Combustion - 國立中央大學機械工程學系
Combustion - 國立中央大學機械工程學系
Combustion - 國立中央大學機械工程學系
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
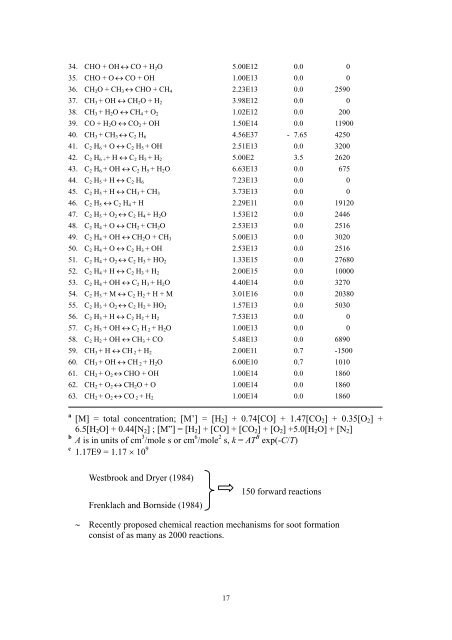
34. CHO + OH ↔ CO + H 2 O 5.00E12 0.0 035. CHO + O ↔ CO + OH 1.00E13 0.0 036. CH 2 O + CH 3 ↔ CHO + CH 4 2.23E13 0.0 259037. CH 3 + OH ↔ CH 2 O + H 2 3.98E12 0.0 038. CH 3 + H 2 O ↔ CH 4 + O 2 1.02E12 0.0 20039. CO + H 2 O ↔ CO 2 + OH 1.50E14 0.0 1190040. CH 3 + CH 3 ↔ C 2 H 6 4.56E37 - 7.65 425041. C 2 H 6 + O ↔ C 2 H 5 + OH 2.51E13 0.0 320042. C 2 H 6 + + H ↔ C 2 H 5 + H 2 5.00E2 3.5 262043. C 2 H 6 + OH ↔ C 2 H 5 + H 2 O 6.63E13 0.0 67544. C 2 H 5 + H ↔ C 2 H 6 7.23E13 0.0 045. C 2 H 5 + H ↔ CH 3 + CH 3 3.73E13 0.0 046. C 2 H 5 ↔ C 2 H 4 + H 2.29E11 0.0 1912047. C 2 H 5 + O 2 ↔ C 2 H 4 + H 2 O 1.53E12 0.0 244648. C 2 H 4 + O ↔ CH 2 + CH 2 O 2.53E13 0.0 251649. C 2 H 4 + OH ↔ CH 2 O + CH 3 5.00E13 0.0 302050. C 2 H 4 + O ↔ C 2 H 3 + OH 2.53E13 0.0 251651. C 2 H 4 + O 2 ↔ C 2 H 3 + HO 2 1.33E15 0.0 2768052. C 2 H 4 + H ↔ C 2 H 3 + H 2 2.00E15 0.0 1000053. C 2 H 4 + OH ↔ C 2 H 3 + H 2 O 4.40E14 0.0 327054. C 2 H 3 + M ↔ C 2 H 2 + H + M 3.01E16 0.0 2038055. C 2 H 3 + O 2 ↔ C 2 H 2 + HO 2 1.57E13 0.0 503056. C 2 H 3 + H ↔ C 2 H 2 + H 2 7.53E13 0.0 057. C 2 H 3 + OH ↔ C 2 H 2 + H 2 O 1.00E13 0.0 058. C 2 H 2 + OH ↔ CH 3 + CO 5.48E13 0.0 689059. CH 3 + H ↔ CH 2 + H 2 2.00E11 0.7 -150060. CH 3 + OH ↔ CH 2 + H 2 O 6.00E10 0.7 101061. CH 2 + O 2 ↔ CHO + OH 1.00E14 0.0 186062. CH 2 + O 2 ↔ CH 2 O + O 1.00E14 0.0 186063. CH 2 + O 2 ↔ CO 2 + H 2 1.00E14 0.0 1860a[M] = total concentration; [M’] = [H 2 ] + 0.74[CO] + 1.47[CO 2 ] + 0.35[O 2 ] +6.5[H 2 O] + 0.44[N 2 ] ; [M”] = [H 2 ] + [CO] + [CO 2 ] + [O 2 ] +5.0[H 2 O] + [N 2 ]bA is in units of cm 3 /mole s or cm 6 /mole 2 s, k = AT B exp(-C/T)c 1.17E9 = 1.17 × 10 9Westbrook and Dryer (1984)Frenklach and Bornside (1984)150 forward reactions∼ Recently proposed chemical reaction mechanisms for soot formationconsist of as many as 2000 reactions.17