AmbioDry2⢠Processed Human Amniotic Membrane - IOP Inc
AmbioDry2⢠Processed Human Amniotic Membrane - IOP Inc
AmbioDry2⢠Processed Human Amniotic Membrane - IOP Inc
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
. .:;<br />
'li<br />
,,j:i<br />
"lt,#<br />
,."<br />
.*lj<br />
.'l g. i:<br />
{, ":.;'lri
<strong>Processed</strong> <strong>Human</strong> <strong>Amniotic</strong> <strong>Membrane</strong><br />
Historical Sackground and Surgical Use<br />
Historical Background<br />
The successful surgical use of<br />
dehydrated, substra{e-iree hunan<br />
amniotic mernbrane - ior the<br />
surgical treatment of the ocular<br />
surface * was first reporled by<br />
both Lavery and Sorsby in 1946.<br />
ln the 1980s, Dr. Juan F. Batlle<br />
re-pioneered the surgical use of<br />
preserved amnrotic membrane for the<br />
treatment of plerygium and other ocular<br />
conditions. Dr. Batlle's research noted<br />
the importance of utilizi,rg preserved<br />
amniotic mernbrane conta ning<br />
devitalized, inlact cellular c0mponents.<br />
hTAD?, the Ap[ e!1y' <strong>Amniotic</strong><br />
<strong>Membrane</strong> technology was introduced<br />
lo the ophthalmic surgical community.<br />
This "generation 1" AmbioDry<br />
technology featured a dehydrated,<br />
decellularized configuration for the<br />
surgical managemenl of ocular wounds.<br />
Today, with c0ntinued direction and<br />
input from industry leaders like Dr. Juan<br />
Satlle, a second gene ration AmbioDry<br />
technology is available. AmbioDry2.<br />
Surgical Application<br />
AmbioDry2 may be used for the<br />
surgical management of :<br />
O Pterygium<br />
O Persislent lpilhelial Defects<br />
I Dermoids<br />
I Fornix HeconstructionlSymblepharon<br />
C Corneal U lcerations/Perforalions<br />
O Bullous Keralopathy<br />
"The surgical use of preserved<br />
amn i atic membra ne tra n spl a ntation<br />
(AMT)has been repo{ted for<br />
decades as a vtable and advantageaus<br />
substitute for conjunctival<br />
a utog rafts. S i g n ifi cantly less<br />
surgical tine is reqaired and<br />
hast canjunctiva is preserved."<br />
Juan F. Ballle, M.D.<br />
Pioneer AMT<br />
President & Foundet<br />
Centro Microcirugia 0cular<br />
Laser {Santo Domingo, DR)
Mio@#e-W<br />
Advancement$, Advantages and Benefits<br />
Continued Innovation for 0ptimal Surgical Utility<br />
Optimized Storage &<br />
Surgical Utility<br />
AmbioDry2' is a processed, dehydrated, substrale-<br />
{ree amnintic membrane tissue graf1. [ach unil is<br />
sterilized and packaged wlth proprietary, device-like<br />
methods, designed to optimize st0rage and utility.<br />
AmbioDryZ can be stored on the shelf for 5 years.<br />
No freezrng or ref rigeration is required,<br />
This unique membrane can be trimme d in its dry<br />
state. applied to the surgical sile, activaled withrn<br />
minute s and the n fixated into place.<br />
lmproved Visual<br />
0rientation ldentification<br />
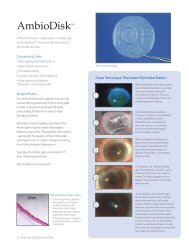
AmbioDry2 features a unique, proprielary<br />
"watermark" impression on the graft surface-for<br />
the simple, error-f ree rdentification of the basement<br />
rnembrane and stronal surfaces. The basement<br />
rnembrane can be easily identified by noting the<br />
prOper vertical orientation 0f the "10P" watermark.<br />
IAP "waterna*" i(lentifies the baserilent nenbrane<br />
More Biostructurally Intact<br />
AmbioDry2 tissue techn0lcgy combines minimallymanrpulative<br />
tissue processing methods with<br />
validaied sterrlization processes to produce a<br />
unique, high-quality amniotic membrane techn0logy.<br />
The generation 1 AmbioDry tissue process produced<br />
a decellularized collagen membrane, void of<br />
epilhelial cells and fibroblasts.<br />
6eneration 1 AnbiaDry: Phalanicrcgraph n7tes ll I the absence ol<br />
epithelial ce!ls an the surface af the nenbrane with (2) aa intact<br />
basenefrl nenbrane.<br />
The new generation AmbioDryZ provides a more<br />
intact, nalive amniotic membrane graft. While the<br />
unique tissue process destroys poten{ial bioburden<br />
and virulency, the membrane's devitalized cellular<br />
comp0nents - along with its dense connective<br />
malrix - ale preserved.<br />
1eneration ? AnbioDry2: Phatani*ograph shavr's {1) an intact epilhelial<br />
cell structurc cn tlle nenbrane.'s surfarc; 12) an iuiad dense basenent<br />
rnetnbranc and l3) lhe presetlce 0f a l00se Nllagen laye! with librcblasts<br />
lloninal thickness: 35 nicrcns.<br />
*-**:lOB, lnc.
Mio&<br />
<strong>Human</strong> <strong>Amniotic</strong> <strong>Membrane</strong> Allografts<br />
Procured, processed and configured with our focus on the surgoon and the patient.<br />
Tissue Safety Danor Selectian, Screening & Testing<br />
All tissue recovered meets stringent specifications during donor<br />
screening and laboratory testing to reduce the risk of transmitting<br />
rnfectious disease. AmbroDry2 allografts are procured and<br />
processed according to standards established by the American<br />
Association ol Tissue Eanks {AATB} and the United States Food<br />
& Drug Administration {FDA). All tissues are re covered under {ull<br />
iniormed 00nsent 0f the donors {represented by the mr:lhers of {he<br />
newborn children). The donors have consented to transfer oflhe<br />
allografts to third parties. A thorough medical and social history<br />
of the donor is also obtained, including detailed family history. The<br />
donor is screened for.<br />
Blood Type & Rh Factor HTLV{ Antibody f-lepatitis Cr:re Antibody<br />
HIV{ Antibody HTLV-2 Antibody Hepatitis Surface Antigen<br />
HIV-2 Antibody Syphrlis Screening l-lepatitis C Antibody<br />
HIV NAT<br />
CMV<br />
All tests results are reviewed prior to the release of the tissue.<br />
0nly tissue from donors that have a normal CBC, test negative for<br />
serology and test negative or non-reactive for infectious dtseases<br />
and conlamination are released.<br />
-..'lOP tn.,<br />
Reimbursement Information<br />
CPT Code 65780<br />
<strong>Amniotic</strong> <strong>Membrane</strong> Tralsplantation, 0cular Surface Rec0nslf ucti0n<br />
wwvr.toptlc.can<br />
Ordering Information<br />
AmbioDry2 Confi gutations<br />
- - ;"*,_11--11?g , no-ilso-' il;440 _[3 rtji<br />
r;;'-AD-irzo lr...Aoa*so-' il;440 At-ai$.'.1<br />
tl:" ;::l{rr l5x2cm _._?r3rt 4x4cnr 15mmd<br />
lt&,!r lrqir -Js''g.p l<br />
Slorage flssm femp: 0o to 30" C {32" to 1 B0' F}<br />
I{ominal Thickness 35 nicrons<br />
3184-B Airway Avenue<br />
Costa Mesa, CA 92626 USA<br />
Tel (714) 549-1185<br />
Fax {714i 549-0557<br />
email: ambiodry@iopinc.com<br />
Amblorfv ard Ambi0l]ryT are lraCemark; sl 0K | 0 0phth0. €)2108 l0P. lic<br />
Customer Service<br />
1-800-535-3545<br />
www.iopinc.com<br />
* t'<strong>IOP</strong> ln..<br />
f * *<br />
llllil "qr{n,ift