(MOPETT) Trial
(MOPETT) Trial
(MOPETT) Trial
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
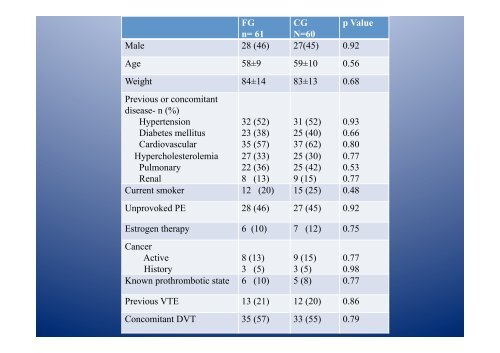
FG CG p Value<br />
n= 61 N=60<br />
Male 28 (46) 27(45) 0.92<br />
Age 58±9 59±10 0.56<br />
Weight 84±14 83±13 0.68<br />
Previous or concomitant<br />
disease- n (%)<br />
Hypertension<br />
Diabetes mellitus<br />
Cardiovascular<br />
Hypercholesterolemia<br />
Pulmonary<br />
Renal<br />
32 (52)<br />
23 (38)<br />
35 (57)<br />
27 (33)<br />
22 (36)<br />
8 (13)<br />
31 (52)<br />
25 (40)<br />
37 (62)<br />
25 (30)<br />
25 (42)<br />
9 (15)<br />
0.93<br />
0.66<br />
0.80<br />
0.77<br />
0.53<br />
0.77<br />
Current smoker 12 (20) 15 (25) 0.48<br />
Unprovoked PE 28 (46) 27 (45) 0.92<br />
Estrogen therapy 6 (10) 7 (12) 0.75<br />
Cancer<br />
Active<br />
History<br />
8 (13)<br />
3 (5)<br />
9 (15)<br />
3 (5)<br />
0.77<br />
0.98<br />
Known prothrombotic state 6 (10) 5 (8) 0.77<br />
Previous VTE 13 (21) 12 (20) 0.86<br />
Concomitant DVT 35 (57) 33 (55) 0.79