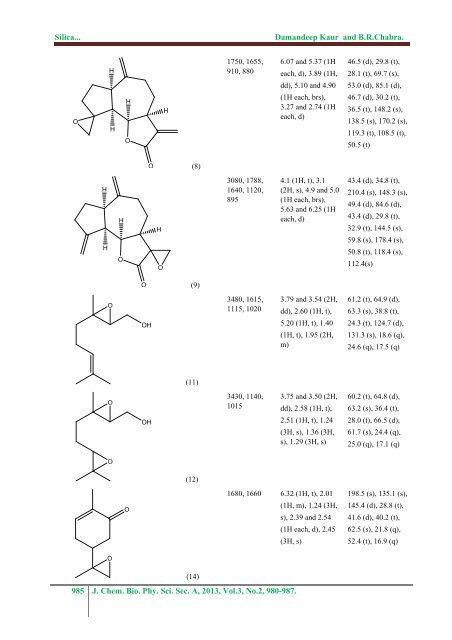
<strong>Silica</strong>...Damandeep Kaur and B.R.Chabra.OHHHOH1750, 1655,910, 8806.07 and 5.37 (1Heach, d), 3.89 (1H,dd), 5.10 and 4.90(1H each, brs),3.27 and 2.74 (1Heach, d)46.5 (d), 29.8 (t),28.1 (t), 69.7 (s),53.0 (d), 85.1 (d),46.7 (d), 30.2 (t),36.5 (t), 148.2 (s),138.5 (s), 170.2 (s),119.3 (t), 108.5 (t),50.5 (t)O(8)HHHOHO3080, 1788,1640, 1120,8954.1 (1H, t), 3.1(2H, s), 4.9 and 5.0(1H each, brs),5.63 and 6.25 (1Heach, d)43.4 (d), 34.8 (t),210.4 (s), 148.3 (s),49.4 (d), 84.6 (d),43.4 (d), 29.8 (t),32.9 (t), 144.5 (s),59.8 (s), 178.4 (s),50.8 (t), 118.4 (s),112.4(s)O(9)OOH3480, 1615,1115, 10203.79 and 3.54 (2H,dd), 2.60 (1H, t),5.20 (1H, t), 1.40(1H, t), 1.95 (2H,m)61.2 (t), 64.9 (d),63.3 (s), 38.8 (t),24.3 (t), 124.7 (d),131.3 (s), 18.6 (q),24.6 (q), 17.5 (q)(11)OOH3430, 1140,10153.75 and 3.50 (2H,dd), 2.58 (1H, t),2.51 (1H, t), 1.24(3H, s), 1.36 (3H,s), 1.29 (3H, s)60.2 (t), 64.8 (d),63.2 (s), 36.4 (t),28.0 (t), 66.5 (d),61.7 (s), 24.4 (q),25.0 (q), 17.1 (q)O(12)O1680, 1660 6.32 (1H, t), 2.01(1H, m), 1.24 (3H,s), 2.39 and 2.54(1H each, d), 2.45(3H, s)198.5 (s), 135.1 (s),145.4 (d), 28.8 (t),41.6 (d), 40.2 (t),62.5 (s), 21.8 (q),52.4 (t), 16.9 (q)O(14)985 J. Chem. Bio. Phy. Sci. Sec. A, 2013, Vol.3, No.2, 980-987.
<strong>Silica</strong>...Damandeep Kaur and B.R.Chabra.OO1720, 1685,3075, 8902.75 (1H, t), 2.51(1H, m), 1.80 (3H,s), 4.90 and 5.10(1H each, d), 1.34(3H, s)205.4 (s), 62.5 (s),62.9 (d), 29.9 (t),35.8 (d), 42.5 (t),147.8 (s), 21.5 (q),110.9 (t), 15.5 (q)(15)RESULTS AND DISCUSSIONThe results obtained under the most favorable conditions using UHP- SiO 2 <strong>as</strong> the oxidising reagent arebetter than those obtained with aqueous H 2 O 2 under the same reaction conditions. The reagent is morestable and could be stored for several months without the loss <strong>of</strong> weight or its activity when compared toUHP <strong>as</strong> such. It does not need any special handling <strong>as</strong> is required in the c<strong>as</strong>e <strong>of</strong> <strong>peroxide</strong>s. This reagentbeing inexpensive, non toxic and e<strong>as</strong>ily available is quite efficient for various organic transformations.Therefore, it is recommended for use in different purposes in organic chemistry which require <strong>hydrogen</strong><strong>peroxide</strong>, <strong>as</strong> the e<strong>as</strong>y recovery <strong>of</strong> the product using this reagent make these processes moreenvironmental friendly.List <strong>of</strong> abbreviations used:<strong>Silica</strong> <strong>supported</strong> <strong>urea</strong>-<strong>hydrogen</strong> <strong>peroxide</strong> UHP-SiO 2Hydrogen <strong>peroxide</strong> H 2 O 2Urea <strong>hydrogen</strong> <strong>peroxide</strong>UHPtetrahydr<strong>of</strong>uranTHFACKNOWLEDGEMENTThe authors thank the Punjab Government for financial support under the project ‘Chemistry andpotential <strong>of</strong> some natural products and synthetic heterocyclics <strong>as</strong> agrochemicals’.REFERENCES1. C. L. Hill, C. M. Prosser-McCartha, Coordination. Chem. Rev., 1995, 143, 407.2. I. V. Kozhevnikov, Chem. Rev., 1998, 98, 171.3. T. Okuhara, N. Mizuno, M. Misono, Adv. Catal., 1996, 41, 113.4. D. K. Lyon, W. K. Miller, T. Novet, P. J. Domaille, E. Evitt, D. C. Johnson, R. G. Finke, J. Am. Chem.Soc., 1991, 113, 7209.5. D. Mansuy, J. F. Bartoli, P. Battioni, D. K. Lyon, R. G. Finke, J. Am. Chem. Soc., 1991, 113, 7222.6. R. Manktala, R. S. Dhillon, B. R. Chhabra, Ind. J. Chem., 2006, 45B, 1591.7. N. K. Kala Raj, A. V. Ram<strong>as</strong>wamy, P. Manikandan, J. Mol. Catal. A : Chem., 2005, 227, 37.8. C. Lu, E. W. Hughes, P. A. Giguere, J. Am. Chem. Soc., 1941, 63, 1507.9. R. B. Milller, E. D. Behare, J Am Chem Soc, 1974, 96, 8102.986 J. Chem. Bio. Phy. Sci. Sec. A, 2013, Vol.3, No.2, 980-987.