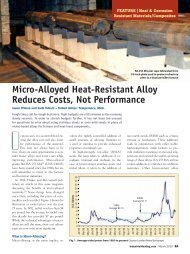
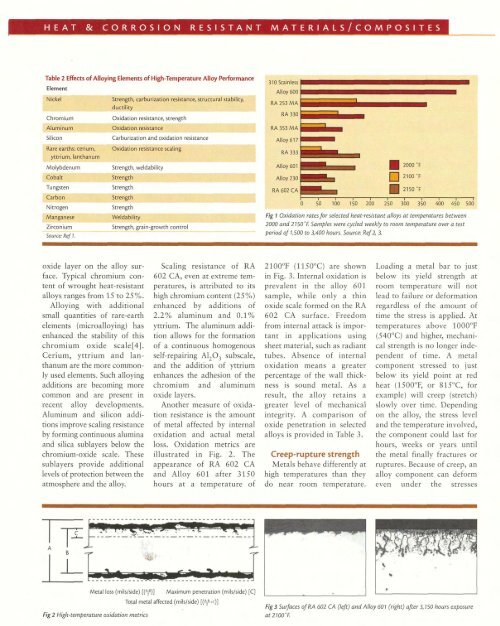
Z~rcon~um- -Strength, gram-growth contrc Source: Ref 1.Fig 1 Oxrdation rates for selected heat-resistant alloys at temperatures between2000 and 2150'E Samples were cycled weekly to room temperature over a testperiod of 7,500 to 3,400 hours. Source: Ref 2,3.oxide layer on the alloy surface.Typical chromium contentof wrought heat-resistantalloys ranges from 15 to 25%.<strong>Alloy</strong>ing with additionalsmall quantities of rare-earthelements (microalloying) hasenhanced the stability of thischromium oxide scale[4].Cerium, yttrium and lanthanumare the more commonlyused elements. Such alloyingadditions are becoming morecommon and are present inrecent alloy developments.Aluminum and silicon additionsimprove scaling resistanceby forming continuous aluminaand silica sublayers below thechromium-oxide scale. Thesesublayers provide additionallevels of protection between theatmosphere and the alloy.Scaling resistance of RA602 CA, even at extreme temperatures,is attributed to itshigh chromium content (25%)enhanced by additions of2.2% aluminum and 0.1%yttrium. The aluminum additionallows for the formationof a continuous homogenousself-repairing A1203 subscale,and the addition of yttriumenhances the adhesion of thechromium and aluminumoxide layers.Another measure of oxidationresistance is the amountof metal affected by internaloxidation and actual metalloss. Oxidation metrics areillustrated in Fig. 2. Theappearance of RA 602 CAand <strong>Alloy</strong> 601 after 3150hours at a temperature of2100°F (1150°C) are shownin Fig. 3. Internal oxidation isprevalent in the alloy 601sample, while only a thinoxide scale formed on the RA602 CA surface. Freedomfrom internal attack is importantin applications usingsheet material, such as radianttubes. Absence of internaloxidation means a greaterpercentage of the wall thicknessis sound metal. As aresult, the alloy retains agreater level of mechanicalintegrity. A comparison ofoxide penetration in selectedalloys is provided in Table 3.Creep-rupture strengthMetals behave differently athigh temperatures than theydo near room temperature.Loading a metal bar to justbelow its yield strength atroom temperature will notlead to failure or deformationregardless of the amount oftime the stress is applied. Attemperatures above 1000°F(540°C) and higher, mechanicalstrength is no longer independentof time. A metalcomponent stressed to justbelow its yield point at redheat (1500°F, or 81S°C, forexample) will creep (stretch)slowly over time. Dependingon the alloy, the stress leveland the temperature involved,the component could last forhours, weeks or years untilthe metal finally fractures orruptures. Because of creep, analloy component can deformeven under the stressesMetal loss (milslside)[(?)I<strong>Maximum</strong> penetration (milslside)[C]Total metal affected (milslside)[(y+c)]Fig2 High-temperature oxidation metricsFig3 Surfaces of RA GO2 CA (left) and <strong>Alloy</strong> GO1 (right) after 3,150 hours exposureat2100"E
imposed by supporting itsown weight. For this reason,the creep rupture strength isan important design criterionfor any alloy intended for useabove 1000°F.The creep rupture propertiesof heat resisting alloysare heavily dependent onalloy content and grain size.In the case of RA 602 CA, itshigh creep-rupture strengthprimarily is a result of alloycontent[S]. The relativelyhigh carbon level ensures theprecipitation of homogeneouslydistributed bulkycarbides. Additions of titaniumand zirconium ensurethat these carbides and alsocarbonitrides are finely dispersed.Even solution annealingat 2230°F (1220°C) doesnot dissolve the carbidescompletely. Thus, the alloyachieves its high creep ruptureproperties from a combinationof solid-solution hardeningand carbide strengthening.The result is an alloyhaving greater creep-ruptureproperties than traditionalalloys such as RA330 andalloy 601 (Tables 4 and 5).Further comparison indicatesthat at extreme temperatures,RA 602 CA compares favorablyto other heat-resistantsuperalloys such as <strong>Alloy</strong> 214and <strong>Alloy</strong> 230 (Table 6).Grain growthBrittle fracture of componentsexposed to extremetemperatures is a commonoccurrence. Operating at temperaturesnear or exceeding1800°F (980°C) will lead tograin growth. A very coarsegrain structure negativelyaffects alloy performance inseveral ways. Reduced resistanceto thermal fatigue undercyclic conditions can causecracking and fracturing.Susceptibility to corrosivemechanisms that preferentiallyattack at the grain boundariesalso is increased. Suchmechanisms include carburization,molten chloride-saltattack and corrosion by halogens.The attack is accelerateddue to the reduced grainboundary volume and themore direct path into thealloy from the surface. Suchtypes of attack reduce ductility.Often, the result is a componentprone to brittle fracture.An example of such afailure is shown in Figure 4.A small addition (approximately0.08%) of zirconium inRA 602 CA is effective in pinninggrain boundaries, thusgreatly slowing the rate ofgrain growth[9]. The results ofa recent grain growth study aredetailed in Table 7. The informationcompares the graingrowth of several heat-resistantalloys that are commonlyused at temperatures above1900°F (1040°C). The datawas compiled from intermittentexposure of mill-annealedsample coupons to a temperatureof 2050°F (1120°C) for atotal of 990 hours.Further evidence of thegrain-growth resistance of RA602 CA came from the examinationof samples used in earliercyclic oxidation testing.After exposure to 2100°F(1150°C) for 3,400 hours and2150°F (1180°C) for morethan 1,500 hours, the grainsize only changed from ASTM7 to ASTM 5.5 in both cases.In contrast, the grain size ofalloy 601 exposed in the sametests increased to greater thanASTM 00.The pick up of carbon fromhigh temperature atmospheresis another potentialreason for an alloy to lose its tures can optimize manyductility during service. Table processes by increasing pro-8 compares the carburization duction, enhancing properresistanceof several heat ties and improving the qualiresistantalloys.ty of parts being heat treated.The properties of traditionalConclusions heat-resistant materials haveHigher operating tempera- limited furnace operators toTable 3 High-temperature internal oxidation of some heat-resistant alloysTemperature, OF2000 2100 2150Time, h Pene., mil Time, h Pene., mil Time, h Pene., milTable 4 Average stress to rupture in 10,000 hStress, psiTemperature, OFlG00 1800 2000<strong>Alloy</strong> 601 6.000 2,700 1.100 GOO(a) Extrapolated. Source. Ref 6.Table 5 Average strws for 1% creep in 1,000 hStress, psiTemperature, OFlG00 1800 2(Table 6 Average stress to rupture in 1,000 h(u) Extrapolated. Source: Ref7,8.3Stress, psiTemperature, O800 190000 GOO



![RA333 Data Sheet [Heat Resistant Alloys] - Rolled Alloys](https://img.yumpu.com/50335849/1/190x245/ra333-data-sheet-heat-resistant-alloys-rolled-alloys.jpg?quality=85)