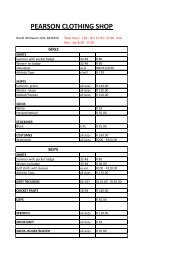
MEMORANDUM - Pearson High School
MEMORANDUM - Pearson High School
MEMORANDUM - Pearson High School
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>MEMORANDUM</strong><br />
ISEBE LE[IFUNDO LEMPI-]MA KOLON<br />
EASTERN CAPE EDLICATION DEPARTMENT<br />
OOS KAAP ONDERWYSDEPARTEITIENT<br />
IMVWO TFBANGA LESHI]M ELLNANYE<br />
GRADE 1] EXAIIINATIONS<br />
GRAAD 11 EKSAMEN<br />
NOVEMBER 2OO8<br />
pHysrcAL sctENcEs - sFcoND PAPER (CHEMISTRY)<br />
FISIESE WETENSKAPPE - TWEEDE VRAESTEL (CHEMIE<br />
rrmmr"nou.<br />
"onsi"tt ol9 pagcsJq erd " n"moranoum besldd" r''o bLad've
2 PTYSTCALSCTENCES/FsrES<br />
SECTION A<br />
QUESTION 1r ONE WORD lTElr4S<br />
VRAAG 1J EENWOORD]TEMS<br />
t1<br />
12<br />
13<br />
(r)<br />
(r)<br />
(r)<br />
(1)<br />
(1)<br />
t5l<br />
QUESTION 2: IIIATCHING ITEMS<br />
21<br />
22<br />
$a<br />
H/<br />
)/<br />
c/<br />
LI<br />
R<br />
(/<br />
(t)<br />
(1)<br />
(1)<br />
(1)<br />
11)<br />
tsl<br />
QUESTION 3: TRUE OR FALSE<br />
VMAG 3: WAAR OF ONWAAR<br />
{2)<br />
FALSE /, no equiibrum n<br />
A^IWAAR geen ewewig it<br />
\2)<br />
(2)<br />
(2)<br />
110l<br />
lt.1,<br />
+3<br />
+5<br />
A<br />
D<br />
I)
ER2DEVRAESTEL(FPSC) 3<br />
OUESTION 4; MULTIPLE-C HOICE QUESTIONS<br />
VRAAG 4: MEERVOUDIGEKEUSE-VRAE<br />
(31<br />
4.2<br />
D,/./r'<br />
(3)<br />
(3)<br />
(3)<br />
(3)<br />
Il sl<br />
51<br />
52<br />
1Ar'<br />
(1)<br />
(1)<br />
(r)<br />
55<br />
(1)<br />
(1)<br />
H I Si /!./<br />
H<br />
(3)<br />
{s)<br />
n1l<br />
6.1<br />
"/^ c = 99,100 = e2 3<br />
c)-T=77r'<br />
,/,H= 9!1,1aa =1.7<br />
. mole rato is 1 1 /<br />
nalvehaudingl :1<br />
n(H)=7 1=7,7 r'<br />
{41
4 PHYsICAL S6IENCES'FISIESE WETENSKAPPE 2ND PAPEfu2DE VRAESTEL IPtsSC!ME O]I ]<br />
Y" ofc in 7aolmo - t9]],7s=72<br />
100<br />
% of H n 73 a/mo -<br />
s lI\ls<br />
100 =6/<br />
Numberoimoecarbon -721<br />
Nlmber ol moe hydrogen = 6/1<br />
12=FJ<br />
Mal verhouding van<br />
C H=6:6<br />
The trle formula s<br />
Dieware famule is<br />
(3)<br />
m<br />
. 6 d1d t6, pF.d . F /'<br />
l4lal ls dre veean/sk3p nrsse, volune en tenPeQtlui<br />
t2)<br />
volume s direcilyproportonaltotehperalureOR<br />
Volume is nversely proporlonalto temperature //<br />
vohme is diek eweredg aan .lre lehpe.aluut OF<br />
votune is anqekeer.l eweredig aan dielenpe@lllu/t (2)<br />
7l
Axes corectly labe led wth units/<br />
Asse kanek benoern net cchhctlc<br />
Cofiect sca es on bolh aYes/<br />
Ko ekte skaalaD bei.le .ssc<br />
Slraighi ine graph drawf throuoh po nis/<br />
Reauittundrcliek oetek deu Dunre<br />
Volume s p.oportona to lemperalLre.///<br />
6)<br />
Valune is ewetedig aan tenpetaluur<br />
7.5 He ium has propenies c oserto those ol an deatgas than oxygen /r'<br />
Heliun hel eienskappe nader aan n ideale gas as suustar<br />
76 See inemarked as Ao. thegraph above.T/<br />
Sbn lyn generk as A ap die qtaliek hieka.<br />
(3)<br />
(2)<br />
(2)<br />
116l<br />
8.1 /<br />
Hsing e bond and O double bond<br />
H enkel bindihg eh a dubbetbindins<br />
a2<br />
12)<br />
lala lTalaal = 1371kJm.r /<br />
(s)
IH ls heat of reacllon orenthalpy change<br />
lH is reaksie wamLe af entalpie vedndeing<br />
^H=E/r,,,, E,.,"",,,r' (4)<br />
111l<br />
9.1<br />
92<br />
9.3<br />
ll s used as a household gas. //<br />
Ward gebruik as huBhoudelike gas t2)<br />
2CaH{ + l3O, + 3CO, + (3)<br />
t2)<br />
Lead em ssions from car efg nes are ioxic and cause lead poisonng finhaled<br />
Load-uiuatihgs wn nato$ is sitig en<br />
deut db hens ihgeasen ward.<br />
Lead replacement i!e!/ LRP<br />
Load vetu an glngsbta ndstal/LR P<br />
veroatsa ak tDodveryft i9in9 in.iien<br />
(2)<br />
(2)<br />
tlil<br />
QUESTION 1O<br />
101 101.1<br />
(2)<br />
101.2<br />
(21<br />
HH<br />
N<br />
H<br />
I<br />
H-C C H<br />
!l<br />
uH<br />
I<br />
I<br />
I<br />
c<br />
I<br />
ll<br />
(3)
1A22 E]m.ator r'/<br />
Bom.e deco ourse //<br />
ER/2DEVRAESIELlPHSC) 7<br />
t2)<br />
t11l<br />
OUESTION 11ruRAAG 11<br />
111<br />
Whal s lhe concenlral on ofv negar n<br />
W.t ts dic kans.nlasic nn.syn rn d).<br />
(2)<br />
lll=!. '<br />
c.,{ j<br />
No oi moeeaiNaOH = n=.v<br />
c=l<br />
- ooo102<br />
o.o05<br />
Il<br />
Die kahsentaste van die<br />
OUESIION 12IVRAAG I2<br />
{7)<br />
(2)<br />
t11l<br />
121<br />
122<br />
123<br />
124<br />
4Fe + 3 A2 .2 Fe2a3t/<br />
1241<br />
Galvanlsnq/ 7/ Coverwlh a coal ol Zf merar //rpa .iinS/SrronSer<br />
Galvansern9// / Bedek met n aag<br />
{2)<br />
t2)<br />
\2)<br />
1r)<br />
1r)<br />
1121
OLJESTION 1]A/RAAG 13<br />
131<br />
Nlirols oxide /. methane / chorofluor..arbonsT<br />
Sriksloloksted mera., .:hlaanhb.txaatst v!tu--<br />
132 Any-lwa // Enigell.o<br />
()<br />
Bnnd ran fossieL hendstawe<br />
Madenre Landb.u rlelades<br />
l2)<br />
Green house eiJect s the trapping ofheat nearea.ths suda.e by gases<br />
n th€ ahosphere padiculary carbon doxde /)<br />
Kweekhuis ellek ts.lie ve6.dehgvah hilte nabl dE aatde s-.<br />
.ppeNlak de . gasse ]n die.rnasleet retalkootslaftljaksja.j<br />
Afy ONE // EnOe EEN<br />
ncrease n temperalureon e.rlh<br />
Rs€ otsea eve OR fo.d ng<br />
f.enahe in letnperatun ap aatde<br />
Styginq van seevbk AF vl.ede<br />
\:l<br />
(21<br />
Reflects hamlul rad ation<br />
Alows eneqy Io radiate nlo space//<br />
Relle kleet sk ade h ke be st r al itg<br />
Laaleheeietaean in.lie tuinte te bestrcal<br />
Any T\t\14 I Enige -.WEE/ /<br />
lJse clerner fu€ls n motorvehces<br />
Redlce emiss on orharnf! sases<br />
EncouEge molorsis to make less use oicars<br />
Scal€ down.. indusira isaton<br />
G e btu ik sk.ner bta ntl stotuwe n not ts<br />
Vetturndet die Lillaal van sk.detkegasse<br />
Moedlq nataiste aanon nhdet nolarctegebtuik<br />
verni ndet h d ustt ial isa sie<br />
l,losl nalons have s'gned the Ky.to Protoco agEeiq] to redlce<br />
Meesle nasteshetdie Kyola Pr.I.kat anden.ken en slent saanldat<br />
hulle k||eekhuts gasse wil vernhder<br />
t2)<br />
tlsl
AUESTION 14l'r'RAAG 14<br />
14.2 Reduc.g agent //<br />
Reduseemid.lcl<br />
t2)<br />
(2)<br />
14.3<br />
Fe,o3 + 3 CO J 2 Fe + 3 Co, (6aan irs)/ (balansetins) (3)<br />
14 4 Making ofsteelr'<br />
Used to franufacture nachnery toosr'<br />
Used in building malenals and cementmaking/<br />
veNaaKtiging van nasjinene, gereedskap<br />
Gebruik in bau-nabnaal en naakvan senent (3)<br />
aFDELtNG/sEcnoNB: Iltol<br />
GROOTTOTAAL/GRAND TOrAL: 150