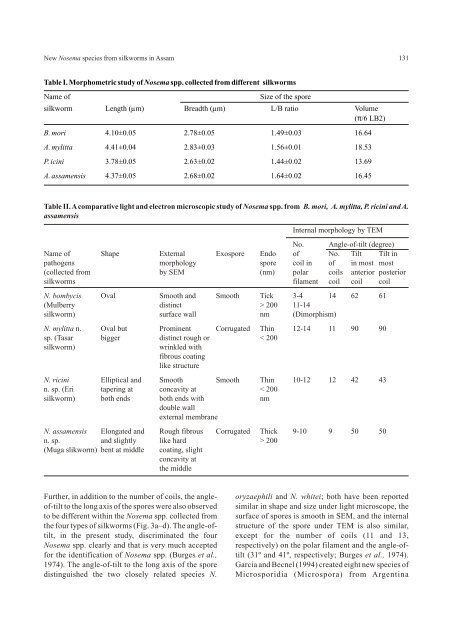
New Nosema species from silkworms in Assam131Table I. Morphometric study of Nosema spp. collected from different silkwormsName ofSize of the sporesilkworm Length (µm) Breadth (µm) L/B ratio Volume(π/6 LB2)B. mori 4.10±0.05 2.78±0.05 1.49±0.03 16.64A. mylitta 4.41±0.04 2.83±0.03 1.56±0.01 18.53P. icini 3.78±0.05 2.63±0.02 1.44±0.02 13.69A. assamensis 4.37±0.05 2.68±0.02 1.64±0.02 16.45Table II. A comparative light and electron microscopic study of Nosema spp. from B. mori, A. mylitta, P. ricini and A.assamensisInternal morphology by TEMNo. Angle-of-tilt (degree)Name of Shape External Exospore Endo of No. Tilt Tilt inpathogens morphology spore coil in of in most most(collected from by SEM (nm) polar coils anterior posteriorsilkworms filament coil coil coilN. bombycis Oval Smooth and Smooth Tick 3-4 14 62 61(Mulberry distinct > 200 11-14silkworm) surface wall nm (Dimorphism)N. mylitta n. Oval but Prominent Corrugated Thin 12-14 11 90 90sp. (Tasar bigger distinct rough or < 200silkworm)wrinkled withfibrous coatinglike structureN. ricini Elliptical and Smooth Smooth Thin 10-12 12 42 43n. sp. (Eri tapering at concavity at < 200silkworm) both ends both ends with nmdouble wallexternal membraneN. assamensis Elongated and Rough fibrous Corrugated Thick 9-10 9 50 50n. sp. and slightly like hard > 200(Muga slikworm) bent at middle coating, slightconcavity atthe middleFurther, in addition to the number of coils, the angle- oryzaephili and N. whitei; both have been reportedof-tilt to the long axis of the spores were also observed similar in shape and size under light microscope, theto be different within the Nosema spp. collected from surface of spores is smooth in SEM, and the internalthe four types of silkworms (Fig. 3a–d). <strong>The</strong> angle-of- structure of the spore under TEM is also similar,tilt, in the present study, discriminated the four except <strong>for</strong> the number of coils (11 and 13,Nosema spp. clearly and that is very much accepted respectively) on the polar filament and the angle-of<strong>for</strong>the identification of Nosema spp. (Burges et al., tilt (31º and 41º, respectively; Burges et al., 1974).1974). <strong>The</strong> angle-of-tilt to the long axis of the spore Garcia and Becnel (1994) created eight new species ofdistinguished the two closely related species N. Microsporidia (Microspora) from Argentina
132Chakrabarti and Mannamosquito (Dipitera: Culicidae) on the basis of the Fowler JL and Reeves EL. 1975. Microsporidian sporenumber of coils in a polar filament and the angle-oftilt.strutcure as revealed by SEM. J Invert Pathol 26:1-6.<strong>The</strong> internal structure of microsporidian spores Fujiwara T. 1980. Three microsporidians (Nosema sp.) fromdiffers greatly among different species; nevertheless, the silkworm, Bombyx mori. J Sericul Sci Japan 49:229-the similarities remain more impressive than the 236.differences (Kudo and Daniels, 1963; Sprague, 1965). Fujiwara T. 1984. A Pleistophora-like microsporidia isolated<strong>The</strong>re<strong>for</strong>e, apparently, all the microsporidian species from the silkworm, Bombyx mori. J Sericul Sci Japanhave a similar basic pattern of spore structure, barring 59:398-402.some minor variations (Sprague et al., 1968).Garcia JJ and Becnel JJ. 1994. Eight new species ofmicrosporidia (Microspora) from Argentine mosquitoesIn conclusion, based on our light microscopy, SEM(Diptera: Culicidae). J Invert Pathol 64:243-252.and TEM obserations, we have demonstrated that thespores of four different Nosema spp. isolated from Graaf DCD, Raes H and Jacobs FJ. 1994. Note: sporefour different silkworms, differ from each other in dimorphism in Nosema apis (Microsporidia: Nosematidae)development cycle. J Invert Pathol 63: 92-94.more than twelve characteristics (Table I and II).<strong>The</strong>re<strong>for</strong>e, the authors propose that these should be Ishihara R and Iwano H. 1991. <strong>The</strong> lawn grass cutworm,considered as three distinct new species of the genus Spodoptera depravata, Butler as a natural reservoir ofNosema, and suggest their names as N. mylitta n. sp. Nosema bombycis Nageli. J Sericul Sci Japan 60:236-239.collected from A. mylitta Druary, N. assamensis n. sp. Iwano H and Ishihara R. 1989. Intracellular germination offrom A. assamensis Westwood and N. ricini n. sp. from spore of a Nosema sp. immediately after their <strong>for</strong>mation inP. ricini Boisd. However, the typical number of coils, culture cell. J Invert Pathol 54:125-127.rather than the length of polar filament in the spore, isslightly different in all the four species. <strong>The</strong>re<strong>for</strong>e, thenumber of coils and the angle-of-tilt to the long axis ofthe spores distinguish them into four different species.ACKNOWLEDGEMENTSIwano H and Ishihara R. 1991. Dimorphic development ofNosema bombycis spore in gut epithelium of larva of thesilkworm Bombyx mori. J Sericul Sci Japan 60:249-256.Jolly MS. 1986. Pebrine and its control. Central SericulturalResearch and Training Institute, Central Silk Board,Manandavadi, Mysore, India.<strong>The</strong> authors gratefully acknowledge Dr. A. K. Kudo R. 1924. A biology and taxonomic study of theChandra, Deputy Director and Dr. A. K. Sarkar, microsporidia, III, Biology Monograph 9:1-268.Director, Central Sericulture Research and TrainingKudo R and Daniels EW. 1963. An electron microscope studyInstitute, Berhampore, West Bengal, <strong>for</strong> their kind of the spore of a microsporidia, <strong>The</strong>lohania cali<strong>for</strong>nica. Jhelp by providing laboratory facilities, suggestions Ptotozool 10:112-120.and encouragement <strong>for</strong> this work. <strong>The</strong> senior author isLarson RJ.1999. Identification of Microsporidia. Actavery much indebted to Dr. B. Saratchandra, DirectorProtozoolo 38:161-197.(Tech.), Central Silk Board, Bangalore and to Dr. RajeUrs, Director, Seri Bio Tech Laboratory, Kodathi, Lom J and Weiser J. 1972. Surface pattern of someBangalore, <strong>for</strong> their interest and constant microsporidan spore as seen in SEM. Foliaparasitolo19:359-363.encouragement. <strong>The</strong> authors also express theirgratitude to the Director, <strong>Indian</strong> Institute of Chemical Milner RJ. 1972a. Nosema whitei, a microsporidia pathogen ofBiology, Kolkata, <strong>for</strong> his kind permission <strong>for</strong> TEM some species of Tribolium. J Invert Pathol 19:230-247.study.Milner RJ. 1972b. Nosema whitei, a microsporidia pathogen ofsome species of Tribolium III. J Invert Pathol 19:248-255.REFERENCESPatil C S. 1993. Review of pebrine, a microsporidian disease inAnja RK. 1993. Experiments in Microbiology, Plant Pathologythe silkworm, Bombyx mori L. Sericologia 33:201-210.and Tissue culture, Wisha Prakashan, New Delhi. pp.31-36.Rausch M and Grunewald J. 1981. Light and electronBurges HD, Canning EU and Hulls IK. 1974. Ultrastructure ofmicroscopic observation of some microsporidia parasitesNosema oryzaephili and the taxonomic value of the polar(Cnidosporidia: Microsporidia) of black fly larva (Diptera:filament. J Invert Pathol 23:135-139.Simuliidae). Zhurnal Parasitenkunde 63:1-2.Cali A. 1970. Morphogenesis in the genus Nosema.Sato R, Kobayashi M and Watanabe H. 1982. Internal structureProceedings of the IVth International Colloquium Insectof Microsporidia isolated from the silkworm Bombyx mori.Pathology, Maryland. pp 431-438.J Invert Pathol 40:260-265.