The anil<strong>in</strong>e dye (synthetic dye) <strong>in</strong>dustry grew rapidly.The first <strong>in</strong>dustrial-scale use was <strong>in</strong> themanufacture of mauve<strong>in</strong>e, a dye of the phenaz<strong>in</strong>e group be<strong>in</strong>g patented <strong>in</strong> 1856 by WHPERKIN (Produc<strong>in</strong>g a new color<strong>in</strong>g matter for dye<strong>in</strong>g with lilac or purple color stuffs of silk,cotton, wool, or other materials, British patent No. 1984, 26 th August 1856); PERKIN WH,1879). Many other dyes created from coal tar (<strong>and</strong> with <strong>in</strong>gredients other than anil<strong>in</strong>e)followed.The first artificial production of a natural dye was alizar<strong>in</strong>, produced by C GRAEBE <strong>and</strong> CLIEBERMANN (1868, 1869). They realized that alizar<strong>in</strong> was a derivative of anthraceneoccurr<strong>in</strong>g <strong>in</strong> coal tar. Industrially, anthraqu<strong>in</strong>one (or alizar<strong>in</strong>) dyes are derived fromanthracene which is a compound of anthracene oil, a coal tar fraction. The synthesis of<strong>in</strong>danthrene dyes (anthraqu<strong>in</strong>one dyes, the name is derived from <strong>in</strong>digo <strong>and</strong> anthracene) isbased on the fusion of 2-am<strong>in</strong>oanthraqu<strong>in</strong>one molecules <strong>in</strong> alkali (BOHN R, 1901). BOHN’s<strong>in</strong>vention of the first anthroqu<strong>in</strong>one vat dye “Indanthrene Blue” was patented <strong>in</strong> 1901(Verfahren zur Darstellung e<strong>in</strong>es blauen Farbstoffs der Anthracenreihe, DRP Nr. 129845, 6 thFebruary 1901). Then, other dyes became synthesized <strong>and</strong> recorded <strong>in</strong> a number of patentspecifications.From the many different classes of dyes, azo dyes form the largest <strong>and</strong> most important groupof synthetic organic dyes. The structure of azo dyes is based on azobenzene <strong>and</strong> <strong>in</strong>cludes thediazo functional group (R-N=N-R’) where the N=N group is the azo group between twoaromatic r<strong>in</strong>gs. Dye synthesis follows reactions studied by P GRIESS (1858) which arediazotation <strong>and</strong> coupl<strong>in</strong>g. The reaction mechanism consists of two steps. In the first step,diazotation of primary/aromatic am<strong>in</strong>es is performed to form diazonium salts (diazoniumkations) <strong>and</strong> called diazo component. Because aliphatic diazonium salts are very unstable,aromatic diazonium compounds are preferred. In a second step, azo coupl<strong>in</strong>g is done with thecoupl<strong>in</strong>g component: the coupler carries an electron-rich carbon atom to which the diazocomponent will become attached (pr<strong>in</strong>ciple of electrophilic substitution) bridg<strong>in</strong>g the gapbetween the two molecules with a new azo group (-N=N-).Start<strong>in</strong>g substances are anil<strong>in</strong>e or appropriately substituted derivatives of benzene. The natureof the aromatic substituents on both sides of the azo group as well as the water-solubility ofthe dyes determ<strong>in</strong>e the colors of the azo compounds (BRUNNER; 1929; HUNGER K et al.,2002). Most azo dyes are red, orange or yellow. The development of azo <strong>and</strong> anthraqu<strong>in</strong>onedyes completed the spectrum of synthetic dyes (HUNGER K <strong>and</strong> HERBST W, 2002; HUNGER Ket al., 2002).Histological specimens are prepared as is usual <strong>in</strong> rout<strong>in</strong>e histopathology which <strong>in</strong>cludespreservation <strong>and</strong> embedd<strong>in</strong>g (i.e. fixation, dehydration <strong>and</strong> embedd<strong>in</strong>g <strong>in</strong> paraff<strong>in</strong> or res<strong>in</strong>).Prior to sta<strong>in</strong><strong>in</strong>g, sections are cut with a microtome. In certa<strong>in</strong> cases, cryostat sections orcelloid<strong>in</strong> preparations are needed. Furthermore, cell smears from suspensions may be used.Sta<strong>in</strong>s are applied to make cell structures more readly visualized than <strong>in</strong> unsta<strong>in</strong>ed specimens,<strong>and</strong>, under certa<strong>in</strong> conditions, they can reveal molecular compounds <strong>and</strong> differencesassociated with evolution or pathological conditions. Thus, <strong>sta<strong>in</strong>s</strong> <strong>in</strong> general are aimed as<strong>special</strong> <strong>probes</strong> which possess variable specificity depend<strong>in</strong>g on the selectivity with which theyreact. Comparable to molecular biology, the term probe may be used to def<strong>in</strong>e a reagentwhich is suitable to detect or to measure the presence of a particular molecule. The termprobe can be applied to all sta<strong>in</strong><strong>in</strong>gs, yet more specicifically <strong>in</strong> cases where reactions withhighly specific reagents are performed such as antibodies, nucleotide sequences or other highaff<strong>in</strong>ity b<strong>in</strong>d<strong>in</strong>g molecules.
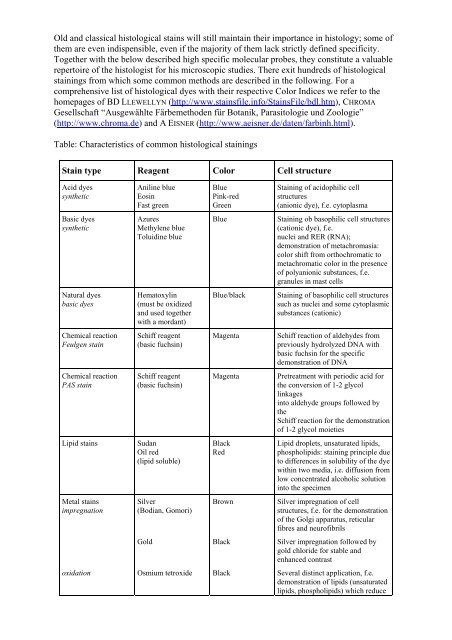
Old <strong>and</strong> classical histological <strong>sta<strong>in</strong>s</strong> will still ma<strong>in</strong>ta<strong>in</strong> their importance <strong>in</strong> <strong>histology</strong>; some ofthem are even <strong>in</strong>dispensible, even if the majority of them lack strictly def<strong>in</strong>ed specificity.Together with the below described high specific molecular <strong>probes</strong>, they constitute a valuablerepertoire of the histologist for his microscopic studies. There exit hundreds of histologicalsta<strong>in</strong><strong>in</strong>gs from which some common methods are described <strong>in</strong> the follow<strong>in</strong>g. For acomprehensive list of histological dyes with their respective Color Indices we refer to thehomepages of BD LLEWELLYN (http://www.<strong>sta<strong>in</strong>s</strong>file.<strong>in</strong>fo/Sta<strong>in</strong>sFile/bdl.htm), CHROMAGesellschaft “Ausgewählte Färbemethoden für Botanik, Parasitologie und Zoologie”(http://www.chroma.de) <strong>and</strong> A EISNER (http://www.aeisner.de/daten/farb<strong>in</strong>h.html).Table: Characteristics of common histological sta<strong>in</strong><strong>in</strong>gsSta<strong>in</strong> type Reagent Color Cell structureAcid dyessyntheticAnil<strong>in</strong>e blueEos<strong>in</strong>Fast greenBlueP<strong>in</strong>k-redGreenSta<strong>in</strong><strong>in</strong>g of acidophilic cellstructures(anionic dye), f.e. cytoplasmaBasic dyessyntheticAzuresMethylene blueToluid<strong>in</strong>e blueBlueSta<strong>in</strong><strong>in</strong>g ob basophilic cell structures(cationic dye), f.e.nuclei <strong>and</strong> RER (RNA);demonstration of metachromasia:color shift from orthochromatic tometachromatic color <strong>in</strong> the presenceof polyanionic substances, f.e.granules <strong>in</strong> mast cellsNatural dyesbasic dyesHematoxyl<strong>in</strong>(must be oxidized<strong>and</strong> used togetherwith a mordant)Blue/blackSta<strong>in</strong><strong>in</strong>g of basophilic cell structuressuch as nuclei <strong>and</strong> some cytoplasmicsubstances (cationic)Chemical reactionFeulgen sta<strong>in</strong>Schiff reagent(basic fuchs<strong>in</strong>)MagentaSchiff reaction of aldehydes frompreviously hydrolyzed DNA withbasic fuchs<strong>in</strong> for the specificdemonstration of DNAChemical reactionPAS sta<strong>in</strong>Schiff reagent(basic fuchs<strong>in</strong>)MagentaPretreatment with periodic acid forthe conversion of 1-2 glycoll<strong>in</strong>kages<strong>in</strong>to aldehyde groups followed bytheSchiff reaction for the demonstrationof 1-2 glycol moietiesLipid <strong>sta<strong>in</strong>s</strong>SudanOil red(lipid soluble)BlackRedLipid droplets, unsaturated lipids,phospholipids: sta<strong>in</strong><strong>in</strong>g pr<strong>in</strong>ciple dueto differences <strong>in</strong> solubility of the dyewith<strong>in</strong> two media, i.e. diffusion fromlow concentrated alcoholic solution<strong>in</strong>to the specimenMetal <strong>sta<strong>in</strong>s</strong>impregnationSilver(Bodian, Gomori)BrownSilver impregnation of cellstructures, f.e. for the demonstrationof the Golgi apparatus, reticularfibres <strong>and</strong> neurofibrilsGoldBlackSilver impregnation followed bygold chloride for stable <strong>and</strong>enhanced contrastoxidationOsmium tetroxideBlackSeveral dist<strong>in</strong>ct application, f.e.demonstration of lipids (unsaturatedlipids, phospholipids) which reduce