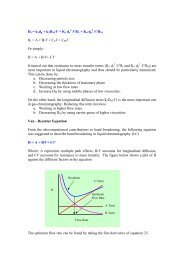
1 Chapter 14 review For the overall hypothetical reaction A + 5B ...
1 Chapter 14 review For the overall hypothetical reaction A + 5B ...
1 Chapter 14 review For the overall hypothetical reaction A + 5B ...
- No tags were found...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Chapter</strong> <strong>14</strong> <strong>review</strong><br />
1<br />
<strong>For</strong> <strong>the</strong> <strong>overall</strong> hypo<strong>the</strong>tical <strong>reaction</strong> A + <strong>5B</strong> → 4C, <strong>the</strong> rate of appearance of C given<br />
by Δ[C]/Δt is <strong>the</strong> same as<br />
A) Δ[A]/Δt<br />
B) -(5/4)(Δ[B]/Δt)<br />
C) -(4/5)(Δ[B]/Δt)<br />
D) -(1/4)(Δ[A]/Δt)<br />
E) none of <strong>the</strong> above.<br />
2<br />
When heated, <strong>the</strong> compound RX 3 decomposes to a mixture of products. The following<br />
data were collected for <strong>the</strong> decomposition at 100º C. What is <strong>the</strong> average rate of<br />
<strong>reaction</strong>, -Δ [RX 3 ]/Δt, over <strong>the</strong> entire experiment?<br />
3<br />
4<br />
A) 0.0028 mol/L . s<br />
B) 0.045 mol/L . s<br />
C) 0.00<strong>14</strong> mol/L . s<br />
D) -0.0027 mol/L . s<br />
E) 0.0057 mol/L . s<br />
The initial rate of <strong>the</strong> <strong>reaction</strong> PCl 5 → PCl 3 + Cl 2 is increased a factor of four when<br />
<strong>the</strong> concentration of PCl 5 is doubled. Therefore, <strong>the</strong> rate<br />
A) depends on <strong>the</strong> concentrations of PCl 3 and Cl 2 .<br />
B) is first order with respect to PCl 5 .<br />
C) is second order with respect to PCl 5 .<br />
D) is fourth order with respect to PCl 5 .<br />
E) is first order with respect to PCl 3 .<br />
Bromine atoms react with iso-butane (iso-C 4 H 10 ) to form hydrogen bromide and <strong>the</strong> t-<br />
butyl radical (t-C 4 H 9 ): Br + iso-C 4 H 10 → HBr + t-C 4 H 9 . The <strong>reaction</strong> is first order<br />
with respect to each of <strong>the</strong> reactants, and at 298 K, <strong>the</strong> rate constant is 1.02 x 10 6 L<br />
mol -1 s -1 . Calculate <strong>the</strong> initial rate of <strong>the</strong> <strong>reaction</strong> in a 4.0 L <strong>reaction</strong> vessel containing<br />
0.60 moles of iso-butane and 0.00010 moles of bromine atoms.<br />
A) 1.5 mol L -1 s -1<br />
B) 3.8 mol L -1 s -1<br />
C) 2.5 x 10 11 mol L -1 s -1<br />
D) 2.5 x 10 2 mol L -1 s -1<br />
E) 6.7 x 10 2 mol L -1 s -1<br />
1
5<br />
6<br />
7<br />
8<br />
9<br />
Consider <strong>the</strong> <strong>the</strong>rmal decomposition of cyclobutane (C 4 H 8 (g)) at 438º C, C 4 H 8 (g) →<br />
2C 2 H 4 (g). The <strong>reaction</strong> follows first-order kinetics and <strong>the</strong> rate constant is k = 2.48 x<br />
10 -4 s -1 . If <strong>the</strong> initial concentration of cyclobutane is 0.800 mol/L, what concentration<br />
will remain after 10.0 min?<br />
A) 0.689 mol/L<br />
B) 0.455 mol/L<br />
C) 0.333 mol/L<br />
D) 0.248 mol/L<br />
E) 0.0061 mol/L<br />
A certain first-order <strong>reaction</strong> is 46 % complete in 68 min at 25°C. What is its rate<br />
constant?<br />
A) 9.1 x 10 -3 min -1<br />
B) 1.1 x 10 -2 min -1<br />
C) 31 min -1<br />
D) 51 min -1<br />
E) none of <strong>the</strong> above<br />
Consider <strong>the</strong> <strong>reaction</strong> A → products. Which, if any, of <strong>the</strong> following plots is<br />
consistent with a second-order <strong>reaction</strong>?<br />
A) ln[A] plotted against time gives a straight line of positive slope.<br />
B) ln[A] plotted against time gives a straight line of negative slope.<br />
C) 1/[A] plotted against time gives a straight line of positive slope.<br />
D) 1/[A] plotted against time gives a straight line of negative slope.<br />
E) None of <strong>the</strong> above plots is consistent with a second-order <strong>reaction</strong>.<br />
Consider <strong>the</strong> <strong>reaction</strong> A → products. Which of <strong>the</strong> following plots is consistent with a<br />
zero-order <strong>reaction</strong>?<br />
A) [A] plotted against time gives a horizontal, straight line.<br />
B) ln[A] plotted against time gives a straight line of negative slope.<br />
C) 1/[A] plotted against time gives a straight line of positive slope.<br />
D) [A] plotted against time gives a straight line of negative slope.<br />
E) [A] plotted against time gives a curved line of negative slope, decreasing in<br />
magnitude as time increases.<br />
What is <strong>the</strong> value of <strong>the</strong> rate constant for a first-order <strong>reaction</strong> for which <strong>the</strong> half-life<br />
is 26.7 min?<br />
A) 18.5 min -1<br />
B) 38.5 min -1<br />
C) 9.25 min -1<br />
D) 19.3 min -1<br />
E) 0.0260 min -1<br />
2
10<br />
The rate constant of a first-order <strong>reaction</strong> is 3.68 x 10 -2 s -1 at 150°C, and <strong>the</strong> activation<br />
energy is 71 kJ/mol. What is <strong>the</strong> value of <strong>the</strong> rate constant at 170°C?<br />
A) 9.2 x 10 -2 s -1<br />
B) 3.7 x 10 -2 s -1<br />
C) 2.49 s -1<br />
D) 4.0 x 10 -2 s -1<br />
E) none of <strong>the</strong> above<br />
11<br />
In order to obtain <strong>the</strong> activation energy of a <strong>reaction</strong> using a graphical method,<br />
__________ is plotted against _________, giving a straight line whose slope is equal<br />
to __________.<br />
A) k; T; -E a<br />
B) k; 1/T; -E a<br />
C) lnk; T; -E a /R<br />
D) k; 1/T; -E a /R<br />
E) lnk; 1/T; -E a /R<br />
12<br />
Select <strong>the</strong> appropriate rate law for <strong>the</strong> elementary process shown below.<br />
2A → B + C<br />
A) Rate = k[2A]<br />
B) Rate = k[A]<br />
C) Rate = k[A] 2<br />
D) Rate = k[A] 1/2<br />
E) Rate = 2k[A]<br />
13<br />
The <strong>reaction</strong><br />
3ClO - (aq) → ClO 3 - (aq)<br />
+ 2Cl - (aq) has been proposed to occur by <strong>the</strong> following mechanism.<br />
ClO - (aq) + ClO - ( aq)→ ClO 2 - ( aq) + Cl - ( aq) (slow)<br />
ClO 2 - (aq) + ClO - (aq) → ClO 3 - (aq) + Cl - (aq) (fast<br />
Which rate law is consistent with this mechanism?<br />
A) rate = k[ClO - ]<br />
B) rate = k[ClO - ] 3<br />
C) rate = k[ClO 2 - ][ClO - ]<br />
D) rate = k[ClO - ] 2<br />
E) rate = k[Cl - ][ClO - ] 2<br />
<strong>14</strong><br />
Consider <strong>the</strong> <strong>reaction</strong>:<br />
2NO(g) + 2H 2 (g) → N 2 (g) + 2H 2 O(g)<br />
A suggested mechanism for this <strong>reaction</strong> follows:<br />
(1) NO(g) + NO(g) → N 2 O 4 (g) (slow)<br />
(2) N 2 O 4 (g) + H 2 (g) → N 2 (g) + H 2 O 2 (g) (fast)<br />
(3) H 2 O 2 (g) + H 2 (g) → 2H 2 O(g) (fast)<br />
Based on this mechanism, which, if any, of <strong>the</strong> following actions will not affect <strong>the</strong><br />
rate of <strong>the</strong> <strong>reaction</strong>?<br />
3
A) adding a catalyst<br />
B) adding more NO(g)<br />
C) adding more H 2 (g)<br />
D) increasing <strong>the</strong> temperature<br />
E) All of <strong>the</strong>se will affect <strong>the</strong> rate of <strong>the</strong> <strong>reaction</strong>.<br />
15<br />
A catalyst speeds up a <strong>reaction</strong> by<br />
A) increasing <strong>the</strong> number of high-energy molecules.<br />
B) increasing <strong>the</strong> temperature of <strong>the</strong> molecules in <strong>the</strong> <strong>reaction</strong>.<br />
C) increasing <strong>the</strong> number of collisions between molecules.<br />
D) increasing <strong>the</strong> activation energy for <strong>the</strong> <strong>reaction</strong>.<br />
E) providing a new <strong>reaction</strong> pathway for molecules.<br />
16<br />
Consider <strong>the</strong> <strong>reaction</strong>:<br />
CH 3 Cl(aq) + OH - (aq) → CH 3 OH(aq) + Cl - (aq)<br />
If <strong>the</strong> rate of appearance of CH 3 OH(aq) remains constant at 1.0 x 10 -4 mol/(L·min),<br />
and <strong>the</strong> initial concentrations of both reactants are 0.10 mol/L, how long will it take to<br />
consume half of <strong>the</strong> reactants?<br />
A) 10,000 min<br />
B) 1,000 min<br />
C) 500 min<br />
D) 100 min<br />
E) none of <strong>the</strong> above<br />
17<br />
Consider <strong>the</strong> <strong>reaction</strong><br />
2A + 2B + C → 2D + E<br />
If <strong>the</strong> rate law for this <strong>reaction</strong> is Rate = k[A][B] 2 , what will be <strong>the</strong> effect on <strong>the</strong> rate if<br />
<strong>the</strong> concentrations of A, B and C are all doubled at <strong>the</strong> same time?<br />
A) The rate will increase by a factor of 2.<br />
B) The rate will increase by a factor of 4.<br />
C) The rate will increase by a factor of 6.<br />
D) The rate will increase by a factor of 8.<br />
E) More information is needed before this question can be answered.<br />
18<br />
Nitric oxide (NO) reacts with hydrogen (H2 ) according to <strong>the</strong> equation:<br />
2NO(g) + 2H 2 (g) → N 2 (g) + 2H 2 O(g)<br />
The following initial rates of <strong>reaction</strong> have been measured for <strong>the</strong> given reactant<br />
concentrations.<br />
Which of <strong>the</strong> following is <strong>the</strong> rate law (rate equation) for this <strong>reaction</strong>?<br />
A) rate = k[NO] 2 [H 2 ]<br />
4
B) rate = k[NO] [H 2 ] 2<br />
C) rate = k[NO] [H 2 ] 4<br />
D) rate = k[NO] [H 2 ]<br />
E) rate = k[NO] 1/2 [H 2 ] 1/4<br />
19<br />
The following initial rate data were collected for <strong>the</strong> <strong>reaction</strong>:<br />
2A + B → C + D<br />
What is <strong>the</strong> value of <strong>the</strong> rate constant?<br />
A) k = 1.42 x 10 -2 s -1<br />
B) k = 2.83 x 10 -3 L mol -1 s -1<br />
C) k = 1.42 L 2 mol -2 s -1<br />
D) k = 0.532 x 10 -3 L 2 mol -2 s -1<br />
E) k = 4.26 x 10 -1 L mol -1 s -1<br />
20<br />
A solution of sucrose in water has an initial concentration of 0.155 mol/L. The<br />
sucrose is being converted to o<strong>the</strong>r sugars in a <strong>reaction</strong> which is first-order with<br />
respect to <strong>the</strong> sucrose concentration. Exactly 30 minutes later, <strong>the</strong> sucrose<br />
concentration has fallen to 0.131 mol/L. Calculate <strong>the</strong> value of <strong>the</strong> rate constant in s -1 .<br />
A) 9.3 x 10 -5 s -1<br />
B) 4.1 x 10 -5 s -1<br />
C) 5.6 x 10 -3 s -1<br />
D) 1.7 x 10 -1 s -1<br />
E) none of <strong>the</strong> above<br />
21<br />
The <strong>reaction</strong> A → products is first-order with respect to A. Which of <strong>the</strong> following<br />
correctly describes a plot of rate data for this <strong>reaction</strong>?<br />
A) [A] plotted vs time gives a straight line of negative slope.<br />
B) [A] plotted vs time gives a straight line of positive slope.<br />
C) ln[A] plotted vs time gives a straight line of positive slope.<br />
D) ln[A] plotted vs time gives a straight line of negative slope.<br />
E) 1/[A] plotted vs time gives a straight line of positive slope.<br />
22<br />
Tetrafluoroe<strong>the</strong>ne (C2 F 4 ) dimerizes at 300º C to form octafluorocyclobutane (cyclo-<br />
C 4 F 8 ) according to <strong>the</strong> equation<br />
2C 2 F 4 (g) → cyclo-C 4 F 8 (g)<br />
The rate law for <strong>the</strong> <strong>reaction</strong> is:<br />
Rate = (0.080 L mol -1 min -1 )[C 2 F 4 ] 2<br />
In an experiment where <strong>the</strong> initial concentration of tetrafluoroethylene is 0.10 mol/L,<br />
what concentration remains after 700 min?<br />
5
A) 8.0 X 10 -3 mol/L<br />
B) 8.0 X 10 -4 mol/L<br />
C) 2.8 X 10 -3 mol/L<br />
D) 0.075 mol/L<br />
E) 0.015 mol/L<br />
23<br />
<strong>For</strong> <strong>the</strong> <strong>reaction</strong> A → products, <strong>the</strong> rate law is Rate = k[A] 2 . Starting with [A] = 0.100<br />
mol/L, its concentration is found to be 0.0652 mol/L after 600. seconds. What is <strong>the</strong><br />
value of <strong>the</strong> rate constant?<br />
A) 5.8 x 10 -5 L . mol -1 . s -1<br />
B) 7.1 x 10 -4 L . mol -1 . s -1<br />
C) 3.2 x 10 -3 L . mol -1 . s -1<br />
D) 8.9 x 10 -3 L . mol -1 . s -1<br />
E) none of <strong>the</strong> above<br />
24<br />
In an enzymatic fermentation, <strong>the</strong> initial concentration of sugar is 0.16 mol/L; after<br />
10.0 hours <strong>the</strong> concentration is 0.080 mol/L; after 20.0 hours <strong>the</strong> concentration is<br />
0.040 mol/L. What is <strong>the</strong> order of this <strong>reaction</strong>, and what is <strong>the</strong> rate constant?<br />
A) zero order; 1.67 x 10 -6 s -1<br />
B) first order; 1.93 x 10 -5 s -1<br />
C) first order; 1.67 x 10 -6 s -1<br />
D) second order; 3.71 x 10 -10 s -1<br />
E) second order; 1.67 x 10 -6 s -1<br />
25<br />
From <strong>the</strong> following data, estimate <strong>the</strong> activation energy for <strong>the</strong> gas-phase <strong>reaction</strong><br />
H 2 + I 2 → 2HI<br />
A) 21.0 kJ/mol<br />
B) 2.10 x 10 4 kJ/mol<br />
C) 1.75 x 10 2 kJ/mol<br />
D) 9.00 x 10 2 kJ/mol<br />
E) none of <strong>the</strong> above<br />
26<br />
A second-order rate constant is given by<br />
k = (6.5 x 10 7 L·mol -1·s -1 -13500 K/T<br />
)e<br />
What is <strong>the</strong> <strong>the</strong> activation energy, E a , for this <strong>reaction</strong>?<br />
A) 135 kJ/mol<br />
B) 56 kJ/mol<br />
C) 112 kJ/mol<br />
D) 162 kJ/mol<br />
E) 782 kJ/mol<br />
6
27<br />
The <strong>reaction</strong> of ethylene (C2 H 4 ) with butadiene (C 4 H 6 ) to form cyclohexene (C 6 H 10 )<br />
has an activation energy (E a ) of 115 kJ/mol. The reverse <strong>reaction</strong> (decomposition of<br />
cyclohexene to ethylene and butadiene) has an activation energy of 287 kJ/mol. What<br />
is <strong>the</strong> heat of <strong>reaction</strong>, Δ H rxn , for <strong>the</strong> forward <strong>reaction</strong>?<br />
A) +115 kJ/mol<br />
B) +287 kJ/mol<br />
C) -287 kJ/mol<br />
D) +172 kJ/mol<br />
E) -172 kJ/mol<br />
28<br />
Consider <strong>the</strong> following mechanism for <strong>the</strong> oxidation of bromide ions by hydrogen<br />
peroxide in aqueous acid solution.<br />
(1) H + + H 2 O 2 → H 3 O 2<br />
+<br />
(rapid equilibrium)<br />
(2) H 3 O 2 + + Br - → HOBr + H 2 O (slow)<br />
(3) HOBr + H + + Br - → Br 2 + H 2 O (fast)<br />
What is <strong>the</strong> <strong>overall</strong> <strong>reaction</strong> equation for this process?<br />
A) 2H + + H 2 O 2 + Br - + HOBr → H 3 O 2<br />
+<br />
+ Br 2 + H 2 O<br />
B) H 3 O 2<br />
+<br />
+ H + + 2Br - → Br 2 + 2H 2 O<br />
C) 2H + + 2Br - + H 2 O 2 → Br 2 + 2H 2 O<br />
D) 2H 3 O 2<br />
+<br />
+ 2Br - → H 2 O 2 + Br 2 + 2H 2 O<br />
E) none of <strong>the</strong> above<br />
29<br />
The mechanism for <strong>the</strong> gas-phase <strong>reaction</strong><br />
2NO + Cl 2 → 2NOCl<br />
is suggested to be:<br />
(1) NO + NO → N 2 O 4 (slow)<br />
(2) N 2 O 4 + Cl 2 → 2NOCl (fast)<br />
Based on this mechanism, <strong>the</strong> rate law for <strong>the</strong> <strong>overall</strong> <strong>reaction</strong> is:<br />
A) Rate = k [NO]<br />
B) Rate = k [NO] 2<br />
C) Rate = k [NO 2 ] 2 [Cl 2 ]<br />
D) Rate = k [Cl 2 ]<br />
E) Rate = k [NO 2 ] 2 [Cl 2 ] 2<br />
30<br />
At elevated temperatures in <strong>the</strong> gas phase, cyclopropane reacts to form propene. The<br />
rate of <strong>reaction</strong> is given by:<br />
rate = Δ[propene]/Δt = k[cyclopropane].<br />
Which one of <strong>the</strong> following actions is least likely to cause a change in <strong>the</strong> rate of this<br />
<strong>reaction</strong>?<br />
A) adding a catalyst<br />
B) raising <strong>the</strong> temperature<br />
C) doubling <strong>the</strong> initial amount of cyclopropane<br />
D) halving <strong>the</strong> volume of <strong>the</strong> <strong>reaction</strong> vessel, but keeping <strong>the</strong> initial amount of<br />
cyclopropane constant.<br />
E) continuously removing propene as it is formed<br />
7