DOUBLE REPLACEMENT PRACTICE REACTIONS example
DOUBLE REPLACEMENT PRACTICE REACTIONS - Library
DOUBLE REPLACEMENT PRACTICE REACTIONS - Library
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
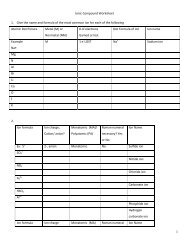
2. Watch out for diatomic molecules. These elements only exist in compounds or asdiatomic molecules.H 2 , N 2 , O 2 , F 2 , Cl 2 , Br 2 , I 2EXAMPLES1. Mg + Zn(NO3)2 ----> Mg(NO3)2 + ZnMg replaces Zn;Mg + 2 AgNO3 -----> Mg(NO3)2 + 2 AgMg replaces Ag;2. Cl2+ 2NaBr ---> 2NaCl + Br2Cl replaces Br. Note that chlorine and bromine are diatomic moleculesFor each reaction predict the products and balance the equation. State thereaction in chemical formulas and in symbols. For <strong>example</strong>:Mg + 2 AgNO3 ----->Magnesium + silver nitrate magnesium nitrate + silverMg + 2 AgNO 3 -----> Mg(NO 3 ) 2 + 2 Ag1. Ag + KNO 3 ----->2. Zn + AgNO 3 ----->3. Al + H 2 SO 4 ----->4. Cl 2 + KI ----->5. Li + H 2 O ----->6. Cu + FeSO 4 ----->Replacement Worksheet-Chem 1214