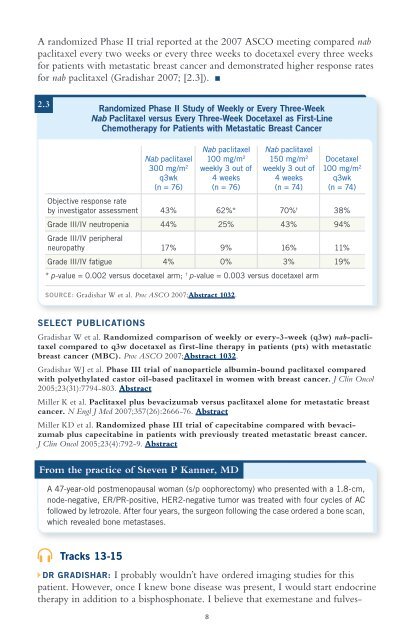
A randomized Phase II trial reported at the 2007 ASCO meeting compared nabpaclitaxel every two weeks or every three weeks to docetaxel every three weeksfor patients with metastatic breast cancer and demonstrated higher response ratesfor nab paclitaxel (Gradishar 2007; [2.3]).2.3Randomized Phase II Study of Weekly or Every Three-WeekNab Paclitaxel versus Every Three-Week Docetaxel as First-LineChemotherapy for Patients with Metastatic Breast CancerNab paclitaxel Nab paclitaxelNab paclitaxel 100 mg/m 2 150 mg/m 2 Docetaxel300 mg/m 2 weekly 3 out of weekly 3 out of 100 mg/m 2q3wk 4 weeks 4 weeks q3wk(n = 76) (n = 76) (n = 74) (n = 74)Objective response rateby investigator assessment 43% 62%* 70% † 38%Grade III/IV neutropenia 44% 25% 43% 94%Grade III/IV peripheralneuropathy 17% 9% 16% 11%Grade III/IV fatigue 4% 0% 3% 19%* p-value = 0.002 versus docetaxel arm; † p-value = 0.003 versus docetaxel armSOURCE: Gradishar W et al. Proc ASCO 2007;Abstract 1032.SELECT PUBLICATIONSGradishar W et al. Randomized comparison of weekly or every-3-week (q3w) nab-paclitaxelcompared to q3w docetaxel as first-line therapy in patients (pts) with metastaticbreast cancer (MBC). Proc ASCO 2007;Abstract 1032.Gradishar WJ et al. Phase III trial of nanoparticle albumin-bound paclitaxel comparedwith polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol2005;23(31):7794-803. AbstractMiller K et al. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breastcancer. N Engl J Med 2007;357(26):2666-76. AbstractMiller KD et al. Randomized phase III trial of capecitabine compared with bevacizumabplus capecitabine in patients with previously treated metastatic breast cancer.J Clin Oncol 2005;23(4):792-9. AbstractFrom the practice of Steven P Kanner, MDA 47-year-old postmenopausal woman (s/p oophorectomy) who presented with a 1.8-cm,node-negative, ER/PR-positive, HER2-negative tumor was treated with four cycles of ACfollowed by letrozole. After four years, the surgeon following the case ordered a bone scan,which revealed bone metastases.Tracks 13-15DR GRADISHAR: I probably wouldn’t have ordered imaging studies for thispatient. However, once I knew bone disease was present, I would start endocrinetherapy in addition to a bisphosphonate. I believe that exemestane and fulves-8
trant are equivalent choices, and a case could be made for tamoxifen as well.Biopsies of bone-only metastases can be problematic because a negative resultcan be caused by a sampling error. So one can make a case for treating empiricallywith endocrine therapy. However, if the patient developed any visceral orsoft tissue disease that could be biopsied easily, I would do so.In cases such as this one treatment is palliative, not curative, so the goals ofselecting an endocrine therapy include delaying the time until chemotherapy isneeded and maintaining quality of life. In managing metastatic disease, we don’thave to see the lesions disappear. Stable disease is an acceptable endpoint, and ithas an outcome that’s equivalent to an objective response.With bone-only disease, evidence supports continuing endocrine therapy as longas you don’t observe disease progression. If the bone scan remains stable, that’sacceptable. I would use endocrine therapy to the point of exhaustion, as long asthe patient doesn’t develop a visceral crisis or another problem that motivates usto use chemotherapy.We’ve spent 20 years trying to figure out which endocrine agent or sequence isbest in these cases. The bottom line is that it probably doesn’t make a big difference.You can switch between classes of endocrine therapy, or within the classof aromatase inhibitors you can switch between a steroidal and a nonsteroidalaromatase inhibitor. In the EFECT study, fulvestrant was compared to exemestaneamong patients whose disease progressed on a nonsteroidal aromataseinhibitor, and the outcomes were identical regarding side effects, clinical benefitrate, objective response rate and time to disease progression.Additional Cases Discussing Metastatic Disease on the Audio ProgramA 49-year-old woman with biopsy-confirmed liver and lung metastases threeyears after adjuvant AC for a triple-negative IDC (from the practice ofMichael A Schwartz, MD)A 47-year-old woman who developed multiple unresectable brain metastases eightyears after adjuvant anthracycline/taxane therapy for an ER/PR-negative, HER2-positive IDC (from the practice of Kenneth R Hoffman, MD, MPH)A 62-year-old woman with a neglected 6-cm, ER/PR-positive, HER2-positive IDCand biopsy-proven lung metastases (from the practice of Dr Hoffman)SELECT PUBLICATIONSChia S et al. Double-blind, randomized placebo controlled trial of fulvestrant comparedwith exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausalwomen with hormone receptor-positive, advanced breast cancer: Results fromEFECT. J Clin Oncol 2008;26(10):1664-70. AbstractCosta L et al. Role of bisphosphonates for the management of skeletal complicationsand bone pain from skeletal metastases. Support Cancer Ther 2006;3(3):143-53. AbstractLyseng-Williamson KA. Zoledronic acid: A review of its use in breast cancer. Drugs2008;68(18):2661-82. AbstractPecherstorfer M. Treatment options for breast cancer and bone metastases. Womens Health(Lond Engl) 2009;5(2):149-63. Abstract9