Ch 14 Mixtures and Solutions Big Idea: Nearly all of the gases ...
Ch 14 Mixtures and Solutions Big Idea: Nearly all of the gases ...
Ch 14 Mixtures and Solutions Big Idea: Nearly all of the gases ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Ch</strong> <strong>14</strong> <strong>Mixtures</strong> <strong>and</strong> <strong>Solutions</strong><strong>Big</strong> <strong>Idea</strong>:<strong>Nearly</strong> <strong>all</strong> <strong>of</strong> <strong>the</strong> <strong>gases</strong>, liquids <strong>and</strong> solidsthat make up our world are mixtures.I CAN:• Describe <strong>the</strong> concentration <strong>of</strong> solutionsusing different units;• Compare properties <strong>of</strong> suspensions,colloids <strong>and</strong> solutions;• Determine colligative properties <strong>of</strong> asolution1
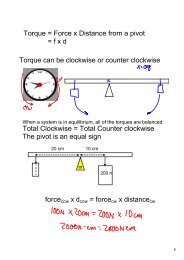
K b = boiling point constant: 0.52 ºC/m (for water)K f = freezing point constant: 1.86 ºC/m (for water)16
Tynd<strong>all</strong> Effect colloid particles scatter light,solution particles do not20
∆T b = K b x m x i∆T f= K f x m x iK b = boiling point constant: 0.52 ºC/m (for water)K f = freezing point constant: 1.86 ºC/m (for water)21