SCHEME NO. 4ASSISTANCE TO AYURVEDA, SIDDHA & UNANI (ASU) DRUG MANUFACTURINGUNITS TO ESTABLISH AN IN-HOUSE QUALITY CONTROL LABORATORY FOR THEPURPOSE OF QUALITY CONTROL TESTING OF ALL RAW MATERIALS/FINISHEDPRODUCTS AS PER PHARMACOPOEIAL PARAMETERS INCLUDING TESTING OFINGREDIENTS/HEAVY METALS/PESTICIDE RESIDUE/MICROBIAL LOAD ETC.Assistance to ASU&H drug manufacturing units having an annual turnover <strong>of</strong> upto Rs.20.00 crores for acquisition <strong>of</strong> prescribed essential quality control equipment for in-house<strong>Quality</strong> <strong>Control</strong> Lab shall be limited to Rs. 30.00 lakhs or 30% <strong>of</strong> expenditure incurred onthe basis <strong>of</strong> a MoU between the manufacturing unit / State Drug <strong>Control</strong>ler and Department<strong>of</strong> AYUSH with a condition that the quality control equipment purchased with Government <strong>of</strong>India assistance shall not be disposed <strong>of</strong> and that Government <strong>of</strong> India will have lien onsuch equipment in case <strong>of</strong> company going into liquidation. The acquisition <strong>of</strong> quality controlequipments would be as per Annexure for setting up an in-house quality control laboratoryfor testing <strong>of</strong> all ingredients and finished products as per Pharmacopoeial and otherstandards laid down by Deptt. <strong>of</strong> AYUSH/WHO from time to time. In the 10 th plan anassistance <strong>of</strong> upto Rs.5.00 lakh as back- ended subsidy was allowed to all ASUmanufacturing units for becoming Good Manufacturing Practices complaint. However, the<strong>of</strong>f take in the scheme was negligible on account <strong>of</strong> assistance being very meager. Further,it has been felt that Good Manufacturing Practices notified under Schedule ‘T’ <strong>of</strong> the Drugs &Cosmetics Act, 1940 and Rules, 1945 does not provide for a mandatory in-house laboratoryfor every manufacturing unit as a result <strong>of</strong> which most <strong>of</strong> the manufacturing units haveobtained Good Manufacturing Practices on the basis <strong>of</strong> having a tie up with an outsidelaboratory. For all practical purposes testing <strong>of</strong> all raw material and finished products is notcommercially viable unless a manufacturing unit has an in-house laboratory. Development<strong>of</strong> quality control laboratory is highly capital intensive. Acquisition <strong>of</strong> equipments like AtomicAbsorption Spectrometer, High Performance Thin Layer Chromatography Spectrometer, etcfor an in-house quality control laboratory entails heavy expenditure. Small and mediumunits find it very burdensome to obtain loans without any subsidy. The Parliamentarystanding Committee <strong>of</strong> Ministry <strong>of</strong> Health & Family Welfare has also recommendedsubstantial increase in assistance to manufacturing units for becoming Good ManufacturingPractices complaint. The grantee institute/ State Govt. may apply in the following performa4 for seeking grant-in-aid under the scheme.
AnnexureLIST OF EQUIPMENT RECOMMENDED FOR IN-HOUSE QUALITY CONTROL SECTIONOF AYUSH MANUFACTURING UNITS(A) Chemistry Section (Area: 600+600 sq. Tentative Costft.)1. Alcohol determination apparatus (completeset)2. volatile Oil Determination Apparatus3. Boiling Point Determination Apparatus4. Melting Point Determination Apparatus5. Refractometer6. Polarimeter7. Viscometer8. Tablet Disintegration Apparatus9. Moisture Meter10. Muffle Furnace11. Electronic Balance12. Magnetic Stirrer13. Hot air Oven14. Refrigerator15. Glass/Steel Distillation Apparatus16. LPG Gas Cylinders with Burners17. Water Bath (Temperature <strong>Control</strong>led)18. Heating Mantles/Hot plates19. TLC Apparatus with all accessories (Manual)20. Paper Chromatography Apparatus withaccessories21. Sieve Size 10 to 120 with Sieve Shaker22. Centrifuge machine23. De-humidifier24. pH Meter25. Limit Test Apparatus26. GLC27. Atomic Absorption Spectrometer with Graphitefurnace with Hydride generator28. HPTLC29. HPLC30. ICPMS Liquid Chromatography-MarsSpectrophotometerRs. 4,000.00Rs. 4,000.00Rs. 2,000.00Rs. 10,000.00Rs. 15,000.00Rs. 20,000.00Rs. 20,000.00Rs. 20,000.00Rs. 20,000.00Rs. 20,000.00Rs. 75,000.00Rs. 10,000.00Rs. 20,000.00Rs. 20,000.00Rs. 20,000.00Rs. 5,000.00Rs. 10,000.00Rs. 10,000.00Rs. 10,000.00Rs. 10,000.00Rs. 10,000.00Rs. 25,000.00Rs. 25,000.00Rs. 10,000.00Rs. 10,000.00Rs. 30,00,000.00Rs. 40,00,000.00Rs. 40,00,000.00Rs. 70,00,000.00Rs.1,00,000,00.00(B) Pharmacognosy Section (600 Sq.ft.)1. Microscope Binocular2. Dissecting Microscope3. Physical Balance4. Aluminum Slide trays5. Stage Micrometer6. Camera Lucida (Prism and Mirror Type)7. Chemicals, Glass-ware etc.(C) Microbiology Laboratory (300 Sq.ft.)1. Laminar flow clean air bench2. Autoclave3. colony counter4. BOD incubator5. incubator6. glassware, media7. temperature bathTotalRs. 1,00,000.00Rs. 50,000.00Rs. 10,000.00Rs. 10,000.00Rs. 35,000.00Rs. 10,000.00Rs. 50,000.00Rs. 50,000.00Rs. 25,000.00Rs. 20,000.00Rs. 30,000.00Rs. 20,000.00Rs. 50,000.00Rs. 10,000.00Rs. 1,58,00,000.00
- Page 1 and 2: Centrally Sponsored Scheme of Quali
- Page 3 and 4: PROFORMA -2PROFORMA OF APPLICANT FO
- Page 5 and 6: SCHEME NO. 3REIMBURSEMENT OF EXPEND
- Page 7: (b) Expenditurecomputerization/ fax
- Page 11 and 12: PROFORMA -4APPLICATION FORM FOR GRA
- Page 13 and 14: SCHEME NO. 5ASSISTANCE TO ASU MANUF
- Page 15 and 16: 8. Recommendation of the StateDrug
- Page 17 and 18: No. Z. 17016/7/2007-DCC (AYUSH)Gove
- Page 19 and 20: (d)The assistance would be restrict
- Page 21 and 22: (b)Add On Interventionsa. Common Ma
- Page 23 and 24: (iii) The SMC without affecting the
- Page 25 and 26: Accounting Standard (AS) 18 of the
- Page 27 and 28: (b)Release of Funds by Department o
- Page 29 and 30: ANNEXURE IIIndicative list of point
- Page 31 and 32: ANNEXURE IIIProject Implementation
- Page 33 and 34: No. Z.17017/5/2007-DCC (AYUSH) (Pt.
- Page 35 and 36: List of Approved Ayurveda, Siddha &
- Page 37 and 38: 35. Punjab Drug Testing Laboratory,
- Page 39 and 40: LIST OF Assisted State Govt. Drug T
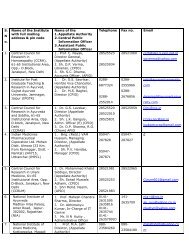
- Page 41 and 42: 20. Deputy Director (ISM&H),Inchage
- Page 43 and 44: 11. Principal/Supdt.,Rishikul State
- Page 45 and 46: 33. Managing Director,Indian Medici
- Page 47 and 48: LIST OF MAJOR MANUFACTURERES OF AYU
- Page 49 and 50: 19 Alathur Pharmaceutical Manufactu
- Page 51 and 52: 35 Amrut PharmaceuticalsAmrut Nagar
- Page 53 and 54: 53 Chadda Trader (Regd)Industrial A
- Page 55 and 56: 73 EURAFRIC Pharma Pvt Ltd25, Manga
- Page 57 and 58: 93 Herbotech IndiaHerbal House,Main
- Page 59 and 60:
113 Jolly PharmaceuticalsGuru Rai N
- Page 61 and 62:
133 Manphar Ayurvedic Drugs# 27-11-
- Page 63 and 64:
151 Nandan BioMatrix LimitedPlot No
- Page 65 and 66:
167 Proctor & Gamble Hygine & Healt
- Page 67 and 68:
187 R.K. PharmacyRama Mandi, Jogind
- Page 69 and 70:
207 Shri Ayurved Seva Sadan,P O Box
- Page 71 and 72:
Designation:Managing Director229 St
- Page 73 and 74:
245 Vital Care Private Ltd304, Spen
- Page 75 and 76:
265 Yogiraj PharmacyJai Maharashtra
- Page 77 and 78:
11.M/s Islahi Laboratories6-21/1/E/
- Page 79 and 80:
Punjab Drug Manufacturers Associati