
Meloxicam tablets CELEBREX (celecoxib)<br />
Meloxicam suspension<br />
MOBIC (meloxicam)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 33 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
NSAIDS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Topical NSAIDs<br />
VOLTAREN GEL (diclofenac) FLECTOR (diclofenac)<br />
PENNSAID (diclofenac)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
OPHTHALMICS, ANTIBIOTIC – STEROID COMBINATIONS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
BLEPHAMIDE<br />
(sulfacetamide/prednisolone)<br />
MAXITROL drops<br />
(neomycin/polymyxin/<br />
dexamethasone)<br />
neomycin/polymyxin/<br />
dexamethasone<br />
sulfacetamide/prednisolone<br />
TOBRADEX<br />
(tobramycin/dexamethasone)<br />
MAXITROL Ointment<br />
(neomycin/polymyxin/<br />
dexamethasone)<br />
neomycin/bacitracin/polymyxin/hydr<br />
ocortisone<br />
neomycin/polymyxin/ hydrocortisone<br />
PRED‐G (gentamicin/prednisolone)<br />
TOBRADEX ST<br />
(tobramycin/dexamethasone)<br />
tobramycin/dexamethasone<br />
ZYLET (tobramycin/loteprednol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 34 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
OPHTHALMICS FOR ALLERGIC CONJUNCTIVITIS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
cromolyn<br />
PATADAY (olopatadine)<br />
ALAMAST (pemirolast)<br />
ALOMIDE (lodoxamide)<br />
ALREX (loteprednol)<br />
ALOCRIL (nedocromil)<br />
azelastine<br />
BEPREVE (bepotastine)<br />
CROLOM (cromolyn)<br />
ELESTAT (epi EMADINE (emedastine)<br />
Epinastine<br />
ketotifen<br />
LASTACAFT (alcaftadine)<br />
OPTIVAR (azelastine)<br />
PATANOL (olopatadine)<br />
ZADITOR (ketotifen)nastine)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
OPHTHALMIC ANTIBIOTICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Aminoglycosides<br />
gentamicin<br />
tobramycin<br />
TOBREX Ointment (tobramycin)<br />
BESIVANCE (besifloxacin)<br />
MOXEZA (moxifloxacin)<br />
ciprofloxacin<br />
ofloxacin<br />
VIGAMOX (moxifloxacin)<br />
GARAMYCIN<br />
TOBREX Solution (tobramycin)<br />
Quinolones<br />
CILOXAN (ciprofloxacin)<br />
IQUIX (levofloxacin)<br />
Levofloxacin<br />
OCUFLOX (ofloxacin)<br />
QUIXIN (levofloxacin)<br />
ZYMAR (gatifloxacin)<br />
ZYMAXID (gatifloxacin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 35 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
OPHTHALMIC ANTIBIOTICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Macrolides<br />
erythromycin AZASITE (azithromycin)<br />
ILOTYCIN<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
bacitracin/polymyxin<br />
neomycin/polymyxin/gramicidin<br />
polymyxin/trimethoprim<br />
sulfacetamide solution<br />
Other<br />
bacitracin<br />
BLEPH‐10 (sulfacetamide)<br />
NATACYN (natamycin)<br />
neomycin/bacitracin/polymyxin<br />
NEOSPORIN<br />
(neomycin/polymyxin/gramicidin)<br />
POLYTRIM (polymyxin/trimethoprim)<br />
sulfacetamide ointment<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
OPHTHALMICS, GLAUCOMA AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Sympathomimetics<br />
brimonidine<br />
IOPIDINE (apraclonidine)<br />
pilocarpine<br />
betaxolol<br />
BETIMOL (timolol)<br />
Carteolol<br />
levobunolol<br />
metipranolol<br />
timolol<br />
ALPHAGAN P (brimonidine)<br />
apraclonidine<br />
brimonidine P<br />
Beta Blockers<br />
BETAGAN (levobunolol)<br />
BETOPTIC S (betaxolol)<br />
ISTALOL (timolol)<br />
OPTIPRANOLOL (metipranolol)<br />
TIMOPTIC (timolol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 36 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
OPHTHALMICS, GLAUCOMA AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Carbonic Anhydrase Inhibitors<br />
AZOPT (brinzolamide)<br />
dorzolamide<br />
TRUSOPT (dorzolamide) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
latanoprost<br />
TRAVATAN‐Z (travoprost)<br />
Prostaglandin Analogs<br />
LUMIGAN (bimatoprost)<br />
XALATAN (latanoprost)<br />
ZIOPTAN (tafluprost)<br />
Combination Agents<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
COMBIGAN (brimonidine/timolol)<br />
dorzolamide/timolol<br />
COSOPT (dorzolamide/timolol) ■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
OPHTHALMICS, ANTI‐INFLAMMATORIES<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
NSAIDS<br />
diclofenac<br />
flurbiprofen<br />
NEVANAC (nepafenac)<br />
ACULAR (ketorolac)<br />
ACULAR LS (ketorolac)<br />
ACUVAIL (ketorolac)<br />
BROMDAY (bromfenac)<br />
bromfenac<br />
ketorolac<br />
ketorolac LS<br />
OCUFEN (flurbiprofen)<br />
XIBROM (bromfenac)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 37 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
OPHTHALMICS, ANTI‐INFLAMMATORIES<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Steroids<br />
dexamethasone<br />
LOTEMAX (loteprednol)<br />
prednisolone<br />
DUREZOL (difluprednate)<br />
FLAREX (fluorometholone)<br />
FML (fluorometholone)<br />
FML FORTE (fluorometholone)<br />
FML S.O.P. (fluorometholone)<br />
MAXIDEX (dexamethasone)<br />
OMNIPRED (prednisolone)<br />
PRED FORTE (prednisolone)<br />
PRED MILD (prednisolone)<br />
VEXOL (rimexolone)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
OPIATE DEPENDENCE TREATMENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Buprenorpine<br />
naltrexone<br />
SUBOXONE FILM<br />
(buprenorphine/naloxone)<br />
SUBOXONE TABLETS<br />
(buprenorphine/naloxone)<br />
OTIC ANTIBIOTICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
CIPRODEX<br />
(ciprofloxacin/dexamethasone)<br />
neomycin/polymyxin/<br />
hydrocortisone<br />
ofloxacin<br />
CIPRO HC<br />
(ciprofloxacin/hydrocortisone)<br />
COLY‐MYCIN S (colistin/neomycin/<br />
hydrocortisone)<br />
CORTISPORIN SOLUTION<br />
(neomycin/polymixin B<br />
sulfates/hydrocortisone)<br />
CORTISPORIN‐TC (colistin/neomycin/<br />
hydrocortisone)<br />
CETRAXAL (ciprofloxacin)<br />
FLOXIN (ofloxacin)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 38 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
OTIC ANTI‐INFECTIVES/ANESTHETICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
acetic acid<br />
antipyrine/benzocaine<br />
OTOZIN (antipyrine/benzocaine/zinc)<br />
VOSOL HC (acetic<br />
acid/hydrocortisone)<br />
acetic acid/hydrocortisone<br />
acetic acid/aluminum<br />
MYOXIN (chloroxylenol/benzocaine/<br />
hydrocortisone)<br />
NEOTIC (antipyrine/benzocaine/<br />
glycerin)<br />
TREAGAN (antipyrine/benzocaine/ u‐<br />
Polycosanol)<br />
TRIOXIN (chloroxylenol/benzocaine/<br />
hydrocortisone)<br />
ZINOTIC<br />
(chloroxylenol/pramoxine/zinc)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
PAH AGENTS (ORAL, INHALATION)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
ADCIRCA (tadalafil)<br />
LETAIRIS (ambrisentan)<br />
TRACLEER (bosentan)<br />
REVATIO (sildenafil)<br />
TYVASO Inhalation (treprostinil)<br />
VENTAVIS Inhalation (iloprost)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
PANCREATIC ENZYMES<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
CREON (pancrelipase)<br />
pancrelipase<br />
ZENPEP (pancrelipase)<br />
PANCREAZE (pancrelipase)<br />
PERTZYE (pancrelipase)<br />
VIOKACE (pancrelipase)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 39 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
PENICILLINS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
amoxicillin<br />
ampicillin<br />
dicloxacillin<br />
penicillin VK<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
PHOSPHATE BINDERS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
calcium acetate<br />
CALPHRON OTC (calcium acetate)<br />
ELIPHOS (calcium acetate)<br />
MAGNEBIND 400 (magnesium,<br />
calcium, folic acid)<br />
PHOSLO (calcium acetate)<br />
PHOSLYRA (calcium acetate)<br />
RENAGEL (sevelamer HCl)<br />
FOSRENOL (lanthanum)<br />
RENVELA (sevelamer carbonate)<br />
Diagnosis of ESRD and<br />
hyperphosphatemia despite dietary<br />
phosphorous restrictions and at least<br />
one of the following:<br />
■ hypercalcemia (corrected serum<br />
calcium >10.2 mg/dL)<br />
■ plasma PTH levels
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
PRENATAL VITAMINS<br />
See Separate Preferred Prenatal Vitamin Listing.<br />
PA Criteria:<br />
■ Prenatal vitamins are covered only for females less than 50 years of age.<br />
PROTON PUMP INHIBITORS (ORAL)<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
pantoprazole<br />
PROTONIX suspension (pantoprazole)<br />
NEXIUM (esomeprazole)<br />
ZEGERID OTC (omeprazole/sodium<br />
bicarbonate)<br />
ACIPHEX (rabeprazole)<br />
DEXILANT (dexlansoprazole)<br />
Lansoprazole<br />
omeprazole<br />
PREVACID (lansoprazole)<br />
PRILOSEC (omeprazole)<br />
PROTONIX (pantoprazole)<br />
■ Treatment failure after no less than<br />
a 30 day trial of each preferred<br />
drug<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Prevacid Solutabs will be approved<br />
for children 10 years of age and<br />
under<br />
SEDATIVE HYPNOTICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Benzodiazepines<br />
flurazepam<br />
temazepam 15, 30 mg<br />
triazolam<br />
DORAL (quazepam)<br />
estazolam<br />
HALCION (triazola)<br />
RESTORIL (temazepam)<br />
temazepam 7.5, 22.5 mg<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 41 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
SEDATIVE HYPNOTICS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Others<br />
chloral hydrate<br />
LUNESTA (eszopiclone)<br />
zolpidem<br />
AMBIEN (zolpidem)<br />
AMBIEN CR (zolpidem)<br />
EDLUAR (zolpidem)<br />
INTERMEZZO (zolpidem)<br />
ROZEREM (ramelteon)<br />
SILENOR (doxepin)<br />
SONATA (zaleplon)<br />
SOMNOTE (chloral hydrate)<br />
zaleplon<br />
zolpidem ER<br />
ZOLPIMIST (zolpidem)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
SKELETAL MUSCLE RELAXANTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
baclofen<br />
carisoprodol (except 250 mg)<br />
carisoprodol compound<br />
chlorzoxazone<br />
cyclobenzaprine IR<br />
methocarbamol<br />
tizanidine tablet<br />
AMRIX (cyclobenzaprine ER)<br />
carisoprodol 250 mg<br />
cyclobenzaprine ER<br />
DANTRIUM (dantrolene)<br />
dantrolene<br />
FEXMID (cyclobenzaprine)<br />
LORZONE (chlorzoxazone)<br />
metaxolone<br />
orphenadrine<br />
orphenadrine compound<br />
PARAFON FORTE DSC (chlorzoxazone)<br />
ROBAXIN (methocarbamol)<br />
SKELAXIN (metaxolone)<br />
SOMA (carisoprodol)<br />
tizanidine capsule<br />
ZANAFLEX (tizanidine)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 42 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
STEROIDS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Low Potency<br />
DERMA‐SMOOTHE/FS (fluocinolone)<br />
desonide cream and ointment<br />
hydrocortisone lotion (OTC)<br />
hydrocortisone cream & ointment<br />
(OTC & RX)<br />
hydrocortisone/aloe cream &<br />
ointiment (OTC)<br />
hydrocortisone/mineral oil ointment<br />
hydrocortisone butyrate<br />
hydrocortisone valerate<br />
fluticasone propionate<br />
mometasone furoate cream &<br />
ointment<br />
ACLOVATE (alclometasone)<br />
Alclometasone<br />
AQUA GLYCOLIC HC (hydrocortisone)<br />
CAPEX (fluocinolone)<br />
DESONATE (desonide)<br />
desonide lotion<br />
DESOWEN (desonide)<br />
fluocinolone oil<br />
hydrocortisone acetate/urea<br />
hydrocortisone lotion (RX)<br />
hydrocortisone/aloe gel<br />
NUZON (hydrocortisone/aloe)<br />
PEDIADERM HC (hydrocortisone)<br />
PEDIADERM TA (triamcinolone)<br />
TEXACORT (hydrocortisone sol)<br />
VERDESO (desonide)<br />
Medium Potency<br />
CLODERM (clocortolone)<br />
CORDRAN (flurandrenolide)<br />
CUTIVATE (fluticasone)<br />
DERMATOP (prednicarbate)<br />
ELECON (mometasone)<br />
flucinolone acetonide<br />
LUXIQ (betamethasone)<br />
mometasone furoate solution<br />
MOMEXIN (mometasone)<br />
P<strong>AND</strong>EL (hydrocortisone probutate)<br />
Prednicarbate<br />
WESTCORT (hydrocortisone valerate)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 43 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
STEROIDS, TOPICAL<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
High Potency<br />
betamethasone valerate<br />
fluocinonide<br />
triamcinolone<br />
clobetasol propionate cream,<br />
ointment, gel, emollient, solution<br />
halobetasol<br />
amcinonide<br />
betamethasone dipropionate<br />
betamethasone dipropionat/<br />
propylene glycol<br />
BETA‐VAL (betamethasone valerate)<br />
desoximetasone<br />
diflorasone<br />
DIPROLENE (betamethasone<br />
dipropionate)<br />
HALOG (halcinonide)<br />
KENALOG aerosol (triamcinolone)<br />
TOPICORT (desoximetasone)<br />
VANOS (fluocinonide)<br />
Very High Potency<br />
APEXICON E (diflorasone)<br />
clobetasol lotion, foam, shampoo<br />
CLOBEX (clobetasol)<br />
HALAC (halobetasol)<br />
HALONATE (halobetasol)<br />
OLUX‐E (clobetasol)<br />
TEMOVATE (clobetasol)<br />
ULTRAVATE (halobetasol)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 44 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
STIMULANTS <strong>AND</strong> RELATED AGENTS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Stimulants<br />
ADDERALL XR (amphetamine salt<br />
combination)<br />
amphetamine salt combination IR<br />
DAYTRANA (methylphenidate)<br />
dexmethylphenidate<br />
dextroamphetamine<br />
FOCALIN XR (dexmethylphenidate)<br />
METHYLIN (methylphenidate)<br />
methylphenidate PPG<br />
methylphenidate SA (generic<br />
Concerta)<br />
methylphenidate solution<br />
PROCENTRA (dextroamphetamine)<br />
VYVANSE (lisdexamfetamine)<br />
INTUNIV (guanfacine ER)<br />
KAPVAY (clonidine ER)<br />
STRATTERA (atomoxetine)<br />
ADDERALL (amphetamine salt<br />
combination)<br />
amphetamine salt combination ER<br />
CONCERTA (methylphenidate)<br />
DEXEDRINE (dextroamphetamine)<br />
DEXEDRINE SPANSULES<br />
(dextroamphetamine)<br />
FOCALIN (dexmethylphenidate)<br />
METADATE CD (methylphenidate)<br />
METHYLIN (methylphenidate)<br />
chewable tablets<br />
NUVIGIL (armodafinil)<br />
PROVIGIL (modafinil)<br />
RITALIN (methylphenidate)<br />
Non‐Stimulants<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 45 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
TETRACYCLINES<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
doxycycline hyclate IR<br />
doxycycline monohydrate capsules<br />
(except 75 mg and 150 mg)<br />
doxycycline monohydrate tablets<br />
minocycline capsules<br />
tetracycline<br />
VIBRAMYCIN suspension<br />
(doxycycline)<br />
ADOXA (doxycycline)<br />
demeclocycline<br />
DORYX (doxycycline)<br />
doxycycline hyclate DR<br />
doxycycline monohydrate capsules75<br />
mg and 150 mg<br />
minocycline ER<br />
minocycline tablets<br />
NUTRIDOX (doxycycline)<br />
ORACEA (doxycycline)<br />
SOLODYN (minocycline)<br />
VIBRAMYCIN capsule and syrup<br />
(doxycycline)<br />
■ Treatment failure with preferred<br />
drugs within any subclass<br />
■ Contraindication to preferred drugs<br />
■ Allergic reaction to preferred drugs<br />
ULCERATIVE COLITIS<br />
Preferred Agents Non‐Preferred Agents PA Criteria<br />
Oral<br />
ASACOL (mesalamine)<br />
balsalazide<br />
sulfasalazine<br />
sulfasalazine DR<br />
SFROWASA (mesalamine)<br />
mesalamine<br />
APRISO (mesalamine)<br />
ASACOL HD (mesalamine)<br />
AZULFIDINE (sulfasalazine)<br />
COLAZAL (balsalazide)<br />
DIPENTUM (olsalazine)<br />
LIALDA (mesalamine)<br />
PENTASA (mesalamine)<br />
Rectal<br />
CANASA (mesalamine)<br />
mesalamine kit<br />
ROWASA (mesalamine)<br />
■ Treatment failure with preferred<br />
drugs within any subclass of same<br />
route<br />
■ Contraindication to preferred drugs<br />
of same route<br />
■ Allergic reaction to preferred drugs<br />
of same route<br />
■ Treatment failure with preferred<br />
drugs within any subclass of same<br />
route<br />
■ Contraindication to preferred drugs<br />
of same route<br />
■ Allergic reaction to preferred drugs<br />
of same route<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 46 of 47
<strong>HEALTH</strong> <strong>AND</strong> <strong>HUMAN</strong> <strong>SERVICES</strong> <strong>COMMISSION</strong><br />
<strong>TEXAS</strong> MEDICAID PREFERRED DRUG LIST (PDL) and PRIOR AUTHORIZATION (PA) CRITERIA<br />
Effective January 23, 2013<br />
PREMIUM PREFERRED GENERIC () MANUFACTURERS: These manufacturers have offered Supplemental Rebates to the state for their PDL drugs dispensed to Medicaid recipients. Pharmacists will<br />
receive an additional $0.50 dispensing fee when they dispense the PDL drugs of these manufacturers.<br />
Generic Manufacturer<br />
Mallinckrodt 00406<br />
Labeler Code(s)<br />
Sun Pharma Glob 41616<br />
Unless otherwise specified, the listing of a particular brand or generic name includes all dosage forms of that drug. January 23, 2013<br />
Page 47 of 47
Cough and Cold Drugs (Oral Only)<br />
PREFERRED Ingredients NON-PREFERRED Ingredients<br />
ALA-HIST PE OTC DEXBROMPHENIRAMIN/PHENYLEPHRIN ALDEX-CT PHENYLEPHRINE/DIPHENHYDRAMINE<br />
ALDEX GS OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL BROTAPP LIQUID OTC PSEUDOEPHEDRINE/BROMPHENIRAMIN<br />
BROVEX PEB PHENYLEPHRINE/BROMPHENIRAMINE CENTERGY PHENYLEPHRINE/CHLORPHENIRAMINE<br />
BROVEX PSB PSEUDOEPHEDRINE/BROMPHENIRAMIN CPM-PSE PSEUDOEPHED/CHLORPHENIRAMINE<br />
BROVEX PSE PSEUDOEPHEDRINE/BROMPHENIRAMIN DALLERGY-JR PHENYLEPHRINE/CHLOR-TAN<br />
CARDEC OTC PHENYLEPHRINE/CHLORPHENIRAMINE DELSYM NIGHTTIME COUGH & COLD OTC PHENYLEPHRINE/DIPHENHYDRAMINE<br />
CHILD DELSYM COUGH-COLD NIGHT OTC PHENYLEPHRINE/DIPHENHYDRAMINE DICEL P-EPD TAN/CHLOR-TAN<br />
CHILD'S MUCINEX OTC GUAIFENESIN DICEL OTC PSEUDOEPHED/CHLORPHENIRAMINE<br />
DALLERGY DROPS OTC PHENYLEPHRINE/CHLORPHENIRAMINE DONATUSSIN OTC GUAIFENESIN/PHENYLEPHRINE HCL<br />
DALLERGY OTC CHLORCYCLIZINE/PHENYLEPHRINE DRYMAX PSEUDOEPHEDRINE/CPM/METHSCOPOL<br />
DECONEX IR OTC GUAIFENESIN/PHENYLEPHRINE HCL EFFERVESCENT COLD RELIEF OTC PHENYLEPHRINE/ACETAMINOPHEN/CP<br />
DECONEX OTC GUAIFENESIN/PHENYLEPHRINE HCL EXEFEN IR GUAIFENESIN/PSEUDOEPHEDRNE HCL<br />
ED A-HIST LIQUID OTC PHENYLEPHRINE/CHLORPHENIRAMINE EXEFIN IR TABLET OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL<br />
ED A-HIST TABLET OTC PHENYLEPHRINE/CHLORPHENIRAMINE GUAIFENESIN 200 MG TABLET OTC GUAIFENESIN<br />
ED BRON GP LIQUID OTC GUAIFENESIN/PHENYLEPHRINE HCL GUAIFENESIN 600 MG TABLET OTC GUAIFENESIN<br />
ED CHLORPED D PHENYLEPHRINE/CHLOR-TAN GUAIFENESIN/PHENYLEPHRINE HCL DROPS GUAIFENESIN/PHENYLEPHRINE HCL<br />
ED CHLORPED D OTC PHENYLEPHRINE/CHLORPHENIRAMINE HIST-PSE SOLUTION OTC P-EPHED HCL/TRIPROLIDINE HCL<br />
ENTEX LQ OTC GUAIFENESIN/PHENYLEPHRINE HCL IBUPROFEN/PSEUDOEPHEDRINE OTC IBUPROFEN/PSEUDOEPHEDRINE HCL<br />
ENTEX T OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL IBUPROFEN/PSEUDOEPHEDRINE SUSPENSION OTC IBUPROFEN/PSEUDOEPHEDRINE HCL<br />
GUAIFENESIN 400 MG TABLET OTC GUAIFENESIN J-MAX GUAIFENESIN/PHENYLEPHRINE HCL<br />
GUAIFENESIN LIQUID OTC GUAIFENESIN J-TAN D ORAL SUSP PHENYLEPHRINE/BROMPHENIRAMIN<br />
GUAIFENESIN/PHENYLEPHRINE TABLET OTC GUAIFENESIN/PHENYLEPHRINE HCL J-TAN D PD OTC PSEUDOEPHEDRINE/BROMPHENIRAMIN<br />
KID'S MUCINEX MINI-MELTS OTC GUAIFENESIN LODRANE D OTC PSEUDOEPHEDRINE/BROMPHENIRAMIN<br />
LOHIST 12D PSEUDOEPHEDRINE/BROMPHENIRAMIN MAXICHLOR PEH PHENYLEPHRINE/CHLORPHENIRAMINE<br />
LOHIST DROPS OTC PHENYLEPHRINE/CHLORPHENIRAMINE MAXIFED GUAIFENESIN/PSEUDOEPHEDRNE HCL<br />
LOHIST-D OTC PSEUDOEPHED/CHLORPHENIRAMINE MAXIFED-G GUAIFENESIN/PSEUDOEPHEDRNE HCL<br />
LOHIST-PEB OTC PHENYLEPHRINE/BROMPHENIRAMINE MAXIPHEN GUAIFENESIN/PHENYLEPHRINE HCL<br />
LOHIST-PSB OTC PSEUDOEPHEDRINE/BROMPHENIRAMIN NEUTRAHIST DROPS OTC PSEUDOEPHED/CHLORPHENIRAMINE<br />
LORTUSS LQ OTC DOXYLAMINE/PSEUDOEPHEDRINE HCL NIGHTTIME SINUS OTC PHENYLEPHRINE/ACETAMIN/DOXYLAM<br />
LUSAIR GUAIFENESIN/PHENYLEPHRINE HCL NOREL SR PHENYLEPH/ACETAMINOP/P-TLOX/CP<br />
MEDI-TUSSIN OTC GUAIFENESIN PAIN RELIEF SINUS PE OTC PHENYLEPHRINE HCL/ACETAMINOPHN<br />
MUCINEX COLD & SINUS OTC GUAIFEN/PHENYLEPH/ACETAMINOPHN PEDIATEX TD LIQUID OTC P-EPHED HCL/TRIPROLIDINE HCL<br />
MUCINEX COLD LIQUID OTC GUAIFENESIN/PHENYLEPHRINE HCL PHENA-S PHENYLEPHRINE/PYRILAMINE MA/CP<br />
MUCINEX D ER OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL PHENA-S 12 PE/PE & PYRILAMINE TANNATE/CPM<br />
MUCINEX D OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL PHENYLEPHRINE/CHLOR-TAN PHENYLEPHRINE/CHLOR-TAN<br />
MUCINEX ER TABLET OTC GUAIFENESIN PHENYLEPHRINE/CHLORPENIRAMINE TABLET OTC PHENYLEPHRINE/CHLORPHENIRAMINE<br />
NASOHIST DROPS OTC PHENYLEPHRINE/CHLORPHENIRAMINE PHENYLTOLOXAMINE PE CPM PHENYLEPHRINE/P-TLOX CI/CP<br />
NASOPEN OTC CHLORCYCLIZINE/PSEUDOEPHEDRINE POLY HIST FORTE TABLET PHENYLEPHRINE/PYRILAMINE<br />
NASOPEN-CH OTC CHLORCYCLIZINE/PHENYLEPHRINE PROHIST LQ PHENYLEPHRINE/TRIPROLIDINE<br />
NOHIST TABLET OTC PHENYLEPHRINE/CHLORPHENIRAMINE PSEUDOEPHEDRINE/ACETAMINOPHEN TABLET OTC PSEUDOEPHEDRINE/ACETAMINOPHEN<br />
NOHIST-LQ LIQUID OTC PHENYLEPHRINE/CHLORPHENIRAMINE PYRICHLOR PE PHENYLEPHRINE/PYRILAMINE MA/CP<br />
P-EPHED/TRIPROLIDINE TAB OTC P-EPHED HCL/TRIPROLIDINE HCL R-TANNA PHENYLEPHRINE/CHLOR-TAN<br />
PHENA-PLUS PHENYLEPHRINE/PYRILAMINE MA/CP R-TANNA PEDIATRIC PHENYLEPHRINE/CHLOR-TAN<br />
PHENYLEPHRINE/BROMPHENIRAMINE ELIXIR OTC PHENYLEPHRINE/BROMPHENIRAMINE RESPA A.R. P-EPHED HCL/CHLOR-MAL/BELL ALK<br />
POLY HIST FORTE TABLET ER PHENYLEPHRINE/PYRILAMINE MA/CP RESPAHIST PSEUDOEPHEDRINE/BROMPHENIRAMIN<br />
POLY-VENT IR OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL RHINACON A PHENYLEPHRINE/P-TLOX CI/CP<br />
POLY-VENT PLUS GUAIFEN/PSEUDOEPHED/ACETAMINOP RU-TUSS P-EPHED HCL/CHLOR-MAL/BELL ALK<br />
PROMETHAZINE VC SYRUP (ACTAVIS) PHENYLEPHRINE HCL/PROMETH HCL RYNATAN PHENYLEPHRINE/CHLOR-TAN<br />
PROMETHAZINE VC SYRUP (QUALITEST) PHENYLEPHRINE HCL/PROMETH HCL RYNATAN PEDIATRIC ORAL SUSP PHENYLEPHRINE/CHLOR-TAN<br />
PSEUDOEPHEDRINE/BROMPHENIRAMINE SOLN OTC PSEUDOEPHEDRINE/BROMPHENIRAMIN RYNATAN PEDIATRIC TAB CHEW PHENYLEPHRINE/CHLOR-TAN<br />
PSEUDOEPHEDRINE/CHLORPHENIRAMINE TABLET OTC PSEUDOEPHED/CHLORPHENIRAMINE SILDEC PSEUDOEPHEDRINE/BROMPHENIRAMIN<br />
PYRIL D PHENYLEPHRINE/PYRILAMINE SINA-12X GUAIFENESIN/PHENYLEPHRINE TANN<br />
RESCON-GG LIQUID OTC GUAIFENESIN/PHENYLEPHRINE HCL SUDATEX G OTC GUAIFENESIN/PSEUDOEPHEDRNE HCL
Cough and Cold Drugs (Oral Only) Continued<br />
PREFERRED Ingredients NON-PREFERRED Ingredients<br />
RESPAIRE-30 GUAIFENESIN/PSEUDOEPHEDRNE HCL TRIP-PSE LIQUID OTC P-EPHED HCL/TRIPROLIDINE HCL<br />
RU-HIST FORTE PHENYLEPHRINE/PYRILAMINE MA/CP TRIPOHIST D P-EPHED HCL/TRIPROLIDINE HCL<br />
RYMED OTC DEXCHLORPHENIR/PHENYLEPHRINE TRITAL SR PHENYLEPH/ACETAMINOP/P-TLOX/CP<br />
SILAFED OTC P-EPHED HCL/TRIPROLIDINE HCL V-HIST PHENYLEPHRINE/BROMPHENIRAMINE<br />
VAZOBID-PD<br />
PHENYLEPHRINE/BROMPHENIRAMINE<br />
VAZOTAB<br />
PHENYLEPHRINE/BROMPHENIRAMIN<br />
VIRDEC DROPS OTC<br />
PHENYLEPHRINE/CHLORPHENIRAMINE<br />
PREFERRED<br />
NRS-NASAL RELIEF NOSE SPRAY<br />
NASAL DECONGESTANT 0.05% SPRAY<br />
NASAL SPRAY X-MOIST<br />
SM NASAL SPRAY 0.05%<br />
MUCINEX FULL FORCE NASAL SPRAY<br />
QC NASAL RELIEF 0.05% SPRAY<br />
Cough and Cold Drugs (Nasal Only)<br />
NON-PREFERRED<br />
TYZINE 0.1% NOSE SPRAY<br />
NOSE DROPS<br />
NASAL SPRAY EXTRA MOISTURIZING<br />
QC NO DRIP NASAL RLF 0.05% SPR<br />
Cough and Cold Drugs - Non-Narcotic<br />
PREFERRED Ingredients NON-PREFERRED Ingredients<br />
ALA-HIST DM OTC BROMPHENIRAMIN/PE/DEXTROMETHOR ATUSS DS D-METHORP TAN/P-EPHED TAN/CP<br />
ALDEX GS DM OTC GUAIFENESIN/DM/PSEUDOEPHEDRINE BICLORA TABLET OTC CHLORCYCLIZINE/CHLOPHEDIANOL<br />
ALLFEN DM TABLETS GUAIFENESIN/DEXTROMETHORPHAN CARBATUSS-12 CAR-PEN/CAR-PEN TAN/PE TAN/PE<br />
BENZONATATE CAPSULE OTC BENZONATATE CENTERGY DM DM/PHENYLEPH/CHLORPHENIRAMINE<br />
BICLORA LIQUID OTC CHLORCYCLIZINE/CHLOPHEDIANOL CLOFERA OTC PSEUDOEPHEDRINE/CHLOPHEDIANOL<br />
BICLORA-D LIQUID OTC CHLORCYCLIZ/PSE/CHLOPHEDIANOL COLD HEAD CONGESTION OTC D-METHORPHAN/PE/ACETAMINOPHEN<br />
BICLORA-D TABLET OTC CHLORCYCLIZ/PSE/CHLOPHEDIANOL CORZALL PLUS PYRIL MAL/P-EPHED HCL/CARBPNTN<br />
BP 8 COUGH OTC GUAIFENESIN/DM/PSEUDOEPHEDRINE CPM-PE-DM DM/PHENYLEPH/CHLORPHENIRAMINE<br />
BPM-PSE-DM LIQUID OTC D-METHORPHAN HB/P-EPD HCL/BPM CPM-PSE DM D-METHORPHAN HB/P-EPHED HCL/CP<br />
BROMFED DM SYRUP D-METHORPHAN HB/P-EPD HCL/BPM D-METHORP TAN/P-EPHED TAN/CP D-METHORP TAN/P-EPHED TAN/CP<br />
BROMPHENIRAMINE/PHENYLEPHRINE/DM ELIXIR OTC BROMPHENIRAMIN/PE/DEXTROMETHOR DAYTIME CAPSULE OTC D-METHORPHAN/PE/ACETAMINOPHEN<br />
BROTAPP DM LIQUID OTC D-METHORPHAN HB/P-EPD HCL/BPM DECONEX DM OTC GUAIFENESIN/D-METHORPHAN HB/PE<br />
BROVEX PEB DM LIQUID OTC BROMPHENIRAMIN/PE/DEXTROMETHOR DECONSAL DM DM-PE-PYRILAMINE TANNATES<br />
BROVEX PSB DM D-METHORPHAN HB/P-EPD HCL/BPM DEXTROMETHORPHAN POLISTIREX SUS ER 12H OTC DEXTROMETHORPHAN POLISTIREX<br />
CARDEC DM OTC DM/PHENYLEPH/CHLORPHENIRAMINE DICEL DM OTC D-METHORPHAN HB/P-EPHED HCL/CP<br />
CHILDREN'S MUCINEX COUGH OTC GUAIFENESIN/DEXTROMETHORPHAN DM/APAP/DOXYLAMINE LIQUID CAPSULE OTC D-METHORPHAN/ACETAMIN/DOXYLAMN<br />
CHILDREN'S MUCINEX OTC GUAIFENESIN/D-METHORPHAN HB/PE DM/APAP/DOXYLAMINE LIQUID OTC D-METHORPHAN/ACETAMIN/DOXYLAMN<br />
CHILDREN'S MUCINEX OTC PHENYLEPHRINE/DM/ACETAMINOP/GG DONATUSSIN DM OTC DM/PHENYLEPH/CHLORPHENIRAMINE<br />
CHLO TUSS EX OTC CHLOPHEDIANOL HCL/GUAIFENESIN DOXYLAMINE DM LIQUID OTC DEXTROMETHORPHAN HB/DOXYLAMINE<br />
CHLO TUSS OTC DEXBROMPHEN/PHENYLEPH/CHLOPHED ENTEX PAC COMPLETE KIT GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
CORFEN-DM LIQUID OTC DM/PHENYLEPH/CHLORPHENIRAMINE ENTRE-S D-METHORP TAN/P-EPHED TAN/CP<br />
DALLERGY DM D-METHORPHAN HB/P-EPD HCL/BPM FLUTABS PSEUDOEPH/DM/GUAIFEN/ACETAMIN<br />
DE-CHLOR DM LIQUID OTC DM/PHENYLEPH/CHLORPHENIRAMINE INDAMIX DM D-METHORP TAN/P-EPD TAN/D-CP<br />
DECONEX DMX OTC GUAIFENESIN/D-METHORPHAN HB/PE MAXICHLOR DM DEXTROMETHORPHAN HBR/CHLOR-MAL<br />
DELSYM COUGH & COLD D-METHORPHAN/ACETAMIN/DOXYLAMN MAXIFED DM GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
DELSYM MULTI-SYMPTOM OTC DM/PE/ACETAMINOPHEN/DOXYLAMINE MAXIFED DMX GUAIFENESIN/DM/PSEUDOEPHEDRINE
Cough and Cold Drugs - Non-Narcotic Continued<br />
PREFERRED Ingredients NON-PREFERRED Ingredients<br />
DELSYM NIGHTTIME MULTI-SYMPTOM OTC D-METHORPHAN/ACETAMIN/DOXYLAMN MAXIFLU DM PSEUDOEPH/DM/GUAIFEN/ACETAMIN<br />
DELSYM SUS ER 12H OTC DEXTROMETHORPHAN POLISTIREX MAXIPHEN DM GUAIFENESIN/D-METHORPHAN HB/PE<br />
DICEL CD OTC BROMPHENIRAM-PSE-CHLOPHEDIANOL NEO DM D-METHORP TAN/PE/BR-PHENIR<br />
DIMETANE-DX LIQUID OTC D-METHORPHAN HB/P-EPD HCL/BPM NEUTRAHIST PDX OTC D-METHORPHAN HB/P-EPHED HCL/CP<br />
DM/P-EPHEDRINE/CHLORPHEN LIQUID OTC D-METHORPHAN HB/P-EPHED HCL/CP NIGHT TIME COLD & FLU LIQUID OTC D-METHORPHAN/ACETAMIN/DOXYLAMN<br />
DM/PSEUDOEPHED/BROMPHEN ELIXIR OTC D-METHORPHAN HB/P-EPD HCL/BPM PHENFLU DM PHENYLEPHRINE/DM/ACETAMINOP/GG<br />
DONATUSSIN PHENYLEPHRINE/CHLOPHEDIANOL/GG PRO-CHLO LIQUID OTC PYRILAMINE/PE/CHLOPHEDIANOL<br />
DURAFLU PSEUDOEPH/DM/GUAIFEN/ACETAMIN PROHIST CD TRIPROLIDINE/PE/CHLOPHEDIANOL<br />
ED-A-HIST DM LIQUID OTC DM/PHENYLEPH/CHLORPHENIRAMINE PROHIST CF TRIPROLIDINE HCL/CHLOPHEDIANOL<br />
ENDACOF OTC DM/PHENYLEPH/CHLORPHENIRAMINE RESPERAL-DM D-METHORPHAN HB/P-EPD HCL/BPM<br />
GUAIFENESIN DM LIQUID OTC GUAIFENESIN/DEXTROMETHORPHAN RYDEX DM OTC GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
GUAIFENESIN DM TABLET OTC GUAIFENESIN/DEXTROMETHORPHAN TANAFED DMX D-METHORP TAN/P-EPD TAN/D-CP<br />
GUAIFENESIN/DM/PHENYLEPHRINE SYRUP OTC GUAIFENESIN/D-METHORPHAN HB/PE TESSALON PERLE BENZONATATE<br />
LOHIST-DM SYRUP BROMPHENIRAMIN/PE/DEXTROMETHOR TL-DEX DM GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
LOHIST PEB DM LIQUID OTC BROMPHENIRAMIN/PE/DEXTROMETHOR TRI-DEX PE LIQUID OTC DM/PHENYLEPH/CHLORPHENIRAMINE<br />
LOHIST-PSB-DM OTC D-METHORPHAN HB/P-EPD HCL/BPM TRIGOFEN DM OTC DM/PHENYLEPH/CHLORPHENIRAMINE<br />
LORTUSS DM OTC DOXYLAMINE/PSEUDOEPHEDRINE/DM TUSSI-12 S CAR-B-PEN TA/CHLOR-TAN<br />
M-END DMX OTC DEXBROMPHEN/PSEUDOEPHEDRINE/DM V-COF BROMPHEN MAL/PE/CARBETAPEN CIT<br />
MUCINEX COLD-FLU & SORE THROAT OTC PHENYLEPHRINE/DM/ACETAMINOP/GG VAZOTAN BROMPHEN/PHENYLEPH/CARBET TANN<br />
MUCINEX COUGH OTC GUAIFENESIN/DEXTROMETHORPHAN Z-COF 8 DM GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
MUCINEX DM OTC GUAIFENESIN/DEXTROMETHORPHAN ZONATUSS BENZONATATE<br />
MUCINEX FAST-MAX DM MAX OTC GUAIFENESIN/DEXTROMETHORPHAN ZOTEX-12D DM/PHENYLEPH/CHLORPHENIRAMINE<br />
MUCINEX SEVERE CONGEST-COUGH OTC<br />
GUAIFENESIN/D-METHORPHAN HB/PE<br />
MYHIST-DM LIQUID<br />
PYRILAMINE/PE/DEXTROMETHORPHAN<br />
NASOHIST DM OTC<br />
DM/PHENYLEPH/CHLORPHENIRAMINE<br />
NEO DM OTC<br />
D-METHORPHAN HB/P-EPD HCL/BPM<br />
NEO DM OTC<br />
DM/PHENYLEPH/CHLORPHENIRAMINE<br />
NOHIST-DM LIQUID OTC<br />
DM/PHENYLEPH/CHLORPHENIRAMINE<br />
PE-HIST DM<br />
DM/PHENYLEPH/CHLORPHENIRAMINE<br />
PE-HIST DM SYRUP<br />
DM/PHENYLEPH/CHLORPHENIRAMINE<br />
PEDIATEX TDM OTC<br />
TRIPROLIDINE/PSEUDOEPHEDRIN/DM<br />
POLY-VENT DM OTC<br />
GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
PROMETHAZINE-DM SYRUP<br />
D-METHORPHAN HB/PROMETH HCL<br />
PSE BROM<br />
D-METHORPHAN HB/P-EPD HCL/BPM<br />
PYRIL DM OTC<br />
PYRILAMINE/PE/DEXTROMETHORPHAN<br />
RESCON-DM LIQUID OTC<br />
D-METHORPHAN HB/P-EPHED HCL/CP<br />
SILPHEN DM OTC<br />
DEXTROMETHORPHAN HBR<br />
SUDATEX-DM OTC<br />
GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
TL-HIST DM<br />
BROMPHENIRAMIN/PE/DEXTROMETHOR<br />
TUSSIN DM SYRUP OTC<br />
GUAIFENESIN/DEXTROMETHORPHAN<br />
VANACOF DX OTC<br />
PSEUDOEPH/CHLOPHEDIANOL/GG<br />
VANACOF G OTC<br />
CHLOPHEDIANOL HCL/GUAIFENESIN<br />
VANACOF OTC<br />
D-CHLORPHENIRA/PSE/CHLOPHEDIAN<br />
VANACOF-PE OTC<br />
CHLORCYCLIZIN-PE-CHLOPHEDIANOL<br />
VANATAB DX OTC<br />
PSEUDOEPH/CHLOPHEDIANOL/GG<br />
Y-COF DMX OTC<br />
BROMPHENIRAMIN/PE/DEXTROMETHOR<br />
Z-COF 12DM<br />
GUAIFENESIN/DM/PSEUDOEPHEDRINE<br />
Z-COF I OTC<br />
GUAIFENESIN/DM/PSEUDOEPHEDRINE
Cough and Cold Drugs - Narcotic<br />
PREFERRED Ingredients NON-PREFERRED Ingredients<br />
CHLORPHENIRAMINE/CODEINE PHOS CHLORPHENIRAMINE/CODEINE PHOS ALA-HIST AC PHENYLEPHRINE HCL/CODEINE<br />
CODAL-DH PHENYLEPHRINE/HYDROCODONE/PYR ALAHIST DHC PHENYLEPHRINE HCL/DIHYDROCO BT<br />
ENDACOF-C CHLORPHENIRAMINE/CODEINE PHOS ALLFEN CD GUAIFENESIN/CODEINE PHOSPHATE<br />
ENDACOF-DC OTC PSEUDOEPHEDRINE HCL/CODEINE ALLFEN CDX GUAIFENESIN/CODEINE PHOSPHATE<br />
GANI-TUSS NR OTC GUAIFENESIN/CODEINE PHOSPHATE AMBIFED CD P-EPHED HCL/CODEINE/GUAIFEN<br />
GUAIFENESIN/CODEINE SYRUP OTC GUAIFENESIN/CODEINE PHOSPHATE AMBIFED CDX P-EPHED HCL/CODEINE/GUAIFEN<br />
HYCODAN TABLET HYDROCODONE BIT/HOMATROP ME-BR AMBIFED-G CD P-EPHED HCL/CODEINE/GUAIFEN<br />
LORTUSS EX OTC P-EPHED HCL/CODEINE/GUAIFEN AMBIFED-G CDX P-EPHED HCL/CODEINE/GUAIFEN<br />
M-CLEAR GUAIFENESIN/CODEINE PHOSPHATE BALTUSSIN PHENYLEPHRINE/DHCODEINE BT/CP<br />
M-CLEAR WC GUAIFENESIN/CODEINE PHOSPHATE BRONTEX GUAIFENESIN/CODEINE PHOSPHATE<br />
M-END WC BROMPHENIRA/PSEUDOEPHED/CODEIN BRONTEX OTC GUAIFENESIN/CODEINE PHOSPHATE<br />
PRO-CLEAR AC OTC CODEINE PHOSPHATE/PYRIL MAL CHERATUSSIN DAC OTC P-EPHED HCL/CODEINE/GUAIFEN<br />
PROMETHAZINE VC/CODEINE SYRUP (ACTAVIS) PHENYLEPHRINE HCL/COD/PROMETH CHLORPHENIRAMINE/CODEINE LIQUID CHLORPHENIRAMINE/CODEINE PHOS<br />
PROMETHAZINE VC/CODEINE SYRUP (QUALITEST) PHENYLEPHRINE HCL/COD/PROMETH COLDCOUGH PD PHENYLEPHRINE/DHCODEINE BT/CP<br />
PROMETHAZINE/CODEINE SYRUP CODEINE/PROMETHAZINE HCL COTAB A CHLORPHENIRAMINE/CODEINE PHOS<br />
TUSSIONEX PENNKINETIC SUSPENSION HYDROCODONE/CHLORPHEN POLIS COTAB AX CHLORPHENIRAMINE/CODEINE PHOS<br />
COTABFLU<br />
CHLOR-MAL/CODEINE/ACETAMINOPHN<br />
CYTUSS HC<br />
PHENYLEPHRINE/HYDROCODONE/CP<br />
DEX-TUSS<br />
GUAIFENESIN/CODEINE PHOSPHATE<br />
DEX-TUSS OTC<br />
GUAIFENESIN/CODEINE PHOSPHATE<br />
DONATUSS DC<br />
PHENYLEPH/DIHYDROCODEINE/GUAIF<br />
DUOHIST DH<br />
PHENYLEPHRINE/DHCODEINE BT/CP<br />
HYDROCODONE/CHLORPHENIRAMINE SUSP<br />
HYDROCODONE/CHLORPHEN POLIS<br />
HYDROCODONE/HOMATROPINE SYRUP<br />
HYDROCODONE BIT/HOMATROP ME-BR<br />
HYDROCODONE/HOMATROPINE TABLET<br />
HYDROCODONE BIT/HOMATROP ME-BR<br />
HYPHED<br />
PSEUDOEPHED/HYDROCODONE/CPM<br />
J-COF DHC<br />
BROMPHENRM/PSEUDOEPH/DIHYDROCD<br />
J-MAX DHC<br />
DIHYDROCODEINE/GUAIFENESIN<br />
M-END MAX D OTC<br />
DEXBROMPHENIRAMINE/PSE/CODEINE<br />
MAXIFED CD<br />
P-EPHED HCL/CODEINE/GUAIFEN<br />
MAXIFED CDX<br />
P-EPHED HCL/CODEINE/GUAIFEN<br />
MAXIFED-G CD<br />
P-EPHED HCL/CODEINE/GUAIFEN<br />
MAXIFED-G CDX<br />
P-EPHED HCL/CODEINE/GUAIFEN<br />
MAXIFLU CD<br />
P/EPHED/CODEINE/ACETAMINOPH/GG<br />
MAXIFLU CDX<br />
P/EPHED/CODEINE/ACETAMINOPH/GG<br />
MAXIPHEN CD<br />
PHENYLEPHRINE/CODEINE/GUAIFEN<br />
MAXIPHEN CDX<br />
PHENYLEPHRINE/CODEINE/GUAIFEN<br />
NASOTUSS OTC<br />
CHLORCYCLIZ/PHENYLEPH/CODEINE<br />
NEO AC OTC<br />
PYRIL MAL/PSEUDOEPHED/CODEINE<br />
PHENFLU CD<br />
PE-CODEINE-ACETAMINOPHEN-GUAIF<br />
PHENFLU CDX<br />
PE-CODEINE-ACETAMINOPHEN-GUAIF<br />
PHENYLHISTINE DH OTC<br />
P-EPHED HCL/COD/CHLORPHENIR<br />
POLY HIST DHC<br />
PYRILAM/PHENYLE/DIHYDROCODEINE<br />
POLY HIST NC<br />
PSEUDOEPHED/CODEINE/TRIPROLIDN<br />
POLY-TUSSIN AC OTC<br />
BROMPHENIRAMIN/PE/CODEINE<br />
POLY-TUSSIN DHC<br />
BROMPHENIRAM/PE/DIHYDROCODEINE<br />
POLY-TUSSIN EX<br />
PHENYLEPH/DIHYDROCODEINE/GUAIF<br />
PSEUDOEPHEDRINE HCL/CODEINE<br />
PSEUDOEPHEDRINE HCL/CODEINE<br />
REZIRA<br />
P-EPHED HCL/HYDROCODONE BIT<br />
SU-TUSS HD<br />
GUAIFENESIN/P-EPHED HCL/HCOD<br />
TL-HIST CD<br />
BROMPHENIRAMIN/PE/CODEINE<br />
TUSSCOUGH DHC<br />
PHENYLEPHRINE/DHCODEINE BT/CP<br />
TUSSICAPS<br />
HYDROCODONE/CHLORPHEN POLIS<br />
TUSSO-C<br />
GUAIFENESIN/CODEINE PHOSPHATE<br />
VANACOF CD<br />
DEXCHLORPHEN/PHENYLEPH/CODEINE
Cough and Cold Drugs - Narcotic Continued<br />
PREFERRED Ingredients NON-PREFERRED Ingredients<br />
I I<br />
ZOTEX-C<br />
ZUTRIPRO<br />
PYRIL MA/PE/CODEINE PHOS<br />
PSEUDOEPHED/HYDROCODONE/CPM
IRON AGENTS<br />
Preferred Agents<br />
Non-Preferred Agents<br />
CENTRATEX<br />
BIFERA RX<br />
DUOFER CORVITA 150<br />
FE FUMARATE/VIT C/B12-IF/FA CORVITE 150<br />
FE FUMARATE/VIT C/VIT B12/FA<br />
CORVITE FE<br />
FERRALET 90 FERRAPLUS 90<br />
FERRALET 90 DUAL-IRON<br />
FERREX 150 PLUS OTC<br />
FERREX 28<br />
FERROFLEX 150 FORTE<br />
FERRIMIN 150 OTC<br />
FOLITAB 500 OTC<br />
FERROUS SULFATE 325 MG TABLET OTC<br />
HEMAX<br />
FETRIN<br />
HEMETAB<br />
FOLITAB 500<br />
ICAR-C OTC<br />
FOLIVANE-F<br />
MAXARON FORTE<br />
FOLIVANE-PLUS<br />
MULTIGEN<br />
FUMATINIC<br />
MULTIGEN PLUS<br />
HEMATINIC PLUS PRUVATE 21-7<br />
HEMATINIC WITH FOLIC ACID<br />
TARON FORTE<br />
HEMATOGEN<br />
VITAFOL<br />
HEMATOGEN FA<br />
HEMOCYTE PLUS<br />
HEMOCYTE-F<br />
IFEREX 150 FORTE<br />
INTEGRA F<br />
INTEGRA OTC<br />
INTEGRA PLUS<br />
IRON 45 MG TABLET OTC<br />
IRON POLYSACCHARIDES COMPLEX OTC<br />
IRON PS CMPLX/VIT B12/FA<br />
IROSPAN<br />
MULTIGEN FOLIC<br />
MV COMB18/FEFM-FEPOL CB1/FA<br />
NEPHRON FA<br />
NOVAFERRUM<br />
SE-TAN PLUS<br />
T<strong>AND</strong>EM DUAL ACTION OTC<br />
T<strong>AND</strong>EM F<br />
T<strong>AND</strong>EM PLUS<br />
TL-FOL 500<br />
TL-HEM 150
PRENATAL VITAMINS<br />
PREFERRED DRUGS<br />
NON-PREFERRED DRUGS<br />
BR<strong>AND</strong> NAME<br />
BR<strong>AND</strong> NAME<br />
CITRANATAL 90 DHA PACK<br />
ADVANCED CARE PLUS TABLET<br />
CITRANATAL ASSURE COMBO PACK<br />
B-NEXA TABLET<br />
CITRANATAL B-CALM COMBO PACK<br />
BP FOLINATAL PLUS B TABLET<br />
CITRANATAL B-CALM PACK<br />
TRIMESIS RX TABLET<br />
CITRANATAL DHA PACK<br />
CAVAN-EC SOD DHA VITAMINS<br />
CITRANATAL RX TABLET<br />
CITRANATAL HARMONY CAPSULE<br />
COMPLETE-RF PRENATAL TABLET<br />
COMPLETE NATAL DHA<br />
ED CYTE F TABLET<br />
COMPLETENATE TABLET CHEW<br />
FE C PLUS TABLET<br />
CONCEPT DHA CAPSULE<br />
ICAR-C PLUS TABLET<br />
CONCEPT OB CAPSULE<br />
MAXINATE TABLET<br />
DUET DHA BALANCED COMBO PACK<br />
NESTABS PRENATAL TABLET<br />
DUET DHA COMPLETE COMBO PACK<br />
NESTABS DHA COMBO PACK<br />
ELITE-OB CAPLET<br />
PAIRE OB PLUS DHA COMBO PACK<br />
FOLCAPS CARE ONE CAPSULE<br />
FOLTABS PRENATAL TABLET<br />
FOLCAPS OMEGA-3 CAPSULE<br />
VINACAL PRENATAL TABLET<br />
FOLIVANE-OB CAPSULE<br />
PRENAPLUS TABLET<br />
FOLTABS 90 PLUS DHA PACK<br />
VOL-PLUS TABLET<br />
GESTICARE DHA COMBO PACK<br />
VOL-PLUS TABLET<br />
ICAR-C PLUS SR CAPSULE<br />
PRENATAL PLUS IRON TABLET<br />
LACTOCAL-F TABLET<br />
PRENATAL PLUS TABLET<br />
MISSION PRENATAL FA TABLET<br />
PRENATAL PLUS TABLET<br />
NATAFORT TABLET<br />
PREFERA-OB PLUS DHA COMBO PACK<br />
NATELLE ONE CAPSULE<br />
PREFERA-OB PLUS DHA COMBO PACK<br />
NATELLE PLUS COMBO PACK<br />
PRENATABS FA TABLET<br />
NEXA SELECT CAPSULE<br />
PRENATABS RX TABLET<br />
OB COMPLETE 400 SOFTGEL<br />
PRENATAL 19 CHEWABLE TABLET<br />
OB COMPLETE DHA SOFTGEL<br />
PRENATAL 19 TABLET<br />
OB COMPLETE ONE SOFTGEL<br />
PRENATAL AD TABLET<br />
OB COMPLETE PREMIER TABLET<br />
TRIADVANCE TABLET<br />
OB COMPLETE CHEWABLE TABLET<br />
VENATAL-FA TABLET<br />
OB COMPLETE CAPLET<br />
PRENATAL TABLET<br />
ELITE OB DHA SOFTGEL<br />
SE-NATAL ONE TABLET<br />
CAVAN ONE OMEGA SOFTGEL<br />
TRINATAL RX 1 TABLET<br />
FOLCAPS OMEGA-3 CAPSULE<br />
VINATE ONE TABLET<br />
ULTIMATECARE ONE CAPSULE<br />
PRENATAL VITAMIN TABLET<br />
ZATEAN-PN TABLET<br />
PRENATAL-U CAPSULE<br />
ZATEAN-PN PLUS SOFTGEL<br />
PROFE FORTE CAPSULE<br />
ROVIN-NV TABLET<br />
SE-NATAL 19 CHEWABLE TABLET<br />
FOLCAL DHA CAPSULE<br />
SE-NATAL 19 TABLET<br />
TL-SELECT CAPSULE<br />
SELECT-OB + DHA PACK<br />
VENA-BAL DHA COMBO PACK<br />
T<strong>AND</strong>EM OB CAPSULE<br />
PR NATAL 400 COMBO PACK
PRENATAL VITAMINS<br />
PREFERRED DRUGS<br />
NON-PREFERRED DRUGS<br />
TARON-BC TABLET<br />
PR NATAL 400 EC COMBO PACK<br />
TARON-C DHA CAPSULE<br />
PR NATAL 430 EC COMBO PACK<br />
TRINATAL GT TABLET<br />
PREFERA OB TABLET<br />
VINATE AZ TABLET<br />
PREFERA-OB ONE SOFTGEL<br />
VINATE CALCIUM PRENATAL TABLET<br />
TRINATAL ULTRA TABLET<br />
VINATE GT TABLET<br />
PRENATE DHA SOFTGEL<br />
VINATE IC CAPSULE<br />
PRENATE ELITE TABLET<br />
VINATE II TABLET<br />
PRENATE ESSENTIAL SOFTGEL<br />
VITAFOL-OB+DHA COMBO PACK<br />
PRENEXA CAPSULE<br />
VITAFOL-ONE CAPSULE<br />
PRENEXA PREMIER CAPSULE<br />
VITASPIRE TABLET<br />
PREQUE 10 TABLET<br />
VOL-NATE TABLET<br />
BP MULTINATAL PLUS CHEW TABLET<br />
VOL-TAB RX TABLET<br />
SE-CARE CHEWABLE TABLET<br />
VINATE CARE CHEWABLE TABLET<br />
BP MULTINATAL PLUS TABLET<br />
SE-CARE CONCEIVE TABLET<br />
VINATE C TABLET<br />
RELNATE DHA PRENATAL SOFTGEL<br />
ROVIN-NV DHA CAPSULE<br />
SE-TAN DHA CAPSULE<br />
SELECT-OB CAPLET<br />
SETONET PRENATAL VITAMIN<br />
SETONET-EC PRENATAL VITAMINS<br />
T<strong>AND</strong>EM DHA CAPSULE<br />
TARON-DUO EC COMB PACK<br />
TL-ASSURE + DHA COMBO PACK<br />
TL-ASSURE ONE CAPSULE<br />
TRICARE PRENATAL TABLET<br />
TRICARE DHA 301 CAPSULE<br />
TRICARE PRENATAL DHA ONE SFTGL<br />
TRIVEEN-DUO DHA COMBO PACK<br />
TRIVEEN-TEN TABLETS<br />
ULTIMATE OB DHA COMBO PACK<br />
ULTIMATECARE ADVANTAGE COMBO<br />
ULTIMATECARE COMBO PACK<br />
VENATAL COMPLETE DHA COMBO<br />
VINATE AZ EXTRA TABLETS<br />
VINATE III TABLET<br />
VINATE PN CARE TABLET<br />
VITAFOL-OB CAPLET<br />
VIVA DHA PRENATAL SOFTGEL<br />
ZATEAN-CH CAPSULE<br />
ZATEAN-PN DHA CAPSULE