Vivid Detail - Bio-Rad
Vivid Detail - Bio-Rad
Vivid Detail - Bio-Rad
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
cover story<br />
cover story<br />
Protein<br />
Separations<br />
Proteomics to Production<br />
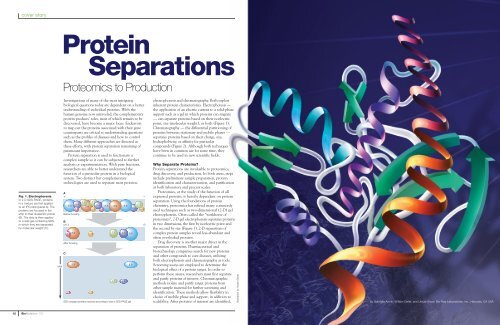
Fig. 1. Electrophoresis.<br />
In 2-D SDS-PAGE, proteins<br />
in a mixture are first applied<br />
to an IPG strip (panel A). The<br />
proteins are focused in the<br />
strip to their isoelectric points<br />
(B). The strip is then applied<br />
to a slab gel containing SDS,<br />
in which they are separated<br />
by molecular weight (C).<br />
MW<br />
Investigations of many of the most intriguing<br />
biological questions today are dependent on a better<br />
understanding of individual proteins. With the<br />
human genome now unraveled, the complementary<br />
protein products’ roles, most of which remain to be<br />
discovered, have become a major focus. Endeavors<br />
to map out the proteins associated with their gene<br />
counterparts are critical to understanding questions<br />
such as the profiles of diseases and how to control<br />
them. Many different approaches are directed at<br />
these efforts, with protein separation remaining of<br />
paramount importance.<br />
Protein separation is used to fractionate a<br />
complex sample so it can be subjected to further<br />
analysis or experimentation. With pure fractions,<br />
researchers are able to better understand the<br />
function of a particular protein in a biological<br />
system. Two distinct but complementary<br />
technologies are used to separate most proteins:<br />
A<br />
+<br />
7.1 7.1<br />
8.4<br />
8.8 8.4<br />
7.1<br />
3.9 8.8 3.9 8.8<br />
8.4<br />
3.7 5.3 3.7<br />
5.3<br />
Before focusing<br />
B<br />
pH 3<br />
3.9<br />
7.1 3.7 5.3<br />
8.4<br />
7.1 8.4<br />
8.8<br />
8.8<br />
After focusing<br />
C<br />
3.7<br />
3.9<br />
5.3<br />
7.1<br />
8.4<br />
SDS-charged proteins resolved according to size in SDS-PAGE gel<br />
8.8<br />
–<br />
10<br />
electrophoresis and chromatography. Both exploit<br />
inherent protein characteristics. Electrophoresis —<br />
the application of an electric current to a solid-phase<br />
support such as a gel in which proteins can migrate<br />
— can separate proteins based on their isoelectric<br />
point, size (molecular weight), or both (Figure 1).<br />
Chromatography — the differential partitioning of<br />
proteins between stationary and mobile phases —<br />
separates proteins based on their charge, size,<br />
hydrophobicity, or affinity for particular<br />
compounds (Figure 2). Although both techniques<br />
have been in common use for some time, they<br />
continue to be used in new scientific fields.<br />
Why Separate Proteins<br />
Protein separations are invaluable to proteomics,<br />
drug discovery, and production. In both areas, steps<br />
include preliminary sample preparation, protein<br />
identification and characterization, and purification<br />
at both laboratory and process scales.<br />
Proteomics, or the study of the function of all<br />
expressed proteins, is heavily dependent on protein<br />
separation. Using the foundations of protein<br />
chemistry, proteomics has refined many commonly<br />
used techniques such as two-dimensional (2-D) gel<br />
electrophoresis. Often called the “workhorse of<br />
proteomics”, 2-D gel electrophoresis separates proteins<br />
in two dimensions, the first by isoelectric point and<br />
the second by size (Figure 1). 2-D separations of<br />
complex protein samples reveal less-abundant and<br />
often overlooked proteins.<br />
Drug discovery is another major driver in the<br />
separation of proteins. Pharmaceutical and<br />
biotechnology companies search for new proteins<br />
and other compounds to cure diseases, utilizing<br />
both electrophoresis and chromatography as tools.<br />
Screening assays are employed to determine the<br />
biological effect of a protein target. In order to<br />
perform these assays, researchers must first separate<br />
and purify proteins of interest. Chromatographic<br />
methods isolate and purify target proteins from<br />
other sample material for further screening and<br />
identification. These methods allow flexibility in<br />
choice of mobile phase and support, in addition to<br />
scalability. After proteins of interest are identified,<br />
Illustration by Audra Geras<br />
by Gabriella Armin, William Gette, and Ursula Snow, <strong>Bio</strong>-<strong>Rad</strong> Laboratories, Inc., Hercules, CA USA<br />
12<br />
<strong>Bio</strong><strong>Rad</strong>iations 112