Drug Delivery, Drug Packaging, Labelling & DispensingA Tool for Designing and EvaluatingPackaging for Healthcare ProductsAbstractAlthough significant resources are devoted to developinghealthcare products that are safe and effectivepharmacologically, significantly less effort is devoted toensuring products perform as intended in the hands ofproviders and patients. Poor designs result in user confusionand frustration, and have the potential to lead to errors andadverse events. The interaction that occurs between peopleand healthcare products is an area in need of study.The human-package interaction framework introduced bythis paper is an analysis tool to support decision-making whiledesigning and evaluating healthcare products. According tothis model, any healthcare product has: a set of intendedusers, varied contexts of use, and a series of tasks that usersmust accomplish (selection of the correct product, opening,administration, reclosing, disposal, etc.). The model providesdesigners with a structured consideration set with regard tothe interactions between people and healthcare products.Considerations to the interface are critically needed in orderto reduce the occurrence of medication errors and increaseadherence. Two examples are described to illustrate theconcepts.KEYWORDS: packaging, usability, adherence, noncompliance,medication errorIntroductionThe American National Coordinating Council forMedication Error Reporting and Prevention (NCCMERP)defines a medication error as “any preventable eventthat may cause or lead to inappropriate medication useor patient harm while the medication is in the control ofthe health care professional, patient, or consumer. Suchevents may be related to professional practice, health careproducts, procedures, and systems, including prescribing;order communication; product labeling, packaging, andnomenclature; compounding; dispensing; distribution;administration; education; monitoring; and use.” 1One way that medication errors manifest themselves isas an adverse drug event (ADE), defined as “injury due tomedication.” 2 By their nature, ADEs can be either preventableor unpreventable. Events that are attributable to productquality defects, medication errors or issues of non-complianceare examples of preventable events, while things that cannotbe foreseen or avoided are unpreventable. Examples ofunpreventable events include things like an unknown allergicreaction to a medication, or unavoidable side-effects, such asnausea and fatigue induced by chemotherapy.Medication errors and non-compliance events are closelyinterrelated. Some researchers have even argued that somenon-compliance events could be considered medical errors. 3Irrespective of how they are classified, both medication errorsand non-compliance issues are, many times, related to poordesign. Consider, for example, the Office of Post-MarketingDrug Risk Assessment (OPDRA, a division of the US Foodand Drug Administration), recommends interventions whichinclude changes in the naming, labelling, or packaging of adrug product. 4Regardless of whether the product is administered by ahealthcare professional, or the patient, there is an importantinteraction which must occur between the human andthe product. However, while a significant amount of timeand resources are devoted to ensuring safety and efficacypharmacologically, the degree to which design impactsthe product’s performance in the hands of patients andcare-givers is given considerably less attention. 5 Healthcareproducts that are designed and evaluated such that theycatalyse medication adherence and minimise errors aresorely needed.The correct and effective use of a drug does not simplydepend on the written information of its labelling and theverbal information given by healthcare professionals; itis supported by a system of physical components such aspackaging, measuring devices, applicators, dosing devices,closure systems, and the drug itself. 6 All these differentparts convey crucial information to patients, and their poordesign is known to be a contributing factor to patient noncompliance6and subsequent adverse events. 7,8 In fact,according to the Medication Error Reporting Program (MERP)operated by US Pharmacopeia (USP) and the Institute forSafe Medication Practices (ISMP), labelling and packagingissues were cited as the primary cause of 33% of medicationerrors, and incited 30% of reported fatalities.A review of the literature suggests that patient confusion andlack of accessibility are at the core of usability problems withmedications:• Confusion due to packaging similarity. 6,8,9• Confusion due to flawed dosing devices. 10• Confusion due to physical drug appearance (e.g., tabletsimilarity). 6• Confusion because of labelling issues: distorted text,non-flat labels, small print, lack of contrast, etc. 6,8• Confusion due to drug naming issues. 8,9,11-13• Non-accessible packaging. 6,14One critical reason for these problems is that the scientists andregulators who have historically made decisions regardingthe design of these systems frequently do not understand thecomplexities of human ability and behaviour. The approachhas generally been product-centered (PCD) in nature. That is,traditional decisions have been driven by the technical needsof the product (i.e., a drug) and manufacturing. Decisionstend to be focused on the manufacturer’s ability to maintain48 Journal For Patient Compliance Strategies to enhance Adherence and Health OutcomesVolume 2 - Issue 4
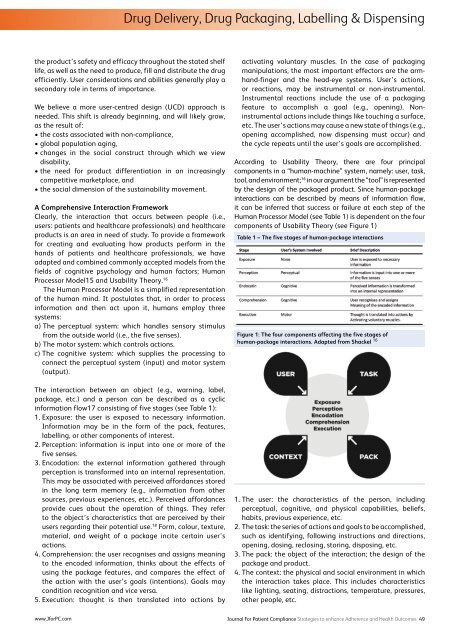
Drug Delivery, Drug Packaging, Labelling & Dispensingthe product’s safety and efficacy throughout the stated shelflife, as well as the need to produce, fill and distribute the drugefficiently. User considerations and abilities generally play asecondary role in terms of importance.We believe a more user-centred design (UCD) approach isneeded. This shift is already beginning, and will likely grow,as the result of:• the costs associated with non-compliance,• global population aging,• changes in the social construct through which we viewdisability,• the need for product differentiation in an increasinglycompetitive marketplace, and• the social dimension of the sustainability movement.A Comprehensive Interaction FrameworkClearly, the interaction that occurs between people (i.e.,users: patients and healthcare professionals) and healthcareproducts is an area in need of study. To provide a frameworkfor creating and evaluating how products perform in thehands of patients and healthcare professionals, we haveadapted and combined commonly accepted models from thefields of cognitive psychology and human factors; HumanProcessor Model15 and Usability Theory. 16The Human Processor Model is a simplified representationof the human mind. It postulates that, in order to processinformation and then act upon it, humans employ threesystems:a) The perceptual system: which handles sensory stimulusfrom the outside world (i.e., the five senses).b) The motor system: which controls actions.c) The cognitive system: which supplies the processing toconnect the perceptual system (input) and motor system(output).The interaction between an object (e.g., warning, label,package, etc.) and a person can be described as a cyclicinformation flow17 consisting of five stages (see Table 1):1. Exposure: the user is exposed to necessary information.Information may be in the form of the pack, features,labelling, or other components of interest.2. Perception: information is input into one or more of thefive senses.3. Encodation: the external information gathered throughperception is transformed into an internal representation.This may be associated with perceived affordances storedin the long term memory (e.g., information from othersources, previous experiences, etc.). Perceived affordancesprovide cues about the operation of things. They referto the object’s characteristics that are perceived by theirusers regarding their potential use. 18 Form, colour, texture,material, and weight of a package incite certain user’sactions.4. Comprehension: the user recognises and assigns meaningto the encoded information, thinks about the effects ofusing the package features, and compares the effect ofthe action with the user’s goals (intentions). Goals maycondition recognition and vice versa.5. Execution: thought is then translated into actions byactivating voluntary muscles. In the case of packagingmanipulations, the most important effectors are the armhand-fingerand the head-eye systems. User’s actions,or reactions, may be instrumental or non-instrumental.Instrumental reactions include the use of a packagingfeature to accomplish a goal (e.g., opening). Noninstrumentalactions include things like touching a surface,etc. The user’s actions may cause a new state of things (e.g.,opening accomplished, now dispensing must occur) andthe cycle repeats until the user’s goals are accomplished.According to Usability Theory, there are four principalcomponents in a “human-machine” system, namely: user, task,tool, and environment; 16 in our argument the “tool” is representedby the design of the packaged product. Since human-packageinteractions can be described by means of information flow,it can be inferred that success or failure at each step of theHuman Processor Model (see Table 1) is dependent on the fourcomponents of Usability Theory (see Figure 1)Table 1 – The five stages of human-package interactionsFigure 1: The four components affecting the five stages ofhuman-package interactions. Adapted from Shackel 161. The user: the characteristics of the person, includingperceptual, cognitive, and physical capabilities, beliefs,habits, previous experience, etc.2. The task: the series of actions and goals to be accomplished,such as identifying, following instructions and directions,opening, dosing, reclosing, storing, disposing, etc.3. The pack: the object of the interaction; the design of thepackage and product.4. The context: the physical and social environment in whichthe interaction takes place. This includes characteristicslike lighting, seating, distractions, temperature, pressures,other people, etc.www.JforPC.comJournal For Patient Compliance Strategies to enhance Adherence and Health Outcomes 49