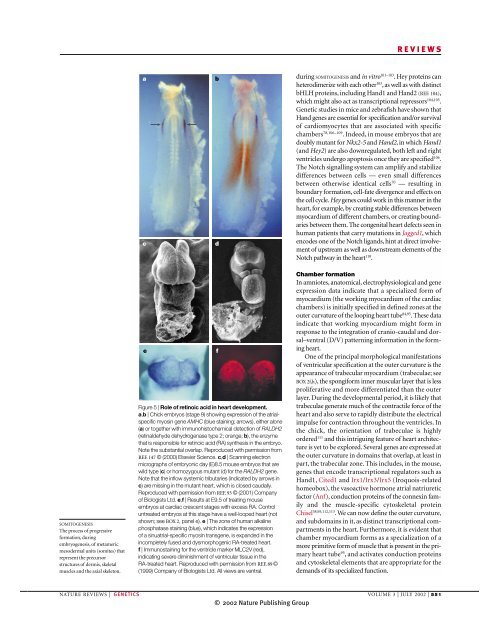
REVIEWSacbdduring SOMITOGENESIS and in vitro 101–103 . Hey proteins canheterodimerize with each other 103 , as well as with distinctbHLH proteins, including Hand1 and Hand2 (REF. 104),which might also act as transcriptional repressors 104,105 .Genetic studies in mice and zebrafish have shown thatHand genes are essential for specification and/or survivalof cardiomyocytes that are associated with specificchambers 78,106–109 . Indeed, in mouse embryos that aredoubly mutant for Nkx2-5 and Hand2, in which Hand1(and Hey2) are also downregulated, both left and rightventricles undergo apoptosis once they are specified 106 .The Notch signalling system can amplify and stabilizedifferences between cells — even small differencesbetween otherwise identical cells 70 — resulting inboundary formation, cell-fate divergence and effects onthe cell cycle. Hey genes could work in this manner in theheart, for example, by creating stable differences betweenmyocardium of different chambers, or creating boundariesbetween them. The congenital heart defects seen inhuman patients that carry mutations in Jagged1, whichencodes one of the Notch ligands, hint at direct involvementof upstream as well as downstream elements of theNotch pathway in the heart 110 .SOMITOGENESISThe process of progressiveformation, duringembryogenesis, of metamericmesodermal units (somites) thatrepresent the precursorstructures of dermis, skeletalmuscles and the axial skeleton.eFigure 5 | Role of retinoic acid in heart development.a,b | Chick embryos (stage 9) showing expression of the atrialspecificmyosin gene AMHC (blue staining; arrows), either alone(a) or together with immunohistochemical detection of RALDH2(retinaldehyde dehydrogenase type 2; orange; b), the enzymethat is responsible for retinoic acid (RA) synthesis in the embryo.Note the substantial overlap. Reproduced with permission fromREF. 147 © (2000) Elsevier Science. c,d | Scanning electronmicrographs of embryonic day (E)8.5 mouse embryos that arewild type (c) or homozygous mutant (d) for the RALDH2 gene.Note that the inflow systemic tributaries (indicated by arrows inc) are missing in the mutant heart, which is closed caudally.Reproduced with permission from REF. 93 © (2001) Companyof Biologists Ltd. e,f | Results at E9.5 of treating mouseembryos at cardiac crescent stages with excess RA. Controluntreated embryos at this stage have a well-looped heart (notshown; see BOX 2, panel e). e | The zone of human alkalinephosphatase staining (blue), which indicates the expressionof a sinuatrial-specific myosin transgene, is expanded in theincompletely fused and dysmorphogenic RA-treated heart.f | Immunostaining for the ventricle marker MLC2V (red),indicating severe diminishment of ventricular tissue in theRA-treated heart. Reproduced with permission from REF. 89 ©(1999) Company of Biologists Ltd. All views are ventral.fChamber formationIn amniotes, anatomical, electrophysiological and geneexpression data indicate that a specialized form ofmyocardium (the working myocardium of the cardiacchambers) is initially specified in defined zones at theouter curvature of the looping heart tube 84,95 . These dataindicate that working myocardium might form inresponse to the integration of cranio-caudal and dorsal–ventral(D/V) patterning information in the formingheart.One of the principal morphological manifestationsof ventricular specification at the outer curvature is theappearance of trabecular myocardium (trabeculae; seeBOX 2f,h), the spongiform inner muscular layer that is lessproliferative and more differentiated than the outerlayer. During the developmental period, it is likely thattrabeculae generate much of the contractile force of theheart and also serve to rapidly distribute the electricalimpulse for contraction throughout the ventricles. Inthe chick, the orientation of trabeculae is highlyordered 111 and this intriguing feature of heart architectureis yet to be explored. Several genes are expressed atthe outer curvature in domains that overlap, at least inpart, the trabecular zone. This includes, in the mouse,genes that encode transcriptional regulators such asHand1, Cited1 and Irx1/Irx3/Irx5 (Iroquois-relatedhomeobox), the vasoactive hormone atrial natriureticfactor (Anf), conduction proteins of the connexin familyand the muscle-specific cytoskeletal proteinChisel 38,95,112,113 . We can now define the outer curvature,and subdomains in it, as distinct transcriptional compartmentsin the heart. Furthermore, it is evident thatchamber myocardium forms as a specialization of amore primitive form of muscle that is present in the primaryheart tube 95 , and activates conduction proteinsand cytoskeletal elements that are appropriate for thedemands of its specialized function.NATURE REVIEWS | GENETICS VOLUME 3 | JULY 2002 | 551© 2002 Nature Publishing Group
REVIEWSHow are these patterns established? The neuregulin/Erbbsignalling system is essential for the formationof trabeculae 114–116 . Neuregulin1 is expressed inthe endocardium and signals to its membrane-boundtyrosine kinase co-receptors Erbb2 and Erbb4, whichare expressed in myocardium. Endocardium is separatedfrom myocardium by a complex proteoglycanand glycosamino-glycan-rich matrix called cardiacjelly, and endocardium makes contact withmyocardium in numerous places through an invasiveprocess 111 . Neuregulin1 signalling might occur at thispoint of contact and/or across the cardiac-jelly matrixthrough its diffusible isoforms. Why trabeculae formonly on the outer curvature of the heart is probablyrelevant to the issue of how working myocardium isspecified. This spatial specificity seems not to be governedby localized expression of the neuregulin coreceptorErbb2, because the global expression of theErbb2 gene in myocardium through targeted integrationinto the Nkx2-5 locus does not seem to expandthe zone of working myocardium 117 . The associationand functional coupling of Erbb2 with CD44,a membranesignalling protein that uses cardiac-jelly componentssuch as hyaluronan as ligands 118 , hints at matrixelements being an important aspect of specification ofthe outer curvature. Indeed, trabeculae are completelylost in mouse embryos that have mutations in thehyaluronan synthetase-2 gene (Has2) 119 . Interestingly,the serotonin 2B receptor is also required for propergrowth and trabeculation of the ventricles, and fornormal levels of Erbb2 expression 120 . This receptor actsthrough the heterotrimeric GTPases Gα qand Gα 11,and embryos that are doubly mutant for their encodinggenes also show a ventricular defect 121 . Studies onthe epidermal growth factor (EGF) receptor, anothermember of the Erbb family, indicate that the intracellulardomain of Erbb receptors can act as an integrativeplatform for signalling through variousligand/receptor types, including G-protein-coupledreceptors, which activate the mitogen-activatedprotein kinase pathway 122 . Understanding theseinteractions might be necessary to fully appreciatechamber formation.Several transcription factors are now implicated inthe formation of the outer-curvature zones that willbecome the working myocardium. The Holt–OramT-box factor, Tbx5, interacts directly with the homeodomainfactor Nkx2-5 and, in vitro, these factorscooperatively activate expression of the Anf (Nppa) 123and connexin 40 (Gja5) genes — markers of workingmyocardium in the embryo 94,95 . Both Tbx5 and Nkx2-5have also been implicated genetically in the formationof working myocardium. In homozygous-null Nkx2-5mutants, all tested markers of the outer curvature,including Nppa, Cited1, Chisel and Hand1, are abolishedor severely downregulated 38,77,112,124–126 . Mutanthearts are unlooped and retain a molecular signaturethat is similar to that of the primary heart tube. Tbx5-mutant hearts also show poorly looped and hypoplastichearts, and expression of the Nppa gene is abolished94 . We have recently shown in the mouse that theneuregulin1 signalling pathway maintains expressionof Nkx2-5 and Tbx5, as well as numerous other cardiactranscription factor and downstream genes,in the developing left and possibly right ventricles(D. Li, M. Zhou and R.P.H., unpublished observations).A rudimentary heart phenotype is also foundin mouse embryos that lack forkhead box H1a(Foxh1a), a gene that encodes Fast2, a winged helixDNA-binding and Smad-interacting factor that actsdownstream in the transforming growth factor-β(Tgf-β)(activin–Nodal) signalling pathway 127,128 .Fast2 has a key role in mesoderm formation andcranio-caudal patterning during gastrulation, andmany mutants do not form a heart at all. However,those that do fail to express markers of workingmyocardium, and the same is true of cardiomyocytesthat are derived by in vitro differentiation of homozygous-mutantembryonic stem cells (I. von Both andJ. Wrana, personal communication).An important repressive role for the T-box factorTbx2 in establishing the expression domains of Nppa inthe working myocardium has recently been shown 129 .Tbx2 is expressed in the myocardium of the formingheart in an evolving pattern that is always reciprocal tothat of Nppa. Furthermore, Tbx2 can form a repressivecomplex with Nkx2-5 on the proximal Nppa promoter,which competes with a Tbx5–Nkx2-5 complex thatpositively regulates Nppa expression. So, integration ofthe positive and negative modalities of the Nkx2-5 andTbx pathways helps to define the working myocardiumin the early heart. How signalling through Fast2 integrateswith these pathways is yet to be explored.The left–right frontierOne of the most vexing aspects of heart biology concernsthe contribution of the embryonic left–right(L/R) axial system to heart patterning and morphogenesis.This system specifies the lateral asymmetriesof the body and its organs. Its precise contributions toheart morphogenesis are poorly characterized; however,on the basis of outcomes of disturbed laterality inhumans and animal models, we can clearly say that thepathway determines L/R differences in the morphologyof the atria and their venous tributaries, and, morespeculatively, the direction and quality of ventricularbending. A spectrum of bizarre abnormalities canoccur if laterality is defective. These include mirrorimagereversals of body asymmetry (situs inversus)and isomerisms, in which both the left and right partsof organs such as the heart and lungs develop withentirely left or right characteristics 130,131 . In the heart,only the atrial appendages (BOX 1) can be truly isomerized,as they derive from left and right cardiac primordia,respectively 132 . By contrast, the left (systemic) andright (pulmonary) ventricles arise in development in acranio-caudal arrangement, and receive contributionsfrom both left and right cardiac progenitor populations132 . The act of looping itself brings the ventriclesinto a L/R configuration. These fundamental differencesin L/R development of the atria and ventriclesare explored further below.552 | JULY 2002 | VOLUME 3 www.nature.com/reviews/genetics© 2002 Nature Publishing Group