Guidelines for intensified tuberculosis case-finding and isoniazid ...
Guidelines for intensified tuberculosis case-finding and isoniazid ...
Guidelines for intensified tuberculosis case-finding and isoniazid ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
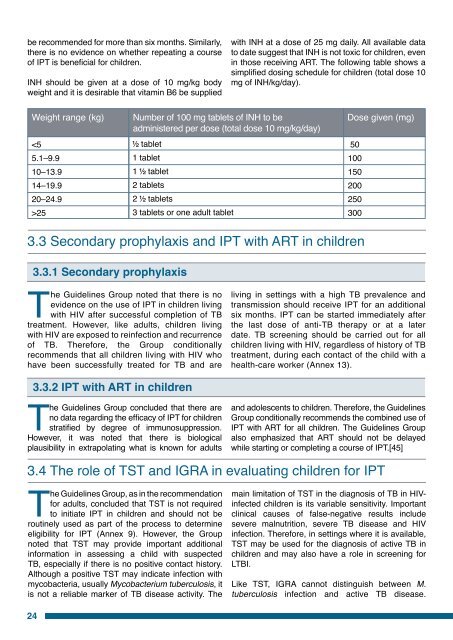
e recommended <strong>for</strong> more than six months. Similarly,there is no evidence on whether repeating a courseof IPT is beneficial <strong>for</strong> children.INH should be given at a dose of 10 mg/kg bodyweight <strong>and</strong> it is desirable that vitamin B6 be suppliedwith INH at a dose of 25 mg daily. All available datato date suggest that INH is not toxic <strong>for</strong> children, evenin those receiving ART. The following table shows asimplified dosing schedule <strong>for</strong> children (total dose 10mg of INH/kg/day).Weight range (kg)25Number of 100 mg tablets of INH to beadministered per dose (total dose 10 mg/kg/day)tablet1 tablet1 tablet2 tablets2 tablets3 tablets or one adult tabletDose given (mg)501001502002503003.3 Secondary prophylaxis <strong>and</strong> IPT with ART in children3.3.1 Secondary prophylaxisThe <strong>Guidelines</strong> Group noted that there is noevidence on the use of IPT in children livingwith HIV after successful completion of TBtreatment. However, like adults, children livingwith HIV are exposed to reinfection <strong>and</strong> recurrenceof TB. There<strong>for</strong>e, the Group conditionallyrecommends that all children living with HIV whohave been successfully treated <strong>for</strong> TB <strong>and</strong> areliving in settings with a high TB prevalence <strong>and</strong>transmission should receive IPT <strong>for</strong> an additionalsix months. IPT can be started immediately afterthe last dose of anti-TB therapy or at a laterdate. TB screening should be carried out <strong>for</strong> allchildren living with HIV, regardless of history of TBtreatment, during each contact of the child with ahealth-care worker (Annex 13).3.3.2 IPT with ART in childrenThe <strong>Guidelines</strong> Group concluded that there areno data regarding the efficacy of IPT <strong>for</strong> childrenstratified by degree of immunosuppression.However, it was noted that there is biologicalplausibility in extrapolating what is known <strong>for</strong> adults<strong>and</strong> adolescents to children. There<strong>for</strong>e, the <strong>Guidelines</strong>Group conditionally recommends the combined use ofIPT with ART <strong>for</strong> all children. The <strong>Guidelines</strong> Groupalso emphasized that ART should not be delayedwhile starting or completing a course of IPT.[45]3.4 The role of TST <strong>and</strong> IGRA in evaluating children <strong>for</strong> IPTThe <strong>Guidelines</strong> Group, as in the recommendation<strong>for</strong> adults, concluded that TST is not requiredto initiate IPT in children <strong>and</strong> should not beroutinely used as part of the process to determineeligibility <strong>for</strong> IPT (Annex 9). However, the Groupnoted that TST may provide important additionalin<strong>for</strong>mation in assessing a child with suspectedTB, especially if there is no positive contact history.Although a positive TST may indicate infection withmycobacteria, usually Mycobacterium <strong>tuberculosis</strong>, itis not a reliable marker of TB disease activity. Themain limitation of TST in the diagnosis of TB in HIVinfectedchildren is its variable sensitivity. Importantclinical causes of false-negative results includesevere malnutrition, severe TB disease <strong>and</strong> HIVinfection. There<strong>for</strong>e, in settings where it is available,TST may be used <strong>for</strong> the diagnosis of active TB inchildren <strong>and</strong> may also have a role in screening <strong>for</strong>LTBI.Like TST, IGRA cannot distinguish between M.<strong>tuberculosis</strong> infection <strong>and</strong> active TB disease.24