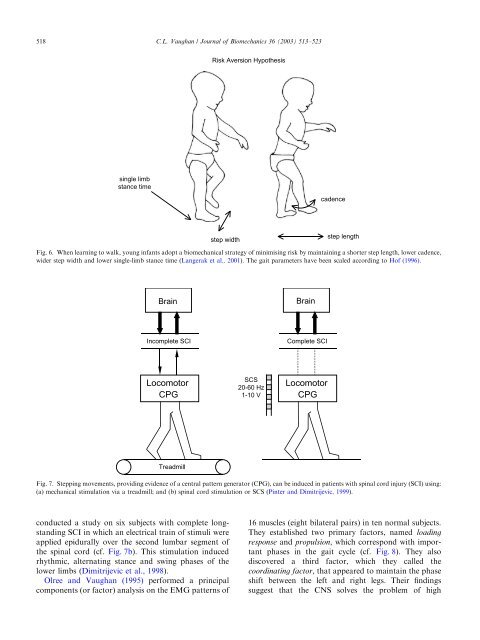
518C.L. Vaugh<strong>an</strong> / Journal <strong>of</strong> Biomech<strong>an</strong>ics 36 (2003) 513–523Risk Aversion Hypothesissingle limbst<strong>an</strong>ce timecadencestep widthstep lengthFig. 6. When learning to walk, young inf<strong>an</strong>ts adopt a biomech<strong>an</strong>ical strategy <strong>of</strong> minimising risk by maintaining a shorter step length, lower cadence,wider step width <strong>an</strong>d lower single-limb st<strong>an</strong>ce time (L<strong>an</strong>gerak et al., 2001). The gait parameters have been scaled according to H<strong>of</strong> (1996).BrainBrainIncomplete SCIComplete SCILocomotorCPGSCS20-60 Hz1-10 VLocomotorCPGTreadmillFig. 7. Stepping movements, providing evidence <strong>of</strong> a central pattern generator (CPG), c<strong>an</strong> be induced in patients with spinal cord injury (SCI) using:(a) mech<strong>an</strong>ical stimulation via a treadmill; <strong>an</strong>d (b) spinal cord stimulation or SCS (Pinter <strong>an</strong>d Dimitrijevic, 1999).conducted a study on six subjects with complete longst<strong>an</strong>dingSCI in which <strong>an</strong> electrical train <strong>of</strong> stimuli wereapplied epidurally over the second lumbar segment <strong>of</strong>the spinal cord (cf. Fig. 7b). This stimulation inducedrhythmic, alternating st<strong>an</strong>ce <strong>an</strong>d swing phases <strong>of</strong> thelower limbs (Dimitrijevic et al., 1998).Olree <strong>an</strong>d Vaugh<strong>an</strong> (1995) performed a principalcomponents (or factor) <strong>an</strong>alysis on the EMG patterns <strong>of</strong>16 muscles (eight bilateral pairs) in ten normal subjects.They established two primary factors, named loadingresponse <strong>an</strong>d propulsion, which correspond with import<strong>an</strong>tphases in the gait cycle (cf. Fig. 8). They alsodiscovered a third factor, which they called thecoordinating factor, that appeared to maintain the phaseshift between the left <strong>an</strong>d right legs. Their findingssuggest that the CNSsolves the problem <strong>of</strong> high
C.L. Vaugh<strong>an</strong> / Journal <strong>of</strong> Biomech<strong>an</strong>ics 36 (2003) 513–523 519Factor Score3210-1-2dimensionality by generating just a few fundamentalsignals which control all the major muscle groups inboth legs. There have been other recent studies utilisingEMG in both inf<strong>an</strong>ts <strong>an</strong>d adults that have providedsupport for the hypothesis that a single locomotor CPGcontrols both forward <strong>an</strong>d backward <strong>walking</strong> (Lamb<strong>an</strong>d Y<strong>an</strong>g, 2000; v<strong>an</strong> Deursen et al., 1998).6. Linking control <strong>an</strong>d effectPropulsion FactorFactor 3Factor 40 20 40 60 80 100% Gait CycleFig. 8. Eigenvectors, or factor <strong>an</strong>alysis scores, plotted as a function <strong>of</strong>the gait cycle (Olree <strong>an</strong>d Vaugh<strong>an</strong>, 1995). Factors 3 <strong>an</strong>d 4 are thepropulsion factors <strong>an</strong>d correspond closely to the electromyographicsignals <strong>of</strong> the left <strong>an</strong>d right gastrocnemius muscles, respectively.Bipedal locomotion is generated through global entrainment<strong>of</strong> the neural system on the one h<strong>an</strong>d, <strong>an</strong>d themusculoskeletal system plus environment on the otherCharles Scott Sherrington, who won a Nobel Prize forhis contributions to neuroscience in 1932, said ‘‘Swiftlythe brain becomes <strong>an</strong> ench<strong>an</strong>ted loom, where millions <strong>of</strong>flashing shuttles weave a dissolving pattern, always ame<strong>an</strong>ingful pattern though never <strong>an</strong> abiding one; ashifting harmony <strong>of</strong> sub-patterns’’ (Sherrington, 1941).This <strong>an</strong>alogy between the functioning <strong>of</strong> the brain <strong>an</strong>d amachine was a mathematical construct described almost60 years ago by McCulloch <strong>an</strong>d Pitts (1943). Theypioneered the field <strong>of</strong> artificial neural networks (ANNs)<strong>an</strong>d, although there was some activity in the 1960s(Rosenblatt, 1961; Minsky <strong>an</strong>d Papert, 1969), ANNsonly became widely accepted as <strong>an</strong> area <strong>of</strong> seriousresearch endeavour during the 1980s with the efforts <strong>of</strong>Hopfield (1982), Anderson (1983), Rumelhart et al.(1986) <strong>an</strong>d others. As the name implies, <strong>an</strong> ANN is agroup <strong>of</strong> m<strong>an</strong>y neurons (or processing elements) that areinterconnected <strong>an</strong>d distributed in layers. An ANNnormally operates in two states: learning, where aprocess <strong>of</strong> ch<strong>an</strong>ges in the synaptic weights occurs; <strong>an</strong>drecall, where <strong>an</strong> input stimulus generates <strong>an</strong> outputsignal (Rumelhart et al., 1986).While ANNs were originally utilised in patternrecognition problems, their application to the study <strong>of</strong>real biological systems beg<strong>an</strong> to appear in the early1990s (Srinivas<strong>an</strong> et al., 1992. Sepulveda et al., 1993).The argument had thus come full circle: ANNs, inspiredby the example <strong>of</strong> the central nervous system, now hadthe potential to help elucidate how the CNSitselffunctioned (Vaugh<strong>an</strong>, 1997). Sepulveda et al. (1993)built <strong>an</strong> ANN in which 16 EMG signals were used as theinput signals <strong>an</strong>d three joint torques were generated asoutput. Once the network had been successfully trained,a series <strong>of</strong> simulations were conducted. A 30% reductionin the magnitude <strong>of</strong> the soleus EMG led to a signific<strong>an</strong>treduction in the pl<strong>an</strong>tar flexor moment at the <strong>an</strong>kle joint(cf. Fig. 9), a prediction that is consistent with ourunderst<strong>an</strong>ding <strong>of</strong> <strong>an</strong>kle biomech<strong>an</strong>ics. Recognising thepotential <strong>of</strong> ANNs to control muscle activation patterns,Popovic et al. (1993) developed a network whichthey tested on chronic spinalised cats. Other researchershave built on this basic science platform <strong>an</strong>d have usedANN models <strong>an</strong>d functional electrical stimulation torehabilitate the gait <strong>of</strong> hum<strong>an</strong> subjects (Graupe <strong>an</strong>dKordylewski, 1995; Sepulveda <strong>an</strong>d Cliquet, 1995).While the aforementioned models linked the controller(neural system) <strong>an</strong>d the effector (mech<strong>an</strong>icalsystem), they lacked true integration with feedbackinfluencing the real-time perform<strong>an</strong>ce <strong>of</strong> the model(Vaugh<strong>an</strong>, 1997). One <strong>of</strong> the most intriguing models <strong>of</strong><strong>bipedal</strong> <strong>walking</strong> is that developed by Taga et al. (1991).A pair <strong>of</strong> CPGs, modelled by <strong>an</strong> ANN, controlled themuscles <strong>of</strong> the trunk plus the left <strong>an</strong>d right hip, knee <strong>an</strong>d<strong>an</strong>kle joints. Their biped consisted <strong>of</strong> eight segments, 10degrees <strong>of</strong> freedom <strong>an</strong>d 19 muscle actuators. A series <strong>of</strong>Fig. 9. An artificial neural network, designed to link 16 electromyographicsignals <strong>an</strong>d three joint moments, predicts the effect <strong>of</strong> a 30%reduction in the soleus activity (cf. dashed line), showing a signific<strong>an</strong>tdecrease in the pl<strong>an</strong>tar flexor moment at the <strong>an</strong>kle during the push-<strong>of</strong>fphase (Sepulveda et al., 1993).