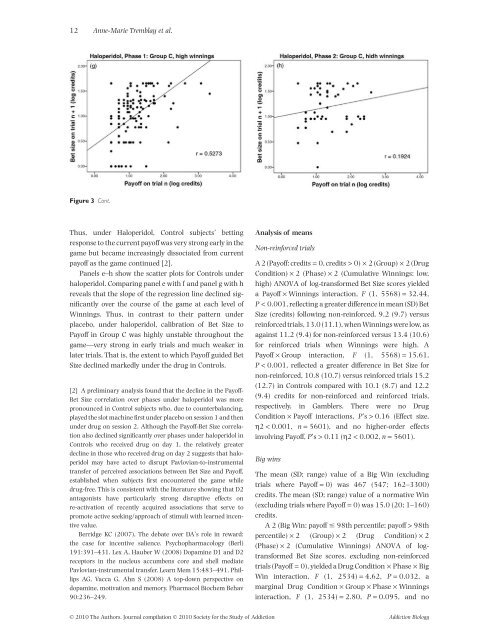
12 Anne-Marie Tremblay et al.Figure 3 Cont.Thus, under <strong>Haloperidol</strong>, Control subjects’ bettingresponse to the current payoff was very strong early in thegame but be<strong>ca</strong>me increasingly dissociated from currentpayoff as the game continued [2].Panels e–h show the s<strong>ca</strong>tter plots for Controls underhaloperidol. Comparing panel e with f <strong>and</strong> panel g with hreveals that the slope of the regression line declined signifi<strong>ca</strong>ntlyover the course of the game at each level ofWinnings. Thus, in contrast to their pattern underplacebo, under haloperidol, <strong>ca</strong>libration of Bet Size toPayoff in Group C was highly unstable throughout thegame—very strong in early trials <strong>and</strong> much weaker inlater trials. That is, the extent to which Payoff guided BetSize declined markedly under the drug in Controls.[2] A preliminary analysis found that the decline in the Payoff-Bet Size correlation over phases under haloperidol was morepronounced in Control subjects who, due to counterbalancing,played the <strong>slot</strong> <strong>machine</strong> first under placebo on session 1 <strong>and</strong> thenunder drug on session 2. Although the Payoff-Bet Size correlationalso declined signifi<strong>ca</strong>ntly over phases under haloperidol inControls who received drug on day 1, the relatively greaterdecline in those who received drug on day 2 suggests that haloperidolmay have acted to disrupt Pavlovian-to-instrumentaltransfer of perceived associations between Bet Size <strong>and</strong> Payoff,established when subjects first encountered the game whiledrug-free. This is consistent with the literature showing that D2antagonists have particularly strong disruptive effects onre-activation of recently acquired associations that serve topromote active seeking/approach of stimuli with learned incentivevalue.Berridge KC (2007), The debate over DA’s role in reward:the <strong>ca</strong>se for incentive salience. Psychopharmacology (Berl)191:391–431. Lex A, Hauber W (2008) Dopamine D1 <strong>and</strong> D2receptors in the nucleus accumbens core <strong>and</strong> shell mediatePavlovian-instrumental transfer. Learn Mem 15:483–491. PhillipsAG, Vac<strong>ca</strong> G, Ahn S (2008) A top-down perspective ondopamine, motivation <strong>and</strong> memory. Pharmacol Biochem Behav90:236–249.Analysis of meansNon-reinforced trialsA 2 (Payoff: credits = 0, credits > 0) ¥ 2 (Group) ¥ 2 (DrugCondition) ¥ 2 (Phase) ¥ 2 (Cumulative Winnings: low,high) ANOVA of log-transformed Bet Size scores yieldeda Payoff ¥ Winnings interaction, F (1, 5568) = 32.44,P < 0.001, reflecting a greater difference in mean (SD) BetSize (credits) following non-reinforced, 9.2 (9.7) versusreinforced trials, 13.0 (11.1), when Winnings were low, asagainst 11.2 (9.4) for non-reinforced versus 13.4 (10.6)for reinforced trials when Winnings were high. APayoff ¥ Group interaction, F (1, 5568) = 15.61,P < 0.001, reflected a greater difference in Bet Size fornon-reinforced, 10.8 (10.7) versus reinforced trials 15.2(12.7) in Controls compared with 10.1 (8.7) <strong>and</strong> 12.2(9.4) credits for non-reinforced <strong>and</strong> reinforced trials,respectively, in Gamblers. There were no DrugCondition ¥ Payoff interactions, P’s > 0.16 (Effect size,h2 < 0.001, n = 5601), <strong>and</strong> no higher-order effectsinvolving Payoff, P’s > 0.11 (h2 < 0.002, n = 5601).Big winsThe mean (SD; range) value of a Big Win (excludingtrials where Payoff = 0) was 467 (547; 162–3300)credits. The mean (SD; range) value of a normative Win(excluding trials where Payoff = 0) was 15.0 (20; 1–160)credits.A 2 (Big Win: payoff 98th percentile; payoff > 98thpercentile) ¥ 2 (Group) ¥ 2 (Drug Condition) ¥ 2(Phase) ¥ 2 (Cumulative Winnings) ANOVA of logtransformedBet Size scores, excluding non-reinforcedtrials (Payoff = 0), yielded a Drug Condition ¥ Phase ¥ BigWin interaction, F (1, 2534) = 4.62, P = 0.032, amarginal Drug Condition ¥ Group ¥ Phase ¥ Winningsinteraction, F (1, 2534) = 2.80, P = 0.095, <strong>and</strong> no© 2010 The Authors. Journal compilation © 2010 Society for the Study of Addiction Addiction Biology
<strong>Haloperidol</strong> modifies instrumental aspects of <strong>slot</strong> <strong>machine</strong> <strong>gambling</strong> 13higher-order interactions, Ps > 0.81 (h2 < 0.001,n = 2564). The three-way interaction reflected a greaterincrease in mean (SD) Bet Size (credits) after a Big Win,22.1 (13.7), than a normative Win, 14.0 (11.8), in Phase1, as opposed to a Big Win, 15.0 (13.1), versus normativeWin, 12.7 (10.4), in Phase 2 under <strong>Haloperidol</strong>. UnderPlacebo, the increase in Bet Size was comparable for a BigWin 24.7 (18.1) versus normative Win 14.5 (11.4) inPhase 1 as for a Big Win 19.1 (9.7) versus normative Win11.3 (8.3) in Phase 2. Thus, Big Wins coincided withincreased Bet Size, <strong>and</strong> this effect was consistent throughoutthe game under Placebo but declined in the second halfof the game under <strong>Haloperidol</strong>.A 2 (Big Win) ¥ 2 (Group) ¥ 2 (Drug Condition) ¥ 2(Cumulative Winnings) ANOVA of Trial number yieldedno signifi<strong>ca</strong>nt effects involving Big Win, Ps > 0.12(h2 < 0.001, n = 2564). Therefore, the timing of a BigWin did not differ as a function of Group, Drug Conditionor Cumulative Winnings.Cumulative winningsA 2 (Group) ¥ 2 (Drug Condition) ¥ 2 (Phase) ANOVA oflog-transformed Cumulative Winnings yielded maineffects of Drug Condition, F (1, 2556) = 8.79, P = 0.003,<strong>and</strong> Phase, F (1, 2556) = 143.96, P < 0.001; a Group ¥Phase interaction, F (1, 2556) = 32.40, P < 0.001, amarginal Drug Condition ¥ Phase interaction, F (1,2556) = 3.68, P = 0.055, <strong>and</strong> no other signifi<strong>ca</strong>nt effects,Ps > 0.16 (h2 < 0.001, n = 2564).The Drug Condition effect reflected greater mean (SD)log-transformed Cumulative Winnings under <strong>Haloperidol</strong>,2.45 (0.42), than Placebo, 2.41 (0.45). However,the raw mean credits were virtually identi<strong>ca</strong>l for <strong>Haloperidol</strong>,392 (347), <strong>and</strong> Placebo, 391 (455). The Phaseeffect reflected greater log-Winnings in Phase 1, 2.51(0.31), than Phase 2, 2.33 (0.52). Corresponding rawmeans were 421 (416) for Phase 1 versus 360 (392) forPhase 2. The Group ¥ Phase effect reflected a largerdecline in log-Winnings from Phase 1, 2.58 (0.30), toPhase 2, 2.28 (0.60), in Controls compared with adecline from 2.48 (0.32) to 2.38 (0.47) in Gamblers. Theraw means for Phases 1 <strong>and</strong> 2 were 513 (597) <strong>and</strong> 392(525) in Controls, as against 371 (254) <strong>and</strong> 346 (293)in Gamblers. The marginal Drug Condition–Phase interactionreflected a somewhat greater decline in log-Winnings from Phase 1, 2.50 (0.28), to Phase 2, 2.30(0.57), under Placebo compared with a decline from2.53 (0.35) to 2.37 (0.47) under <strong>Haloperidol</strong>. The lackof three-way interaction, P = 0.163 (h2 = 0.001,n = 2564), ensured that group differences in the patternof Winnings under drug <strong>and</strong> placebo did not account forthe groups’ different betting profiles under each level ofDrug Condition.DISCUSSIONThis study investigated three main questions: Does <strong>slot</strong><strong>machine</strong> <strong>gambling</strong> conform to the basic principles ofinstrumental reinforcement? Does DA mediate thisprocess? Do PG subjects differ from Controls in theirbetting profile under placebo <strong>and</strong> their response tomanipulation of DA? The correlation between Payoff on agiven trial <strong>and</strong> Bet Size on the next trial operationallydefined reinforcement: the stronger the correlation, thegreater the reinforcement.Evidence from animals suggested that a modest dose ofhaloperidol would induce a within-session decline inreinforced responding in healthy subjects (Sanger & Perrault1995). Neuroimaging evidence from otherwisehealthy subjects with PG showing hypo-responsiveness tomonetary reward in the ventral striatum, the terminalregion for mesolimbic DA neurons, suggested that a givenPayoff would be less reinforcing for PG subjects than Controlsunder placebo (Reuter et al. 2005). Parallel profilesof reward-related responding in patients with Parkinson’sdisease <strong>and</strong> healthy subjects under a modest dose ofhaloperidol suggested that haloperidol would simulatepatterns seen in Parkinson’s patients in response to <strong>gambling</strong>(Frank & O’Reilly 2006; Steeves et al. 2009). Specifi<strong>ca</strong>lly,blockade of inhibitory D2 receptors under amodest dose of a D2 antagonist should increase rewardrelatedDA release in PG subjects, with a correspondingincrease in instrumental responding to rewarding stimuliunder the drug.In line with our first hypothesis, Payoff reliably predictedBet Size on the subsequent trial regardless ofother factors. The proportional increase in the criterionresponse (Betting) as a function of variations in theoutcome of that response (Payoff) indi<strong>ca</strong>tes that <strong>slot</strong><strong>machine</strong> <strong>gambling</strong> conforms to the basic principles ofinstrumental conditioning. In Control subjects, whowere essentially naive to <strong>slot</strong> <strong>machine</strong>s, the Payoff–BetSize correlation was highly consistent over the course oftrials under placebo <strong>and</strong> seemingly unaffected by availableresources in the form of Cumulative Winnings. Incontrast, PG subjects’ betting response to reward underplacebo varied over the course of the game dependingon Cumulative Winnings, becoming less responsive overtrials when Winnings were low but more responsiveover trials when Winnings were high. This pattern suggeststhat PG subjects were betting strategi<strong>ca</strong>lly to maximizetheir opportunity to extend play based on theiravailable resources. Based on findings from co<strong>ca</strong>ineabusers (Goldstein et al. 2007), PG subjects underplacebo were expected to be relatively insensitive tovariations in monetary reward throughout the gamecompared with healthy subjects. Accordingly, thePayoff–Bet Size correlation under placebo, averaged over© 2010 The Authors. Journal compilation © 2010 Society for the Study of Addiction Addiction Biology