FDA AERS Mefloquine, Judicial Watch FOIA report~20120403
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
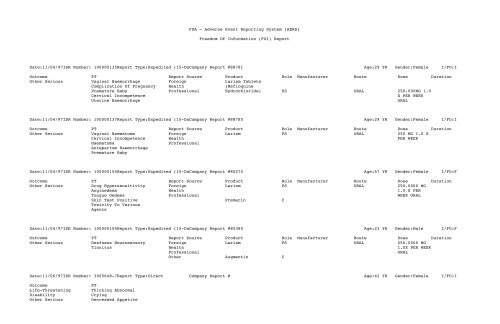
Date:11/04/97ISR Number: 100000135Report Type:Expedited (15-DaCompany Report #88781 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vaginal Haemorrhage Foreign Lariam Tablets<br />
Complication Of Pregnancy Health (<strong>Mefloquine</strong><br />
Premature Baby Professional Hydrochloride) PS ORAL 250.000MG 1.0<br />
Cervical Incompetence<br />
X PER WEEK<br />
Uterine Haemorrhage<br />
ORAL<br />
Date:11/04/97ISR Number: 100000137Report Type:Expedited (15-DaCompany Report #88780 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vaginal Haematoma Foreign Lariam PS ORAL 250 MG 1.0 X<br />
Cervical Incompetence Health PER WEEK<br />
Haematoma<br />
Professional<br />
Antepartum Haemorrhage<br />
Premature Baby<br />
Date:11/04/97ISR Number: 100000155Report Type:Expedited (15-DaCompany Report #80275 Age:57 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Hypersensitivity Foreign Lariam PS ORAL 250.0000 MG<br />
Angioedema Health 1.0 X PER<br />
Tongue Oedema Professional WEEK ORAL<br />
Skin Test Positive Premarin C<br />
Toxicity To Various<br />
Agents<br />
Date:11/04/97ISR Number: 100000155Report Type:Expedited (15-DaCompany Report #85380 Age:23 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Neurosensory Foreign Lariam PS ORAL 250.0000 MG<br />
Tinnitus Health 1.0X PER WEEK<br />
Professional<br />
ORAL<br />
Other Augmentin C<br />
Date:11/06/97ISR Number: 3005648-7Report Type:Direct Company Report # Age:42 YR Gender:Female I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Disability<br />
Other Serious<br />
PT<br />
Thinking Abnormal<br />
Crying<br />
Decreased Appetite
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Panic Disorder<br />
Insomnia Related To<br />
Another Mental Condition<br />
Anxiety<br />
Personality Change<br />
Fear<br />
Insomnia<br />
Paraesthesia<br />
03-Apr-2012 09:37 AM Page: 1
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dysarthria<br />
Confusional State<br />
Suicidal Ideation Report Source Product Role Manufacturer Route Dose Duration<br />
Asthenia Lariam PS Roche ORAL TABS; 250 MG<br />
Depression<br />
TAB PO ONCE<br />
Chills<br />
WEEKLY;(PT<br />
Abnormal Dreams<br />
D'CD DRUG)<br />
Date:11/10/97ISR Number: 3000073-7Report Type:Expedited (15-DaCompany Report #86849 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cough Foreign Lariam PS ORAL 250.000 MG<br />
Initial or Prolonged Dyspnoea Other 1.0 X PER<br />
Asthenia<br />
WEEK ORAL<br />
Bronchitis Dpt C<br />
Dizziness<br />
Yellow Fever Vaccine C<br />
Red Blood Cell Havrix C<br />
Sedimentation Rate Typhim C<br />
Increased Mercalm C<br />
Vomiting Genhevac B C<br />
Nausea<br />
Pyrexia<br />
Date:11/10/97ISR Number: 3005888-7Report Type:Direct Company Report # Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Restlessness Lariam PS<br />
Depression <strong>Mefloquine</strong> SS<br />
Confusional State<br />
Emotional Disorder<br />
Memory Impairment<br />
Visual Impairment<br />
Anxiety<br />
Date:11/12/97ISR Number: 3000157-3Report Type:Expedited (15-DaCompany Report #89097 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Health Lariam Tablets<br />
Initial or Prolonged Lung Infiltration Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL<br />
Date:11/12/97ISR Number: 3000321-3Report Type:Expedited (15-DaCompany Report #89246 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Uterine Contractions Foreign Lariam Tablets
Intervention to Abnormal Health (<strong>Mefloquine</strong><br />
Prevent Permanent Professional Hydrochloride) PS ORAL 250MG 1 X PER<br />
Impairment/Damage Other WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 2
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/12/97ISR Number: 3000325-0Report Type:Expedited (15-DaCompany Report #89247 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Amylase Increased Foreign Lariam Tablets<br />
Initial or Prolonged Pancreatitis Health (<strong>Mefloquine</strong><br />
Lipase Increased Professional Hydrochloride) PS ORAL 250MG 1 X PER<br />
Other<br />
WEEK, ORAL<br />
Imodium (Loperamide<br />
Hydrochloride) C<br />
Flagyl<br />
(Metronidazole) C<br />
Date:11/12/97ISR Number: 3000331-6Report Type:Expedited (15-DaCompany Report #82871 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tachycardia Foreign Lariam Tablets<br />
Hyperventilation Health (Mefloquiine<br />
Alkalosis Hypokalaemic Professional Hydrochloride) PS ORAL 250MG 1 X PER<br />
Paraesthesia Other WEEK ORAL<br />
Cyclo-Menorette<br />
(Estradiol Valerate/<br />
Estradiol Succinate/<br />
Levonorgestrel) C<br />
Date:11/12/97ISR Number: 3000529-7Report Type:Expedited (15-DaCompany Report #80739 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Astrocytoma Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Convulsion Health 1.0 X PER<br />
Complex Partial Seizures Professional WEEK ORAL<br />
Grand Mal Convulsion Phenhydan C<br />
Paresis<br />
Hallucination<br />
Hemiplegia<br />
Aphasia<br />
Speech Disorder<br />
Blood Glucose Decreased<br />
Blood Lactic Acid<br />
Decreased<br />
Brain Neoplasm<br />
Glioma<br />
Simple Partial Seizures<br />
Paralysis<br />
Blood Albumin Abnormal<br />
Hemiparesis<br />
Date:11/17/97ISR Number: 3000972-6Report Type:Expedited (15-DaCompany Report #85820 Age:26 YR Gender:Male I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Migraine<br />
Headache<br />
Gastroenteritis Bacterial<br />
Trance<br />
Illusion<br />
Paraesthesia<br />
03-Apr-2012 09:37 AM Page: 3
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diarrhoea<br />
Disturbance In Attention<br />
Restlessness Report Source Product Role Manufacturer Route Dose Duration<br />
Psychotic Disorder Foreign Lariam PS ORAL 250.0000 MG<br />
Health<br />
1.0 X PER<br />
Professional<br />
WEEK ORAL<br />
Ciprobay<br />
C<br />
Date:11/17/97ISR Number: 3006058-9Report Type:Direct Company Report # Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia <strong>Mefloquine</strong> PS ORAL 1 TABLET PER<br />
Fatigue<br />
WEEK<br />
Depression Prozac C<br />
Immune System Disorder<br />
Weight Decreased<br />
Decreased Appetite<br />
Date:11/17/97ISR Number: 3006086-3Report Type:Direct Company Report # Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety <strong>Mefloquine</strong> PS ORAL Q WEEK PO<br />
Gastrooesophageal Reflux<br />
Disease<br />
Date:11/19/97ISR Number: 3001851-0Report Type:Expedited (15-DaCompany Report #85820 Age:26 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Restlessness Foreign Lariam PS ORAL 250.000 MG<br />
Initial or Prolonged Sleep Disorder Health 1.0 X PER<br />
Trance Professional WEEK ORAL<br />
Headache Ciprobay C<br />
Psychotic Disorder<br />
Paraesthesia<br />
Migraine<br />
Illusion<br />
Gastroenteritis Bacterial<br />
Suicidal Ideation<br />
Disturbance In Attention<br />
Diarrhoea<br />
Date:11/21/97ISR Number: 3002261-2Report Type:Expedited (15-DaCompany Report #88615 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Foreign Lariam PS ORAL 250.0000 MG
Tachycardia Health 1.0 X PER<br />
Arrhythmia Professional WEEK ORAL<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 4
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/26/97ISR Number: 3002889-XReport Type:Expedited (15-DaCompany Report #89882 Age:82 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paralysis Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Sensory Disturbance Health 1.0 X PER<br />
Gait Disturbance Professional WEEK ORAL<br />
Hypoaesthesia Insuline C<br />
Coordination Abnormal<br />
Feeling Drunk<br />
Sciatica<br />
Date:11/26/97ISR Number: 3002892-XReport Type:Expedited (15-DaCompany Report #89097 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Health Lariam PS ORAL 10.0000 MG/KG<br />
Initial or Prolonged Lung Infiltration Professional DAILY ORAL<br />
Date:12/01/97ISR Number: 3003013-XReport Type:Expedited (15-DaCompany Report #86213 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Muscle Spasms Health Lariam PS ORAL 1.2500 GRAM<br />
Medication Error Professional 1.0 X PER<br />
Pain Other ODOS ORAL<br />
Nausea Tetanus Vaccine C<br />
Tremor Hepatitis Vaccine C<br />
Dizziness Birth Control Pills C<br />
Agitation<br />
Confusional State<br />
Anxiety<br />
Condition Aggravated<br />
Date:12/01/97ISR Number: 3003015-3Report Type:Expedited (15-DaCompany Report #90214 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bradycardia Foreign Lariam PS ORAL 250.0000 MG<br />
Abdominal Pain Other 1.0 X PER<br />
Syncope<br />
WEEK ORAL<br />
Dizziness<br />
Date:12/01/97ISR Number: 3003374-1Report Type:Expedited (15-DaCompany Report #90289 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Infection Foreign Lariam PS ORAL<br />
Urticaria<br />
Consumer<br />
Erysipeloid
Date:12/01/97ISR Number: 3003432-1Report Type:Expedited (15-DaCompany Report #92090 Age:72 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Skin Infection<br />
Urticaria<br />
03-Apr-2012 09:37 AM Page: 5
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Erysipeloid<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL<br />
Health<br />
Professional<br />
Date:12/01/97ISR Number: 3003541-7Report Type:Expedited (15-DaCompany Report #89097 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Health Lariam PS ORAL 10.0000 MG/KG<br />
Initial or Prolonged Lung Infiltration Professional DAILY ORAL<br />
Date:12/01/97ISR Number: 3082443-4Report Type:Expedited (15-DaCompany Report #90612 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Parkinsonism Health Lariam PS ORAL ORAL<br />
Extrapyramidal Disorder Professional<br />
Date:12/02/97ISR Number: 3003685-XReport Type:Expedited (15-DaCompany Report #87481 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Health Lariam PS ORAL 250.0000 MG<br />
Other Serious Hypertension Professional 1.0 X PER DAY<br />
Medication Error<br />
ORAL<br />
Vomiting<br />
Fatigue<br />
Oedema<br />
Visual Impairment<br />
Hypoaesthesia<br />
Paraesthesia<br />
Nausea<br />
Dysgeusia<br />
Date:12/02/97ISR Number: 3003799-4Report Type:Expedited (15-DaCompany Report #90767 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Toxic Skin Eruption Foreign Lariam PS ORAL 250.0000MG<br />
Initial or Prolonged Other 1.0 X PER<br />
WEEK ORAL<br />
Date:12/02/97ISR Number: 3003874-4Report Type:Expedited (15-DaCompany Report #90601 Age:36 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depressed Mood Foreign Lariam Tablets<br />
Mood Swings Other (<strong>Mefloquine</strong><br />
Ear Infection<br />
Hydrochloride)<br />
Depression Suicidal 250.000 Mg PS ORAL 250.000 MG<br />
Panic Attack<br />
1.0 X PER<br />
Suicidal Ideation<br />
WEEK ORAL<br />
Yellow Fever Vaccine C<br />
03-Apr-2012 09:37 AM Page: 6
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Rabies Vaccine<br />
Havrix<br />
C<br />
C<br />
Date:12/02/97ISR Number: 3003979-8Report Type:Expedited (15-DaCompany Report #85820 Age:26 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Diarrhoea Health 1.0 X PER<br />
Insomnia Professional WEEK ORAL<br />
Illusion<br />
TAB<br />
Paraesthesia Ciprobay SS ORAL 500.0000 MG<br />
Gastroenteritis Bacterial<br />
2.0 X PER DAY<br />
Disturbance In Attention<br />
ORAL<br />
Restlessness<br />
Headache<br />
Psychotic Disorder<br />
Hypoaesthesia<br />
Trance<br />
Date:12/02/97ISR Number: 3003982-8Report Type:Expedited (15-DaCompany Report #59906 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious No Adverse Event Foreign Lariam PS ORAL 250 MG<br />
Health Paludrine C<br />
Professional Typhim Vi C<br />
Nivaquine<br />
C<br />
Date:12/02/97ISR Number: 3004047-1Report Type:Expedited (15-DaCompany Report #90736 Age:75 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hostility Other Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Delusion 1.0 X PER<br />
Nervousness<br />
WEEK ORAL<br />
Psychotic Disorder<br />
Agitation<br />
Toxicity To Various<br />
Agents<br />
Hypersensitivity<br />
Hallucination, Auditory<br />
Trance<br />
Aggression<br />
Date:12/02/97ISR Number: 3004049-5Report Type:Expedited (15-DaCompany Report #90607 Age:78 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Dementia Foreign Lariam PS ORAL ORAL<br />
Health<br />
Professional<br />
03-Apr-2012 09:37 AM Page: 7
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/02/97ISR Number: 3004114-2Report Type:Expedited (15-DaCompany Report #89618 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Decreased Appetite Consumer Lariam PS ORAL 250.0000 MG<br />
Abnormal Dreams<br />
1.0 X PER<br />
Crying<br />
WEEK ORAL<br />
Palpitations<br />
Hyperreflexia<br />
Hypoaesthesia<br />
Insomnia<br />
Abnormal Behaviour<br />
Muscle Contractions<br />
Involuntary<br />
Motor Dysfunction<br />
Panic Reaction<br />
Drug Hypersensitivity<br />
Paraesthesia<br />
Date:12/03/97ISR Number: 3003946-4Report Type:Expedited (15-DaCompany Report #88694 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paraesthesia Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Dizziness Consumer 1.0 X PER<br />
Asthenia Health WEEK ORAL 56 DAY<br />
Blood Calcium Increased Professional<br />
Alopecia<br />
Arthralgia<br />
Headache<br />
Hypoaesthesia<br />
Hypoparathyroidism<br />
Hyperhidrosis<br />
Visual Impairment<br />
Date:12/03/97ISR Number: 3007297-3Report Type:Direct Company Report # Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Panic Attack Lariam PS 1 PER WEEK<br />
Intervention to<br />
Insomnia<br />
Prevent Permanent Anxiety<br />
Impairment/Damage Agoraphobia<br />
Dyspnoea<br />
Date:12/08/97ISR Number: 3004513-9Report Type:Expedited (15-DaCompany Report #90887 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Muscle Rigidity Foreign Lariam PS ORAL 250.000 MG<br />
Initial or Prolonged Muscle Twitching Other 1.0 X PER
Blood Glucose Decreased WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 8
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/08/97ISR Number: 3004514-0Report Type:Expedited (15-DaCompany Report #90886 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Photosensitivity Reaction Foreign Lariam PS ORAL 250.000 MG<br />
Rash Pustular Other 1.0 X PER<br />
Skin Infection<br />
WEEK ORAL<br />
Sakmonella Vaccine C<br />
Immunoglobulins C<br />
Poliomyelitis<br />
Vaccine<br />
C<br />
Date:12/08/97ISR Number: 3004766-7Report Type:Expedited (15-DaCompany Report #90692 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cough Consumer Lariam PS ORAL 250.0000 MG<br />
Deafness Neurosensory<br />
1.0 X PER<br />
Tinnitus<br />
WEEK ORAL<br />
Date:12/08/97ISR Number: 3004769-2Report Type:Expedited (15-DaCompany Report #89328 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Missed Foreign Lariam PS ORAL 250.0000MG<br />
Health<br />
1.0X PER WEEK<br />
Professional<br />
ORAL<br />
Date:12/09/97ISR Number: 3004654-6Report Type:Expedited (15-DaCompany Report #911054 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Electrocardiogram Qrs Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Complex Abnormal Literature 1.0 X PER<br />
Chest Pain<br />
WEEK ORAL<br />
Atrial Flutter<br />
Condition Aggravated<br />
Atrioventricular Block<br />
Palpitations<br />
Heart Rate Increased<br />
Conduction Disorder<br />
Tachycardia<br />
Date:12/09/97ISR Number: 3004657-1Report Type:Expedited (15-DaCompany Report #90945 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Foreign Lariam PS ORAL 1.5000 MG<br />
Initial or Prolonged Jaundice Literature<br />
Asthenia
Pyrexia<br />
Malaria<br />
Chills<br />
Chromaturia<br />
Blood Creatinine<br />
Increased<br />
Haemolysis<br />
03-Apr-2012 09:37 AM Page: 9
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/09/97ISR Number: 3004686-8Report Type:Expedited (15-DaCompany Report #91054 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Palpitations Literature 1.0 X PER<br />
Atrial Flutter<br />
WEEK ORAL<br />
Heart Rate Increased<br />
Tachycardia<br />
Date:12/11/97ISR Number: 3007641-7Report Type:Direct Company Report # Age:8 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coordination Abnormal <strong>Mefloquine</strong> PS Roche ORAL 125 MG WEEKLY<br />
Initial or Prolonged Photophobia<br />
Gait Disturbance<br />
Asthenia<br />
Headache<br />
Date:12/12/97ISR Number: 3006775-0Report Type:Expedited (15-DaCompany Report #91314 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Abnormal Behaviour Foreign Lariam PS ORAL 250.0000MG<br />
Completed Suicide Other 1.0 X PER<br />
WEEK ORAL<br />
Chlorquine SS UNKNOWN<br />
Date:12/16/97ISR Number: 3008113-6Report Type:Expedited (15-DaCompany Report #91370 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Ulna Fracture Foreign Lariam PS ORAL 250.0000MG<br />
Initial or Prolonged Delusional Disorder, Other 1.0 X PER<br />
Persecutory Type<br />
WEEK ORAL<br />
Suicide Attempt<br />
Radius Fracture<br />
Psychotic Disorder<br />
Pelvic Fracture<br />
Date:12/16/97ISR Number: 3008116-1Report Type:Expedited (15-DaCompany Report #89882 Age:82 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gait Disturbance Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Paralysis Health 1.0 X PER<br />
Hypoaesthesia Professional WEEK ORAL<br />
Muscular Weakness Daonil C<br />
Insulin<br />
C
Date:12/23/97ISR Number: 3010716-XReport Type:Expedited (15-DaCompany Report #91691 Age:50 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Hallucination, Tactile<br />
Hyperhidrosis<br />
03-Apr-2012 09:37 AM Page: 10
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Malaise<br />
Hallucination, Visual<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250.0000MG<br />
Other<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:12/30/97ISR Number: 3097260-9Report Type:Direct Company Report # Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nervousness Lariam PS 250MG /WK X3<br />
Feeling Jittery<br />
Malaise<br />
Insomnia<br />
Panic Attack<br />
Date:01/02/98ISR Number: 3014141-7Report Type:Expedited (15-DaCompany Report #JAFRA-36938 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Staphylococcal Infection Foreign Imodium PS Janssen ORAL ORAL<br />
Initial or Prolonged Arthralgia Ercefuryl SS ORAL ORAL<br />
Pyrexia Smecta SS 30 DAY<br />
Aspartate Engerix B SS INTRAMUSCULAR 20 MCG TOTAL<br />
Aminotransferase<br />
INTRAMUSCULAR<br />
Increased Lariam SS ORAL 1 WEEKLY ORAL 7 WK<br />
Arthritis Reactive Tiorfan C ORAL 100 MG ORAL 30 DAY<br />
Diarrhoea<br />
Hepatomegaly<br />
Conjunctivitis<br />
Alanine Aminotransferase<br />
Increased<br />
Date:01/07/98ISR Number: 3015608-8Report Type:Expedited (15-DaCompany Report #90736 Age:75 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Other Lariam PS ORAL 250.0000MG<br />
Initial or Prolonged Delusion 1.0 X PER<br />
Aggression<br />
WEEK ORAL<br />
Nervousness<br />
Toxicity To Various<br />
Agents<br />
Agitation<br />
Trance<br />
Date:01/08/98ISR Number: 3015567-8Report Type:Expedited (15-DaCompany Report #92037 Age: Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam PS 250.0000MG<br />
Initial or Prolonged Suicidal Ideation Consumer 1.0 X PER<br />
Anxiety<br />
WEEK<br />
03-Apr-2012 09:37 AM Page: 11
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/08/98ISR Number: 3016847-2Report Type:Expedited (15-DaCompany Report #92171 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myalgia Foreign Lariam PS ORAL 250 MG 1.0 X<br />
Initial or Prolonged Sick Sinus Syndrome Other PER WEEK ORAL<br />
Post Viral Fatigue Thyroxine C<br />
Syndrome<br />
Date:01/12/98ISR Number: 3016579-0Report Type:Expedited (15-DaCompany Report #92444 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Small For Dates Baby Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Maternal Drugs Affecting Consumer 1.0 X PER<br />
Congenital Anomaly Foetus Other WEEK ORAL<br />
Premature Baby Adepal SS ORAL 1.0000 DOSE<br />
Foetal Growth Restriction<br />
FORM 1.0 X<br />
PER DAY ORAL<br />
Yellow Fever Vaccine SS SUBCUTANEOUS 1.0000 DOSE<br />
FORM 1.0 X<br />
PER ODOS<br />
SUBCUTANEOUS<br />
Hepatitis A Vaccine SS INTRAMUSCULAR 1.0 X PER<br />
ODOS<br />
INTRAMUSCULAR<br />
Hepatitis B Vaccine SS INTRAMUSCULAR 1.0 X PER<br />
ODOS<br />
INTRAMUSCULAR<br />
Date:01/15/98ISR Number: 3018437-4Report Type:Direct Company Report # Age:80 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fall Lariam PS Laroche 1 WEEKLY FOR<br />
Gait Disturbance<br />
8 WKS<br />
Balance Disorder Novaldex C<br />
Date:01/15/98ISR Number: 3053811-1Report Type:Periodic Company Report #WAES 97041992 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
PT<br />
Chills<br />
Anxiety<br />
Fatigue<br />
Dizziness<br />
Headache<br />
Myalgia<br />
Nausea<br />
Palpitations<br />
Asthenia
Tinnitus<br />
Accommodation Disorder<br />
Diarrhoea<br />
Decreased Appetite<br />
Abdominal Pain<br />
Depression<br />
Pruritus<br />
Drug Interaction<br />
03-Apr-2012 09:37 AM Page: 12
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dysgeusia<br />
Emotional Disorder<br />
Salivary Hypersecretion Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer Crixivan PS Merck Research<br />
Health<br />
Laboratories Div<br />
Professional Merck Co Inc ORAL 800 MG/Q8H/PO<br />
Lariam Unk SS ORAL PO<br />
Halicon<br />
C<br />
Triazolam<br />
C<br />
Retin-A<br />
C<br />
Valtrex<br />
C<br />
Zerit<br />
C<br />
Cimetidine<br />
C<br />
Epivir<br />
C<br />
(Beta)-Carotene C<br />
Nizoral<br />
C<br />
Date:01/21/98ISR Number: 3017831-5Report Type:Expedited (15-DaCompany Report #92564 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Bradycardia Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Gastroenteritis Health 1.0 X PER<br />
Professional<br />
WEEK ORAL<br />
Date:01/22/98ISR Number: 3018708-1Report Type:Direct Company Report # Age:80 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Lariam PS La Roche<br />
Gait Disturbance Pharmaceutical Co 1 PER WEEK<br />
FOR 8 WEEKS<br />
Nolvadex<br />
C<br />
Date:01/27/98ISR Number: 3019299-1Report Type:Expedited (15-DaCompany Report #92891 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Foreign Lariam PS ORAL 250.000 MG<br />
Balance Disorder Health 1.0 X PER<br />
Gait Disturbance Professional WEEK ORAL<br />
Geluprane<br />
C<br />
Date:01/27/98ISR Number: 3019406-0Report Type:Expedited (15-DaCompany Report #91568 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Foreign Lariam PS ORAL 250.0000 MG<br />
Health<br />
1.0 X PER
Professional WEEK ORAL 28 DAY<br />
Premarin<br />
C<br />
Salazopyrine<br />
C<br />
03-Apr-2012 09:37 AM Page: 13
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/02/98ISR Number: 3021867-8Report Type:Direct Company Report # Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations <strong>Mefloquine</strong> PS ORAL 250 MG PO 8<br />
Initial or Prolonged Fatigue DOSES<br />
Chest Pain<br />
Date:02/02/98ISR Number: 3021875-7Report Type:Expedited (15-DaCompany Report #80873 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Thymus Enlargement Foreign Lariam PS ORAL 250.000 MG<br />
Trisomy 21 Health 1.0 X PER<br />
Congenital Anomaly Professional WEEK ORAL<br />
Ventricular Septal Defect<br />
Acquired<br />
Dysmorphism<br />
Maternal Drugs Affecting<br />
Foetus<br />
Abortion Induced<br />
Date:02/02/98ISR Number: 3021893-9Report Type:Direct Company Report # Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Health Lariam PS ORAL 250 MG WEEKLY 4 WK<br />
Initial or Prolonged Chest Discomfort Professional<br />
Decreased Appetite<br />
Cardiac Disorder<br />
Asthenia<br />
Heart Rate Increased<br />
Date:02/02/98ISR Number: 3021894-0Report Type:Expedited (15-DaCompany Report #89618 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Palpitations Consumer Lariam PS ORAL 250.OOO MG<br />
Insomnia<br />
1.0 X PER<br />
Crying<br />
WEEK ORAL<br />
Decreased Appetite<br />
Hyperreflexia<br />
Panic Attack<br />
Motor Dysfunction<br />
Paraesthesia<br />
Abnormal Dreams<br />
Date:02/02/98ISR Number: 3022891-1Report Type:Expedited (15-DaCompany Report #87335 Age:25 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Foreign Lariam PS ORAL 250.00 MG 1.0<br />
Initial or Prolonged Pyrexia Other X PER WEEK<br />
Embolism Arterial<br />
ORAL<br />
Headache Marvelon C<br />
Hemiparesis<br />
03-Apr-2012 09:37 AM Page: 14
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/04/98ISR Number: 3022672-9Report Type:Expedited (15-DaCompany Report #90763 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Leukopenia Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Hyperhidrosis Health 1.0 X PER<br />
Myalgia Professional WEEK ORAL<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Fatigue<br />
Hepatic Enzyme Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Skin Discolouration<br />
Myopathy<br />
Liver Function Test<br />
Abnormal<br />
Alanine Aminotransferase<br />
Increased<br />
Chest Pain<br />
Pain In Extremity<br />
Diarrhoea<br />
Hypogammaglobulinaemia<br />
Tongue Coated<br />
Cardiovascular Disorder<br />
Muscular Weakness<br />
Myocarditis<br />
Hepatic Steatosis<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Headache<br />
Myoglobin Blood Increased<br />
Date:02/04/98ISR Number: 3022673-0Report Type:Expedited (15-DaCompany Report #92891 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Balance Disorder Foreign Lariam PS ORAL 250.0000 MG<br />
Vertigo Health 1.0 X PER<br />
Gait Disturbance Professional WEEK ORAL<br />
Dizziness Geluprane C<br />
Date:02/04/98ISR Number: 3023912-2Report Type:Expedited (15-DaCompany Report #87481 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
Other Serious<br />
PT<br />
Amnesia<br />
Fatigue<br />
Nausea<br />
Hypoaesthesia<br />
Nervous System Disorder
Overdose<br />
Dysgeusia<br />
Hypertension<br />
Oedema<br />
Disturbance In Attention<br />
Visual Field Defect<br />
Paraesthesia<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 15
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer Lariam PS ORAL 250.000 MG<br />
Health<br />
1.0 X PER DAY<br />
Professional<br />
ORAL<br />
Date:02/09/98ISR Number: 3025971-XReport Type:Expedited (15-DaCompany Report #88651 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diplopia Foreign Lariam PS ORAL 250.0000 MG<br />
Vith Nerve Paralysis Health 1.0 X PER<br />
Nausea Professional WEEK ORAL<br />
Headache<br />
TAB<br />
Date:02/09/98ISR Number: 3026005-3Report Type:Expedited (15-DaCompany Report #93517 Age:73 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Heart Rate Decreased Foreign Lariam PS ORAL 250.0000 MG<br />
Hospitalization - Atrioventricular Block Other 1.0 X PER<br />
Initial or Prolonged Complete WEEK ORAL<br />
Required<br />
Dizziness<br />
Intervention to<br />
Pallor<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:02/10/98ISR Number: 3027135-2Report Type:Expedited (15-DaCompany Report #87481 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Mental Impairment Health Lariam PS ORAL 250.000 MG<br />
Medication Error Professional 2.0 X WEEK<br />
Visual Impairment Other ORAL<br />
Vomiting<br />
Overdose<br />
Hypertension<br />
Drug Level Above<br />
Therapeutic<br />
Dysgeusia<br />
Dizziness<br />
Brain Injury<br />
Visual Acuity Reduced<br />
Amnesia<br />
Fatigue<br />
Paraesthesia<br />
Nausea<br />
Oedema
Date:02/12/98ISR Number: 3027269-2Report Type:Expedited (15-DaCompany Report #92564 Age:16 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Bradycardia<br />
Gastroenteritis<br />
Asthenia<br />
03-Apr-2012 09:37 AM Page: 16
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hypotension<br />
Red Blood Cell<br />
Sedimentation Rate Report Source Product Role Manufacturer Route Dose Duration<br />
Increased Foreign Lariam PS ORAL 250.0000 MG<br />
Health<br />
1.0 X PER<br />
Professional<br />
WEEK ORAL<br />
Date:02/12/98ISR Number: 3028339-5Report Type:Expedited (15-DaCompany Report #93802 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam PS ORAL 50.000 MG 1.0<br />
Initial or Prolonged Anxiety Other X PER WEEK<br />
Thinking Abnormal<br />
Tremor<br />
Personality Disorder<br />
Abnormal Behaviour<br />
Date:02/12/98ISR Number: 3030309-8Report Type:Expedited (15-DaCompany Report #89882 Age:82 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoaesthesia Foreign Lariam PS ORAL 250.000 MG<br />
Initial or Prolonged Feeling Drunk Health 1.0 X PER<br />
Coordination Abnormal Professional WEEK ORAL<br />
Paralysis Daonil C<br />
Balance Disorder Insuline C<br />
Muscular Weakness<br />
Monoparesis<br />
Date:02/12/98ISR Number: 3030313-XReport Type:Expedited (15-DaCompany Report #87481 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypoaesthesia Consumer Lariam PS ORAL 250.0000 MG<br />
Other Serious Dysgeusia Health 1.0 X PER DAY<br />
Hypertension Professional ORAL<br />
Medication Error<br />
Nausea<br />
Fatigue<br />
Visual Impairment<br />
Dizziness<br />
Disturbance In Attention<br />
Vomiting<br />
Memory Impairment<br />
Brain Injury<br />
Oedema
Date:02/13/98ISR Number: 3030113-0Report Type:Expedited (15-DaCompany Report #JAFRA-36938 Age:25 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Diarrhoea<br />
Hepatic Function Abnormal<br />
Hepatomegaly<br />
Pyrexia<br />
Arthralgia<br />
03-Apr-2012 09:37 AM Page: 17
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Conjunctivitis<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Imodium PS Janssen ORAL ORAL<br />
Health Ercefuryl SS ORAL ORAL<br />
Professional Smecta SS 30 DAY<br />
Engerix B SS INTRAMUSCULAR 20 MCG TOTAL,<br />
INTRAMUSCULAR<br />
SUSPENSION 20<br />
MCG<br />
Lariam SS ORAL 1 TABLET<br />
WEEKLY ORAL 7 WK<br />
Tiorfan C ORAL 100MG<br />
CAPSULES 30 DAY<br />
Date:02/18/98ISR Number: 3032216-3Report Type:Expedited (15-DaCompany Report #94117 Age:67 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Polyneuropathy Foreign Lariam PS ORAL 250.0000 MG<br />
Intervention to Gait Disturbance Health 1.0 X PER<br />
Prevent Permanent Spinal Osteoarthritis Professional WEEK ORAL<br />
Impairment/Damage Sciatica<br />
Back Pain<br />
Lumbar Spinal Stenosis<br />
Date:02/18/98ISR Number: 3032221-7Report Type:Expedited (15-DaCompany Report #93891 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Renal Failure Foreign Lariam PS ORAL 250 MG<br />
Cardiac Arrest Consumer ORAL;TAB<br />
Malaria<br />
Date:02/18/98ISR Number: 3036413-2Report Type:Direct Company Report # Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Panic Attack Lariam PS PRESCRIBED<br />
Required Convulsion AMOUNTS<br />
Intervention to<br />
Dizziness<br />
Prevent Permanent<br />
Impairment/Damage<br />
Tremor<br />
Vision Blurred<br />
Anxiety<br />
Date:02/19/98ISR Number: 3032366-1Report Type:Expedited (15-DaCompany Report #94074 Age:30 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Depression Foreign Lariam PS ORAL ORAL<br />
Suicidal Ideation<br />
Other<br />
03-Apr-2012 09:37 AM Page: 18
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/23/98ISR Number: 3035857-2Report Type:Expedited (15-DaCompany Report #93243 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Alkaline Foreign Lariam PS ORAL 1500.0000 MG<br />
Phosphatase Increased Health ORAL<br />
Gamma-Glutamyltransferase Professional<br />
Increased<br />
Platelet Count Decreased<br />
White Blood Cell Count<br />
Decreased<br />
Thrombocytopenia<br />
Alanine Aminotransferase<br />
Increased<br />
Date:02/23/98ISR Number: 3035884-5Report Type:Expedited (15-DaCompany Report #94429 Age:4 MON Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Pneumonia Viral Literature Lariam PS ORAL 250 MG 1 X<br />
Maternal Drugs Affecting<br />
PER WEEK ORAL<br />
Foetus<br />
Date:02/23/98ISR Number: 3035887-0Report Type:Expedited (15-DaCompany Report #94430 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Literature Lariam PS ORAL 250 MG 1 X<br />
Foetus<br />
PER WEEK ORAL<br />
Benign Hydatidiform Mole<br />
Date:02/23/98ISR Number: 3036234-0Report Type:Expedited (15-DaCompany Report #94382 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Coordination Abnormal Foreign Lariam PS ORAL 250.0000MG<br />
Agraphia Other 1.0 X PER<br />
WEEK ORAL<br />
Date:02/23/98ISR Number: 3037257-8Report Type:Expedited (15-DaCompany Report #93562 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cardiolipin Antibody Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Positive Health 1.0 X PER<br />
Abortion Spontaneous Professional WEEK ORAL<br />
Dna Antibody Positive<br />
Systemic Lupus<br />
Erythematosus<br />
Pre-Eclampsia
Maternal Drugs Affecting<br />
Foetus<br />
03-Apr-2012 09:37 AM Page: 19
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/23/98ISR Number: 3037262-1Report Type:Expedited (15-DaCompany Report #94371 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Foreign Lariam PS ORAL 250 MG 1 X<br />
Health<br />
PER WEEK ORAL<br />
Professional<br />
Date:02/24/98ISR Number: 3035602-0Report Type:Expedited (15-DaCompany Report #94035 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Foreign Lariam PS ORAL 250.000 MG<br />
Dyskinesia Other ORAL<br />
Insomnia<br />
Dizziness<br />
Nausea<br />
Date:02/24/98ISR Number: 3035604-4Report Type:Expedited (15-DaCompany Report #94037 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urticaria Foreign Lariam PS ORAL 250.000 MG<br />
Angioedema Other ORAL<br />
Date:02/24/98ISR Number: 3035607-XReport Type:Expedited (15-DaCompany Report #947072 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Foreign Lariam PS ORAL 250.000 MG<br />
Suicidal Ideation Other ORAL<br />
Anxiety<br />
Date:02/24/98ISR Number: 3035611-1Report Type:Expedited (15-DaCompany Report #94073 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Foreign Lariam PS ORAL 250.000 MG<br />
Agitation Other ORAL<br />
Anxiety<br />
Palpitations<br />
Atrial Fibrillation<br />
Date:02/24/98ISR Number: 3035613-5Report Type:Expedited (15-DaCompany Report #94074 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Foreign Lariam PS ORAL 250.000 MG<br />
Suicidal Ideation Other ORAL
Date:02/24/98ISR Number: 3035616-0Report Type:Expedited (15-DaCompany Report #94092 Age:24 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Depression<br />
Psychotic Disorder<br />
03-Apr-2012 09:37 AM Page: 20
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Suicidal Ideation<br />
Anxiety<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250.000 MG<br />
Other<br />
ORAL<br />
Date:02/24/98ISR Number: 3035620-2Report Type:Expedited (15-DaCompany Report #94093 Age:73 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Foreign Lariam PS ORAL 250.000 MG<br />
Renal Failure Acute Other ORAL<br />
Dermatitis<br />
Disorientation<br />
Date:02/24/98ISR Number: 3035623-8Report Type:Expedited (15-DaCompany Report #94101 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Foreign Lariam PS ORAL 250.000 MG<br />
Dyskinesia Other ORAL<br />
Agitation<br />
Date:02/24/98ISR Number: 3035628-7Report Type:Expedited (15-DaCompany Report #04106 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hyperhidrosis Foreign Lariam PS ORAL 250.000 MG<br />
Vomiting Other ORAL<br />
Palpitations<br />
Psychotic Disorder<br />
Diarrhoea<br />
Hallucination<br />
Paralysis<br />
Date:02/24/98ISR Number: 3035644-5Report Type:Expedited (15-DaCompany Report #94124 Age:5 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anaemia Foreign Lariam PS ORAL 250.000 MG<br />
Coombs Direct Test Other ORAL<br />
Positive<br />
Date:02/24/98ISR Number: 3035647-0Report Type:Expedited (15-DaCompany Report #94126 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Foreign Lariam PS ORAL 250.000 MG
Angina Pectoris Other 1.0 X PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 21
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/24/98ISR Number: 3035650-0Report Type:Expedited (15-DaCompany Report #94128 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bronchospasm Foreign Lariam PS ORAL 250.0000 MG<br />
Other<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:02/24/98ISR Number: 3035652-4Report Type:Expedited (15-DaCompany Report #94131 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cardiac Failure Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Other 1.0 X PER<br />
WEEK ORAL<br />
Date:02/24/98ISR Number: 3035657-3Report Type:Expedited (15-DaCompany Report #94137 Age:78 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Cholestatic Foreign Lariam PS ORAL 250.0000 MG<br />
Other<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:02/24/98ISR Number: 3035660-3Report Type:Expedited (15-DaCompany Report #94146 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoglycaemia Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Other 1.0 X PER<br />
WEEK ORAL<br />
Date:02/24/98ISR Number: 3103313-9Report Type:Expedited (15-DaCompany Report #94065 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Angioedema Foreign Lariam PS ORAL 250.000 MG<br />
Dermatitis Other ORAL<br />
Depressed Mood<br />
Date:03/02/98ISR Number: 3050236-XReport Type:Direct Company Report # Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare <strong>Mefloquine</strong> PS ORAL 250 MG PO<br />
Date:03/03/98ISR Number: 3040313-1Report Type:Expedited (15-DaCompany Report #94569 Age:12 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Obsessive-Compulsive Health Lariam PS ORAL<br />
Disorder<br />
Professional<br />
Emotional Disorder<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 22
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/03/98ISR Number: 3049890-8Report Type:Direct Company Report # Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Depressed Level Of Lariam PS Hoffmann-Laroche 250MG WKLY<br />
Hospitalization - Consciousness<br />
Initial or Prolonged Hallucination, Visual<br />
Disability<br />
Myalgia<br />
Required<br />
Muscular Weakness<br />
Intervention to<br />
Depression<br />
Prevent Permanent Pain In Extremity<br />
Impairment/Damage Hypersensitivity<br />
Dizziness<br />
Headache<br />
Paralysis<br />
Bone Pain<br />
Injury<br />
Pain<br />
Pyrexia<br />
Dyspnoea<br />
Psychotic Disorder<br />
Agitation<br />
Paraesthesia<br />
Tinnitus<br />
Educational Problem<br />
Heart Rate Irregular<br />
Coordination Abnormal<br />
Loss Of Consciousness<br />
Insomnia<br />
Chills<br />
Abnormal Behaviour<br />
Sleep Disorder<br />
Hallucination, Auditory<br />
Abnormal Dreams<br />
Palpitations<br />
Cold Sweat<br />
Speech Disorder<br />
Visual Impairment<br />
Influenza Like Illness<br />
Date:03/05/98ISR Number: 3050365-0Report Type:Direct Company Report # Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam PS Hoffman La Roche 1 TAB/WEEK<br />
Pain In Extremity<br />
Scab<br />
Insomnia<br />
Dermatitis Bullous<br />
Rash Papular<br />
Urticaria
Date:03/05/98ISR Number: 3050366-2Report Type:Direct Company Report # Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sleep Disorder Consumer Lariam PS Hoffman Laroche 1 TAB/WK<br />
Dermatitis Bullous<br />
03-Apr-2012 09:37 AM Page: 23
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/10/98ISR Number: 3049117-7Report Type:Direct Company Report # Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Abnormal Dreams Lariam PS ORAL 1 PILL WEEKLY<br />
Photosensitivity Reaction Vitamins C<br />
Scleral Discolouration<br />
Tooth Discolouration<br />
Sleep Disorder<br />
Date:03/10/98ISR Number: 3054941-0Report Type:Expedited (15-DaCompany Report #88694 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Visual Impairment Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Asthenia Consumer 1.0 X PER<br />
Blood Calcium Decreased Health WEEK ORAL<br />
Infectious Mononucleosis Professional<br />
Paraesthesia<br />
Arthralgia<br />
Dizziness<br />
Hyperhidrosis<br />
Mental Disorder<br />
Hypoaesthesia<br />
Arrhythmia<br />
Tension Headache<br />
Alopecia<br />
Sensory Disturbance<br />
Hypoparathyroidism<br />
Headache<br />
Date:03/12/98ISR Number: 3058545-5Report Type:Direct Company Report # Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS 1 PER WEEK 5<br />
Required Panic Attack DOSES<br />
Intervention to Birth Control Pills C<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:03/12/98ISR Number: 3059187-8Report Type:Direct Company Report # Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Health Sibutamine PS Knoll ORAL 10 MG QD<br />
Agitation Professional <strong>Mefloquine</strong> SS Roche ORAL 250 MG Q WEEK<br />
Nervousness
Date:03/16/98ISR Number: 3055731-5Report Type:Expedited (15-DaCompany Report #94329 Age:2 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Inappropriate<br />
Antidiuretic Hormone<br />
Secretion<br />
Diabetes Insipidus<br />
Polyuria<br />
03-Apr-2012 09:37 AM Page: 24
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Polydipsia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250 MG 1.0 X<br />
Health<br />
PER WEEK ORAL<br />
Professional<br />
Date:03/16/98ISR Number: 3055732-7Report Type:Expedited (15-DaCompany Report #95577 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Foreign Lariam PS ORAL 1 DOSE FORM 1<br />
Initial or Prolonged Dehydration Other X PER DAY<br />
Abdominal Pain Upper<br />
ORAL<br />
Cholecystitis<br />
Vomiting<br />
Gastroenteritis<br />
Date:03/17/98ISR Number: 3055864-3Report Type:Expedited (15-DaCompany Report #940202190001 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Foetal Growth Restriction Other Lariam PS ORAL 250.000 MG<br />
Premature Baby<br />
1.0 X PER<br />
Maternal Drugs Affecting<br />
WEEK ORAL<br />
Foetus Iron C<br />
Orasthin<br />
C<br />
Vitamins<br />
C<br />
Date:03/17/98ISR Number: 3055868-0Report Type:Expedited (15-DaCompany Report #940202190002 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Premature Labour Literature Lariam PS ORAL 250.0000 MG<br />
Polyhydramnios Other 1.0 X PER<br />
WEEK ORAL<br />
Meningococci Vaccine C<br />
Cholera Vaccine C<br />
Plague Vaccine C<br />
Immunoglobulins C<br />
Typhoid Vaccine C<br />
Date:03/17/98ISR Number: 3055872-2Report Type:Expedited (15-DaCompany Report #940202190003 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Jaundice Neonatal Literature Lariam PS ORAL 250.0000 MG<br />
Other<br />
1.0 X PER<br />
WEEK ORAL
Iron C<br />
Tuberkulin Tine Test<br />
Ppd<br />
C<br />
Yellow Fever Vaccine C<br />
Influenza Virus C<br />
Immunoglobulins C<br />
Vitamins<br />
C<br />
Oral Contraceptive C<br />
03-Apr-2012 09:37 AM Page: 25
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/17/98ISR Number: 3055892-8Report Type:Expedited (15-DaCompany Report #92796 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Health Lariam PS ORAL 1.0 X PER<br />
Initial or Prolonged Flight Of Ideas Professional WEEK ORAL<br />
Psychotic Disorder Birth Control Pills C<br />
Diarrhoea<br />
Paranoia<br />
Hallucination, Auditory<br />
Delusional Disorder,<br />
Unspecified Type<br />
Dystonia<br />
Thinking Abnormal<br />
Agitation<br />
Mood Swings<br />
Date:03/18/98ISR Number: 3056397-0Report Type:Direct Company Report # Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Tooth Discolouration Lariam PS 1 PILL WEEKLY<br />
Scleral Discolouration Vitamines C<br />
Photosensitivity Reaction<br />
Abnormal Dreams<br />
Sleep Disorder<br />
Date:03/19/98ISR Number: 3057866-XReport Type:Direct Company Report # Age:15 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hallucinations, Mixed Lariam PS 3 TABLETS<br />
Hospitalization - Hyperventilation SINGLE DOSE<br />
Initial or Prolonged Chest Pain<br />
Disability<br />
Insomnia<br />
Required<br />
Abnormal Behaviour<br />
Intervention to<br />
Suicidal Ideation<br />
Prevent Permanent Psychomotor Hyperactivity<br />
Impairment/Damage<br />
Date:03/24/98ISR Number: 3059313-0Report Type:Expedited (15-DaCompany Report #95970 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancreatitis Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Other 1.0 X PER<br />
WEEK ORAL<br />
Date:03/24/98ISR Number: 3059361-0Report Type:Expedited (15-DaCompany Report #96259 Age:26 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Foreign Lariam PS ORAL 250.0000 MG<br />
Nystagmus Other 1.0 X PER<br />
Bronchospasm<br />
WEEK ORAL<br />
Panic Attack Beecham'S Powders C<br />
Tyrozets<br />
C<br />
Kalms Herbal<br />
C<br />
03-Apr-2012 09:37 AM Page: 26
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/24/98ISR Number: 3059363-4Report Type:Expedited (15-DaCompany Report #96078 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Epilepsy Foreign Lariam PS ORAL 250.0000 MG<br />
Bradycardia Health 1.0 X PER<br />
Loss Of Consciousness Professional WEEK<br />
Paraesthesia<br />
Colitofalk<br />
Amnesia (Mesalamine) C<br />
Date:03/24/98ISR Number: 3059365-8Report Type:Expedited (15-DaCompany Report #95984 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaise Foreign Lariam PS ORAL 250.0000 MG<br />
Insomnia Other 1.0 X PER<br />
Balance Disorder<br />
WEEK ORAL<br />
Oropharyngeal Pain Logynon C<br />
Pyrexia<br />
Coordination Abnormal<br />
Anosmia<br />
Fatigue<br />
Confusional State<br />
Anxiety<br />
Dysgeusia<br />
Date:03/24/98ISR Number: 3060160-4Report Type:Expedited (15-DaCompany Report #96194 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Foreign Lariam PS ORAL 250.0000 MG<br />
Dizziness Postural Health 1.0 X PER DAY<br />
Depression Professional ORAL<br />
Medication Error<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Nausea<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Diarrhoea<br />
Tinnitus<br />
Date:03/24/98ISR Number: 3060164-1Report Type:Expedited (15-DaCompany Report #96200 Age:23 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Bleeding Time Prolonged<br />
Abdominal Pain<br />
C-Reactive Protein<br />
Increased
Headache<br />
Dizziness<br />
Panic Reaction<br />
Encephalitis<br />
Psychotic Disorder<br />
Myalgia<br />
Gastrointestinal Disorder<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 27
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Transferrin Increased<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250.0000 MG<br />
Other<br />
1.O X PER<br />
WEEK ORAL<br />
Hepatitis A<br />
Vaccination<br />
C<br />
Flagyl<br />
C<br />
Yellow Fever Vaccine C<br />
Date:03/24/98ISR Number: 3060304-4Report Type:Expedited (15-DaCompany Report #91489 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cerebral Ischaemia Foreign Lariam PS ORAL 250.0000 MG<br />
Clonic Convulsion Health 1.0 X PER<br />
Spinal Disorder Professional WEEK ORAL<br />
Flu Vaccine<br />
(Influenza Virus<br />
Vaccine)<br />
C<br />
Flixotide<br />
C<br />
Salbutamol<br />
C<br />
Prozac<br />
C<br />
Tetracyclin<br />
C<br />
Date:03/24/98ISR Number: 3061536-1Report Type:Expedited (15-DaCompany Report #92796 Age:23 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Thinking Abnormal Health Lariam PS ORAL 1.0 X PER<br />
Initial or Prolonged Diarrhoea Professional WEEK ORAL<br />
Dystonia Birth Control Pills C<br />
Suicidal Ideation<br />
Psychotic Disorder<br />
Delusion<br />
Hallucination, Auditory<br />
Paranoia<br />
Agitation<br />
Mood Altered<br />
Toxicologic Test Abnormal<br />
Date:03/25/98ISR Number: 3058316-XReport Type:Expedited (15-DaCompany Report #96157 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Diplopia Health Lariam PS ORAL 250.0000 MG<br />
Condition Aggravated Professional 1.0 X PER<br />
WEEK ORAL<br />
Estratest<br />
C
Synthroid C<br />
03-Apr-2012 09:37 AM Page: 28
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/27/98ISR Number: 3061119-3Report Type:Direct Company Report # Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Palpitations <strong>Mefloquine</strong> PS Hoffman-La Roche ORAL 250MG ONCE A<br />
Dizziness<br />
WEEK PO ONLY<br />
ONE DOSE<br />
Prastitata Natural<br />
Vitamin<br />
C<br />
Date:03/27/98ISR Number: 3065019-4Report Type:Direct Company Report # Age:51 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nervous System Disorder Lariam PS ORAL TAB; 250 MG<br />
Initial or Prolonged Dissociation PO Q 7 DAYS-8<br />
Abnormal Dreams<br />
DOSES , LAST<br />
Ileus Paralytic<br />
2 MONTHS<br />
Date:03/31/98ISR Number: 3060069-6Report Type:Expedited (15-DaCompany Report #93604 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Fatigue Health 1.0 X PER DAY 28 DAY<br />
Vomiting<br />
Professional<br />
Overdose<br />
Headache<br />
Dysarthria<br />
Vision Blurred<br />
Somnolence<br />
Depression<br />
Confusional State<br />
Diarrhoea<br />
Date:03/31/98ISR Number: 3063290-6Report Type:Direct Company Report # Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia Larium PS Hoffman-La Roche LARIUM -1 A<br />
Urinary Bladder<br />
WEEK<br />
Haemorrhage Primaquine SS PRIMAQUINE -<br />
Haematochezia 1 X DAY 2 WK<br />
Ear Haemorrhage Ogen C<br />
Weight Decreased Synthroid C<br />
Diarrhoea<br />
Date:03/31/98ISR Number: 3067124-5Report Type:Direct Company Report # Age: Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam PS AS<br />
Mood Swings<br />
INSTRUCTED-DO<br />
Hallucination<br />
NT REMEMBER<br />
03-Apr-2012 09:37 AM Page: 29
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/02/98ISR Number: 3059988-6Report Type:Expedited (15-DaCompany Report #96729 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Clonic Convulsion Foreign Lariam PS ORAL 250.000 MG<br />
Other<br />
1.0 X PER<br />
WEEK ORAL<br />
Influenza Vaccine C<br />
Flixotide<br />
C<br />
Salbutamol<br />
C<br />
Prozac<br />
C<br />
Tetracycline<br />
C<br />
Date:04/02/98ISR Number: 3059990-4Report Type:Expedited (15-DaCompany Report #94329 Age:2 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diabetes Insipidus Foreign Lariam PS ORAL 50.0000 MG<br />
Polydipsia Health 1.0 X PER<br />
Polyuria Professional WEEK ORAL<br />
Date:04/02/98ISR Number: 3059992-8Report Type:Expedited (15-DaCompany Report #96767 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Pyrexia Health Lariam PS ORAL 250.0000 MG<br />
Intraocular Pressure Test Professional 1.0 X PER<br />
Chills Other WEEK ORAL<br />
Retinal Detachment<br />
Eye Pain<br />
Date:04/03/98ISR Number: 3063293-1Report Type:Expedited (15-DaCompany Report #96727 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Platelet Count Decreased Foreign Lariam PS ORAL 1500.0000 MG<br />
Leukopenia Other DAILY ORAL<br />
Thrombocytopenia<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Thrombocytosis<br />
White Blood Cell Count<br />
Increased<br />
Aspartate<br />
Aminotransferase<br />
Increased
Date:04/03/98ISR Number: 3063295-5Report Type:Expedited (15-DaCompany Report #967761 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haemangioma Of Liver Consumer Lariam PS ORAL 250.0000 MG<br />
Nephrolithiasis<br />
1.0 X PER<br />
WEEK ORAL<br />
Tetanus Booster C<br />
Polio Booster<br />
C<br />
03-Apr-2012 09:37 AM Page: 30
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Poliomyelitis<br />
Vaccines<br />
Hepatitis A Vaccine<br />
Hepatitis B Vaccine<br />
C<br />
C<br />
C<br />
Date:04/07/98ISR Number: 3061744-XReport Type:Expedited (15-DaCompany Report #90692 Age:44 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cough Consumer Lariam PS ORAL 250.0000 MG<br />
Influenza<br />
1.0 X PER<br />
Tinnitus<br />
WEEK ORAL<br />
Deafness Neurosensory<br />
Date:04/07/98ISR Number: 3061746-3Report Type:Expedited (15-DaCompany Report #83677 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrial Fibrillation Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Maternal Drugs Affecting Health 1.0 X PER<br />
Required Foetus Professional WEEK ORAL<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:04/07/98ISR Number: 3061749-9Report Type:Expedited (15-DaCompany Report #93562 Age:32 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Systemic Lupus Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Erythematosus Health 1.0 X PER<br />
Dna Antibody Positive Professional WEEK ORAL<br />
Abortion Spontaneous<br />
Other<br />
Cardiolipin Antibody<br />
Positive<br />
Pre-Eclampsia<br />
Date:04/09/98ISR Number: 3062290-XReport Type:Expedited (15-DaCompany Report #96373 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Ecchymosis Health Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Professional 1.0 X PER<br />
WEEK ORAL<br />
Flonase<br />
C<br />
Ventolin<br />
C<br />
Amoxicillin<br />
C<br />
Ortho-Cept<br />
C
Date:04/10/98ISR Number: 3061854-7Report Type:Expedited (15-DaCompany Report #97149 Age:52 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Ileus Paralytic<br />
Dehydration<br />
Feeling Abnormal<br />
03-Apr-2012 09:37 AM Page: 31
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
White Blood Cell Count<br />
Increased<br />
Abnormal Dreams Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Appetite Health Lariam PS ORAL 250.0000 MG<br />
Nausea Professional 1.0 X PER<br />
Abdominal Pain<br />
WEEK ORAL<br />
Date:04/10/98ISR Number: 3061856-0Report Type:Expedited (15-DaCompany Report #96157 Age:58 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Diplopia Health Lariam PS ORAL 250 MG 1 X<br />
Professional<br />
PER WEEK ORAL<br />
Estratest<br />
C<br />
Synthroid<br />
C<br />
Date:04/14/98ISR Number: 3073100-9Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam PS Roche ONCE PER<br />
Anxiety<br />
WEEEK<br />
Flushing<br />
Thinking Abnormal<br />
Pyrexia<br />
Insomnia<br />
Suicidal Ideation<br />
Diarrhoea<br />
Cold Sweat<br />
Date:04/15/98ISR Number: 3066252-8Report Type:Expedited (15-DaCompany Report #97094 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Allergic Granulomatous Other Lariam PS ORAL 250.0000 MG<br />
Angiitis<br />
1.0 X PER<br />
WEEK PO<br />
Date:04/15/98ISR Number: 3066253-XReport Type:Expedited (15-DaCompany Report #96103 Age:67 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brain Scan Abnormal Foreign Lariam PS ORAL 250.0000 MG<br />
Delirium<br />
Health<br />
Psychotic Disorder<br />
Professional<br />
Encephalopathy<br />
Mania
Date:04/15/98ISR Number: 3071734-9Report Type:Direct Company Report # Age:64 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Fatigue<br />
Malaise<br />
Night Sweats<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 32
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disturbance In Attention<br />
Confusional State<br />
Blood Pressure Report Source Product Role Manufacturer Route Dose Duration<br />
Fluctuation Lariam PS Roche ORAL 250MGM ; ORAL<br />
Asthenia<br />
;ONCE A WEEK<br />
Paraesthesia Mevacor C<br />
Tenormin<br />
C<br />
Moduretic<br />
C<br />
Date:04/20/98ISR Number: 3065208-9Report Type:Expedited (15-DaCompany Report #97097 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Coma Foreign Lariam PS ORAL 19.3000 MG/KG<br />
Literature<br />
1.O X PER<br />
Health<br />
ODOS ORAL<br />
Professional Pyrimethamine C<br />
Sulfadoxine<br />
C<br />
Date:04/20/98ISR Number: 3066091-8Report Type:Expedited (15-DaCompany Report #97118 Age:5 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rash Morbilliform Foreign Lariam PS ORAL 50.0000 MG<br />
Initial or Prolonged Neurotoxicity Literature 1.0 X PER<br />
Pyrexia Health WEEK ORAL<br />
Drug Interaction Professional Quinine SS INTRAVENOUS 21.0000 MG/KG<br />
Status Epilepticus<br />
DAILY<br />
Convulsion INTRAVENOUS 7 DAY<br />
Date:04/20/98ISR Number: 3069248-5Report Type:Expedited (15-DaCompany Report #97566 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Health Lariam PS ORAL 250 MG<br />
Deafness Professional (TABLET) 1.0<br />
X PER WEEK<br />
Premarin<br />
C<br />
Date:04/20/98ISR Number: 3073073-9Report Type:Direct Company Report # Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia Lariam PS Roche Laboratories 1 PER WEEK<br />
Other Serious<br />
Hyperhidrosis<br />
Anxiety<br />
Disturbance In Attention<br />
Restlessness<br />
Weight Decreased
Eating Disorder<br />
03-Apr-2012 09:37 AM Page: 33
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/20/98ISR Number: 3073074-0Report Type:Direct Company Report # Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Ulcer Lariam PS 1 TABLET A<br />
Hospitalization - Blood Pressure Increased DAY FOR ONE<br />
Initial or Prolonged<br />
WEEK THEN ONE<br />
EVERY WEEK<br />
Date:04/21/98ISR Number: 3074855-XReport Type:Direct Company Report # Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Headache Larium PS ORAL ONE PILL EACH<br />
Balance Disorder<br />
WEEK<br />
Hearing Impaired<br />
Visual Impairment<br />
Date:04/23/98ISR Number: 3071860-4Report Type:Direct Company Report # Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Health Lariam PS ORAL (1) TAB Q/WK<br />
Initial or Prolonged Delusion Professional ORAL 1 DAY<br />
Other Serious Psychotic Disorder Cipro C<br />
Date:04/24/98ISR Number: 3068735-3Report Type:Expedited (15-DaCompany Report #97094 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Allergic Granulomatous Other Lariam PS ORAL 250 MG 1XPER<br />
Angiitis<br />
WEEK<br />
Unknown<br />
C<br />
Date:04/24/98ISR Number: 3068760-2Report Type:Expedited (15-DaCompany Report #97775 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pneumonia Viral Foreign Lariam PS ORAL 250.0000 MG<br />
Initial or Prolonged Pneumonitis Health 1.0 X PER<br />
Alveolitis Allergic Professional WEEK ORAL<br />
Other<br />
Date:04/24/98ISR Number: 3068768-7Report Type:Expedited (15-DaCompany Report #97779 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Foreign Lariam PS ORAL 250 MG 1X PER<br />
Foetus Health WEEK ORAL
Pre-Eclampsia Professional<br />
03-Apr-2012 09:37 AM Page: 34
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/24/98ISR Number: 3068811-5Report Type:Expedited (15-DaCompany Report #83939 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Pre-Eclampsia Foreign Lariam PS ORAL 250.000 MG<br />
Maternal Drugs Affecting Health 1.0 X PER<br />
Foetus Professional WEEK ORAL<br />
Cleft Palate<br />
Date:04/24/98ISR Number: 3072341-4Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paranoia Lariam PS Roche ONCE PER WEEK<br />
Depression<br />
Thinking Abnormal<br />
Dermatitis<br />
Feeling Of Body<br />
Temperature Change<br />
Anxiety<br />
Diarrhoea<br />
Suicidal Ideation<br />
Insomnia<br />
Pyrexia<br />
Date:04/27/98ISR Number: 3069033-4Report Type:Expedited (15-DaCompany Report #83939 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Cleft Palate Foreign Lariam PS ORAL 250 MG 1 X<br />
Maternal Drugs Affecting Health PER WEEK ORAL<br />
Foetus<br />
Professional<br />
Date:04/27/98ISR Number: 3069037-1Report Type:Expedited (15-DaCompany Report #97885 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dry Mouth Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Oedema Mouth Health PER WEEK ORAL<br />
Pharyngeal Oedema Professional Levlen C<br />
Date:04/27/98ISR Number: 3070615-4Report Type:Expedited (15-DaCompany Report #96103 Age:67 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Foreign Lariam PS ORAL 250 MG 1 X<br />
Delirium Health PER WEEK ORAL<br />
Progressive Multifocal Professional<br />
Leukoencephalopathy<br />
Mania
Date:04/27/98ISR Number: 3072501-2Report Type:Direct Company Report # Age:40 YR Gender:Male I/FU:I<br />
Outcome<br />
Disability<br />
Required<br />
03-Apr-2012 09:37 AM Page: 35
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Personality Disorder <strong>Mefloquine</strong> PS ORAL 250MG WEEKLY<br />
Disorientation<br />
PO<br />
Acute Psychosis<br />
Paranoia<br />
Hallucination, Auditory<br />
Visual Impairment<br />
Agitation<br />
Nausea<br />
Disturbance In Attention<br />
Headache<br />
Date:04/29/98ISR Number: 3072953-8Report Type:Direct Company Report # Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fear Health Lariam PS La Roche ORAL 1 TABLET<br />
Other Serious Heat Exhaustion Professional WEEKLY<br />
Paranoia<br />
Insomnia<br />
Nausea<br />
Vomiting<br />
Decreased Appetite<br />
Panic Attack<br />
Alopecia<br />
Palpitations<br />
Date:04/30/98ISR Number: 3074393-4Report Type:Expedited (15-DaCompany Report #98097 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Skull Fracture Foreign Lariam PS ORAL 250 MG 1X PER<br />
Facial Bones Fracture Health WEEK ORAL<br />
Completed Suicide Professional TAB<br />
Aortic Injury<br />
Multiple Injuries<br />
Fall<br />
Date:04/30/98ISR Number: 3074395-8Report Type:Expedited (15-DaCompany Report #98092 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paralysis Foreign Lariam PS ORAL ORAL<br />
Health<br />
Professional
Date:05/04/98ISR Number: 3073979-0Report Type:Expedited (15-DaCompany Report #93422 Age:28 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Rash Pruritic<br />
Insomnia<br />
Hypersensitivity<br />
Swelling<br />
Spider Naevus<br />
03-Apr-2012 09:37 AM Page: 36
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Restlessness<br />
Mental Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Asthma Other Lariam PS ORAL 250 MG 1 X<br />
Nausea<br />
PER WEEK ORAL<br />
Anxiety Hepatitis A Vaccine C<br />
Vision Blurred Albuterol Inhaler C<br />
Headache<br />
Triphasic Birth<br />
Control Pill<br />
C<br />
Date:05/04/98ISR Number: 3073980-7Report Type:Expedited (15-DaCompany Report #96923 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Foreign Lariam PS ORAL ORAL<br />
Amnesia<br />
Health<br />
Brain Neoplasm<br />
Professional<br />
Mental Disorder<br />
Date:05/05/98ISR Number: 3073586-XReport Type:Expedited (15-DaCompany Report #91747 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Foreign Lariam PS ORAL 250 MG 1 X<br />
Foetus Health PER WEEK<br />
Dysmorphism Professional Vaccines Nos C<br />
Date:05/05/98ISR Number: 3073768-7Report Type:Expedited (15-DaCompany Report #98430 Age:74 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Lariam PS ORAL ORAL<br />
Pulmonary Fibrosis<br />
Date:05/05/98ISR Number: 3073808-5Report Type:Expedited (15-DaCompany Report #97885 Age:30 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoaesthesia Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Dry Mouth Health PER WEEK ORAL<br />
Oedema Mouth Professional Levlen C<br />
Pharyngeal Oedema<br />
Liver Function Test<br />
Abnormal<br />
Date:05/12/98ISR Number: 3075378-4Report Type:Direct Company Report # Age:21 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Larium PS Roche 1 DOSE<br />
Initial or Prolonged Abnormal Behaviour<br />
03-Apr-2012 09:37 AM Page: 37
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/12/98ISR Number: 3076411-6Report Type:Expedited (15-DaCompany Report #9905511 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Psychotic Disorder Foreign Depakene PS Abbott ORAL PO<br />
Epilepsy Consumer <strong>Mefloquine</strong> SS ORAL PO<br />
Drug Level Below Carbamazepine C<br />
Therapeutic<br />
Drug Interaction<br />
Abnormal Behaviour<br />
Date:05/12/98ISR Number: 3077372-6Report Type:Expedited (15-DaCompany Report #98566 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thinking Abnormal Foreign Lariam PS Global Development ORAL 250 MG 1 X<br />
Mania Other PER DAY ORAL<br />
Date:05/12/98ISR Number: 3077375-1Report Type:Expedited (15-DaCompany Report #87481 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Health Lariam PS ORAL 250 MG 2 X<br />
Hypertension Professional PER WEEK ORAL<br />
Oedema<br />
Other<br />
Nausea<br />
Sleep Apnoea Syndrome<br />
Overdose<br />
Cough<br />
Eye Irritation<br />
Visual Acuity Reduced<br />
Dizziness<br />
Hypoaesthesia<br />
Bone Pain<br />
Chest Pain<br />
Paraesthesia<br />
Medication Error<br />
Brain Injury<br />
Neck Pain<br />
Nocturia<br />
Dysgeusia<br />
Thinking Abnormal<br />
Vomiting<br />
Upper Respiratory Tract<br />
Infection<br />
Date:05/12/98ISR Number: 3077579-8Report Type:Expedited (15-DaCompany Report #91489 Age:57 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Spinal Cord Injury Foreign Lariam PS ORAL 250 MG 1X PER
Cervical Study WEEK<br />
Cerebral Ischaemia Health Flu Vaccine<br />
Clonic Convulsion Professional (Influenza Virus<br />
Spinal Cord Injury Other Vaccine) C<br />
Thoracic<br />
Flixotide(Fluticason<br />
e Propionate)<br />
C<br />
Salbutamol<br />
(Albuterol)<br />
C<br />
03-Apr-2012 09:37 AM Page: 38
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Prozac (Fluoxetine<br />
Hydrochloride)<br />
Tetracyclin<br />
(Tetracycline)<br />
C<br />
C<br />
Date:05/15/98ISR Number: 3079401-2Report Type:Expedited (15-DaCompany Report #96103 Age:67 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mania Foreign Lariam PS ORAL 250 MG X PER<br />
Encephalopathy Health WEEK ORAL<br />
Delirium<br />
Professional<br />
Psychotic Disorder<br />
Date:05/15/98ISR Number: 3079404-8Report Type:Expedited (15-DaCompany Report #98742 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myocardial Infarction Foreign Lariam PS ORAL 250 MG 1X PER<br />
Initial or Prolonged Health WEEK ORAL<br />
Professional<br />
Date:05/15/98ISR Number: 3079416-4Report Type:Expedited (15-DaCompany Report #89246 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Premature Labour Foreign Lariam PS ORAL 250 MG 1XPER<br />
Intervention to Health WEEK ORAL<br />
Prevent Permanent<br />
Professional<br />
Impairment/Damage<br />
Date:05/15/98ISR Number: 3079420-6Report Type:Expedited (15-DaCompany Report #96729 Age:57 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Clonic Convulsion Foreign Lariam PS ORAL 250MG 1XPER<br />
Other<br />
WEEK ORAL<br />
Influenza Vaccine C<br />
Flixotide<br />
C<br />
Salbutamol<br />
C<br />
Prozac<br />
C<br />
Tetracycline<br />
C<br />
Date:05/15/98ISR Number: 3079450-4Report Type:Expedited (15-DaCompany Report #93562 Age:32 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Foreign Larium PS ORAL 250MG X 1 PER
Initial or Prolonged Systemic Lupus Health WEEK ORAL<br />
Erythematosus<br />
Professional<br />
Pre-Eclampsia<br />
Other<br />
Abortion Spontaneous<br />
03-Apr-2012 09:37 AM Page: 39
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/15/98ISR Number: 3079460-7Report Type:Expedited (15-DaCompany Report #98539 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dysmorphism Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Oligohydramnios Other PER WEEK ORAL<br />
Congenital Anomaly Placental Disorder<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:05/15/98ISR Number: 3079461-9Report Type:Expedited (15-DaCompany Report #97094 Age:66 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oedema Peripheral Other Lariam PS ORAL 250 MG 1 X<br />
Allergic Granulomatous<br />
PER WEEK ORAL<br />
Angiitis Proventil Inhaler C<br />
Skin Mass Aminophylline C<br />
Fosamax<br />
C<br />
Premarin<br />
C<br />
Flovent<br />
C<br />
Vitamin E<br />
C<br />
Fish Oil<br />
C<br />
Calcium<br />
C<br />
Seliran<br />
C<br />
Multivitamins<br />
C<br />
Accolate<br />
C<br />
Vitamin C<br />
C<br />
Date:05/19/98ISR Number: 3081150-1Report Type:Expedited (15-DaCompany Report #83711 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Health Lariam PS ORAL 250 MG 1 X<br />
Visual Impairment Professional PER WEEK ORAL<br />
Anxiety<br />
Schizophrenia<br />
Abnormal Dreams<br />
Psychotic Disorder<br />
Hallucination, Visual<br />
Date:05/19/98ISR Number: 3081153-7Report Type:Expedited (15-DaCompany Report #80215 Age:53 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Company Lariam PS ORAL 250 MG 1 X<br />
Nausea Representative PER WEEK ORAL<br />
Visual Impairment<br />
Blindness
Date:05/19/98ISR Number: 3081447-5Report Type:Expedited (15-DaCompany Report #85335 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abnormal Behaviour Health Lariam PS ORAL 1 DOSE FROM 1<br />
Initial or Prolonged Aggression Professional X PER WEEK<br />
Hallucination<br />
ORAL<br />
Schizophrenia<br />
03-Apr-2012 09:37 AM Page: 40
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/20/98ISR Number: 3080425-XReport Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Visual Impairment Lariam PS 250 MG 1 X<br />
Other Serious Abnormal Dreams WEEK<br />
Mood Altered<br />
Anxiety<br />
Panic Attack<br />
Date:05/20/98ISR Number: 3081830-8Report Type:Expedited (15-DaCompany Report #98794 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haemolysis Foreign Lariam PS ORAL 250 MG 1 X<br />
Liver Function Test Health PER WEEK ORAL<br />
Abnormal<br />
Professional<br />
Platelet Count Decreased<br />
Date:05/22/98ISR Number: 3082507-5Report Type:Expedited (15-DaCompany Report #96767 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Retinal Detachment Health Lariam PS ORAL 250 MG 1XPER<br />
Malaria Professional WEEK ORAL<br />
Intraocular Pressure<br />
Other<br />
Increased<br />
Pyrexia<br />
Eye Pain<br />
Chills<br />
Hyperhidrosis<br />
Date:05/22/98ISR Number: 3082511-7Report Type:Expedited (15-DaCompany Report #88781 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Premature Baby Foreign Lariam PS ORAL 250 MG 1 X<br />
Uterine Haemorrhage Health PER WEEK ORAL<br />
Respiratory Disorder<br />
Professional<br />
Neonatal<br />
Infection<br />
Uterine Injury<br />
Maternal Drugs Affecting<br />
Foetus<br />
Cervical Incompetence<br />
Haematoma<br />
Vaginal Haemorrhage<br />
Date:05/22/98ISR Number: 3082513-0Report Type:Expedited (15-DaCompany Report #88780 Age:29 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Premature Baby Foreign Lariam PS ORAL 250 MG 1 X<br />
Premature Labour Health PER WEEK ORAL<br />
Vaginal Haemorrhage<br />
Professional<br />
Infection<br />
Cervical Incompetence<br />
Haematoma<br />
03-Apr-2012 09:37 AM Page: 41
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/26/98ISR Number: 3084316-XReport Type:Direct Company Report # Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Health <strong>Mefloquine</strong> PS ORAL 1 TAB Q WK ;<br />
Dizziness Professional 250 MG/WK<br />
Feeling Abnormal P.O. 4 WK<br />
Anxiety<br />
Date:05/28/98ISR Number: 3084620-5Report Type:Expedited (15-DaCompany Report #99368 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Infectious Mononucleosis Foreign Lariam PS ORAL 250 MG 1 X<br />
Mouth Ulceration Other PER WEEK ORAL<br />
Post Viral Fatigue<br />
Syndrome<br />
Lymphopenia<br />
Date:06/01/98ISR Number: 3087636-8Report Type:Expedited (15-DaCompany Report #85972 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Foreign Lariam PS ORAL 250 MG 1 X<br />
Foetus Health PER WEEK ORAL<br />
Jaundice Neonatal Professional Cabergoline C<br />
Date:06/01/98ISR Number: 3088034-3Report Type:Expedited (15-DaCompany Report #98742 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myocardial Infarction Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Health PER WEEK ORAL<br />
Professional<br />
Date:06/01/98ISR Number: 3088035-5Report Type:Expedited (15-DaCompany Report #99181 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Hypersensitivity Health PER WEEK ORAL<br />
Asthma<br />
Professional<br />
Date:06/01/98ISR Number: 3088675-3Report Type:Expedited (15-DaCompany Report #80215 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam PS ORAL 250 MG 1 X<br />
Visual Acuity Reduced<br />
PER WEEK ORAL
Nightmare Estrogen C<br />
03-Apr-2012 09:37 AM Page: 42
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/02/98ISR Number: 3088297-4Report Type:Expedited (15-DaCompany Report #98097 Age:37 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Depression Foreign Lariam PS ORAL 250 MG 1X PER<br />
Completed Suicide Health WEEK ORAL<br />
Professional<br />
Other<br />
Date:06/04/98ISR Number: 3089762-6Report Type:Expedited (15-DaCompany Report #99833 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Arthralgia Foreign Lariam PS ORAL 250 MG 1 X<br />
Myalgia Other PER WEEK ORAL<br />
Date:06/04/98ISR Number: 3090175-1Report Type:Expedited (15-DaCompany Report #68408 Age:25 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Spinal Fracture Health Lariam PS ORAL 250 MG 1 X<br />
Hallucination Professional PER WEEK ORAL<br />
Paraplegia<br />
Other<br />
Mental Disorder<br />
Panic Attack<br />
Fall<br />
Date:06/04/98ISR Number: 3090177-5Report Type:Expedited (15-DaCompany Report #99753 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Other Lariam PS ORAL 250 MG 1 X<br />
Agitation<br />
PER WEEK ORAL<br />
Suicide Attempt<br />
Suicidal Ideation<br />
Paranoia<br />
Flushing<br />
Restlessness<br />
Diarrhoea<br />
Date:06/05/98ISR Number: 3179164-6Report Type:Periodic Company Report #95701 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam Tablets<br />
Nausea<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK 21 DAY
Date:06/08/98ISR Number: 3159668-2Report Type:Periodic Company Report #81017 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Alopecia Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 43
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Synthroid<br />
C<br />
Date:06/08/98ISR Number: 3159671-2Report Type:Periodic Company Report #81018 Age:75 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pruritus Consumer Lariam Tablets<br />
Dizziness Health (<strong>Mefloquine</strong><br />
Alopecia Professional Hydrochloride)<br />
Abnormal Dreams 250.000 Mg PS ORAL 250.000 MG<br />
Eye Irritation<br />
1.0 X PER<br />
WEEK (ORAL)<br />
Dyazide<br />
C<br />
Date:06/08/98ISR Number: 3159674-8Report Type:Periodic Company Report #82001 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Thirst<br />
(<strong>Mefloquine</strong><br />
Eye Irritation<br />
Hydrochloride)<br />
Hypoaesthesia Oral 250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Zestril<br />
C<br />
Date:06/08/98ISR Number: 3159676-1Report Type:Periodic Company Report #82176 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam Tablets<br />
Heart Rate Increased<br />
(<strong>Mefloquine</strong><br />
Nervousness<br />
Hydrochloride)<br />
Paranoia 250.000 Mg PS ORAL 250.000 MG<br />
Hallucinations, Mixed<br />
1.0 X PER<br />
Anxiety<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159680-3Report Type:Periodic Company Report #82334 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Phosphorus Health Lariam Tablets<br />
Increased Professional (<strong>Mefloquine</strong><br />
Gamma-Glutamyltransferase<br />
Hydrochloride)<br />
Increased 250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL
Date:06/08/98ISR Number: 3159682-7Report Type:Periodic Company Report #82521 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
03-Apr-2012 09:37 AM Page: 44
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159683-9Report Type:Periodic Company Report #82632 Age:72 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pain In Extremity Health Lariam Tablets<br />
Pain Professional (<strong>Mefloquine</strong><br />
Arthralgia<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159684-0Report Type:Periodic Company Report #82726 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation Other Lariam Tablets<br />
Tremor<br />
(<strong>Mefloquine</strong><br />
Anxiety<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159685-2Report Type:Periodic Company Report #83053 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Other Lariam Tablets<br />
Judgement Impaired<br />
(<strong>Mefloquine</strong><br />
Panic Disorder<br />
Hydrochloride)<br />
Confusional State 250.000 Mg PS ORAL 1.0000 DOSE<br />
Crying<br />
FORM 1.0 X<br />
PER WEEK ORAL<br />
Date:06/08/98ISR Number: 3159686-4Report Type:Periodic Company Report #83231 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam Tablets<br />
Sedation<br />
(<strong>Mefloquine</strong><br />
Memory Impairment<br />
Hydrochloride)<br />
Vision Blurred 250.000 Mg PS ORAL 250.000 MG<br />
Fatigue<br />
1.0 X PER<br />
Dizziness<br />
WEEK ORAL<br />
Tachycardia<br />
Visual Impairment
Date:06/08/98ISR Number: 3159687-6Report Type:Periodic Company Report #83412 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urinary Incontinence Other Lariam Tablets<br />
Muscle Spasms<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
03-Apr-2012 09:37 AM Page: 45
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Yellow Fever Vaccine C<br />
Rabies Vaccine C<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159688-8Report Type:Periodic Company Report #83622 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gynaecomastia Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Sporanox<br />
(Itraconazole)<br />
250.000 Mg SS ORAL 3.0 X PER MON<br />
ORAL<br />
Vitamin E<br />
C<br />
Vitamin D<br />
C<br />
Vitamin B Complex C<br />
Date:06/08/98ISR Number: 3159690-6Report Type:Periodic Company Report #83624 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Acuity Reduced Consumer Lariam Tablets<br />
Alopecia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Premarin<br />
C<br />
Synthroid<br />
C<br />
Date:06/08/98ISR Number: 3159691-8Report Type:Periodic Company Report #83790 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Impairment Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.O X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159692-XReport Type:Periodic Company Report #83947 Age:58 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hyporeflexia Consumer Lariam Tablets<br />
Paraesthesia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Mevacor (Lovastatin) SS ORAL 20.0000 MG<br />
03-Apr-2012 09:37 AM Page: 46
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Premarin<br />
Provera<br />
C<br />
C<br />
DAILY ORAL<br />
Date:06/08/98ISR Number: 3159694-3Report Type:Periodic Company Report #83953 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Alopecia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.O X PER<br />
WEEK ORAL<br />
Maxidex<br />
C<br />
Date:06/08/98ISR Number: 3159695-5Report Type:Periodic Company Report #84120 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159697-9Report Type:Periodic Company Report #84292 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Consumer Lariam Tablets<br />
Palpitations<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 PER WEEK<br />
ORAL<br />
Date:06/08/98ISR Number: 3159699-2Report Type:Periodic Company Report #84462 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Health Lariam Tablets (<br />
Palpitations Professional <strong>Mefloquine</strong><br />
Flashback<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL
Date:06/08/98ISR Number: 3159701-8Report Type:Periodic Company Report #84488 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Other Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
03-Apr-2012 09:37 AM Page: 47
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159702-XReport Type:Periodic Company Report #84805 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Exfoliative Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL ORAL<br />
Date:06/08/98ISR Number: 3159703-1Report Type:Periodic Company Report #84870 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Diarrhoea<br />
(<strong>Mefloquine</strong><br />
Asthenia Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Estrogen (Estrogens<br />
Nos)<br />
C<br />
Date:06/08/98ISR Number: 3159705-5Report Type:Periodic Company Report #84928 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Sectral (Acebutolol) C<br />
Date:06/08/98ISR Number: 3159707-9Report Type:Periodic Company Report #84953 Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Increased Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159709-2Report Type:Periodic Company Report #84991 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Dyspnoea Other Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 48
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3159710-9Report Type:Periodic Company Report #85215 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Headache<br />
(<strong>Mefloquine</strong><br />
Dizziness Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159711-0Report Type:Periodic Company Report #85238 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS OCCLUSIVE<br />
DRESSING<br />
250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159712-2Report Type:Periodic Company Report #85358 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Systolic Consumer Lariam Tablets<br />
Decreased<br />
(<strong>Mefloquine</strong><br />
Eye Irritation<br />
Hydrochloride)<br />
Nausea 250.000 Mg PS ORAL 250.0000 MG<br />
Insomnia<br />
1.0 X PER<br />
Conjunctivitis Allergic<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159714-6Report Type:Periodic Company Report #85373 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159716-XReport Type:Periodic Company Report #85494 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psoriasis Other Lariam Tablets<br />
Abnormal Dreams<br />
(<strong>Mefloquine</strong><br />
Pruritus Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 49
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3159719-5Report Type:Periodic Company Report #85538 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Metrorrhagia Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Synthroid<br />
(Levothyroxine<br />
Sodium)<br />
C<br />
Date:06/08/98ISR Number: 3159721-3Report Type:Periodic Company Report #85602 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ecchymosis Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Cipro (Ciprofloxacin<br />
Hydrochloride) C<br />
Date:06/08/98ISR Number: 3159722-5Report Type:Periodic Company Report #85669 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Other Lariam Tablets<br />
Decreased Appetite<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 0.5000 DOSE<br />
FORM 1.0 X<br />
PER WEEK ORAL<br />
Date:06/08/98ISR Number: 3159725-0Report Type:Periodic Company Report #85699 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oligomenorrhoea Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Hepatitis Vaccine<br />
(Hepatitis Vaccine) C<br />
Typhoid Vaccine<br />
(Typhoid Vaccines) C<br />
Polio Vaccination<br />
(Poliomyelitis<br />
Vaccines)<br />
C
Amoxicillin<br />
(Amoxicillin)<br />
Anaprox (Naproxen<br />
Sodium)<br />
C<br />
C<br />
03-Apr-2012 09:37 AM Page: 50
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3159726-2Report Type:Periodic Company Report #85807 Age:3 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urticaria Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159727-4Report Type:Periodic Company Report #85892 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
Contraceptives<br />
(Contraceptives)<br />
C<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159729-8Report Type:Periodic Company Report #86520 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chills Health Lariam Tablets<br />
Pyrexia Professional (<strong>Mefloquine</strong><br />
Myalgia Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159730-4Report Type:Periodic Company Report #86747 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Disorder Other Lariam Tablets<br />
Aggression<br />
(<strong>Mefloquine</strong><br />
Abnormal Dreams Hydrochloride) PS ORAL 250.0000 MG<br />
Crying<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3159731-6Report Type:Periodic Company Report #86858 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Alopecia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 51
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3159732-8Report Type:Periodic Company Report #87231 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pruritus Health Lariam Tablets<br />
Rash Maculo-Papular Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Ortho-Novum (Ethinyl<br />
Estradiol Or<br />
Menstranol/<br />
Norethindrone) C<br />
Date:06/08/98ISR Number: 3159734-1Report Type:Periodic Company Report #87256 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Flushing Consumer Lariam Tablets<br />
Insomnia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 125.0000 MG<br />
2.0 X PER<br />
WEEK ORAL 49 DAY<br />
Zantac (Ranitidine) C<br />
Vivelle Transdermal<br />
System (Estradiol) C<br />
Date:06/08/98ISR Number: 3160383-XReport Type:Periodic Company Report #87379 Age:68 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Consumer Lariam Tablets<br />
Insomnia Health (<strong>Mefloquine</strong><br />
Dizziness Professional Hydrochloride)<br />
Nausea 250.000 Mg PS ORAL 250.000 MG<br />
Nasal Congestion<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3160387-7Report Type:Periodic Company Report #87446 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Iron Increased Consumer Lariam Tablets<br />
Hepatomegaly<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL
Date:06/08/98ISR Number: 3160390-7Report Type:Periodic Company Report #87479 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Health Lariam Tablets<br />
Nausea Professional (<strong>Mefloquine</strong><br />
Hair Disorder<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 PER WEEK<br />
03-Apr-2012 09:37 AM Page: 52
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
ORAL<br />
Date:06/08/98ISR Number: 3160394-4Report Type:Periodic Company Report #87727 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blindness Other Lariam Tablets<br />
Deafness<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/08/98ISR Number: 3160396-8Report Type:Periodic Company Report #87777 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspepsia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 1.0 PER<br />
WEEK ORAL<br />
Demulen<br />
C<br />
Date:06/08/98ISR Number: 3160398-1Report Type:Periodic Company Report #87993 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pollakiuria Health Lariam Tablets<br />
Thirst Professional (<strong>Mefloquine</strong><br />
Hydrochloride) 250.<br />
000 Mg PS ORAL 250.000 MG<br />
1.0 PER WEEK<br />
ORAL<br />
Date:06/08/98ISR Number: 3160401-9Report Type:Periodic Company Report #87999 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3160403-2Report Type:Periodic Company Report #88273 Age:65 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Health Lariam Tablets
Professional (<strong>Mefloquine</strong><br />
Hydrochloride) 250.<br />
000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 53
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3160405-6Report Type:Periodic Company Report #88393 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Consumer Lariam Tablets<br />
Dizziness<br />
(<strong>Mefloquine</strong><br />
Lymphadenopathy<br />
Hydrochloride)<br />
Irritability 250.00 Mg PS ORAL 250.000 MG<br />
Nausea<br />
1.0 PER WEEK<br />
Headache<br />
ORAL<br />
Cipro<br />
C<br />
Nordette<br />
C<br />
Date:06/08/98ISR Number: 3160406-8Report Type:Periodic Company Report #89194 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250.<br />
000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3160409-3Report Type:Periodic Company Report #89203 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspepsia Consumer Lariam Tablets<br />
Nightmare<br />
(<strong>Mefloquine</strong><br />
Insomnia<br />
Hydrochloride)<br />
Headache 250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3160410-XReport Type:Periodic Company Report #89287 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3160412-3Report Type:Periodic Company Report #89617 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Vomiting Consumer Lariam Tablets<br />
Pyrexia<br />
(<strong>Mefloquine</strong><br />
Panic Attack<br />
Hydrochloride)<br />
Decreased Appetite 250.000 Mg PS ORAL 250.00 MG 1.0<br />
Dizziness<br />
X PER WEEK<br />
Disturbance In Attention<br />
ORAL<br />
Ortho Novum<br />
C<br />
Hepatitis Vaccine C<br />
03-Apr-2012 09:37 AM Page: 54
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Valium<br />
General Anesthesia<br />
Lortab<br />
Erytromycin<br />
C<br />
C<br />
C<br />
C<br />
Date:06/08/98ISR Number: 3160413-5Report Type:Periodic Company Report #90262 Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Estrace<br />
C<br />
Prilosec<br />
C<br />
Date:06/08/98ISR Number: 3175254-2Report Type:Periodic Company Report #90313 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Health Lariam Tablets<br />
Insomnia Professional (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3175260-8Report Type:Periodic Company Report #90406 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Disorder Literature Lariam Tablets<br />
Anxiety Consumer (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Synthroid<br />
C<br />
Date:06/08/98ISR Number: 3175262-1Report Type:Periodic Company Report #91164 Age:69 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viral Infection Consumer Lariam Tablets<br />
Dyspepsia<br />
(<strong>Mefloquine</strong><br />
Tachycardia<br />
Hydrochloride)<br />
Tremor 250.00 Mg PS ORAL 250.000 MG
Pyrexia 1.0 X PER<br />
WEEK, ORAL<br />
Glucophage<br />
C<br />
Glyburide<br />
C<br />
Flu Shot<br />
C<br />
Lotensin<br />
C<br />
03-Apr-2012 09:37 AM Page: 55
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3175264-5Report Type:Periodic Company Report #91344 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Consumer Lariam Tablets<br />
Diarrhoea<br />
(<strong>Mefloquine</strong><br />
Crying<br />
Hydrochloride)<br />
Dysphoria 250.00 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Date:06/08/98ISR Number: 3175267-0Report Type:Periodic Company Report #91417 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Other Lariam Tablets<br />
Pressure Of Speech<br />
(<strong>Mefloquine</strong><br />
Restlessness<br />
Hydrochloride)<br />
Paranoia 250.00 Mg PS ORAL 250.00 MG 1.0<br />
X PER WEEK,<br />
ORAL<br />
Date:06/08/98ISR Number: 3175269-4Report Type:Periodic Company Report #91527 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Menstruation Irregular Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.0000 MG<br />
1.0 PER WEEK,<br />
ORAL<br />
Vitamin C<br />
C<br />
Date:06/08/98ISR Number: 3175271-2Report Type:Periodic Company Report #91582 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Consumer Lariam Tablets<br />
Heart Rate Increased<br />
(<strong>Mefloquine</strong><br />
Insomnia Hydrochloride) 250<br />
Anxiety Mg PS ORAL 250 MG 1.0 X<br />
PER WEEK,<br />
ORAL<br />
Ortho-Novum 7/7/7 C<br />
Date:06/08/98ISR Number: 3175273-6Report Type:Periodic Company Report #91731 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Restlessness Other Lariam Tablets<br />
Nausea<br />
(<strong>Mefloquine</strong><br />
Diarrhoea<br />
Hydrochloride)<br />
Dyspepsia 250.00 Mg PS ORAL 250.00 MG 1.0<br />
Flatulence<br />
X PER WEEK,<br />
Dermatitis<br />
ORAL<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 56
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3175275-XReport Type:Periodic Company Report #92041 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Prothrombin Time Other Lariam Tablets<br />
Prolonged<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.00 MG 1.0<br />
PER WEEK,<br />
ORAL<br />
Coumadin<br />
C<br />
Date:06/08/98ISR Number: 3175277-3Report Type:Periodic Company Report #92512 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Diarrhoea<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.00 MG 1.0<br />
X PER WEEK,<br />
ORAL<br />
Date:06/08/98ISR Number: 3175279-7Report Type:Periodic Company Report #92525 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Health Lariam Tablets<br />
Insomnia Professional (<strong>Mefloquine</strong><br />
Visual Acuity Reduced<br />
Hydrochloride)<br />
Paraesthesia 250.00 Mg PS ORAL 250.00 MG 1.0<br />
Thinking Abnormal<br />
X PER ODOS,<br />
Hallucination<br />
ORAL<br />
Date:06/08/98ISR Number: 3175282-7Report Type:Periodic Company Report #92697 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Health Lariam Tablets<br />
Nausea Professional (<strong>Mefloquine</strong><br />
Emotional Disorder<br />
Hydrochloride)<br />
Amoebic Dysentery 250.00 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Flagyl<br />
C<br />
Date:06/08/98ISR Number: 3175284-0Report Type:Periodic Company Report #92713 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Anxiety Consumer Lariam Tablets<br />
Insomnia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.00 MG 1.0<br />
X PER WEEK<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 57
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3175286-4Report Type:Periodic Company Report #92738 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Date:06/08/98ISR Number: 3175288-8Report Type:Periodic Company Report #92825 Age:13 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vomiting Other Lariam Tablets<br />
Agitation<br />
(<strong>Mefloquine</strong><br />
Dizziness<br />
Hydrochloride)<br />
Photosensitivity Reaction 250.00 Mg PS ORAL 250.000 MG<br />
Insomnia<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3175290-6Report Type:Periodic Company Report #93510 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Arthralgia Consumer Lariam Tablets<br />
Myalgia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Date:06/08/98ISR Number: 3175291-8Report Type:Periodic Company Report #93511 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chromaturia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.00 MG 1.0<br />
X PER WEEK,<br />
ORAL<br />
Ortho-Novum 1/35 C<br />
Date:06/08/98ISR Number: 3175292-XReport Type:Periodic Company Report #93513 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Laryngitis Consumer Lariam Tablets
Health (<strong>Mefloquine</strong><br />
Professional<br />
Hydrochloride)<br />
250.00 Mg PS ORAL 250.00 MG 1.0<br />
X PER WEEK<br />
ORAL<br />
Amoxycillin<br />
C<br />
03-Apr-2012 09:37 AM Page: 58
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3175294-3Report Type:Periodic Company Report #93921 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Syncope Other Lariam Tablets<br />
Abdominal Pain<br />
(<strong>Mefloquine</strong><br />
Dizziness<br />
Hydrochloride)<br />
Vomiting 250.00 Mg PS ORAL 250.00 MG 1.0<br />
X PER ODOS,<br />
ORAL<br />
Date:06/08/98ISR Number: 3175296-7Report Type:Periodic Company Report #93923 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam Tablets<br />
Anxiety Professional (<strong>Mefloquine</strong><br />
Nervousness Hydrochloride) 250<br />
Mg PS ORAL 250.00 MG 1.0<br />
X PER WEEK<br />
ORAL<br />
Date:06/08/98ISR Number: 3179135-XReport Type:Periodic Company Report #76339 Age:54 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Neurogenic Bladder Health Lariam Tablets<br />
Pollakiuria Professional (<strong>Mefloquine</strong><br />
Micturition Urgency<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179137-3Report Type:Periodic Company Report #77417 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Typhoid Vaccine<br />
(Thphoid Vaccines) C<br />
Dramamine<br />
(Dimenhydrinate) C<br />
Measles, Mumps, &<br />
Rubella Vaccine<br />
(Measles Virus<br />
Vaccine Live/Mumps<br />
Virus Vaccine<br />
C
Date:06/08/98ISR Number: 3179139-7Report Type:Periodic Company Report #78055 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Health Lariam Tablets<br />
Hypoaesthesia Oral Professional (<strong>Mefloquine</strong><br />
Headache<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
03-Apr-2012 09:37 AM Page: 59
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179140-3Report Type:Periodic Company Report #80073 Age:10 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sedation Other Lariam Tablets<br />
Petit Mal Epilepsy<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Date:06/08/98ISR Number: 3179141-5Report Type:Periodic Company Report #80172 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Prostatic Specific Health Lariam Tablets<br />
Antigen Increased Professional (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Hytrin (Terazosin<br />
Hydrochloride) C<br />
Date:06/08/98ISR Number: 3179143-9Report Type:Periodic Company Report #80199 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Other Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Accupril (Quinapril<br />
Hydrochloride) C<br />
Prempro (Estrogens,<br />
Conjugated/Medroxypr<br />
ogesterone Acetate) C<br />
Date:06/08/98ISR Number: 3179145-2Report Type:Periodic Company Report #80216 Age:65 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Abdominal Pain Upper<br />
(<strong>Mefloquine</strong><br />
Muscle Spasms<br />
Hydrochloride)
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Klonopin<br />
(Clonazepam)<br />
C<br />
Ritalin<br />
(Methylphenidate<br />
Hydrochloride) C<br />
03-Apr-2012 09:37 AM Page: 60
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Inderal (Propranolol<br />
Hydrochloride)<br />
Paxil (Paroxetine)<br />
C<br />
C<br />
Date:06/08/98ISR Number: 3179146-4Report Type:Periodic Company Report #96623 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Alopecia Consumer Lariam Tablets<br />
Depression Other (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179147-6Report Type:Periodic Company Report #97072 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Upper Consumer Lariam Tablets<br />
Blood Cholesterol<br />
(<strong>Mefloquine</strong><br />
Increased Hidrochloride) 250.<br />
000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL 42 DAY<br />
Lipitor<br />
(Atorvastatin<br />
Calcium) SS ORAL ORAL<br />
Hepatitis Injection<br />
(Hepatitis Injection<br />
Nos)<br />
C<br />
Calcium (Calcium<br />
Nos)<br />
C<br />
Magnesium (Magnesium<br />
Nos)<br />
C<br />
Glucosamine Sulfate<br />
(Glucosamine<br />
Sulfate)<br />
C<br />
Vitamin E (Vitamin<br />
E) C<br />
Multivitamins<br />
(Multivitamin Nos) C<br />
Vitamin C (Ascorbid<br />
Acid)<br />
C<br />
Date:06/08/98ISR Number: 3179148-8Report Type:Periodic Company Report #97208 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Health Lariam Tablets
Professional (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER DAY<br />
ORAL<br />
Kaolin (Kaolin) C<br />
03-Apr-2012 09:37 AM Page: 61
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179149-XReport Type:Periodic Company Report #97647 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Consumer Lariam Tablets<br />
Pruritus<br />
(<strong>Mefloquine</strong><br />
Flushing<br />
Hydrochloride)<br />
Rhinorrhoea 250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Premarin (Estrogens,<br />
Conjugated)<br />
C<br />
Date:06/08/98ISR Number: 3179150-6Report Type:Periodic Company Report #97681 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Thinking Abnormal<br />
(<strong>Mefloquine</strong><br />
Fatigue<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179151-8Report Type:Periodic Company Report #63751 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Consumer Lariam Tablets<br />
Initial or Prolonged Nausea (<strong>Mefloquine</strong><br />
Disability Vertigo Hydrochloride) PS ORAL ORAL<br />
Visual Impairment<br />
Depression<br />
Decreased Appetite<br />
Anxiety<br />
Date:06/08/98ISR Number: 3179152-XReport Type:Periodic Company Report #67732 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucinations, Mixed Health Lariam Tablets<br />
Initial or Prolonged Professional (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179153-1Report Type:Periodic Company Report #54965 Age:51 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urticaria Consumer Lariam Tablets<br />
Abnormal Dreams User Facility (<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Cotrim<br />
03-Apr-2012 09:37 AM Page: 62
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
(Sulfamethoxazole/Tr<br />
imethoprim)<br />
C<br />
Date:06/08/98ISR Number: 3179154-3Report Type:Periodic Company Report #62060 Age:64 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179155-5Report Type:Periodic Company Report #74666 Age:53 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Estrogens Nos<br />
(Estrogens Nos) C<br />
Date:06/08/98ISR Number: 3179156-7Report Type:Periodic Company Report #77480 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nervousness Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179157-9Report Type:Periodic Company Report #78054 Age:34 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Exfoliative Rash Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Metakelfin<br />
(Pyrimethamine/Sulfa<br />
lene) SS ORAL ORAL
Date:06/08/98ISR Number: 3179158-0Report Type:Periodic Company Report #81052 Age:75 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pruritus Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
03-Apr-2012 09:37 AM Page: 63
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179159-2Report Type:Periodic Company Report #94231 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Consumer Lariam Tablets<br />
Dermatitis<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK<br />
Date:06/08/98ISR Number: 3179160-9Report Type:Periodic Company Report #94304 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vestibular Disorder Health Lariam Tablets<br />
Dizziness Professional (<strong>Mefloquine</strong><br />
Tinnitus Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER DAY<br />
Date:06/08/98ISR Number: 3179161-0Report Type:Periodic Company Report #94510 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK<br />
Date:06/08/98ISR Number: 3179162-2Report Type:Periodic Company Report #95230 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 M<br />
1.0 X PER<br />
WEEK<br />
Amlodipine<br />
(Amlodipine<br />
Besylate)<br />
C<br />
Cozaar (Losartan<br />
Potassium)<br />
C
Date:06/08/98ISR Number: 3179163-4Report Type:Periodic Company Report #95668 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL<br />
03-Apr-2012 09:37 AM Page: 64
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179165-8Report Type:Periodic Company Report #95731 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Impairment Health Lariam Tablets<br />
Dizziness Professional (<strong>Mefloquine</strong><br />
Depression Hydrochloride) PS ORAL 250.0000 MG<br />
Insomnia<br />
1.0 X PER<br />
Gait Disturbance<br />
WEEK<br />
Cough<br />
Confusional State<br />
Date:06/08/98ISR Number: 3179166-XReport Type:Periodic Company Report #96253 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Flushing Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK<br />
Synthroid<br />
(Levothyroxine<br />
Sodium)<br />
C<br />
Date:06/08/98ISR Number: 3179167-1Report Type:Periodic Company Report #96289 Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Palpitations Health Lariam Tablets<br />
Dizziness Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK<br />
Date:06/08/98ISR Number: 3179168-3Report Type:Periodic Company Report #96334 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam Tablets<br />
Anxiety<br />
(<strong>Mefloquine</strong><br />
Depression Hydrochloride) PS ORAL 250.000 MG<br />
Disturbance In Attention<br />
1.0 X PER<br />
Chills<br />
WEEK<br />
Insomnia Paxil (Paroxetine) C<br />
Confusional State<br />
Flushing<br />
Dizziness
Date:06/08/98ISR Number: 3179169-5Report Type:Periodic Company Report #96379 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 8 DAY<br />
03-Apr-2012 09:37 AM Page: 65
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179170-1Report Type:Periodic Company Report #96418 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Cold Other Lariam Tablets<br />
Flushing<br />
(<strong>Mefloquine</strong><br />
Nausea Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK<br />
Date:06/08/98ISR Number: 3179171-3Report Type:Periodic Company Report #96622 Age:51 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Spasms Other Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 PER WEEK<br />
Date:06/08/98ISR Number: 3179172-5Report Type:Periodic Company Report #80314 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscular Weakness Other Lariam Tablets<br />
Syncope<br />
(<strong>Mefloquine</strong><br />
Pallor<br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL<br />
Date:06/08/98ISR Number: 3179174-9Report Type:Periodic Company Report #80475 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride)<br />
250.000 Mg PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK, ORAL 49 DAY<br />
Date:06/08/98ISR Number: 3179175-0Report Type:Periodic Company Report #80485 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
Nightmare Health (<strong>Mefloquine</strong><br />
Neuropathy Peripheral Professional Hydrochloride) PS ORAL 250.0000 MG<br />
Confusional State<br />
1.0 X PER<br />
Palpitations<br />
WEEK, ORAL
Birth Control Pills<br />
(Oral Contraceptive<br />
Nos)<br />
C<br />
03-Apr-2012 09:37 AM Page: 66
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179968-XReport Type:Periodic Company Report #77585 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Other Lariam Tablets<br />
Malaria<br />
(<strong>Mefloquine</strong><br />
Nausea Hydrochloride) PS ORAL 250.0000 MG<br />
Dysgeusia<br />
1.O X PER<br />
Sunburn<br />
WEEK ORAL<br />
Headache<br />
Abdominal Pain<br />
Diarrhoea<br />
Sensory Disturbance<br />
Paraesthesia<br />
Date:06/08/98ISR Number: 3179969-1Report Type:Periodic Company Report #80918 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Encephalopathy Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/08/98ISR Number: 3179970-8Report Type:Periodic Company Report #80968 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Epilepsy Consumer Lariam Tablets<br />
Diarrhoea<br />
(<strong>Mefloquine</strong><br />
Chills Hydrochloride) PS ORAL 250.0000 MG<br />
Glossodynia<br />
1.0 X PER<br />
Loss Of Consciousness<br />
WEEK ORAL<br />
Hepatic Function Abnormal<br />
Imipramine<br />
Headache<br />
(Imipramine<br />
Pyrexia Hydrochloride) C<br />
Platelet Count Decreased<br />
Premarin (Estrogens,<br />
Conjugated)<br />
C<br />
Date:06/08/98ISR Number: 3179971-XReport Type:Periodic Company Report #81879 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Health Lariam Tablets<br />
Initial or Prolonged Productive Cough Professional (<strong>Mefloquine</strong><br />
Aspartate Hydrochloride) PS ORAL 250.000 MG<br />
Aminotransferase<br />
1.0 X PER<br />
Increased<br />
WEEK ORAL<br />
Chills<br />
Pulmonary Congestion
Date:06/08/98ISR Number: 3179972-1Report Type:Periodic Company Report #82256 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Consumer Lariam Tablets<br />
Initial or Prolonged<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
03-Apr-2012 09:37 AM Page: 67
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Robitussin<br />
(Guaifenesin)<br />
Premarin (Estrogens,<br />
Conjugated)<br />
C<br />
C<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179973-3Report Type:Periodic Company Report #83032 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER DAY<br />
ORAL<br />
Date:06/08/98ISR Number: 3179974-5Report Type:Periodic Company Report #84444 Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Other Lariam Tablets<br />
Initial or Prolonged Diarrhoea (<strong>Mefloquine</strong><br />
Pyrexia Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Luvox (Fluvoxamine) SS<br />
Date:06/08/98ISR Number: 3179975-7Report Type:Periodic Company Report #85156 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Consumer Lariam Tablets<br />
Erythema Multiforme Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Oruvail (Ketoprofen) SS ORAL ORAL<br />
Fosamax (Alendronate<br />
Sodium)<br />
C<br />
Mevacor (Lovastatin) C<br />
Date:06/08/98ISR Number: 3179976-9Report Type:Periodic Company Report #86443 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Neuropathy Peripheral Health Lariam Tablets<br />
Paraesthesia Professional (<strong>Mefloquine</strong><br />
Pain Hydrochloride) PS ORAL 250.0000 MG<br />
Tonic Convulsion<br />
1.0 X PER<br />
Hypoaesthesia<br />
WEEK ORAL
Propulsid<br />
(Cisapride)<br />
Proscar<br />
(Finasteride)<br />
C<br />
C<br />
03-Apr-2012 09:37 AM Page: 68
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179977-0Report Type:Periodic Company Report #86526 Age:15 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Health Lariam Tablets<br />
Initial or Prolonged Major Depression Professional (<strong>Mefloquine</strong><br />
Mood Altered Other Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179978-2Report Type:Periodic Company Report #90808 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Consumer Lariam Tablets<br />
Aggression Health (<strong>Mefloquine</strong><br />
Confusional State Professional Hydrochloride) PS ORAL 250.0000 MG<br />
Tinnitus<br />
1.0 X PER<br />
Fatigue<br />
WEEK ORAL<br />
Depression<br />
Nightmare<br />
Hallucination<br />
Date:06/08/98ISR Number: 3179979-4Report Type:Periodic Company Report #92025 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179980-0Report Type:Periodic Company Report #92026 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER DAY<br />
ORAL<br />
Ambien (Zolpidem<br />
Tartrate)<br />
C<br />
Norvasc (Amlodipine<br />
Besylate)<br />
C<br />
Alcohol (Alcohol) C<br />
Date:06/08/98ISR Number: 3179981-2Report Type:Periodic Company Report #92537 Age:46 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Increased Consumer Lariam Tablets<br />
Visual Impairment<br />
(<strong>Mefloquine</strong><br />
Hyperventilation Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 69
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179982-4Report Type:Periodic Company Report #94951 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chills Consumer Lariam Tablets<br />
Initial or Prolonged Chest Pain Health (<strong>Mefloquine</strong><br />
Diarrhoea Professional Hydrochloride) PS ORAL 250.0000 MG<br />
Nausea<br />
1.0 X PER<br />
Malaise<br />
WEEK ORAL<br />
Anxiety<br />
Palpitations<br />
Date:06/08/98ISR Number: 3179983-6Report Type:Periodic Company Report #96220 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mania Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/08/98ISR Number: 3179984-8Report Type:Periodic Company Report #74928 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Acuity Reduced Consumer Lariam Tablets<br />
Coordination Abnormal<br />
(<strong>Mefloquine</strong><br />
Tremor Hydrochloride) PS ORAL 250.0000 MG<br />
Muscle Contractions<br />
1.0 X PER<br />
Involuntary<br />
WEEK ORAL<br />
Date:06/08/98ISR Number: 3179985-XReport Type:Periodic Company Report #75973 Age:9 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Arthralgia Consumer Lariam Tablets<br />
Upper Respiratory Tract<br />
(<strong>Mefloquine</strong><br />
Infection Hydrochloride) PS ORAL 187.5000 MG<br />
Dermatitis<br />
1.0 X PER<br />
WEEK ORAL<br />
Imodium (Loperamide<br />
Hydrochloride) C<br />
Augmentin<br />
(Amoxicillin/Clavula<br />
nate Potassium) C<br />
Date:06/08/98ISR Number: 3179986-1Report Type:Periodic Company Report #76016 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Consumer Lariam Tablets
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 70
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/98ISR Number: 3179987-3Report Type:Periodic Company Report #76173 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Health Lariam Tablets<br />
Pruritus Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250.0000 MG<br />
1.0 X PER<br />
WEEK ORAL<br />
Date:06/09/98ISR Number: 3091276-4Report Type:Expedited (15-DaCompany Report #99832 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pneumonitis Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Other PER ONE DOSE<br />
ORAL<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
Date:06/09/98ISR Number: 3091279-XReport Type:Expedited (15-DaCompany Report #99823 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maculopathy Foreign Lariam PS ORAL 250 MG 1 X<br />
Other<br />
PER WEEK ORAL<br />
Scopolamine<br />
(Scopolamine<br />
Hydrobromide)<br />
C<br />
Date:06/09/98ISR Number: 3091862-1Report Type:Expedited (15-DaCompany Report #99981 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pneumonitis Health Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Professional PER WEEK ORAL<br />
Date:06/09/98ISR Number: 3091863-3Report Type:Expedited (15-DaCompany Report #98430 Age:72 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pulmonary Fibrosis Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Dyspnoea Health PER WEEK ORAL<br />
Professional Naproxen C<br />
Date:06/09/98ISR Number: 3091876-1Report Type:Expedited (15-DaCompany Report #99913 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Agitation Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Nightmare Other PER WEEK ORAL<br />
Paranoia Cannabis C<br />
Influenza Like Illness<br />
03-Apr-2012 09:37 AM Page: 71
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/10/98ISR Number: 3092795-7Report Type:Direct Company Report # Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Memory Impairment Larium PS AS PRESCRIBED<br />
Hospitalization - Decompression Sickness<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:06/11/98ISR Number: 3092547-8Report Type:Expedited (15-DaCompany Report #88780 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Premature Baby Foreign Lariam PS ORAL 250 MG 1 X<br />
Cervical Incompetence Health PER WEEK ORAL<br />
Haematoma<br />
Professional<br />
Vaginal Infection<br />
Vaginal Haemorrhage<br />
Premature Labour<br />
Date:06/16/98ISR Number: 3094153-8Report Type:Direct Company Report # Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disorientation Lariam PS ONCE A WEEK 6 WK<br />
Initial or Prolonged Panic Attack<br />
Required<br />
Suicidal Ideation<br />
Intervention to<br />
Myocardial Infarction<br />
Prevent Permanent Headache<br />
Impairment/Damage Depression<br />
Date:06/16/98ISR Number: 3094934-0Report Type:Expedited (15-DaCompany Report #94569 Age:12 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Prophylaxis Health Lariam PS ORAL ORAL<br />
Anxiety<br />
Professional<br />
Panic Attack<br />
Emotional Disorder<br />
Obsessive-Compulsive<br />
Disorder<br />
Date:06/16/98ISR Number: 3094936-4Report Type:Expedited (15-DaCompany Report #65435 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Consumer Lariam PS ORAL 250MG 1XPER
Apathy Health WEEK ORAL<br />
Hallucination<br />
Professional<br />
Mental Impairment<br />
Pain<br />
Lethargy<br />
03-Apr-2012 09:37 AM Page: 72
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/19/98ISR Number: 3096992-6Report Type:Expedited (15-DaCompany Report #98742 Age:39 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myocardial Infarction Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Health PER WEEK<br />
Professional<br />
Date:06/19/98ISR Number: 3097002-7Report Type:Expedited (15-DaCompany Report #100462 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Carpal Tunnel Syndrome Foreign Lariam PS ORAL 250 MG 1 X<br />
Arthralgia Consumer PER WEEK ORAL<br />
Myalgia<br />
Date:06/23/98ISR Number: 3097532-8Report Type:Expedited (15-DaCompany Report #98486 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hearing Impaired Foreign Lariam PS ORAL 250 MG 1 X<br />
Audiogram Abnormal Health PER WEEK<br />
Vestibular Disorder Professional ORAL<br />
Dizziness<br />
Date:06/29/98ISR Number: 3100448-1Report Type:Expedited (15-DaCompany Report #101184 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Foreign Lariam PS ORAL 250 MG 1 X<br />
Health<br />
PER WEEK ORAL<br />
Professional<br />
Date:06/29/98ISR Number: 3102153-4Report Type:Periodic Company Report #7395898 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Interaction Health Norvir PS ORAL 300.000 MG PO<br />
Vision Blurred Professional BID<br />
Dizziness Company Lariam SS<br />
Representative Zovirax C<br />
Other Dt4 C<br />
Ddi<br />
C<br />
Lariam<br />
C<br />
Date:07/02/98ISR Number: 3101371-9Report Type:Expedited (15-DaCompany Report #101337 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Viith Nerve Paralysis Foreign Lariam PS ORAL 250 MG X 1<br />
Health<br />
PER WEEK<br />
Professional<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 73
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/02/98ISR Number: 3101373-2Report Type:Expedited (15-DaCompany Report #96761 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nephrolithiasis Consumer Lariam PS ORAL 250 MG 1 X<br />
Haemangioma Of Liver<br />
PER WEEK ORAL<br />
Infection Parasitic<br />
Tetanus Booster<br />
(Tetanus Toxoid) C<br />
Polio Booster<br />
(Poliomyelitis<br />
Vaccines)<br />
C<br />
Hepatitis A Vaccine C<br />
Hepatitis B Vaccine C<br />
Date:07/02/98ISR Number: 3101779-1Report Type:Expedited (15-DaCompany Report #98097 Age:37 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Foreign Lariam PS ORAL 250 MG 1 X<br />
Depression Health PER WEEK ORAL<br />
Abnormal Behaviour<br />
Professional<br />
Other<br />
Date:07/07/98ISR Number: 3102623-9Report Type:Expedited (15-DaCompany Report #101725 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation Foreign Lariam PS ORAL 6 DOSE FORM<br />
Dyslexia Health DAILY ORAL<br />
Headache Professional Chloroquine<br />
Aphasia (Choroquine) C<br />
Dizziness<br />
Toxicity To Various<br />
Agents<br />
Date:07/13/98ISR Number: 3104477-3Report Type:Expedited (15-DaCompany Report #74171 Age:47 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paresis Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Dizziness Health PER ONE DOSE<br />
Required Ileus Paralytic Professional ORAL<br />
Intervention to Guillain-Barre Syndrome Other Epaxal SS INTRAMUSCULAR .5 ML 1 X PER<br />
Prevent Permanent Gait Disturbance ONE DOSE<br />
Impairment/Damage Abdominal Distension INTRAMUSCULAR<br />
Insomnia Arilvax SS SUBCUTANEOUS .5 ML 1 X PER<br />
Gastrointestinal Disorder<br />
ONE DOSE<br />
SUBCUTANEOUS
Date:07/13/98ISR Number: 3105539-7Report Type:Expedited (15-DaCompany Report #98430 Age:72 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Foreign Lariam PS ORAL 250 MG 1XPER<br />
Initial or Prolonged Pulmonary Fibrosis Health WEEK ORAL<br />
Professional Naproxen C<br />
03-Apr-2012 09:37 AM Page: 74
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/17/98ISR Number: 3106187-5Report Type:Expedited (15-DaCompany Report #102436 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Foreign Lariam PS<br />
Fatigue<br />
Consumer<br />
Chest Pain<br />
Pain In Extremity<br />
Dyspnoea<br />
Date:07/23/98ISR Number: 3109359-9Report Type:Direct Company Report #4-756-039-6 Age:67 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Accolate PS<br />
Arthralgia Lariam SS<br />
Allergic Granulomatous Dyazide C<br />
Angiitis Prednisone C<br />
Gait Disturbance<br />
Myalgia<br />
Granuloma<br />
Date:07/24/98ISR Number: 3109393-9Report Type:Expedited (15-DaCompany Report #96767 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Retinal Detachment Health Lariam PS ORAL 250 MG 1 X<br />
Eye Pain Professional PER WEEK<br />
Hyperhidrosis Other ,ORAL<br />
Malaria<br />
Chills<br />
Intraocular Pressure<br />
Increased<br />
Pyrexia<br />
Date:07/29/98ISR Number: 3110717-7Report Type:Expedited (15-DaCompany Report #100462 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Myalgia Foreign Lariam PS ORAL 250 MG 1 X<br />
Arthralgia Consumer PER WEEK<br />
Paraesthesia<br />
Carpal Tunnel Syndrome<br />
Date:07/29/98ISR Number: 3113067-8Report Type:Direct Company Report # Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Rash Vesicular Lariam PS ORAL PO 1/WEEK 1 WK<br />
Intervention to<br />
Scar
Prevent Permanent<br />
Impairment/Damage<br />
Insomnia<br />
Haemorrhage<br />
Erythema<br />
Wound Secretion<br />
Dermatitis Exfoliative<br />
Pruritus<br />
Discomfort<br />
03-Apr-2012 09:37 AM Page: 75
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/29/98ISR Number: 3123285-0Report Type:Direct Company Report # Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Larium PS Roche ORAL 250 MG ORAL<br />
Initial or Prolonged Anxiety ONCE PER WEEK<br />
Restlessness<br />
Date:08/03/98ISR Number: 3112339-0Report Type:Expedited (15-DaCompany Report #103417 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Foreign Lariam PS ORAL 250 MG DAILY<br />
Gastrointestinal Consumer ORAL<br />
Hypermotility<br />
Restlessness<br />
Muscular Weakness<br />
Alopecia<br />
Decreased Appetite<br />
Periorbital Oedema<br />
Anxiety<br />
Feeling Abnormal<br />
Oedema<br />
Psychotic Disorder<br />
Abnormal Dreams<br />
Memory Impairment<br />
Lethargy<br />
Date:08/14/98ISR Number: 3117013-2Report Type:Direct Company Report # Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness <strong>Mefloquine</strong> PS ORAL 250 MG PO Q<br />
Diarrhoea<br />
1T WEEK<br />
Vomiting Lariam SS<br />
Vertigo<br />
Anxiety<br />
Date:08/18/98ISR Number: 3118491-5Report Type:Expedited (15-DaCompany Report #101725 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Foreign Lariam PS ORAL 6 DOSE FORM<br />
Headache Health DAILY ORAL<br />
Disorientation Professional Chloroquine<br />
Dyslexia (Chloroquine) C<br />
Aphasia<br />
Toxicity To Various<br />
Agents
Date:08/19/98ISR Number: 3118885-8Report Type:Expedited (15-DaCompany Report #90892 Age:28 YR Gender:Female I/FU:I<br />
Outcome<br />
Congenital Anomaly<br />
PT<br />
Feeding Disorder Neonatal<br />
Maternal Drugs Affecting<br />
Foetus<br />
Congenital Jaw<br />
03-Apr-2012 09:37 AM Page: 76
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Malformation<br />
Dysmorphism<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250 MG 1 X<br />
Health<br />
PER WEEK ORAL<br />
Professional<br />
Date:08/19/98ISR Number: 3118889-5Report Type:Expedited (15-DaCompany Report #104255 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Congenital Foreign Lariam PS ORAL 250 MG 1 X<br />
Musculoskeletal Anomaly Health PER WEEK ORAL<br />
Cholestasis<br />
Professional<br />
Dysmorphism<br />
Maternal Drugs Affecting<br />
Foetus<br />
Premature Labour<br />
Liver Function Test<br />
Abnormal<br />
Date:09/02/98ISR Number: 3124912-4Report Type:Expedited (15-DaCompany Report #104882 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Anxiety Consumer PER WEEK<br />
Dizziness<br />
ORAL<br />
Tachycardia<br />
Arrhythmia<br />
Convulsion<br />
Cardiovascular Disorder<br />
Balance Disorder<br />
Restlessness<br />
Circulatory Collapse<br />
Ventricular Extrasystoles<br />
Asthenia<br />
Date:09/02/98ISR Number: 3124913-6Report Type:Expedited (15-DaCompany Report #104935 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam PS ORAL 250 MG 1 X<br />
Foetus Health PER WEEK ORAL<br />
Cleft Palate<br />
Professional<br />
Date:09/04/98ISR Number: 3125941-7Report Type:Expedited (15-DaCompany Report #105053 Age:27 YR Gender:Female I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Tachycardia<br />
Panic Attack<br />
Nausea<br />
Dizziness<br />
Bradycardia<br />
Insomnia<br />
Palpitations<br />
03-Apr-2012 09:37 AM Page: 77
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250 MG 1 X<br />
Other<br />
PER WEEK;<br />
ORAL<br />
Date:09/04/98ISR Number: 3126058-8Report Type:Expedited (15-DaCompany Report #104663 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Osteoarthritis Health Lariam PS ORAL 250 MG 1 X<br />
Disturbance In Attention Professional PER WEEK ORAL<br />
Joint Swelling<br />
Typhoid Vaccine<br />
Arthralgia (Typhoid Vaccines) SS INTRAMUSCULAR 1 X PER ONE<br />
Fatigue<br />
DOSE<br />
Serum Sickness<br />
INTRAMUSCULAR<br />
Anxiety<br />
Date:09/10/98ISR Number: 3128393-6Report Type:Direct Company Report # Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams <strong>Mefloquine</strong> PS ORAL 250MG/WEEK<br />
Depression<br />
ORAL<br />
Anxiety<br />
Hearing Impaired<br />
Aggression<br />
Fatigue<br />
Hallucination<br />
Date:09/14/98ISR Number: 3128291-8Report Type:Expedited (15-DaCompany Report #105284 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Foreign Lariam PS ORAL 250 MG, 750<br />
Hospitalization - Depression Health MG 1 X PER<br />
Initial or Prolonged Restlessness Professional ONE DOSE ORAL<br />
Insomnia<br />
Tranxilium<br />
(Colorazepate<br />
Dipotassium)<br />
C<br />
Atosil (Promethazine<br />
Hydrochloride) C<br />
Resochin<br />
(Chloroquine<br />
Phosphate)<br />
C<br />
Date:09/25/98ISR Number: 3135453-2Report Type:Expedited (15-DaCompany Report #105895 Age:27 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Arrested Labour Foreign Lariam PS ORAL 250 MG 1 X<br />
Intervention to Maternal Drugs Affecting Health PER WEEK ORAL<br />
Prevent Permanent Foetus Professional Clarityne<br />
Impairment/Damage (Loratadine) C<br />
Engerix B (Hepatitis<br />
B Virus Vaccine<br />
03-Apr-2012 09:37 AM Page: 78
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Inactivated)<br />
Atarax (Hydroxyzine<br />
Hydrochloride)<br />
Typhim (Typhoif<br />
Vaccines)<br />
Dpt (Diphteria<br />
Toxoid/Pertussis<br />
Vaccine/Tetaus<br />
Toxoid)<br />
C<br />
C<br />
C<br />
C<br />
Date:09/30/98ISR Number: 3136796-9Report Type:Expedited (15-DaCompany Report #106188 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Fatigue Foreign Lariam PS ORAL 250 MG 1 X<br />
Hospitalization - Haemolytic Anaemia Other PER WEEK ORAL<br />
Initial or Prolonged Influenza Like Illness Aspirin (Aspirin) C<br />
Anxiety<br />
Grippostad C<br />
(Acetaminophen/Ascor<br />
bic<br />
Acid/Caffeine/Chlorp<br />
heniramine Maleate) C<br />
Date:10/02/98ISR Number: 3260324-0Report Type:Periodic Company Report #8-97337-022L Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Erythema Multiforme Health Oruvail PS ORAL ORAL<br />
Professional Lariam SS ORAL 250 MG WEEKLY<br />
ORAL<br />
Mevacor<br />
C<br />
Cortisone Injection C<br />
Fosamax<br />
C<br />
Date:10/13/98ISR Number: 3142010-0Report Type:Expedited (15-DaCompany Report #106318 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dystonia Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Panic Attack Other PER WEEK ORAL<br />
Grand Mal Convulsion<br />
Date:10/13/98ISR Number: 3142011-2Report Type:Expedited (15-DaCompany Report #106675 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ovarian Cancer Health Lariam PS ORAL 250 MG 1 X<br />
Professional<br />
PER WEEK ORAL
Date:10/23/98ISR Number: 3145988-4Report Type:Expedited (15-DaCompany Report #106970 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Mania Foreign Lariam PS ORAL 250 MG 1 X<br />
Completed Suicide Other PER WEEK ORAL<br />
Hypomania<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 79
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/23/98ISR Number: 3145989-6Report Type:Expedited (15-DaCompany Report #107015 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dissociative Identity Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Disorder Other PER WEEK ORAL<br />
Hallucination<br />
Panic Attack<br />
Date:11/02/98ISR Number: 3152111-9Report Type:Expedited (15-DaCompany Report #107663 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Foreign Lariam PS ORAL ORAL<br />
Health<br />
Professional<br />
Date:11/05/98ISR Number: 3152322-2Report Type:Expedited (15-DaCompany Report #92444 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Maternal Drugs Affecting Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Foetus Health PER WEEK ORAL<br />
Congenital Anomaly Premature Baby Professional Adepal SS ORAL 1 DOSE FORM 1<br />
Foetal Growth Restriction Other X PER DAY<br />
Small For Dates Baby<br />
ORAL<br />
Stamaril SS SUBCUTANEOUS 1 DOSE FORM 1<br />
X PER ONE<br />
DOSE<br />
SUBCUTANEOUS<br />
Havrix SS INTRAMUSCULAR 1440 UNIT/ML<br />
1 X PER ONE<br />
DOSE<br />
INTRAMUSCULAR<br />
Hepatitis SS INTRAMUSCULAR 1 X PER ONE<br />
DOSE<br />
INTRAMUSCULAR<br />
Date:11/10/98ISR Number: 3155903-5Report Type:Expedited (15-DaCompany Report #107875 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam PS ORAL 250 MG 1 X<br />
Foetus Other PER WEEK ORAL<br />
Benign Congenital<br />
Hypotonia<br />
Congenital Anomaly<br />
Microcephaly<br />
Thalassaemia Alpha<br />
Mental Retardation<br />
Dysmorphism
Strabismus<br />
Developmental Delay<br />
03-Apr-2012 09:37 AM Page: 80
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/10/98ISR Number: 3155959-XReport Type:Expedited (15-DaCompany Report #107840 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Colitis Foreign Lariam PS ORAL TABLETS; ORAL<br />
Initial or Prolonged<br />
Health<br />
Professional<br />
Date:11/12/98ISR Number: 3156945-6Report Type:Expedited (15-DaCompany Report #107872 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fall Foreign Lariam PS ORAL 250MG 1 X PER<br />
Circulatory Collapse Other WEEK ORAL<br />
Fatigue<br />
Grand Mal Convulsion<br />
Loss Of Consciousness<br />
Anhedonia<br />
Epilepsy<br />
Lethargy<br />
Date:11/13/98ISR Number: 3157862-8Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Crying Consumer Larium PS<br />
Depression<br />
Emotional Disorder<br />
Date:11/17/98ISR Number: 3158657-1Report Type:Expedited (15-DaCompany Report #107663 Age:61 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Foreign Lariam PS ORAL 250 MG 1 X<br />
Health<br />
PER WEEK ORAL<br />
Professional<br />
Date:11/20/98ISR Number: 3160902-3Report Type:Expedited (15-DaCompany Report #65435 Age:33 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT<br />
Apathy<br />
Pollakiuria<br />
Haematuria<br />
Anxiety<br />
Cholecystitis<br />
Syncope<br />
Constipation<br />
Nausea<br />
Cough
Lethargy<br />
Night Sweats<br />
Nightmare<br />
Chills<br />
Back Pain<br />
Micturition Urgency<br />
Culture Urine Positive<br />
Condition Aggravated<br />
03-Apr-2012 09:37 AM Page: 81
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Headache<br />
Mental Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucinations, Mixed Consumer Lariam PS ORAL 250 MG 1 X<br />
Pain Health PER WEEK ORAL 14 DAY<br />
Calculus Urinary Professional Lorazepam C<br />
Other Anaprox Ds C<br />
Motrin<br />
C<br />
Toradol<br />
C<br />
Wellbutrin<br />
C<br />
Zovirax<br />
C<br />
Date:11/24/98ISR Number: 3161905-5Report Type:Direct Company Report # Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Lariam PS 250MG,1 PER<br />
Hallucination WEEK FOR 5<br />
Dermatitis WEEKS 5 WK<br />
Convulsion<br />
Flushing<br />
Anxiety<br />
Insomnia<br />
Date:11/30/98ISR Number: 3164083-1Report Type:Expedited (15-DaCompany Report #108878 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cerebral Infarction Foreign Lariam PS ORAL 250 MG 1XPER<br />
Hemianopia Other WEEK ORAL<br />
Hepatitis A Vaccine C<br />
Yellow Fever Vaccine C<br />
Mengivac<br />
C<br />
Typhim<br />
C<br />
Date:11/30/98ISR Number: 3164100-9Report Type:Expedited (15-DaCompany Report #108916 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Foreign Lariam PS ORAL 250 MG 1 X<br />
Other<br />
PER WEEK ORAL<br />
Date:11/30/98ISR Number: 3164195-2Report Type:Expedited (15-DaCompany Report #81879 Age:23 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Chills<br />
Tongue Disorder<br />
Suicidal Ideation
Medication Error<br />
Blood Cholesterol<br />
Increased<br />
Bipolar I Disorder<br />
Pulmonary Congestion<br />
Schizophreniform Disorder<br />
Culture Stool Positive<br />
Hepatic Enzyme Increased<br />
03-Apr-2012 09:37 AM Page: 82
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Aspartate<br />
Aminotransferase<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Psychotic Disorder Health Lariam PS 250 MG 1 X<br />
Oral Candidiasis Professional PER WEEK ORAL<br />
Cough Typhoid Vaccine C<br />
Diphtheria And<br />
Tetaus Vaccine C<br />
Yellow Fever Vaccine C<br />
Date:12/01/98ISR Number: 3164881-4Report Type:Expedited (15-DaCompany Report #107840 Age:30 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - White Blood Cell Count Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Increased Health PER WEEK ORAL<br />
Red Blood Cell<br />
Professional<br />
Sedimentation Rate<br />
Increased<br />
Thrombocytosis<br />
Colitis Ulcerative<br />
Diarrhoea Haemorrhagic<br />
Date:12/01/98ISR Number: 3164986-8Report Type:Expedited (15-DaCompany Report #108638 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Breast Mass Foreign Lariam PS 250 MG 1 X<br />
Breast Cancer Female Consumer PER WEEK<br />
Premature Labour Other OTHER<br />
Date:12/03/98ISR Number: 3171304-8Report Type:Expedited (15-DaCompany Report #98676 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Psychotic Disorder Foreign Lariam PS ORAL 250 MG 1 X<br />
Hospitalization - Paresis Consumer PER ONE DOSE<br />
Initial or Prolonged Cholera Health ORAL<br />
Headache<br />
Professional<br />
Drug Level Above<br />
Therapeutic<br />
Paraesthesia<br />
Hallucination<br />
Hypoaesthesia<br />
Suicidal Ideation<br />
Restlessness<br />
Date:12/03/98ISR Number: 3171305-XReport Type:Expedited (15-DaCompany Report #109425 Age:46 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Optic Neuritis Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Visual Impairment Other PER WEEK ORAL<br />
Papilloedema Typhim Vi SS INTRAMUSCULAR 1 X PER ONE<br />
DOSE<br />
INTRAMUSCULAR<br />
Engerix B SS INTRAMUSCULAR 1 X PER ONE<br />
03-Apr-2012 09:37 AM Page: 83
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
DOSE<br />
INTRAMUSCULAR<br />
Profenid SS ORAL 1 X PER PRN<br />
ORAL<br />
Propofan SS ORAL 1 X PER PRN<br />
ORAL<br />
Sanmigran<br />
C<br />
Date:12/03/98ISR Number: 3171306-1Report Type:Expedited (15-DaCompany Report #109197 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ejection Fraction Foreign Lariam PS ORAL 25 MG/KG ORAL<br />
Abnormal Health Lariam SS ORAL 250 MG 1X PER<br />
Tachycardia Professional WEEK ORAL<br />
Medication Error<br />
Dyspnoea<br />
Date:12/08/98ISR Number: 3168310-6Report Type:Expedited (15-DaCompany Report #104882 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Restlessness Consumer PER WEEK ORAL<br />
Feeling Abnormal<br />
Health<br />
Asthenia<br />
Professional<br />
Paraesthesia<br />
Circulatory Collapse<br />
Balance Disorder<br />
Dizziness<br />
Confusional State<br />
Tachycardia<br />
Anxiety<br />
Cardiovascular Disorder<br />
Ventricular Extrasystoles<br />
Arrhythmia<br />
Date:12/08/98ISR Number: 3168484-7Report Type:Expedited (15-DaCompany Report #109778 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypersensitivity Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Other PER WEEK ORAL<br />
Microgynon (Ethinyl<br />
Estradiol/Levonorges<br />
trel)<br />
C<br />
Date:12/08/98ISR Number: 3168488-4Report Type:Expedited (15-DaCompany Report #109038 Age:48 YR Gender:Female I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Influenza Like Illness<br />
Palpitations<br />
Urticaria<br />
Pyrexia<br />
Chills<br />
Anxiety<br />
Lung Consolidation<br />
03-Apr-2012 09:37 AM Page: 84
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Q Fever<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS ORAL 250 MG 1 X<br />
Health<br />
PER WEEK ORAL<br />
Professional<br />
Date:12/08/98ISR Number: 3168721-9Report Type:Expedited (15-DaCompany Report #108541 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperpyrexia Foreign Lariam PS ORAL 250 MG 1XPER<br />
Initial or Prolonged Gastroenteritis Shigella Study WEEK ORAL<br />
White Blood Cell Count Health Orlistat SS ORAL 3XPER DAY<br />
Increased Professional ORAL<br />
Date:12/10/98ISR Number: 3168961-9Report Type:Direct Company Report # Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Lariam PS ORAL 1 PILL PER<br />
Diarrhoea<br />
WEEK<br />
Malaise<br />
Date:12/10/98ISR Number: 3169049-3Report Type:Direct Company Report # Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Weight Decreased Larium 250mg<br />
Initial or Prolonged Hallucination (<strong>Mefloquine</strong>) PS Roche Rh 674414 ORAL ORAL 250MG 1T<br />
Disability Hyperhidrosis TAB WEEKLY<br />
Required Delusion Benztropinue C<br />
Intervention to Bipolar I Disorder Imxiete C<br />
Prevent Permanent Panic Attack Haloperidol C<br />
Impairment/Damage Convulsion<br />
Date:12/10/98ISR Number: 3169225-XReport Type:Expedited (15-DaCompany Report #8-98334-066A Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Papilloedema Foreign Profenid<br />
Initial or Prolonged Optic Neuritis Health (Ketoprofen) PS ORAL AS NEEDED<br />
Visual Impairment Professional ORAL YR<br />
Hepatitis B Vaccine<br />
Injection SS INJECTABLE<br />
<strong>Mefloquine</strong> Tablet SS ORAL ONE TABLET<br />
WEEKLY; ORAL<br />
Propavan<br />
(Propiomazine
Maleate) SS AS NEEDED YR<br />
Typhoid Vaccine<br />
Injection SS ONE<br />
INJECTION;<br />
INJECTABLE<br />
Cafeine +Paracetamol<br />
+Chlorphenamine<br />
Maleate<br />
03-Apr-2012 09:37 AM Page: 85
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
+Dextropropoxyphene<br />
+Carbasalate Calcium C<br />
Date:12/10/98ISR Number: 3169279-0Report Type:Expedited (15-DaCompany Report #108731 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Atrial Fibrillation Foreign Lariam PS ORAL 250MG 1 X PER<br />
Arrhythmia Health WEEK ORAL<br />
Professional<br />
Date:12/10/98ISR Number: 3169280-7Report Type:Expedited (15-DaCompany Report #96761 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Liver Disorder Consumer Lariam PS ORAL 250MG 1 X PER<br />
Haemangioma Of Liver<br />
WEEK ORAL<br />
Nephrolithiasis Tetanus Booster C<br />
Infection Parasitic Polio Booster C<br />
Hepatitis A Vaccine C<br />
Hepatitis B Vaccine C<br />
Date:12/14/98ISR Number: 3170021-8Report Type:Expedited (15-DaCompany Report #B0062044 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Decreased Appetite Foreign Zovirax Tablet PS ORAL ORAL<br />
Initial or Prolonged Vision Blurred <strong>Mefloquine</strong><br />
Depression<br />
(Formulation<br />
Vertigo Unknown) SS ORAL ORAL<br />
Asthenia Pristinamycin C<br />
Gait Disturbance<br />
Abdominal Pain<br />
Nausea<br />
Malaise<br />
Date:12/14/98ISR Number: 3170131-5Report Type:Expedited (15-DaCompany Report #110216 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychomotor Hyperactivity Foreign Lariam<br />
PS ORAL 250 MG 1XPER<br />
Initial or Prolonged Muscle Rigidity Other WEEK ORAL<br />
Psychotic Disorder<br />
Speech Disorder<br />
Depersonalisation<br />
Insomnia<br />
Screaming<br />
Crying<br />
Nightmare
Sedation<br />
Confusional State<br />
Delirium<br />
03-Apr-2012 09:37 AM Page: 86
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/14/98ISR Number: 3170215-1Report Type:Expedited (15-DaCompany Report #109968 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Decreased Appetite Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Vision Blurred Other PER WEEK ORAL<br />
Depression Zovirax SS ORAL 800 MG DAILY<br />
Nausea<br />
ORAL<br />
Asthenia Pyostacine C<br />
Vertigo<br />
Gait Disturbance<br />
Abdominal Pain<br />
Fall<br />
Date:12/14/98ISR Number: 3170217-5Report Type:Expedited (15-DaCompany Report #110362 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depersonalisation Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Dizziness Other PER WEEK ORAL<br />
Date:12/14/98ISR Number: 3170236-9Report Type:Expedited (15-DaCompany Report #104407 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Discomfort Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Lethargy Health Hydrochloride) PS ORAL 250 MG 1 X<br />
Pain Professional PER WEEK ORAL<br />
Loss Of Libido<br />
Restlessness<br />
Anxiety<br />
Abdominal Pain<br />
Panic Attack<br />
Date:12/14/98ISR Number: 3170525-8Report Type:Expedited (15-DaCompany Report #104882 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Paraesthesia Consumer PER WEEK ORAL<br />
Tachycardia<br />
Health<br />
Confusional State<br />
Professional<br />
Condition Aggravated<br />
Convulsion<br />
Dizziness<br />
Depersonalisation<br />
Balance Disorder<br />
Cardiovascular Disorder<br />
Ventricular Extrasystoles<br />
Circulatory Collapse<br />
Restlessness
Anxiety<br />
Panic Attack<br />
Arrhythmia<br />
03-Apr-2012 09:37 AM Page: 87
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/16/98ISR Number: 3170982-7Report Type:Expedited (15-DaCompany Report #110115 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Abscess Other PER ONE DOSE<br />
Nausea<br />
ORAL<br />
Abdominal Pain<br />
C-Reactive Protein<br />
Increased<br />
Dysuria<br />
Diverticulum Intestinal<br />
Diverticulitis<br />
Pyrexia<br />
White Blood Cell Count<br />
Increased<br />
Diarrhoea<br />
Date:12/16/98ISR Number: 3170984-0Report Type:Expedited (15-DaCompany Report #106675 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ovarian Cancer Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 X<br />
PER WEEK ORAL<br />
Date:12/16/98ISR Number: 3170987-6Report Type:Expedited (15-DaCompany Report #110313 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Loss Of Consciousness Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Grand Mal Convulsion Literature PER WEEK ORAL 21 DAY<br />
Atrial Fibrillation<br />
Health<br />
Blood Cholesterol<br />
Professional<br />
Increased<br />
Urinary Incontinence<br />
Heart Rate Irregular<br />
Date:12/24/98ISR Number: 3174151-6Report Type:Expedited (15-DaCompany Report #58978 Age:31 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Heart Rate Increased<br />
Vertigo<br />
Psychotic Disorder<br />
Feeling Abnormal<br />
Visual Acuity Reduced<br />
Fatigue<br />
Circulatory Collapse
Asthenia<br />
Nervous System Disorder<br />
Nystagmus<br />
Drug Level Above<br />
Therapeutic<br />
Dyspnoea<br />
Amnesia<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 88
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disturbance In Attention<br />
Head Injury<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer<br />
Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1XPER<br />
WEEK ORAL<br />
Norflex<br />
C<br />
Date:12/29/98ISR Number: 3176456-1Report Type:Expedited (15-DaCompany Report #102527 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Apallic Syndrome Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Electroencephalogram Health PER WEEK ORAL<br />
Abnormal Professional Chinin C<br />
Nightmare<br />
Other<br />
Amnesia<br />
Dystonia<br />
Depressed Level Of<br />
Consciousness<br />
Drug Effect Decreased<br />
Restlessness<br />
Pyrexia<br />
Asthenia<br />
Thrombocytopenia<br />
Disturbance In Attention<br />
Sinusitis<br />
Malaria<br />
Nervous System Disorder<br />
Date:12/30/98ISR Number: 3176659-6Report Type:Expedited (15-DaCompany Report #108731 Age:58 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Condition Aggravated Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Atrial Fibrillation Health PER WEEK ORAL<br />
Malaise Professional Tetanus Vaccine<br />
Tachycardia (Tetanus Toxoid) C<br />
Arrhythmia<br />
Immunoglobulin<br />
(Globulin, Immune) C<br />
Polio Vaccine<br />
(Poliomyelitis<br />
Vaccines)<br />
C<br />
Date:12/30/98ISR Number: 3176664-XReport Type:Expedited (15-DaCompany Report #95713 Age:29 YR Gender:Female I/FU:I<br />
Outcome<br />
PT
Hospitalization -<br />
Initial or Prolonged<br />
Congenital Anomaly<br />
White Blood Cell Count<br />
Increased<br />
Congenital Anomaly<br />
Blood Glucose Decreased<br />
Hyperbilirubinaemia<br />
Neonatal<br />
Foetal Haemoglobin<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 89
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Platelet Count Decreased<br />
Talipes<br />
Pyrexia Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Hip Deformity Foreign Lariam PS ORAL 250 MG 1 X<br />
Maternal Drugs Affecting Health PER WEEK ORAL<br />
Foetus<br />
Professional<br />
Hypoglycaemia<br />
Type 1 Diabetes Mellitus<br />
Blood Bilirubin Increased<br />
Date:12/30/98ISR Number: 3176807-8Report Type:Expedited (15-DaCompany Report #107634 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Health Lariam PS ORAL 250 MG 1 X<br />
Ear Pain Professional PER WEEK ORAL<br />
Hemiparesis<br />
Multiple Sclerosis<br />
Eustachian Tube<br />
Obstruction<br />
Paraesthesia<br />
Date:01/08/99ISR Number: 3178806-9Report Type:Expedited (15-DaCompany Report #111360 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Missed Foreign Lariam PS ORAL 250 MG 1 X<br />
Health<br />
PER WEEK ORAL<br />
Professional Clotrimazol C<br />
Date:01/08/99ISR Number: 3178807-0Report Type:Expedited (15-DaCompany Report #111442 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Neuropathy Peripheral Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Headache Other PER DOSE,<br />
Nightmare<br />
ORAL<br />
Dysgeusia<br />
Date:01/08/99ISR Number: 3178853-7Report Type:Expedited (15-DaCompany Report #109968 Age:27 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Asthenia Other Hydrochloride) PS ORAL 250 1 X PER<br />
Abdominal Pain<br />
WEEK ORAL<br />
Vertigo<br />
Zovirax (Acyclovir)<br />
Fall 200 Mg SS ORAL 800 MG DAILY<br />
Decreased Appetite<br />
ORAL
Depression Pyostacine<br />
Gait Disturbance (Pristinamycin) C<br />
Vision Blurred<br />
03-Apr-2012 09:37 AM Page: 90
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/08/99ISR Number: 3179344-XReport Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Lariam PS 1 PILL PER<br />
Tremor<br />
WEEK<br />
Headache<br />
Chills<br />
Date:01/11/99ISR Number: 3179642-XReport Type:Direct Company Report # Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Convulsion Lariam PS Hoffman Laroche 250 MG, I<br />
Insomnia<br />
TABLET PER<br />
Nausea WEEK FOR 10<br />
Vision Blurred<br />
WEEKS<br />
Paraesthesia<br />
Photophobia<br />
Fear<br />
Mania<br />
Psychotic Disorder<br />
Toxicity To Various<br />
Agents<br />
Feeling Of Body<br />
Temperature Change<br />
Fatigue<br />
Paranoia<br />
Dyspnoea<br />
Headache<br />
Decreased Appetite<br />
Dizziness<br />
Date:01/13/99ISR Number: 3180240-2Report Type:Direct Company Report # Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Consumer Lariam PS 1 TABLET PER<br />
Irritability<br />
WEEK<br />
Mood Altered<br />
Insomnia<br />
Palpitations<br />
Dizziness<br />
Date:01/13/99ISR Number: 3180261-XReport Type:Direct Company Report # Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Loss Of Consciousness <strong>Mefloquine</strong> PS ORAL 250MG /WK PO<br />
Partial Seizures<br />
Disorientation
Date:01/13/99ISR Number: 3180629-1Report Type:Expedited (15-DaCompany Report #93243 Age:58 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Thrombocytopenia<br />
White Blood Cell Count<br />
03-Apr-2012 09:37 AM Page: 91
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Decreased<br />
Liver Function Test<br />
Abnormal<br />
Thrombocytosis Report Source Product Role Manufacturer Route Dose Duration<br />
Malaria Foreign Lariam PS ORAL 1500 MG DAILY<br />
Health<br />
ORAL<br />
Professional<br />
Other<br />
Date:01/14/99ISR Number: 3180688-6Report Type:Expedited (15-DaCompany Report #96727 Age:58 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thrombocytopenia Foreign Lariam PS ORAL 1500 MG DAILY<br />
Liver Function Test Other ORAL<br />
Abnormal<br />
Blood Alkaline<br />
Phosphatase Increased<br />
White Blood Cell Count<br />
Decreased<br />
Leukopenia<br />
Date:01/14/99ISR Number: 3180722-3Report Type:Expedited (15-DaCompany Report #96727 Age:58 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Leukopenia Foreign Lariam PS ORAL 1500 MG DAILY<br />
White Blood Cell Count Other ORAL<br />
Decreased<br />
Thrombocytopenia<br />
Liver Function Test<br />
Abnormal<br />
Thrombocytosis<br />
Date:01/19/99ISR Number: 3182196-5Report Type:Expedited (15-DaCompany Report #111775 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Cleft Palate Foreign Lariam PS ORAL 250 MG 1 X<br />
Maternal Drugs Affecting Other PER WEEK ORAL<br />
Foetus<br />
Date:01/19/99ISR Number: 3182565-3Report Type:Expedited (15-DaCompany Report #104407 Age:39 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lethargy Foreign Lariam PS ORAL 250 MG 1 X<br />
Initial or Prolonged Abdominal Pain Health PER WEEK ORAL<br />
Anxiety<br />
Professional<br />
Panic Attack
Chest Discomfort<br />
Restlessness<br />
03-Apr-2012 09:37 AM Page: 92
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/22/99ISR Number: 3184622-4Report Type:Expedited (15-DaCompany Report #92891 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Foreign Lariam PS ORAL 250MG 1 X PER<br />
Gait Disturbance Health WEEK ORAL<br />
Balance Disorder Professional Geluprane C<br />
Dizziness<br />
Date:01/25/99ISR Number: 3185409-9Report Type:Expedited (15-DaCompany Report #112058 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Foreign Lariam PS ORAL 250 MG 1X PER<br />
Initial or Prolonged Anxiety Consumer WEEK ORAL 28 DAY<br />
Dizziness<br />
Gastritis<br />
Date:02/04/99ISR Number: 3191986-4Report Type:Expedited (15-DaCompany Report #112683 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicide Attempt Foreign Lariam Tablets<br />
Initial or Prolonged Intentional Overdose Health (<strong>Mefloquine</strong><br />
Delusional Disorder, Professional Hydrochloride) PS ORAL 250 MG 1 X<br />
Persecutory Type<br />
PER WEEK ORAL<br />
Psychotic Disorder<br />
Date:02/08/99ISR Number: 3193356-1Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Delusion Lariam PS ORAL 8/98 ORAL<br />
Adjustment Disorder<br />
Depression<br />
Date:02/09/99ISR Number: 3194845-6Report Type:Direct Company Report # Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Memory Impairment <strong>Mefloquine</strong> PS 1 PILL ONCE A<br />
Dermatitis WEEK 2 YR<br />
Alopecia<br />
Date:02/10/99ISR Number: 3195192-9Report Type:Direct Company Report # Age:32 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
PT<br />
Panic Attack
Initial or Prolonged<br />
Saliva Altered<br />
Hypoaesthesia<br />
Balance Disorder<br />
Toxicity To Various<br />
Agents<br />
Muscular Weakness<br />
Paraesthesia<br />
Burning Sensation<br />
03-Apr-2012 09:37 AM Page: 93
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Chest Pain<br />
Anxiety Report Source Product Role Manufacturer Route Dose Duration<br />
Headache Lariam PS Roche Labs ORAL 250 MG WEEKLY<br />
Coordination Abnormal<br />
BY MOUTH<br />
Insomnia<br />
Gait Disturbance<br />
Movement Disorder<br />
Malaria<br />
Fatigue<br />
Nightmare<br />
Salivary Hypersecretion<br />
Abdominal Pain<br />
Asthenia<br />
Sensation Of Heaviness<br />
Myalgia<br />
Diarrhoea<br />
Arthralgia<br />
Date:02/16/99ISR Number: 3199935-XReport Type:Expedited (15-DaCompany Report #98676 Age:36 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bipolar I Disorder Foreign Lariam Tablets<br />
Hospitalization - Loss Of Employment Consumer (<strong>Mefloquine</strong><br />
Initial or Prolonged Ulnar Nerve Injury Health Hydrochloride) 250<br />
Depression Professional Mg PS ORAL 250 MG 1 PER<br />
Catatonia<br />
DAY ORAL<br />
Mood Altered<br />
Restlessness<br />
Head Injury<br />
Perseveration<br />
Suicidal Ideation<br />
Paraesthesia<br />
Extrapyramidal Disorder<br />
Hypoaesthesia<br />
Schizophreniform Disorder<br />
Flat Affect<br />
Delusion<br />
Stress<br />
Paresis<br />
Anger<br />
Paranoia<br />
Headache<br />
Abnormal Behaviour<br />
Lethargy<br />
Bradyphrenia<br />
Hallucination<br />
Date:02/17/99ISR Number: 3200611-5Report Type:Expedited (15-DaCompany Report #113215 Age:82 YR Gender:Female I/FU:I
Outcome<br />
Disability<br />
PT<br />
Speech Disorder<br />
Dizziness<br />
Confusional State<br />
Medication Error<br />
Agitation<br />
Deafness<br />
03-Apr-2012 09:37 AM Page: 94
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Coordination Abnormal<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Literature<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL ORAL 61 DAY<br />
Date:02/17/99ISR Number: 3200641-3Report Type:Expedited (15-DaCompany Report #113209 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Overdose Literature Lariam Tablets<br />
Initial or Prolonged Nausea Health (<strong>Mefloquine</strong><br />
Jaundice Professional Hydrochloride) 250<br />
Hallucination Mg PS ORAL 1250 MG 1 PER<br />
Hepatic Failure<br />
ONE DOSE ORAL<br />
Chromaturia<br />
Primaquine<br />
Abdominal Pain<br />
(Primaqine<br />
Liver Function Test Phosphate) SS 1260 MG 1 PER<br />
Abnormal<br />
ONE DOSE<br />
Date:02/17/99ISR Number: 3200745-5Report Type:Expedited (15-DaCompany Report #104407 Age:39 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Pain Foreign Lariam Tablets<br />
Initial or Prolonged Restlessness Health (<strong>Mefloquine</strong><br />
Anxiety Professional Hydrochloride) 250<br />
Panic Attack Mg PS ORAL 250 MG 1 X<br />
Chest Discomfort<br />
PER WEEK ORAL<br />
Lethargy<br />
Date:02/18/99ISR Number: 3201276-9Report Type:Direct Company Report # Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination <strong>Mefloquine</strong> PS Roche Laboratories<br />
Chest Pain<br />
Paraesthesia<br />
Fatigue<br />
Headache<br />
Chills<br />
Date:02/18/99ISR Number: 3201365-9Report Type:Direct Company Report # Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hallucination Lariam PS 1 PILL PER<br />
Hospitalization - Mania WEEK
Initial or Prolonged<br />
Panic Attack<br />
Tachycardia<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 95
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/18/99ISR Number: 3203383-3Report Type:Expedited (15-DaCompany Report #200209 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Foreign Lariam Tablets<br />
Suicidal Ideation Other (<strong>Mefloquine</strong><br />
Paranoia Hydrochloride) PS ORAL 250 MG DAILY<br />
Agitation<br />
ORAL<br />
Date:02/18/99ISR Number: 3203385-7Report Type:Expedited (15-DaCompany Report #200139 Age:4 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Level Above Foreign Lariam Tablets<br />
Initial or Prolonged Therapeutic Health (<strong>Mefloquine</strong><br />
Liver Transplant Professional Hydrochloride) 250<br />
Rejection Mg PS ORAL 250 MG 1 PER<br />
Drug Interaction<br />
WEEK ORAL<br />
Fk506 (Tacrolimus) SS<br />
Date:02/19/99ISR Number: 3203427-9Report Type:Expedited (15-DaCompany Report #113217 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Decreased Appetite Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Nervousness Literature Hydrochloride) PS ORAL 250 MG 1 PER<br />
Oedema Health WEEK ORAL<br />
Blood Albumin Decreased Professional<br />
Vomiting<br />
Dehydration<br />
Inflammation<br />
Blood Calcium Decreased<br />
Weight Decreased<br />
Hepatomegaly<br />
Endoscopy Upper<br />
Gastrointestinal Tract<br />
Abnormal<br />
Hepatic Failure<br />
Oedema Peripheral<br />
Compartment Syndrome<br />
Abdominal Distension<br />
Liver Function Test<br />
Abnormal<br />
Depression<br />
Blood Potassium Decreased<br />
Diarrhoea<br />
Hepatic Steatosis<br />
Gastroenteritis<br />
Date:02/19/99ISR Number: 3204070-8Report Type:Direct Company Report # Age:51 YR Gender:Unknown I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Interest Health Lariam PS<br />
Insomnia Professional Halcion C<br />
Disturbance In Attention<br />
Cognitive Disorder<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 96
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/22/99ISR Number: 3204969-2Report Type:Expedited (15-DaCompany Report #113209 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chromaturia Literature Lariam Tablets<br />
Initial or Prolonged Abdominal Pain Health (<strong>Mefloquine</strong><br />
Jaundice Professional Hydrochloride) 250<br />
Nausea Mg PS ORAL 1250 MG 1 PER<br />
Liver Function Test<br />
ONE DOSE<br />
Abnormal<br />
ORAL<br />
Hepatic Failure<br />
Primaquine<br />
Hallucination<br />
(Primaquine<br />
Overdose Phosphate) SS 1260 MG 1 PER<br />
ONE DOSE<br />
Date:02/22/99ISR Number: 3205088-1Report Type:Expedited (15-DaCompany Report #98676 Age:36 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Foreign Lariam Tablets<br />
Hospitalization - Paresis Consumer (<strong>Mefloquine</strong><br />
Initial or Prolonged Extrapyramidal Disorder Health Hydrochloride) 250<br />
Restlessness Professional Mg PS ORAL 250 MG 1 PER<br />
Peripheral Nerve Lesion<br />
DAY ORAL<br />
Disturbance In Attention<br />
Lethargy<br />
Hypoaesthesia<br />
Paranoia<br />
Schizophreniform Disorder<br />
Delusion<br />
Paraesthesia<br />
Bipolar I Disorder<br />
Catatonia<br />
Hallucination<br />
Date:02/24/99ISR Number: 3206756-8Report Type:Expedited (15-DaCompany Report #200139 Age:4 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Level Above Foreign Lariam PS ORAL TABLETS, 1<br />
Initial or Prolonged Therapeutic Health PER WEEK ORAL<br />
Transplant Rejection Professional Fk506 (Tacrolimus) SS<br />
Drug Interaction<br />
Date:02/26/99ISR Number: 3208126-5Report Type:Expedited (15-DaCompany Report #200527 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Toxoplasmosis Foreign Lariam PS ORAL 250MG 1 PER<br />
Health<br />
WEEK ORAL<br />
Professional Beriglobin C
Typhoral C<br />
03-Apr-2012 09:37 AM Page: 97
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/26/99ISR Number: 3208128-9Report Type:Expedited (15-DaCompany Report #200442 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Jaundice Health Lariam PS ORAL 250MG 1 PER<br />
Condition Aggravated Professional WEEK ORAL<br />
Haemolytic Anaemia<br />
Date:03/01/99ISR Number: 3209203-5Report Type:Expedited (15-DaCompany Report #200139 Age:4 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Foreign Lariam Tablets<br />
Initial or Prolonged Liver Transplant Health (<strong>Mefloquine</strong><br />
Rejection Professional Hydrochloride) PS ORAL 1 PER WEEK<br />
Drug Level Above<br />
ORAL<br />
Therapeutic Fk506 (Tacrolimus) SS<br />
Date:03/01/99ISR Number: 3209298-9Report Type:Expedited (15-DaCompany Report #201094 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Speech Disorder Other Lariam Tablets<br />
Coordination Abnormal<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:03/01/99ISR Number: 3209366-1Report Type:Expedited (15-DaCompany Report #200926 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Foreign Laraim Tablets<br />
Initial or Prolonged Delusion Health (<strong>Mefloquine</strong><br />
Hepatitis Professional Hydrochloride) PS ORAL 250 MG 1 PER<br />
Schizophrenia<br />
WEEK ORAL<br />
Jaundice<br />
Date:03/03/99ISR Number: 3210714-7Report Type:Direct Company Report # Age:69 YR Gender:Female I/FU:I<br />
Outcome<br />
Disability<br />
Other Serious<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT<br />
Disturbance In Attention<br />
Feeling Abnormal<br />
Joint Stiffness<br />
Abdominal Pain Upper<br />
Insomnia<br />
Disorientation<br />
Dizziness<br />
Decreased Appetite<br />
Vision Blurred<br />
Chills
Tremor<br />
Fatigue<br />
Abdominal Pain<br />
Vertigo<br />
Arthralgia<br />
Dysgraphia<br />
Nausea<br />
Asthenia<br />
03-Apr-2012 09:37 AM Page: 98
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Confusional State<br />
Anxiety<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Hoffman-La Roche Inc ORAL 250MG ONCE<br />
/WEEK ORALLY<br />
/8 WEEKS<br />
Date:03/03/99ISR Number: 3214303-XReport Type:Direct Company Report # Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Hypersomnia Larium PS ORAL TAB 1T ORAL 2 DAY<br />
Intervention to<br />
Hyperhidrosis<br />
Prevent Permanent Agoraphobia<br />
Impairment/Damage Malaria<br />
Depression<br />
Bipolar Disorder<br />
Date:03/05/99ISR Number: 3214272-2Report Type:Expedited (15-DaCompany Report #201217 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Fibrosis Foreign Lariam PS ORAL<br />
Hepatic Steatosis Other Hepatitis A Vaccine<br />
Ventricular Hypertrophy<br />
(Hepatitis A<br />
Obstruction Vaccine) SS INTRAMUSCULAR<br />
Hepatosplenomegaly<br />
Malaise<br />
Ventricular Fibrillation<br />
Cardiomegaly<br />
Date:03/05/99ISR Number: 3214273-4Report Type:Expedited (15-DaCompany Report #201099 Age:69 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Neurosensory Health Lariam PS ORAL 250 MG 1 PER<br />
Deafness Professional WEEK<br />
Beta-Blocker (Beta<br />
Blocker Nos)<br />
C<br />
Date:03/08/99ISR Number: 3214076-0Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypoaesthesia Lariam PS ONCE.WK<br />
Palpitations<br />
Anxiety<br />
Pain<br />
Dermatitis
Dizziness<br />
03-Apr-2012 09:37 AM Page: 99
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/08/99ISR Number: 3215137-2Report Type:Expedited (15-DaCompany Report #72710 Age:82 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Deafness Literature Lariam PS ORAL 250 MG 1 PER<br />
Speech Disorder Health DAY ORAL<br />
Agitation Professional Cardizem C<br />
Asthenia Propoxyphene C<br />
Coordination Abnormal<br />
Desyrel (Trazodone<br />
Bundle Branch Block Hydrochloride) C<br />
Medication Error Ultram C<br />
Confusional State Depakote C<br />
Respiratory Disorder Apresoline C<br />
Alanine Aminotransferase<br />
Increased<br />
Accidental Overdose<br />
Dizziness<br />
Date:03/08/99ISR Number: 3215139-6Report Type:Expedited (15-DaCompany Report #113215 Age:82 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Deafness Literature Lariam PS ORAL ORAL 61 DAY<br />
Agitation<br />
Health<br />
Confusional State<br />
Professional<br />
Dizziness<br />
Medication Error<br />
Speech Disorder<br />
Coordination Abnormal<br />
Date:03/08/99ISR Number: 3215142-6Report Type:Expedited (15-DaCompany Report #200926 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abnormal Dreams Foreign Lariam PS ORAL 250 MG 1 PER<br />
Initial or Prolonged Aggression Health WEEK ORAL<br />
Feeling Abnormal<br />
Professional<br />
Injury<br />
Jaundice<br />
Stress<br />
Hepatitis<br />
Psychotic Disorder<br />
Schizophrenia<br />
Hepatic Enzyme Increased<br />
Delusion<br />
Asthenia<br />
Mental Disorder<br />
Hallucination<br />
Date:03/08/99ISR Number: 3218790-2Report Type:Direct Company Report # Age: Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Lariam PS 1TAB PER WEEK<br />
Restlessness FOR 3 WEEKS ;<br />
Dizziness<br />
GIVEN SAMPLE<br />
IN MD'S<br />
OFFICE<br />
Celbrex<br />
C<br />
03-Apr-2012 09:37 AM Page: 100
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diamox<br />
Prempro<br />
C<br />
C<br />
Date:03/17/99ISR Number: 3223037-7Report Type:Expedited (15-DaCompany Report #200209 Age:19 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Agitation Foreign Lariam Tablets<br />
Suicidal Ideation Other (<strong>Mefloquine</strong><br />
Nightmare Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
Paranoia<br />
WEEK ORAL<br />
Date:03/24/99ISR Number: 3225735-8Report Type:Expedited (15-DaCompany Report #200442 Age:32 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anaemia Haemolytic Health Lariam Tablets<br />
Autoimmune Professional (<strong>Mefloquine</strong><br />
Jaundice Hydrochloride) 250<br />
Diarrhoea Mg PS ORAL 250 MG 1 PER<br />
Condition Aggravated<br />
WEEK ORAL<br />
Date:03/25/99ISR Number: 3226364-2Report Type:Expedited (15-DaCompany Report #202374 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lethargy Other Lariam Tablets<br />
Initial or Prolonged Dizziness (<strong>Mefloquine</strong><br />
Colonic Polyp Hydrochloride) 250<br />
Haemoglobin Decreased Mg PS ORAL 250 MG 1 PER<br />
Chromaturia<br />
WEEK ORAL<br />
Haemolytic Anaemia<br />
Abdominal Pain Lower<br />
Decreased Appetite<br />
Jaundice<br />
Haematocrit Decreased<br />
Neoplasm Malignant<br />
Pain<br />
Body Temperature<br />
Decreased<br />
Dehydration<br />
Date:03/30/99ISR Number: 3230064-2Report Type:Expedited (15-DaCompany Report #202571 Age:65 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haematuria Foreign Lariam PS ORAL 250 MG 1 PER<br />
Haematoma Health 1 WEEK ORAL<br />
International Normalised Professional Previscan
Ratio Increased (Fluindione) SS ORAL ORAL<br />
Drug Level Above<br />
Therapeutic<br />
Nephrolithiasis<br />
Drug Interaction<br />
03-Apr-2012 09:37 AM Page: 101
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/31/99ISR Number: 3230866-2Report Type:Expedited (15-DaCompany Report #200926 Age:57 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abnormal Dreams Foreign Lariam Tablets<br />
Initial or Prolonged Abnormal Behaviour Health (<strong>Mefloquine</strong><br />
Schizophrenia Professional Hydrochloride) PS ORAL 250 MG 1 PER<br />
Hallucination<br />
WEEK ORAL<br />
Asthenia<br />
Hepatitis<br />
Delusion<br />
Jaundice<br />
Aggression<br />
Stress<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Psychotic Disorder<br />
Feeling Abnormal<br />
Date:04/01/99ISR Number: 3231215-6Report Type:Direct Company Report # Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dizziness Lariam PS<br />
Pyrexia<br />
Deafness<br />
Headache<br />
Malaise<br />
Insomnia<br />
Fatigue<br />
Dermatitis<br />
Nasal Congestion<br />
Pulmonary Congestion<br />
Hallucination<br />
Influenza Like Illness<br />
Pruritus<br />
Fear<br />
Paranoia<br />
Hyperhidrosis<br />
Chills<br />
Vomiting<br />
Decreased Appetite<br />
Aggression<br />
Date:04/02/99ISR Number: 3232453-9Report Type:Expedited (15-DaCompany Report #202998 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Foreign Lariam Tablets<br />
Initial or Prolonged Dizziness Health (<strong>Mefloquine</strong>
Anxiety Professional Hydrochloride) 250<br />
Hearing Impaired Mg PS ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 102
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/08/99ISR Number: 3236025-1Report Type:Expedited (15-DaCompany Report #203042 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Abnormal Labour Foreign Lariam Tablets<br />
Intervention to Premature Rupture Of Consumer (<strong>Mefloquine</strong><br />
Prevent Permanent Membranes Hydrochloride) 250<br />
Impairment/Damage Maternal Drugs Affecting Mg PS ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Uterine Spasm<br />
Prenatal Vitamins<br />
Metrorrhagia<br />
Premature Baby<br />
(Minerals<br />
Nos/Multivitamins<br />
Nos)<br />
C<br />
Date:04/13/99ISR Number: 3239019-5Report Type:Expedited (15-DaCompany Report #202374 Age:64 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haemolytic Anaemia Consumer Lariam Tablets<br />
Initial or Prolonged Chromaturia Health (<strong>Mefloquine</strong><br />
Reticulocyte Count Professional Hydrochloride) 250<br />
Increased Other Mg PS ORAL 250 MG 1 PER<br />
Colonic Polyp<br />
WEEK ORAL<br />
Dehydration<br />
Decreased Appetite<br />
Lethargy<br />
Jaundice<br />
Haematocrit Decreased<br />
Pain<br />
Decreased Activity<br />
Dizziness<br />
Abdominal Pain Lower<br />
Haemoglobin Decreased<br />
Neoplasm Malignant<br />
Amnesia<br />
Pyrexia<br />
Date:04/13/99ISR Number: 3239023-7Report Type:Expedited (15-DaCompany Report #105284 Age:30 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Depression Foreign Lariam Tablets<br />
Hospitalization - Hemiparesis Health (<strong>Mefloquine</strong><br />
Initial or Prolonged Insomnia Professional Hydrochloride) PS ORAL 750 MG 1 PER<br />
Head Injury<br />
ONE DOSE ORAL<br />
Restlessness Tranxilium C<br />
Suicide Attempt Atosil C<br />
Resochin<br />
C<br />
Date:04/13/99ISR Number: 3239042-0Report Type:Expedited (15-DaCompany Report #203230 Age: Gender:Female I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT<br />
Pyrexia<br />
Foreign Travel<br />
Panic Attack<br />
Vomiting<br />
Chest Discomfort<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 103
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dehydration<br />
Cough<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Health<br />
Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:04/13/99ISR Number: 3239440-5Report Type:Expedited (15-DaCompany Report #58978 Age:31 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Consumer Lariam Tablets<br />
Dizziness<br />
(<strong>Mefloquine</strong><br />
Brain Contusion Hydrochloride) PS ORAL 250 MG 1 PER<br />
Amnesia<br />
WEEK ORAL<br />
Disturbance In Attention Norflex C<br />
Feeling Abnormal<br />
Asthenia<br />
Emotional Distress<br />
Nystagmus<br />
Dyspnoea<br />
Heart Rate Increased<br />
Nervous System Disorder<br />
Psychotic Disorder<br />
Injury<br />
Mental Disability<br />
Decreased Activity<br />
Dysgraphia<br />
Fatigue<br />
Visual Impairment<br />
Reading Disorder<br />
Date:04/14/99ISR Number: 3239847-6Report Type:Direct Company Report # Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Palpitations Health Lariam Tab PS Ud 1/ WK 6 WKS<br />
Required Vertigo Professional<br />
Intervention to<br />
Anxiety<br />
Prevent Permanent Hypertension<br />
Impairment/Damage Myalgia<br />
Date:04/14/99ISR Number: 3239864-6Report Type:Expedited (15-DaCompany Report #107634 Age:37 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Hypertension<br />
Nuclear Magnetic<br />
Resonance Imaging
Abnormal<br />
Blood Immunoglobulin G<br />
Increased<br />
Flushing<br />
Paraesthesia<br />
Hearing Impaired<br />
Hypoaesthesia<br />
Muscular Weakness<br />
03-Apr-2012 09:37 AM Page: 104
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Ear Pain<br />
Sensory Disturbance<br />
Multiple Sclerosis Report Source Product Role Manufacturer Route Dose Duration<br />
Dysgraphia Health Lariam Tablets<br />
Coordination Abnormal Professional (<strong>Mefloquine</strong><br />
Demyelination Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
Brain Stem Auditory<br />
WEEK ORAL<br />
Evoked Response Abnormal<br />
Motor Dysfunction<br />
Foreign Travel<br />
Ear Discomfort<br />
Cerebral Ischaemia<br />
Date:04/14/99ISR Number: 3239951-2Report Type:Expedited (15-DaCompany Report #203042 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Premature Baby Foreign Lariam PS TRANSPLACENTAL 250 MG 1 PER<br />
Intervention to Maternal Drugs Affecting Consumer WEEK ORAL<br />
Prevent Permanent Foetus Prenatal Vitamins C<br />
Impairment/Damage Muscle Spasms<br />
Metrorrhagia<br />
Premature Rupture Of<br />
Membranes<br />
Abnormal Labour<br />
Date:04/15/99ISR Number: 3240621-5Report Type:Expedited (15-DaCompany Report #102721 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Other Lariam Tablets<br />
Foetus<br />
(<strong>Mefloquine</strong><br />
Deafness Congenital Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Vitamins<br />
C<br />
Date:04/15/99ISR Number: 3240622-7Report Type:Expedited (15-DaCompany Report #202571 Age:59 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nephrolithiasis Foreign Lariam Tablet<br />
Epistaxis Health (<strong>Mefloquine</strong><br />
Drug Interaction Professional Hydrochloride) PS ORAL 250 MG 1 PER<br />
Haematuria<br />
WEEK ORAL<br />
Haematoma<br />
Previscan<br />
Subcutaneous Haematoma (Fluindione) SS ORAL 20 MG 1 PER 1<br />
Renal Colic<br />
DAY ORAL<br />
International Normalised Nitrofurantoine C<br />
Ratio Increased
Date:04/20/99ISR Number: 3243445-8Report Type:Expedited (15-DaCompany Report #203837 Age: Gender:Male I/FU:I<br />
Outcome<br />
Death<br />
PT<br />
Hallucination<br />
Nervous System Disorder<br />
Medication Error<br />
03-Apr-2012 09:37 AM Page: 105
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Completed Suicide<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Other<br />
Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:04/26/99ISR Number: 3246927-8Report Type:Expedited (15-DaCompany Report #204205 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Foreign Lariam Tablets<br />
Asthma Health (<strong>Mefloquine</strong><br />
Lethargy Professional Hydrochloride) PS ORAL ORAL<br />
Anxiety<br />
Other<br />
Date:04/26/99ISR Number: 3247086-8Report Type:Expedited (15-DaCompany Report #202374 Age:64 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Biopsy Bone Marrow Consumer Lariam Tablets<br />
Initial or Prolonged Abnormal Health (<strong>Mefloquine</strong><br />
Dizziness Professional Hydrochloride) 250<br />
Dehydration Other Mg PS ORAL 250 MG 1 PER<br />
Reticulocyte Count<br />
WEEK ORAL<br />
Increased<br />
Abdominal Pain Lower<br />
Pyrexia<br />
Haemolytic Anaemia<br />
Decreased Appetite<br />
Jaundice<br />
Lethargy<br />
Colon Cancer<br />
Colonic Polyp<br />
Haemoglobin Decreased<br />
Chromaturia<br />
Haematocrit Decreased<br />
Pain<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Amnesia<br />
Blood Bilirubin Increased<br />
Date:04/27/99ISR Number: 3247569-0Report Type:Expedited (15-DaCompany Report #203042 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Premature Labour Foreign Lariam Tablets<br />
Intervention to Premature Baby Consumer (<strong>Mefloquine</strong><br />
Prevent Permanent Maternal Drugs Affecting Hydrochloride) 250
Impairment/Damage Foetus Mg PS ORAL 250 MG 1 PER<br />
Premature Rupture Of<br />
WEEK ORAL<br />
Membranes Prenatal Vitamins C<br />
Small For Dates Baby<br />
Delayed Delivery<br />
Arrested Labour<br />
03-Apr-2012 09:37 AM Page: 106
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/27/99ISR Number: 3247571-9Report Type:Expedited (15-DaCompany Report #204366 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Missed Foreign Lariam Tablets<br />
Abortion Spontaneous Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL ORAL<br />
Date:04/28/99ISR Number: 3249015-XReport Type:Expedited (15-DaCompany Report #104882 Age:25 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tachycardia Foreign Lariam Tablets<br />
Initial or Prolonged Circulatory Collapse Consumer (<strong>Mefloquine</strong><br />
Balance Disorder Health Hydrochloride) PS ORAL ORAL<br />
Paraesthesia<br />
Professional<br />
Ventricular Extrasystoles<br />
Panic Attack<br />
Cardiovascular Disorder<br />
Muscle Spasms<br />
Anxiety<br />
Arrhythmia<br />
Restlessness<br />
Asthenia<br />
Dizziness<br />
Confusional State<br />
Convulsion<br />
Feeling Abnormal<br />
Date:04/30/99ISR Number: 3251229-XReport Type:Expedited (15-DaCompany Report #106318 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Grand Mal Convulsion Foreign Lariam Tablets<br />
Initial or Prolonged Dystonia (<strong>Mefloquine</strong><br />
Panic Attack Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK<br />
Date:04/30/99ISR Number: 3251360-9Report Type:Expedited (15-DaCompany Report #58978 Age:31 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Malaise<br />
Circulatory Collapse<br />
Asthenia<br />
Emotional Distress<br />
Decreased Activity<br />
Feeling Abnormal<br />
Mental Impairment<br />
Disturbance In Attention<br />
Heart Rate Increased
Vertigo<br />
Dyspnoea<br />
Amnesia<br />
Psychotic Disorder<br />
Dizziness<br />
Fatigue<br />
Nervous System Disorder<br />
Injury<br />
03-Apr-2012 09:37 AM Page: 107
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Nystagmus<br />
Visual Impairment<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Health<br />
Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Norflex<br />
C<br />
Date:05/04/99ISR Number: 3253962-2Report Type:Expedited (15-DaCompany Report #202374 Age:64 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Decreased Appetite Consumer Lariam Tablets<br />
Initial or Prolonged Fatigue Health (<strong>Mefloquine</strong><br />
Jaundice Professional Hydrochloride) 250<br />
Abdominal Pain Lower Other Mg PS ORAL 250 MG 1 PER<br />
Coombs Direct Test<br />
WEEK ORAL<br />
Positive<br />
Chromaturia<br />
Pain<br />
Body Temperature<br />
Increased<br />
Dizziness<br />
Dehydration<br />
Amnesia<br />
Colonic Polyp<br />
Lethargy<br />
Haemolytic Anaemia<br />
Anaemia Haemolytic<br />
Autoimmune<br />
Colon Cancer<br />
Date:05/11/99ISR Number: 3258994-6Report Type:Expedited (15-DaCompany Report #99483 Age:36 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Nausea<br />
Balance Disorder<br />
Diarrhoea<br />
Paralysis<br />
Tachycardia<br />
Abdominal Pain Upper<br />
Hypoaesthesia<br />
Ear Disorder<br />
Visual Impairment<br />
Pruritus<br />
Panic Attack<br />
Paraesthesia<br />
Tinnitus
Tremor<br />
Dizziness<br />
Flushing<br />
Condition Aggravated<br />
Depression<br />
Anxiety<br />
Toxicity To Various<br />
Agents<br />
03-Apr-2012 09:37 AM Page: 108
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hearing Impaired<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
Other<br />
ONE DOSE ORAL<br />
Contraceptive<br />
C<br />
Date:05/11/99ISR Number: 3258996-XReport Type:Expedited (15-DaCompany Report #205111 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Foreign Lariam Tablets<br />
Initial or Prolonged Dyspnoea Health (<strong>Mefloquine</strong><br />
Disability Psychotic Disorder Professional Hydrochloride) 250<br />
Hypoaesthesia Mg PS ORAL 150 MG 1 PER<br />
Confusional State<br />
WEEK ORAL<br />
Anxiety<br />
Muscle Spasms<br />
Date:05/11/99ISR Number: 3259222-8Report Type:Expedited (15-DaCompany Report #205170 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Date:05/13/99ISR Number: 3261143-1Report Type:Expedited (15-DaCompany Report #205270 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Musculoskeletal Stiffness Foreign Lariam Tablets<br />
Initial or Prolonged Visual Impairment Consumer (<strong>Mefloquine</strong><br />
Insomnia Hydrochloride) 250<br />
Panic Attack Mg PS ORAL 250 MG 1 PER<br />
Depression<br />
WEEK ORAL<br />
Dizziness<br />
Ketoprofene<br />
Suicidal Ideation (Ketoprofen) C<br />
Fatigue<br />
Ibuprofene<br />
(Ibuprofen)<br />
C<br />
Date:05/20/99ISR Number: 3266172-XReport Type:Expedited (15-DaCompany Report #205574 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disturbance In Attention Foreign Lariam Tablets
Initial or Prolonged Coordination Abnormal Consumer (<strong>Mefloquine</strong><br />
Vertigo Hydrochloride) 250<br />
Visual Field Defect Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 109
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/21/99ISR Number: 3267645-6Report Type:Expedited (15-DaCompany Report #205170 Age:31 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Foreign Lariam Tablets<br />
Consumer<br />
(<strong>Mefloquine</strong><br />
Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:05/21/99ISR Number: 3268957-2Report Type:Direct Company Report # Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dissociation Health <strong>Mefloquine</strong> PS ONCE PER WEEK<br />
Dizziness Professional X 6 WEEKS<br />
Emotional Disorder Ocp'S C<br />
Crying<br />
Date:06/02/99ISR Number: 3275253-6Report Type:Expedited (15-DaCompany Report #99483 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Flushing Foreign Lariam Tablets<br />
Initial or Prolonged Blood Cholesterol Health (<strong>Mefloquine</strong><br />
Increased Professional Hydrochloride) 250<br />
Diarrhoea Other Mg PS ORAL 250 MG 1 PER<br />
Vestibular Ataxia<br />
ONE DOSE ORAL<br />
Tachycardia Contraceptive C<br />
Pruritus<br />
Balance Disorder<br />
Refraction Disorder<br />
Hypoaesthesia<br />
Paralysis<br />
Tremor<br />
Depression<br />
Anxiety<br />
Abdominal Pain Upper<br />
Tinnitus<br />
Panic Attack<br />
Dizziness<br />
Visual Impairment<br />
Paraesthesia<br />
Ear Discomfort<br />
Date:06/07/99ISR Number: 3285451-3Report Type:Periodic Company Report #200588 Age:18 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coma Health Lariam Tablets<br />
Initial or Prolonged Malaria Professional (<strong>Mefloquine</strong>
Chills Hydrochloride) 250<br />
Drug Ineffective Mg PS ORAL 62.5 MG 1 PER<br />
Pyrexia<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 110
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285452-5Report Type:Periodic Company Report #202262 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Consumer Lariam Tablets<br />
Dizziness<br />
(<strong>Mefloquine</strong><br />
Vertigo Hydrochloride) 250<br />
Nystagmus Mg PS ORAL 250 MG 1 PER<br />
Insomnia<br />
WEEK ORAL<br />
Coma Hormone Replacement C<br />
Anxiety<br />
Flatulence<br />
Diarrhoea<br />
Restlessness<br />
Date:06/07/99ISR Number: 3285453-7Report Type:Periodic Company Report #202382 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Other Lariam Tablets<br />
Paranoia<br />
(<strong>Mefloquine</strong><br />
Hallucination, Auditory Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285454-9Report Type:Periodic Company Report #203056 Age:6 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Other Lariam Tablets<br />
Convulsion<br />
(<strong>Mefloquine</strong><br />
Confusional State Hydrochloride ) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285455-0Report Type:Periodic Company Report #97937 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cyst Other Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride ) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285456-2Report Type:Periodic Company Report #98216 Age:70 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dry Skin Other Lariam Tablets
Rash Erythematous (Mefloquiine<br />
Hydrochlorine) 250<br />
Mg PS 250MG<br />
03-Apr-2012 09:37 AM Page: 111
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285457-4Report Type:Periodic Company Report #98284 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vitreous Detachment Consumer Lariam Tablets<br />
Abnormal Behaviour Health (<strong>Mefloquine</strong><br />
Vitreous Floaters Professional Hydrochloride) 250<br />
Photopsia Mg PS ORAL 250 MG 1 PER<br />
Dermatitis<br />
Estrogen/Progesteron<br />
e<br />
C<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285458-6Report Type:Periodic Company Report #99463 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Burning Sensation Health Lariam (<strong>Mefloquine</strong><br />
Headache Professional Hydrochloride) 250<br />
Vomiting Mg PS ORAL 250 MG 1 PER<br />
Nausea<br />
WEEK ORAL<br />
Premarin<br />
C<br />
Date:06/07/99ISR Number: 3285459-8Report Type:Periodic Company Report #99776 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Respiratory Disorder Other Lariam Tablets (<br />
Panic Attack<br />
<strong>Mefloquine</strong><br />
Hyperhidrosis Hydrochloride) 250<br />
Delirium Mg PS ORAL 250 MG 1 PER<br />
Nightmare<br />
WEEK ORAL<br />
Hallucination<br />
Pyrexia<br />
Date:06/07/99ISR Number: 3285460-4Report Type:Periodic Company Report #99969 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psoriasis Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrocholride) 25omg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285461-6Report Type:Periodic Company Report #101038 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Prostatitis Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL 42 DAY<br />
03-Apr-2012 09:37 AM Page: 112
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285462-8Report Type:Periodic Company Report #101838 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypertension Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285463-XReport Type:Periodic Company Report #102345 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cystitis Other Lariam Tablets<br />
Nausea<br />
(<strong>Mefloquine</strong><br />
Pyrexia Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285464-1Report Type:Periodic Company Report #102654 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Face Oedema Health Lariam Tablets<br />
Urticaria Professional (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285465-3Report Type:Periodic Company Report #102931 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Face Oedema Health Lariam Tablets<br />
Lip Disorder Professional (<strong>Mefloquine</strong><br />
Skin Irritation Hydrochloride) 250<br />
Rash Erythematous Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285466-5Report Type:Periodic Company Report #103254 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Menstruation Irregular Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:06/07/99ISR Number: 3285467-7Report Type:Periodic Company Report #103331 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Throat Irritation Consumer Lariam Tablets<br />
Alopecia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride ) 250<br />
Mg<br />
PS<br />
03-Apr-2012 09:37 AM Page: 113
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Ortho-Novum<br />
C<br />
Date:06/07/99ISR Number: 3285468-9Report Type:Periodic Company Report #103366 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hyperhidrosis Other Lariam Tablets<br />
Asthenia<br />
(<strong>Mefloquine</strong><br />
Vomiting Hydrochloride) 250<br />
Nausea Mg PS ORAL 250 MG 1 PER<br />
Feeling Jittery<br />
WEEK ORAL<br />
Pyrexia Prempro C<br />
Retching<br />
Chills<br />
Diarrhoea<br />
Date:06/07/99ISR Number: 3285469-0Report Type:Periodic Company Report #103404 Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Health Lariam Tablets<br />
Decreased Appetite Professional (<strong>Mefloquine</strong><br />
Depression Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL 42 DAY<br />
Date:06/07/99ISR Number: 3285470-7Report Type:Periodic Company Report #103535 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eye Disorder Health Lariam Tablets<br />
Nervous System Disorder Professional (<strong>Mefloquine</strong><br />
Burning Sensation Hydrochloride) 250<br />
Panic Disorder Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285471-9Report Type:Periodic Company Report #98501 Age:56 YR Gender:Female I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Insomnia<br />
Anxiety<br />
Tremor<br />
Myalgia<br />
Balance Disorder<br />
Palpitations<br />
Hallucination<br />
Coma<br />
Muscle Twitching
Pyrexia<br />
Headache<br />
Depression<br />
Cough<br />
Dyspnoea<br />
Fatigue<br />
Extrasystoles<br />
Tachycardia<br />
03-Apr-2012 09:37 AM Page: 114
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Paranoia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Aspirin<br />
C<br />
Biaxin<br />
C<br />
Date:06/07/99ISR Number: 3285472-0Report Type:Periodic Company Report #99092 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Health Lariam Tablets<br />
Initial or Prolonged Professional (Mefoquine<br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285473-2Report Type:Periodic Company Report #100159 Age:75 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Disorder Consumer Lariam Tablets<br />
Depression Health (<strong>Mefloquine</strong><br />
Amnesia Professional Hydrochloride) 250<br />
Gait Disturbance Other Mg PS ORAL 250 MG 1 PER<br />
Fatigue<br />
WEEK ORAL<br />
Dizziness Cimetidine C<br />
Back Disorder Prempro C<br />
Questran<br />
C<br />
Mevacor<br />
C<br />
Date:06/07/99ISR Number: 3285474-4Report Type:Periodic Company Report #101158 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dry Mouth Consumer Lariam Tablets<br />
Dehydration Other (<strong>Mefloquine</strong><br />
Dyspnoea Hydrochloride) 250<br />
Paraesthesia Mg PS ORAL 250 MG 1 PER<br />
Diarrhoea<br />
WEEK ORAL<br />
Dizziness Provera C<br />
Chest Discomfort Cipro C<br />
Coma Calcium C<br />
Herbal<br />
C<br />
Estrace<br />
C<br />
Multivitamin W Iron C
Date:06/07/99ISR Number: 3285475-6Report Type:Periodic Company Report #104590 Age:27 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Anxiety<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 115
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mania<br />
Psychotic Disorder<br />
Psychiatric Symptom Report Source Product Role Manufacturer Route Dose Duration<br />
Coma Health Lariam Tablets<br />
Paranoia Professional (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285476-8Report Type:Periodic Company Report #104852 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Balance Disorder Consumer Lariam Tablets<br />
Initial or Prolonged Mental Impairment (<strong>Mefloquine</strong><br />
Asthenia Hydrochloride) 250<br />
Confusional State Mg PS ORAL 250 MG 1 PER<br />
Constipation<br />
WEEK ORAL<br />
Amnesia Allegra C<br />
Myalgia Wellbutrin C<br />
Hyperhidrosis<br />
Fatigue<br />
Arthralgia<br />
Rash Pustular<br />
Sedation<br />
Coma<br />
Agitation<br />
Hypersensitivity<br />
Date:06/07/99ISR Number: 3285477-XReport Type:Periodic Company Report #105631 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Consumer Lariam Tablets<br />
Initial or Prolonged Coma (<strong>Mefloquine</strong><br />
Vomiting Hydrochloride) 250<br />
Dehydration Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285478-1Report Type:Periodic Company Report #106904 Age:77 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypertension Consumer Lariam Tablets<br />
Initial or Prolonged Tremor Other (<strong>Mefloquine</strong><br />
Tachycardia Hydrochloride) PS ORAL 80 MG 1 PER<br />
Ventricular Extrasystoles<br />
WEEK ORAL<br />
Multivitamin<br />
C<br />
Folic Acid<br />
C<br />
Flomax<br />
C
Baby Aspirin C<br />
03-Apr-2012 09:37 AM Page: 116
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285479-3Report Type:Periodic Company Report #107363 Age:77 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coma Consumer Lariam Tablets<br />
Initial or Prolonged Drug Ineffective Health (<strong>Mefloquine</strong><br />
Malaria Professional Hydrochloride) 250<br />
Fatigue Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Accutane<br />
C<br />
Aspirin<br />
C<br />
Tenormin<br />
C<br />
Axid<br />
C<br />
Date:06/07/99ISR Number: 3285480-XReport Type:Periodic Company Report #109779 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Coma Other Lariam Tablets<br />
Bipolar I Disorder<br />
(<strong>Mefloquine</strong><br />
Mood Swings Hydrochloride) 250<br />
Hallucination Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285481-1Report Type:Periodic Company Report #110102 Age:67 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Health Lariam Tablets<br />
Initial or Prolonged Confusional State Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285482-3Report Type:Periodic Company Report #111592 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lethargy Other Lariam Tablets<br />
Initial or Prolonged Dizziness (<strong>Mefloquine</strong><br />
Movement Disorder Hydrochloride) 250<br />
Coma Mg PS ORAL ORAL<br />
Vision Blurred Prevacid C<br />
Malaise Zestril C<br />
Tremor<br />
Date:06/07/99ISR Number: 3285483-5Report Type:Periodic Company Report #112271 Age:19 MON Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Health Lariam Tablets
Initial or Prolonged Abdominal Pain Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 50 MG 1 PER<br />
WEEK ORAL<br />
Bactrim Paediatric C<br />
03-Apr-2012 09:37 AM Page: 117
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285484-7Report Type:Periodic Company Report #112461 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coma Consumer Lariam Tablets<br />
Initial or Prolonged Hyperhidrosis (<strong>Mefloquine</strong><br />
Malaria Hydrochloride) PS ORAL 250 MG 1 PER<br />
Drug Ineffective<br />
WEEK ORAL<br />
Dehydration Prilosec C<br />
Leukocytosis<br />
Vomiting<br />
Pyrexia<br />
Chills<br />
Abdominal Pain<br />
Diarrhoea<br />
Lethargy<br />
Date:06/07/99ISR Number: 3285485-9Report Type:Periodic Company Report #112492 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Other Lariam Tablets<br />
Initial or Prolonged Vomiting (<strong>Mefloquine</strong><br />
Abdominal Pain Hydrochloride) PS ORAL 250 MG 1 PER<br />
Chills<br />
WEEK ORAL<br />
Chest Pain<br />
Coma<br />
Diarrhoea<br />
Hyperhidrosis<br />
Drug Ineffective<br />
Date:06/07/99ISR Number: 3285486-0Report Type:Periodic Company Report #112582 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Weight Decreased Health Lariam Tablets<br />
Personality Change Professional (<strong>Mefloquine</strong><br />
Depression Hydrochloride) PS ORAL 1 DOSE FORM<br />
Anxiety<br />
DAILY ORAL<br />
Coma<br />
Date:06/07/99ISR Number: 3285487-2Report Type:Periodic Company Report #113216 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Health Lariam Tablets<br />
Asthenia Professional (<strong>Mefloquine</strong><br />
Paraesthesia Hydrochloride) PS ORAL 250 MG DAILY<br />
Depression<br />
ORAL<br />
Fatigue Lamisil C<br />
Disorientation
Date:06/07/99ISR Number: 3285488-4Report Type:Periodic Company Report #200381 Age:35 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaria<br />
Coma<br />
03-Apr-2012 09:37 AM Page: 118
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Drug Ineffective<br />
Chills Report Source Product Role Manufacturer Route Dose Duration<br />
Health<br />
Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Other Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285489-6Report Type:Periodic Company Report #200388 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Health Lariam Tablets<br />
Coma Professional (<strong>Mefloquine</strong><br />
Pyrexia Hydrochloride) 250<br />
Panic Attack Mg PS ORAL 250 MG 1 PER<br />
Tremor<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285530-0Report Type:Periodic Company Report #200733 Age:65 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam Tablets<br />
Headache<br />
(<strong>Mefloquine</strong><br />
Palpitations Hydrochloride) 250<br />
Dizziness Mg PS ORAL 250 MG 1 PER<br />
Chills<br />
WEEK ORAL<br />
Panic Attack<br />
Estrogen (Estrogens<br />
Alopecia Nos) C<br />
Nausea<br />
Progestin (Progestin<br />
Hyperhidrosis Nos) C<br />
Chest Pain<br />
Tremor<br />
Anxiety<br />
Decreased Appetite<br />
Vomiting<br />
Paraesthesia<br />
Date:06/07/99ISR Number: 3285531-2Report Type:Periodic Company Report #200897 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Asa (Aspirin)<br />
C<br />
Ibuprofen
(Ibuprofen) C<br />
03-Apr-2012 09:37 AM Page: 119
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285532-4Report Type:Periodic Company Report #201043 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Other Lariam Tablets<br />
Asthenia<br />
(<strong>Mefloquine</strong><br />
Ear Pain Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285533-6Report Type:Periodic Company Report #202058 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urticaria Consumer Lariam Tablets<br />
Ageusia<br />
(<strong>Mefloquine</strong><br />
Pruritus Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285534-8Report Type:Periodic Company Report #202788 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Apathy Consumer Lariam Tablets<br />
Flat Affect<br />
(<strong>Mefloquine</strong><br />
Aggression Hydrochloride) 250<br />
Mg PS ORAL 1 PER WEEK<br />
ORAL<br />
Date:06/07/99ISR Number: 3285535-XReport Type:Periodic Company Report #202801 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aggression Other Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285536-1Report Type:Periodic Company Report #202966 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Consumer Lariam Tablets<br />
Dizziness<br />
(<strong>Mefloquine</strong><br />
Nausea Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Hydrochlorothiazide
(Hydrochlorothiazide<br />
) C<br />
03-Apr-2012 09:37 AM Page: 120
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285537-3Report Type:Periodic Company Report #55872 Age:38 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Consumer Lariam Tablets<br />
Chest Pain Health (<strong>Mefloquine</strong><br />
Vomiting Professional Hydrochloride) 250<br />
Decreased Appetite Mg PS ORAL 250 MG 1 PER<br />
Muscle Contractions<br />
WEEK ORAL<br />
Involuntary<br />
Hepatitis A Vaccine<br />
Headache<br />
(Hepatitis A<br />
Palpitations Vaccine) C<br />
Paraesthesia<br />
Extrasystoles<br />
Lethargy<br />
Cough<br />
Upper Respiratory Tract<br />
Infection<br />
Pyrexia<br />
Insomnia<br />
Depression<br />
Oropharyngeal Pain<br />
Temperature Intolerance<br />
Weight Decreased<br />
Influenza Like Illness<br />
Date:06/07/99ISR Number: 3285538-5Report Type:Periodic Company Report #72278 Age:24 YR Gender: I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Consumer Lariam Tablets<br />
Gastrointestinal Disorder<br />
(<strong>Mefloquine</strong><br />
Amnesia Hyrdrochloride) 250<br />
Pain Mg PS ORAL 250 MG 1 PER<br />
Pyrexia WEEK ORAL 35 DAY<br />
Ear Pain<br />
Headache<br />
Dizziness<br />
Dyspnoea<br />
Fatigue<br />
Asthenia<br />
Confusional State<br />
Malaise<br />
Palpitations<br />
Date:06/07/99ISR Number: 3285539-7Report Type:Periodic Company Report #97647 Age:58 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Flushing Consumer Lariam Tablets<br />
Pruritus<br />
(<strong>Mefloquine</strong><br />
Dermatitis Hydrochloride) 250
Rhinorrhoea Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Premarin (Estrogens,<br />
Conjugated)<br />
C<br />
03-Apr-2012 09:37 AM Page: 121
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285592-0Report Type:Periodic Company Report #103538 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Face Oedema Health Lariam Tablets<br />
Coma Professional (<strong>Mefloquine</strong><br />
Oropharyngeal Blistering Hydrochloride) 250<br />
Diarrhoea Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Vivotif "Berna"<br />
(Typhoid Vaccines) SS ORAL 1 DOSE FORM 1<br />
PER 2 DAY<br />
ORAL<br />
Atarax<br />
C<br />
Date:06/07/99ISR Number: 3285593-2Report Type:Periodic Company Report #103551 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Consumer Lariam Tablets<br />
Chills<br />
(<strong>Mefloquine</strong><br />
Nausea Hydrochloride) 250<br />
Cold Sweat Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Cytotec<br />
C<br />
Hormone Replacement<br />
Therapy<br />
C<br />
Date:06/07/99ISR Number: 3285594-4Report Type:Periodic Company Report #103573 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Other Lariam Tablets<br />
Cold Sweat<br />
(<strong>Mefloquine</strong><br />
Hypertension Hydrochloride) 250<br />
Dizziness Mg PS ORAL 250 MG 1 PER<br />
Nausea<br />
WEEK ORAL<br />
Cardizem<br />
C<br />
Vasotec<br />
C<br />
Date:06/07/99ISR Number: 3285595-6Report Type:Periodic Company Report #104147 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Decreased Appetite Other Lariam Tablets<br />
Nausea<br />
(<strong>Mefloquine</strong><br />
Tremor Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:06/07/99ISR Number: 3285596-8Report Type:Periodic Company Report #104270 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Consumer Lariam Tablets<br />
Gastrointestinal Disorder<br />
(<strong>Mefloquine</strong><br />
Flatulence Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
03-Apr-2012 09:37 AM Page: 122
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285597-XReport Type:Periodic Company Report #104454 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Spasms Consumer Lariam Tablets<br />
Fatigue Health (<strong>Mefloquine</strong><br />
Neck Pain Professional Hydrochloride) 250<br />
Chest Pain Mg PS ORAL 250 MG 1 PER<br />
Pain In Extremity<br />
WEEK ORAL<br />
Tri-Levlen<br />
C<br />
Date:06/07/99ISR Number: 3285598-1Report Type:Periodic Company Report #105183 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Flushing Other Lariam Tablets<br />
Hypertension<br />
(<strong>Mefloquine</strong><br />
Burning Sensation Hydrochloride) 250<br />
Abdominal Pain Mg PS ORAL 250 MG 1 PER<br />
Headache<br />
WEEK ORAL<br />
Palpitations<br />
Date:06/07/99ISR Number: 3285599-3Report Type:Periodic Company Report #105877 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Consumer Lariam Tablets<br />
Breast Discharge<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285600-7Report Type:Periodic Company Report #106528 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Corneal Dystrophy Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 26 PER<br />
YEAR ORAL<br />
Date:06/07/99ISR Number: 3285601-9Report Type:Periodic Company Report #106670 Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypertension Consumer Lariam Tablets
Tinnitus Health (<strong>Mefloquine</strong><br />
Tachycardia Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
DOSE ORAL<br />
03-Apr-2012 09:37 AM Page: 123
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285602-0Report Type:Periodic Company Report #106792 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dysuria Consumer Lariam Tablets<br />
Burning Sensation<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/07/99ISR Number: 3285603-2Report Type:Periodic Company Report #106988 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Serum Ferritin Abnormal Consumer Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Baycol (Cerivastatin<br />
Sodium) SS ORAL .3 MG DAILY<br />
ORAL<br />
Date:06/07/99ISR Number: 3285604-4Report Type:Periodic Company Report #108987 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Consumer Lariam Tablets<br />
Nausea Other (<strong>Mefloquine</strong><br />
Myalgia Hydrochloride) 250<br />
Anxiety Mg PS ORAL 250 MG 1 PER<br />
Arthralgia<br />
WEEK ORAL<br />
Aggression<br />
Cough<br />
Pyrexia<br />
Date:06/07/99ISR Number: 3285605-6Report Type:Periodic Company Report #109650 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Consumer Lariam Tablets<br />
Joint Swelling Health (<strong>Mefloquine</strong><br />
Fatigue Professional Hydrochloride) 250<br />
Dizziness Mg PS ORAL 250 MG 1 PER<br />
Asthenia<br />
WEEK ORAL<br />
Lipitor<br />
C<br />
Lopressor<br />
C<br />
Date:06/07/99ISR Number: 3285606-8Report Type:Periodic Company Report #110173 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Metrorrhagia Consumer Lariam Tablets<br />
Menorrhagia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Ortho-Cyclen<br />
C<br />
03-Apr-2012 09:37 AM Page: 124
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/07/99ISR Number: 3285607-XReport Type:Periodic Company Report #110773 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Distress Other Lariam Tablets<br />
Emotional Disorder<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285608-1Report Type:Periodic Company Report #111195 Age:60 YR Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Hyperpigmentation Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285609-3Report Type:Periodic Company Report #112630 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Other Lariam Tablets<br />
Visual Impairment<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/07/99ISR Number: 3285610-XReport Type:Periodic Company Report #200634 Age:10 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Antinuclear Antibody Health Lariam Tablets<br />
Positive Professional (<strong>Mefloquine</strong><br />
Neutropenia Hydrochloride) PS ORAL ORAL<br />
Date:06/07/99ISR Number: 3285611-1Report Type:Periodic Company Report #200728 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Alkaline Consumer Lariam Tablets<br />
Phosphatase Increased<br />
(<strong>Mefloquine</strong><br />
Hepatic Enzyme Increased Hydrochloride) 250<br />
Condition Aggravated Mg PS ORAL 250 MG 1 PER<br />
Gamma-Glutamyltransferase<br />
WEEK ORAL<br />
Increased
Date:06/09/99ISR Number: 3279221-XReport Type:Expedited (15-DaCompany Report #105648 Age: Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Cardiac Murmur<br />
Maternal Drugs Affecting<br />
Foetus<br />
Antepartum Haemorrhage<br />
03-Apr-2012 09:37 AM Page: 125
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Premature Baby<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/15/99ISR Number: 3285102-8Report Type:Expedited (15-DaCompany Report #104882 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Circulatory Collapse Foreign Lariam Tablets<br />
Initial or Prolonged Asthenia Consumer (Melfloquine<br />
Tachycardia Health Hydrochloride) 250<br />
Confusional State Professional Mg PS ORAL ORAL<br />
Dizziness<br />
Pyrexia<br />
Disturbance In Attention<br />
Helicobacter Gastritis<br />
Feeling Abnormal<br />
Muscle Spasms<br />
Anxiety<br />
Haematochezia<br />
Cardiovascular Disorder<br />
Arrhythmia<br />
Paraesthesia<br />
Abdominal Pain<br />
Balance Disorder<br />
Restlessness<br />
Ventricular Extrasystoles<br />
Panic Attack<br />
Date:06/15/99ISR Number: 3285108-9Report Type:Expedited (15-DaCompany Report #207710 Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Condition Aggravated Foreign Lariam Tablets<br />
Initial or Prolonged Menometrorrhagia Other (<strong>Mefloquine</strong><br />
Required Hydrochloride) 250<br />
Intervention to Mg PS ORAL 250 MG 1 PER<br />
Prevent Permanent<br />
WEEK ORAL<br />
Impairment/Damage<br />
Date:06/17/99ISR Number: 3286062-6Report Type:Expedited (15-DaCompany Report #207703 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abortion Spontaneous Foreign Lariam Tablets
Initial or Prolonged Other (<strong>Mefloquine</strong><br />
Hydrochloride) 50 Mg PS ORAL 50 MG DAILY<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 126
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/17/99ISR Number: 3286067-5Report Type:Expedited (15-DaCompany Report #207919 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Affective Disorder Foreign Lariam Tablets<br />
Nausea Other (<strong>Mefloquine</strong><br />
Abnormal Behaviour Hydrochloride) 250<br />
Crying Mg PS ORAL 250 MG 1 PER<br />
Dizziness<br />
WEEK ORAL<br />
Mental Disorder<br />
Suicidal Ideation<br />
Visual Impairment<br />
Mania<br />
Date:06/17/99ISR Number: 3286250-9Report Type:Expedited (15-DaCompany Report #207220 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Purulent Discharge Foreign Lariam Tablets<br />
Initial or Prolonged Lymphocyte Count Health (<strong>Mefloquine</strong><br />
Decreased Professional Hydrochloride) 250mg PS ORAL 250MG 1 PER<br />
Monocyte Count Decreased Other WEEK ORAL<br />
Neutrophil Count<br />
Increased<br />
Headache<br />
Haematocrit Decreased<br />
Nasal Congestion<br />
Lymphocyte Morphology<br />
Abnormal<br />
Bacterial Infection<br />
Foreign Travel<br />
Sinusitis<br />
Pyrexia<br />
Date:06/17/99ISR Number: 3287250-5Report Type:Expedited (15-DaCompany Report #207840 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Torticollis Foreign Lariam Tablets<br />
Initial or Prolonged Gastritis Health (<strong>Mefloquine</strong><br />
Influenza Like Illness Professional Hydrochloride) 250<br />
Lymphadenopathy Other Mg PS ORAL 250 MG 1 PER<br />
Flushing WEEK ORAL 122 DAY<br />
Heart Rate Increased<br />
Inflammation<br />
White Blood Cell Count<br />
Increased<br />
Pharyngeal Oedema<br />
Myalgia<br />
Oropharyngeal Pain<br />
Ear Disorder<br />
Malaise
Bacterial Infection<br />
03-Apr-2012 09:37 AM Page: 127
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/22/99ISR Number: 3289600-2Report Type:Expedited (15-DaCompany Report #112331 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Premature Rupture Of Foreign Lariam Tablets<br />
Initial or Prolonged Membranes Health (<strong>Mefloquine</strong><br />
Haemoglobin Decreased Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK<br />
Date:06/25/99ISR Number: 3292126-3Report Type:Expedited (15-DaCompany Report #208431 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Visual Acuity Reduced Consumer Lariam Tablets<br />
Required Vision Blurred Health (<strong>Mefloquine</strong><br />
Intervention to Cataract Operation Professional Hydrochloride) 250<br />
Prevent Permanent Foreign Travel Mg PS ORAL 250 MG 1 PER<br />
Impairment/Damage Cataract WEEK ORAL<br />
Date:06/29/99ISR Number: 3294566-5Report Type:Expedited (15-DaCompany Report #208430 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hearing Impaired Foreign Lariam Tablets<br />
Other<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/29/99ISR Number: 3294570-7Report Type:Expedited (15-DaCompany Report #208480 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Foreign Lariam Tablets<br />
Drug Ineffective Health (<strong>Mefloquine</strong><br />
Haemolytic Anaemia Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/30/99ISR Number: 3295144-4Report Type:Expedited (15-DaCompany Report #208901 Age:61 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Decreased Appetite<br />
Gastric Haemorrhage<br />
Mucosal Erosion<br />
Pyrexia<br />
Constipation<br />
Malaria<br />
Haematemesis<br />
Dyspnoea
Ascites<br />
Oesophageal Disorder<br />
Abdominal Distension<br />
Chills<br />
Drug Ineffective<br />
Haematocrit Decreased<br />
Oedema Peripheral<br />
Blood Albumin Decreased<br />
03-Apr-2012 09:37 AM Page: 128
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Gastritis Erosive<br />
Haemoglobin Decreased<br />
Condition Aggravated Report Source Product Role Manufacturer Route Dose Duration<br />
Faeces Discoloured Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Rezulin<br />
C<br />
Aspirin<br />
C<br />
Glucotrol<br />
C<br />
Date:07/09/99ISR Number: 3300329-4Report Type:Expedited (15-DaCompany Report #209222 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Supraventricular Foreign Lariam Tablets<br />
Initial or Prolonged Tachycardia Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:07/09/99ISR Number: 3300636-5Report Type:Expedited (15-DaCompany Report #203230 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paraesthesia Health Lariam Tablets<br />
Initial or Prolonged Cough Professional (<strong>Mefloquine</strong><br />
Required Nausea Hydrochloride) 250<br />
Intervention to Anxiety Mg PS ORAL 250 MG 1 PER<br />
Prevent Permanent Asthma WEEK ORAL<br />
Impairment/Damage Panic Attack<br />
Dyspnoea<br />
Tinnitus<br />
Dehydration<br />
Chest Discomfort<br />
Vomiting<br />
Pyrexia<br />
Date:07/13/99ISR Number: 3302667-8Report Type:Expedited (15-DaCompany Report #208480 Age:21 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Ineffective Foreign Lariam PS ORAL 250 MG 1 PER<br />
Pyrexia Health WEEK ORAL<br />
Haemolytic Anaemia<br />
Professional<br />
Malaria
Date:07/14/99ISR Number: 3303518-8Report Type:Direct Company Report # Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation Lariam 250mg PS ORAL 1 PILL WEEKLY<br />
Skin Discolouration<br />
ORALLY<br />
Confusional State Estra C<br />
Nail Disorder<br />
Pain In Extremity<br />
03-Apr-2012 09:37 AM Page: 129
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/16/99ISR Number: 3305371-5Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Consumer Lariam PS ORAL 250 MG 1X WK<br />
Neurological Symptom<br />
ORALLY<br />
Hallucination, Tactile<br />
Restlessness<br />
Palpitations<br />
Agitation<br />
Date:07/21/99ISR Number: 3308458-6Report Type:Expedited (15-DaCompany Report #210488 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Thrombotic Foreign Lariam Tablets<br />
Initial or Prolonged Thrombocytopenic Purpura Other (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
PER ONE DOSE<br />
ORAL<br />
Date:07/23/99ISR Number: 3309996-2Report Type:Expedited (15-DaCompany Report #203230 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tinnitus Health Lariam Tablets<br />
Initial or Prolonged Paraesthesia Professional (<strong>Mefloquine</strong><br />
Required Dehydration Hydrochloride) 250<br />
Intervention to Chest Discomfort Mg PS ORAL 250 MG 1 PER<br />
Prevent Permanent Anxiety WEEK ORAL<br />
Impairment/Damage Vomiting<br />
Cough<br />
Pyrexia<br />
Dyspnoea<br />
Date:07/30/99ISR Number: 3315299-2Report Type:Expedited (15-DaCompany Report #210600 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Consumer Lariam Tablets<br />
Initial or Prolonged Coordination Abnormal Health (<strong>Mefloquine</strong><br />
Bipolar Disorder Professional Hydrochloride) 250<br />
Hypersensitivity Mg PS ORAL 250 MG 1 PER<br />
Tremor<br />
WEEK ORAL<br />
Pyrexia<br />
Lithium (Lithium<br />
Flatulence Nos) SS ORAL 300 MG DAILY<br />
Circulatory Collapse<br />
ORAL<br />
Balance Disorder Lorazepam C<br />
Chest Discomfort Atenolol C<br />
Palpitations<br />
Diarrhoea
Disorientation<br />
Psychotic Disorder<br />
Abdominal Pain<br />
Chest Pain<br />
Vomiting<br />
Syncope<br />
03-Apr-2012 09:37 AM Page: 130
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/02/99ISR Number: 3316743-7Report Type:Expedited (15-DaCompany Report #211408 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Foreign <strong>Mefloquine</strong><br />
Dizziness Other Hydrochloride(Mefloq<br />
Suicidal Ideation<br />
uine Hydrochloride)<br />
250 Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Antibiotics Nos C<br />
Date:08/02/99ISR Number: 3316746-2Report Type:Expedited (15-DaCompany Report #211405 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaria Foreign Lariam Tablets<br />
Initial or Prolonged Mood Swings Other (<strong>Mefloquine</strong><br />
Abnormal Dreams Hydrochloride) PS ORAL 500 MG 1 PER<br />
Paraesthesia<br />
WEEK ORAL<br />
Condition Aggravated Flucloxacillin C<br />
Pyrexia Amoxycillin C<br />
Malaise<br />
Toxicity To Various<br />
Agents<br />
Chest Pain<br />
Foreign Travel<br />
Agitation<br />
Personality Change<br />
Chills<br />
Headache<br />
Diarrhoea<br />
Anxiety<br />
Date:08/03/99ISR Number: 3316753-XReport Type:Expedited (15-DaCompany Report #210600 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Consumer Lariam Tablets<br />
Initial or Prolonged Abdominal Pain Health (<strong>Mefloquine</strong><br />
Pruritus Professional Hydrochloride) 250<br />
White Blood Cell Count Mg PS ORAL 250 MG 1 PER<br />
Increased<br />
WEEK ORAL<br />
Pyrexia<br />
Lithium (Lithium<br />
Mania Nos) SS ORAL 300 MG DAILY<br />
Drug Hypersensitivity<br />
ORAL<br />
Urticaria Lorazepam C<br />
Nausea Atenolol C<br />
Circulatory Collapse<br />
Coordination Abnormal<br />
Vomiting Projectile<br />
Chest Pain<br />
Medication Error
Chest Discomfort<br />
Diarrhoea<br />
Bipolar Disorder<br />
Palpitations<br />
Tremor<br />
Flatulence<br />
03-Apr-2012 09:37 AM Page: 131
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/03/99ISR Number: 3316767-XReport Type:Expedited (15-DaCompany Report #211570 Age:1 HR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Heart Valve Replacement Foreign Lariam Tablets<br />
Maternal Drugs Affecting Health (<strong>Mefloquine</strong><br />
Foetus Professional Hydrochloride) 250<br />
Heart Disease Congenital Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:08/10/99ISR Number: 3322744-5Report Type:Expedited (15-DaCompany Report #211814 Age:67 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diabetes Mellitus Foreign Lariam Tablets<br />
Initial or Prolonged Diarrhoea Health (<strong>Mefloquine</strong><br />
Nephrolithiasis Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:08/10/99ISR Number: 3322745-7Report Type:Expedited (15-DaCompany Report #211998 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pain In Extremity Foreign Lariam Tablets<br />
Gastritis Erosive Health (<strong>Mefloquine</strong><br />
Gamma-Glutamyltransferase Professional Hydrochloride) 250<br />
Increased Mg PS ORAL 250 MG 1 PER<br />
Polyneuropathy<br />
WEEK ORAL<br />
Diverticulum<br />
Date:08/10/99ISR Number: 3322775-5Report Type:Expedited (15-DaCompany Report #206437 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Retinopathy Consumer Lariam Tablets<br />
Iris Atrophy Health (<strong>Mefloquine</strong><br />
Visual Impairment Professional Hydrochloride) 250<br />
Vitreous Disorder Mg PS ORAL 250 MG 1 PER<br />
Medication Error<br />
WEEK ORAL<br />
Visual Field Defect<br />
Date:08/12/99ISR Number: 3324870-3Report Type:Expedited (15-DaCompany Report #208901 Age:61 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Ascites<br />
Oesophageal Disorder<br />
Drug Ineffective<br />
Pyrexia
Malaria<br />
Oedema Peripheral<br />
Gastric Haemorrhage<br />
Blood Albumin Decreased<br />
Chills<br />
Abdominal Distension<br />
Dyspnoea<br />
Haematemesis<br />
03-Apr-2012 09:37 AM Page: 132
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Phlebitis<br />
Hepatitis<br />
Constipation Report Source Product Role Manufacturer Route Dose Duration<br />
Faeces Discoloured Consumer Lariam Tablets<br />
Intervertebral Disc<br />
(<strong>Mefloquine</strong><br />
Disorder Hydrochloride) 250<br />
Mucosal Erosion Mg PS ORAL 250 MG 1 PER<br />
Haemoglobin Decreased<br />
WEEK ORAL<br />
Haematocrit Decreased Rezulin C<br />
Gastrointestinal Aspirin C<br />
Haemorrhage Glucotrol C<br />
Date:08/12/99ISR Number: 3324875-2Report Type:Expedited (15-DaCompany Report #212598 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haematuria Health Lariam Tablets<br />
Nephrolithiasis Professional (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:08/17/99ISR Number: 3327902-1Report Type:Expedited (15-DaCompany Report #205574 Age:46 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Foreign Lariam Tablets<br />
Initial or Prolonged Eye Movement Disorder Consumer (<strong>Mefloquine</strong><br />
Visual Field Defect Health Hydrochloride) 250<br />
Vision Blurred Professional Mg PS ORAL 250 MG 1 PER<br />
Nausea<br />
WEEK ORAL<br />
Nystagmus<br />
Dizziness<br />
Arthropod Bite<br />
Vertigo<br />
Cerebellar Syndrome<br />
Disturbance In Attention<br />
Coordination Abnormal<br />
Phobia<br />
Date:08/17/99ISR Number: 3327972-0Report Type:Expedited (15-DaCompany Report #202262 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viiith Nerve Lesion Consumer Lariam Tablets<br />
Deafness<br />
(<strong>Mefloquine</strong><br />
Diarrhoea Hydrochloride) 250<br />
Restlessness Mg PS ORAL 250 MG 1 PER<br />
Vertigo<br />
WEEK ORAL<br />
Anxiety Hormone Replacement C<br />
Intervertebral Disc<br />
Protrusion
Ototoxicity<br />
Insomnia<br />
Flatulence<br />
Nystagmus<br />
Abdominal Pain Lower<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 133
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/17/99ISR Number: 3328019-2Report Type:Expedited (15-DaCompany Report #211301 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brachial Plexopathy Foreign Lariam Tablets<br />
Paralysis Health (<strong>Mefloquine</strong><br />
Foreign Travel Professional Hydrochloride) 250<br />
Neuropathy Peripheral Mg PS ORAL 250 MG 1 PER<br />
Muscular Weakness<br />
Desmopressin<br />
(Desmopressin<br />
Acetate)<br />
C<br />
WEEK ORAL<br />
Date:08/17/99ISR Number: 3328996-XReport Type:Direct Company Report # Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia Health Mefloquin / 125 Mg PS 1TBL/ 1 TBL<br />
Dyspepsia<br />
Professional<br />
Asthenia<br />
Fatigue<br />
Decreased Appetite<br />
Malaise<br />
Diarrhoea<br />
Weight Decreased<br />
Apathy<br />
Depression<br />
Dysphoria<br />
Mental Impairment<br />
Decreased Activity<br />
Date:08/19/99ISR Number: 3330225-8Report Type:Expedited (15-DaCompany Report #213079 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Myelodysplastic Syndrome Foreign Lariam Tablets<br />
Seronegative Arthritis Other (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Ciprofloxacin<br />
(Ciprofloxacin) SS ORAL 500 MG DAILY<br />
ORAL<br />
Doxycycline<br />
C<br />
Erythromycin<br />
C<br />
Date:08/24/99ISR Number: 3333682-6Report Type:Expedited (15-DaCompany Report #213259 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Foreign Lariam Tablets
Initial or Prolonged Erythema Health (<strong>Mefloquine</strong><br />
Urticaria Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 134
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/25/99ISR Number: 3333861-8Report Type:Direct Company Report # Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Larium / 250 Mg /<br />
Initial or Prolonged Sedation Hoffman La Roche PS Hoffman Laroche 250MG ONCE A<br />
Other Serious Bite WEEK<br />
Movement Disorder<br />
Loss Of Consciousness<br />
Eye Movement Disorder<br />
Crying<br />
Date:08/26/99ISR Number: 3334601-9Report Type:Direct Company Report # Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Lariam, 250mg, La<br />
Amnesia Roche PS La Roche 1 PILL/WK<br />
Fatigue<br />
(LATE<br />
Abnormal Dreams JUNE-JULY) 6 WK<br />
Dizziness Immunization For Tb C<br />
Cold Sweat<br />
Immunization For<br />
Malaise Polio C<br />
Chills<br />
Immunization For<br />
Pyrexia Yellow Fever C<br />
Panic Attack<br />
Immunization For Hep<br />
Anxiety A C<br />
Date:08/26/99ISR Number: 3335379-5Report Type:Expedited (15-DaCompany Report #213489 Age:1 HR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Deafness Congenital Other Lariam Tablets<br />
Maternal Drugs Affecting<br />
(<strong>Mefloquine</strong><br />
Foetus Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:08/31/99ISR Number: 3338457-XReport Type:Expedited (15-DaCompany Report #213701 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation Foreign Lariam Tablets<br />
Initial or Prolonged Malaria Other (<strong>Mefloquine</strong><br />
Personality Change Hydrochloride) 250<br />
Drug Ineffective Mg PS ORAL 250 MG 1 PER<br />
Sedation<br />
WEEK ORAL<br />
Toxicity To Various Polio Vaccination C<br />
Agents<br />
Yellow Fever Vaccine C<br />
Tetanus Vaccines C<br />
Havrix<br />
C
Typhoid Vaccines C<br />
03-Apr-2012 09:37 AM Page: 135
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/31/99ISR Number: 3338460-XReport Type:Expedited (15-DaCompany Report #213852 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Tinnitus Foreign Lariam Tablets<br />
Intervention to Hearing Impaired Health (<strong>Mefloquine</strong><br />
Prevent Permanent Professional Hydrochloride) 250<br />
Impairment/Damage Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Thyroid Medication C<br />
Date:09/02/99ISR Number: 3339917-8Report Type:Expedited (15-DaCompany Report #210600 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Circulatory Collapse Consumer Lariam Tablets<br />
Initial or Prolonged White Blood Cell Count Health (<strong>Mefloquine</strong><br />
Increased Professional Hydrochloride) 250<br />
Chest Discomfort Mg PS ORAL 250 MG 1 PER<br />
Vomiting Projectile<br />
WEEK ORAL<br />
Drug Hypersensitivity<br />
Lithium (Lithium<br />
Diarrhoea Nos) SS ORAL 300 MG DAILY<br />
Coordination Abnormal<br />
ORAL<br />
Mania Lorazepam C<br />
Palpitations Atenolol C<br />
Urticaria<br />
Pyrexia<br />
Flatulence<br />
Insomnia<br />
Abdominal Pain<br />
Pruritus<br />
Chest Pain<br />
Bipolar Disorder<br />
Tremor<br />
Date:09/03/99ISR Number: 3342519-0Report Type:Expedited (15-DaCompany Report #214112 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Intracranial Pressure Foreign Lariam Tablets<br />
Initial or Prolonged Increased Health (<strong>Mefloquine</strong><br />
Fatigue Professional Hydrochloride) 250<br />
Papilloedema Mg PS ORAL 250 MG 1 PER<br />
Diplopia<br />
WEEK ORAL<br />
Oedema Peripheral<br />
Contraceptive<br />
Headache (Contraceptive Nos) C<br />
Date:09/09/99ISR Number: 3344621-6Report Type:Expedited (15-DaCompany Report #214408 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Muscle Rigidity Consumer Lariam Tablets<br />
Malaise<br />
(<strong>Mefloquine</strong><br />
Pain Hydrochloride) 250<br />
Tremor Mg PS ORAL 250 MG 1 PER<br />
Foreign Travel<br />
WEEK ORAL<br />
Parkinson'S Disease<br />
03-Apr-2012 09:37 AM Page: 136
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/14/99ISR Number: 3347516-7Report Type:Expedited (15-DaCompany Report #214499 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Alopecia Other Lariam Tablets<br />
Amenorrhoea<br />
(<strong>Mefloquine</strong><br />
Abdominal Pain Hydrochloride) 250<br />
Infertility Mg PS ORAL 250 MG 1 PER<br />
Diarrhoea<br />
WEEK ORAL<br />
Date:09/14/99ISR Number: 3347543-XReport Type:Expedited (15-DaCompany Report #214801 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis C Other Lariam Tablets<br />
Oedema<br />
(<strong>Mefloquine</strong><br />
Dyspnoea Hydrochloride) 250<br />
Condition Aggravated Mg PS ORAL 250 MG 1 PER<br />
Heart Rate Irregular<br />
WEEK ORAL<br />
Date:09/14/99ISR Number: 3347546-5Report Type:Expedited (15-DaCompany Report #214510 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myocarditis Foreign Lariam Tablets<br />
Initial or Prolonged Sweat Gland Disorder Health (<strong>Mefloquine</strong><br />
Hormone Level Abnormal Professional Hydrochloride) 250<br />
Thyroid Atrophy Mg PS ORAL 250 MG 1 PER<br />
Constipation<br />
WEEK ORAL<br />
Amenorrhoea<br />
Echo Virus Infection<br />
Viral Infection<br />
Decreased Appetite<br />
Alcohol Intolerance<br />
Asthenia<br />
Oligomenorrhoea<br />
Paraesthesia<br />
Feeling Cold<br />
Hypotension<br />
Blood Thyroid Stimulating<br />
Hormone Abnormal<br />
Hypothyroidism<br />
Bradycardia<br />
Autonomic Nervous System<br />
Imbalance<br />
Syncope<br />
Date:09/22/99ISR Number: 3354848-5Report Type:Expedited (15-DaCompany Report #215354 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Embolism Arterial Foreign Lariam Tablets<br />
Initial or Prolonged Peripheral Ischaemia Health (<strong>Mefloquine</strong><br />
Cardiac Fibrillation Professional Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 137
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/22/99ISR Number: 3354987-9Report Type:Expedited (15-DaCompany Report #215735 Age:47 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Prochlorperazine C<br />
Date:09/23/99ISR Number: 3356130-9Report Type:Expedited (15-DaCompany Report #215428 Age:66 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Consumer Lariam Tablets<br />
Initial or Prolonged Oliguria Other (<strong>Mefloquine</strong><br />
Panic Reaction Hydrochloride) 250<br />
Agitation Mg PS ORAL 250 MG 1<br />
Dehydration<br />
PER WEEK<br />
Urinary Retention<br />
ORAL<br />
Tamoxifen<br />
C<br />
Fosamax<br />
C<br />
Date:09/23/99ISR Number: 3356132-2Report Type:Expedited (15-DaCompany Report #215861 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Circulatory Collapse Foreign Lariam Tablets<br />
Electrocardiogram Qt Health (<strong>Mefloquine</strong><br />
Prolonged Professional Hydrochloride) 250<br />
Heart Rate Decreased Mg PS ORAL 250 MG 1<br />
PER WEEK<br />
ORAL<br />
Date:09/23/99ISR Number: 3356134-6Report Type:Expedited (15-DaCompany Report #214112 Age:18 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Oedema Peripheral Foreign Lariam Tablets<br />
Initial or Prolonged Benign Intracranial Health (<strong>Mefloquine</strong><br />
Hypertension Professional Hydrochloride) 250<br />
Vith Nerve Paralysis Mg PS ORAL 250 MG 1<br />
Papilloedema<br />
PER WEEK<br />
Diplopia<br />
ORAL<br />
Contraceptive<br />
C<br />
Date:09/23/99ISR Number: 3356136-XReport Type:Expedited (15-DaCompany Report #215743 Age:47 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Foreign Lariam Tablets<br />
Initial or Prolonged Malaria Health (<strong>Mefloquine</strong><br />
Abdominal Pain Professional Hydrochloride) 250<br />
Nodal Arrhythmia Mg PS ORAL 250 MG 1<br />
Paraesthesia<br />
PER WEEK<br />
Syncope<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 138
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/23/99ISR Number: 3356139-5Report Type:Expedited (15-DaCompany Report #215573 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Screaming Foreign Lariam Tablets<br />
Initial or Prolonged Anxiety Other (<strong>Mefloquine</strong><br />
Dizziness Hydrochloride) 250<br />
Disturbance In Attention Mg PS ORAL 250 MG 1<br />
Crying<br />
PER WEEK<br />
Tremor<br />
ORAL<br />
Arrhythmia<br />
Emotional Disorder<br />
Confusional State<br />
Hyperventilation<br />
Nausea<br />
Arthralgia<br />
Headache<br />
Pain In Extremity<br />
Date:09/27/99ISR Number: 3359736-6Report Type:Expedited (15-DaCompany Report #215834 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thrombotic Foreign Lariam Tablets<br />
Thrombocytopenic Purpura Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:09/28/99ISR Number: 3360051-5Report Type:Expedited (15-DaCompany Report #215428 Age:66 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Oliguria Consumer Lariam Tablets<br />
Initial or Prolonged Insomnia Health (<strong>Mefloquine</strong><br />
Dehydration Professional Hydrochloride) 250<br />
Panic Reaction Mg PS ORAL 250 MG 1 PER<br />
Anxiety<br />
WEEK<br />
Urinary Retention<br />
Tamoxifen (Tamoxifen<br />
Agitation Citrate) C<br />
Circadian Rhythm Sleep<br />
Fosamax (Alendronate<br />
Disorder Sodium) C<br />
Date:09/28/99ISR Number: 3360064-3Report Type:Expedited (15-DaCompany Report #210600 Age:59 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Chest Pain<br />
Vomiting Projectile<br />
Mania<br />
Coordination Abnormal
Bipolar Disorder<br />
White Blood Cell Count<br />
Increased<br />
Drug Hypersensitivity<br />
Psychotic Disorder<br />
Circulatory Collapse<br />
Pruritus<br />
Pyrexia<br />
03-Apr-2012 09:37 AM Page: 139
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Chest Discomfort<br />
Urticaria<br />
Insomnia Report Source Product Role Manufacturer Route Dose Duration<br />
Abdominal Pain Consumer Lariam Tablets<br />
Palpitations Health (<strong>Mefloquine</strong><br />
Disorientation Professional Hydrochloride) 250<br />
Tremor Mg PS ORAL 250 MG 1 PER<br />
Vomiting<br />
Diarrhoea<br />
WEEK<br />
Lithium (Lithium<br />
Nos) SS ORAL 300 MG DAILY<br />
Lorazepam<br />
(Lorazepam)<br />
Atenolol (Atenolol)<br />
C<br />
C<br />
Date:09/28/99ISR Number: 3360067-9Report Type:Expedited (15-DaCompany Report #216020 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eclampsia Foreign Lariam Tablets<br />
Pregnancy Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
1 WEEK<br />
Moneva (Ethinyl<br />
Estradiol/Gestodine) C<br />
Date:09/28/99ISR Number: 3360071-0Report Type:Expedited (15-DaCompany Report #215907 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Speech Disorder Health Lariam Tablets<br />
Nervousness Professional (<strong>Mefloquine</strong><br />
Psychotic Disorder Other Hydrochloride) 250<br />
Agitation Mg PS ORAL 250 MG 1 PER<br />
Suicidal Ideation<br />
Anxiety<br />
Oral Contraception<br />
(Oral Contraceptive<br />
Nos)<br />
C<br />
WEEK<br />
Date:09/28/99ISR Number: 3361765-3Report Type:Expedited (15-DaCompany Report #215928 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypervitaminosis Health Lariam Tablets<br />
Initial or Prolonged Hepatitis Professional (<strong>Mefloquine</strong><br />
Liver Function Test Hydrochloride) 250<br />
Abnormal Mg PS ORAL 250 MG 1 PER<br />
Blood Bilirubin Increased<br />
WEEK ORAL<br />
Foreign Travel<br />
Vitamin Nos (Vitamin<br />
Jaundice Nos) SS
Kernicterus<br />
03-Apr-2012 09:37 AM Page: 140
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/28/99ISR Number: 3361769-0Report Type:Expedited (15-DaCompany Report #211998 Age:61 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diverticulum Intestinal Foreign Lariam Tablets<br />
Initial or Prolonged Gastritis Erosive Health (<strong>Mefloquine</strong><br />
Required Paraesthesia Professional Hydrochloride) 250<br />
Intervention to Csf Protein Increased Mg PS ORAL 250 MG 1 PER<br />
Prevent Permanent Peripheral Sensory WEEK ORAL<br />
Impairment/Damage Neuropathy<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Pain In Extremity<br />
Date:10/01/99ISR Number: 3362900-3Report Type:Expedited (15-DaCompany Report #210600 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Pain Consumer Lariam Tablets<br />
Initial or Prolonged Hypersensitivity Health (<strong>Mefloquine</strong><br />
Condition Aggravated Professional Hydrochloride) 250<br />
Urticaria Mg PS ORAL 250 MG 1 PER<br />
Pyrexia<br />
WEEK ORAL<br />
Dry Skin<br />
Lithium (Lithium<br />
Circulatory Collapse Nos) SS ORAL 300 MG DAILY<br />
Coordination Abnormal<br />
ORAL<br />
Chest Discomfort<br />
Lorazepam<br />
Vomiting (Lorazepam) C<br />
Bipolar Disorder Atenolol (Atenolol) C<br />
Tremor<br />
White Blood Cell Count<br />
Increased<br />
Diarrhoea<br />
Pruritus<br />
Flatulence<br />
Insomnia<br />
Palpitations<br />
Vomiting Projectile<br />
Date:10/01/99ISR Number: 3363208-2Report Type:Expedited (15-DaCompany Report #216164 Age:13 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Foreign Lariam Tablets<br />
Initial or Prolonged Neuropathy Peripheral Health (<strong>Mefloquine</strong><br />
Fatigue Professional Hydrochloride) PS ORAL 250 MG 3 PER<br />
Aggression<br />
DAY ORAL<br />
Sleep Disorder<br />
Gait Disturbance<br />
Fansidar<br />
(Pyrimethamine/Sulad<br />
oxine)<br />
C
Date:10/01/99ISR Number: 3363211-2Report Type:Expedited (15-DaCompany Report #211301 Age:16 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paralysis Foreign Lariam Tablets<br />
Muscular Weakness Health (<strong>Mefloquine</strong><br />
Neuropathy Peripheral Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1 PER<br />
03-Apr-2012 09:37 AM Page: 141
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Desmopressin<br />
(Desmopressin<br />
Acetate)<br />
C<br />
WEEK ORAL<br />
Date:10/01/99ISR Number: 3363214-8Report Type:Expedited (15-DaCompany Report #213623 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Foreign Lariam Tablets<br />
Foetus Health (<strong>Mefloquine</strong><br />
Pregnancy Professional Hydrochloride) 250<br />
Foetal Distress Syndrome Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Jodetten (Potassium<br />
Iodine)<br />
C<br />
Vitaferro (Ferrous<br />
Sulfate)<br />
C<br />
Lafol (Folic Acid) C<br />
Date:10/05/99ISR Number: 3364678-6Report Type:Expedited (15-DaCompany Report #215428 Age:66 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Consumer Lariam Tablets<br />
Initial or Prolonged Urinary Retention Other (<strong>Mefloquine</strong><br />
Disability Agitation Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
Panic Attack<br />
WEEK ORAL<br />
Oliguria<br />
Tamoxifen (Tamoxifen<br />
Urinary Tract Infection Citrate) C<br />
Dehydration<br />
Fosamax (Alendronate<br />
Anxiety Sodium) C<br />
Date:10/05/99ISR Number: 3364679-8Report Type:Expedited (15-DaCompany Report #215735 Age:47 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Foreign Lariam Tablets<br />
Decreased Activity Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Prochlorperazine<br />
(Prochlorperazine) C<br />
Date:10/08/99ISR Number: 3369296-1Report Type:Expedited (15-DaCompany Report #216832 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Back Pain Health Lariam Tablets<br />
Initial or Prolonged Nervous System Disorder Professional (<strong>Mefloquine</strong>
Grand Mal Convulsion Hydrochloride) 250<br />
Movement Disorder Mg PS ORAL 250 MG 1<br />
Gait Disturbance<br />
PER WEEK<br />
Foreign Travel<br />
ORAL<br />
Loss Of Consciousness<br />
03-Apr-2012 09:37 AM Page: 142
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/13/99ISR Number: 3371543-7Report Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Skin Discolouration Other Lariam Tablets<br />
Emotional Disorder<br />
(<strong>Mefloquine</strong><br />
Injury Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
Lichen Planus<br />
WEEK ORAL<br />
Date:10/13/99ISR Number: 3371544-9Report Type:Expedited (15-DaCompany Report #216810 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Emotional Disorder Other Lariam Tablets<br />
Lichen Planus<br />
(<strong>Mefloquine</strong><br />
Skin Discolouration Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:10/13/99ISR Number: 3371546-2Report Type:Expedited (15-DaCompany Report #206437 Age:37 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Scintillating Scotoma Health Lariam Tablets<br />
Retinopathy Professional (<strong>Mefloquine</strong><br />
Retinal Depigmentation Other Hydrochloride) 250<br />
Iris Atrophy Mg PS ORAL 250 MG 1 PER<br />
Macular Oedema<br />
WEEK ORAL<br />
Visual Field Defect<br />
Pigmentation Disorder<br />
Visual Impairment<br />
Vitreous Disorder<br />
Date:10/13/99ISR Number: 3371556-5Report Type:Expedited (15-DaCompany Report #216859 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Skin Discolouration Other Lariam Tablets<br />
Lichen Planus<br />
(<strong>Mefloquine</strong><br />
Emotional Disorder Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:10/14/99ISR Number: 3372847-4Report Type:Expedited (15-DaCompany Report #217046 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Sudden Death Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250
Mg PS ORAL ORAL<br />
Na<br />
SS<br />
Paludrine<br />
(Choroguanide<br />
Hydrochloride) C<br />
Diane 35<br />
(Cyproterone<br />
Acetate/Ethinyl<br />
03-Apr-2012 09:37 AM Page: 143
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Estradiol)<br />
C<br />
Date:10/14/99ISR Number: 3372852-8Report Type:Expedited (15-DaCompany Report #216957 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Foreign Lariam Tablets<br />
Initial or Prolonged Fatigue Other (<strong>Mefloquine</strong><br />
Arthralgia Hydrochloride) 250<br />
Hepatic Fibrosis Mg PS ORAL 250 MG 1 PER<br />
Autoimmune Hepatitis<br />
WEEK ORAL<br />
Liver Function Test Na SS<br />
Abnormal<br />
Dpt Injection<br />
Bile Duct Stone<br />
(Diphtheria<br />
Blood Bilirubin Increased<br />
Toxoid/Pertussis<br />
Hepatic Pain<br />
Vaccine/Tetanus<br />
Jaundice Toxoid) C<br />
Hepatomegaly<br />
Immunoglobulin Human<br />
Normal (Globulin,<br />
Immune)<br />
C<br />
Date:10/14/99ISR Number: 3372855-3Report Type:Expedited (15-DaCompany Report #216944 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Abortion Spontaneous Foreign Lariam Tablets<br />
Maternal Drugs Affecting Other (<strong>Mefloquine</strong><br />
Foetus Hydrochloride) 250<br />
Limb Malformation Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Na<br />
SS<br />
Date:10/15/99ISR Number: 3372126-5Report Type:Direct Company Report # Age:67 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Health Lariam 250mg Po PS ORAL 250 MG PO QWK<br />
Acute Stress Disorder Professional TOOK 1/2<br />
TABLET BIW 3 MON<br />
Date:10/15/99ISR Number: 3374199-2Report Type:Expedited (15-DaCompany Report #216908 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Autoimmune Hepatitis Health Lariam Tablets<br />
Initial or Prolonged Blood Bilirubin Increased Professional (<strong>Mefloquine</strong><br />
Gamma-Glutamyltransferase Hydrochloride) 250mg PS ORAL 250 MG 1 PER<br />
Increased<br />
1 WEEK ORAL<br />
Jaundice<br />
Polio Vaccine<br />
Aspartate<br />
(Poliomyelitis
Aminotransferase Vaccines) C<br />
Increased<br />
Hepatitis B Vaccine<br />
Alanine Aminotransferase<br />
(Hepatitis B Virus<br />
Increased<br />
Vaccine Inactivated) C<br />
03-Apr-2012 09:37 AM Page: 144
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/18/99ISR Number: 3377417-XReport Type:Expedited (15-DaCompany Report #200442 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haemolytic Anaemia Health Lariam Tablets<br />
Anaemia Professional (<strong>Mefloquine</strong>/Hydrochl<br />
Foreign Travel oride) 250 Mg PS ORAL 250 MG 1 PER<br />
Haptoglobin Decreased<br />
WEEK ORAL<br />
Blood Bilirubin Increased<br />
Jaundice<br />
Diarrhoea<br />
Reticulocyte Count<br />
Increased<br />
Date:10/19/99ISR Number: 3375976-4Report Type:Expedited (15-DaCompany Report #215628 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cytogenetic Abnormality Foreign Lariam Tablets<br />
Pregnancy Health (<strong>Mefloquine</strong><br />
Neck Deformity Professional Hydrochloride) 250<br />
Maternal Drugs Affecting Mg PS ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Ultrasound Scan Abnormal<br />
Date:10/19/99ISR Number: 3376135-1Report Type:Expedited (15-DaCompany Report #215861 Age:51 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pulse Absent Foreign Lariam Tablets<br />
Initial or Prolonged Circulatory Collapse Health (<strong>Mefloquine</strong><br />
Electrocardiogram Qt Professional Hydrochloride) 250<br />
Prolonged Other Mg PS ORAL 250 MG 1 PER<br />
Cholinergic Syndrome<br />
WEEK ORAL<br />
Date:10/19/99ISR Number: 3376138-7Report Type:Expedited (15-DaCompany Report #217368 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chills Foreign Lariam Tablets<br />
Initial or Prolonged Red Blood Cell Other (<strong>Mefloquine</strong><br />
Sedimentation Rate Hydrochloride) 250<br />
Increased Mg PS ORAL 250 MG 1 PER<br />
Liver Function Test<br />
WEEK ORAL<br />
Abnormal<br />
Arthralgia<br />
Pyrexia<br />
Date:10/21/99ISR Number: 3378793-4Report Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Injury Other Lariam Tablets<br />
Lichen Planus<br />
(<strong>Mefloquine</strong><br />
Emotional Disorder Hydrochloride) 250<br />
Family Stress Mg PS ORAL 250 MG 1 PER<br />
Skin Discolouration<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 145
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/21/99ISR Number: 3378796-XReport Type:Expedited (15-DaCompany Report #216859 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Skin Discolouration Other Lariam Tablets<br />
Emotional Disorder<br />
(<strong>Mefloquine</strong><br />
Lichen Planus Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:10/25/99ISR Number: 3381772-4Report Type:Expedited (15-DaCompany Report #215928 Age:35 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Jaundice Health Lariam Tablets<br />
Initial or Prolonged Liver Function Test Professional (<strong>Mefloquine</strong><br />
Abnormal Hydrochloride) 250<br />
Hypervitaminosis Mg PS ORAL 250 MG 1 PER<br />
Blood Bilirubin Increased<br />
Vitamin Nos (Vitamin<br />
Nos)<br />
SS<br />
WEEK ORAL<br />
Date:10/25/99ISR Number: 3382580-0Report Type:Expedited (15-DaCompany Report #217696 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Sleep Disorder Foreign Lariam Tablets<br />
Balance Disorder Other (<strong>Mefloquine</strong><br />
Weight Decreased Hydrochloride) 250<br />
Memory Impairment Mg PS ORAL 250 MG 1 PER<br />
Decreased Appetite<br />
WEEK ORAL<br />
Visual Impairment<br />
Date:10/27/99ISR Number: 3383793-4Report Type:Expedited (15-DaCompany Report #217808 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Foreign Lariam Tablets<br />
Initial or Prolonged Depressed Mood Health (<strong>Mefloquine</strong><br />
Hallucination Professional Hydrochloride) 250<br />
Memory Impairment Other Mg PS ORAL 250 MG 1 PER<br />
Insomnia<br />
WEEK ORAL<br />
Confusional State<br />
Anxiety<br />
Decreased Appetite<br />
Restlessness<br />
Foreign Travel<br />
Visual Impairment<br />
Balance Disorder<br />
Diarrhoea
Date:10/28/99ISR Number: 3383896-4Report Type:Expedited (15-DaCompany Report #206437 Age:37 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Iris Atrophy<br />
Retinopathy<br />
Macular Oedema<br />
03-Apr-2012 09:37 AM Page: 146
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Iris Disorder<br />
Visual Impairment<br />
Retinal Pigmentation Report Source Product Role Manufacturer Route Dose Duration<br />
Vitreous Disorder Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Other Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK<br />
Date:10/29/99ISR Number: 3386323-6Report Type:Expedited (15-DaCompany Report #217046 Age:27 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Brain Oedema Foreign Lariam Tablets<br />
Sudden Death Health (<strong>Mefloquine</strong><br />
Pulmonary Oedema Professional Hydrochloride) 250<br />
Toxicity To Various Mg PS ORAL 250 MG 1 PER<br />
Agents<br />
1 WEEK ORAL<br />
Cyanosis Diane 35<br />
Pulmonary Congestion<br />
(Cyproterone<br />
Toxicologic Test Abnormal<br />
Acetate/Ethinyl<br />
Estradiol)<br />
C<br />
Ethyl Alcohol<br />
(Alcohol)<br />
C<br />
Paludrine<br />
(Chloroguanide<br />
Hydrochloride) C<br />
Date:10/29/99ISR Number: 3386327-3Report Type:Expedited (15-DaCompany Report #218344 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Haemangioma Foreign Lariam Tablets<br />
Abortion Induced Other (<strong>Mefloquine</strong><br />
Foetal Disorder Hydrochloride) 250<br />
Maternal Drugs Affecting Mg PS ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Date:11/01/99ISR Number: 3387632-7Report Type:Expedited (15-DaCompany Report #202330 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam Tablets<br />
Foetus Health (<strong>Mefloquine</strong><br />
Pregnancy Professional Hydrochloride) 250<br />
Ventricular Septal Defect Mg PS ORAL 250 MG 1<br />
PER WEEK<br />
ORAL
Date:11/04/99ISR Number: 3388785-7Report Type:Expedited (15-DaCompany Report #209222 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Supraventricular Foreign Lariam Tablets<br />
Initial or Prolonged Tachycardia Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1<br />
03-Apr-2012 09:37 AM Page: 147
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
PER WEEK<br />
ORAL<br />
Date:11/04/99ISR Number: 3388786-9Report Type:Expedited (15-DaCompany Report #104160 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Tricuspid Valve Foreign Lariam Tablets<br />
Required Incompetence Health (<strong>Mefloquine</strong><br />
Intervention to Cardiomegaly Professional Hydrochloride) 250<br />
Prevent Permanent Heart Disease Congenital Mg PS ORAL 250 MG 1<br />
Impairment/Damage Pregnancy PER WEEK<br />
Maternal Drugs Affecting<br />
ORAL<br />
Foetus<br />
Cyanosis Neonatal<br />
Date:11/04/99ISR Number: 3388787-0Report Type:Expedited (15-DaCompany Report #218871 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam Tablets<br />
Initial or Prolonged Visual Field Defect Health (<strong>Mefloquine</strong><br />
Anxiety Professional Hydrochloride) 250<br />
Nightmare Mg PS ORAL 250 MG 1<br />
PER WEEK<br />
ORAL<br />
Pondocillin<br />
(Pivampicillin<br />
Hydrochloride) C<br />
Klorokinfosfat<br />
(Chloroquine<br />
Phosphate)<br />
C<br />
Date:11/05/99ISR Number: 3390188-6Report Type:Expedited (15-DaCompany Report #218942 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Foreign Lariam Tablets<br />
Electroencephalogram Health (<strong>Mefloquine</strong><br />
Abnormal Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:11/09/99ISR Number: 3391768-4Report Type:Expedited (15-DaCompany Report #218954 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Foreign Lariam PS ORAL 250 MG 1 PER<br />
Initial or Prolonged Delusion Health WEEK<br />
Muscle Disorder<br />
Professional
Hypotonia<br />
Cachexia<br />
Personality Change<br />
03-Apr-2012 09:37 AM Page: 148
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/09/99ISR Number: 3391823-9Report Type:Expedited (15-DaCompany Report #218956 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paralysis Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:11/09/99ISR Number: 3391874-4Report Type:Expedited (15-DaCompany Report #218938 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam Tablets<br />
Initial or Prolonged Diarrhoea Health (<strong>Mefloquine</strong><br />
Blood Bilirubin Increased Professional Hydrochloride) 250<br />
Dizziness Mg PS ORAL ORAL 1 DAY<br />
Leukocytosis<br />
Pyrexia<br />
Date:11/09/99ISR Number: 3391875-6Report Type:Expedited (15-DaCompany Report #218945 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Arrhythmia Foreign Lariam Tablets<br />
Depression Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:11/09/99ISR Number: 3391907-5Report Type:Expedited (15-DaCompany Report #218933 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brain Scan Abnormal Foreign Lariam Tablets<br />
Nervous System Disorder Health (<strong>Mefloquine</strong><br />
Dyspnoea Professional Hydrochloride) 250<br />
Paraesthesia Mg PS ORAL ORAL<br />
Date:11/12/99ISR Number: 3396228-2Report Type:Expedited (15-DaCompany Report #218934 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aphasia Foreign Lariam Tablets<br />
Mental Disorder Health (<strong>Mefloquine</strong><br />
Grand Mal Convulsion Professional Hydrochloride) 250<br />
Dyskinesia Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:11/12/99ISR Number: 3396231-2Report Type:Expedited (15-DaCompany Report #218937 Age:59 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Dizziness<br />
Pallor<br />
Gastric Ulcer Haemorrhage<br />
03-Apr-2012 09:37 AM Page: 149
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Melaena<br />
Abdominal Distension<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Vaccine Nos (Vaccine<br />
Nos)<br />
C<br />
Date:11/12/99ISR Number: 3396477-3Report Type:Expedited (15-DaCompany Report #8-98334-066A Age:46 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Visual Impairment Health Profenid Capsule<br />
Initial or Prolonged Papilloedema Professional (Ketoprofen) PS ORAL ASS NEEDED<br />
Optic Ischaemic Other Propavan SS AS NEEDED<br />
Neuropathy <strong>Mefloquine</strong> SS ORAL ONE TABLET<br />
Optic Neuritis WEEKLY 3 WK<br />
Hepatitis B Vaccine SS PARENTERAL 1 MON<br />
<strong>Mefloquine</strong><br />
C<br />
Hepatitis B Vaccine C<br />
Caffeine<br />
+Paracetamol<br />
+Chlorphenamine<br />
Maleate<br />
+Dextropropoxyphene C<br />
Propavan<br />
(Propiomazine<br />
Maleate)<br />
C<br />
Typhoid Vaccine C<br />
Date:11/15/99ISR Number: 3398080-8Report Type:Expedited (15-DaCompany Report #219447 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Syndrome Foreign Lariam Tablets<br />
Initial or Prolonged Other (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg<br />
PS<br />
Date:11/15/99ISR Number: 3398082-1Report Type:Expedited (15-DaCompany Report #205111 Age:30 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Anxiety<br />
Palpitations<br />
Suicidal Ideation<br />
Fatigue
Insomnia<br />
Foreign Travel<br />
Muscle Spasms<br />
Dyspnoea<br />
Hypoaesthesia<br />
Psychotic Disorder<br />
Visual Impairment<br />
Confusional State<br />
03-Apr-2012 09:37 AM Page: 150
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Depression<br />
Musculoskeletal Stiffness Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam Tablets<br />
Consumer<br />
(<strong>Mefloquine</strong><br />
Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Ketoprofene<br />
(Ketoprofene)<br />
C<br />
Ibuprofene<br />
(Ibuprofene)<br />
C<br />
Date:11/15/99ISR Number: 3398085-7Report Type:Expedited (15-DaCompany Report #219442 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abnormal Behaviour Foreign Lariam Tablets<br />
Initial or Prolonged Visual Impairment Other (<strong>Mefloquine</strong><br />
Aggression Hydrochloride) 250<br />
Delirium Mg PS ORAL 250 MG 1 PER<br />
Foreign Travel<br />
WEEK ORAL<br />
Schizophrenia<br />
Speech Disorder<br />
Anxiety<br />
Date:11/15/99ISR Number: 3398090-0Report Type:Expedited (15-DaCompany Report #205270 Age:30 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam Tablets<br />
Initial or Prolonged Insomnia Consumer (<strong>Mefloquine</strong><br />
Musculoskeletal Stiffness Hydrochloride) 250<br />
Fatigue Mg PS ORAL 250 MG 1 PER<br />
Foreign Travel<br />
WEEK ORAL<br />
Panic Attack<br />
Ketoprofene<br />
Suicidal Ideation (Ketoprofene) C<br />
Visual Impairment<br />
Ibuprofene<br />
Dizziness (Ibuprofene) C<br />
Date:11/15/99ISR Number: 3398476-4Report Type:Expedited (15-DaCompany Report #218871 Age:45 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam Tablets<br />
Initial or Prolonged Anxiety Health (<strong>Mefloquine</strong><br />
Visual Field Defect Professional Hydrochloride) 250<br />
Nightmare Mg PS ORAL ORAL<br />
Pondocillin<br />
(Pivampicillin
Hydrochloride) C<br />
Klorokinfosfat<br />
(Chloroquine<br />
Phosphate)<br />
C<br />
03-Apr-2012 09:37 AM Page: 151
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/16/99ISR Number: 3399224-4Report Type:Expedited (15-DaCompany Report #109425 Age:46 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Visual Impairment Foreign Lariam Tablets<br />
Initial or Prolonged Optic Neuritis Other (<strong>Mefloquine</strong><br />
Papilloedema Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Profenid<br />
(Ketoprofen) SS ORAL 1 PER PRN<br />
ORAL<br />
Propofan<br />
(Acetaminophen/Caffe<br />
ine/Calcium<br />
Carbimide/Chlorpheni<br />
ramine SS ORAL 1 PER PRN<br />
ORAL<br />
Engerix B (Hepatitis<br />
B Virus Vaccine<br />
Inactivated) 20 Mcg<br />
Ml SS INTRAMUSCULAR INTRAMUSCULAR<br />
Typhim Vi<br />
C<br />
Sanmigram<br />
C<br />
Date:11/17/99ISR Number: 3398607-6Report Type:Direct Company Report # Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Mefloquene PS ORAL WEEKLY 250MG<br />
Insomnia ORAL 1 WK<br />
Panic Attack<br />
Depression<br />
Heart Rate Increased<br />
Dehydration<br />
Date:11/18/99ISR Number: 3399120-2Report Type:Direct Company Report # Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia Lariam PS<br />
Crying<br />
Suicidal Ideation<br />
Psychotic Disorder<br />
Abnormal Behaviour<br />
Flat Affect<br />
Foreign Travel<br />
Depression<br />
Date:11/18/99ISR Number: 3402652-1Report Type:Expedited (15-DaCompany Report #219962 Age:65 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG<br />
1 PER WEEK<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 152
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/19/99ISR Number: 3402792-7Report Type:Expedited (15-DaCompany Report #219706 Age:71 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Discolouration Consumer Lariam Tablets<br />
Oedema<br />
(<strong>Mefloquine</strong><br />
Pyrexia Hydrochloride) 250<br />
Upper Respiratory Tract Mg PS ORAL 250 MG 1 PER<br />
Infection<br />
WEEK ORAL<br />
Rash Erythematous<br />
Cipro (Ciprofloxacin<br />
Tenderness Hydrochloride) 500<br />
Burning Sensation Mg SS ORAL 500 MG DAILY<br />
Tendonitis<br />
ORAL<br />
Erythema Multiforme<br />
Aspirin (Baby<br />
Arthralgia Aspirin) C<br />
Cough<br />
Fosamax (Alendronate<br />
Drug Interaction Sodium) C<br />
Pallor<br />
Premarin (Estrogens,<br />
Conjugated)<br />
C<br />
Date:11/22/99ISR Number: 3405855-5Report Type:Expedited (15-DaCompany Report #220064 Age:13 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Skin Exfoliation Consumer Lariam Tablets<br />
Initial or Prolonged Dermatitis Exfoliative (<strong>Mefloquine</strong><br />
Hypersensitivity Hydrochloride) 250<br />
Vomiting Mg PS ORAL 250 MG 1 PER<br />
Dehydration<br />
WEEK ORAL<br />
Rash Maculo-Papular<br />
Pain In Extremity<br />
Face Oedema<br />
Viral Infection<br />
Rash Pruritic<br />
Rash Erythematous<br />
Date:11/23/99ISR Number: 3405398-9Report Type:Direct Company Report # Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Jaundice Health Larium PS ORAL ORAL TABLET<br />
Initial or Prolonged Liver Function Test Professional PER WEEK<br />
Required Abnormal (047)<br />
Intervention to Toxicity To Various Polio + Engenix B<br />
Prevent Permanent Agents Vaccines C<br />
Impairment/Damage Blood Bilirubin Increased<br />
Date:11/23/99ISR Number: 3415180-4Report Type:Expedited (15-DaCompany Report #215471 Age:79 YR Gender:Male I/FU:I<br />
Outcome<br />
PT
Other Serious<br />
Sciatica<br />
Abdominal Pain<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Blood Uric Acid Increased<br />
Diarrhoea<br />
Nausea<br />
Malaise<br />
03-Apr-2012 09:37 AM Page: 153
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Asthenia<br />
Blood Cholesterol<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Appetite Foreign Lariam Tablets<br />
Myalgia Consumer (<strong>Mefloquine</strong><br />
Joint Ankylosis Health Hydrochloride) 250<br />
Retching Professional Mg PS ORAL 250 MG 1 PER<br />
Abnormal Faeces<br />
WEEK ORAL<br />
Date:11/23/99ISR Number: 3415182-8Report Type:Expedited (15-DaCompany Report #220347 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Arrest Foreign Lariam Tablets<br />
Other<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:11/23/99ISR Number: 3415185-3Report Type:Expedited (15-DaCompany Report #220219 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eclampsia Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Ibuprofen<br />
(Ibuprofen)<br />
C<br />
Duspatal (Mebeverine<br />
Hydrochloride) C<br />
Hepatitis A Vaccine C<br />
Date:11/26/99ISR Number: 3409794-5Report Type:Expedited (15-DaCompany Report #104160 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Pregnancy Foreign Lariam Tablets<br />
Required Maternal Drugs Affecting Health (<strong>Mefloquine</strong><br />
Intervention to Foetus Professional Hydrochloride) 250<br />
Prevent Permanent Tricuspid Valve Mg PS TRANSPLACENTAL 250 MG 1 PER<br />
Impairment/Damage Incompetence WEEK ORAL<br />
Cyanosis Neonatal<br />
Heart Disease Congenital<br />
Date:11/26/99ISR Number: 3409969-5Report Type:Expedited (15-DaCompany Report #213726 Age:25 YR Gender:Female I/FU:I<br />
Outcome<br />
PT
Disability<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Scotoma<br />
Brain Oedema<br />
Haemoglobin Decreased<br />
Blood Potassium Increased<br />
Blood Urea Increased<br />
Blood Creatinine<br />
Increased<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 154
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dialysis<br />
Alanine Aminotransferase<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Visual Acuity Reduced Foreign Lariam PS ORAL 1000 MG DAILY<br />
Visual Impairment Health ORAL<br />
White Blood Cell Count Professional Gabexate Mesilate<br />
Increased (Gabexate) SS INTRAVENOUS 800 MG 2 PER<br />
Platelet Count Decreased<br />
DAY<br />
Blood Bilirubin Increased<br />
INTRAVENOUS<br />
Colour Blindness<br />
Minocycline<br />
Cardiovascular Disorder<br />
Hydrochloride<br />
Nuclear Magnetic<br />
(Minocycline<br />
Resonance Imaging Hydrochloride) SS INTRAVENOUS 100 MG 2 PER<br />
Abnormal<br />
DAY<br />
Renal Disorder<br />
INTRAVENOUS<br />
Aspartate<br />
Quinimax<br />
Aminotransferase<br />
Increased<br />
(Cinchonidine/Cincho<br />
nine/Quinidine/Quini<br />
ne) SS INTRAVENOUS 500 MG 2 PER<br />
DAY<br />
INTRAVENOUS<br />
Date:12/01/99ISR Number: 3411908-8Report Type:Expedited (15-DaCompany Report #215628 Age: Gender: I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Cystic Lymphangioma Foreign Lariam Tablets<br />
Foetal Disorder Health (<strong>Mefloquine</strong><br />
Pregnancy Professional Hydrochloride) 250<br />
Maternal Drugs Affecting Mg PS ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Abortion Induced<br />
Trisomy 21<br />
Date:12/03/99ISR Number: 3413305-8Report Type:Direct Company Report # Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Lariam Standard (For<br />
Convulsion Anti-Malarial PS STRADARD USE<br />
Neuropathy Peripheral<br />
Abnormal Dreams<br />
Anxiety<br />
Dizziness<br />
Panic Attack<br />
Paranoia<br />
Depression<br />
Palpitations<br />
Disorientation<br />
Foreign Travel<br />
Visual Impairment
Muscle Spasms<br />
Emotional Distress<br />
Panic Disorder<br />
03-Apr-2012 09:37 AM Page: 155
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/03/99ISR Number: 3413543-4Report Type:Expedited (15-DaCompany Report #210600 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania Consumer Lariam Tablets<br />
Initial or Prolonged Diarrhoea Health (<strong>Mefloquine</strong><br />
Disorientation Professional Hydrochloride) 250<br />
Leukocytosis Mg PS ORAL 250 MG 1 PER<br />
Pruritus<br />
WEEK ORAL<br />
Bipolar Disorder<br />
Lithium (Lithium<br />
Palpitations Nos) SS ORAL 300 MG DAILY<br />
Hypertension<br />
ORAL<br />
Drug Hypersensitivity<br />
Lorazepam<br />
Vomiting Projectile (Lorazepam) C<br />
Tremor Atenolol (Atenolol) C<br />
Circulatory Collapse<br />
Abdominal Pain<br />
Chest Discomfort<br />
Urticaria<br />
Dry Skin<br />
Pyrexia<br />
Insomnia<br />
Coordination Abnormal<br />
Flatulence<br />
Date:12/03/99ISR Number: 3413545-8Report Type:Expedited (15-DaCompany Report #220944 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Mental Disorder Foreign Lariam Tablets<br />
Suicide Attempt Health (<strong>Mefloquine</strong><br />
Paranoia Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:12/06/99ISR Number: 3414688-5Report Type:Expedited (15-DaCompany Report #204459 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Foetal Malposition Foreign Lariam Tablets<br />
Intervention to Prolonged Labour Health (<strong>Mefloquine</strong><br />
Prevent Permanent Ultrasound Scan Abnormal Professional Hydrochloride) 250<br />
Impairment/Damage Maternal Drugs Affecting Mg PS ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Na<br />
SS<br />
Date:12/07/99ISR Number: 3415578-4Report Type:Expedited (15-DaCompany Report #203725 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Premature Baby Foreign Lariam Tablets
Initial or Prolonged Maternal Drugs Affecting Health (<strong>Mefloquine</strong><br />
Foetus Professional Hydrochcloride) 250<br />
Premature Labour Mg PS ORAL 250 MG 1 PER<br />
WEEK<br />
03-Apr-2012 09:37 AM Page: 156
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/09/99ISR Number: 3417792-0Report Type:Expedited (15-DaCompany Report #221602 Age:47 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusional Disorder, Foreign Lariam Tablets<br />
Persecutory Type Other (<strong>Mefloquine</strong><br />
Hepatitis Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:12/09/99ISR Number: 3417793-2Report Type:Expedited (15-DaCompany Report #220219 Age:31 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eclampsia Foreign Lariam Tablets<br />
Pregnancy Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Ibuprofen<br />
(Ibuprofen)<br />
C<br />
Duspatal (Mebeverine<br />
Hydrochloride) C<br />
Hepatitis A Vaccine<br />
(Hepatitis A<br />
Vaccine)<br />
C<br />
Date:12/09/99ISR Number: 3417794-4Report Type:Expedited (15-DaCompany Report #200926 Age:57 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Feeling Abnormal Foreign Lariam Tablets<br />
Initial or Prolonged Jaundice Health (<strong>Mefloquine</strong><br />
Stress Professional Hydrochloride) PS ORAL 250 MG 1 PER<br />
Asthenia<br />
WEEK ORAL<br />
Hallucination<br />
Lariam Tablets<br />
Abnormal Dreams<br />
(<strong>Mefloquine</strong><br />
Foreign Travel Hydrochloride) SS ORAL 250 MG 1 PER<br />
Psychotic Disorder<br />
WEEK ORAL<br />
Aggression<br />
Delusion<br />
Hepatitis<br />
Hepatic Enzyme Increased<br />
Date:12/14/99ISR Number: 3421134-4Report Type:Expedited (15-DaCompany Report #216908 Age:35 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Toxicity To Various Health Lariam Tablets<br />
Initial or Prolonged Agents Professional (<strong>Mefloquine</strong><br />
Autoimmune Hepatitis Hydrochloride) 250<br />
Liver Function Test Mg PS ORAL 250 MG 1 PER
Abnormal 1 WEEK ORAL<br />
Hepatocellular Injury<br />
Polio Vaccine<br />
Jaundice<br />
(Poliomyelitis<br />
Vaccines)<br />
C<br />
Hepatitis B Vaccine<br />
(Hepatitis B Virus<br />
Vaccine Inactivated) C<br />
03-Apr-2012 09:37 AM Page: 157
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/14/99ISR Number: 3421219-2Report Type:Expedited (15-DaCompany Report #215861 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Circulatory Collapse Foreign Lariam Tablets<br />
Hospitalization - Heart Rate Decreased Consumer (<strong>Mefloquine</strong><br />
Initial or Prolonged Cholinergic Syndrome Health Hydrochloride) 200<br />
Pulse Absent Professional Mg PS ORAL 200 MG 1 PER<br />
Apnoea Other 1 WEEK ORAL<br />
Electrocardiogram Qt<br />
Prolonged<br />
Date:12/14/99ISR Number: 3422046-2Report Type:Expedited (15-DaCompany Report #215928 Age:35 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Scleral Discolouration Consumer Lariam Tablets<br />
Initial or Prolonged Hypervitaminosis Health (<strong>Mefloquine</strong><br />
Jaundice Professional Hydrochloride) 250<br />
Hepatitis Mg PS ORAL 250 MG 1 PER<br />
Hepatocellular Injury<br />
WEEK ORAL<br />
Toxicity To Various<br />
Vitamin Nos (Vitamin<br />
Agents Nos) SS<br />
Blood Bilirubin Increased<br />
Liver Function Test<br />
Abnormal<br />
Spleen Disorder<br />
Autoimmune Disorder<br />
Hepatic Necrosis<br />
Date:12/20/99ISR Number: 3426590-3Report Type:Expedited (15-DaCompany Report #222412 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaria Foreign Laroxyl<br />
Initial or Prolonged Neuropathy Peripheral Health (Amitriptyline<br />
Professional Hydrochloride) PS UNK<br />
Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) SS ORAL 1 DOSE FORM 1<br />
PER WEEK ORAL<br />
Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) SS ORAL ORAL<br />
Rohypnol<br />
(Flunitrazepam) SS ORAL ORAL<br />
Fumafer (Ferrous<br />
Fumarate)<br />
C<br />
Stilnox (Zolpidem<br />
Tartrate)<br />
C<br />
Date:12/21/99ISR Number: 3426942-1Report Type:Expedited (15-DaCompany Report #221071 Age:73 YR Gender:Male I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaise<br />
Electroencephalogram<br />
Abnormal<br />
Pallor<br />
Fatigue<br />
Bradycardia<br />
03-Apr-2012 09:37 AM Page: 158
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sedation<br />
Balance Disorder<br />
Cardiomegaly Report Source Product Role Manufacturer Route Dose Duration<br />
Candidiasis Foreign Lariam Tablets<br />
Myalgia Consumer (<strong>Mefloquine</strong><br />
Depressed Level Of Hydrochloride) 250<br />
Consciousness Mg PS ORAL 250 MG 1 PER<br />
Weight Decreased<br />
WEEK ORAL<br />
Pyrexia Timolol (Timolol) C<br />
Hypertension<br />
Influenza Vaccine<br />
Vomiting<br />
(Influenza Virus<br />
Headache Vaccine) C<br />
Bradyphrenia<br />
Atarax (Hydroxyine<br />
Oesophagitis Hydrochloride) C<br />
Osteoarthritis<br />
Bradypnoea<br />
Date:12/21/99ISR Number: 3426947-0Report Type:Expedited (15-DaCompany Report #214472 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cerebellar Atrophy Foreign Lariam Tablets<br />
Dysgraphia Health (<strong>Mefloquine</strong><br />
Fear Professional Hydrochloride) 250<br />
Balance Disorder Mg PS ORAL 250 MG 1 PER<br />
Dysarthria<br />
WEEK ORAL<br />
Movement Disorder<br />
Coordination Abnormal<br />
Cerebellar Syndrome<br />
Clumsiness<br />
Date:12/29/99ISR Number: 3434420-9Report Type:Expedited (15-DaCompany Report #223142 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Biopsy Bone Marrow Health Lariam Tablets<br />
Initial or Prolonged Abnormal Professional (<strong>Mefloquine</strong><br />
Platelet Count Decreased Hydrochloride) 250<br />
Mouth Haemorrhage Mg PS ORAL 250 MG 1 PER<br />
White Blood Cell Count<br />
WEEK ORAL<br />
Decreased<br />
Bone Marrow Failure<br />
Epistaxis<br />
Petechiae<br />
Haematocrit Decreased<br />
Date:01/04/00ISR Number: 3435387-XReport Type:Direct Company Report # Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Depression Lariam (Mefoquine)<br />
Tremor 250mg-La Roche PS La-Roche 1 TABLET EACH<br />
Insomnia<br />
WEEK<br />
Movement Disorder<br />
Panic Attack<br />
Heart Rate Increased<br />
03-Apr-2012 09:37 AM Page: 159
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/06/00ISR Number: 3439115-3Report Type:Expedited (15-DaCompany Report #220064 Age:13 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palmar Erythema Health Lariam Tablets<br />
Initial or Prolonged Pain In Extremity Professional (<strong>Mefloquine</strong><br />
Dermatitis Hydrochloride) 250<br />
Rash Generalised Mg PS ORAL 250 MG 1 PER<br />
Face Oedema<br />
WEEK ORAL<br />
Vomiting<br />
Dehydration<br />
Pruritus<br />
Rash Pruritic<br />
Rash Papular<br />
Skin Exfoliation<br />
Date:01/06/00ISR Number: 3439121-9Report Type:Expedited (15-DaCompany Report #223797 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Abortion Induced Foreign Lariam Tablets<br />
Trisomy 21 Health (<strong>Mefloquine</strong><br />
Pregnancy Professional Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:01/12/00ISR Number: 3443106-6Report Type:Direct Company Report # Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Lariam (<strong>Mefloquine</strong>) PS 250MG, MOUTH<br />
Decreased Appetite<br />
ONCE WEEKLY<br />
Chills<br />
Depression<br />
Anxiety<br />
Panic Attack<br />
Paraesthesia<br />
Parosmia<br />
Hypoaesthesia<br />
Muscle Spasms<br />
Insomnia<br />
Dermatitis<br />
Headache<br />
Date:01/12/00ISR Number: 3443550-7Report Type:Expedited (15-DaCompany Report #215212 Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Flatulence Foreign <strong>Mefloquine</strong><br />
Pancreatitis Other Hydrochloride<br />
Cholelithiasis<br />
(<strong>Mefloquine</strong><br />
Pancreatic Disorder Hydrochloride) 250
Abdominal Pain Mg PS ORAL ORAL<br />
Dyspepsia<br />
Blood Amylase Increased<br />
03-Apr-2012 09:37 AM Page: 160
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/12/00ISR Number: 3443552-0Report Type:Expedited (15-DaCompany Report #215378 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chest Pain Foreign Lariam Tablets<br />
Chest Discomfort Other (<strong>Mefloquine</strong><br />
Palpitations Hydrochloride) 250<br />
Ventricular Extrasystoles Mg PS ORAL 250 MG 1 PER<br />
Dyspnoea<br />
Brevinor-1 (Ethinyl<br />
Estradiol /<br />
Norethindrone)<br />
C<br />
WEEK ORAL<br />
Date:01/12/00ISR Number: 3443559-3Report Type:Expedited (15-DaCompany Report #223961 Age:67 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myasthenic Syndrome Foreign Lariam Tablets<br />
Initial or Prolonged Antibody Test Positive Other (<strong>Mefloquine</strong><br />
Eye Disorder Hydrochloride) 250<br />
Eyelid Ptosis Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Sotalex (Sotalol<br />
Hydrochloride) C<br />
Piascledine<br />
(Avocado/Soybean<br />
Oil)<br />
C<br />
Date:01/12/00ISR Number: 3443767-1Report Type:Expedited (15-DaCompany Report #215513 Age:16 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Foreign Lariam Tablets<br />
Depersonalisation Other (<strong>Mefloquine</strong><br />
Depression Hydrochloride) 250<br />
Paranoia Mg PS ORAL 250 MG 1 PER<br />
Dissociative Disorder<br />
WEEK ORAL<br />
Anxiety<br />
Fear<br />
Date:01/12/00ISR Number: 3443770-1Report Type:Expedited (15-DaCompany Report #215542 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Foreign Lariam Tablets<br />
Hepatitis Cholestatic Other (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Noroxin<br />
(Norfloxacin) SS 1 DOSE FORM
DAILY<br />
Vancomycin<br />
Hydrochloride<br />
(Vancomycin<br />
Hydrochloride) SS<br />
Imodium (Loperamide<br />
Hydrochloride) SS 1 DOSE FORM<br />
DAILY<br />
03-Apr-2012 09:37 AM Page: 161
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/19/00ISR Number: 3445475-XReport Type:Expedited (15-DaCompany Report #106318 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blister Foreign Lariam Tablets<br />
Initial or Prolonged Dystonia Other (<strong>Mefloquine</strong><br />
Hallucination Hydrochloride) 250<br />
Panic Attack Mg PS ORAL 250 MG 1 PER<br />
Dermatitis<br />
WEEK ORAL<br />
Grand Mal Convulsion<br />
Oral Contraceptive<br />
Insomnia<br />
Nos (Oral<br />
Contraceptive Nos) C<br />
Sedative Nos<br />
(Sedative Nos) C<br />
Date:01/19/00ISR Number: 3445477-3Report Type:Expedited (15-DaCompany Report #215861 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Cardiac Arrest Foreign Lariam Tablets<br />
Hospitalization - Cholinergic Syndrome Consumer (<strong>Mefloquine</strong><br />
Initial or Prolonged Circulatory Collapse Health Hydrochloride) 250<br />
Pulse Absent Professional Mg PS ORAL 250 MG 1 PER<br />
Electrocardiogram Qt Other 1 WEEK ORAL<br />
Prolonged<br />
Apnoea<br />
Date:01/19/00ISR Number: 3445854-0Report Type:Expedited (15-DaCompany Report #224407 Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Herpes Simplex Foreign Lariam Tablets<br />
Hepatitis Consumer (<strong>Mefloquine</strong><br />
Blood Immunoglobulin G Health Hydrochloride) 250<br />
Increased Professional Mg PS ORAL 250 MG 1 PER<br />
Liver Function Test<br />
WEEK ORAL<br />
Abnormal<br />
Date:01/19/00ISR Number: 3446071-0Report Type:Expedited (15-DaCompany Report #224923 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Foreign Lariam Tablets<br />
Initial or Prolonged Suicide Attempt Health (<strong>Mefloquine</strong><br />
Theft Professional Hydrochloride) PS ORAL 1.5 DOSE FORM<br />
Acute Psychosis<br />
1 PER WEEK<br />
ORAL 14 DAY<br />
Date:01/19/00ISR Number: 3446254-XReport Type:Expedited (15-DaCompany Report #220347 Age:49 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Arrest Foreign Lariam Tablets<br />
Other<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 162
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/21/00ISR Number: 3446442-2Report Type:Expedited (15-DaCompany Report #218871 Age:45 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam Tablets<br />
Initial or Prolonged Nightmare Health (<strong>Mefloquine</strong><br />
Visual Field Defect Professional Hydrochloride) 250<br />
Paranoia Mg PS ORAL ORAL<br />
Anxiety<br />
Kodimagnyl<br />
(Aspirin/Codeine<br />
Phosphate)<br />
C<br />
Panodil<br />
(Acetaminophen) C<br />
Klorokinfosfat<br />
(Chloroquine<br />
Phosphate)<br />
C<br />
Pondocillin<br />
(Pivampicillin<br />
Hydrochloride) C<br />
Date:01/28/00ISR Number: 3447825-7Report Type:Direct Company Report # Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Gait Disturbance Larium (Mefloquin) PS<br />
Nervous System Disorder<br />
Paralysis<br />
Feeling Abnormal<br />
Date:01/28/00ISR Number: 3448097-XReport Type:Expedited (15-DaCompany Report #223142 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Thrombocytopenia Health Lariam Tablets<br />
Hospitalization - Petechiae Professional (<strong>Mefloquine</strong><br />
Initial or Prolonged Epistaxis Hydrochloride) 250<br />
Disability Leukopenia Mg PS ORAL 250 MG 1 PER<br />
Haematocrit Decreased<br />
WEEK ORAL<br />
Mouth Haemorrhage<br />
Megakaryocytes Abnormal<br />
Bone Marrow Failure<br />
Date:01/28/00ISR Number: 3448154-8Report Type:Expedited (15-DaCompany Report #225350 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Tinnitus Foreign Lariam Tablets<br />
Intervention to Deafness Consumer (<strong>Mefloquine</strong><br />
Prevent Permanent Hydrochloride) 250<br />
Impairment/Damage Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:02/02/00ISR Number: 3451029-1Report Type:Expedited (15-DaCompany Report #226681 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source<br />
Disability Haemolysis Foreign<br />
Health<br />
03-Apr-2012 09:37 AM Page: 163
Professional<br />
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:02/04/00ISR Number: 3452318-7Report Type:Expedited (15-DaCompany Report #216832 Age:30 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Loss Of Consciousness Health Lariam Tablets<br />
Initial or Prolonged Back Pain Professional (<strong>Mefloquine</strong><br />
Grand Mal Convulsion Hydrochloride) 250<br />
Neurological Symptom Mg PS ORAL 250 MG 1 PER<br />
Movement Disorder<br />
WEEK ORAL<br />
Gait Disturbance<br />
Date:02/08/00ISR Number: 3454673-0Report Type:Expedited (15-DaCompany Report #227211 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oesophagitis Foreign Lariam Tablets<br />
Oesophageal Pain Other (<strong>Mefloquine</strong><br />
Sensation Of Pressure Hydrochloride) 250<br />
Bundle Branch Block Right Mg PS ORAL 250 MG 1 PER<br />
Supraventricular 1 WEEK, ORAL 28 DAY<br />
Extrasystoles<br />
Dyspnoea<br />
Date:02/08/00ISR Number: 3454675-4Report Type:Expedited (15-DaCompany Report #227013 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Foreign Lariam Tablets<br />
Initial or Prolonged Cerebrovascular Accident Health (<strong>Mefloquine</strong><br />
Disability Feeling Abnormal Professional Hudrochloride) 250<br />
Thrombocytopenia Mg PS ORAL 250 MG 1 PER<br />
Cerebral Infarction<br />
1 WEEK, ORAL<br />
Date:02/08/00ISR Number: 3455315-0Report Type:Expedited (15-DaCompany Report #220944 Age:59 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Foreign Lariam Tablets<br />
Paranoia Health (<strong>Mefloquine</strong><br />
Mental Disorder Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 164
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/09/00ISR Number: 3455449-0Report Type:Expedited (15-DaCompany Report #215471 Age:79 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pain In Extremity Foreign Lariam Tablets<br />
Blood Cholesterol Consumer (<strong>Mefloquine</strong><br />
Increased Health Hydrochloride) 250<br />
Decreased Appetite Professional Mg PS ORAL 250 MG 1 PER<br />
Retching<br />
WEEK ORAL<br />
Polymyalgia Rheumatica<br />
Joint Ankylosis<br />
Abnormal Faeces<br />
Diarrhoea<br />
Arthralgia<br />
Malaise<br />
Asthenia<br />
Sciatica<br />
Myalgia<br />
Foreign Travel<br />
Nausea<br />
Abdominal Pain<br />
Date:02/11/00ISR Number: 3457006-9Report Type:Expedited (15-DaCompany Report #215928 Age:35 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypervitaminosis Consumer Lariam Tablets<br />
Initial or Prolonged Hepatitis Health (<strong>Mefloquine</strong><br />
Liver Function Test Professional Hydrochloride) 250<br />
Abnormal Mg PS ORAL 250 MG 1 PER<br />
Autoimmune Disorder<br />
WEEK ORAL<br />
Spleen Disorder<br />
Vitamin Nos (Vitamin<br />
Blood Bilirubin Increased Nos) SS<br />
Hepatic Necrosis<br />
Jaundice<br />
Date:02/11/00ISR Number: 3457009-4Report Type:Expedited (15-DaCompany Report #217772 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Condition Aggravated Foreign Lariam Tablets<br />
Initial or Prolonged Ventricular Hypertrophy Other (<strong>Mefloquine</strong><br />
Bradycardia Hydrochloride) 250<br />
Drug Interaction Mg PS ORAL 250 MG 1 PER<br />
Hypertension<br />
WEEK ORAL<br />
Muscular Weakness<br />
Timoptol Eye Drops<br />
Ejection Fraction (Timolol Maleate) 5<br />
Abnormal Mg/Ml SS OPHTHALMIC .05 DOSE FORM<br />
Dizziness<br />
DAILY<br />
OPHTHALMIC<br />
Bethahistine<br />
Hydrochloride
(Bethahistine) C<br />
03-Apr-2012 09:37 AM Page: 165
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/11/00ISR Number: 3457011-2Report Type:Expedited (15-DaCompany Report #215889 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Foreign Travel Foreign Lariam Tablets<br />
Initial or Prolonged Eye Pain Health (<strong>Mefloquine</strong><br />
Csf Lymphocyte Count Professional Hydrochloride) 250<br />
Increased Mg PS ORAL 250 MG 1 PER<br />
Csf White Blood Cell WEEK ORAL 56 DAY<br />
Count Positive<br />
Optic Neuritis<br />
Headache<br />
Vision Blurred<br />
Visual Acuity Reduced<br />
C-Reactive Protein<br />
Increased<br />
Blood Glucose Increased<br />
Sinusitis<br />
Blood Triglycerides<br />
Increased<br />
Glucose Tolerance<br />
Impaired<br />
Meningitis<br />
Date:02/14/00ISR Number: 3457666-2Report Type:Expedited (15-DaCompany Report #224407 Age:57 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Liver Function Test Foreign Lariam Tablets<br />
Abnormal Health (<strong>Mefloquine</strong><br />
Blood Immunoglobulin G Professional Hydrochloride) 250<br />
Increased Mg PS ORAL 250 MG 1 PER<br />
Hepatitis<br />
WEEK ORAL<br />
Herpes Simplex<br />
Date:02/15/00ISR Number: 3458375-6Report Type:Expedited (15-DaCompany Report #227783 Age:8 WK Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Hypotonia<br />
Maternal Drugs Affecting<br />
Foetus<br />
Chondromalacia<br />
Congenital Gastric<br />
Anomaly<br />
Congenital Hair Disorder<br />
Microphthalmos<br />
Gastrooesophageal Reflux<br />
Disease<br />
Finger Deformity<br />
Oesophageal Dilatation<br />
Feeding Disorder Neonatal
Microcephaly<br />
Congenital Anomaly<br />
Congenital Nose<br />
Malformation<br />
Vomiting<br />
Dyskinesia Oesophageal<br />
Dysmorphism<br />
Blood Pressure Increased<br />
03-Apr-2012 09:37 AM Page: 166
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Oesophageal Achalasia<br />
Congenital Central<br />
Nervous System Anomaly Report Source Product Role Manufacturer Route Dose Duration<br />
Neck Deformity Foreign Lariam Tablets<br />
Developmental Delay Literature (<strong>Mefloquine</strong><br />
Salivary Hypersecretion Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Convulsion Professional WEEK ORAL<br />
Joint Contracture<br />
Micrognathia<br />
Respiratory Arrest<br />
Ear Malformation<br />
Date:02/16/00ISR Number: 3458485-3Report Type:Direct Company Report # Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Panic Attack Lariam/Roche PS Roche BUCCAL 6 TABLE<br />
Disability Dizziness BUCCAL<br />
Asthenia<br />
Disturbance In Attention<br />
Eye Pain<br />
Date:02/16/00ISR Number: 3458909-1Report Type:Expedited (15-DaCompany Report #217772 Age:53 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Ventricular Hypertrophy Foreign Lariam Tablets<br />
Initial or Prolonged Muscular Weakness Other (<strong>Mefloquine</strong><br />
Bradycardia Hydrochloride) 250<br />
Hypertension Mg PS ORAL 250 MG 1 PER<br />
Ejection Fraction<br />
WEEK ORAL<br />
Abnormal<br />
Timoptol Eye Drops<br />
Dizziness<br />
(Timolol Maleate)<br />
Drug Interaction 5mg/Ml SS OPHTHALMIC .05 MG DOSE<br />
FORM DAILY<br />
OPHTHALMIC<br />
Betahistine<br />
Hydrochloride<br />
(Betahistine)<br />
C<br />
Date:02/17/00ISR Number: 3459446-0Report Type:Expedited (15-DaCompany Report #207736 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam Tablets<br />
Foetus Health (<strong>Mefloquine</strong><br />
Congenital Cystic Kidney Professional Hydrochloride) 250<br />
Disease Mg PS ORAL 250 MG 1 PER<br />
Congenital Anomaly<br />
WEEK ORAL<br />
Renal Hypertrophy
Date:02/18/00ISR Number: 3459354-5Report Type:Expedited (15-DaCompany Report #58978 Age:31 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Back Pain<br />
Vestibular Disorder<br />
03-Apr-2012 09:37 AM Page: 167
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sensation Of Pressure<br />
Dyspnoea<br />
Hyperhidrosis Report Source Product Role Manufacturer Route Dose Duration<br />
Balance Disorder Consumer Lariam Tablets<br />
Anxiety Health (<strong>Mefloquine</strong><br />
Ear Discomfort Professional Hydrochloride) 250<br />
Drug Hypersensitivity Mg PS ORAL 250 MG 1 PER<br />
Cerebellar Ataxia<br />
WEEK ORAL<br />
Nystagmus<br />
Norflex<br />
Thrombocytopenia<br />
(Orphenadrine<br />
Headache Citrate) C<br />
Tachycardia<br />
Accident<br />
Chest Pain<br />
Road Traffic Accident<br />
Palpitations<br />
Vomiting<br />
Asthenia<br />
Confusional State<br />
Vertigo<br />
Spinal Fracture<br />
Syncope<br />
Clonic Convulsion<br />
Anaemia<br />
Fatigue<br />
Neck Pain<br />
Amnesia<br />
Abdominal Pain<br />
Sinus Headache<br />
Dizziness<br />
Gait Disturbance<br />
Disturbance In Attention<br />
Nausea<br />
Feeling Abnormal<br />
Platelet Count Abnormal<br />
Scoliosis<br />
Epistaxis<br />
Date:02/18/00ISR Number: 3459955-4Report Type:Expedited (15-DaCompany Report #228385 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania Health Lariam Tablets<br />
Initial or Prolonged Psychotic Disorder Professional (<strong>Mefloquine</strong><br />
Thinking Abnormal Hydrochloride) 250<br />
Bipolar Disorder Mg PS ORAL 250 MG 1 PER<br />
Weight Decreased<br />
WEEK ORAL<br />
Psychomotor Hyperactivity<br />
Chloroquine<br />
Magical Thinking (Chloroquine) C<br />
Foreign Travel<br />
Abnormal Behaviour
Date:02/18/00ISR Number: 3459957-8Report Type:Expedited (15-DaCompany Report #224949 Age:67 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaise<br />
Hypoxia<br />
Oedema Peripheral<br />
03-Apr-2012 09:37 AM Page: 168
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pruritus<br />
Pain In Extremity<br />
Dyspnoea Exertional Report Source Product Role Manufacturer Route Dose Duration<br />
Dehydration Foreign Lariam Tablets<br />
Subcutaneous Emphysema Consumer (<strong>Mefloquine</strong><br />
Vein Pain Hydrochloride) 250<br />
Dry Skin Mg PS ORAL 250 MG 1 PER<br />
Pneumothorax Traumatic<br />
1 WEEK ORAL<br />
Pleural Effusion<br />
Pain<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Jaundice<br />
Lung Consolidation<br />
Diarrhoea<br />
Ecchymosis<br />
Blood Sodium Increased<br />
Fall<br />
Bacterial Test Positive<br />
Rib Fracture<br />
Bronchospasm<br />
Blood Bilirubin Increased<br />
Decreased Appetite<br />
Injury<br />
Liver Function Test<br />
Abnormal<br />
Haemorrhoids<br />
Cholestasis<br />
Chromaturia<br />
Wound Infection<br />
Pulmonary Oedema<br />
Dyspnoea<br />
Staphylococcal Infection<br />
Date:02/18/00ISR Number: 3459961-XReport Type:Expedited (15-DaCompany Report #213259 Age:18 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Pharyngitis Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Drug Hypersensitivity Professional 1 WEEK ORAL<br />
Cough<br />
Clamoxyl Duo<br />
Erythema<br />
(Amoxicillin<br />
Urticaria<br />
Sodium/*Clavulanate<br />
Pyrexia Potassium) SS ORAL 2 GRAM DAILY<br />
Eyelid Oedema<br />
Dyspnoea<br />
ORAL<br />
Imudon (Lysates,<br />
Bacterial) SS ORAL ORAL<br />
Doliprane<br />
(Acetaminophen)<br />
Hexomedine<br />
Collutoire<br />
C
(Hexamidine/Tetracai<br />
ne Hydrochloride)<br />
C<br />
03-Apr-2012 09:37 AM Page: 169
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/23/00ISR Number: 3461482-5Report Type:Expedited (15-DaCompany Report #228826 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aplastic Anaemia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL 42 DAY<br />
Date:02/24/00ISR Number: 3462565-6Report Type:Expedited (15-DaCompany Report #228880 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Euphoric Mood Consumer Lariam Tablets<br />
Initial or Prolonged Dizziness (<strong>Mefloquine</strong><br />
Nausea Hydrochloride) 250<br />
Photophobia Mg PS ORAL 250 MG 1 PER<br />
Palpitations<br />
Dimetapp<br />
(Brompheniramine<br />
Maleate/*Phenylephri<br />
ne<br />
Hydrochloride/Phenyl C<br />
WEEK ORAL<br />
Date:02/24/00ISR Number: 3462569-3Report Type:Expedited (15-DaCompany Report #228555 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Splenomegaly Foreign Lariam Tablets<br />
Diarrhoea Literature (<strong>Mefloquine</strong><br />
Fear Health Hydrochloride) 250<br />
Dizziness Professional Mg PS ORAL 500 MG 1 PER<br />
Oropharyngeal Pain<br />
ONE DOSE ORAL<br />
Hypoaesthesia<br />
Co-Trimoxazol<br />
Burning Sensation<br />
(Sulfamethoxazole/Tr<br />
Panic Disorder imethoprim) C<br />
Peripheral Coldness<br />
Penicillin V<br />
Psychotic Disorder (Penicillin V) C<br />
Chills<br />
Intravenous Glucose<br />
Confusional State (Glucose) C<br />
Intentional Self-Injury<br />
Paracetamol<br />
Depression (Acetaminophen) C<br />
Anxiety<br />
Codeine Phosphate<br />
Myocarditis (Codeine Phosphate) C<br />
Pyrexia<br />
Fansidar<br />
Bone Pain<br />
(Pyrimethamine/Sulfa<br />
Completed Suicide doxine) C<br />
Dyspnoea Quinine (Quinine) C<br />
Disorientation<br />
Headache<br />
Antimalarial Agent<br />
Nos (Antimalarial<br />
Agent Nos)<br />
C
Aspirin (Aspirin) C<br />
03-Apr-2012 09:37 AM Page: 170
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/24/00ISR Number: 3462570-XReport Type:Expedited (15-DaCompany Report #225350 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Audiogram Abnormal Foreign Lariam Tablets<br />
Intervention to Tinnitus Consumer (<strong>Mefloquine</strong><br />
Prevent Permanent Deafness Health Hydrochloride) 250<br />
Impairment/Damage Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:02/24/00ISR Number: 3462575-9Report Type:Expedited (15-DaCompany Report #223797 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Anomaly Foreign Lariam Tablets<br />
Trisomy 21 Health (<strong>Mefloquine</strong><br />
Pregnancy Professional Hydrochloride) 250<br />
Abortion Induced Mg PS ORAL 250 MG 1 PER<br />
Maternal Drugs Affecting<br />
WEEK ORAL<br />
Foetus<br />
Date:02/24/00ISR Number: 3462577-2Report Type:Expedited (15-DaCompany Report #211640 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Foreign Lariam Tablets<br />
Initial or Prolonged Paralysis Other (<strong>Mefloquine</strong><br />
Disability Gait Disturbance Hydrochloride) 250<br />
Myalgia Mg PS ORAL 250 MG 1 PER<br />
Facial Pain<br />
WEEK ORAL<br />
Pain In Extremity<br />
Headache<br />
Asthenia<br />
Polyneuropathy<br />
Date:02/24/00ISR Number: 3462580-2Report Type:Expedited (15-DaCompany Report #228268 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Premature Separation Of Foreign Lariam Tablets<br />
Initial or Prolonged Placenta Health (<strong>Mefloquine</strong><br />
Menometrorrhagia Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
ONE DOSE ORAL<br />
Date:02/28/00ISR Number: 3464073-5Report Type:Direct Company Report # Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Photophobia Lariam (<strong>Mefloquine</strong>) PS TABLET WEEKLY
Headache FOR 7 WEEKS<br />
Ear Discomfort<br />
Hypersensitivity<br />
03-Apr-2012 09:37 AM Page: 171
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/28/00ISR Number: 3464099-1Report Type:Direct Company Report # Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Lariam (<strong>Mefloquine</strong>) PS ORAL 1 TABLET<br />
WEEKLY FOR 7<br />
WEEKS (ORAL)<br />
Date:02/29/00ISR Number: 3465856-8Report Type:Expedited (15-DaCompany Report #228826 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aplastic Anaemia Consumer Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL 42 DAY<br />
Date:02/29/00ISR Number: 3465857-XReport Type:Expedited (15-DaCompany Report #229472 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fall Foreign Lariam Tablets<br />
Neuropathy Peripheral Health (<strong>Mefloquine</strong><br />
Diarrhoea Professional Hydrochloride) PS ORAL 250 MG 1 PER<br />
Coordination Abnormal<br />
WEEK ORAL<br />
Paraesthesia Oral Atenolol (Atenolol) C<br />
Gait Disturbance<br />
Date:03/01/00ISR Number: 3467533-6Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Disturbance In Attention Health Laraim PS TABLET<br />
Other Serious Anxiety Professional WEEKLY<br />
Hallucination<br />
Feeling Abnormal<br />
Restlessness<br />
Muscle Twitching<br />
Depression<br />
Sleep Disorder<br />
Paranoia<br />
Alopecia<br />
Date:03/02/00ISR Number: 3468068-7Report Type:Expedited (15-DaCompany Report #223959 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Foreign Lariam Tablets<br />
Initial or Prolonged Deja Vu Other (<strong>Mefloquine</strong><br />
Illusion Hydrochloride) 250<br />
Anxiety Mg PS ORAL 250 MG 1 PER
Panic Attack WEEK ORAL<br />
Insomnia<br />
Delirium<br />
Depersonalisation<br />
Abnormal Behaviour<br />
Hallucination, Auditory<br />
Confusional State<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 172
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/03/00ISR Number: 3468986-XReport Type:Expedited (15-DaCompany Report #222412 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Neuropathy Peripheral Foreign Laroxyl<br />
Initial or Prolonged Asthenia Health (Amitriptyline<br />
Insomnia Professional Hydrochloride) 25 Mg PS ORAL 25 MG DAILY<br />
Red Blood Cell<br />
ORAL<br />
Sedimentation Rate<br />
Lariam Tablets<br />
Increased<br />
(<strong>Mefloquine</strong><br />
Hyporeflexia Hydrochloride) 250<br />
Depression Mg SS ORAL 250 MG 1 PER<br />
Malaria<br />
WEEK ORAL<br />
Stilnox (Zolpidem<br />
Tartrate)<br />
C<br />
Fumafer (Ferrous<br />
Fumarate)<br />
C<br />
Quinine (Quinine) C<br />
Rohypnol<br />
(Flunitrazepam) C<br />
Date:03/08/00ISR Number: 3471240-3Report Type:Direct Company Report # Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nausea Lariam PS 1 TABLET /<br />
Influenza Like Illness<br />
ONCE A WEEK<br />
Pyrexia<br />
Vertigo<br />
Tremor<br />
Medication Error<br />
Depression<br />
Headache<br />
Dizziness<br />
Lethargy<br />
Date:03/09/00ISR Number: 3472238-1Report Type:Direct Company Report # Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare <strong>Mefloquine</strong> PS ORAL 250MG 1 PO QD<br />
Insomnia<br />
Anxiety<br />
Date:03/09/00ISR Number: 3473493-4Report Type:Expedited (15-DaCompany Report #230147 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Consumer Lariam Tablets<br />
Initial or Prolonged Monoparesis (<strong>Mefloquine</strong><br />
Hydrochloride) PS
Date:03/09/00ISR Number: 3473498-3Report Type:Expedited (15-DaCompany Report #227783 Age:8 WK Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Congenital<br />
Musculoskeletal Anomaly<br />
03-Apr-2012 09:37 AM Page: 173
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Vomiting<br />
Microcephaly<br />
Salivary Hypersecretion Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Nose Foreign Lariam Tablets<br />
Malformation Literature (<strong>Mefloquine</strong><br />
Oesophageal Dilatation Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Growth Retardation Professional WEEK ORAL<br />
Developmental<br />
Coordination Disorder<br />
Dyskinesia Oesophageal<br />
Gastrooesophageal Reflux<br />
Disease<br />
Congenital Jaw<br />
Malformation<br />
Micrognathia<br />
Congenital Hair Disorder<br />
Developmental Delay<br />
Hypertension<br />
Congenital Oral<br />
Malformation<br />
Convulsion<br />
Microphthalmos<br />
Respiratory Disorder<br />
Neonatal<br />
Hypercapnia<br />
Respiratory Arrest<br />
Hypotonia<br />
Hypertonia<br />
Feeding Disorder<br />
Maternal Drugs Affecting<br />
Foetus<br />
Oesophageal Achalasia<br />
Congenital Gastric<br />
Anomaly<br />
Congenital Central<br />
Nervous System Anomaly<br />
Mental Impairment<br />
Joint Contracture<br />
Microstomia<br />
Autosomal Chromosome<br />
Anomaly<br />
Dysmorphism<br />
Congenital Foot<br />
Malformation<br />
Ear Malformation<br />
Date:03/10/00ISR Number: 3474446-2Report Type:Expedited (15-DaCompany Report #75640 Age: Gender:Unknown I/FU:F<br />
Outcome<br />
Congenital Anomaly<br />
PT<br />
Hypercapnia<br />
Clumsy Child Syndrome
Oesophageal Achalasia<br />
Feeding Disorder<br />
Muscular Dystrophy<br />
Pregnancy<br />
Congenital Jaw<br />
Malformation<br />
Hypertension<br />
Obstructive Airways<br />
03-Apr-2012 09:37 AM Page: 174
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorder<br />
Maternal Drugs Affecting<br />
Foetus<br />
Growth Retardation Report Source Product Role Manufacturer Route Dose Duration<br />
Autoimmune Disorder Foreign Lariam Tablets<br />
Hypotonia Health (<strong>Mefloquine</strong><br />
Dysmorphism Professional Hydrochloride) 250<br />
Mental Disorder Mg PS ORAL 250 MG 1 PER<br />
Microcephaly<br />
WEEK ORAL<br />
Multiple Congenital<br />
Abnormalities<br />
Malnutrition<br />
Oesophageal Disorder<br />
Gastrooesophageal Reflux<br />
Disease<br />
Date:03/10/00ISR Number: 3474449-8Report Type:Expedited (15-DaCompany Report #207736 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Anomaly Foreign Lariam Tablets<br />
Congenital Cystic Kidney Consumer (<strong>Mefloquine</strong><br />
Disease Health Hydrochloride) 250<br />
Renal Hypertrophy Professional Mg PS ORAL 250 MG 1 PER<br />
Pregnancy<br />
WEEK ORAL<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:03/13/00ISR Number: 3474365-1Report Type:Direct Company Report # Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lethargy Lariam 250mg<br />
Initial or Prolonged Headache Hoffmann-La Roche<br />
Other Serious Dysentery Inc PS Hoffmann-La Roche<br />
Asthenia Inc ORAL 250MG WEEKLY<br />
Decreased Appetite<br />
ORAL<br />
Nausea Metrodozole C<br />
Vertigo Tinidozole C<br />
Dizziness Anti-Biotics C<br />
Pyrexia<br />
Diarrhoea<br />
Date:03/14/00ISR Number: 3475522-0Report Type:Expedited (15-DaCompany Report #230503 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dysphonia Foreign Lariam Tablets<br />
Depression Other (<strong>Mefloquine</strong><br />
Gait Disturbance Hydrochloride) PS ORAL 250 MG ORAL<br />
Speech Disorder
Date:03/14/00ISR Number: 3475572-4Report Type:Expedited (15-DaCompany Report #230342 Age: Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Overdose<br />
Nervous System Disorder<br />
03-Apr-2012 09:37 AM Page: 175
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL ORAL 7 DAY<br />
Date:03/14/00ISR Number: 3475596-7Report Type:Expedited (15-DaCompany Report #224949 Age:67 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Jaundice Foreign Lariam Tablets<br />
Initial or Prolonged Ecchymosis Consumer (<strong>Mefloquine</strong><br />
Haemorrhoids Hydrochloride ) 250<br />
Dyspnoea Exertional Mg PS ORAL 250 MG 1 PER<br />
Decreased Appetite<br />
1 WEEK ORAL<br />
Wound Infection<br />
Pulmonary Oedema<br />
Fall<br />
Subcutaneous Emphysema<br />
Chromaturia<br />
Pruritus<br />
Pain<br />
Pneumothorax<br />
Blood Bilirubin Increased<br />
Hypoxia<br />
Lung Consolidation<br />
Pleural Effusion<br />
Bronchospasm<br />
Cholestasis<br />
Malaise<br />
Diarrhoea<br />
Liver Function Test<br />
Abnormal<br />
Rib Fracture<br />
Staphylococcal Infection<br />
Oedema Peripheral<br />
Benign Prostatic<br />
Hyperplasia<br />
Haemothorax<br />
Vein Pain<br />
Dry Skin<br />
Injury<br />
Dehydration<br />
Date:03/14/00ISR Number: 3475600-6Report Type:Expedited (15-DaCompany Report #229994 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dyspnoea Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong>
Hydrochloride) PS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 176
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/15/00ISR Number: 3475487-1Report Type:Direct Company Report # Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination, Visual <strong>Mefloquine</strong> (Lariam)<br />
Initial or Prolonged Paranoia Roche Pharm. 250 Mg PS Roche Pharm. ORAL ORAL 1/ WEEK<br />
Disability<br />
Arthralgia<br />
Required<br />
Confusional State<br />
Intervention to<br />
Panic Attack<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:03/16/00ISR Number: 3476231-4Report Type:Direct Company Report # Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam PS<br />
Anxiety<br />
Professional<br />
Date:03/20/00ISR Number: 3478147-6Report Type:Expedited (15-DaCompany Report #231049 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Foreign Lariam Tablets<br />
Initial or Prolonged Vomiting Health (<strong>Mefloquine</strong><br />
Nausea Professional Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Nystagmus<br />
PER WEEK ORAL<br />
Dizziness<br />
Date:03/21/00ISR Number: 3478539-5Report Type:Expedited (15-DaCompany Report #225350 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Tinnitus Foreign Lariam Tablets<br />
Intervention to Deafness Consumer (<strong>Mefloquine</strong><br />
Prevent Permanent Health Hydrochloride) 250<br />
Impairment/Damage Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:03/23/00ISR Number: 3479213-1Report Type:Expedited (15-DaCompany Report #224949 Age:67 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Bronchospasm<br />
Chest X-Ray Abnormal<br />
Vein Pain<br />
Chromaturia<br />
Wound Infection<br />
Gait Disturbance<br />
Dry Skin
Injury<br />
Tenderness<br />
Pain<br />
Jaundice<br />
Ecchymosis<br />
Alanine Aminotransferase<br />
Increased<br />
Cholestasis<br />
03-Apr-2012 09:37 AM Page: 177
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hypoxia<br />
Diarrhoea<br />
Emphysema Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Appetite Foreign Lariam Tablets<br />
Pruritus Consumer (<strong>Mefloquine</strong><br />
Oedema Peripheral Hydrochloride) 250<br />
Dyspnoea Exertional Mg PS ORAL 250 MG 1 PER<br />
Pulmonary Oedema<br />
1 WEEK ORAL<br />
Haemorrhoids<br />
Staphylococcal Infection<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Pleural Effusion<br />
Pneumothorax<br />
Blood Bilirubin Increased<br />
Haemorrhage<br />
Rib Fracture<br />
Dehydration<br />
Date:03/23/00ISR Number: 3479216-7Report Type:Expedited (15-DaCompany Report #231053 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Muscular Weakness Foreign Lariam Tablets<br />
Initial or Prolonged Paraesthesia Other (<strong>Mefloquine</strong><br />
Sensory Disturbance Hydrochloride) 250<br />
Hyporeflexia Mg PS ORAL 250 MG 1 PER<br />
Bacterial Infection<br />
WEEK ORAL<br />
Myalgia<br />
Proscar<br />
Gait Disturbance (Finasteride) C<br />
Csf Protein Increased<br />
Dyspnoea<br />
Guillain-Barre Syndrome<br />
Antibody Test Positive<br />
Dizziness<br />
Date:03/23/00ISR Number: 3479288-XReport Type:Direct Company Report # Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Lariam (<strong>Mefloquine</strong>)<br />
Initial or Prolonged Dyspnoea 250mg PS Hoffman Laroche 1 WEEKLY FOR<br />
Disability Headache 7 WKS<br />
Other Serious<br />
Vomiting<br />
Panic Attack<br />
Fatigue<br />
Chills<br />
Palpitations<br />
Paraesthesia
Diarrhoea<br />
Hallucination<br />
Tremor<br />
03-Apr-2012 09:37 AM Page: 178
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/23/00ISR Number: 3479360-4Report Type:Direct Company Report # Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare <strong>Mefloquine</strong><br />
Hyperhidrosis<br />
Hydrochloride<br />
Palpitations (Lariam) PS 1T WEEKLY<br />
Fear<br />
Date:03/27/00ISR Number: 3480434-2Report Type:Expedited (15-DaCompany Report #214472 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dysgraphia Foreign Lariam<br />
Clumsiness Health Tablets(<strong>Mefloquine</strong><br />
Cerebellar Atrophy Professional Hydrochloride) 250<br />
Neurological Examination Mg PS ORAL 250 MG 1 PER<br />
Abnormal<br />
WEEK ORAL<br />
Coordination Abnormal<br />
Dysarthria<br />
Cerebellar Syndrome<br />
Movement Disorder<br />
Balance Disorder<br />
Date:03/30/00ISR Number: 3482118-3Report Type:Expedited (15-DaCompany Report #231507 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haemolysis Foreign Lariam Tablets<br />
Initial or Prolonged Inborn Error Of Literature (<strong>Mefloquine</strong><br />
Required Metabolism Health Hydrochloride) PS ORAL 1.5 GRAM<br />
Intervention to Malaria Professional DAILY 1 PER 8<br />
Prevent Permanent Fatigue HOUR ORAL<br />
Impairment/Damage Anaemia<br />
Pyrexia<br />
Chromaturia<br />
Date:04/05/00ISR Number: 3484145-9Report Type:Expedited (15-DaCompany Report #231049 Age:32 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Foreign Lariam Tablets<br />
Initial or Prolonged Vomiting Health (<strong>Mefloquine</strong><br />
Vertigo Professional Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Nystagmus<br />
PER WEEK ORAL<br />
Alanine Aminotransferase<br />
Increased<br />
Ventoline (Albuterol<br />
Or Albuterol<br />
Sulfate)<br />
C
Date:04/05/00ISR Number: 3484146-0Report Type:Expedited (15-DaCompany Report #231053 Age:62 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Dyspnoea<br />
Csf Protein Increased<br />
Gait Disturbance<br />
Guillain-Barre Syndrome<br />
Bacterial Infection<br />
03-Apr-2012 09:37 AM Page: 179
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sensory Disturbance<br />
Myalgia<br />
Dizziness Report Source Product Role Manufacturer Route Dose Duration<br />
Blood Immunoglobulin G Foreign Lariam Tablets<br />
Increased Other (<strong>Mefloquine</strong><br />
Hyporeflexia Hydrochloride) 250<br />
Muscular Weakness Mg PS ORAL 250 MG 1 PER<br />
Paraesthesia<br />
Proscar<br />
(Finasteride)<br />
C<br />
WEEK ORAL<br />
Date:04/05/00ISR Number: 3484147-2Report Type:Expedited (15-DaCompany Report #218956 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Guillain-Barre Syndrome Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL 70 DAY<br />
Date:04/05/00ISR Number: 3484148-4Report Type:Expedited (15-DaCompany Report #218954 Age:23 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Personality Change Foreign Lariam Tablets<br />
Initial or Prolonged Pyrexia Health (<strong>Mefloquine</strong><br />
Delusion Professional Hydrochloride) 250<br />
Neurodermatitis Other Mg PS ORAL 250 MG 1 PER<br />
Hypotonia WEEK ORAL 61 DAY<br />
Dermatitis<br />
Psychotic Disorder<br />
Cachexia<br />
Diarrhoea<br />
Abscess<br />
Muscle Disorder<br />
Date:04/05/00ISR Number: 3484149-6Report Type:Expedited (15-DaCompany Report #218938 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Leukocytosis Foreign Lariam Tablets<br />
Initial or Prolonged Diarrhoea Health (<strong>Mefloquine</strong><br />
Dizziness Professional Hydrochloride) 250<br />
Pyrexia Other Mg PS ORAL ORAL<br />
Vomiting<br />
Blood Bilirubin Increased
Date:04/05/00ISR Number: 3484150-2Report Type:Expedited (15-DaCompany Report #218934 Age:32 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Expressive Language Foreign Lariam Tablets<br />
Disorder Health (<strong>Mefloquine</strong><br />
Dyskinesia Professional Hydrochloride) 250<br />
Mental Disorder Other Mg PS ORAL 250 MG 1 PER<br />
Grand Mal Convulsion<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 180
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/05/00ISR Number: 3484151-4Report Type:Expedited (15-DaCompany Report #218937 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gastric Ulcer Haemorrhage Foreign Lariam Tablets<br />
Melaena Health (<strong>Mefloquine</strong><br />
Dizziness Professional Hydrochloride) 250<br />
Pallor Other Mg PS ORAL 250 MG 1 PER<br />
Abdominal Distension<br />
Vaccines Nos<br />
(Vaccine Nos)<br />
C<br />
WEEK ORAL<br />
Date:04/05/00ISR Number: 3484152-6Report Type:Expedited (15-DaCompany Report #218933 Age:65 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Foreign Lariam Tablets<br />
Paraesthesia Health (<strong>Mefloquine</strong><br />
Nervous System Disorder Professional Hydrochloride) 250<br />
Brain Scan Abnormal Other Mg PS ORAL ORAL<br />
Date:04/05/00ISR Number: 3484159-9Report Type:Expedited (15-DaCompany Report #232718 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Psychotic Disorder Foreign Lariam Tablets<br />
Intervention to Dystonia Other (<strong>Mefloquine</strong><br />
Prevent Permanent Hydrochloride) PS ORAL 1 PER 1 WEEK<br />
Impairment/Damage<br />
Chlorpromazine<br />
(Chlorpromazine)<br />
SS<br />
ORAL<br />
Date:04/05/00ISR Number: 3484160-5Report Type:Expedited (15-DaCompany Report #232293 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sedation Foreign Lariam Tablets<br />
Initial or Prolonged Depression Health (<strong>Mefloquine</strong><br />
Circulatory Collapse Professional Hydrochloride) PS ORAL ORAL 7 DAY<br />
Date:04/05/00ISR Number: 3484188-5Report Type:Expedited (15-DaCompany Report #218942 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Other Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:04/06/00ISR Number: 3485079-6Report Type:Expedited (15-DaCompany Report #218953 Age:30 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Blood Glucose Decreased<br />
Circadian Rhythm Sleep<br />
Disorder<br />
03-Apr-2012 09:37 AM Page: 181
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Drug Interaction<br />
Loss Of Consciousness<br />
Bite Report Source Product Role Manufacturer Route Dose Duration<br />
Muscle Spasms Foreign Lariam Tablets<br />
Prostatitis Health (<strong>Mefloquine</strong><br />
Grand Mal Convulsion Professional Hydrochloride) 250<br />
Tremor Other Mg PS ORAL ORAL<br />
Urethritis<br />
Tongue Disorder<br />
Rytmonorm<br />
(Propafenone<br />
Hydrochloride) 300<br />
Mg SS ORAL ORAL<br />
Polio Vaccine<br />
(Poliomyelitis<br />
Vaccines)<br />
Hepatitis A Vaccine<br />
(Hepatitis A<br />
Vaccine)<br />
Diphtheria Vaccine<br />
(Diphtheria Toxoid)<br />
Tetanus Vaccine<br />
(Tetanus Toxoid)<br />
C<br />
C<br />
C<br />
C<br />
Date:04/07/00ISR Number: 3485171-6Report Type:Direct Company Report # Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Lariam 250 Mg Roche PS Roche ORAL 1 WEEK ORAL<br />
Congenital Anomaly Pyrexia<br />
Required<br />
Dysuria<br />
Intervention to<br />
Headache<br />
Prevent Permanent Insomnia<br />
Impairment/Damage Diarrhoea<br />
Arthralgia<br />
Hypoaesthesia<br />
Paraesthesia<br />
Back Pain<br />
Oropharyngeal Pain<br />
Neck Pain<br />
Date:04/07/00ISR Number: 3485372-7Report Type:Expedited (15-DaCompany Report #20000400108 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hemiplegia Foreign Prilosec PS ORAL 40 MG QD PO<br />
Initial or Prolonged Haemorrhage Health Lariam SS<br />
Cerebral Artery Occlusion Professional Imovane SS<br />
Aphasia<br />
Sedation<br />
Atrial Fibrillation
Date:04/07/00ISR Number: 3485913-XReport Type:Expedited (15-DaCompany Report #232768 Age:32 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Feeling Hot<br />
Paraesthesia<br />
Dizziness<br />
Antinuclear Antibody<br />
03-Apr-2012 09:37 AM Page: 182
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Positive<br />
Nausea<br />
Weight Increased<br />
Disturbance In Attention Report Source Product Role Manufacturer Route Dose Duration<br />
Vision Blurred Foreign Lariam Tablets<br />
Balance Disorder Health (<strong>Mefloquine</strong><br />
Vertigo Professional Hydrochloride) PS ORAL ORAL<br />
Decreased Appetite<br />
Wolff-Parkinson-White<br />
Syndrome<br />
Hypophagia<br />
Peripheral Coldness<br />
Headache<br />
Abdominal Pain<br />
Demyelination<br />
Hyperacusis<br />
Date:04/07/00ISR Number: 3485989-XReport Type:Expedited (15-DaCompany Report #232633 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebral Artery Occlusion Foreign Lariam Tablets<br />
Initial or Prolonged Atrial Flutter Other (<strong>Mefloquine</strong><br />
Aphasia Hydrochloride) 250<br />
Cerebral Infarction Mg PS ORAL 250 MG 1 PER<br />
Sedation<br />
ONE DOSE ORAL<br />
Cerebrovascular Accident<br />
Mopral (Omeprazole)<br />
Hemiplegia 20 Mg SS ORAL 40 MG DAILY<br />
Pyrexia<br />
ORAL<br />
Imovane (Zopiclone)<br />
7.5 Mg SS ORAL 7.5 MG DAILY<br />
ORAL<br />
Date:04/07/00ISR Number: 3485993-1Report Type:Expedited (15-DaCompany Report #232902 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lacrimation Decreased Foreign Lariam Tablets<br />
Dizziness Health (<strong>Mefloquine</strong><br />
Accommodation Disorder Professional Hydrochloride) 250<br />
Vertigo Other Mg PS ORAL ORAL<br />
Eye Pain<br />
Resochin<br />
Feeling Abnormal<br />
(Chloroquine<br />
Condition Aggravated Phosphate) C<br />
Headache<br />
Vision Blurred<br />
Nystagmus<br />
Gait Disturbance<br />
Date:04/13/00ISR Number: 3487950-8Report Type:Expedited (15-DaCompany Report #233138 Age:22 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Foreign Lariam Tablets<br />
Depression Health (<strong>Mefloquine</strong><br />
Suicidal Ideation Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 183
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/13/00ISR Number: 3488028-XReport Type:Expedited (15-DaCompany Report #229994 Age:19 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dyspnoea Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:04/14/00ISR Number: 3488468-9Report Type:Direct Company Report # Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Larium PS ORAL 1 TABLE ONCE<br />
Nightmare<br />
PER ORAL<br />
Anxiety<br />
Fear<br />
Panic Attack<br />
Hallucination<br />
Date:04/17/00ISR Number: 3489298-4Report Type:Expedited (15-DaCompany Report #228385 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania Health Lariam Tablets<br />
Initial or Prolonged Weight Decreased Professional (<strong>Mefloquine</strong><br />
Psychotic Disorder Hydrochloride) 250<br />
Abnormal Behaviour Mg PS ORAL 250 MG 1PER<br />
Psychomotor Hyperactivity<br />
WEEK ORAL<br />
Magical Thinking Chloroquine C<br />
Date:04/17/00ISR Number: 3489299-6Report Type:Expedited (15-DaCompany Report #233759 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Consumer Lariam Tablets<br />
Depression<br />
(<strong>Mefloquine</strong><br />
Pulmonary Oedema Hydrochloride) PS ORAL ORAL<br />
Mountain Sickness Acute<br />
Abnormal Dreams<br />
Lethargy<br />
Nightmare<br />
Date:04/18/00ISR Number: 3488728-1Report Type:Direct Company Report # Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Convulsion Lariam PS 250MG WEEKLY<br />
Dilantin SS 400MG DAILY
Date:04/18/00ISR Number: 3489912-3Report Type:Expedited (15-DaCompany Report #233467 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Foreign Lariam Tablets<br />
Visual Impairment Other (<strong>Mefloquine</strong><br />
Reading Disorder Hydrochloride) PS<br />
03-Apr-2012 09:37 AM Page: 184
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/18/00ISR Number: 3489913-5Report Type:Expedited (15-DaCompany Report #233599 Age:70 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrial Fibrillation Foreign Lariam Tablets<br />
Initial or Prolonged Syncope Other (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL DOSE FORM 1<br />
PER WEEK,<br />
ORAL<br />
Date:04/19/00ISR Number: 3490412-5Report Type:Expedited (15-DaCompany Report #233987 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Foreign Lariam Tablets<br />
Initial or Prolonged Feeling Abnormal Other (<strong>Mefloquine</strong><br />
Visual Impairment Hydrochloride) 250<br />
Disturbance In Attention Mg PS ORAL 250 MG 1 PER<br />
Fatigue<br />
WEEK ORAL<br />
Orthostatic Hypotension<br />
Ibuprofen<br />
Memory Impairment (Ibuprofen) C<br />
Hypoaesthesia<br />
Retching<br />
Speech Disorder<br />
Date:04/20/00ISR Number: 3490517-9Report Type:Expedited (15-DaCompany Report #233963 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haptoglobin Decreased Foreign Lariam Tablets<br />
Initial or Prolonged Haemolytic Anaemia Health (<strong>Mefloquine</strong><br />
Medication Error Professional Hydrochloride) PS ORAL ORAL<br />
Date:04/24/00ISR Number: 3492276-2Report Type:Expedited (15-DaCompany Report #232768 Age:32 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaise<br />
Feeling Hot<br />
Blood Glucose Decreased<br />
Wolff-Parkinson-White<br />
Syndrome<br />
Multiple Sclerosis<br />
Dizziness<br />
Vision Blurred<br />
Weight Increased<br />
Dehydration<br />
Pyrexia<br />
Antinuclear Antibody<br />
Positive<br />
Lethargy
Demyelination<br />
Hyperacusis<br />
Headache<br />
Asthenia<br />
Vertigo<br />
Vomiting<br />
Disorientation<br />
Peripheral Coldness<br />
03-Apr-2012 09:37 AM Page: 185
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Balance Disorder<br />
Migraine<br />
Nausea Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Foreign Lariam Tablets<br />
Abdominal Pain Health (<strong>Mefloquine</strong><br />
Paraesthesia Professional Hydrochloride) PS ORAL ORAL<br />
Sensation Of Foreign Body<br />
Disturbance In Attention<br />
Dysphagia<br />
Date:04/24/00ISR Number: 3492277-4Report Type:Expedited (15-DaCompany Report #234272 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cough Foreign Lariam Tablets<br />
Loss Of Consciousness Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL ORAL<br />
Date:04/26/00ISR Number: 3494185-1Report Type:Expedited (15-DaCompany Report #234319 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Foreign Lariam Tablets<br />
Paresis Health (<strong>Mefloquine</strong><br />
Delusion Professional Hydrochloride) PS ORAL 250 MG 1 PER<br />
Anxiety<br />
WEEK ORAL<br />
Sleep Disorder<br />
Alopecia<br />
Date:04/28/00ISR Number: 3494447-8Report Type:Expedited (15-DaCompany Report #234391 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia Consumer Lariam Tablets<br />
Accidental Overdose<br />
(<strong>Mefloquine</strong><br />
Loss Of Control Of Legs Hydrochloride) 250<br />
Blindness Transient Mg PS ORAL 250 MG 2 PER<br />
Nausea<br />
DAY ORAL<br />
Medication Error<br />
Date:05/02/00ISR Number: 3495586-8Report Type:Expedited (15-DaCompany Report #231507 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Foreign Lariam Tablets<br />
Initial or Prolonged Haemolytic Anaemia Literature (<strong>Mefloquine</strong><br />
Required Anaemia Health Hydrochloride) PS ORAL 1.5 GRAM<br />
Intervention to Malaria Professional DAILY 1 PER 8<br />
Prevent Permanent Inborn Error Of HOUR ORAL
Impairment/Damage<br />
Metabolism<br />
Fatigue<br />
Chromaturia<br />
03-Apr-2012 09:37 AM Page: 186
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/03/00ISR Number: 3495850-2Report Type:Direct Company Report # Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Bronchitis Consumer Lariam (<strong>Mefloquine</strong>)<br />
Intervention to Dyspnoea 250 Mg Once A Week PS Hoffman-Roche<br />
Prevent Permanent Tachycardia Pharmaceuticals ORAL PO ONCE A<br />
Impairment/Damage Anxiety WEEK 250 MG<br />
Levoquin<br />
C<br />
Date:05/03/00ISR Number: 3495910-6Report Type:Direct Company Report # Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Fatigue Health Larium (<strong>Mefloquine</strong><br />
Intervention to Headache Professional Hcl) 250 Mg PS 1 TABLET<br />
Prevent Permanent Insomnia (250MG) PER<br />
Impairment/Damage Abnormal Dreams WEEK<br />
Skin Discolouration<br />
Malaise<br />
Palpitations<br />
Hypertension<br />
Date:05/03/00ISR Number: 3497235-1Report Type:Expedited (15-DaCompany Report #234271 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Syncope Foreign Lariam Tablets<br />
Initial or Prolonged Malaise Other (<strong>Mefloquine</strong><br />
Blood Cholesterol Hydrochloride) PS ORAL ORAL<br />
Increased<br />
Pleural Fibrosis<br />
Renal Cyst<br />
Hypertension<br />
Sinoatrial Block<br />
Nausea<br />
Date:05/04/00ISR Number: 3495964-7Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Consumer Lariam PS ORAL ONCE A 3<br />
Fear<br />
TIMES ORAL<br />
Fear Of Disease<br />
Date:05/04/00ISR Number: 3502895-2Report Type:Direct Company Report # Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination <strong>Mefloquine</strong> PS
Abnormal Dreams<br />
03-Apr-2012 09:37 AM Page: 187
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/05/00ISR Number: 3497017-0Report Type:Expedited (15-DaCompany Report #234723 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Foreign <strong>Mefloquine</strong><br />
Drug Interaction Literature Hydrochloride<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS<br />
Ofloxacin<br />
(Ofloxacin)<br />
SS<br />
Date:05/05/00ISR Number: 3497133-3Report Type:Expedited (15-DaCompany Report #234722 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Foreign <strong>Mefloquine</strong><br />
Drug Interaction Literature Hydrochloride<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS<br />
Sparfloxacin<br />
(Sparfloxacin) SS<br />
Date:05/05/00ISR Number: 3497134-5Report Type:Expedited (15-DaCompany Report #234734 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Foreign <strong>Mefloquine</strong><br />
Drug Interaction Literature Hydrochloride<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS<br />
Ciprofloxacin<br />
(Ciprofloxacin) SS<br />
Date:05/05/00ISR Number: 3497136-9Report Type:Expedited (15-DaCompany Report #234751 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cough Foreign Lariam Tablets<br />
Initial or Prolonged Loss Of Consciousness Consumer (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:05/08/00ISR Number: 3497838-4Report Type:Expedited (15-DaCompany Report #234954 Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vith Nerve Paralysis Foreign Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Other Mg PS ORAL ORAL
Date:05/08/00ISR Number: 3497843-8Report Type:Direct Company Report # Age:35 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Anxiety<br />
Constipation<br />
03-Apr-2012 09:37 AM Page: 188
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorientation<br />
Fatigue<br />
Dizziness Report Source Product Role Manufacturer Route Dose Duration<br />
Larium 250mg Roche PS Roche ORAL ORALLY<br />
Date:05/08/00ISR Number: 3498350-9Report Type:Expedited (15-DaCompany Report #233329 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Feeling Drunk Health Inc ORAL 250 MG 1 PER<br />
Dizziness Professional WEEK ORAL<br />
Balance Disorder<br />
Memory Impairment<br />
Nystagmus<br />
Date:05/08/00ISR Number: 3498353-4Report Type:Expedited (15-DaCompany Report #234272 Age:44 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Loss Of Consciousness Foreign Lariam PS Hoffmann La Roche<br />
Cough Health Inc ORAL ORAL<br />
Professional<br />
Allopurinol<br />
(Allopurinol)<br />
C<br />
Date:05/11/00ISR Number: 3498920-8Report Type:Direct Company Report # Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Consumer Lariam PS ORAL 1 PER WEEK<br />
Crying Other ORAL<br />
Hallucination<br />
Gastrointestinal Disorder<br />
Panic Attack<br />
Sedation<br />
Mood Altered<br />
Balance Disorder<br />
Visual Impairment<br />
Date:05/16/00ISR Number: 3500997-8Report Type:Expedited (15-DaCompany Report #235343 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Epileptic Aura Foreign Lariam Tablets<br />
Aphasia Health (<strong>Mefloquine</strong><br />
Loss Of Consciousness Professional Hydrochloride) 250<br />
Other Mg PS Hoffmann La Roche<br />
Inc ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:05/17/00ISR Number: 3501085-7Report Type:Direct Company Report # Age:39 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
03-Apr-2012 09:37 AM Page: 189
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disability<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nausea Consumer Lariam-<strong>Mefloquine</strong><br />
Back Pain 250mg Roche PS Roche ORAL 250MG 1 PER<br />
Emotional Disorder<br />
WEEK ORAL<br />
Dizziness<br />
Diarrhoea<br />
Muscle Spasms<br />
Loss Of Consciousness<br />
Anxiety<br />
Hypoaesthesia<br />
Paraesthesia<br />
Panic Attack<br />
Pain In Extremity<br />
Tinnitus<br />
Abnormal Dreams<br />
Neck Pain<br />
Headache<br />
Abdominal Pain<br />
Date:05/24/00ISR Number: 3504590-2Report Type:Direct Company Report # Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Health <strong>Mefloquine</strong>-Lariam-/<br />
Paranoia Professional 250 Mg. PS ORAL 1 WEEK ORAL 1 WK<br />
Paraesthesia<br />
Panic Attack<br />
Pyrexia<br />
Dyspnoea<br />
Palpitations<br />
Sleep Disorder<br />
Diarrhoea<br />
Dizziness<br />
Decreased Appetite<br />
Weight Decreased<br />
Nervousness<br />
Cold Sweat<br />
Date:05/26/00ISR Number: 3505463-1Report Type:Expedited (15-DaCompany Report #234272 Age:44 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cough Foreign Lariam PS Hoffmann La Roche<br />
Syncope Health Inc ORAL ORAL<br />
Professional<br />
Allopurinol<br />
(Allopurinol)<br />
C
Date:05/31/00ISR Number: 3506616-9Report Type:Expedited (15-DaCompany Report #236845 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Pregnancy Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Abortion Incomplete Health Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Antepartum Haemorrhage Professional WEEK ORAL<br />
Impairment/Damage Maternal Drugs Affecting<br />
Foetus<br />
03-Apr-2012 09:37 AM Page: 190
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/01/00ISR Number: 3507011-9Report Type:Expedited (15-DaCompany Report #236875 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Abdominal Pain Upper Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL ORAL<br />
Professional<br />
Other<br />
Date:06/02/00ISR Number: 3507714-6Report Type:Expedited (15-DaCompany Report #940320860001 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anaemia Foreign Lariam PS Hoffmann La Roche<br />
Hepatitis A Antibody Consumer Inc ORAL 250 MG 1 PER<br />
Positive Health WEEK ORAL<br />
Pain<br />
Professional<br />
Complement Factor<br />
Decreased<br />
Red Blood Cell<br />
Sedimentation Rate<br />
Decreased<br />
Gastrointestinal Disorder<br />
Sleep Disorder<br />
Condition Aggravated<br />
Tinnitus<br />
Dyspnoea<br />
Arrhythmia<br />
Immunosuppression<br />
Swelling<br />
Blood Cholesterol<br />
Increased<br />
Blepharitis<br />
Pain In Jaw<br />
Immunology Test Abnormal<br />
Trigeminal Nerve Disorder<br />
Lymphadenopathy<br />
Fatigue<br />
Blood Immunoglobulin G<br />
Increased<br />
Angiopathy<br />
Immune System Disorder<br />
Laryngeal Disorder<br />
Autoimmune Disorder<br />
Antinuclear Antibody<br />
Positive<br />
Mononucleosis Heterophile<br />
Test Positive<br />
Cd4 Lymphocytes Decreased<br />
Arterial Disorder<br />
Hyperaesthesia<br />
Arthropathy
Date:06/02/00ISR Number: 3507717-1Report Type:Expedited (15-DaCompany Report #236161 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Purpura Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Renal Impairment Health Inc ORAL 1 DOSE FORM 1<br />
Hepatic Enzyme Increased Professional PER WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 191
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Previscan<br />
(Fluindione) SS ORAL ORAL<br />
Lasilix (Furosemide) C<br />
Seloken (Metoprolol<br />
Tartrate)<br />
C<br />
Pyostacine<br />
(Pristinamycin) C<br />
Cordarone<br />
(Amiodarone)<br />
C<br />
Levothyrox<br />
(Levothyroxine<br />
Sodium)<br />
C<br />
Date:06/02/00ISR Number: 3507718-3Report Type:Expedited (15-DaCompany Report #233138 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Electroencephalogram Health Inc ORAL 250 MG 1 PER<br />
Abnormal Professional WEEK ORAL<br />
Depression<br />
Social Avoidant Behaviour<br />
Hallucination, Auditory<br />
Gait Disturbance<br />
Disturbance In Attention<br />
Anxiety<br />
Personality Disorder<br />
Rash Erythematous<br />
Mood Swings<br />
Decreased Appetite<br />
Psychotic Disorder<br />
Feeling Abnormal<br />
Cognitive Disorder<br />
Insomnia<br />
Delusion<br />
Blood Bilirubin Increased<br />
Date:06/05/00ISR Number: 3507999-6Report Type:Direct Company Report # Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypertension Lariam 500mg PS<br />
Initial or Prolonged Pyrexia<br />
Migraine<br />
Toxicity To Various<br />
Agents<br />
Depressed Mood<br />
Diarrhoea<br />
Nightmare
Date:06/06/00ISR Number: 3509002-0Report Type:Expedited (15-DaCompany Report #237147 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Urinary Retention Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Dermatitis Other Inc ORAL 250 MG ORAL<br />
Suspected<br />
Unspecified Drug<br />
03-Apr-2012 09:37 AM Page: 192
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
(Generic<br />
Component(S)<br />
Unknown) SS ORAL 1 DOSE FORM<br />
DAILY ORAL<br />
Date:06/06/00ISR Number: 3509005-6Report Type:Expedited (15-DaCompany Report #236953 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatic Enzyme Increased Lariam PS Hoffmann La Roche<br />
Myopathy Inc ORAL 250 MG 1 PER<br />
Nerve Root Lesion WEEK ORAL 35 DAY<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Paraesthesia<br />
Drug Level Above<br />
Therapeutic<br />
Insomnia<br />
Date:06/06/00ISR Number: 3510082-7Report Type:Periodic Company Report #211352 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Interaction Health Lariam PS Hoffmann La Roche<br />
Chills Professional Inc ORAL 250 MG 1 PER<br />
Pyrexia<br />
1 WEEK ORAL<br />
Cipro SS ORAL 1 DOSE FORM<br />
ORAL<br />
Date:06/06/00ISR Number: 3510083-9Report Type:Periodic Company Report #211684 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Acne Consumer Lariam PS Hoffmann La Roche<br />
Nightmare Inc ORAL 250 MG 1 PER<br />
Photophobia<br />
WEEK ORAL<br />
Ibuprofen<br />
C<br />
Date:06/06/00ISR Number: 3510084-0Report Type:Periodic Company Report #212626 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Other Lariam PS Hoffmann La Roche<br />
Dyspepsia Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/06/00ISR Number: 3510085-2Report Type:Periodic Company Report #212649 Age:35 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Consumer Lariam PS Hoffmann La Roche<br />
Eosinophilia Inc ORAL 250 MG 1 PER<br />
Haematuria<br />
WEEK ORAL<br />
Vision Blurred<br />
Blood Pressure Increased<br />
03-Apr-2012 09:37 AM Page: 193
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510086-4Report Type:Periodic Company Report #212713 Age:54 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Heart Rate Increased Other Lariam PS Hoffmann La Roche<br />
Coordination Abnormal Inc ORAL 250 MG 1 PER<br />
Tremor<br />
WEEK ORAL<br />
Hyperhidrosis Progesterone C<br />
Anxiety Ogen C<br />
Headache<br />
Date:06/06/00ISR Number: 3510087-6Report Type:Periodic Company Report #215004 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Consumer Lariam PS Hoffmann La Roche<br />
Anxiety Inc ORAL 250 MG 1 PER<br />
Arthralgia<br />
WEEK ORAL<br />
Abnormal Dreams<br />
Date:06/06/00ISR Number: 3510088-8Report Type:Periodic Company Report #216929 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gingival Erosion Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Calcium Supplements C<br />
Date:06/06/00ISR Number: 3510089-XReport Type:Periodic Company Report #217582 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Consumer Lariam PS Hoffmann La Roche<br />
Fatigue Inc ORAL ORAL<br />
Dizziness<br />
Oropharyngeal Pain<br />
Date:06/06/00ISR Number: 3510090-6Report Type:Periodic Company Report #218907 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amenorrhoea Consumer Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 250 MG 1 PER<br />
Professional<br />
WEEK ORAL<br />
Birth Control Pills SS ORAL ORAL<br />
Date:06/06/00ISR Number: 3510091-8Report Type:Periodic Company Report #219666 Age:55 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaise Health Lariam PS Hoffmann La Roche<br />
Vertigo Professional Inc ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 194
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510094-3Report Type:Periodic Company Report #219667 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Health Lariam PS Hoffmann La Roche<br />
Asthenia Professional Inc ORAL ORAL<br />
Mental Impairment<br />
Balance Disorder<br />
Date:06/06/00ISR Number: 3510097-9Report Type:Periodic Company Report #219671 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Health Lariam PS Hoffmann La Roche<br />
Malaise Professional Inc ORAL ORAL<br />
Date:06/06/00ISR Number: 3510098-0Report Type:Periodic Company Report #219674 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Health Lariam PS Hoffmann La Roche<br />
Malaise Professional Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/06/00ISR Number: 3510100-6Report Type:Periodic Company Report #220897 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sedation Consumer Lariam PS Hoffmann La Roche<br />
Mood Swings Inc ORAL 250 MG DAILY<br />
Red Blood Cell Count<br />
ORAL<br />
Increased<br />
Oral Contraceptive<br />
Dizziness Nos C<br />
Electrocardiogram<br />
Abnormal<br />
Conversion Disorder<br />
Electrocardiogram Change<br />
Nightmare<br />
Psychotic Disorder<br />
Confusional State<br />
Disorientation<br />
Paranoia<br />
Insomnia<br />
Crying<br />
Night Sweats<br />
Hallucination<br />
Date:06/06/00ISR Number: 3510103-1Report Type:Periodic Company Report #220939 Age:36 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Consumer Lariam PS Hoffmann La Roche<br />
Anxiety Other Inc ORAL 250 MG 1 PER<br />
Depression<br />
WEEK ORAL<br />
Dehydration Lotensin C<br />
Panic Attack<br />
Heart Rate Increased<br />
03-Apr-2012 09:37 AM Page: 195
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510106-7Report Type:Periodic Company Report #222392 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Body Temperature Consumer Lariam PS Hoffmann La Roche<br />
Increased Inc ORAL 250 MG 1 PER<br />
Nasopharyngitis<br />
WEEK ORAL<br />
Fatigue<br />
Blood Pressure Diastolic<br />
Increased<br />
Urinary Tract Infection<br />
Tachycardia<br />
Anxiety<br />
Dizziness<br />
Decreased Appetite<br />
Productive Cough<br />
Dyspnoea<br />
Oropharyngeal Pain<br />
Pyrexia<br />
Date:06/06/00ISR Number: 3510107-9Report Type:Periodic Company Report #223365 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Health Lariam PS Hoffmann La Roche<br />
Diarrhoea Professional Inc ORAL 500 MG DAILY<br />
ORAL 49 DAY<br />
Date:06/06/00ISR Number: 3510109-2Report Type:Periodic Company Report #223368 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oedema Peripheral Consumer Lariam PS Hoffmann La Roche<br />
Lethargy Inc ORAL 250 MG 1 PER<br />
WEEK ORAL 28 DAY<br />
Date:06/06/00ISR Number: 3510111-0Report Type:Periodic Company Report #224970 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Health Lariam PS Hoffmann La Roche<br />
Tachycardia Professional Inc ORAL 250 MG 1 PER<br />
Dizziness<br />
WEEK ORAL<br />
Pyrexia Fosamax C<br />
Anxiety<br />
Diarrhoea<br />
Hypoaesthesia<br />
Upper Respiratory Tract<br />
Infection<br />
Fatigue<br />
Dysphoria
Hypoaesthesia Oral<br />
Disturbance In Attention<br />
Dyspnoea<br />
Abdominal Pain<br />
Hypoventilation<br />
03-Apr-2012 09:37 AM Page: 196
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510113-4Report Type:Periodic Company Report #227110 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Health Lariam PS Hoffmann La Roche<br />
Chest Discomfort Professional Inc ORAL 250 MG 1 PER<br />
Chest Pain<br />
WEEK ORAL<br />
Anxiety<br />
Date:06/06/00ISR Number: 3510116-XReport Type:Periodic Company Report #200733 Age:65 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Attack Consumer Lariam PS Hoffmann La Roche<br />
Nausea Inc ORAL 250 MG 1 PER<br />
Headache<br />
WEEK ORAL<br />
Anxiety<br />
Estrogen (Estrogens<br />
Paraesthesia Nos) C<br />
Tremor<br />
Progestin (Progestin<br />
Dizziness Nos) C<br />
Hyperhidrosis<br />
Palpitations<br />
Abnormal Dreams<br />
Decreased Appetite<br />
Chills<br />
Chest Pain<br />
Alopecia<br />
Vomiting<br />
Date:06/06/00ISR Number: 3510118-3Report Type:Periodic Company Report #201043 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Other Lariam PS Hoffmann La Roche<br />
Ear Disorder Inc ORAL 250 MG 1 PER<br />
Eye Disorder<br />
WEEK ORAL<br />
Discomfort<br />
Headache<br />
Date:06/06/00ISR Number: 3510120-1Report Type:Periodic Company Report #930201053001 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Health Lariam PS Hoffmann La Roche<br />
Asthenia Professional Inc ORAL ORAL<br />
Tachycardia<br />
Irritability<br />
Hyperacusis<br />
Insomnia<br />
Disturbance In Attention<br />
Paranoia
Date:06/06/00ISR Number: 3510124-9Report Type:Periodic Company Report #208413 Age:36 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Tachycardia<br />
Flushing<br />
03-Apr-2012 09:37 AM Page: 197
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Syncope<br />
Hypotension Report Source Product Role Manufacturer Route Dose Duration<br />
Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 1 PER WEEK<br />
ORAL<br />
Synthroid<br />
(Levothyroxine<br />
Sodium)<br />
C<br />
Date:06/06/00ISR Number: 3510127-4Report Type:Periodic Company Report #209051 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nuclear Magnetic Health Lariam PS Hoffmann La Roche<br />
Resonance Imaging Professional Inc ORAL 250 MG 1 PER<br />
Abnormal<br />
WEEK ORAL<br />
Tinnitus<br />
Malaria<br />
Alopecia<br />
Vision Blurred<br />
Drug Ineffective<br />
Date:06/06/00ISR Number: 3510131-6Report Type:Periodic Company Report #211281 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Spasms Other Lariam PS Hoffmann La Roche<br />
Syncope Inc ORAL 250 MG 1 PER<br />
Abdominal Pain<br />
WEEK ORAL<br />
Coordination Abnormal<br />
Diarrhoea<br />
Dizziness<br />
Date:06/06/00ISR Number: 3510134-1Report Type:Periodic Company Report #212816 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Condition Aggravated Consumer Lariam PS Hoffmann La Roche<br />
Balance Disorder Health Inc ORAL 250 MG 1 PER<br />
Panic Attack Professional WEEK ORAL<br />
Phobia<br />
Date:06/06/00ISR Number: 3510137-7Report Type:Periodic Company Report #213466 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eye Movement Disorder Health Lariam PS Hoffmann La Roche<br />
Loss Of Consciousness Professional Inc ORAL 250 MG 1 PER
Crying ONE DOSE ORAL<br />
Convulsion<br />
Bite<br />
Tremor<br />
Amnesia<br />
Fall<br />
03-Apr-2012 09:37 AM Page: 198
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510140-7Report Type:Periodic Company Report #214497 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Other Lariam PS Hoffmann La Roche<br />
Drug Ineffective Inc ORAL 250 MG 1 PER<br />
Malaria<br />
WEEK ORAL<br />
Diarrhoea<br />
Headache<br />
Date:06/06/00ISR Number: 3510142-0Report Type:Periodic Company Report #217162 Age:65 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Health Lariam PS Hoffmann La Roche<br />
Pyrexia Professional Inc ORAL 250 MG 1 PER<br />
Drug Ineffective<br />
1 WEEK ORAL<br />
Haematocrit Decreased<br />
Chills<br />
White Blood Cell Count<br />
Decreased<br />
Platelet Count Decreased<br />
Date:06/06/00ISR Number: 3510147-XReport Type:Periodic Company Report #223678 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Alopecia Consumer Lariam PS Hoffmann La Roche<br />
Dizziness Inc ORAL 250 MG 1 PER<br />
Hallucination<br />
WEEK ORAL<br />
Vomiting<br />
Suicidal Ideation<br />
Date:06/06/00ISR Number: 3510150-XReport Type:Periodic Company Report #227106 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dyspnoea Health Lariam PS Hoffmann La Roche<br />
Constipation Professional Inc ORAL 250 MG 1 PER<br />
Choking Other WEEK ORAL<br />
Muscle Twitching<br />
Ortho-Novum (Ethinyl<br />
Diarrhoea<br />
Estradiol Or<br />
Panic Attack<br />
Mestranol/Norethidro<br />
ne)<br />
C<br />
Aspirin (Aspirin) C<br />
Date:06/06/00ISR Number: 3510154-7Report Type:Periodic Company Report #230635 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Syncope Health Lariam PS Hoffmann La Roche<br />
Hyperhidrosis Professional Inc ORAL 20 MG 1 PER<br />
Dyspnoea<br />
WEEK ORAL<br />
Oxygen Saturation<br />
Decreased<br />
Bradycardia<br />
03-Apr-2012 09:37 AM Page: 199
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510156-0Report Type:Periodic Company Report #205795 Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Iritis Health Lariam PS Hoffmann La Roche<br />
Scotoma Professional Inc ORAL ORAL<br />
Eye Pain<br />
Dermatitis Atopic<br />
Blindness<br />
Date:06/06/00ISR Number: 3510158-4Report Type:Periodic Company Report #205967 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tongue Disorder Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Relafen (Nabmetone) C<br />
Prednisone<br />
(Prednisone)<br />
C<br />
Plaquenil<br />
(Hydrochloroquine<br />
Sulfate)<br />
C<br />
Date:06/06/00ISR Number: 3510160-2Report Type:Periodic Company Report #207060 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Other Lariam PS Hoffmann La Roche<br />
Petechiae Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/06/00ISR Number: 3510164-XReport Type:Periodic Company Report #208862 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Health Lariam PS Hoffmann La Roche<br />
Pyrexia Professional Inc ORAL 250 MG 1 PER<br />
Blood Thyroid Stimulating<br />
WEEK ORAL<br />
Hormone Increased<br />
Vitamin C (Ascorbic<br />
Liver Function Test Acid) C<br />
Abnormal<br />
Inhaler Nos<br />
Chills (Inhalant Nos) C<br />
Multivitamin<br />
(Multivitamin Nos) C<br />
Beta-Carotene (Beta<br />
Carotene)<br />
C<br />
Unknown Drug<br />
(Generic<br />
Component(S)<br />
Unknown)<br />
C
Ventolin Inhaler<br />
(Albuterol)<br />
Calcium (Calcium<br />
Nos)<br />
C<br />
C<br />
03-Apr-2012 09:37 AM Page: 200
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/06/00ISR Number: 3510165-1Report Type:Periodic Company Report #208888 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Acuity Reduced Other Lariam PS Hoffmann La Roche<br />
Gout Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/06/00ISR Number: 3510169-9Report Type:Periodic Company Report #209840 Age:16 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haematuria Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Date:06/06/00ISR Number: 3510172-9Report Type:Periodic Company Report #211009 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Increased Health Lariam PS Hoffmann La Roche<br />
Pallor Professional Inc ORAL 250 MG 1 PER<br />
Dizziness<br />
WEEK ORAL<br />
Date:06/06/00ISR Number: 3510191-2Report Type:Periodic Company Report #228486 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Consumer Lariam PS Hoffmann La Roche<br />
Vertigo Inc ORAL 1.25 GRAM 1<br />
Memory Impairment<br />
PER ONE DOSE<br />
Disturbance In Attention<br />
ORAL<br />
Chills<br />
Headache<br />
Tinnitus<br />
Insomnia<br />
Date:06/06/00ISR Number: 3510193-6Report Type:Periodic Company Report #231666 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Consumer Lariam PS Hoffmann La Roche<br />
Depression<br />
Inc<br />
Fatigue Ativan (Lorazepam) C<br />
Celexa (Citalopram) C<br />
Date:06/06/00ISR Number: 3510194-8Report Type:Periodic Company Report #232888 Age:21 YR Gender:Male I/FU:I<br />
Outcome<br />
PT
Other Serious<br />
Feeling Abnormal<br />
Asthma<br />
Vertigo<br />
Panic Attack<br />
Paranoia<br />
Anxiety<br />
Fatigue<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 201
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Restlessness<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 1 PER WEEK<br />
ORAL<br />
Date:06/06/00ISR Number: 3510196-1Report Type:Periodic Company Report #112461 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lethargy Consumer Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Chills Health Inc ORAL 250 MG 1 PER<br />
Drug Ineffective Professional WEEK ORAL<br />
Diarrhoea<br />
Prilosec<br />
Hyperhidrosis (Omeprazole) C<br />
Vomiting<br />
Malaria<br />
Abdominal Pain<br />
Dehydration<br />
Leukocytosis<br />
Pyrexia<br />
Date:06/06/00ISR Number: 3510198-5Report Type:Periodic Company Report #202382 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Other Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Depression Inc ORAL 250 MG 1 PER<br />
Delusion<br />
WEEK ORAL<br />
Date:06/06/00ISR Number: 3510200-0Report Type:Periodic Company Report #203056 Age:6 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Confusional State Other Lariam PS Hoffmann La Roche<br />
Convulsion Inc ORAL 250 MG 1 PER<br />
Amnesia<br />
WEEK ORAL<br />
Date:06/08/00ISR Number: 3510582-XReport Type:Expedited (15-DaCompany Report #237264 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Spinal Fracture Foreign Lariam PS Hoffmann La Roche<br />
Convulsion Consumer Inc ORAL ORAL<br />
Dizziness<br />
Fall<br />
Drug Ineffective
Date:06/08/00ISR Number: 3510583-1Report Type:Expedited (15-DaCompany Report #237590 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Foreign Lariam PS Hoffmann La Roche<br />
Accidental Exposure Health Inc<br />
Professional<br />
03-Apr-2012 09:37 AM Page: 202
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/00ISR Number: 3510584-3Report Type:Expedited (15-DaCompany Report #237544 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaria Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Malaise Health Inc<br />
Professional<br />
Date:06/08/00ISR Number: 3510585-5Report Type:Expedited (15-DaCompany Report #237143 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Hallucination Other Inc ORAL 1 DOSE FORM<br />
Arthralgia<br />
ORAL<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Jaundice<br />
Nausea<br />
Asthenia<br />
Date:06/08/00ISR Number: 3510586-7Report Type:Expedited (15-DaCompany Report #236875 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Pain Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL ORAL<br />
Professional<br />
Other<br />
Date:06/09/00ISR Number: 3510997-XReport Type:Expedited (15-DaCompany Report #237587 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Foreign Lariam PS Hoffmann La Roche<br />
Accidental Exposure Health Inc<br />
Professional<br />
Date:06/09/00ISR Number: 3517524-1Report Type:Periodic Company Report #73316 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Rash Pruritic Consumer Accutane PS Hlr Technology ORAL 20 MG 3 PER<br />
Dermatitis<br />
DAY ORAL<br />
Skin Wrinkling<br />
Lariam Tablets<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250
Mg SS ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Ortho Novum (Ethinyl<br />
Estradiol Or<br />
Mestranol/Norethindr<br />
one)<br />
C<br />
Synthroid<br />
(Levothyroxine<br />
03-Apr-2012 09:37 AM Page: 203
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sodium)<br />
C<br />
Date:06/13/00ISR Number: 3513543-XReport Type:Expedited (15-DaCompany Report #237062 Age:65 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Agitation Other Inc ORAL 1 DOSE FORM 1<br />
Anxiety<br />
PER WEEK ORAL<br />
Delirium<br />
Date:06/15/00ISR Number: 3514429-7Report Type:Expedited (15-DaCompany Report #237672 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Foreign Lariam PS Hoffmann La Roche<br />
Cataract Consumer Inc ORAL ORAL<br />
Visual Impairment<br />
Disturbance In Attention<br />
Date:06/20/00ISR Number: 3516339-8Report Type:Expedited (15-DaCompany Report #215628 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Ultrasound Antenatal Foreign Lariam PS Hoffmann La Roche<br />
Screen Abnormal Health Inc ORAL 250 MG 1 PER<br />
Trisomy 21 Professional WEEK ORAL<br />
Amniocentesis Abnormal<br />
Abortion Induced<br />
Foetal Disorder<br />
Maternal Drugs Affecting<br />
Foetus<br />
Cystic Lymphangioma<br />
Pregnancy<br />
Date:06/20/00ISR Number: 3516343-XReport Type:Expedited (15-DaCompany Report #237264 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Foreign Lariam PS Hoffmann La Roche<br />
Malaria Consumer Inc ORAL ORAL<br />
Paraesthesia<br />
Spinal Fracture<br />
Fall<br />
Drug Ineffective<br />
Feeling Abnormal<br />
Dizziness<br />
Headache
Date:06/21/00ISR Number: 3517156-5Report Type:Expedited (15-DaCompany Report #237147 Age:39 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Anuria<br />
Thrombocytosis<br />
Urinary Retention<br />
Pyrexia<br />
03-Apr-2012 09:37 AM Page: 204
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dysuria<br />
Erythema<br />
Hypokalaemia Report Source Product Role Manufacturer Route Dose Duration<br />
Exfoliative Rash Foreign Lariam PS Hoffmann La Roche<br />
C-Reactive Protein Other Inc ORAL 250 MG ORAL<br />
Increased<br />
Suspected<br />
Skin Disorder<br />
Unspecified Drug<br />
Cystitis<br />
(Generic<br />
Neurogenic Bladder<br />
Component(S)<br />
Pyelonephritis Unknown) SS 6 DOSE FORM<br />
Dermatitis<br />
DAILY<br />
Suspected<br />
Unspecified Drug<br />
(Generic<br />
Component(S)<br />
Unknown) SS ORAL 1 DOSE FORM<br />
DAILY ORAL<br />
Date:06/21/00ISR Number: 3517158-9Report Type:Expedited (15-DaCompany Report #238384 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Liver Function Test Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Abnormal Health Inc ORAL ORAL<br />
Abdominal Pain<br />
Professional<br />
Date:06/23/00ISR Number: 3518681-3Report Type:Expedited (15-DaCompany Report #238706 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tendon Rupture Foreign Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL 70 DAY<br />
Atenolol (Atenolol) C<br />
Date:06/26/00ISR Number: 3520477-3Report Type:Expedited (15-DaCompany Report #238943 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoproteinaemia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Blood Bilirubin Increased Health Inc ORAL 250 MG 1 PER<br />
Jaundice Professional WEEK ORAL<br />
Ascites<br />
Date:06/27/00ISR Number: 3520664-4Report Type:Expedited (15-DaCompany Report #238749 Age: Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaria<br />
Tremor
Disability<br />
Hallucination<br />
Sensory Disturbance<br />
Pupils Unequal<br />
Chest Pain<br />
Coordination Abnormal<br />
Fatigue<br />
Drug Ineffective<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 205
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Visual Impairment<br />
Emotional Disorder<br />
Depression Report Source Product Role Manufacturer Route Dose Duration<br />
Pain In Extremity Foreign Lariam PS Hoffmann La Roche<br />
Blood Pressure Increased Consumer Inc ORAL ORAL<br />
Paranoia<br />
Muscle Twitching<br />
Disturbance In Attention<br />
Circulatory Collapse<br />
Nightmare<br />
Abnormal Dreams<br />
Asthenia<br />
Nervous System Disorder<br />
Feeling Of Body<br />
Temperature Change<br />
Date:06/27/00ISR Number: 3520667-XReport Type:Expedited (15-DaCompany Report #237264 Age:50 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Paraesthesia Foreign Lariam PS Hoffmann La Roche<br />
Dizziness Consumer Inc ORAL 250 MG 1 PER<br />
Headache Health WEEK ORAL<br />
Malaria<br />
Professional<br />
Feeling Abnormal<br />
Drug Ineffective<br />
Spinal Fracture<br />
Pain<br />
Fall<br />
Convulsion<br />
Date:06/27/00ISR Number: 3520673-5Report Type:Expedited (15-DaCompany Report #237544 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Malaria Health Inc ORAL ORAL<br />
Professional<br />
Date:06/28/00ISR Number: 3521532-4Report Type:Expedited (15-DaCompany Report #232833 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sleep Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Nausea Health Inc ORAL ORAL<br />
Tremor<br />
Professional<br />
Depression<br />
Mental Disorder<br />
Neuropathy Peripheral<br />
Depressed Mood
Date:06/28/00ISR Number: 3521542-7Report Type:Expedited (15-DaCompany Report #231053 Age:62 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Hyporeflexia<br />
Csf Protein Increased<br />
03-Apr-2012 09:37 AM Page: 206
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Lumbar Puncture Abnormal<br />
Dizziness<br />
Gait Disturbance Report Source Product Role Manufacturer Route Dose Duration<br />
Antibody Test Positive Foreign Lariam PS Hoffmann La Roche<br />
Myalgia Other Inc ORAL 250 MG 1 PER<br />
Muscular Weakness<br />
WEEK ORAL<br />
Polyneuropathy<br />
Proscar<br />
Balance Disorder (Finasteride) C<br />
Dyspnoea<br />
Paraesthesia<br />
Date:06/29/00ISR Number: 3522435-1Report Type:Expedited (15-DaCompany Report #238730 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Pressure Systolic Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Increased Other Inc ORAL 45 MG 1 PER<br />
Required Calcinosis WEEK ORAL<br />
Intervention to Enterococcal Bacteraemia Budesonide<br />
Prevent Permanent Computerised Tomogram (Budesonide) C<br />
Impairment/Damage Abnormal Warfarin (Warfarin<br />
Vomiting Sodium) C<br />
Low Cardiac Output<br />
Bumetanide<br />
Syndrome (Bumetanide) C<br />
Malaise<br />
Metformin<br />
Red Blood Cell (Metformin) C<br />
Sedimentation Rate<br />
Amiodarone<br />
Increased (Amiodarone) C<br />
Pyrexia<br />
Loss Of Consciousness<br />
Hepatitis B Antibody<br />
Positive<br />
Urinary Tract Infection<br />
Hepatitis<br />
Dehydration<br />
C-Reactive Protein<br />
Increased<br />
Liver Function Test<br />
Abnormal<br />
Hepatic Function Abnormal<br />
Asthenia<br />
Torsade De Pointes<br />
Culture Urine Positive<br />
Circulatory Collapse<br />
Electrocardiogram Qt<br />
Prolonged<br />
Date:06/29/00ISR Number: 3522437-5Report Type:Expedited (15-DaCompany Report #239189 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Amyotrophic Lateral Foreign Lariam PS Hoffmann La Roche<br />
Sclerosis Other Inc ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 207
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/05/00ISR Number: 3524097-6Report Type:Direct Company Report # Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia<br />
Lariam 250mg<br />
Vomiting Hoffman/Roche PS Hoffman/Roche ORAL 250MG ONCE<br />
Feeling Cold<br />
WEEKLY ORAL<br />
Flushing<br />
Tylenol-Acetaminioph<br />
Pain en C<br />
Pyrexia<br />
Cold Sweat<br />
Diarrhoea<br />
Decreased Appetite<br />
Date:07/05/00ISR Number: 3524160-XReport Type:Expedited (15-DaCompany Report #236161 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Purpura Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Hepatic Infarction Health Inc ORAL 1 DOSE FORM 1<br />
Renal Impairment Professional PER WEEK ORAL<br />
Hepatic Enzyme Increased<br />
Previscan<br />
(Fluindione) SS ORAL ORAL<br />
Lasilix<br />
C<br />
Seloken<br />
C<br />
Pyostacine<br />
C<br />
Praxilene<br />
C<br />
Cordarone<br />
C<br />
Levothyrox<br />
C<br />
Date:07/05/00ISR Number: 3524263-XReport Type:Direct Company Report # Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gastrointestinal Pain Lariam 250mg<br />
Fatigue<br />
(Hoffman Laroche<br />
Back Pain Ltd) PS Hoffman Laroche Ltd ORAL 1 PILL , 1 X<br />
WEEK, ORAL<br />
047<br />
Date:07/05/00ISR Number: 3524267-7Report Type:Direct Company Report # Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Gastrointestinal Pain<br />
Lariam -250mg<br />
Eye Pain<br />
(Hoffmann-La Roche,<br />
Vomiting Ltd) PS Hoffmann-La Roche,<br />
Dizziness Ltd ORAL 1 PILL, 1X<br />
Headache<br />
WEEK, ORAL<br />
Diarrhoea 047<br />
Balance Disorder
Rash Pruritic<br />
Abdominal Pain Upper<br />
Discomfort<br />
03-Apr-2012 09:37 AM Page: 208
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/05/00ISR Number: 3524360-9Report Type:Expedited (15-DaCompany Report #237264 Age:50 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Spinal Fracture Foreign Lariam PS Hoffmann La Roche<br />
Pain Consumer Inc ORAL 250 MG 1 PER<br />
Malaria Health WEEK ORAL<br />
Convulsion<br />
Professional<br />
Paraesthesia<br />
Drug Ineffective<br />
Fall<br />
Headache<br />
Feeling Abnormal<br />
Dizziness<br />
Date:07/05/00ISR Number: 3524794-2Report Type:Expedited (15-DaCompany Report #239438 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Deafness Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Drug Level Above Other Inc ORAL ORAL<br />
Therapeutic<br />
Quinimax<br />
Medication Error<br />
(Cinchonidine/Cincho<br />
Tinnitus<br />
nine/Quinidine/Quini<br />
Electrocardiogram Qt ne) SS ORAL ORAL<br />
Prolonged<br />
Vomiting<br />
Date:07/07/00ISR Number: 3526201-2Report Type:Direct Company Report # Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Consumer Larium / Mefloquin<br />
Initial or Prolonged Depression 250mg PS ORAL 250MG/<br />
Amnesia<br />
WEEKLY/ ORAL<br />
Headache<br />
Insomnia<br />
Abnormal Behaviour<br />
Cranial Nerve Disorder<br />
Judgement Impaired<br />
Dislocation Of Vertebra<br />
Emotional Disorder<br />
Toxicity To Various<br />
Agents<br />
Nervous System Disorder<br />
Decreased Appetite<br />
Date:07/07/00ISR Number: 3526638-1Report Type:Expedited (15-DaCompany Report #228555 Age:33 YR Gender:Male I/FU:F<br />
Outcome<br />
PT
Death<br />
Splenomegaly<br />
Psychotic Disorder<br />
Completed Suicide<br />
Pyrexia<br />
Malaise<br />
Bone Pain<br />
Oropharyngeal Pain<br />
Burning Sensation<br />
03-Apr-2012 09:37 AM Page: 209
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Panic Disorder<br />
Dyspnoea<br />
Peripheral Coldness Report Source Product Role Manufacturer Route Dose Duration<br />
Dizziness Foreign Lariam PS Hoffmann La Roche<br />
Disorientation Literature Inc ORAL 500 MG 1 PER<br />
Headache Health ONE DOSE ORAL<br />
Malaria Professional Co-Trimoxazol C<br />
Depression Other Penicillin V C<br />
Anxiety Intravenous Glucose C<br />
Hypoaesthesia Paracetamol C<br />
Fear Codeine Phosphate C<br />
Cardiomyopathy Fansidar C<br />
Chills Quinine C<br />
Confusional State Antimalarial Agent C<br />
Myocarditis Aspirin C<br />
Diarrhoea<br />
Date:07/11/00ISR Number: 3527822-3Report Type:Expedited (15-DaCompany Report #239809 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Obsessive-Compulsive Foreign Lariam PS Hoffmann La Roche<br />
Disorder Other Inc ORAL 250 MG 1 PER<br />
Anxiety<br />
WEEK ORAL<br />
Aggression<br />
Date:07/11/00ISR Number: 3527823-5Report Type:Expedited (15-DaCompany Report #203837 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
PT<br />
Insomnia<br />
Decreased Appetite<br />
Agitation<br />
Chills<br />
Flushing<br />
Staring<br />
Lethargy<br />
Obsessive-Compulsive<br />
Disorder<br />
Nervous System Disorder<br />
Depression<br />
Hallucination<br />
Hyperhidrosis<br />
Weight Decreased<br />
Coma<br />
Fatigue<br />
Completed Suicide<br />
Urinary Hesitation<br />
Feeling Abnormal<br />
Anxiety<br />
Delusional Disorder,
Persecutory Type<br />
Amnesia<br />
Cognitive Disorder<br />
Musculoskeletal Stiffness<br />
Delirium<br />
Adjustment Disorder<br />
Polydipsia<br />
Hallucinations, Mixed<br />
03-Apr-2012 09:37 AM Page: 210
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mental Disorder<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer Lariam PS Hoffmann La Roche<br />
Inc ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Date:07/11/00ISR Number: 3527825-9Report Type:Expedited (15-DaCompany Report #235479 Age:76 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Corneal Disorder Health Lariam PS Hoffmann La Roche<br />
Corneal Oedema Professional Inc ORAL ORAL<br />
Date:07/12/00ISR Number: 3528437-3Report Type:Expedited (15-DaCompany Report #236649 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Fatigue Health Inc ORAL 1 DOSE FORM 1<br />
Decreased Appetite Professional PER WEEK ORAL<br />
Abdominal Pain<br />
Oesophageal Candidiasis<br />
Gastritis<br />
Date:07/14/00ISR Number: 3529989-XReport Type:Expedited (15-DaCompany Report #239896 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis E Foreign Lariam PS Hoffmann La Roche<br />
Hypertriglyceridaemia Health Inc ORAL 250 MG 1 PER<br />
Blood Immunoglobulin A Professional WEEK ORAL<br />
Increased<br />
Sortis (Atorvastatin<br />
Blood Immunoglobulin M Calcium) SS 30 MG DAILY<br />
Increased<br />
Havrix (Hepatitis A<br />
Serum Ferritin Increased Vaccine) C<br />
High Density Lipoprotein<br />
Omeprazole<br />
Decreased (Omeprazole) C<br />
Jaundice<br />
Hb-Vax-Dna<br />
Cholestasis<br />
(Hepatitis B Virus<br />
Blood Bilirubin Increased<br />
Vaccine Inactivated) C<br />
Liver Function Test<br />
Hepatitis B<br />
Abnormal<br />
Immunoglobulin<br />
Hypercholesterolaemia<br />
(Hepatitis B Immune<br />
Blood Immunoglobulin G Globulin) C<br />
Increased<br />
Beloc Zok Mite<br />
Monocytosis<br />
(Metoprolol<br />
Hepatitis Succinate) C<br />
Delix (Ramipril) C<br />
Ass (Aspirin)<br />
C
Date:07/20/00ISR Number: 3532363-3Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome<br />
Required<br />
Intervention to<br />
03-Apr-2012 09:37 AM Page: 211
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Dizziness Lariam 250mg Laroche PS Laroche ORAL 250MG WEEKLY<br />
Lethargy<br />
ORAL<br />
Disturbance In Attention Bi-Est C<br />
Mental Impairment Estriol/Estradiol C<br />
Nausea Progesterone C<br />
Emotional Distress<br />
Date:07/21/00ISR Number: 3534058-9Report Type:Expedited (15-DaCompany Report #240830 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Foreign Lariam PS Hoffmann La Roche<br />
Injury Other Inc ORAL ORAL<br />
Date:07/24/00ISR Number: 3533956-XReport Type:Direct Company Report # Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Mefloquin PS ORAL SMALL WEEKLY<br />
Initial or Prolonged Paranoia / LARGE<br />
ONE TIME /<br />
ORAL<br />
Deployment Vaccines<br />
For Us Army And Gulf<br />
War 90-91<br />
C<br />
Date:07/24/00ISR Number: 3533962-5Report Type:Direct Company Report # Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Mefloqine Lariam<br />
Initial or Prolonged Decreased Appetite Roche PS Roche ORAL 1 PILL WEEK<br />
Required Weight Decreased ORAL<br />
Intervention to<br />
Depression<br />
Prevent Permanent Anxiety<br />
Impairment/Damage<br />
Date:07/25/00ISR Number: 3534612-4Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia Lariam Mephloquine PS Roche Laboratories ORAL 1/10 DAYS<br />
Fatigue<br />
ORAL<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder
Headache<br />
Disturbance In Attention<br />
Anxiety<br />
Amnesia<br />
Communication Disorder<br />
Tinnitus<br />
Hearing Impaired<br />
03-Apr-2012 09:37 AM Page: 212
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/25/00ISR Number: 3535273-0Report Type:Expedited (15-DaCompany Report #213831 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Premature Baby Foreign Lariam PS Hoffmann La Roche<br />
Maternal Drugs Affecting Health Inc ORAL 250 MG 1 PER<br />
Foetus Professional WEEK ORAL<br />
Zovirax<br />
C<br />
Date:07/26/00ISR Number: 3535783-6Report Type:Expedited (15-DaCompany Report #241048 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Syndrome Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Csf White Blood Cell Other Inc ORAL 150 MG 1 PER<br />
Count Positive<br />
ONE DOSE ORAL<br />
Physical Examination<br />
Abnormal<br />
Coma<br />
Pyrexia<br />
Blood Pressure Diastolic<br />
Decreased<br />
Csf Protein Increased<br />
Bradycardia<br />
Confusional State<br />
Encephalitis<br />
Agitation<br />
Hallucination, Visual<br />
Musculoskeletal Stiffness<br />
Abnormal Behaviour<br />
Date:07/27/00ISR Number: 3536477-3Report Type:Expedited (15-DaCompany Report #219219 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Pregnancy Foreign Lariam PS Hoffmann La Roche<br />
Maternal Drugs Affecting Health Inc ORAL 250 MG 1 PER<br />
Foetus Professional WEEK ORAL<br />
Neonatal Disorder<br />
Cardiac Murmur<br />
Congenital Anomaly<br />
Date:07/28/00ISR Number: 3537250-2Report Type:Direct Company Report # Age:71 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Lariam PS<br />
Abnormal Dreams
Date:08/01/00ISR Number: 3539212-8Report Type:Expedited (15-DaCompany Report #240839 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypomania Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 250 MG 1 PER<br />
Disability Professional WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 213
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/01/00ISR Number: 3539316-XReport Type:Expedited (15-DaCompany Report #240830 Age:24 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Foreign Lariam PS Hoffmann La Roche<br />
Haemorrhage Other Inc ORAL ORAL<br />
Nightmare<br />
Injury<br />
Amnesia<br />
Date:08/01/00ISR Number: 3539356-0Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Health Lariam PS Hoffmann La Roche<br />
Headache Professional Inc ORAL 250 MG 1 PER<br />
Vertigo<br />
WEEK ORAL<br />
Dizziness<br />
Depression<br />
Panic Attack<br />
Date:08/01/00ISR Number: 3539359-6Report Type:Expedited (15-DaCompany Report #241539 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 250 MG 1 PER<br />
Professional WEEK ORAL 14 DAY<br />
Date:08/01/00ISR Number: 3539361-4Report Type:Expedited (15-DaCompany Report #241249 Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Foreign Lariam PS Hoffmann La Roche<br />
Social Avoidant Behaviour Health Inc ORAL ORAL<br />
Intentional Overdose Professional Paracetamol/Pseudoep<br />
Suicide Attempt<br />
hedrine<br />
(Acetaminophen/Pseud<br />
oephedrine<br />
Hydrochloride) SS<br />
Date:08/01/00ISR Number: 3539370-5Report Type:Expedited (15-DaCompany Report #241194 Age:3 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Congenital Foreign Lariam PS Hoffmann La Roche<br />
Maternal Drugs Affecting Health Inc ORAL ORAL 61 DAY<br />
Foetus<br />
Professional<br />
Psychomotor Retardation
Date:08/02/00ISR Number: 3539668-0Report Type:Direct Company Report # Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Medication Error Consumer Lariam PS Roche 250MG TABLET<br />
03-Apr-2012 09:37 AM Page: 214
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/07/00ISR Number: 3544183-4Report Type:Expedited (15-DaCompany Report #233963 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Lactate Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Dehydrogenase Increased Health Inc ORAL ORAL<br />
Leukopenia Professional Prodafalgan C<br />
Haptoglobin Decreased Potassium C<br />
Haemolytic Anaemia Quinimax C<br />
Overdose<br />
Balance Disorder<br />
Medication Error<br />
Thrombocytopenia<br />
Dizziness<br />
Hypokalaemia<br />
Date:08/07/00ISR Number: 3544187-1Report Type:Expedited (15-DaCompany Report #241950 Age:11 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Malaria Foreign Lariam PS Hoffmann La Roche<br />
Hospitalization - Malaise Health Inc ORAL 250 MG 1 PER<br />
Initial or Prolonged Anaphylactic Shock Professional WEEK ORAL<br />
Date:08/10/00ISR Number: 3547668-XReport Type:Expedited (15-DaCompany Report #232139 Age:33 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Astigmatism<br />
Staphylococcal Infection<br />
Hypoaesthesia<br />
Feeling Abnormal<br />
Vertigo<br />
Pyrexia<br />
Alanine Aminotransferase<br />
Increased<br />
Gastritis<br />
Visual Impairment<br />
Nystagmus<br />
Temperature Regulation<br />
Disorder<br />
Facial Nerve Disorder<br />
Tinnitus<br />
Malaise<br />
Palpitations<br />
Hearing Impaired<br />
Nervous System Disorder<br />
Arthritis<br />
Diarrhoea<br />
Conduction Disorder<br />
Binocular Eye Movement<br />
Disorder
Antibody Test Negative<br />
Neck Pain<br />
Gait Disturbance<br />
Hyporeflexia<br />
Csf Test Abnormal<br />
Balance Disorder<br />
Eye Movement Disorder<br />
Diplopia<br />
03-Apr-2012 09:37 AM Page: 215
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Folliculitis<br />
Eye Disorder<br />
Hyperreflexia Report Source Product Role Manufacturer Route Dose Duration<br />
Hypertension Foreign Lariam PS Hoffmann La Roche<br />
Paraesthesia Health Inc ORAL 250 MG 1 PER<br />
Optic Disc Disorder Professional 15 DAY ORAL<br />
Influenza Like Illness<br />
Hepatosplenomegaly<br />
Rash Pustular<br />
Date:08/11/00ISR Number: 3549527-5Report Type:Expedited (15-DaCompany Report #238191 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Foreign Lariam PS Hoffmann La Roche<br />
Fatigue Health Inc ORAL 1 DOSE FORM 1<br />
Syncope Professional PER WEEK ORAL<br />
Diarrhoea<br />
Vertigoheel<br />
Dizziness<br />
(Ambra/Cocculus/Coni<br />
Disturbance In Attention um/Petrolatum) C<br />
Balance Disorder<br />
Date:08/11/00ISR Number: 3549531-7Report Type:Expedited (15-DaCompany Report #241757 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disturbance In Attention Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Conjunctival Hyperaemia Consumer Inc ORAL ORAL<br />
Fatigue<br />
Other<br />
Sedation<br />
Eyelid Oedema<br />
Abdominal Pain<br />
Panic Attack<br />
Decreased Activity<br />
Visual Impairment<br />
Nervous System Disorder<br />
Date:08/11/00ISR Number: 3549536-6Report Type:Expedited (15-DaCompany Report #237672 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Balance Disorder Foreign Lariam PS Hoffmann La Roche<br />
Condition Aggravated Consumer Inc ORAL 250 MG 1 PER<br />
Cataract Other WEEK ORAL<br />
Visual Impairment<br />
Vision Blurred<br />
Confusional State<br />
Memory Impairment<br />
Alopecia<br />
Disturbance In Attention
Fatigue<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 216
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/15/00ISR Number: 3551061-3Report Type:Expedited (15-DaCompany Report #242221 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Retinal Haemorrhage Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL ORAL<br />
Professional<br />
Date:08/15/00ISR Number: 3551062-5Report Type:Expedited (15-DaCompany Report #238943 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoproteinaemia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Jaundice Health Inc ORAL 250 MG 1 PER<br />
Ascites Professional WEEK ORAL<br />
Blood Bilirubin Increased<br />
Date:08/16/00ISR Number: 3551060-1Report Type:Expedited (15-DaCompany Report #240839 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypomania Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 250 MG 1 PER<br />
Disability Professional WEEK ORAL<br />
Date:08/16/00ISR Number: 3551475-1Report Type:Expedited (15-DaCompany Report #242474 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Hydrocephalus Foreign Lariam PS Hoffmann La Roche<br />
Cerebellar Hypoplasia Health Inc ORAL ORAL 28 DAY<br />
Hearing Impaired<br />
Professional<br />
Visual Impairment<br />
Congenital Anomaly<br />
Convulsion<br />
Electroencephalogram<br />
Abnormal<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:08/17/00ISR Number: 3552270-XReport Type:Expedited (15-DaCompany Report #58978 Age:31 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Nausea<br />
Back Pain<br />
Fatigue<br />
Affective Disorder<br />
Stress<br />
Anger
Spinal Disorder<br />
Clonic Convulsion<br />
Amnesia<br />
Multiple Sclerosis<br />
Emotional Disorder<br />
Balance Disorder<br />
Nervous System Disorder<br />
Diarrhoea<br />
03-Apr-2012 09:37 AM Page: 217
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hypoaesthesia<br />
Ligament Sprain<br />
Asthenia Report Source Product Role Manufacturer Route Dose Duration<br />
Irritability Consumer Lariam PS Hoffmann La Roche<br />
Fasciitis Health Inc ORAL 250 MG 1 PER<br />
Infarction Professional WEEK ORAL<br />
Arthropathy<br />
Norflex<br />
Panic Attack<br />
(Orphenadrine<br />
Gait Disturbance Citrate) C<br />
Dyspnoea<br />
Vertigo<br />
Syncope<br />
Coordination Abnormal<br />
Nervousness<br />
Dermatitis<br />
Throat Tightness<br />
Road Traffic Accident<br />
Hearing Impaired<br />
Drug Hypersensitivity<br />
Spinal Fracture<br />
Derealisation<br />
Arthralgia<br />
Neck Pain<br />
Cerebellar Ataxia<br />
Anxiety<br />
Sinus Headache<br />
Dizziness<br />
Disturbance In Attention<br />
Feeling Abnormal<br />
Vestibular Disorder<br />
Sensation Of Pressure<br />
Disorientation<br />
Headache<br />
Opticokinetic Nystagmus<br />
Tests Abnormal<br />
Confusional State<br />
Oropharyngeal Pain<br />
Heart Rate Increased<br />
Pain<br />
Nystagmus<br />
Vomiting<br />
Dysphagia<br />
Date:08/17/00ISR Number: 3552322-4Report Type:Expedited (15-DaCompany Report #242515 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Vomiting Other Inc ORAL 250 MG 1 PER<br />
Hallucination<br />
1 WEEK ORAL<br />
Depression<br />
Sedation
Confusional State<br />
03-Apr-2012 09:37 AM Page: 218
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/18/00ISR Number: 3553097-5Report Type:Direct Company Report # Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaise Lariam 250mg Roche PS Roche ORAL 250MG ONCE<br />
Bipolar I Disorder<br />
PER ORAL<br />
Depression<br />
Mood Swings<br />
Anxiety<br />
Hallucination<br />
Lethargy<br />
Psychiatric Symptom<br />
Visual Acuity Reduced<br />
Hypoaesthesia<br />
Date:08/18/00ISR Number: 3553806-5Report Type:Expedited (15-DaCompany Report #242746 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depressed Level Of Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Consciousness Other Inc ORAL ORAL<br />
Encephalitis<br />
Stamaril Unidose<br />
Disorientation<br />
(Yellow Fever<br />
Lymphocytosis Vaccine) SS SUBCUTANEOUS 1 DOSE FORM 1<br />
PER DAY<br />
SUBCUTANEOUS<br />
Date:08/22/00ISR Number: 3554807-3Report Type:Direct Company Report # Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Panic Attack<br />
Lariam-<strong>Mefloquine</strong><br />
Insomnia<br />
Hcl-250mg Hoffman-La<br />
Depression Roche PS Hoffman-La Roche ORAL 1 TABLET WEEK<br />
Suicidal Ideation<br />
ORAL<br />
Paranoia<br />
Headache<br />
Anxiety<br />
Date:08/22/00ISR Number: 3555499-XReport Type:Expedited (15-DaCompany Report #215928 Age:35 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Jaundice Consumer Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Hepatic Necrosis Health Inc ORAL 250 MG 1 PER<br />
Liver Function Test Professional WEEK ORAL<br />
Abnormal Vitamin Nos SS<br />
Toxicity To Various<br />
Agents<br />
Spleen Disorder<br />
Autoimmune Disorder
Blood Bilirubin Increased<br />
03-Apr-2012 09:37 AM Page: 219
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/23/00ISR Number: 3556414-5Report Type:Expedited (15-DaCompany Report #242880 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Health Lariam PS Hoffmann La Roche<br />
Fear Professional Inc ORAL 1 DOSE FORM 1<br />
Alopecia Other PER WEEK ORAL<br />
Depression<br />
Crying<br />
Dizziness<br />
Nausea<br />
Toxicity To Various<br />
Agents<br />
Thinking Abnormal<br />
Panic Reaction<br />
Feeling Jittery<br />
Delusion<br />
Nightmare<br />
Abdominal Distension<br />
Diarrhoea<br />
Anxiety<br />
Date:08/24/00ISR Number: 3557763-7Report Type:Expedited (15-DaCompany Report #243099 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Consumer Lariam PS Hoffmann La Roche<br />
Other<br />
Inc<br />
Date:08/24/00ISR Number: 3557764-9Report Type:Expedited (15-DaCompany Report #243090 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nervousness Consumer Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Urinary Tract Infection Other Inc ORAL ORAL<br />
Headache<br />
Flushing<br />
Tremor<br />
Muscle Spasms<br />
Palpitations<br />
Fear<br />
Diarrhoea<br />
Phobia<br />
Myalgia<br />
Asthenia<br />
Weight Decreased<br />
Malaise<br />
Dizziness<br />
Date:08/25/00ISR Number: 3557765-0Report Type:Expedited (15-DaCompany Report #243085 Age:28 YR Gender:Male I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Panic Attack<br />
Circulatory Collapse<br />
Dizziness<br />
Disturbance In Attention<br />
Chest Pain<br />
Pyrexia<br />
03-Apr-2012 09:37 AM Page: 220
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Syncope<br />
Palpitations<br />
Anxiety Report Source Product Role Manufacturer Route Dose Duration<br />
Paraesthesia Consumer Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL<br />
Date:08/25/00ISR Number: 3559237-6Report Type:Expedited (15-DaCompany Report #243079 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Extrasystoles Foreign Lariam PS Hoffmann La Roche<br />
Maternal Drugs Affecting Health Inc 35 DAY<br />
Foetus<br />
Professional<br />
Polyhydramnios<br />
Pregnancy<br />
Date:08/25/00ISR Number: 3559244-3Report Type:Expedited (15-DaCompany Report #243061 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Stillbirth Consumer Lariam PS Hoffmann La Roche<br />
Accidental Exposure Other Inc<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:08/25/00ISR Number: 3559246-7Report Type:Expedited (15-DaCompany Report #243107 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pregnancy Consumer Lariam PS Hoffmann La Roche<br />
Intra-Uterine Death Other Inc ORAL ORAL<br />
Accidental Exposure<br />
Date:08/25/00ISR Number: 3559248-0Report Type:Expedited (15-DaCompany Report #243034 Age:80 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Sedation Foreign Lariam PS Hoffmann La Roche<br />
Life-Threatening Gastric Perforation Health Inc ORAL 2 DOSE FORM 1<br />
Nuclear Magnetic Professional PER ONE DOSE<br />
Resonance Imaging<br />
ORAL<br />
Abnormal<br />
Stupor<br />
Cardio-Respiratory Arrest<br />
Date:08/30/00ISR Number: 3561960-4Report Type:Expedited (15-DaCompany Report #237587 Age: Gender:Unknown I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Foreign Lariam PS Hoffmann La Roche<br />
Accidental Exposure Health Inc ORAL ORAL<br />
Professional<br />
Other<br />
03-Apr-2012 09:37 AM Page: 221
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/30/00ISR Number: 3561965-3Report Type:Expedited (15-DaCompany Report #243061 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Consumer Lariam PS Hoffmann La Roche<br />
Stillbirth Other Inc<br />
Pregnancy<br />
Accidental Exposure<br />
Date:08/30/00ISR Number: 3561967-7Report Type:Expedited (15-DaCompany Report #243107 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Accidental Exposure Consumer Lariam PS Hoffmann La Roche<br />
Intra-Uterine Death Other Inc ORAL ORAL<br />
Date:08/30/00ISR Number: 3561969-0Report Type:Expedited (15-DaCompany Report #224923 Age:22 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicide Attempt Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Abnormal Behaviour Consumer Inc ORAL 1.5 DOSE FORM<br />
Hallucination Health 1 PER WEEK<br />
Acute Psychosis Professional ORAL 14 DAY<br />
Other<br />
Date:08/30/00ISR Number: 3561972-0Report Type:Expedited (15-DaCompany Report #243661 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Dizziness Consumer Inc ORAL ORAL<br />
Fatigue Other Chloroquine Hcl<br />
Dissociation<br />
(Chloroquine<br />
Gait Disturbance<br />
Hydrochloride<br />
Eating Disorder (Injection)) C<br />
Anxiety<br />
Vomiting<br />
Speech Disorder<br />
Panic Attack<br />
Hallucination<br />
Balance Disorder<br />
Dyspnoea<br />
Date:08/30/00ISR Number: 3562000-3Report Type:Expedited (15-DaCompany Report #243398 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Mental Disorder Health Inc ORAL 7 DOSE FORM 1
Abnormal Behaviour Professional PER ONE DOSE<br />
Nervous System Disorder<br />
ORAL<br />
Hemiparesis<br />
Cerebral Venous<br />
Thrombosis<br />
Malaria<br />
03-Apr-2012 09:37 AM Page: 222
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/30/00ISR Number: 3562002-7Report Type:Expedited (15-DaCompany Report #243587 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Bipolar Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Mania Consumer Inc ORAL ORAL<br />
Other<br />
Date:08/30/00ISR Number: 3562005-2Report Type:Expedited (15-DaCompany Report #237590 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Foreign Lariam PS Hoffmann La Roche<br />
Pregnancy Health Inc ORAL ORAL<br />
Accidental Exposure<br />
Professional<br />
Other<br />
Date:08/31/00ISR Number: 3562208-7Report Type:Direct Company Report # Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia Lariam PS ORAL 1 PER WEEK<br />
Heart Rate Increased<br />
ORAL<br />
Nausea<br />
Abdominal Pain<br />
Date:09/01/00ISR Number: 3562944-2Report Type:Direct Company Report # Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Abnormal Dreams Larium PS<br />
Anxiety<br />
Paraesthesia<br />
Flushing<br />
Insomnia<br />
Chills<br />
Date:09/01/00ISR Number: 3564122-XReport Type:Expedited (15-DaCompany Report #243633 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dysgraphia Foreign Lariam PS Hoffmann La Roche<br />
Migraine Consumer Inc ORAL ORAL<br />
Reading Disorder<br />
Other<br />
Visual Acuity Reduced<br />
Disturbance In Attention<br />
Date:09/01/00ISR Number: 3564123-1Report Type:Expedited (15-DaCompany Report #243603 Age: Gender:Female I/FU:I
Outcome<br />
Other Serious<br />
PT<br />
Vomiting<br />
Hepatitis<br />
Headache<br />
Nausea<br />
Disorientation<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 223
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sedation<br />
Hyperhidrosis<br />
Hepatocellular Injury Report Source Product Role Manufacturer Route Dose Duration<br />
Weight Decreased Consumer Lariam PS Hoffmann La Roche<br />
Haematemesis Other Inc ORAL ORAL<br />
Nightmare<br />
Tremor<br />
Date:09/01/00ISR Number: 3564124-3Report Type:Expedited (15-DaCompany Report #243535 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Bradycardia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Paraesthesia Other Inc ORAL 250 MG 1 PER<br />
Chest Pain<br />
WEEK ORAL<br />
Date:09/06/00ISR Number: 3565002-6Report Type:Expedited (15-DaCompany Report #242880 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thinking Abnormal Lariam Tablets PS Roche 15 DAY<br />
Feeling Jittery<br />
Diarrhoea<br />
Alopecia<br />
Abdominal Pain<br />
Crying<br />
Dizziness<br />
Delusion<br />
Malaise<br />
Depression<br />
Nightmare<br />
Nausea<br />
Anxiety<br />
Fear<br />
Amnesia<br />
Panic Reaction<br />
Abdominal Distension<br />
Date:09/06/00ISR Number: 3565005-1Report Type:Expedited (15-DaCompany Report #243099 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Lariam Tablets PS Roche<br />
Personality Change<br />
Date:09/06/00ISR Number: 3565007-5Report Type:Expedited (15-DaCompany Report #243090 Age: Gender:Female I/FU:F<br />
Outcome<br />
PT
Hospitalization -<br />
Initial or Prolonged<br />
Asthenia<br />
Flushing<br />
Muscle Spasms<br />
Malaise<br />
Diarrhoea<br />
Weight Decreased<br />
Fear<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 224
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Nervousness<br />
Urinary Tract Infection<br />
Tremor Report Source Product Role Manufacturer Route Dose Duration<br />
Headache Lariam Tablets PS Roche<br />
Palpitations<br />
Myalgia<br />
Phobia<br />
Date:09/06/00ISR Number: 3565032-4Report Type:Expedited (15-DaCompany Report #243085 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Syncope Lariam Tablets PS Roche<br />
Initial or Prolonged Chest Pain<br />
Palpitations<br />
Paraesthesia<br />
Panic Attack<br />
Tension<br />
Dizziness<br />
Disturbance In Attention<br />
Circulatory Collapse<br />
Anxiety<br />
Date:09/06/00ISR Number: 3565049-XReport Type:Expedited (15-DaCompany Report #244099 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Neuralgia Lariam Tablets PS Roche<br />
Pruritus Zoloft C<br />
Urticaria Neurontin C<br />
Muscular Weakness Vioxx C 123 DAY<br />
Balance Disorder Provera C FOR 12 DAYS<br />
Fatigue<br />
BETWEEN JUL<br />
Haematoma<br />
1999 AND JULY<br />
Cartilage Injury 2000 AT 2.5<br />
Ecchymosis<br />
MG.<br />
CURRENTLY<br />
Muscle Disorder Synthroid C<br />
Dizziness Premarin C<br />
Date:09/07/00ISR Number: 3565856-3Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Lariam Tablets PS Roche ESTIMATED 5<br />
Initial or Prolonged Anxiety DOSES IN<br />
Disability Panic Attack TOTAL. 21 DAY<br />
Headache<br />
Vertigo<br />
Dizziness
Date:09/07/00ISR Number: 3565886-1Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Headache<br />
Panic Attack<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 225
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Vertigo<br />
Dizziness<br />
Anxiety Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche ESTIMATED 5<br />
DOSES IN<br />
TOTAL. 21 DAY<br />
Date:09/07/00ISR Number: 3565888-5Report Type:Expedited (15-DaCompany Report #243398 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Personality Change Due To Lariam Tablets PS Roche<br />
Initial or Prolonged A General Medical<br />
Condition<br />
Gastroenteritis<br />
Nervous System Disorder<br />
Diarrhoea<br />
Encephalitis<br />
Faecal Incontinence<br />
Fatigue<br />
Extensor Plantar Response<br />
Psychomotor Retardation<br />
Confusional State<br />
Vomiting<br />
Urinary Incontinence<br />
Hemiparesis<br />
Mental Disorder<br />
Date:09/07/00ISR Number: 3565942-8Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Lariam Tablets PS Roche ESTIMATED 5<br />
Initial or Prolonged Headache DOSES IN<br />
Disability Dizziness TOTAL. 21 DAY<br />
Anxiety<br />
Depression<br />
Vertigo<br />
Date:09/07/00ISR Number: 3565949-0Report Type:Expedited (15-DaCompany Report #243398 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Vomiting<br />
Faecal Incontinence<br />
Urinary Incontinence<br />
Encephalitis<br />
Gastroenteritis<br />
Diarrhoea<br />
Personality Change Due To
A General Medical<br />
Condition<br />
Hemiparesis<br />
Mental Disorder<br />
Fatigue<br />
Confusional State<br />
Nervous System Disorder<br />
Psychomotor Retardation<br />
03-Apr-2012 09:37 AM Page: 226
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Extensor Plantar Response<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche<br />
Date:09/07/00ISR Number: 3566017-4Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam Tablets PS Roche ESTIMATED 5<br />
Initial or Prolonged Headache DOSES IN<br />
Disability Vertigo TOTAL. 21 DAY<br />
Depression<br />
Panic Attack<br />
Date:09/07/00ISR Number: 3565858-7Report Type:Expedited (15-DaCompany Report #243398 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Oliguria Lariam Tablets PS Roche<br />
Initial or Prolonged Nervous System Disorder<br />
Fatigue<br />
Hemiparesis<br />
Gastroenteritis<br />
Encephalitis Viral<br />
Extensor Plantar Response<br />
Overdose<br />
Personality Change Due To<br />
A General Medical<br />
Condition<br />
Diarrhoea<br />
Cerebral Venous<br />
Thrombosis<br />
Confusional State<br />
Toxicity To Various<br />
Agents<br />
Vomiting<br />
Mental Disorder<br />
Date:09/08/00ISR Number: 3566999-0Report Type:Expedited (15-DaCompany Report #72278 Age:24 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Visual Impairment<br />
Abdominal Pain<br />
Skin Irritation<br />
Hearing Impaired<br />
Pain<br />
Full Blood Count Abnormal<br />
Syncope
Campbell De Morgan Spots<br />
Deafness<br />
Neutropenia<br />
Lymphadenopathy<br />
Vision Blurred<br />
Diarrhoea<br />
Eosinophil Count<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 227
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Skin Papilloma<br />
Renal Colic<br />
Pco2 Abnormal<br />
Muscle Spasms<br />
Dizziness<br />
Cough<br />
Neck Pain<br />
Bone Marrow Failure<br />
Coordination Abnormal<br />
Migraine<br />
Depersonalisation<br />
Nausea<br />
Chills<br />
Asthenia<br />
Cyst<br />
Oropharyngeal Pain<br />
Amnesia<br />
Biopsy Bone Marrow<br />
Abnormal<br />
Lipids Abnormal<br />
Blood Potassium Decreased<br />
Arthralgia<br />
Abscess<br />
Basophilia<br />
Penile Ulceration<br />
Hyperhidrosis<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Abdominal Tenderness<br />
Pharyngitis<br />
Bladder Pain<br />
Dissociation<br />
Liver Function Test<br />
Abnormal<br />
Amoebiasis<br />
Haemorrhoids<br />
Hypokalaemia<br />
Paraesthesia<br />
Haematocrit Increased<br />
Asthenopia<br />
Vertigo<br />
Blood Heavy Metal<br />
Increased<br />
Herpes Simplex<br />
Dysphagia<br />
Rash Pustular<br />
Drug Level Below<br />
Therapeutic<br />
Hyperglycaemia<br />
Hypersensitivity<br />
Hypoglycaemia<br />
Fatigue<br />
Confusional State
Hypersomnia<br />
Pain In Extremity<br />
Swelling<br />
Red Blood Cell Count<br />
Increased<br />
Disorientation<br />
Tinnitus<br />
Tremor<br />
03-Apr-2012 09:37 AM Page: 228
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dyspnoea<br />
Ear Pain<br />
Schistosomiasis Report Source Product Role Manufacturer Route Dose Duration<br />
Hepatic Pain Lariam Tablets PS Roche 35 DAY<br />
Malaria<br />
Thinking Abnormal<br />
Back Pain<br />
Feeling Abnormal<br />
Bradycardia<br />
Palpitations<br />
Blood Phosphorus<br />
Decreased<br />
Vestibular Disorder<br />
Pyrexia<br />
Flatulence<br />
Malaise<br />
Blood Urea Increased<br />
Muscular Weakness<br />
Balance Disorder<br />
Gastrointestinal Disorder<br />
Anxiety<br />
Blood Triglycerides<br />
Increased<br />
Headache<br />
Date:09/08/00ISR Number: 3567004-2Report Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Skin Discolouration Lariam Tablets PS Roche 50 DAY<br />
Skin Infection<br />
Scar<br />
Agitation<br />
Emotional Disorder<br />
Skin Necrosis<br />
Lichen Planus<br />
Fear<br />
Psychosexual Disorder<br />
Date:09/08/00ISR Number: 3568264-4Report Type:Expedited (15-DaCompany Report #244099 Age:48 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Haematoma<br />
Myalgia<br />
Muscle Disorder<br />
Muscle Spasms<br />
Dizziness<br />
Asthenia<br />
Ecchymosis<br />
Pruritus
Urticaria<br />
Depression<br />
Ammonia Increased<br />
Balance Disorder<br />
Cartilage Injury<br />
Muscular Weakness<br />
Paraesthesia<br />
Asthenopia<br />
03-Apr-2012 09:37 AM Page: 229
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fatigue<br />
Neuralgia<br />
Burning Sensation Report Source Product Role Manufacturer Route Dose Duration<br />
Tetany Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Zoloft<br />
C<br />
Vioxx<br />
C<br />
Premarin<br />
C<br />
Provera<br />
C<br />
Synthroid<br />
C<br />
Neurontin<br />
C<br />
Date:09/08/00ISR Number: 3568266-8Report Type:Expedited (15-DaCompany Report #242880 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Health Lariam PS Hoffmann La Roche<br />
Toxicity To Various Professional Inc ORAL 1 DOSE FORM 1<br />
Agents Other PER WEEK ORAL<br />
Feeling Jittery<br />
Anxiety<br />
Thinking Abnormal<br />
Depression<br />
Nightmare<br />
Amnesia<br />
Nausea<br />
Dizziness<br />
Abdominal Distension<br />
Phobia<br />
Panic Reaction<br />
Pain<br />
Diarrhoea<br />
Alopecia<br />
Abdominal Pain<br />
Date:09/08/00ISR Number: 3568316-9Report Type:Expedited (15-DaCompany Report #72278 Age:24 YR Gender:Unknown I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Coordination Abnormal<br />
Malaria<br />
Vision Blurred<br />
Basophil Count Increased<br />
Blood Potassium Decreased<br />
Liver Function Test<br />
Abnormal<br />
Blood Heavy Metal<br />
Increased<br />
Dyspnoea<br />
Hearing Impaired<br />
Asthenopia
Nausea<br />
Muscular Weakness<br />
Anxiety<br />
Palpitations<br />
Lipids Abnormal<br />
Diarrhoea<br />
Tinnitus<br />
Amoebiasis<br />
03-Apr-2012 09:37 AM Page: 230
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Syncope<br />
Penile Ulceration<br />
Depersonalisation<br />
Malaise<br />
Visual Impairment<br />
Feeling Abnormal<br />
Bradycardia<br />
Antibody Test Positive<br />
Amnesia<br />
Skin Irritation<br />
Paraesthesia<br />
Hypersensitivity<br />
Vestibular Disorder<br />
Vertigo<br />
Disorientation<br />
Schistosomiasis<br />
Abscess<br />
Photopsia<br />
Hyperhidrosis<br />
Fatigue<br />
Abdominal Pain<br />
Balance Disorder<br />
Confusional State<br />
Oropharyngeal Pain<br />
Blood Triglycerides<br />
Increased<br />
Swelling<br />
Eye Disorder<br />
Pharyngitis<br />
Muscle Spasms<br />
Dysphagia<br />
Pyrexia<br />
Rash Pustular<br />
Hypoglycaemia<br />
Low Density Lipoprotein<br />
Increased<br />
Migraine<br />
Drug Level Below<br />
Therapeutic<br />
Pain In Extremity<br />
Lymphocyte Count<br />
Increased<br />
Bladder Pain<br />
Pain<br />
Tremor<br />
Ear Pain<br />
Flatulence<br />
Headache<br />
Lymphadenopathy<br />
Asthenia<br />
Hypersomnia<br />
Biopsy Bone Marrow<br />
Abnormal
Neutrophil Count<br />
Decreased<br />
Blood Phosphorus<br />
Decreased<br />
Blood Cholesterol<br />
Increased<br />
Full Blood Count Abnormal<br />
Neck Pain<br />
03-Apr-2012 09:37 AM Page: 231
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Campbell De Morgan Spots<br />
Thinking Abnormal<br />
Back Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Chills Consumer Lariam PS Hoffmann La Roche<br />
Cyst Inc ORAL 250 MG 1 PER<br />
Gastrointestinal Disorder WEEK ORAL 35 DAY<br />
White Blood Cell Count<br />
Decreased<br />
Monocyte Count Increased<br />
Herpes Simplex<br />
Arthralgia<br />
Dizziness<br />
Cough<br />
Date:09/08/00ISR Number: 3568345-5Report Type:Expedited (15-DaCompany Report #230098 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Crying Consumer Inc ORAL 250 MG ORAL<br />
Conversion Disorder Health Hismanal C<br />
Hypertension Professional Jodthyrox C<br />
Furuncle<br />
Psychotic Disorder<br />
Dizziness<br />
Fatigue<br />
Abdominal Discomfort<br />
Depression<br />
Agoraphobia<br />
Retching<br />
Tinnitus<br />
Hyperventilation<br />
Hyperhidrosis<br />
Nausea<br />
Paraesthesia<br />
Mental Disorder<br />
Blood Pressure Systolic<br />
Increased<br />
Chills<br />
Date:09/08/00ISR Number: 3568346-7Report Type:Expedited (15-DaCompany Report #219219 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Neonatal Disorder Foreign Lariam PS Hoffmann La Roche<br />
Cardiac Murmur Health Inc ORAL 250 MG 1 PER<br />
Maternal Drugs Affecting Professional WEEK ORAL<br />
Foetus<br />
Congenital Anomaly
Date:09/08/00ISR Number: 3568522-3Report Type:Expedited (15-DaCompany Report #243099 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Foreign Lariam PS Hoffmann La Roche<br />
Consumer<br />
Inc<br />
Other<br />
03-Apr-2012 09:37 AM Page: 232
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/08/00ISR Number: 3568523-5Report Type:Expedited (15-DaCompany Report #243085 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disturbance In Attention Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Paraesthesia Consumer Inc ORAL ORAL<br />
Dizziness<br />
Other<br />
Anxiety<br />
Circulatory Collapse<br />
Panic Attack<br />
Syncope<br />
Chest Pain<br />
Anhedonia<br />
Pyrexia<br />
Malaise<br />
Palpitations<br />
Date:09/08/00ISR Number: 3568524-7Report Type:Expedited (15-DaCompany Report #243090 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myalgia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Dizziness Consumer Inc ORAL ORAL<br />
Phobia<br />
Other<br />
Malaise<br />
Weight Decreased<br />
Tremor<br />
Headache<br />
Flushing<br />
Diarrhoea<br />
Urinary Tract Infection<br />
Muscle Spasms<br />
Asthenia<br />
Palpitations<br />
Date:09/08/00ISR Number: 3569032-XReport Type:Expedited (15-DaCompany Report #234391 Age:56 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia Consumer Lariam PS Hoffmann La Roche<br />
Emotional Distress Inc ORAL 250 MG 2 PER<br />
Overdose DAY 42 DAY<br />
Nausea<br />
Loss Of Control Of Legs<br />
Medication Error<br />
Blindness Transient<br />
Brain Injury<br />
Decreased Activity<br />
Date:09/08/00ISR Number: 3567013-3Report Type:Expedited (15-DaCompany Report #244150 Age:11 YR Gender:Female I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Tachycardia<br />
Malaise<br />
Dizziness<br />
Fatigue<br />
Chills<br />
Loss Of Consciousness<br />
03-Apr-2012 09:37 AM Page: 233
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hypotension<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche 15 DAY<br />
Date:09/08/00ISR Number: 3567021-2Report Type:Expedited (15-DaCompany Report #244148 Age: Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Lariam Tablets PS Roche 34 DAY<br />
Foetus<br />
Talipes<br />
Klinefelter'S Syndrome<br />
Pregnancy<br />
Abortion Induced<br />
Date:09/11/00ISR Number: 3568894-XReport Type:Direct Company Report # Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Consumer Lariam 250 Mg Tablet<br />
Paraesthesia Roche PS Roche ORAL 1 TABLET ONCE<br />
Burning Sensation<br />
A WEEK ORAL<br />
Headache<br />
Panic Attack<br />
Crying<br />
Melaena<br />
Depression<br />
Lethargy<br />
Dizziness<br />
Anxiety<br />
Disturbance In Attention<br />
Diarrhoea<br />
Date:09/11/00ISR Number: 3569224-XReport Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Emotional Disorder Other Lariam PS Hoffmann La Roche<br />
Fear Inc ORAL 250 MG 1 PER<br />
Injury<br />
WEEK ORAL<br />
Agitation<br />
Lichen Planus<br />
Infection<br />
Skin Discolouration<br />
Date:09/11/00ISR Number: 3569299-8Report Type:Expedited (15-DaCompany Report #244150 Age:11 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Fatigue Other Inc ORAL 250 MG 1PER<br />
Tachycardia<br />
WEEK ORAL<br />
Hypotension<br />
Malaise<br />
Chills<br />
Loss Of Consciousness<br />
03-Apr-2012 09:37 AM Page: 234
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/11/00ISR Number: 3569301-3Report Type:Expedited (15-DaCompany Report #244148 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Talipes Foreign Lariam PS Hoffmann La Roche<br />
Maternal Drugs Affecting Other Inc ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Klinefelter'S Syndrome<br />
Abortion Induced<br />
Pregnancy<br />
Date:09/11/00ISR Number: 3569673-XReport Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Skin Discolouration Other Lariam PS Hoffmann La Roche<br />
Injury Inc ORAL 250 MG 1 PER<br />
Agitation<br />
WEEK ORAL<br />
Infection<br />
Fear<br />
Emotional Disorder<br />
Lichen Planus<br />
Date:09/11/00ISR Number: 3570099-3Report Type:Expedited (15-DaCompany Report #72278 Age:24 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Coordination Abnormal<br />
Lymphadenopathy<br />
Asthenia<br />
Gastrointestinal Disorder<br />
Lipids Abnormal<br />
Diarrhoea<br />
Swelling<br />
Neck Pain<br />
Hypouricaemia<br />
Lymphadenitis<br />
Neutrophil Count<br />
Decreased<br />
Hypercholesterolaemia<br />
Thinking Abnormal<br />
Back Pain<br />
Malaise<br />
Balance Disorder<br />
Anxiety<br />
Blood Phosphorus<br />
Decreased<br />
Disorientation<br />
Tinnitus<br />
Ear Pain<br />
Headache<br />
Rash Pustular
Ear Discomfort<br />
Skin Papilloma<br />
Gait Disturbance<br />
Penile Ulceration<br />
Malaria<br />
Confusional State<br />
Vertigo<br />
Schistosomiasis<br />
03-Apr-2012 09:37 AM Page: 235
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Feeling Drunk<br />
Haematocrit Increased<br />
Abdominal Tenderness<br />
Haemoglobin Increased<br />
Arthralgia<br />
Vision Blurred<br />
Hypersomnia<br />
Palpitations<br />
Muscle Spasms<br />
Haemorrhoids<br />
Cough<br />
Pyrexia<br />
Campbell De Morgan Spots<br />
Bone Marrow Failure<br />
Nasopharyngitis<br />
Hyperglycaemia<br />
Depersonalisation<br />
Migraine<br />
Drug Ineffective<br />
Oropharyngeal Pain<br />
Skin Irritation<br />
Paraesthesia<br />
Hypersensitivity<br />
Asthenopia<br />
Full Blood Count Abnormal<br />
Liver Function Test<br />
Abnormal<br />
Dizziness<br />
Hypokalaemia<br />
Syncope<br />
Abscess<br />
Toxicity To Various<br />
Agents<br />
Eosinophil Count<br />
Increased<br />
Enthesopathy<br />
Amoebiasis<br />
Nausea<br />
Chills<br />
Muscular Weakness<br />
Hearing Impaired<br />
Tenderness<br />
Gastrointestinal Pain<br />
Chronic Fatigue Syndrome<br />
Hypoglycaemia<br />
Herpes Simplex<br />
Vestibular Disorder<br />
Bladder Pain<br />
Tremor<br />
Dysphagia<br />
Dyspnoea<br />
Flatulence<br />
Spleen Disorder
High Density Lipoprotein<br />
Increased<br />
Viral Infection<br />
Condition Aggravated<br />
Monocyte Count Increased<br />
Blood Heavy Metal<br />
Increased<br />
Hyperhidrosis<br />
03-Apr-2012 09:37 AM Page: 236
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fatigue<br />
Visual Impairment<br />
Pharyngitis Streptococcal Report Source Product Role Manufacturer Route Dose Duration<br />
Feeling Abnormal Consumer Lariam PS Hoffmann La Roche<br />
Abdominal Pain Health Inc ORAL 250 MG 1 PER<br />
Bradycardia Professional WEEK ORAL 35 DAY<br />
Cyst<br />
Other<br />
Amnesia<br />
Pain In Extremity<br />
Pain<br />
Red Blood Cell Count<br />
Increased<br />
Nystagmus<br />
Date:09/12/00ISR Number: 3568900-2Report Type:Expedited (15-DaCompany Report #244099 Age:48 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam Tablets PS Roche<br />
Haematoma Zoloft C<br />
Urticaria Vioxx C 123 DAY<br />
Ecchymosis Premarin C<br />
Paraesthesia Provera C FOR 12 DAYS<br />
Myalgia<br />
BETWEEN JUL<br />
Muscular Weakness<br />
1999 AND JULY<br />
Asthenia 2000 AT 2.5<br />
Balance Disorder<br />
MG.<br />
CURRENTLY<br />
Dizziness Synthroid C<br />
Cartilage Injury Neurontin C<br />
Burning Sensation<br />
Fatigue<br />
Tetany<br />
Pruritus<br />
Muscle Disorder<br />
Muscle Spasms<br />
Date:09/12/00ISR Number: 3568893-8Report Type:Expedited (15-DaCompany Report #241248 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Barotrauma Lariam Tablets PS Roche<br />
Initial or Prolonged<br />
Date:09/13/00ISR Number: 3569968-XReport Type:Expedited (15-DaCompany Report #241757 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Eyelid Oedema Lariam Tablets PS Roche 27 DAY<br />
Initial or Prolonged Panic Attack
Abdominal Pain<br />
Conjunctival Hyperaemia<br />
Visual Impairment<br />
Disturbance In Attention<br />
Fatigue<br />
Sedation<br />
03-Apr-2012 09:37 AM Page: 237
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/13/00ISR Number: 3571074-5Report Type:Expedited (15-DaCompany Report #243398 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Mental Disorder Health Inc ORAL 7 DOSE FORM 1<br />
Urinary Incontinence Professional PER ONE DOSE<br />
Personality Change Due To<br />
ORAL<br />
A General Medical<br />
Condition<br />
Faecal Incontinence<br />
Toxicity To Various<br />
Agents<br />
Vomiting<br />
Hemiparesis<br />
Psychomotor Retardation<br />
Confusional State<br />
Nervous System Disorder<br />
Diarrhoea<br />
Gastroenteritis<br />
Overdose<br />
Encephalitis<br />
Date:09/13/00ISR Number: 3571321-XReport Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Health Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Depression Professional Inc ORAL 250 MG 1 PER<br />
Disability Headache WEEK PO<br />
Anxiety<br />
Panic Attack<br />
Date:09/14/00ISR Number: 3571509-8Report Type:Expedited (15-DaCompany Report #241248 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Barotrauma Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL ORAL<br />
Professional<br />
Date:09/14/00ISR Number: 3571842-XReport Type:Expedited (15-DaCompany Report #244099 Age:48 YR Gender:Female I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Muscular Weakness<br />
Balance Disorder<br />
Burning Sensation<br />
Urticaria<br />
Tetany<br />
Cartilage Injury
Depression<br />
Ecchymosis<br />
Myalgia<br />
Haematoma<br />
Injury<br />
Neuralgia<br />
Paraesthesia<br />
Ammonia Increased<br />
03-Apr-2012 09:37 AM Page: 238
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pruritus<br />
Asthenia<br />
Muscle Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Asthenopia Health Lariam PS Hoffmann La Roche<br />
Muscle Spasms Professional Inc ORAL ORAL<br />
Fatigue Zoloft C<br />
Dizziness Vioxx C<br />
Premarin<br />
C<br />
Provera<br />
(Medroxyprogesterone<br />
) C<br />
Synthroid<br />
C<br />
Neurontin<br />
C<br />
Date:09/14/00ISR Number: 3570925-8Report Type:Expedited (15-DaCompany Report #220387 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Induced Labour Lariam Tablets PS Roche 58 DAY<br />
Postmature Baby Logynon C<br />
Pregnancy<br />
Abnormal Labour<br />
Date:09/14/00ISR Number: 3570928-3Report Type:Expedited (15-DaCompany Report #244551 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aplastic Anaemia Lariam Tablets PS Roche 29 DAY<br />
Fatigue<br />
Date:09/15/00ISR Number: 3572844-XReport Type:Expedited (15-DaCompany Report #241757 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Conjunctival Hyperaemia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Eyelid Oedema Consumer Inc ORAL ORAL<br />
Visual Impairment<br />
Health<br />
Disturbance In Attention Professional<br />
Fatigue<br />
Sedation<br />
Abdominal Pain<br />
Panic Attack<br />
Date:09/15/00ISR Number: 3571353-1Report Type:Expedited (15-DaCompany Report #244484 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Lariam Tablets PS Roche 71 DAY<br />
Viith Nerve Paralysis
Date:09/18/00ISR Number: 3572388-5Report Type:Expedited (15-DaCompany Report #244551 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Lariam Tablets PS Roche 29 DAY<br />
Aplastic Anaemia<br />
03-Apr-2012 09:37 AM Page: 239
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/18/00ISR Number: 3574528-0Report Type:Expedited (15-DaCompany Report #220387 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Caesarean Section Foreign Lariam PS Hoffmann La Roche<br />
Pregnancy Health Inc ORAL 250 MG 1 PER<br />
Abnormal Labour Professional WEEK ORAL<br />
Logynon (Ethinyl<br />
Estradiol /<br />
Levonorgestrel) C<br />
Date:09/19/00ISR Number: 3573038-4Report Type:Expedited (15-DaCompany Report #235479 Age:76 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Corneal Oedema Lariam Tablets PS Roche<br />
Corneal Disorder<br />
Vision Blurred<br />
Date:09/19/00ISR Number: 3573039-6Report Type:Expedited (15-DaCompany Report #242474 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Hydrocephalus Lariam Tablets PS Roche 28 DAY<br />
Maternal Drugs Affecting<br />
Foetus<br />
Congenital Anomaly<br />
Cerebellar Hypoplasia<br />
Pregnancy<br />
Ultrasound Antenatal<br />
Screen Abnormal<br />
Convulsion<br />
Visual Impairment<br />
Hearing Impaired<br />
Date:09/19/00ISR Number: 3573049-9Report Type:Expedited (15-DaCompany Report #243079 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bradycardia Lariam Tablets PS Roche 35 DAY<br />
Arrhythmia Neonatal<br />
Palpitations<br />
Maternal Drugs Affecting<br />
Foetus<br />
Polyhydramnios<br />
Extrasystoles<br />
Neonatal Disorder<br />
Date:09/19/00ISR Number: 3573350-9Report Type:Direct Company Report # Age:32 YR Gender:Female I/FU:I
Outcome<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT<br />
Nausea<br />
Anxiety<br />
Fear<br />
Diarrhoea<br />
Dissociation<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 240
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Headache<br />
Asthenia<br />
Paraesthesia Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination Larium PS<br />
Balance Disorder<br />
Somnambulism<br />
Abnormal Dreams<br />
Depression<br />
Date:09/19/00ISR Number: 3574500-0Report Type:Expedited (15-DaCompany Report #244484 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Tinnitus Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Viith Nerve Paralysis Health Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Professional WEEK ORAL<br />
Impairment/Damage<br />
Date:09/19/00ISR Number: 3575054-5Report Type:Expedited (15-DaCompany Report #235479 Age:76 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Corneal Oedema Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Date:09/19/00ISR Number: 3575202-7Report Type:Expedited (15-DaCompany Report #245061 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Physical Assault Foreign Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL<br />
Date:09/19/00ISR Number: 3575223-4Report Type:Expedited (15-DaCompany Report #244870 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Foreign Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL<br />
Tetavax (Adsorbed<br />
Tetanus Vaccine) C<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
Typhim Iv (Typhoid<br />
Vaccines)<br />
C<br />
Date:09/19/00ISR Number: 3575225-8Report Type:Expedited (15-DaCompany Report #244736 Age:30 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Idiopathic Foreign Lariam PS Hoffmann La Roche<br />
Thrombocytopenic Purpura Health Inc ORAL ORAL<br />
Purpura<br />
Professional<br />
03-Apr-2012 09:37 AM Page: 241
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/19/00ISR Number: 3575226-XReport Type:Expedited (15-DaCompany Report #243079 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Palpitations Foreign Lariam PS Hoffmann La Roche<br />
Polyhydramnios Health Inc ORAL 250 MG 1 PER<br />
Extrasystoles Professional WEEK ORAL 35 DAY<br />
Pregnancy<br />
Bradycardia<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:09/19/00ISR Number: 3575228-3Report Type:Expedited (15-DaCompany Report #242474 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Hearing Impaired Foreign Lariam PS Hoffmann La Roche<br />
Congenital Hydrocephalus Health Inc ORAL ORAL 28 DAY<br />
Congenital Anomaly<br />
Professional<br />
Vision Abnormal Neonatal<br />
Convulsion Neonatal<br />
Electroencephalogram<br />
Abnormal<br />
Apgar Score Low<br />
Developmental Delay<br />
Maternal Drugs Affecting<br />
Foetus<br />
Cerebellar Hypoplasia<br />
Date:09/19/00ISR Number: 3573037-2Report Type:Expedited (15-DaCompany Report #244736 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Idiopathic Lariam Tablets PS Roche<br />
Thrombocytopenic Purpura<br />
Date:09/19/00ISR Number: 3573045-1Report Type:Expedited (15-DaCompany Report #244870 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Lariam Tablets PS Roche<br />
Havrix C 1 DAY<br />
Typhim Vi C 1 DAY<br />
Tetavax C 1 DAY<br />
Date:09/19/00ISR Number: 3573048-7Report Type:Expedited (15-DaCompany Report #245061 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Physical Assault Lariam Tablets PS Roche
Human Bite<br />
Injury<br />
03-Apr-2012 09:37 AM Page: 242
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/20/00ISR Number: 3575612-8Report Type:Expedited (15-DaCompany Report #245128 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Extrapyramidal Disorder Foreign Lariam PS Hoffmann La Roche<br />
Gastrooesophageal Reflux Health Inc ORAL 1 DOSE FORM 1<br />
Disease Professional PER WEEK ORAL<br />
Primperan<br />
C<br />
Date:09/20/00ISR Number: 3576586-6Report Type:Direct Company Report # Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fear Lariam Cant Remember<br />
Psychotic Disorder<br />
500mg?? Hoffman<br />
Disturbance In Attention Laroche PS Hoffman Laroche ORAL ONE PER ORAL<br />
Hallucination<br />
Abnormal Dreams<br />
Irritability<br />
Panic Attack<br />
Sleep Disorder<br />
Date:09/20/00ISR Number: 3573885-9Report Type:Expedited (15-DaCompany Report #245128 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gastrooesophageal Reflux Lariam Tablets PS Roche 63 DAY<br />
Disease Primperan C 1 DAY<br />
Extrapyramidal Disorder<br />
Date:09/21/00ISR Number: 3575082-XReport Type:Expedited (15-DaCompany Report #200841 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Accommodation Disorder<br />
Fatigue<br />
Insomnia<br />
Nausea<br />
Vision Blurred<br />
Hyperventilation<br />
Lacrimation Increased<br />
Paraesthesia<br />
Hallucination, Visual<br />
Psychomotor Retardation<br />
Diplopia<br />
Arrhythmia<br />
Emotional Disorder<br />
Crying<br />
Asthenia<br />
Paraplegia<br />
Tinnitus
Malaise<br />
Dry Mouth<br />
Balance Disorder<br />
Diarrhoea<br />
Fall<br />
Dyspnoea<br />
Speech Disorder<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 243
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Respiratory Disorder<br />
Convulsion<br />
Memory Impairment Report Source Product Role Manufacturer Route Dose Duration<br />
Stress Lariam Tablets PS Roche CUMULATIVE<br />
Movement Disorder DOSAGE= 750<br />
Cognitive Disorder MG 380 DAY<br />
Hydrocephalus<br />
Meningococcal<br />
Mental Disorder Vaccine C 1 DAY<br />
Heterophoria Flu Vaccine C 1 DAY<br />
Disturbance In Attention<br />
Visual Impairment<br />
Sedation<br />
Panic Attack<br />
Tremor<br />
Dysphagia<br />
Reading Disorder<br />
Date:09/21/00ISR Number: 3576532-5Report Type:Direct Company Report # Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam PS ORAL 1 PILL PER<br />
Depersonalisation<br />
WEEK ORAL<br />
Anxiety<br />
Abnormal Dreams<br />
Dizziness<br />
Nausea<br />
Date:09/25/00ISR Number: 3578070-2Report Type:Expedited (15-DaCompany Report #245128 Age:15 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nervousness Primperan PS 1 DAY<br />
Gastrooesophageal Reflux Lariam Tablets SS Roche 63 DAY<br />
Disease<br />
Throat Tightness<br />
Tremor<br />
Extrapyramidal Disorder<br />
Date:09/25/00ISR Number: 3578162-8Report Type:Expedited (15-DaCompany Report #245128 Age:15 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Extrapyramidal Disorder Primperan PS 1 DAY<br />
Tremor Lariam Tablets SS Roche 63 DAY<br />
Nervousness<br />
Throat Tightness<br />
Gastrooesophageal Reflux<br />
Disease
Date:09/25/00ISR Number: 3578203-8Report Type:Expedited (15-DaCompany Report #245128 Age:15 YR Gender:Female I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Extrapyramidal Disorder<br />
Tremor<br />
Gastrooesophageal Reflux<br />
03-Apr-2012 09:37 AM Page: 244
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disease<br />
Nervousness<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche 63 DAY<br />
Primperan SS 1 DAY<br />
Date:09/25/00ISR Number: 3578365-2Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Larium 250 Mg PS Q WEEK PO<br />
Initial or Prolonged Dyspnoea<br />
Dizziness<br />
Date:09/25/00ISR Number: 3579511-7Report Type:Expedited (15-DaCompany Report #245128 Age:15 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Throat Tightness Foreign Lariam PS Hoffmann La Roche<br />
Extrapyramidal Disorder Health Inc ORAL 1 DOSE FORM 1<br />
Tremor Professional PER WEEK ORAL<br />
Gastrooesophageal Reflux Primperan SS ORAL 1 DOSE FORM 2<br />
Disease<br />
PER DAY ORAL<br />
Nervousness<br />
Date:09/26/00ISR Number: 3578972-7Report Type:Expedited (15-DaCompany Report #237672 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Lariam Tablets PS Roche 57 DAY<br />
Confusional State<br />
Memory Impairment<br />
Alopecia<br />
Cataract<br />
Disturbance In Attention<br />
Fatigue<br />
Visual Impairment<br />
Visual Field Defect<br />
Dizziness<br />
Balance Disorder<br />
Date:09/26/00ISR Number: 3580409-9Report Type:Expedited (15-DaCompany Report #237672 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Memory Impairment Foreign Lariam PS Hoffmann La Roche<br />
Vision Blurred Consumer Inc ORAL 250 MG 1 PER<br />
Fatigue Health WEEK; ORAL<br />
Confusional State<br />
Professional
Visual Field Defect Other<br />
Balance Disorder<br />
Disturbance In Attention<br />
Dizziness<br />
Cataract<br />
Visual Impairment<br />
Alopecia<br />
03-Apr-2012 09:37 AM Page: 245
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/26/00ISR Number: 3580555-XReport Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fear Lariam PS 1<br />
Anxiety<br />
Panic Disorder<br />
Date:09/27/00ISR Number: 3581501-5Report Type:Expedited (15-DaCompany Report #245340 Age:16 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyskinesia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL ORAL<br />
Professional<br />
Date:09/27/00ISR Number: 3580067-3Report Type:Expedited (15-DaCompany Report #245340 Age:16 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyskinesia Lariam Tablets PS Roche<br />
Initial or Prolonged<br />
Date:09/29/00ISR Number: 3582445-5Report Type:Expedited (15-DaCompany Report #245721 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety <strong>Mefloquine</strong><br />
Initial or Prolonged Depressed Mood Hydrochloride PS Roche<br />
Panic Attack Fluoxetine C<br />
Date:10/02/00ISR Number: 3584867-5Report Type:Direct Company Report # Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Lariam PS ORAL 1TAB PO Q WK<br />
Hypoaesthesia<br />
FOR 16 WKS<br />
Date:10/02/00ISR Number: 3584214-9Report Type:Expedited (15-DaCompany Report #245885 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Lariam Tablets PS Roche 1462 DAY<br />
Depression<br />
Decreased Activity<br />
Anxiety
Date:10/02/00ISR Number: 3584219-8Report Type:Expedited (15-DaCompany Report #245766 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Acuity Reduced Lariam Tablets PS Roche<br />
Papilloedema<br />
03-Apr-2012 09:37 AM Page: 246
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/03/00ISR Number: 3586759-4Report Type:Expedited (15-DaCompany Report #245766 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Acuity Reduced Foreign Lariam PS Hoffmann La Roche<br />
Papilloedema Health Inc ORAL ORAL<br />
Professional<br />
Other<br />
Date:10/03/00ISR Number: 3586870-8Report Type:Expedited (15-DaCompany Report #245974 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Syncope Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Anaphylactic Reaction Consumer Inc ORAL ORAL<br />
Prevent Permanent Ill-Defined Disorder<br />
Impairment/Damage<br />
Date:10/03/00ISR Number: 3586878-2Report Type:Expedited (15-DaCompany Report #245885 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Foreign Lariam PS Hoffmann La Roche<br />
Amnesia Other Inc ORAL ORAL<br />
Depression<br />
Date:10/03/00ISR Number: 3585187-5Report Type:Expedited (15-DaCompany Report #245974 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anaphylactic Shock Lariam Tablets PS Roche<br />
Date:10/04/00ISR Number: 3586112-3Report Type:Expedited (15-DaCompany Report #245994 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Ischaemic Stroke <strong>Mefloquine</strong><br />
Initial or Prolonged Haemorrhage Intracranial Hydrochloride PS Roche<br />
Hemianopia Homonymous Aspirin Cardio C<br />
Cerebral Infarction<br />
Date:10/04/00ISR Number: 3587874-1Report Type:Expedited (15-DaCompany Report #246098 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Impairment Foreign Lariam PS Hoffmann La Roche<br />
Aphasia Other Inc ORAL ORAL 28 DAY<br />
Anxiety<br />
Paranoia
Date:10/04/00ISR Number: 3586113-5Report Type:Expedited (15-DaCompany Report #246098 Age:22 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Anxiety<br />
Visual Impairment<br />
03-Apr-2012 09:37 AM Page: 247
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paranoia<br />
Aphasia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche 28 DAY<br />
Date:10/05/00ISR Number: 3587508-6Report Type:Expedited (15-DaCompany Report #241950 Age:11 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Malaria Lariam Tablets PS Roche<br />
Hospitalization - Tachycardia<br />
Initial or Prolonged Hypotension<br />
Loss Of Consciousness<br />
Malaise<br />
Date:10/06/00ISR Number: 3588421-0Report Type:Expedited (15-DaCompany Report #61443 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delirium Lariam Tablets PS Roche 22 DAY<br />
Initial or Prolonged Mental Disorder Lysanxia C<br />
Depression<br />
Anxiety<br />
Suicide Attempt<br />
Overdose<br />
Abnormal Behaviour<br />
Date:10/06/00ISR Number: 3589373-XReport Type:Direct Company Report # Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Autism Spectrum Disorder Larium PS WEEKLY<br />
Initial or Prolonged Maternal Drugs Affecting<br />
Disability<br />
Foetus<br />
Congenital Anomaly Nervous System Disorder<br />
Grand Mal Convulsion<br />
Date:10/06/00ISR Number: 3590758-6Report Type:Expedited (15-DaCompany Report #61443 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Depression Other Inc ORAL 250 MG 1 PER<br />
Anxiety<br />
WEEK ORAL<br />
Persecutory Delusion Lysanxia (Prazepam) C<br />
Partner Stress<br />
Abnormal Behaviour<br />
Delirium<br />
Suicide Attempt
Date:10/10/00ISR Number: 3591920-9Report Type:Expedited (15-DaCompany Report #216859 Age:20 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Emotional Disorder<br />
Rhinorrhoea<br />
03-Apr-2012 09:37 AM Page: 248
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Lymphadenopathy<br />
Erythema<br />
Bronchospasm Report Source Product Role Manufacturer Route Dose Duration<br />
Headache Health Lariam PS Hoffmann La Roche<br />
Pharyngitis Streptococcal Professional Inc ORAL 250 MG 1 PER<br />
Pharyngitis Other WEEK ORAL<br />
Lichen Planus<br />
Skin Discolouration<br />
Date:10/11/00ISR Number: 3593063-7Report Type:Expedited (15-DaCompany Report #246417 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Foreign Lariam PS Hoffmann La Roche<br />
Mania Other Inc ORAL 1 PER WEEK<br />
Psychotic Disorder<br />
ORAL<br />
Depression<br />
Schizophreniform Disorder<br />
Delusion<br />
Paranoia<br />
Date:10/11/00ISR Number: 3593064-9Report Type:Expedited (15-DaCompany Report #245340 Age:17 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Grand Mal Convulsion Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Speech Disorder Health Inc ORAL ORAL<br />
Malaria<br />
Professional<br />
Clonic Convulsion<br />
Disorientation<br />
Pyrexia<br />
Chorea<br />
Vomiting<br />
Dyskinesia<br />
Agitation<br />
Abnormal Behaviour<br />
Date:10/11/00ISR Number: 3593110-2Report Type:Expedited (15-DaCompany Report #246415 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Foreign Lariam PS Hoffmann La Roche<br />
Tremor Other Inc ORAL 1 DOSE FORM 1<br />
Delirium<br />
PER WEEK ORAL<br />
Asthenia<br />
Date:10/12/00ISR Number: 3592579-7Report Type:Expedited (15-DaCompany Report #245340 Age:17 YR Gender:Male I/FU:F
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaria<br />
Chorea<br />
Speech Disorder<br />
Dyskinesia<br />
Disorientation<br />
Agitation<br />
Clonic Convulsion<br />
03-Apr-2012 09:37 AM Page: 249
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Abnormal Behaviour<br />
Vomiting Report Source Product Role Manufacturer Route Dose Duration<br />
Health Lariam Tablets PS Roche 2 DAY<br />
Professional<br />
Date:10/12/00ISR Number: 3592642-0Report Type:Expedited (15-DaCompany Report #216859 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lymphadenopathy Health Lariam Tablets PS Roche 50 DAY<br />
Nasal Congestion<br />
Professional<br />
Pharyngitis<br />
Bronchospasm<br />
Emotional Disorder<br />
Emotional Distress<br />
Erythema<br />
Lichen Planus<br />
Skin Discolouration<br />
Rash Erythematous<br />
Oropharyngeal Pain<br />
Rhinorrhoea<br />
Headache<br />
Date:10/12/00ISR Number: 3593005-4Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Sleep Terror Larium , F.<br />
Disability Delusion Hoffmann-La Roche<br />
Other Serious Suicidal Ideation Ltd PS F. Hoffmann-La Roche<br />
Required Disturbance In Attention Ltd ORAL 250-MG, WEEK,<br />
Intervention to Paranoia ORAL<br />
Prevent Permanent Screaming<br />
Impairment/Damage Anxiety<br />
Date:10/12/00ISR Number: 3594160-2Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tachycardia Lariam-Mefloquin PS 1 TABLET PER<br />
Hyperventilation<br />
WEEK<br />
Anxiety<br />
Influenza<br />
Depression<br />
Disturbance In Attention<br />
Insomnia<br />
Feeling Abnormal<br />
Hypertension<br />
Irritable Bowel Syndrome
Paranoia<br />
Panic Attack<br />
Fatigue<br />
03-Apr-2012 09:37 AM Page: 250
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/12/00ISR Number: 3594368-6Report Type:Direct Company Report # Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Lariam PS Roche ORAL ONE PER 8 WKS<br />
Initial or Prolonged Restlessness ORAL<br />
Disability<br />
Myocardial Infarction<br />
Required<br />
Hypertension<br />
Intervention to<br />
Musculoskeletal Stiffness<br />
Prevent Permanent<br />
Impairment/Damage<br />
Fatigue<br />
Confusional State<br />
Tachycardia<br />
Headache<br />
Sedation<br />
Muscle Spasms<br />
Depression<br />
Weight Increased<br />
Panic Attack<br />
Insomnia<br />
Muscle Disorder<br />
Chest Pain<br />
Date:10/12/00ISR Number: 3592569-4Report Type:Expedited (15-DaCompany Report #246415 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Lariam Tablets PS Roche<br />
Pyrexia<br />
Delirium<br />
Asthenia<br />
Date:10/12/00ISR Number: 3592586-4Report Type:Expedited (15-DaCompany Report #246417 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Lariam Tablets PS Roche<br />
Delusion<br />
Schizophreniform Disorder<br />
Depression<br />
Paranoia<br />
Mania<br />
Hallucination<br />
Date:10/13/00ISR Number: 3594815-XReport Type:Expedited (15-DaCompany Report #246414 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Foreign Lariam PS Hoffmann La Roche<br />
Pyrexia Other Inc ORAL ORAL<br />
Delirium<br />
Dermatitis
Delusion<br />
Rash Maculo-Papular<br />
Paranoia<br />
Neurosis<br />
Confusional State<br />
03-Apr-2012 09:37 AM Page: 251
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/16/00ISR Number: 3594523-5Report Type:Expedited (15-DaCompany Report #216859 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pharyngitis Lariam Tablets PS Roche 50 DAY<br />
Thermal Burn<br />
Lichen Planus<br />
Skin Discolouration<br />
Emotional Disorder<br />
Lymphadenopathy<br />
Bronchospasm<br />
Pharyngitis Streptococcal<br />
Erythema<br />
Rhinorrhoea<br />
Headache<br />
Date:10/16/00ISR Number: 3594525-9Report Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Discolouration Lariam Tablets PS Roche 50 DAY<br />
Infection<br />
Skin Exfoliation<br />
Economic Problem<br />
Fibrous Histiocytoma<br />
Agitation<br />
Eczema<br />
Hyperkeratosis<br />
Lichen Planus<br />
Marital Problem<br />
Libido Decreased<br />
Emotional Distress<br />
Lichenification<br />
Depression<br />
Fear<br />
Neurosis<br />
Neurodermatitis<br />
Dermatitis<br />
Blister<br />
Folliculitis<br />
Emotional Disorder<br />
Pruritus<br />
Skin Disorder<br />
Condition Aggravated<br />
Date:10/16/00ISR Number: 3594532-6Report Type:Expedited (15-DaCompany Report #216810 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Discolouration Lariam Tablets PS Roche 50 DAY<br />
Lichen Planus<br />
Emotional Distress
Skin Disorder<br />
Emotional Disorder<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 252
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/16/00ISR Number: 3595588-7Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Lariam 250 Mg Roche<br />
Abnormal Dreams Laboratories PS Roche Laboratories ORAL 250 MG 1X<br />
Joint Stiffness<br />
WEEK/4 ORAL<br />
Dermatitis<br />
Dizziness<br />
Syncope<br />
Back Pain<br />
Diarrhoea<br />
Date:10/16/00ISR Number: 3595592-9Report Type:Direct Company Report # Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Pain Larium Roche PS Roche 1 OTHER<br />
Required<br />
Phobia<br />
Intervention to<br />
Panic Reaction<br />
Prevent Permanent Dizziness<br />
Impairment/Damage Paranoia<br />
Headache<br />
Abnormal Dreams<br />
Lethargy<br />
Hallucination<br />
Hypersomnia<br />
Date:10/16/00ISR Number: 3595866-1Report Type:Expedited (15-DaCompany Report #216810 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Discolouration Other Lariam PS Hoffmann La Roche<br />
Skin Disorder Inc ORAL 250 MG 1 PER<br />
Depression<br />
WEEK; ORAL<br />
Emotional Disorder<br />
Lichen Planus<br />
Date:10/16/00ISR Number: 3595867-3Report Type:Expedited (15-DaCompany Report #216859 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lymphadenopathy Health Lariam PS Hoffmann La Roche<br />
Skin Discolouration Professional Inc ORAL 250 MG 1 PER<br />
Rhinorrhoea Other WEEK; ORAL<br />
Pharyngitis Streptococcal<br />
Headache<br />
Rash Erythematous<br />
Emotional Disorder<br />
Pharyngitis<br />
Oropharyngeal Pain
Lichen Planus<br />
Skin Disorder<br />
Bronchospasm<br />
Thermal Burn<br />
03-Apr-2012 09:37 AM Page: 253
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/16/00ISR Number: 3595868-5Report Type:Expedited (15-DaCompany Report #216747 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lichen Planus Other Lariam PS Hoffmann La Roche<br />
Agitation Inc ORAL 250 MG 1 PER<br />
Ulcer<br />
WEEK ORAL<br />
Blister<br />
Folliculitis<br />
Skin Lesion<br />
Pruritus<br />
Medication Error<br />
Partner Stress<br />
Skin Exfoliation<br />
Eczema<br />
Necrosis<br />
Marital Problem<br />
Skin Hyperpigmentation<br />
Hyperkeratosis<br />
Infection<br />
Neurodermatitis<br />
Skin Discolouration<br />
Condition Aggravated<br />
Emotional Disorder<br />
Fear<br />
Date:10/16/00ISR Number: 3596305-7Report Type:Expedited (15-DaCompany Report #246861 Age:54 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deep Vein Thrombosis Foreign Lariam PS Hoffmann La Roche<br />
Other Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:10/16/00ISR Number: 3594515-6Report Type:Expedited (15-DaCompany Report #246414 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Lariam Tablets PS Roche 1 DAY<br />
Confusional State<br />
Pyrexia<br />
Delirium<br />
Paranoia<br />
Delusion<br />
Neurosis<br />
Rash Maculo-Papular<br />
Date:10/16/00ISR Number: 3594534-XReport Type:Expedited (15-DaCompany Report #246861 Age:54 YR Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Deep Vein Thrombosis <strong>Mefloquine</strong><br />
Hydrochloride PS Roche 29 DAY<br />
03-Apr-2012 09:37 AM Page: 254
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/17/00ISR Number: 3595469-9Report Type:Expedited (15-DaCompany Report #245766 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Acuity Reduced Lariam Tablets PS Roche<br />
Scotoma<br />
Optic Nerve Glioma<br />
Leukaemia<br />
Visual Field Defect<br />
Optic Neuropathy<br />
Lipoma<br />
Meningioma<br />
Sarcoidosis<br />
Empty Sella Syndrome<br />
Optic Ischaemic<br />
Neuropathy<br />
Lymphoma<br />
Papilloedema<br />
Metastasis<br />
Date:10/17/00ISR Number: 3596671-2Report Type:Direct Company Report # Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Lariam <strong>Mefloquine</strong><br />
Restlessness 250 Mg PS Roche Lab. ORAL ONE<br />
Panic Disorder<br />
TABLE/WEEKLY/<br />
ORAL<br />
Date:10/19/00ISR Number: 3597344-2Report Type:Expedited (15-DaCompany Report #235163 Age: Gender:Not SpecifiI/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Missed Lariam Tablets PS Roche 32 DAY<br />
Pregnancy<br />
Date:10/20/00ISR Number: 3598149-9Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Lariam Tablets PS Roche ESTIMATED 5<br />
Initial or Prolonged Anxiety DOSES IN<br />
Disability Panic Attack TOTAL. 21 DAY<br />
Dizziness<br />
Headache<br />
Depression<br />
Date:10/20/00ISR Number: 3598174-8Report Type:Expedited (15-DaCompany Report #211570 Age:1 HR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Cardiac Valve Disease Lariam Tablets PS Roche<br />
Mitral Valve Incompetence<br />
Heart Disease Congenital<br />
Maternal Drugs Affecting<br />
Foetus<br />
03-Apr-2012 09:37 AM Page: 255
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/20/00ISR Number: 3599182-3Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Level Above Health Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Therapeutic Professional Inc ORAL 250 MG 1 PER<br />
Disability Anxiety WEEK ORAL<br />
Panic Attack<br />
Vertigo<br />
Dizziness<br />
Depression<br />
Headache<br />
Date:10/20/00ISR Number: 3599647-4Report Type:Expedited (15-DaCompany Report #235163 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Pregnancy Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Abortion Missed Health Inc ORAL 1 DOSE FORM<br />
Prevent Permanent Professional 1PER WEEK<br />
Impairment/Damage<br />
ORAL<br />
Date:10/20/00ISR Number: 3599649-8Report Type:Expedited (15-DaCompany Report #247262 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicide Attempt Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:10/20/00ISR Number: 3599650-4Report Type:Expedited (15-DaCompany Report #211570 Age:1 HR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Mitral Valve Foreign Lariam PS Hoffmann La Roche<br />
Incompetence Health Inc ORAL 250 MG 1 PER<br />
Heart Disease Congenital Professional WEEK ORAL<br />
Cardiac Valve Disease<br />
Maternal Drugs Affecting<br />
Foetus<br />
Heart Valve Replacement<br />
Date:10/20/00ISR Number: 3599651-6Report Type:Expedited (15-DaCompany Report #247267 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoxia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Lung Infiltration Other Inc ORAL ORAL<br />
Pneumonitis<br />
Pyrexia
Dyspnoea<br />
03-Apr-2012 09:37 AM Page: 256
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/20/00ISR Number: 3599766-2Report Type:Expedited (15-DaCompany Report #245766 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Papilloedema Foreign Lariam PS Hoffmann La Roche<br />
Visual Field Defect Health Inc ORAL ORAL<br />
Visual Acuity Reduced<br />
Professional<br />
Empty Sella Syndrome<br />
Lipoma<br />
Optic Neuritis<br />
Retinal Exudates<br />
Optic Neuropathy<br />
Optic Nerve Disorder<br />
Date:10/20/00ISR Number: 3598163-3Report Type:Expedited (15-DaCompany Report #247262 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicide Attempt Lariam Tablets PS Roche<br />
Initial or Prolonged<br />
Date:10/20/00ISR Number: 3598168-2Report Type:Expedited (15-DaCompany Report #247267 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoxia Lariam Tablets PS Roche 2 DAY<br />
Initial or Prolonged Pneumonitis<br />
Dyspnoea<br />
Pyrexia<br />
Date:10/23/00ISR Number: 3599859-XReport Type:Direct Company Report # Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia Consumer Larium -<strong>Mefloquine</strong>-<br />
Nightmare Roche PS Roche ORAL ONE PILL WEEK<br />
Insomnia<br />
ORAL<br />
Fatigue<br />
Vertigo<br />
Dizziness<br />
Confusional State<br />
Listless<br />
Paranoia<br />
Disturbance In Attention<br />
Lethargy<br />
Headache<br />
Depression<br />
Date:10/23/00ISR Number: 3600682-8Report Type:Expedited (15-DaCompany Report #245745 Age:36 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Menorrhagia Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Consumer Inc ORAL 250 MG 1 PER<br />
Prevent Permanent<br />
WEEK ORAL<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 257
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/23/00ISR Number: 3599102-1Report Type:Expedited (15-DaCompany Report #245745 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vaginal Haemorrhage Lariam Tablets PS Roche 36 DAY<br />
Menorrhagia<br />
Date:10/24/00ISR Number: 3600623-3Report Type:Direct Company Report # Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Larium Once A Week PS ORAL ONCE A WEEK<br />
Mental Impairment<br />
ORAL<br />
Amnesia<br />
Disturbance In Attention<br />
Date:10/24/00ISR Number: 3601395-9Report Type:Expedited (15-DaCompany Report #247406 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Foreign Lariam PS Hoffmann La Roche<br />
Dyskinesia Other Inc ORAL 250 MG 1 PER<br />
Eye Movement Disorder<br />
WEEK ORAL<br />
Movement Disorder<br />
Salmonella Vaccine<br />
Disorientation (Typhoid Vaccines) C<br />
Date:10/24/00ISR Number: 3599672-3Report Type:Expedited (15-DaCompany Report #247406 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation Lariam Tablets PS Roche 15 DAY<br />
Abnormal Dreams Salmonella Vaccine C 1 DAY<br />
Eye Movement Disorder<br />
Movement Disorder<br />
Date:10/26/00ISR Number: 3601036-0Report Type:Expedited (15-DaCompany Report #243034 Age:80 YR Gender:Female I/FU:F<br />
Outcome<br />
Death<br />
Life-Threatening<br />
PT<br />
Stupor<br />
Abdominal Pain<br />
Gastrointestinal Disorder<br />
Extensor Plantar Response<br />
X-Ray Gastrointestinal<br />
Tract Abnormal<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal<br />
Mental Disorder Due To A<br />
General Medical Condition
Gastric Perforation<br />
Cardio-Respiratory Arrest<br />
Angiopathy<br />
Dyspnoea<br />
Gastrointestinal Sounds<br />
Abnormal<br />
Sedation<br />
Cardiac Failure<br />
03-Apr-2012 09:37 AM Page: 258
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abdominal Tenderness<br />
C-Reactive Protein<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Hypophagia Lariam Tablets PS Roche 1 DAY<br />
Arrhythmia<br />
Shock<br />
Respiratory Failure<br />
Flatulence<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Depressed Level Of<br />
Consciousness<br />
Asthenia<br />
Confusional State<br />
Cardiac Murmur<br />
Oedema<br />
Date:10/26/00ISR Number: 3601049-9Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Lariam Tablets PS Roche 280 DAY<br />
Date:10/27/00ISR Number: 3602925-3Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 1 PER WEEK<br />
ORAL<br />
Date:10/27/00ISR Number: 3603403-8Report Type:Expedited (15-DaCompany Report #243034 Age:80 YR Gender:Female I/FU:F<br />
Outcome<br />
Death<br />
Life-Threatening<br />
PT<br />
Asthenia<br />
Endoscopy Abnormal<br />
Abdominal Pain<br />
Extensor Plantar Response<br />
Gastric Perforation<br />
Confusional State<br />
Abdominal Tenderness<br />
Respiratory Failure<br />
Cardiac Failure<br />
X-Ray Gastrointestinal<br />
Tract Abnormal<br />
Respiratory Disorder<br />
Shock<br />
Blood Lactate<br />
Dehydrogenase Increased
Ill-Defined Disorder<br />
Mental Disorder Due To A<br />
General Medical Condition<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal<br />
C-Reactive Protein<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 259
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Condition Aggravated<br />
Ileus Paralytic<br />
Cardio-Respiratory Arrest Report Source Product Role Manufacturer Route Dose Duration<br />
Dyspnoea Foreign Lariam PS Hoffmann La Roche<br />
Arrhythmia Health Inc ORAL 2 DOSE FORM 1<br />
Professional<br />
PER ONE DOSE<br />
ORAL<br />
Date:10/30/00ISR Number: 3602885-5Report Type:Expedited (15-DaCompany Report #87481 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Thinking Abnormal Lariam Tablets PS Roche 180 DAY<br />
Bone Pain Lamisil C<br />
Feeling Abnormal<br />
Job Dissatisfaction<br />
Sleep Apnoea Syndrome<br />
Vomiting<br />
Loss Of Employment<br />
Brain Injury<br />
Amnesia<br />
Burning Sensation<br />
Oedema<br />
Respiratory Disorder<br />
Disorientation<br />
Dizziness<br />
Drug Level Above<br />
Therapeutic<br />
Disturbance In Attention<br />
Visual Impairment<br />
Hypertension<br />
Medication Error<br />
Pain<br />
Mental Impairment<br />
Mastoid Disorder<br />
Neck Pain<br />
Nausea<br />
Asthenia<br />
Accidental Overdose<br />
Vision Blurred<br />
Depression<br />
Sleep Disorder<br />
Upper Respiratory Tract<br />
Infection<br />
Visual Acuity Reduced<br />
Paraesthesia<br />
Neurological Symptom<br />
Cough<br />
Hypoaesthesia<br />
Nocturia<br />
Fatigue<br />
Dysgeusia
Oedema Peripheral<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 260
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/30/00ISR Number: 3602919-8Report Type:Expedited (15-DaCompany Report #200841 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tremor Lariam Tablets PS Roche CUMULATIVE<br />
Initial or Prolonged Asthenia DOSAGE= 750<br />
Disability Disturbance In Attention MG 380 DAY<br />
Visual Impairment<br />
Meningococcal<br />
Convulsion Vaccine C 1 DAY<br />
Abnormal Behaviour Flu Vaccine C 1 DAY<br />
Mental Disorder<br />
Condition Aggravated<br />
Lacrimation Increased<br />
Stress<br />
Arrhythmia<br />
Hallucination<br />
Dyspnoea<br />
Diplopia<br />
Malaise<br />
Sedation<br />
Paraesthesia<br />
Fall<br />
Movement Disorder<br />
Crying<br />
Fatigue<br />
Hallucination, Visual<br />
Panic Attack<br />
Nausea<br />
Speech Disorder<br />
Balance Disorder<br />
Dysphagia<br />
Tinnitus<br />
Hyperventilation<br />
Vision Blurred<br />
Accommodation Disorder<br />
Dry Mouth<br />
Diarrhoea<br />
Hydrocephalus<br />
Heterophoria<br />
Date:10/30/00ISR Number: 3603006-5Report Type:Direct Company Report # Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam-<strong>Mefloquine</strong> PS 1 / WEEK<br />
Required<br />
Nightmare<br />
Intervention to<br />
Insomnia<br />
Prevent Permanent Mania<br />
Impairment/Damage Mental Disorder<br />
Depression<br />
Nervous System Disorder
Date:10/30/00ISR Number: 3603605-0Report Type:Direct Company Report # Age:36 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Psychotic Disorder<br />
Paranoia<br />
Hallucinations, Mixed<br />
Feeling Abnormal<br />
03-Apr-2012 09:37 AM Page: 261
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Persecutory Delusion<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Larium PS 1 PER WEEK<br />
Date:10/30/00ISR Number: 3603668-2Report Type:Expedited (15-DaCompany Report #247733 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Field Defect Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Other<br />
Date:10/30/00ISR Number: 3603957-1Report Type:Expedited (15-DaCompany Report #87481 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypoaesthesia Literature Lariam PS Hoffmann La Roche<br />
Nausea Health Inc ORAL 250 MG 2 PER<br />
Depression Professional WEEK ORAL<br />
Paraesthesia<br />
Lamisil<br />
Vomiting (Terbinafine) C<br />
Disorientation<br />
Visual Impairment<br />
Asthenia<br />
Medication Error<br />
Dysgeusia<br />
Oedema<br />
Hypertension<br />
Dizziness<br />
Fatigue<br />
Accidental Overdose<br />
Date:10/30/00ISR Number: 3602890-9Report Type:Expedited (15-DaCompany Report #247733 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Field Defect Lariam Tablets PS Roche THE PATIENT<br />
RECEIVED A<br />
TOTAL OF 2<br />
TABLETS. 16 DAY<br />
Date:11/01/00ISR Number: 3604078-4Report Type:Expedited (15-DaCompany Report #245340 Age:17 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Stupor<br />
Insomnia
Ear Disorder<br />
Ventricular Hypertrophy<br />
Clonic Convulsion<br />
Blood Bilirubin Increased<br />
Tinnitus<br />
Dizziness<br />
Neutrophilia<br />
C-Reactive Protein<br />
03-Apr-2012 09:37 AM Page: 262
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Increased<br />
Headache<br />
Speech Disorder<br />
Confusional State Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety Lariam Tablets PS Roche 2 DAY<br />
Dyskinesia Advil C<br />
Agitation<br />
Drug Tolerance Decreased<br />
Electroencephalogram<br />
Abnormal<br />
Abnormal Behaviour<br />
Vomiting<br />
Pyrexia<br />
Diplopia<br />
Thrombocytosis<br />
Chorea<br />
Date:11/01/00ISR Number: 3605361-9Report Type:Direct Company Report # Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Larium <strong>Mefloquine</strong> PS OROPHARINGEAL 1 PILL 1 TIME<br />
Fatigue<br />
W<br />
Dizziness<br />
OROPHARINGEAL<br />
Chills<br />
Vision Blurred<br />
Drug Hypersensitivity<br />
Hypoaesthesia<br />
Anxiety<br />
Headache<br />
Nausea<br />
Dehydration<br />
Pyrexia<br />
Date:11/01/00ISR Number: 3605668-5Report Type:Expedited (15-DaCompany Report #245340 Age:17 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Clonic Convulsion<br />
Insomnia<br />
Malaria<br />
Confusional State<br />
Anxiety<br />
C-Reactive Protein<br />
Increased<br />
Leukocytosis<br />
Stupor<br />
Electroencephalogram<br />
Abnormal<br />
Headache<br />
Chorea<br />
Blood Lactate
Dehydrogenase Increased<br />
Thrombocytosis<br />
Ventricular Hypertrophy<br />
Dizziness<br />
Pyrexia<br />
Diplopia<br />
Haematocrit Decreased<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 263
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tinnitus<br />
Agitation<br />
Depressed Level Of Report Source Product Role Manufacturer Route Dose Duration<br />
Consciousness Foreign Lariam PS Hoffmann La Roche<br />
White Blood Cells Urine Health Inc ORAL ORAL<br />
Positive Professional Advil (Ibuprofen) C<br />
Blood Bilirubin Increased Other<br />
Disorientation<br />
Speech Disorder<br />
Abnormal Behaviour<br />
Cardiomegaly<br />
Dyskinesia<br />
Date:11/02/00ISR Number: 3605199-2Report Type:Expedited (15-DaCompany Report #247893 Age:66 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Syncope Consumer Lariam Tablets PS Roche 61 DAY<br />
Initial or Prolonged Muscle Spasms Plendil C<br />
Epistaxis Moduretic C<br />
Mouth Haemorrhage<br />
Date:11/03/00ISR Number: 3605736-8Report Type:Expedited (15-DaCompany Report #245128 Age:15 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Primperan PS 1 DAY<br />
Tremor Lariam Tablets SS Roche 63 DAY<br />
Extrapyramidal Disorder<br />
Gastrooesophageal Reflux<br />
Disease<br />
Throat Tightness<br />
Date:11/03/00ISR Number: 3605741-1Report Type:Expedited (15-DaCompany Report #230098 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperhidrosis Lariam Tablets PS Roche 22 DAY<br />
Initial or Prolonged Tinnitus Jodthyrox C 731 DAY<br />
Depression Hismanal C<br />
Psychotic Disorder<br />
Paraesthesia<br />
Furuncle<br />
Mental Disorder<br />
Agoraphobia<br />
Retching<br />
Dizziness<br />
Blood Pressure Systolic<br />
Increased<br />
Nausea
Chills<br />
03-Apr-2012 09:37 AM Page: 264
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/06/00ISR Number: 3608144-9Report Type:Expedited (15-DaCompany Report #245128 Age:15 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Foreign Lariam PS Hoffmann La Roche<br />
Extrapyramidal Disorder Health Inc ORAL 1 DOSE FORM<br />
Gastrooesophageal Reflux Professional 1 PER WEEK<br />
Disease<br />
Nervousness<br />
ORAL<br />
Primperan<br />
(Metoclopramide<br />
Hydrochloride) SS ORAL 1 DOSE FORM<br />
2 PER DAY<br />
ORAL<br />
Date:11/06/00ISR Number: 3608146-2Report Type:Expedited (15-DaCompany Report #230098 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperhidrosis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Fatigue Consumer Inc ORAL 250 MG ORAL<br />
Retching Health Hismanal<br />
Crying Professional (Astemizole) C<br />
Agoraphobia<br />
Hismanal<br />
Phobia (Astemizole) C<br />
Dizziness<br />
Jodthyrox<br />
Anxiety<br />
(Levothyroxine<br />
Tinnitus<br />
Sodium/Potassium<br />
Agitation Iodide) C<br />
Conversion Disorder<br />
Abdominal Discomfort<br />
Nausea<br />
Chills<br />
Depression<br />
Obsessive-Compulsive<br />
Disorder<br />
Mental Disorder<br />
Hyperventilation<br />
Psychotic Disorder<br />
Blood Pressure Systolic<br />
Increased<br />
Stress<br />
Paraesthesia<br />
Furuncle<br />
Date:11/06/00ISR Number: 3608353-9Report Type:Expedited (15-DaCompany Report #244563 Age:47 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Depression<br />
Paraesthesia<br />
Mental Impairment<br />
Syncope
Adjustment Disorder<br />
Orgasm Abnormal<br />
Emotional Disorder<br />
Weight Decreased<br />
Fear<br />
Fatigue<br />
Asthenia<br />
Stress<br />
03-Apr-2012 09:37 AM Page: 265
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disturbance In Attention<br />
Vision Blurred<br />
Feeling Abnormal Report Source Product Role Manufacturer Route Dose Duration<br />
Emotional Distress Health Lariam PS Hoffmann La Roche<br />
Abnormal Dreams Professional Inc ORAL ORAL<br />
Anxiety<br />
Other<br />
Crying<br />
Psychiatric Symptom<br />
Panic Attack<br />
Obsessive-Compulsive<br />
Personality Disorder<br />
Dizziness<br />
Tremor<br />
Insomnia<br />
Libido Decreased<br />
Mood Swings<br />
Bradyphrenia<br />
Date:11/06/00ISR Number: 3606583-3Report Type:Expedited (15-DaCompany Report #244563 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Impairment Lariam Tablets PS Roche<br />
Initial or Prolonged Feeling Abnormal<br />
Tremor<br />
Vision Blurred<br />
Fatigue<br />
Anxiety<br />
Orgasm Abnormal<br />
Obsessive-Compulsive<br />
Disorder<br />
Emotional Disorder<br />
Disturbance In Attention<br />
Asthenia<br />
Abnormal Dreams<br />
Dizziness<br />
Sleep Disorder<br />
Depression<br />
Syncope<br />
Weight Decreased<br />
Libido Decreased<br />
Panic Attack<br />
Paraesthesia<br />
Date:11/08/00ISR Number: 3608713-6Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam / <strong>Mefloquine</strong><br />
Required Heart Rate Increased / 250mg / Roche PS Roche ORAL 250MG ONCE A<br />
Intervention to Feeling Abnormal WEEK ORAL
Prevent Permanent<br />
Impairment/Damage<br />
Social Phobia<br />
Dizziness<br />
Insomnia<br />
Hypertension<br />
Confusional State<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 266
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/08/00ISR Number: 3608796-3Report Type:Direct Company Report # Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Melfoquine Lariam PS ORAL 1 1PER WEEK<br />
Initial or Prolonged Angina Pectoris ORAL<br />
Disability Fatigue Felodipine C<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:11/08/00ISR Number: 3608799-9Report Type:Direct Company Report # Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain <strong>Mefloquine</strong> (Lariam) PS ORAL 1 1 PER WEEK<br />
Initial or Prolonged<br />
ORAL<br />
Disability Felodipine C<br />
Required Morphine C<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:11/13/00ISR Number: 3610011-1Report Type:Expedited (15-DaCompany Report #60555 Age:45 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Lariam Tablets PS Roche 8 DAY<br />
Initial or Prolonged Chest Pain Dormonoct C<br />
Anxiety Prothiaden C<br />
Palpitations<br />
Date:11/13/00ISR Number: 3610049-4Report Type:Expedited (15-DaCompany Report #85415 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dizziness Lariam Tablets PS Roche 22 DAY<br />
Nightmare<br />
Alopecia<br />
Depression<br />
Suicidal Ideation<br />
Nausea<br />
Date:11/13/00ISR Number: 3611050-7Report Type:Expedited (15-DaCompany Report #60555 Age:45 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Palpitations Other Inc ORAL 250 MG 1 PER
Depression WEEK ORAL<br />
Anxiety<br />
Dormonoct<br />
(Loprazolam)<br />
C<br />
Prothiaden<br />
(Dothiepin<br />
Hydrochloride) C<br />
03-Apr-2012 09:37 AM Page: 267
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/13/00ISR Number: 3610058-5Report Type:Expedited (15-DaCompany Report #910100239001 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Lariam Tablets PS Roche 15 DAY<br />
Hospitalization - Depression<br />
Initial or Prolonged<br />
Disability<br />
Date:11/14/00ISR Number: 3612201-0Report Type:Expedited (15-DaCompany Report #216859 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Lymphadenopathy Health Lariam PS Hoffmann La Roche<br />
Intervention to Pharyngitis Professional Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Rhinorrhoea Other WEEK ORAL<br />
Impairment/Damage Thermal Burn<br />
Skin Discolouration<br />
Oropharyngeal Pain<br />
Emotional Disorder<br />
Emotional Distress<br />
Lichen Planus<br />
Pharyngitis Streptococcal<br />
Erythema<br />
Refraction Disorder<br />
Bronchospasm<br />
Sinus Congestion<br />
Headache<br />
Date:11/15/00ISR Number: 3611274-9Report Type:Expedited (15-DaCompany Report #216859 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Lariam Tablets PS Roche 50 DAY<br />
Skin Discolouration<br />
Thermal Burn<br />
Lichen Planus<br />
Rhinorrhoea<br />
Refraction Disorder<br />
Lymphadenopathy<br />
Emotional Disorder<br />
Erythema<br />
Pharyngitis<br />
Date:11/15/00ISR Number: 3611309-3Report Type:Expedited (15-DaCompany Report #237590 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Lariam Tablets PS Roche 550 DAY
Date:11/15/00ISR Number: 3611310-XReport Type:Expedited (15-DaCompany Report #237587 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Lariam Tablets PS Roche 550 DAY<br />
03-Apr-2012 09:37 AM Page: 268
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/15/00ISR Number: 3611397-4Report Type:Direct Company Report # Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam 250mg Roche<br />
Dizziness Lab Inc PS Roche Lab Inc ORAL 3 1 PER WEEK<br />
Insomnia<br />
ORAL<br />
Paranoia<br />
Abnormal Dreams<br />
Abnormal Behaviour<br />
Mood Altered<br />
Confusional State<br />
Depression<br />
Date:11/15/00ISR Number: 3612397-0Report Type:Expedited (15-DaCompany Report #85415 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nausea Foreign Lariam PS Hoffmann La Roche<br />
Alopecia Other Inc ORAL 250 MG 1 PER<br />
Depression<br />
WEEK ORAL<br />
Dizziness<br />
Nightmare<br />
Suicidal Ideation<br />
Date:11/15/00ISR Number: 3612515-4Report Type:Expedited (15-DaCompany Report #237587 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Accidental Exposure Foreign Lariam PS Hoffmann La Roche<br />
Abortion Missed Consumer Inc ORAL ORAL<br />
Intra-Uterine Death<br />
Health<br />
Professional<br />
Other<br />
Date:11/15/00ISR Number: 3612728-1Report Type:Expedited (15-DaCompany Report #910100239001 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Health Lariam PS Hoffmann La Roche<br />
Hospitalization - Overdose Professional Inc ORAL 1 DOSE FORM<br />
Initial or Prolonged Dysthymic Disorder 1 PER WEEK<br />
Disability Tremor ORAL<br />
Suicide Attempt<br />
Agitation<br />
Nervousness<br />
Depression<br />
Date:11/15/00ISR Number: 3612784-0Report Type:Expedited (15-DaCompany Report #237590 Age: Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Missed Foreign Lariam PS Hoffmann La Roche<br />
Intra-Uterine Death Consumer Inc ORAL ORAL<br />
Maternal Drugs Affecting Health<br />
Foetus<br />
Professional<br />
Accidental Exposure<br />
Other<br />
03-Apr-2012 09:37 AM Page: 269
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/16/00ISR Number: 3612864-XReport Type:Expedited (15-DaCompany Report #244563 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Decreased Appetite Health Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pneumonia Professional Inc ORAL ORAL<br />
Required Tremor Other<br />
Intervention to<br />
Chills<br />
Prevent Permanent<br />
Impairment/Damage<br />
Orgasm Abnormal<br />
Panic Attack<br />
Eosinophilia<br />
Haemoglobin Decreased<br />
Feeling Abnormal<br />
Musculoskeletal Stiffness<br />
Lymphadenopathy<br />
Dry Mouth<br />
Emotional Disorder<br />
Sleep Disorder<br />
Cognitive Disorder<br />
Headache<br />
Vision Blurred<br />
Asthenia<br />
Bradycardia<br />
Paraesthesia<br />
Obsessive-Compulsive<br />
Disorder<br />
Diarrhoea<br />
Mental Impairment<br />
Dehydration<br />
Syncope<br />
Disturbance In Attention<br />
Bronchitis<br />
Skin Lesion<br />
Abnormal Dreams<br />
Fatigue<br />
Depression<br />
Anxiety<br />
Dizziness<br />
Upper Respiratory Tract<br />
Infection<br />
Libido Decreased<br />
Mood Swings<br />
Stress<br />
Dysuria<br />
Weight Decreased<br />
Date:11/16/00ISR Number: 3613028-6Report Type:Expedited (15-DaCompany Report #234234 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Stillbirth Foreign Lariam PS Hoffmann La Roche<br />
Premature Labour Health Inc ORAL 1 PER WEEK<br />
Placental Insufficiency Professional ORAL
Caesarean Section Meningococcal<br />
Maternal Drugs Affecting<br />
Vaccine<br />
Foetus<br />
(Meningococcal<br />
Vaccines Nos)<br />
Diphtheria Vaccine<br />
(Diphtheria Toxoid)<br />
Cholera Vaccine<br />
(Cholera Vaccine)<br />
C<br />
C<br />
C<br />
03-Apr-2012 09:37 AM Page: 270
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Typhus Vaccine<br />
(Typhus Vaccine)<br />
Hepatitis A Vaccine<br />
(Hepatitis A<br />
Vaccine)<br />
C<br />
C<br />
Date:11/16/00ISR Number: 3613117-6Report Type:Expedited (15-DaCompany Report #248735 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Interaction Foreign Lariam PS Hoffmann La Roche<br />
International Normalised Health Inc ORAL ORAL<br />
Ratio Increased Professional Warfarin (Warfarin<br />
Sodium)<br />
SS<br />
Co Proxamol<br />
(Acetaminophen /<br />
Prophoxyphene<br />
Hydrochloride) C<br />
Date:11/17/00ISR Number: 3613227-3Report Type:Direct Company Report # Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arthropathy Lariam 1 Pill Weekly PS 1 PILL<br />
Initial or Prolonged Diarrhoea<br />
Other Serious<br />
Vomiting<br />
Abdominal Pain<br />
Arthritis<br />
Date:11/21/00ISR Number: 3613999-8Report Type:Expedited (15-DaCompany Report #85415 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Lariam Tablets PS Roche 22 DAY<br />
Alopecia<br />
Nightmare<br />
Depression<br />
Dizziness<br />
Nausea<br />
Date:11/21/00ISR Number: 3614000-2Report Type:Expedited (15-DaCompany Report #89618 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Contractions Lariam Tablets PS Roche 8 DAY<br />
Involuntary<br />
Paraesthesia<br />
Insomnia<br />
Abnormal Dreams
Psychotic Disorder<br />
Crying<br />
Hyperreflexia<br />
Panic Reaction<br />
Palpitations<br />
Hypoaesthesia<br />
Hyperhidrosis<br />
Decreased Appetite<br />
03-Apr-2012 09:37 AM Page: 271
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/21/00ISR Number: 3614035-XReport Type:Expedited (15-DaCompany Report #910100239001 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Lariam Tablets PS Roche 15 DAY<br />
Hospitalization - Depression<br />
Initial or Prolonged<br />
Disability<br />
Date:11/21/00ISR Number: 3614074-9Report Type:Expedited (15-DaCompany Report #930202261001 Age:41 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Reaction Lariam PS Roche 11 DAY<br />
Initial or Prolonged Diarrhoea<br />
Disability<br />
Decreased Appetite<br />
Vertigo<br />
Abnormal Behaviour<br />
Hyperhidrosis<br />
Visual Impairment<br />
Dermatitis<br />
Chromaturia<br />
Weight Decreased<br />
White Blood Cell Count<br />
Increased<br />
Suicidal Ideation<br />
Gastritis<br />
Nausea<br />
Fatigue<br />
Hyperaesthesia<br />
Depression<br />
Balance Disorder<br />
Pollakiuria<br />
Anxiety<br />
Crying<br />
Dyspepsia<br />
Malaise<br />
Pharyngeal Disorder<br />
Date:11/21/00ISR Number: 3615482-2Report Type:Expedited (15-DaCompany Report #910100239001 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Overdose Health Lariam PS Hoffmann La Roche<br />
Hospitalization - Suicide Attempt Professional Inc ORAL 1 DOSE FORM 1<br />
Initial or Prolonged Suicidal Ideation PER WEEK ORAL<br />
Disability<br />
Tremor<br />
Agitation<br />
Nervousness<br />
Depression<br />
Dysthymic Disorder
Date:11/21/00ISR Number: 3615623-7Report Type:Expedited (15-DaCompany Report #930202261001 Age:41 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Hyperhidrosis<br />
Fatigue<br />
Dyspepsia<br />
03-Apr-2012 09:37 AM Page: 272
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Injury<br />
Gastritis<br />
White Blood Cell Count Report Source Product Role Manufacturer Route Dose Duration<br />
Increased Consumer Lariam PS Hoffmann La Roche<br />
Suicidal Ideation Health Inc ORAL 250 MG 1 PER<br />
Balance Disorder Professional WEEK ORAL<br />
Diarrhoea<br />
Other<br />
Weight Decreased<br />
Abnormal Faeces<br />
Insomnia<br />
Chromaturia<br />
Dermatitis<br />
Depression<br />
Anxiety<br />
Malaise<br />
Visual Impairment<br />
Abnormal Behaviour<br />
Paraesthesia<br />
Pollakiuria<br />
Decreased Appetite<br />
Nausea<br />
Foreign Body<br />
Feeling Abnormal<br />
Crying<br />
Vertigo<br />
Date:11/21/00ISR Number: 3615624-9Report Type:Expedited (15-DaCompany Report #89618 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Movement Disorder Consumer Lariam PS Hoffmann La Roche<br />
Dyspnoea Other Inc ORAL 250 MG 1 PER<br />
Decreased Appetite<br />
WEEK ORAL<br />
Psychotic Disorder<br />
Insomnia<br />
Hyperhidrosis<br />
Hypophagia<br />
Hyperreflexia<br />
Hypoaesthesia<br />
Palpitations<br />
Panic Attack<br />
Abnormal Dreams<br />
Crying<br />
Paraesthesia<br />
Muscle Contractions<br />
Involuntary<br />
Date:11/21/00ISR Number: 3615641-9Report Type:Expedited (15-DaCompany Report #248917 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Neuritis Foreign Lariam PS Hoffmann La Roche<br />
Inner Ear Disorder Consumer Inc ORAL ORAL<br />
Dizziness<br />
Dermatitis<br />
03-Apr-2012 09:37 AM Page: 273
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/21/00ISR Number: 3615646-8Report Type:Expedited (15-DaCompany Report #85415 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Depression Suicidal Foreign Lariam PS Hoffmann La Roche<br />
Dizziness Other Inc ORAL 250 MG 1 PER<br />
Alopecia<br />
WEEK ORAL<br />
Nausea<br />
Nightmare<br />
Date:11/21/00ISR Number: 3614037-3Report Type:Expedited (15-DaCompany Report #249131 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gamma-Glutamyltransferase Lariam Tablets PS Roche<br />
Increased<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Date:11/21/00ISR Number: 3614045-2Report Type:Expedited (15-DaCompany Report #249135 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Respiratory Arrest Lariam Tablets PS Roche<br />
Doxycyclin<br />
C<br />
Date:11/21/00ISR Number: 3614079-8Report Type:Expedited (15-DaCompany Report #248917 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ear Disorder Lariam Tablets PS Roche TWO DOSES OF<br />
Dermatitis<br />
LARIAM TAKEN,<br />
ONE ON 7 JAN<br />
00 AND ONE ON<br />
14 JAN 00. 9 DAY<br />
Date:11/22/00ISR Number: 3615108-8Report Type:Expedited (15-DaCompany Report #203837 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
PT<br />
Weight Decreased<br />
Agitation<br />
Flushing<br />
Delusional Disorder,<br />
Persecutory Type<br />
Nervous System Disorder<br />
Cognitive Disorder<br />
Urinary Hesitation<br />
Hyperhidrosis<br />
Fatigue
Musculoskeletal Stiffness<br />
Depression<br />
Polydipsia<br />
Delirium<br />
Anxiety<br />
Insomnia<br />
Abnormal Dreams<br />
Completed Suicide<br />
03-Apr-2012 09:37 AM Page: 274
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Amnesia<br />
Chills<br />
Headache Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Appetite Lariam Tablets PS Roche 50 DAY<br />
Obsessive-Compulsive<br />
Disorder<br />
Hallucinations, Mixed<br />
Adjustment Disorder<br />
Date:11/22/00ISR Number: 3616466-0Report Type:Expedited (15-DaCompany Report #248917 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Foreign Lariam PS Hoffmann La Roche<br />
Ear Disorder Consumer Inc ORAL ORAL<br />
Neuritis<br />
Dermatitis<br />
Balance Disorder<br />
Vestibular Disorder<br />
Date:11/22/00ISR Number: 3616467-2Report Type:Expedited (15-DaCompany Report #85415 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nausea Foreign Lariam PS Hoffmann La Roche<br />
Dizziness Other Inc ORAL 250 MG 1 PER<br />
Alopecia<br />
WEEK ORAL<br />
Nightmare<br />
Depression<br />
Suicidal Ideation<br />
Date:11/22/00ISR Number: 3616469-6Report Type:Expedited (15-DaCompany Report #249131 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Red Blood Cell Foreign Lariam PS Hoffmann La Roche<br />
Sedimentation Rate Health Inc ORAL ORAL<br />
Increased<br />
Professional<br />
Liver Function Test<br />
Abnormal<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Blood Uric Acid Increased<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Blood Iron Decreased<br />
Condition Aggravated
Date:11/22/00ISR Number: 3616470-2Report Type:Expedited (15-DaCompany Report #249135 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Respiratory Arrest Foreign Lariam PS Hoffmann La Roche<br />
Convulsion Other Inc ORAL ORAL<br />
Circulatory Collapse<br />
Doxycyclin<br />
(Doxycycline)<br />
C<br />
03-Apr-2012 09:37 AM Page: 275
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/22/00ISR Number: 3617622-8Report Type:Expedited (15-DaCompany Report #203837 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Sleep Disorder Consumer Lariam PS Hoffmann La Roche<br />
Nervous System Disorder Other Inc ORAL 250 MG 1 PER<br />
Insomnia<br />
WEEK ORAL<br />
Fatigue<br />
Depression<br />
Musculoskeletal Stiffness<br />
Polydipsia<br />
Anxiety<br />
Social Problem<br />
Dissociation<br />
Hallucinations, Mixed<br />
Delusion<br />
Flushing<br />
Delirium<br />
Obsessive-Compulsive<br />
Disorder<br />
Decreased Appetite<br />
Chills<br />
Memory Impairment<br />
Mental Impairment<br />
Weight Decreased<br />
Delusional Disorder,<br />
Persecutory Type<br />
Amnesia<br />
Lethargy<br />
Personality Disorder<br />
Nightmare<br />
Dysgraphia<br />
Staring<br />
Cognitive Disorder<br />
Adjustment Disorder<br />
Hyperhidrosis<br />
Abnormal Dreams<br />
Agitation<br />
Completed Suicide<br />
Urinary Hesitation<br />
Fear<br />
Headache<br />
Date:11/24/00ISR Number: 3617901-4Report Type:Expedited (15-DaCompany Report #249282 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Atrioventricular Block Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 250 MG 1 PER<br />
Professional<br />
WEEK ORAL
Date:11/27/00ISR Number: 3616037-6Report Type:Expedited (15-DaCompany Report #249282 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Atrioventricular Block Lariam Tablets PS Roche 45 DAY<br />
03-Apr-2012 09:37 AM Page: 276
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/28/00ISR Number: 3616670-1Report Type:Expedited (15-DaCompany Report #225972 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Kidney Malformation Health Lariam Tablets PS Roche 42 DAY<br />
Hypospadias Professional Magne-B6 C<br />
Maternal Drugs Affecting Tardyferon C<br />
Foetus Speciafoldine C<br />
Aspegic<br />
C<br />
Date:11/28/00ISR Number: 3616675-0Report Type:Expedited (15-DaCompany Report #232768 Age:27 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vision Blurred Health Lariam Tablets PS Roche<br />
Initial or Prolonged Demyelination Professional<br />
Peripheral Coldness<br />
Disturbance In Attention<br />
Pyrexia<br />
Fatigue<br />
Vertigo<br />
Abdominal Pain<br />
Lethargy<br />
Headache<br />
Wolff-Parkinson-White<br />
Syndrome<br />
Dizziness<br />
Balance Disorder<br />
Hyperacusis<br />
Nausea<br />
Weight Increased<br />
Antinuclear Antibody<br />
Positive<br />
Migraine<br />
Eating Disorder<br />
Disorientation<br />
Vomiting<br />
Paraesthesia<br />
Feeling Hot<br />
Dehydration<br />
Dysphagia<br />
Date:11/28/00ISR Number: 3617657-5Report Type:Expedited (15-DaCompany Report #225972 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Lariam Tablets PS Roche 42 DAY<br />
Foetus Magne-B6 C<br />
Kidney Malformation Tardyferon C<br />
Hypospadias Speciafoldine C<br />
Aspegic<br />
C
Date:11/28/00ISR Number: 3617658-7Report Type:Expedited (15-DaCompany Report #214112 Age:18 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Oedema Peripheral<br />
Headache<br />
Nightmare<br />
03-Apr-2012 09:37 AM Page: 277
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Weight Decreased<br />
Vith Nerve Paralysis<br />
Fatigue Report Source Product Role Manufacturer Route Dose Duration<br />
Csf Pressure Decreased Lariam Tablets PS Roche 71 DAY<br />
Benign Intracranial Contraceptive C<br />
Hypertension<br />
Csf Protein Increased<br />
Papilloedema<br />
Diplopia<br />
Date:11/28/00ISR Number: 3617664-2Report Type:Expedited (15-DaCompany Report #232768 Age:27 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperacusis Lariam Tablets PS Roche<br />
Initial or Prolonged Dehydration<br />
Weight Increased<br />
Fatigue<br />
Wolff-Parkinson-White<br />
Syndrome<br />
Migraine<br />
Disorientation<br />
Abdominal Pain<br />
Paraesthesia<br />
Pyrexia<br />
Dizziness<br />
Lethargy<br />
Balance Disorder<br />
Vision Blurred<br />
Antinuclear Antibody<br />
Positive<br />
Anxiety<br />
Angiopathy<br />
Demyelination<br />
Vomiting<br />
Depression<br />
Peripheral Coldness<br />
Dysphagia<br />
Vertigo<br />
Headache<br />
Disturbance In Attention<br />
Eating Disorder<br />
Malaise<br />
Nausea<br />
Feeling Hot<br />
Date:11/28/00ISR Number: 3617700-3Report Type:Expedited (15-DaCompany Report #248975 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Neuropathy Peripheral Lariam Tablets PS Roche 42 DAY
Lethargy<br />
Paraesthesia<br />
Myalgia<br />
03-Apr-2012 09:37 AM Page: 278
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/28/00ISR Number: 3617710-6Report Type:Expedited (15-DaCompany Report #249339 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lichen Planus Lariam Tablets PS Roche 80 DAY<br />
Initial or Prolonged Pruritus<br />
Blister<br />
Date:11/28/00ISR Number: 3617714-3Report Type:Expedited (15-DaCompany Report #249491 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cognitive Disorder Lariam Tablets PS Roche<br />
Gait Disturbance<br />
Amnesia<br />
Dyspraxia<br />
Date:11/28/00ISR Number: 3617717-9Report Type:Expedited (15-DaCompany Report #249476 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Petit Mal Epilepsy Lariam Tablets PS Roche 8 DAY<br />
Date:11/28/00ISR Number: 3618062-8Report Type:Expedited (15-DaCompany Report #225972 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Anomaly Foreign Lariam PS Hoffmann La Roche<br />
Hypospadias Health Inc ORAL 250 MG 1 PER<br />
Maternal Drugs Affecting Professional WEEK ORAL<br />
Foetus<br />
Magen-86 (Magnesium<br />
Renal Disorder<br />
Nos/Pyridoxine<br />
Placental Disorder Hydrochloride) C<br />
Tardyferon (Ferrous<br />
Sulfate)<br />
C<br />
Speciafolidine<br />
(Folic Acid)<br />
C<br />
Aspegic (Aspirin<br />
Dl-Lysine)<br />
C<br />
Date:11/28/00ISR Number: 3618065-3Report Type:Expedited (15-DaCompany Report #232768 Age:27 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Paraesthesia<br />
Vomiting<br />
Hypoaesthesia<br />
Feeling Hot<br />
Hyperacusis<br />
Peripheral Coldness
Vision Blurred<br />
Balance Disorder<br />
Lethargy<br />
Weight Increased<br />
Eating Disorder<br />
Disturbance In Attention<br />
Antinuclear Antibody<br />
Positive<br />
03-Apr-2012 09:37 AM Page: 279
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Migraine<br />
Malaise<br />
Abdominal Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Vertigo Foreign Lariam PS Hoffmann La Roche<br />
Blood Glucose Decreased Health Inc ORAL ORAL<br />
Dizziness<br />
Professional<br />
Dehydration<br />
Disorientation<br />
Dysphagia<br />
Headache<br />
Fatigue<br />
Anxiety<br />
Wolff-Parkinson-White<br />
Syndrome<br />
Pyrexia<br />
Demyelination<br />
Date:11/29/00ISR Number: 3619066-1Report Type:Expedited (15-DaCompany Report #248735 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious International Normalised Foreign Lariam PS Hoffmann La Roche<br />
Ratio Increased Health Inc ORAL 1 PER WEEK<br />
Drug Interaction Professional ORAL<br />
Warfarin (Warfarin<br />
Sodium)<br />
SS<br />
Co Proxamol<br />
(Acetaminophen/<br />
Propoxyphene<br />
Hydrochloride) C<br />
Date:11/29/00ISR Number: 3619067-3Report Type:Expedited (15-DaCompany Report #248975 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Neuropathy Peripheral Foreign Lariam PS Hoffmann La Roche<br />
Lethargy Health Inc ORAL 1 PER WEEK<br />
Pain In Extremity Professional ORAL<br />
Paraesthesia<br />
Date:11/29/00ISR Number: 3619068-5Report Type:Expedited (15-DaCompany Report #214112 Age:18 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Csf Protein Increased Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Diplopia Health Inc ORAL 250 MG 1 PER<br />
Nightmare Professional WEEK ORAL<br />
Fatigue<br />
Contraceptive<br />
Vith Nerve Paralysis (Contraceptive Nos) C<br />
Oedema Peripheral
Papilloedema<br />
Benign Intracranial<br />
Hypertension<br />
03-Apr-2012 09:37 AM Page: 280
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/30/00ISR Number: 3618871-5Report Type:Expedited (15-DaCompany Report #249379 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arrhythmia Lariam Tablets PS Roche 36 DAY<br />
Initial or Prolonged Bradycardia<br />
Hypertension<br />
Date:11/30/00ISR Number: 3618882-XReport Type:Expedited (15-DaCompany Report #249874 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Agitation Lariam Tablets PS Roche 31 DAY<br />
Electroencephalogram<br />
Abnormal<br />
Psychotic Disorder<br />
Balance Disorder<br />
Amnesia<br />
Insomnia<br />
Emotional Distress<br />
Nightmare<br />
Headache<br />
Visual Impairment<br />
Educational Problem<br />
Date:12/01/00ISR Number: 3620945-XReport Type:Expedited (15-DaCompany Report #249874 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Headache Other Lariam PS Hoffmann La Roche<br />
Insomnia Inc ORAL ORAL<br />
Emotional Distress<br />
Electroencephalogram<br />
Abnormal<br />
Nightmare<br />
Amnesia<br />
Psychotic Disorder<br />
Learning Disorder<br />
Agitation<br />
Visual Impairment<br />
Balance Disorder<br />
Date:12/01/00ISR Number: 3621130-8Report Type:Expedited (15-DaCompany Report #249379 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Bradycardia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Arrhythmia Health Inc ORAL 250 MG 1 PER<br />
Hypertension Professional WEEK ORAL<br />
Other
Date:12/06/00ISR Number: 3622972-5Report Type:Expedited (15-DaCompany Report #242660 Age:53 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Parosmia Lariam Tablets PS Roche<br />
Nasopharyngitis Stamaril Unidose C 1 DAY<br />
Ageusia Havrix C 1 DAY<br />
03-Apr-2012 09:37 AM Page: 281
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Prophylactic<br />
Medication<br />
C<br />
Date:12/06/00ISR Number: 3622973-7Report Type:Expedited (15-DaCompany Report #249748 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation Lariam Tablets PS Roche<br />
Initial or Prolonged Confusional State<br />
Mutism<br />
Hypertonia<br />
Delusion<br />
Acute Psychosis<br />
Speech Disorder<br />
Disorientation<br />
Date:12/07/00ISR Number: 3623547-4Report Type:Expedited (15-DaCompany Report #249977 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Dizziness Lariam Tablets PS Roche 1 DAY<br />
Decreased Appetite Havrix C 1 DAY<br />
Malaise Typhim C 1 DAY<br />
Skull Fracture<br />
Drowning<br />
Head Injury<br />
Hypophagia<br />
Fatigue<br />
Restlessness<br />
Haemorrhage Subcutaneous<br />
Mutism<br />
Scratch<br />
Confusional State<br />
Personality Change<br />
Fall<br />
Influenza Like Illness<br />
Brain Contusion<br />
Depression<br />
Aggression<br />
Skull Fractured Base<br />
Date:12/07/00ISR Number: 3624751-1Report Type:Expedited (15-DaCompany Report #249748 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Confusional State Literature Inc<br />
Persecutory Delusion<br />
Health<br />
Pyrexia<br />
Professional<br />
Delusion
Hypertonia<br />
Mutism<br />
Disorientation<br />
Speech Disorder<br />
Acute Psychosis<br />
03-Apr-2012 09:37 AM Page: 282
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/07/00ISR Number: 3624753-5Report Type:Expedited (15-DaCompany Report #242660 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nasopharyngitis Foreign Lariam PS Hoffmann La Roche<br />
Anosmia Consumer Inc ORAL 1 DOSE FORM<br />
Ageusia Health 1 PER WEEK<br />
Professional<br />
Stamaril Unidose<br />
(Yellow Fever<br />
Vaccine)<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
Prophylactic<br />
Medication<br />
(Prophylactic<br />
Medication Nos)<br />
C<br />
C<br />
C<br />
ORAL<br />
Date:12/08/00ISR Number: 3624451-8Report Type:Expedited (15-DaCompany Report #249746 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Decreased Appetite <strong>Mefloquine</strong><br />
Initial or Prolonged Persecutory Delusion Hydrochloride PS Roche<br />
Depressed Mood Zopiclone C<br />
Abnormal Behaviour Bromazepam C<br />
Depression<br />
Delusion Of Reference<br />
Hallucinations, Mixed<br />
Soliloquy<br />
Insomnia<br />
Anger<br />
Suicidal Ideation<br />
Crying<br />
Anxiety<br />
Date:12/08/00ISR Number: 3625505-2Report Type:Expedited (15-DaCompany Report #249977 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Dizziness Foreign Lariam PS Hoffmann La Roche<br />
Skull Fractured Base Other Inc ORAL 250 MG 1 PER<br />
Depression<br />
WEEK ORAL<br />
Brain Contusion<br />
Havrix (Hepatitis A<br />
Mutism Vaccine) C<br />
Fall<br />
Typhim (Typhoid<br />
Head Injury Vaccines) C<br />
Restlessness<br />
Skull Fracture<br />
Fatigue<br />
Confusional State<br />
Victim Of Crime
Scratch<br />
Drowning<br />
Haemorrhage Subcutaneous<br />
Decreased Appetite<br />
Malaise<br />
Personality Change<br />
03-Apr-2012 09:37 AM Page: 283
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/11/00ISR Number: 3625248-5Report Type:Expedited (15-DaCompany Report #250469 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pain In Extremity Lariam Tablets PS Roche<br />
Hypoaesthesia<br />
Optic Neuritis<br />
Neurological Symptom<br />
Date:12/11/00ISR Number: 3625253-9Report Type:Expedited (15-DaCompany Report #250135 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coma Lariam Tablets PS Roche 8 DAY<br />
Initial or Prolonged Convulsion Neuroleptic Nos C 1 DAY<br />
Overdose Gardenal C 1 DAY<br />
Agitation<br />
Unknown<br />
Toxicity To Various Benzodiazepines C 1 DAY<br />
Agents<br />
Date:12/11/00ISR Number: 3627203-8Report Type:Expedited (15-DaCompany Report #249746 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Depression Literature Inc<br />
Decreased Appetite Health Zopiclone<br />
Delusion Professional (Zopiclone) C<br />
Hallucinations, Mixed<br />
Bromazepam<br />
(Bromazepam)<br />
C<br />
Date:12/12/00ISR Number: 3626644-2Report Type:Expedited (15-DaCompany Report #250525 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Weight Decreased Lariam Tablets PS Roche 22 DAY<br />
Renal Failure<br />
Date:12/12/00ISR Number: 3627119-7Report Type:Direct Company Report # Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psychotic Disorder Lariam PS ORAL ONE WEE ORAL<br />
Date:12/12/00ISR Number: 3627669-3Report Type:Expedited (15-DaCompany Report #250469 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Neurological Symptom Health Lariam PS Hoffmann La Roche
Hypoaesthesia Professional Inc ORAL ORAL<br />
Pain In Extremity<br />
Optic Neuritis<br />
03-Apr-2012 09:37 AM Page: 284
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/12/00ISR Number: 3628092-8Report Type:Expedited (15-DaCompany Report #250135 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Coma Other Inc ORAL 250 MG 1 PER<br />
Agitation<br />
WEEK ORAL<br />
Neuroleptic Nos<br />
(Neuroleptic Nos) C<br />
Gardenal<br />
(Phenobarbital) C<br />
Unknown<br />
Benzodiazepines<br />
(Benzodiazepine Nos) C<br />
Date:12/13/00ISR Number: 3627583-3Report Type:Expedited (15-DaCompany Report #228520 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bradycardia Lariam Tablets PS Roche CUMULATIVE<br />
Maternal Drugs Affecting DOSE 2000 MG. 60 DAY<br />
Foetus<br />
Date:12/13/00ISR Number: 3629372-2Report Type:Expedited (15-DaCompany Report #250525 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Renal Failure Foreign Lariam PS Hoffmann La Roche<br />
Weight Decreased Health Inc ORAL ORAL<br />
Professional<br />
Date:12/14/00ISR Number: 3628891-2Report Type:Expedited (15-DaCompany Report #250525 Age:15 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Renal Failure Health Lariam Tablets PS Roche 22 DAY<br />
Weight Decreased<br />
Professional<br />
Blood Albumin Increased<br />
Date:12/14/00ISR Number: 3630418-6Report Type:Expedited (15-DaCompany Report #228520 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Bradycardia Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Maternal Drugs Affecting Consumer Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Foetus WEEK ORAL<br />
Impairment/Damage
Date:12/15/00ISR Number: 3630208-4Report Type:Direct Company Report # Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paranoia Lariam 250 Mg<br />
Pyrexia Hoffman-La-Roche PS Hoffman-La-Roche ORAL 1-250 MG 10<br />
Delirium<br />
DAYS ORAL<br />
Tremor<br />
03-Apr-2012 09:37 AM Page: 285
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/15/00ISR Number: 3630214-XReport Type:Direct Company Report # Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Lariam PS ORAL ONCE ORAL<br />
Panic Reaction<br />
Mental Impairment<br />
Date:12/15/00ISR Number: 3630289-8Report Type:Expedited (15-DaCompany Report #250670 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Personality Change Lariam Tablets PS Roche<br />
Agitation Antimalarial Nos SS<br />
Triludan<br />
SS<br />
Date:12/15/00ISR Number: 3631006-8Report Type:Expedited (15-DaCompany Report #248735 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious International Normalised Lariam Tablets PS Roche<br />
Ratio Increased Co Proxamol C<br />
Drug Interaction Warfarin I<br />
Date:12/15/00ISR Number: 3631966-5Report Type:Expedited (15-DaCompany Report #250525 Age:15 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Weight Decreased Foreign Lariam PS Hoffmann La Roche<br />
Renal Failure Health Inc ORAL 1 DOSE FORM<br />
Professional<br />
1 PER WEEK<br />
ORAL<br />
Date:12/18/00ISR Number: 3633587-7Report Type:Direct Company Report # Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Inappropriate Affect Lariam PS Hoffmann-Laroche ORAL 1 TABLE DAY<br />
Other Serious Crying ORAL<br />
Insomnia<br />
Anxiety<br />
Mental Impairment<br />
Panic Reaction<br />
Date:12/19/00ISR Number: 3632679-6Report Type:Expedited (15-DaCompany Report #250403 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bradycardia Lariam Tablets PS Roche 21 DAY
Hospitalization - Ventricular Fibrillation Moduretic C<br />
Initial or Prolonged Blood Potassium Decreased<br />
Head Injury<br />
Condition Aggravated<br />
Syncope<br />
Torsade De Pointes<br />
Electrocardiogram Qt<br />
Prolonged<br />
03-Apr-2012 09:37 AM Page: 286
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/20/00ISR Number: 3633799-2Report Type:Expedited (15-DaCompany Report #250469 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Lariam Tablets PS Roche REPORTED THE<br />
Emotional Disorder<br />
PATIENT TOOK<br />
Pain In Extremity<br />
THREE LARIAM<br />
Neurological Symptom<br />
TABLETS.<br />
Date:12/20/00ISR Number: 3633802-XReport Type:Expedited (15-DaCompany Report #251020 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myositis Lariam Tablets PS Roche<br />
Initial or Prolonged Phlebitis Contraceptive Pill C<br />
Date:12/20/00ISR Number: 3634802-6Report Type:Expedited (15-DaCompany Report #250403 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Ventricular Fibrillation Foreign Lariam PS Hoffmann La Roche<br />
Hospitalization - Torsade De Pointes Health Inc ORAL 250 MG 1 PER<br />
Initial or Prolonged Condition Aggravated Professional WEEK ORAL<br />
Electrocardiogram Qt Other Moduretic (Amiloride<br />
Prolonged<br />
Hydrochloride/Hydroc<br />
Heart Disease Congenital hlorothiazide) C<br />
Bradycardia<br />
Blood Potassium Decreased<br />
Syncope<br />
Head Injury<br />
Date:12/20/00ISR Number: 3635602-3Report Type:Expedited (15-DaCompany Report #250670 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Personality Change Foreign Lariam PS Hoffmann La Roche<br />
Agitation Health Inc<br />
Professional<br />
Antimalarial Nos<br />
(Antimalarial Agent<br />
Nos)<br />
SS<br />
Triludan<br />
(Terfenadine)<br />
SS<br />
Date:12/21/00ISR Number: 3634463-6Report Type:Expedited (15-DaCompany Report #910100239001 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Depression Lariam Tablets PS Roche 15 DAY<br />
Hospitalization - Nervousness<br />
Initial or Prolonged Tremor
Disability<br />
Dysthymic Disorder<br />
Suicide Attempt<br />
03-Apr-2012 09:37 AM Page: 287
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/22/00ISR Number: 3635906-4Report Type:Expedited (15-DaCompany Report #251020 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Swelling Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pain Health Inc ORAL 250MG, 1 PER<br />
Erythema Professional WEEK ORAL<br />
Phlebitis<br />
Myositis<br />
Contraceptive Pill<br />
(Oral Contraceptive<br />
Nos)<br />
C<br />
Date:12/22/00ISR Number: 3636105-2Report Type:Expedited (15-DaCompany Report #250469 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Health Lariam PS Hoffmann La Roche<br />
Pain In Extremity Professional Inc ORAL ORAL<br />
Neurological Symptom<br />
Date:12/22/00ISR Number: 3637192-8Report Type:Periodic Company Report #A031374 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Consumer Zithromax PS Pfizer Chemicals Div<br />
Vertigo Pfizer Inc ORAL 500.00 MG<br />
TOTAL: ORAL<br />
Lariam<br />
SS<br />
Date:12/26/00ISR Number: 3638713-1Report Type:Expedited (15-DaCompany Report #910100239001 Age:20 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Depression Health Lariam PS Hoffmann La Roche<br />
Hospitalization - Tremor Professional Inc ORAL 1 DOSE FORM 1<br />
Initial or Prolonged Condition Aggravated WEEK ORAL<br />
Disability<br />
Nervousness<br />
Obsessive-Compulsive<br />
Disorder<br />
Suicidal Ideation<br />
Agitation<br />
Dysthymic Disorder<br />
Suicide Attempt<br />
Date:01/02/01ISR Number: 3639580-2Report Type:Expedited (15-DaCompany Report #247262 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Health Lariam Tablets PS Roche 46 DAY<br />
Initial or Prolonged Spinal Fracture Professional Prozac I<br />
Suicide Attempt Xanax I
Depression<br />
03-Apr-2012 09:37 AM Page: 288
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/02/01ISR Number: 3639591-7Report Type:Expedited (15-DaCompany Report #247262 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Spinal Fracture Health Lariam Tablets PS Roche 46 DAY<br />
Initial or Prolonged Depression Professional Prozac C<br />
Suicide Attempt Xanax C<br />
Date:01/02/01ISR Number: 3639595-4Report Type:Expedited (15-DaCompany Report #251020 Age:28 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myositis Health Lariam Tablets PS Roche<br />
Initial or Prolonged Phlebitis Professional Contraceptive Pill C<br />
Vasculitis<br />
Date:01/02/01ISR Number: 3639605-4Report Type:Expedited (15-DaCompany Report #251435 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sleep Disorder Lariam Tablets PS Roche 750 MG AND<br />
Headache<br />
THEN SIX<br />
Tremor<br />
HOURS LATER<br />
Dizziness 500 MG. 1 DAY<br />
Stupor<br />
Flushing<br />
Vomiting<br />
Date:01/02/01ISR Number: 3639613-3Report Type:Expedited (15-DaCompany Report #251577 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Epilepsy Lariam Tablets PS Roche 18 DAY<br />
Initial or Prolonged Confusional State<br />
Date:01/02/01ISR Number: 3639619-4Report Type:Expedited (15-DaCompany Report #251466 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Lariam Tablets PS Roche PATIENT<br />
Diarrhoea<br />
RECEIVED 1 X<br />
Decreased Appetite 750 MG AND 6<br />
Vomiting HOURS LATER 1<br />
Anxiety X 500 MG. 1 DAY<br />
Shock<br />
Dizziness<br />
Date:01/02/01ISR Number: 3640343-2Report Type:Direct Company Report # Age:53 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam 250 Mg PS ORAL 1 PER 7 WEEKS<br />
Required<br />
ORAL<br />
Intervention to Lariam SS ORAL 1 PER 2 WEEKS<br />
Prevent Permanent<br />
ORAL<br />
Impairment/Damage Benzodiazapines C<br />
Paxil<br />
C<br />
03-Apr-2012 09:37 AM Page: 289
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sleeping Aids<br />
C<br />
Date:01/03/01ISR Number: 3641446-9Report Type:Expedited (15-DaCompany Report #251577 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Epilepsy Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Confusional State Health Inc ORAL ORAL<br />
Professional<br />
Date:01/03/01ISR Number: 3641477-9Report Type:Expedited (15-DaCompany Report #247262 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fracture Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Drug Interaction Health Inc ORAL 1 DOSE FORM<br />
Suicide Attempt Professional 1 PER 1 WEEK<br />
Depression<br />
ORAL<br />
Prozac (Fluoxetine<br />
Hydrochloride) SS ORAL 1 DOSE FORM<br />
DAILY ORAL<br />
Xanax (Alprazolam)<br />
0.25 Mg SS ORAL 0.5 MG DAILY<br />
ORAL<br />
Date:01/03/01ISR Number: 3641530-XReport Type:Expedited (15-DaCompany Report #247262 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicide Attempt Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Spinal Fracture Health Inc ORAL 1 DOSE FORM 1<br />
Depression Professional PER WEEK<br />
ORAL<br />
Prozac (Fluoxetine<br />
Hydrochloride) C<br />
Xanax (Alprazolam) C<br />
Date:01/03/01ISR Number: 3641657-2Report Type:Expedited (15-DaCompany Report #251466 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Foreign Lariam PS Hoffmann La Roche<br />
Diarrhoea Health Inc ORAL 1250 MG,<br />
Shock Professional DAILY, ORAL<br />
Anxiety<br />
Decreased Appetite<br />
Vomiting<br />
Dizziness
Date:01/03/01ISR Number: 3641658-4Report Type:Expedited (15-DaCompany Report #251435 Age:36 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Vomiting<br />
Dizziness<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 290
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Headache<br />
Stupor<br />
Flushing Report Source Product Role Manufacturer Route Dose Duration<br />
Sleep Disorder Foreign Lariam PS Hoffmann La Roche<br />
Tremor Other Inc ORAL 1250 MG<br />
DAILY, ORAL<br />
Date:01/03/01ISR Number: 3641659-6Report Type:Expedited (15-DaCompany Report #251020 Age:28 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vasculitis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Myositis Health Inc ORAL 250 MG, 1 PER<br />
Phlebitis Professional WEEK, ORAL<br />
Contraceptive Pill<br />
(Oral Contraceptive<br />
Nos)<br />
C<br />
Date:01/04/01ISR Number: 3640928-3Report Type:Expedited (15-DaCompany Report #251435 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sleep Disorder Consumer Lariam Tablets PS Roche 750 MG AND<br />
Flushing<br />
THEN SIX<br />
Tremor<br />
HOURS LATER<br />
Nausea 500 MG. 1 DAY<br />
Dizziness<br />
Headache<br />
Stupor<br />
Vomiting<br />
Date:01/05/01ISR Number: 3642323-XReport Type:Expedited (15-DaCompany Report #249929 Age:38 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Face Oedema<br />
Depression<br />
Road Traffic Accident<br />
Jaundice<br />
Gastritis<br />
Thinking Abnormal<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Diarrhoea<br />
Urticaria<br />
Weight Decreased<br />
Insomnia<br />
Malaise<br />
Constipation<br />
Drug Hypersensitivity
Respiratory Rate<br />
Increased<br />
Hepatomegaly<br />
Haematemesis<br />
Haemoglobin Decreased<br />
Liver Tenderness<br />
Decreased Appetite<br />
Abnormal Dreams<br />
03-Apr-2012 09:37 AM Page: 291
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fatigue<br />
Chills<br />
Feeling Hot Report Source Product Role Manufacturer Route Dose Duration<br />
Night Sweats Lariam Tablets PS Roche 32 DAY<br />
Heart Rate Increased<br />
Urinary Tract Infection<br />
Date:01/05/01ISR Number: 3642795-0Report Type:Expedited (15-DaCompany Report #251435 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Stupor Foreign Lariam PS Hoffmann La Roche<br />
Flushing Other Inc ORAL 1250 MG DAILY<br />
Dizziness<br />
ORAL<br />
Headache<br />
Sleep Disorder<br />
Vomiting<br />
Tremor<br />
Date:01/08/01ISR Number: 3642763-9Report Type:Expedited (15-DaCompany Report #251768 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Abortion Induced Lariam Tablets PS Roche<br />
Pregnancy<br />
Maternal Drugs Affecting<br />
Foetus<br />
Limb Malformation<br />
Date:01/08/01ISR Number: 3643675-7Report Type:Expedited (15-DaCompany Report #249929 Age:38 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT<br />
Haemoglobin Decreased<br />
Abnormal Dreams<br />
Disturbance In Attention<br />
Depression<br />
Feeling Hot<br />
Respiratory Rate<br />
Increased<br />
Blister<br />
Hallucination, Visual<br />
Night Sweats<br />
Urticaria<br />
Hepatomegaly<br />
Liver Disorder<br />
Heart Rate Increased<br />
Body Temperature<br />
Increased<br />
Thinking Abnormal
Diarrhoea<br />
Haematemesis<br />
Insomnia<br />
Chills<br />
Face Oedema<br />
Gastritis<br />
Malaise<br />
Urinary Tract Infection<br />
03-Apr-2012 09:37 AM Page: 292
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Constipation<br />
Road Traffic Accident<br />
Decreased Appetite Report Source Product Role Manufacturer Route Dose Duration<br />
Rash Pruritic Foreign Lariam PS Hoffmann La Roche<br />
Consumer Inc ORAL 250 MG 1 PER<br />
Other<br />
WEEK ORAL<br />
Date:01/09/01ISR Number: 3643443-6Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Health Lariam Tablets PS Roche 280 DAY<br />
Gait Disturbance<br />
Professional<br />
Date:01/09/01ISR Number: 3643457-6Report Type:Expedited (15-DaCompany Report #247733 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Field Defect Health Lariam Tablets PS Roche THE PATIENT<br />
Pituitary Tumour Benign Professional RECEIVED A<br />
TOTAL OF 2<br />
TABLETS. 16 DAY<br />
Date:01/09/01ISR Number: 3644647-9Report Type:Expedited (15-DaCompany Report #251768 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam PS Hoffmann La Roche<br />
Foetus Health Inc ORAL 1 DOSE FORM<br />
Foetal Disorder Professional 1 PER WEEK<br />
Pregnancy<br />
ORAL<br />
Abortion Induced<br />
Limb Reduction Defect<br />
Date:01/10/01ISR Number: 3645682-7Report Type:Expedited (15-DaCompany Report #247733 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pituitary Tumour Benign Foreign Lariam PS Hoffmann La Roche<br />
Visual Field Defect Health Inc ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:01/10/01ISR Number: 3645686-4Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Decreased Activity Health Lariam PS Hoffmann La Roche<br />
Psoriasis Professional Inc ORAL 1 PER WEEK
03-Apr-2012 09:37 AM Page: 293<br />
ORAL
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/11/01ISR Number: 3645661-XReport Type:Expedited (15-DaCompany Report #251020 Age:28 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Phlebitis Health Lariam Tablets PS Roche<br />
Initial or Prolonged Myositis Professional Contraceptive Pill C<br />
Vasculitis<br />
Date:01/12/01ISR Number: 3647272-9Report Type:Expedited (15-DaCompany Report #251020 Age:28 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vasculitis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Myositis Health Inc ORAL 250 MG 1 PER<br />
Phlebitis Professional WEEK ORAL<br />
Contraceptive Pill<br />
(Oral Contraceptive<br />
Nos)<br />
C<br />
Date:01/16/01ISR Number: 3647463-7Report Type:Expedited (15-DaCompany Report #252294 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Back Pain Lariam Tablets PS Roche 43 DAY<br />
Prostatic Specific Multivitamins C<br />
Antigen Increased<br />
Dysuria<br />
Haematuria<br />
Date:01/16/01ISR Number: 3647467-4Report Type:Expedited (15-DaCompany Report #234319 Age:20 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Lariam Tablets PS Roche 112 DAY<br />
Anxiety<br />
Paresis<br />
Hallucination<br />
Illusion<br />
Alopecia<br />
Sensory Disturbance<br />
Sleep Disorder<br />
Delusion<br />
Mental Disorder<br />
Nightmare<br />
Date:01/16/01ISR Number: 3647469-8Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Lariam Tablets PS Roche TWO DOSES
Initial or Prolonged Hyperventilation TAKEN. 9 DAY<br />
Loss Of Consciousness<br />
Convulsion<br />
Dyspnoea<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 294
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/17/01ISR Number: 3648112-4Report Type:Direct Company Report # Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Face Oedema <strong>Mefloquine</strong> PS ORAL 1 TAB Q 7<br />
Intervention to Urticaria DAYS P.O.;<br />
Prevent Permanent Eyelid Oedema FROM ONSET OF<br />
Impairment/Damage Joint Swelling RASH FOR 10<br />
Pruritus DAYS 10 DAY<br />
Anaphylactoid Reaction<br />
Date:01/17/01ISR Number: 3649402-1Report Type:Expedited (15-DaCompany Report #234319 Age:20 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Foreign Lariam PS Hoffmann La Roche<br />
Disturbance In Attention Health Inc ORAL 250 MG 1 PER<br />
Delusion Professional WEEK ORAL<br />
Illusion<br />
Other<br />
Alopecia<br />
Sensory Disturbance<br />
Nightmare<br />
Anxiety<br />
Mental Disorder<br />
Paresis<br />
Sleep Disorder<br />
Date:01/17/01ISR Number: 3649403-3Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pyrexia Health Inc ORAL 1 PER 1 WEEK<br />
Hyperventilation Professional ORAL<br />
Tetany<br />
Dyspnoea<br />
Convulsion<br />
Loss Of Consciousness<br />
Date:01/17/01ISR Number: 3649448-3Report Type:Expedited (15-DaCompany Report #252294 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Prostatic Specific Consumer Lariam PS Hoffmann La Roche<br />
Antigen Increased Inc ORAL 250 MG 1 PER<br />
Dysuria<br />
WEEK ORAL<br />
Haematuria<br />
Multivitamins Nos<br />
Back Pain (Multivitamins Nos) C
Date:01/22/01ISR Number: 3651513-1Report Type:Expedited (15-DaCompany Report #252739 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Chest Pain Lariam Tablets PS Roche 54 DAY<br />
Hospitalization - Dyspnoea Climen C<br />
Initial or Prolonged Bradycardia<br />
03-Apr-2012 09:37 AM Page: 295
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/23/01ISR Number: 3652329-2Report Type:Expedited (15-DaCompany Report #252778 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Persecutory Delusion Lariam Tablets PS Roche 11 DAY<br />
Mood Altered Stamaril Unidose C 1 DAY<br />
Paranoia<br />
Suicide Attempt<br />
Hallucination, Auditory<br />
Insomnia<br />
Date:01/23/01ISR Number: 3652402-9Report Type:Direct Company Report # Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Medication Error <strong>Mefloquine</strong> PS<br />
Date:01/23/01ISR Number: 3653816-3Report Type:Expedited (15-DaCompany Report #252739 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Back Pain Foreign Lariam PS Hoffmann La Roche<br />
Hospitalization - Dyspnoea Health Inc ORAL ORAL<br />
Initial or Prolonged Chest Pain Professional Climen C<br />
Bradycardia<br />
Date:01/24/01ISR Number: 3653477-3Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Lariam Tablets PS Roche<br />
Delusion<br />
Date:01/24/01ISR Number: 3653480-3Report Type:Expedited (15-DaCompany Report #252779 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Uterine Leiomyoma Lariam Tablets PS Roche 29 DAY<br />
Gestational Diabetes<br />
Gonorrhoea<br />
Appendicitis<br />
Date:01/24/01ISR Number: 3654504-XReport Type:Expedited (15-DaCompany Report #252778 Age:35 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
PT<br />
Asthenia<br />
Nightmare<br />
Paranoia
Hallucination, Auditory<br />
Headache<br />
Psychotic Disorder<br />
Anxiety<br />
Sleep Disorder<br />
Dizziness<br />
Irritability<br />
Suicide Attempt<br />
03-Apr-2012 09:37 AM Page: 296
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Persecutory Delusion<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS Hoffmann La Roche<br />
Other Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Stamaril Unidose<br />
(Yellow Fever<br />
Vaccine)<br />
C<br />
Date:01/25/01ISR Number: 3654162-4Report Type:Expedited (15-DaCompany Report #249232 Age:22 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Affective Disorder Lariam Tablets PS Roche TOTAL DOSE OF<br />
Anxiety<br />
LARIAM TAKEN<br />
Diarrhoea<br />
BY THE<br />
Delusion PATIENT: 2<br />
Vision Blurred TABLETS. 8 DAY<br />
Depression<br />
Paranoia<br />
Asthenia<br />
Insomnia<br />
Nightmare<br />
Date:01/25/01ISR Number: 3655969-XReport Type:Expedited (15-DaCompany Report #252779 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Appendicitis Foreign Lariam PS Hoffmann La Roche<br />
Pregnancy Health Inc ORAL DOSE FORM 1<br />
Gestational Diabetes Professional PER WEEK ORAL<br />
Gonorrhoea<br />
Uterine Leiomyoma<br />
Date:01/25/01ISR Number: 3655974-3Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Foreign Lariam PS Hoffmann La Roche<br />
Delusion Health Inc ORAL 1 PER WEEK<br />
Professional<br />
ORAL<br />
Date:01/26/01ISR Number: 3654951-6Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Lariam Tablets PS Roche
Date:01/29/01ISR Number: 3656105-6Report Type:Expedited (15-DaCompany Report #235479 Age:76 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Corneal Oedema Lariam Tablets PS Roche<br />
03-Apr-2012 09:37 AM Page: 297
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/29/01ISR Number: 3656134-2Report Type:Expedited (15-DaCompany Report #252730 Age:70 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Increased Lariam Tablets PS Roche<br />
Ventricular Hypertrophy<br />
Date:01/29/01ISR Number: 3657437-8Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Foreign Lariam PS Hoffmann La Roche<br />
Abnormal Behaviour Health Inc ORAL 1 PER WEEK<br />
Professional<br />
ORAL<br />
Date:01/30/01ISR Number: 3657556-6Report Type:Expedited (15-DaCompany Report #252730 Age:70 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ventricular Hypertrophy Foreign Lariam PS Hoffmann La Roche<br />
Blood Pressure Increased Health Inc 250 MG 1 PER<br />
Professional<br />
WEEK<br />
Date:01/30/01ISR Number: 3659264-4Report Type:Expedited (15-DaCompany Report #235479 Age:76 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Corneal Oedema Health Lariam PS Hoffmann La Roche<br />
Corneal Transplant Professional Inc ORAL ORAL<br />
Date:01/31/01ISR Number: 3657664-XReport Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Lariam Tablets PS Roche TWO DOSES<br />
Initial or Prolonged Loss Of Consciousness TAKEN. 9 DAY<br />
Convulsion<br />
Mental Disorder<br />
Pyrexia<br />
Date:02/01/01ISR Number: 3659145-6Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Loss Of Consciousness Health Inc ORAL 1 PER 1 WEEK<br />
Mental Disorder Professional ORAL<br />
Tetany<br />
Dyspnoea
Hyperventilation<br />
03-Apr-2012 09:37 AM Page: 298
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/02/01ISR Number: 3658970-5Report Type:Expedited (15-DaCompany Report #249748 Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disorientation Lariam Tablets PS Roche<br />
Initial or Prolonged Delusion<br />
Hypertonia<br />
Persecutory Delusion<br />
Agitation<br />
Acute Psychosis<br />
Confusional State<br />
Speech Disorder<br />
Mutism<br />
Date:02/02/01ISR Number: 3658974-2Report Type:Expedited (15-DaCompany Report #253219 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Atrial Fibrillation Lariam Tablets PS Roche<br />
Date:02/02/01ISR Number: 3658987-0Report Type:Expedited (15-DaCompany Report #253380 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Blood Pressure Increased Lariam Tablets PS Roche 70 DAY<br />
Retinal Haemorrhage<br />
Unspecified<br />
Vision Blurred Immunizations C<br />
Date:02/02/01ISR Number: 3658988-2Report Type:Expedited (15-DaCompany Report #244551 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aplastic Anaemia Lariam Tablets PS Roche 29 DAY<br />
Date:02/02/01ISR Number: 3659017-7Report Type:Expedited (15-DaCompany Report #253574 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypoaesthesia Lariam Tablets PS Roche 57 DAY<br />
Hallucination<br />
Tinnitus<br />
Agoraphobia<br />
Paraesthesia<br />
Palpitations<br />
Paranoia<br />
Date:02/02/01ISR Number: 3659019-0Report Type:Expedited (15-DaCompany Report #253632 Age:53 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Clonic Convulsion Lariam Tablets PS Roche 33 DAY<br />
Hypercholesterolaemia<br />
03-Apr-2012 09:37 AM Page: 299
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/02/01ISR Number: 3660323-0Report Type:Expedited (15-DaCompany Report #249232 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Foreign Lariam PS Hoffmann La Roche<br />
Paranoia Consumer Inc ORAL 1 DOSE FORM 1<br />
Depression<br />
PER 1 WEEK<br />
Asthenia<br />
ORAL<br />
Affective Disorder<br />
Anxiety<br />
Nightmare<br />
Diarrhoea<br />
Delusion<br />
Vision Blurred<br />
Date:02/02/01ISR Number: 3660344-8Report Type:Expedited (15-DaCompany Report #249748 Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disorientation Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Persecutory Delusion Literature Inc<br />
Psychotic Disorder<br />
Health<br />
Agitation<br />
Professional<br />
Mutism<br />
Confusional State<br />
Hypertonia<br />
Pyrexia<br />
Speech Disorder<br />
Date:02/05/01ISR Number: 3659704-0Report Type:Direct Company Report # Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dry Eye Lariam PS ORAL 1 PER WEEK<br />
Fatigue<br />
ORAL<br />
Dry Mouth<br />
Feeling Abnormal<br />
Balance Disorder<br />
Tremor<br />
Nausea<br />
Date:02/05/01ISR Number: 3662891-1Report Type:Expedited (15-DaCompany Report #253574 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hallucination Foreign Lariam PS Hoffmann La Roche<br />
Nausea Other Inc ORAL DOSE FORM<br />
Paraesthesia<br />
ORAL<br />
Palpitations<br />
Dizziness<br />
Hypoaesthesia
Agoraphobia<br />
Visual Impairment<br />
Tinnitus<br />
Paranoia<br />
03-Apr-2012 09:37 AM Page: 300
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/05/01ISR Number: 3662893-5Report Type:Expedited (15-DaCompany Report #253380 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypertension Foreign Lariam PS Hoffmann La Roche<br />
Vision Blurred Consumer Inc ORAL ORAL 70 DAY<br />
Retinal Haemorrhage<br />
Unspecified<br />
Immunizations<br />
(Immunisation) C<br />
Date:02/05/01ISR Number: 3662894-7Report Type:Expedited (15-DaCompany Report #253632 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Cholesterol Foreign Lariam PS Hoffmann La Roche<br />
Increased Health Inc ORAL 250 MG 1 PER<br />
Clonic Convulsion Professional WEEK ORAL<br />
Date:02/06/01ISR Number: 3660174-7Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Lariam Tablets PS Roche ESTIMATED 5<br />
Initial or Prolonged Panic Attack DOSES IN<br />
Disability Depression TOTAL. 21 DAY<br />
Headache<br />
Date:02/07/01ISR Number: 3661122-6Report Type:Expedited (15-DaCompany Report #220210 Age:17 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Lariam Tablets PS Roche<br />
Initial or Prolonged Obesity<br />
Hypertension<br />
Chlamydial Infection<br />
Vision Blurred<br />
Visual Impairment<br />
Pupillary Disorder<br />
Measles Antibody Positive<br />
Antinuclear Antibody<br />
Positive<br />
Inflammation<br />
Drug Hypersensitivity<br />
Accommodation Disorder<br />
Folate Deficiency<br />
Binocular Eye Movement<br />
Disorder<br />
Smooth Muscle Antibody<br />
Positive<br />
Cytogenetic Abnormality<br />
Laboratory Test Abnormal
Diplopia<br />
Coagulopathy<br />
Platelet Count Decreased<br />
03-Apr-2012 09:37 AM Page: 301
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/07/01ISR Number: 3661137-8Report Type:Expedited (15-DaCompany Report #247340 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Vertigo Positional Lariam Tablets PS Roche FOR MANY<br />
Motor Dysfunction<br />
YEARS.<br />
Nightmare<br />
Dizziness<br />
Date:02/07/01ISR Number: 3661805-8Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Health Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Vertigo Professional Inc ORAL 250 MG 1 PER<br />
Disability Panic Attack WEEK ORAL<br />
Dizziness<br />
Headache<br />
Anxiety<br />
Date:02/08/01ISR Number: 3662801-7Report Type:Expedited (15-DaCompany Report #220210 Age:17 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Antinuclear Antibody Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Positive Consumer Inc ORAL 250 MG 1 PER<br />
Hypertension Health WEEK ORAL<br />
Visual Acuity Reduced<br />
Professional<br />
Vision Blurred<br />
Platelet Count Decreased<br />
Inflammation<br />
Headache<br />
Laboratory Test Abnormal<br />
Iris Disorder<br />
Diplopia<br />
Csf Test Abnormal<br />
Binocular Eye Movement<br />
Disorder<br />
Dna Antibody Positive<br />
Measles Antibody Positive<br />
Pupillary Reflex Impaired<br />
Chlamydial Infection<br />
Sinusitis<br />
Herpes Simplex<br />
Visual Impairment<br />
Folate Deficiency<br />
Infectious Mononucleosis<br />
Accommodation Disorder<br />
Herpes Zoster
Date:02/08/01ISR Number: 3662860-1Report Type:Expedited (15-DaCompany Report #247340 Age:42 YR Gender:Male I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Nightmare<br />
Gait Disturbance<br />
Vertigo Positional<br />
Vomiting<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 302
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Motor Dysfunction<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer Lariam PS Hoffmann La Roche<br />
Inc ORAL ORAL<br />
Date:02/09/01ISR Number: 3662550-5Report Type:Expedited (15-DaCompany Report #254219 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam Tablets PS Roche 49 DAY<br />
Suicidal Ideation<br />
Disturbance In Attention<br />
Sinusitis<br />
Hallucination<br />
Influenza Like Illness<br />
Abnormal Dreams<br />
Anxiety<br />
Insomnia<br />
Lethargy<br />
Date:02/12/01ISR Number: 3663406-4Report Type:Expedited (15-DaCompany Report #254186 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tongue Oedema Lariam Tablets PS Roche 42 DAY<br />
Angioedema Vit B12 C LAST<br />
Face Oedema<br />
INJECTION WAS<br />
02 JAN 2001.<br />
Zestoretic<br />
C<br />
Ranitidine<br />
C<br />
Date:02/12/01ISR Number: 3663685-3Report Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oropharyngeal Pain Larium PS<br />
Mood Swings<br />
Decreased Activity<br />
Nausea<br />
Pain In Extremity<br />
Rickettsiosis<br />
Insomnia<br />
Sleep Attacks<br />
Fatigue<br />
Fibromyalgia<br />
Arthralgia
Date:02/12/01ISR Number: 3664102-XReport Type:Expedited (15-DaCompany Report #254219 Age: Gender:Male I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Sinusitis<br />
Anxiety<br />
Suicidal Ideation<br />
Depression<br />
Hallucination<br />
03-Apr-2012 09:37 AM Page: 303
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Insomnia<br />
Disturbance In Attention<br />
Influenza Like Illness Report Source Product Role Manufacturer Route Dose Duration<br />
Abnormal Dreams Consumer Lariam PS Hoffmann La Roche<br />
Lethargy Inc ORAL ORAL<br />
Date:02/13/01ISR Number: 3664562-4Report Type:Expedited (15-DaCompany Report #254226 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Inborn Error Of Lariam Tablets PS Roche<br />
Initial or Prolonged Metabolism<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Biopsy Bronchus Abnormal<br />
Haemoglobin Decreased<br />
Lung Disorder<br />
Haematocrit Decreased<br />
White Blood Cell Count<br />
Increased<br />
Pneumonitis<br />
Lymphocyte Count<br />
Decreased<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Neutrophil Count<br />
Increased<br />
Malaise<br />
Laboratory Test Abnormal<br />
Date:02/13/01ISR Number: 3666432-4Report Type:Expedited (15-DaCompany Report #254186 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Angioedema Foreign Lariam Tablets<br />
Intervention to Face Oedema Health (<strong>Mefloquine</strong><br />
Prevent Permanent Tongue Oedema Professional Hydrochloride) 250<br />
Impairment/Damage Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL 42 DAY<br />
Vit B12<br />
(Cyanocobalamin) C<br />
Zestoretic<br />
(Hydrochlorothiazide<br />
/Lisinopril)<br />
C<br />
Ranitidine<br />
(Ranitidine)<br />
C<br />
Date:02/14/01ISR Number: 3664745-3Report Type:Expedited (15-DaCompany Report #254230 Age:68 YR Gender:Male I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Nausea<br />
Asthenia<br />
Cardiac Murmur<br />
Venous Pressure Jugular<br />
Increased<br />
Hepatomegaly<br />
Blood Urea Increased<br />
03-Apr-2012 09:37 AM Page: 304
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Red Blood Cell<br />
Sedimentation Rate<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Hepatitis<br />
<strong>Mefloquine</strong><br />
Decreased Appetite Hydrochloride PS Roche 42 DAY<br />
Atrial Flutter Allopurinol SS Roche<br />
Fatigue Spironolactone C<br />
Blood Creatinine Warfarin C<br />
Increased Propafenone C<br />
Jaundice Digoxin C<br />
Chest X-Ray Abnormal Furosemide C<br />
Electrocardiogram<br />
Abnormal<br />
Dehydration<br />
Condition Aggravated<br />
Date:02/14/01ISR Number: 3667666-5Report Type:Expedited (15-DaCompany Report #254226 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pneumonia Foreign Lariam Tablets<br />
Initial or Prolonged Inborn Error Of Literature (<strong>Mefloquine</strong><br />
Metabolism Health Hydrochloride) 250<br />
Lung Disorder Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:02/15/01ISR Number: 3665573-5Report Type:Expedited (15-DaCompany Report #249135 Age:61 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Respiratory Arrest Lariam Tablets PS Roche<br />
Hospitalization - Loss Of Consciousness Ibuprofen C 20 DAY<br />
Initial or Prolonged Condition Aggravated Doxycyclin C 20 DAY<br />
Electroencephalogram<br />
Abnormal<br />
Pulse Absent<br />
Ventricular Extrasystoles<br />
Hypertension<br />
Echocardiogram Abnormal<br />
Electrocardiogram T Wave<br />
Amplitude Decreased<br />
Convulsion<br />
Electrocardiogram St<br />
Segment Depression<br />
Supraventricular<br />
Extrasystoles<br />
Date:02/15/01ISR Number: 3665580-2Report Type:Expedited (15-DaCompany Report #254480 Age: Gender:Not SpecifiI/FU:I
Outcome<br />
Congenital Anomaly<br />
PT<br />
Dysmorphism<br />
Skull Malformation<br />
Congenital Foot<br />
Malformation<br />
Cleft Palate<br />
Congenital Anomaly<br />
Syndactyly<br />
03-Apr-2012 09:37 AM Page: 305
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Low Set Ears<br />
Pregnancy<br />
Maternal Drugs Affecting Report Source Product Role Manufacturer Route Dose Duration<br />
Foetus Lariam Tablets PS Roche 28 DAY<br />
Abortion Induced<br />
Yellow Fever Vaccine C<br />
Congenital Eye Disorder<br />
Thymus Disorder<br />
Congenital Central<br />
Nervous System Anomaly<br />
Date:02/15/01ISR Number: 3666602-5Report Type:Expedited (15-DaCompany Report #254230 Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Foreign <strong>Mefloquine</strong><br />
Initial or Prolonged Venous Pressure Jugular Literature Hydrochloride<br />
Increased Health (<strong>Mefloquine</strong><br />
Asthenia Professional Hydrochloride) PS 250 MG 1 PER<br />
Dehydration WEEK 42 DAY<br />
Blood Urea Increased<br />
Allopurinol<br />
Nausea (Allopurinol) SS 100 MG DAILY<br />
International Normalised<br />
Warfarin (Warfarin<br />
Ratio Increased Sodium) C<br />
White Blood Cell Count<br />
Propafenone<br />
Increased<br />
(Propafenone<br />
Hepatotoxicity Hydrochloride) C<br />
Decreased Appetite<br />
Spironolactone<br />
Cardiac Murmur (Spironolactone) C<br />
Platelet Count Decreased<br />
Chest X-Ray Abnormal<br />
Hepatomegaly<br />
Atrial Flutter<br />
Red Blood Cell<br />
Sedimentation Rate<br />
Increased<br />
Hepatitis<br />
Date:02/16/01ISR Number: 3668044-5Report Type:Expedited (15-DaCompany Report #254480 Age: Gender:Unknown I/FU:I<br />
Outcome<br />
Congenital Anomaly<br />
PT<br />
Thymus Disorder<br />
Congenital Jaw<br />
Malformation<br />
Congenital Nose<br />
Malformation<br />
Abortion Induced<br />
Cleft Palate<br />
Dysmorphism<br />
Maternal Drugs Affecting<br />
Foetus
Skull Malformation<br />
Congenital Foot<br />
Malformation<br />
Multiple Congenital<br />
Abnormalities<br />
Congenital Central<br />
Nervous System Anomaly<br />
Congenital Eye Disorder<br />
03-Apr-2012 09:37 AM Page: 306
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Syndactyly<br />
Ear Malformation<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign Lariam PS Hoffmann La Roche<br />
Other Inc ORAL 250 MG 1 PER<br />
WEEK ORAL 28 DAY<br />
Yellow Fever Vaccine<br />
(Yellow Fever<br />
Vaccine)<br />
C<br />
Date:02/16/01ISR Number: 3668050-0Report Type:Expedited (15-DaCompany Report #249135 Age:61 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Convulsive Threshold Foreign Lariam PS Hoffmann La Roche<br />
Hospitalization - Lowered Other Inc ORAL 250 MG 1 PER<br />
Initial or Prolonged Respiratory Arrest WEEK ORAL<br />
Supraventricular<br />
Ibuprofen<br />
Extrasystoles (Ibuprofen) C<br />
Coronary Artery Disease<br />
Doxycyclin<br />
Convulsion (Doxycycline) C<br />
Myocardial Ischaemia<br />
Circulatory Collapse<br />
Cardiac Disorder<br />
Ventricular Extrasystoles<br />
Hypertension<br />
Date:02/20/01ISR Number: 3666693-1Report Type:Expedited (15-DaCompany Report #220210 Age:17 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Dysgraphia<br />
Reading Disorder<br />
Accommodation Disorder<br />
Visual Impairment<br />
Diplopia<br />
Drug Hypersensitivity<br />
Thrombocytosis<br />
Lipids Increased<br />
Antibody Test Positive<br />
Binocular Eye Movement<br />
Disorder<br />
Folate Deficiency<br />
Smooth Muscle Antibody<br />
Positive<br />
Csf Test Abnormal<br />
Obesity<br />
Dna Antibody Positive<br />
Nuclear Magnetic<br />
Resonance Imaging Brain<br />
Abnormal
Visual Evoked Potentials<br />
Abnormal<br />
Hypertension<br />
Laboratory Test Abnormal<br />
Pupillary Disorder<br />
Measles Antibody Positive<br />
Headache<br />
Inflammation<br />
03-Apr-2012 09:37 AM Page: 307
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Viral Infection<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche<br />
Date:02/20/01ISR Number: 3667169-8Report Type:Direct Company Report # Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Lariam PS ORAL 1 A WEEK ORAL<br />
Panic Attack<br />
Muscular Weakness<br />
Feeling Of Body<br />
Temperature Change<br />
Vision Blurred<br />
Balance Disorder<br />
Date:02/20/01ISR Number: 3669532-8Report Type:Expedited (15-DaCompany Report #254595 Age:54 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dermatomyositis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 250 MG 1 PER<br />
Disability Professional WEEK ORAL<br />
Date:02/21/01ISR Number: 3667469-1Report Type:Expedited (15-DaCompany Report #254186 Age:50 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Angioedema Health Lariam Tablets PS Roche<br />
Professional Ranitidine SS<br />
Vit B12 C LAST<br />
INJECTION WAS<br />
02 JAN 2001.<br />
Zestoretic<br />
C<br />
Date:02/21/01ISR Number: 3669477-3Report Type:Expedited (15-DaCompany Report #220210 Age:17 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Inflammation<br />
Diplopia<br />
Infectious Mononucleosis<br />
Apolipoprotein A-I<br />
Increased<br />
Vision Blurred<br />
Iris Disorder<br />
Binocular Eye Movement<br />
Disorder
Folate Deficiency<br />
Blood Immunoglobulin G<br />
Increased<br />
Accommodation Disorder<br />
Herpes Zoster<br />
Hypertension<br />
Headache<br />
Dysgraphia<br />
03-Apr-2012 09:37 AM Page: 308
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Viral Infection<br />
Chlamydial Infection<br />
Thrombocytosis Report Source Product Role Manufacturer Route Dose Duration<br />
Reading Disorder Foreign Lariam PS Hoffmann La Roche<br />
Geniculate Ganglionitis Consumer Inc ORAL 250 MG 1 PER<br />
Health<br />
WEEK ORAL<br />
Professional<br />
Date:02/22/01ISR Number: 3669931-4Report Type:Expedited (15-DaCompany Report #254186 Age:50 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Angioedema Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Health Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Professional WEEK ORAL<br />
Impairment/Damage<br />
Ranitidine<br />
(Ranitidine) SS ORAL ORAL<br />
Vit B12<br />
(Cyanocobalamin) C<br />
Zestoretic<br />
(Hydrohlorothiazide/<br />
Lisnopril)<br />
C<br />
Date:02/26/01ISR Number: 3669114-8Report Type:Expedited (15-DaCompany Report #252730 Age:70 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Increased Lariam Tablets PS Roche 9 DAY<br />
Ventricular Hypertrophy<br />
Date:02/27/01ISR Number: 3670332-3Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
PT<br />
Amnesia<br />
Flushing<br />
Hepatic Steatosis<br />
Agitation<br />
Weight Decreased<br />
Cardiomegaly<br />
Nightmare<br />
Chills<br />
Depression<br />
Adjustment Disorder<br />
Obsessive-Compulsive<br />
Disorder<br />
Urinary Hesitation<br />
Delusional Disorder,<br />
Persecutory Type<br />
Decreased Appetite
Nervous System Disorder<br />
Musculoskeletal Stiffness<br />
Polydipsia<br />
Ventricular Hypertrophy<br />
Insomnia<br />
Abnormal Dreams<br />
Completed Suicide<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 309
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucinations, Mixed<br />
Headache<br />
Hyperhidrosis Report Source Product Role Manufacturer Route Dose Duration<br />
Delirium Consumer Lariam Tablets PS Roche 50 DAY<br />
Cognitive Disorder<br />
Arteriosclerosis<br />
Date:02/27/01ISR Number: 3671565-2Report Type:Expedited (15-DaCompany Report #252730 Age:70 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypertension Foreign Lariam PS Hoffmann La Roche<br />
Ventricular Hypertrophy Health Inc ORAL 250 MG 1 PER<br />
Professional<br />
WEEK ORAL<br />
Date:02/28/01ISR Number: 3671793-6Report Type:Expedited (15-DaCompany Report #200111100GDS Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Interaction Foreign Avelox PS Bayer Corp<br />
Hypertension Other Pharmaceutical Div ORAL 400 MG DAILY<br />
Heart Rate Increased<br />
ORAL<br />
<strong>Mefloquine</strong> SS ORAL 250 MG DAILY<br />
ORAL<br />
Altace<br />
C<br />
Moduret<br />
C<br />
Date:02/28/01ISR Number: 3671960-1Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
PT<br />
Depression<br />
Hyperhidrosis<br />
Arterial Injury<br />
Hallucinations, Mixed<br />
Musculoskeletal Stiffness<br />
Hepatic Steatosis<br />
Staring<br />
Agitation<br />
Lethargy<br />
Spinal Fracture<br />
Subarachnoid Haemorrhage<br />
Abnormal Behaviour<br />
Psychotic Disorder<br />
Decreased Appetite<br />
Polydipsia<br />
Flushing<br />
Urinary Hesitation<br />
Delusional Disorder,<br />
Persecutory Type
Hepatic Necrosis<br />
Insomnia<br />
Delirium<br />
Obsessive-Compulsive<br />
Disorder<br />
Abnormal Dreams<br />
Amnesia<br />
Cardiomegaly<br />
03-Apr-2012 09:37 AM Page: 310
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Ventricular Hypertrophy<br />
Arteriosclerosis<br />
Respiratory Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Injury Consumer Lariam PS Hoffmann La Roche<br />
Chills Other Inc ORAL 250 MG 1 PER<br />
Anxiety<br />
1 WEEK ORAL<br />
Cognitive Disorder<br />
Weight Decreased<br />
Headache<br />
Fatigue<br />
Completed Suicide<br />
Nervous System Disorder<br />
Nightmare<br />
Adjustment Disorder<br />
Date:03/01/01ISR Number: 3672141-8Report Type:Direct Company Report # Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Lariam / 250 Mg PS ORAL 1 TABLET<br />
Initial or Prolonged Dry Mouth WEEKLY ORAL<br />
Other Serious<br />
Anxiety<br />
Dyspnoea<br />
Pain<br />
Fatigue<br />
Tinnitus<br />
Insomnia<br />
Migraine<br />
Visual Impairment<br />
Muscular Weakness<br />
Headache<br />
Coordination Abnormal<br />
Heart Rate Increased<br />
Date:03/07/01ISR Number: 3674831-XReport Type:Expedited (15-DaCompany Report #255461 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dystonia Lariam Tablets PS Roche<br />
Initial or Prolonged Contraceptive Pill C<br />
Date:03/07/01ISR Number: 3674843-6Report Type:Expedited (15-DaCompany Report #250525 Age:14 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Speech Disorder<br />
Gastroenteritis<br />
Insomnia<br />
Back Pain<br />
Sedation
Memory Impairment<br />
Psychiatric Symptom<br />
Nightmare<br />
Renal Failure<br />
Irritability<br />
Laboratory Test Abnormal<br />
Learning Disorder<br />
Eczema<br />
03-Apr-2012 09:37 AM Page: 311
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diarrhoea<br />
Alpha 2 Globulin<br />
Decreased Report Source Product Role Manufacturer Route Dose Duration<br />
Educational Problem Lariam Tablets PS Roche THE PATIENT<br />
Hypogammaglobulinaemia RECEIVED 8<br />
Crying<br />
TABLETS IN<br />
Asthenia TOTAL. 50 DAY<br />
Blood Albumin Increased<br />
Alpha 1 Globulin<br />
Decreased<br />
Fatigue<br />
Condition Aggravated<br />
Protein Total Increased<br />
Anxiety<br />
Bronchitis<br />
Herpes Virus Infection<br />
Weight Decreased<br />
Date:03/07/01ISR Number: 3675235-6Report Type:Direct Company Report # Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dizziness Lariam<br />
Hospitalization - Anxiety Hoffman/Laroche PS Hoffman / Larouche ORAL ONE PILL<br />
Initial or Prolonged Suicidal Ideation ONCE WEEKLY<br />
Disability Diarrhoea ORAL<br />
Required<br />
Panic Reaction<br />
Intervention to<br />
Hallucinations, Mixed<br />
Prevent Permanent<br />
Impairment/Damage<br />
Nightmare<br />
Crying<br />
Insomnia<br />
Fear<br />
Decreased Activity<br />
Date:03/08/01ISR Number: 3676534-4Report Type:Expedited (15-DaCompany Report #250525 Age:14 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Back Pain Foreign Lariam PS Hoffmann La Roche<br />
Weight Decreased Health Inc ORAL 1 DOSE FORM<br />
Bronchitis Professional 1 PER WEEK<br />
Nightmare<br />
ORAL<br />
Fatigue<br />
Herpes Virus Infection<br />
Gastroenteritis<br />
Eczema<br />
Renal Failure<br />
Irritability<br />
Anxiety<br />
Asthenia<br />
Insomnia
Pyrexia<br />
Crying<br />
Psychiatric Symptom<br />
03-Apr-2012 09:37 AM Page: 312
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/08/01ISR Number: 3676700-8Report Type:Expedited (15-DaCompany Report #200111100GDS Age:46 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Foreign Avelox PS Bayer Corp<br />
Drug Interaction Health Pharmaceutical Div ORAL 400 MG DAILY<br />
Vomiting Professional ORAL<br />
Headache Other <strong>Mefloquine</strong> SS ORAL 250 MG DAILY<br />
Hypertension<br />
ORAL<br />
Heart Rate Increased Altace C<br />
Sensation Of Pressure Moduret C<br />
Dry Mouth<br />
Date:03/08/01ISR Number: 3676818-XReport Type:Expedited (15-DaCompany Report #255461 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dystonia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Other Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Contraceptive Pill<br />
(Oral Contraceptive<br />
Nos)<br />
C<br />
Date:03/09/01ISR Number: 3676793-8Report Type:Expedited (15-DaCompany Report #255903 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hallucination, Visual Lariam Tablets PS Roche 29 DAY<br />
Panic Reaction<br />
Amnesia<br />
Nightmare<br />
Insomnia<br />
Headache<br />
Date:03/09/01ISR Number: 3676802-6Report Type:Expedited (15-DaCompany Report #255897 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Lariam Tablets PS Roche 27 DAY<br />
Ethambutol C 154 DAY<br />
Pirazinamide C 154 DAY<br />
Date:03/12/01ISR Number: 3678135-0Report Type:Expedited (15-DaCompany Report #254595 Age:54 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pain Lariam Tablets PS Roche 32 DAY<br />
Initial or Prolonged Blood Creatinine<br />
Disability<br />
Increased
Myalgia<br />
Dermatomyositis<br />
Malaise<br />
Dermatitis<br />
03-Apr-2012 09:37 AM Page: 313
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/12/01ISR Number: 3680792-XReport Type:Expedited (15-DaCompany Report #255903 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Headache Foreign Lariam PS Hoffmann La Roche<br />
Amnesia Other Inc ORAL 250 MG 1 PER<br />
Insomnia<br />
WEEK ORAL<br />
Panic Reaction<br />
Nightmare<br />
Hallucination<br />
Date:03/12/01ISR Number: 3680802-XReport Type:Expedited (15-DaCompany Report #255897 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Jaundice Foreign Lariam PS Hoffmann La Roche<br />
Hepatitis Other Inc ORAL 250 MG DAILY<br />
ORAL<br />
Ethambutol<br />
(Ethambutol<br />
Hydrochloride) C<br />
Pirazinamide<br />
(Pyrazinamide) C<br />
Date:03/13/01ISR Number: 3680675-5Report Type:Expedited (15-DaCompany Report #254595 Age:54 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dermatomyositis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Malaise Health Inc ORAL 250 MG 1 PER<br />
Disability Dermatitis Professional WEEK ORAL<br />
Blood Creatinine<br />
Increased<br />
Myalgia<br />
Date:03/14/01ISR Number: 3680020-5Report Type:Expedited (15-DaCompany Report #253034 Age:49 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Erythema Nodosum Lariam Tablets PS Roche 33 DAY<br />
Initial or Prolonged Pyrexia<br />
Date:03/15/01ISR Number: 3681154-1Report Type:Direct Company Report # Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucinations, Mixed <strong>Mefloquine</strong> PS ORAL PO ( 3 TABS<br />
Initial or Prolonged Psychotic Disorder TOTAL)<br />
Panic Attack<br />
Delusional Disorder,
Persecutory Type<br />
03-Apr-2012 09:37 AM Page: 314
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/15/01ISR Number: 3682140-8Report Type:Expedited (15-DaCompany Report #253034 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Erythema Nodosum Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pyrexia Health Inc ORAL ORAL<br />
Professional<br />
Date:03/16/01ISR Number: 3681869-5Report Type:Expedited (15-DaCompany Report #235171 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delivery Lariam Tablets PS Roche THE PATIENT<br />
Abnormal Labour<br />
TOOK A TOTAL<br />
OF 7 TABLETS<br />
WHILE SHE WAS<br />
A FEW WEEKS 50 DAY<br />
Date:03/20/01ISR Number: 3684540-9Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam Tablets PS Roche TWO DOSES<br />
Initial or Prolonged Loss Of Consciousness TAKEN (THREE<br />
Dyspnoea<br />
TABLETS IN<br />
Hyperventilation<br />
TOTAL).<br />
Convulsion<br />
Pyrexia<br />
Date:03/20/01ISR Number: 3684549-5Report Type:Expedited (15-DaCompany Report #256591 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Alveolitis Lariam Tablets PS Roche<br />
Initial or Prolonged Prozac C<br />
Date:03/20/01ISR Number: 3685099-2Report Type:Direct Company Report # Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Social Avoidant Behaviour Lariam PS Roche ORAL 1 WEEKLY ORAL<br />
Agitation<br />
Ear Disorder<br />
Mental Disorder<br />
Emotional Disorder<br />
Depression<br />
Date:03/20/01ISR Number: 3685682-4Report Type:Direct Company Report # Age:58 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Asthma Lariam PS 1 TABLET<br />
Intervention to Hypoaesthesia WEEKLY<br />
Prevent Permanent<br />
Impairment/Damage<br />
Insomnia<br />
Discomfort<br />
Feeling Jittery<br />
03-Apr-2012 09:37 AM Page: 315
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/21/01ISR Number: 3687541-XReport Type:Expedited (15-DaCompany Report #256591 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Alveolitis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 250 MG 1 PER<br />
Professional<br />
Prozac (Fluoxetine<br />
Hydrochloride)<br />
C<br />
1 WEEK ORAL<br />
Date:03/21/01ISR Number: 3687543-3Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pyrexia Health Inc ORAL 1 PER 1 WEEK<br />
Po2 Decreased Professional ORAL<br />
Agitation<br />
Fear<br />
Depressed Level Of<br />
Consciousness<br />
Arthralgia<br />
Respiratory Disorder<br />
Depression<br />
Convulsion<br />
Dyspnoea<br />
Malaise<br />
Fatigue<br />
Stress<br />
Blood Ph Increased<br />
Tenderness<br />
Apnoea<br />
Panic Attack<br />
Hyperventilation<br />
Electroencephalogram<br />
Abnormal<br />
Nausea<br />
Loss Of Consciousness<br />
Restlessness<br />
Autonomic Nervous System<br />
Imbalance<br />
Date:03/22/01ISR Number: 3688672-0Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Lariam 250mg Roche<br />
Initial or Prolonged Panic Attack Lab PS Roche Lab ORAL 250MG ONCE A<br />
Balance Disorder<br />
WEEK ORAL<br />
Anxiety<br />
Nausea<br />
Diarrhoea
Disorientation<br />
03-Apr-2012 09:37 AM Page: 316
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/23/01ISR Number: 3688177-7Report Type:Expedited (15-DaCompany Report #256731 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Sudden Death Lariam Tablets PS Roche<br />
Drug Interaction Acetylcysteine C<br />
Hypertrophic Dirithromycin I<br />
Cardiomyopathy Halofantrine I<br />
Date:03/26/01ISR Number: 3690128-6Report Type:Expedited (15-DaCompany Report #256731 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hypertrophic Literature Lariam PS Hoffmann La Roche<br />
Cardiomyopathy Health Inc ORAL ORAL<br />
Malaria Professional Halofantrine<br />
Sudden Death<br />
(Halofantrine<br />
Drug Interaction Hydrochloride) SS ORAL ORAL<br />
Bronchopneumonia<br />
Dirithromycin<br />
Circulatory Collapse (Dirithromycin) SS ORAL ORAL<br />
Headache<br />
Acetylcysteine<br />
(Acetylcysteine) C<br />
Date:03/29/01ISR Number: 3691252-4Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Mental Disorder Foreign Lariam Tablets PS Roche 4 TABLETS<br />
Hospitalization - Pyrexia Health TAKEN IN<br />
Initial or Prolonged Loss Of Consciousness Professional TOTAL. 21 DAY<br />
Influenza Like Illness<br />
Convulsion<br />
Dyspnoea<br />
Hyperventilation<br />
Date:03/29/01ISR Number: 3691267-6Report Type:Expedited (15-DaCompany Report #256952 Age:65 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tachycardia Lariam Tablets PS Roche 6 DAY<br />
Atrial Fibrillation<br />
Anxiety<br />
Date:03/29/01ISR Number: 3692700-6Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Autonomic Neuropathy<br />
Restlessness<br />
Convulsion
Pyrexia<br />
Agitation<br />
Cyanosis<br />
Psychotic Disorder<br />
Malaise<br />
Fatigue<br />
Hyperventilation<br />
Electroencephalogram<br />
03-Apr-2012 09:37 AM Page: 317
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abnormal<br />
Stress<br />
Panic Attack<br />
Apnoea Report Source Product Role Manufacturer Route Dose Duration<br />
Influenza Like Illness Foreign Lariam PS Hoffmann La Roche<br />
Neck Pain Health Inc ORAL 250 MG 1PER<br />
Fear Professional WEEK ORAL<br />
Drug Effect Decreased<br />
Other<br />
Blood Ph Increased<br />
Depressed Level Of<br />
Consciousness<br />
Depression<br />
Mental Disorder<br />
Muscle Spasms<br />
Nausea<br />
Loss Of Consciousness<br />
Pco2 Decreased<br />
Arthralgia<br />
Dyspnoea<br />
Date:03/30/01ISR Number: 3692381-1Report Type:Expedited (15-DaCompany Report #256952 Age:65 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam Tablets PS Roche 6 DAY<br />
Tachycardia<br />
Atrial Fibrillation<br />
Date:03/30/01ISR Number: 3693076-0Report Type:Direct Company Report # Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain Lariam PS Roche ORAL ONCE ORAL<br />
Initial or Prolonged Abnormal Dreams<br />
Other Serious<br />
Dizziness<br />
Required<br />
Gait Disturbance<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:04/02/01ISR Number: 3693472-1Report Type:Expedited (15-DaCompany Report #257483 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Lariam Tablets PS Roche<br />
Initial or Prolonged<br />
Date:04/02/01ISR Number: 3693489-7Report Type:Expedited (15-DaCompany Report #257610 Age:10 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Myalgia Lariam Tablets PS Roche REPORTED DOSE<br />
Nausea = 192.5<br />
Blood Creatine<br />
MG/WEEK,<br />
Phosphokinase Increased TOTAL DOSE =<br />
5 TABLETS. 22 DAY<br />
03-Apr-2012 09:37 AM Page: 318
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/03/01ISR Number: 3694495-9Report Type:Expedited (15-DaCompany Report #256591 Age:58 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Alveolitis Foreign Lariam Tablets PS Roche 29 DAY<br />
Initial or Prolonged Health Prozac C<br />
Professional<br />
Date:04/03/01ISR Number: 3694515-1Report Type:Expedited (15-DaCompany Report #257527 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Foreign Lariam Tablets PS Roche 29 DAY<br />
Confusional State<br />
Consumer<br />
Balance Disorder<br />
Anxiety<br />
Amnesia<br />
Crying<br />
Date:04/03/01ISR Number: 3694523-0Report Type:Expedited (15-DaCompany Report #257719 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dissociation Health Lariam Tablets PS Roche SEVEN TABLETS<br />
Dizziness Professional TAKEN IN<br />
Amnesia<br />
TOTAL.<br />
Suicidal Ideation<br />
Headache<br />
Balance Disorder<br />
Confusional State<br />
Hypertension<br />
Anxiety<br />
Agoraphobia<br />
Date:04/03/01ISR Number: 3694813-1Report Type:Direct Company Report # Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Impairment Lariam Hoffman<br />
Palpitations Laroche PS Hoffman Laroche ORAL 1 WEEK ORAL<br />
Dizziness<br />
Panic Attack<br />
Tremor<br />
Date:04/03/01ISR Number: 3695222-1Report Type:Expedited (15-DaCompany Report #257483 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Epilepsy Health Inc ORAL DOSE FORM 1
Dialysis Professional PER WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 319
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/03/01ISR Number: 3695224-5Report Type:Expedited (15-DaCompany Report #257610 Age:10 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aspartate Foreign Lariam PS Hoffmann La Roche<br />
Aminotransferase Other Inc ORAL 192.5 MG ORAL<br />
Increased<br />
Muscle Spasms<br />
Myalgia<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Nausea<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Date:04/04/01ISR Number: 3695248-8Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Lariam Tablets PS Roche 280 DAY<br />
Date:04/04/01ISR Number: 3695261-0Report Type:Expedited (15-DaCompany Report #256317 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Lariam Tablets PS Roche 3288 DAY<br />
Diarrhoea Unknown Medication C MEDICATIONS<br />
Myalgia<br />
TAKEN IN<br />
Alopecia<br />
1994,1995 AND<br />
Mental Impairment 1999.<br />
Depression Doxycycline C<br />
Anxiety<br />
Sleep Disorder<br />
Mood Swings<br />
Weight Decreased<br />
Nightmare<br />
Date:04/04/01ISR Number: 3696973-5Report Type:Expedited (15-DaCompany Report #257719 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Confusional State Consumer Lariam PS Hoffmann La Roche<br />
Hypertension Health Inc ORAL ORAL<br />
Feeling Abnormal<br />
Professional<br />
Headache<br />
Amnesia<br />
Dissociative Disorder<br />
Anxiety<br />
Agoraphobia<br />
Balance Disorder<br />
Dizziness
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 320
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/04/01ISR Number: 3697929-9Report Type:Expedited (15-DaCompany Report #257527 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Amnesia Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Disturbance In Attention Other Inc ORAL 1 DOSE FORM 1<br />
Prevent Permanent Balance Disorder PER WEEK ORAL<br />
Impairment/Damage Crying<br />
Confusional State<br />
Anxiety<br />
Date:04/04/01ISR Number: 3697932-9Report Type:Expedited (15-DaCompany Report #256591 Age:58 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Alveolitis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 250 MG 1 PER<br />
Professional<br />
Prozac (Fluoxetine<br />
Hydrochloride)<br />
C<br />
1 WEEK ORAL<br />
Date:04/05/01ISR Number: 3699597-9Report Type:Expedited (15-DaCompany Report #256317 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Disturbance In Attention Other Lariam PS Hoffmann La Roche<br />
Myalgia<br />
Inc<br />
Fatigue<br />
Unknown Medication<br />
Irritability<br />
(Generic<br />
Anxiety<br />
Component(S)<br />
Sleep Disorder Unknown) C<br />
Mood Swings<br />
Doxycycline<br />
Alopecia (Doxycycline) C<br />
Mental Impairment<br />
Muscular Weakness<br />
Nightmare<br />
Diarrhoea<br />
Weight Decreased<br />
Depression<br />
Date:04/05/01ISR Number: 3699598-0Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Health Lariam PS Hoffmann La Roche<br />
Gait Disturbance Professional Inc ORAL 1 PER WEEK<br />
ORAL<br />
Date:04/05/01ISR Number: 3699935-7Report Type:Direct Company Report # Age:45 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Myalgia Lariam / 250 Mg. /<br />
Required Influenza Like Illness Roche PS Roche ORAL 1 TABLET 1 PR<br />
Intervention to Malaise WK/8 ORAL<br />
Prevent Permanent Feeling Abnormal<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 321
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/09/01ISR Number: 3701040-8Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Vertigo Positional Lariam (Tablets<br />
Depression 250mg) PS 1 TAB ONCE<br />
Dehydration WEEKLY 10 WK<br />
Sleep Disorder Milk Thistle C<br />
Headache Vitamins C<br />
Circadian Rhythm Sleep Ginger C<br />
Disorder Tums C<br />
Chest Pain Aspirin C<br />
Weight Increased<br />
Dizziness<br />
Abnormal Dreams<br />
Ear Infection<br />
Balance Disorder<br />
Stress<br />
Fall<br />
Mountain Sickness Acute<br />
Anxiety<br />
Date:04/09/01ISR Number: 3701071-8Report Type:Direct Company Report # Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain Lariam Roche PS Roche ORAL ONCE ORAL<br />
Initial or Prolonged Dizziness<br />
Other Serious<br />
Abnormal Dreams<br />
Required<br />
Feeling Abnormal<br />
Intervention to<br />
Gait Disturbance<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:04/10/01ISR Number: 3701155-4Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Lariam Tablets PS Roche 280 DAY<br />
Date:04/11/01ISR Number: 3702283-XReport Type:Expedited (15-DaCompany Report #258083 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Lariam Tablets PS Roche 42 DAY<br />
Abdominal Pain<br />
Evening Primrose Oil C<br />
Calcium Carbonate C<br />
Ibuprofen<br />
C
Date:04/11/01ISR Number: 3703089-8Report Type:Direct Company Report # Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Lariam PS ORAL 1 TABLE<br />
Insomnia<br />
WEEKLY ORAL<br />
Pain<br />
Dehydration<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 322
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/11/01ISR Number: 3703237-XReport Type:Direct Company Report # Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Affective Disorder Lariam / 250mg PS ORAL 250 MG ONCE A<br />
Decreased Appetite<br />
WEEK ORAL<br />
Lethargy<br />
Headache<br />
Dermatitis<br />
Date:04/11/01ISR Number: 3703278-2Report Type:Direct Company Report # Age:75 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urinary Incontinence Lariam (<strong>Mefloquine</strong>)<br />
Circulatory Collapse<br />
250 Mg. Roche<br />
Loss Of Consciousness Laboratories PS Roche Laboratories 1 TABLET<br />
Fall<br />
WEEKLY<br />
Dyskinesia BEGINNING 1<br />
WEEK BEFORE<br />
TRIP<br />
Date:04/11/01ISR Number: 3703298-8Report Type:Expedited (15-DaCompany Report #246474 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Health Lariam PS Hoffmann La Roche<br />
Skin Lesion Professional Inc ORAL 1 PER WEEK<br />
ORAL<br />
Date:04/12/01ISR Number: 3704261-3Report Type:Expedited (15-DaCompany Report #258083 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Foreign Lariam PS Hoffmann La Roche<br />
Hepatitis Other Inc ORAL 250 MG DAILY<br />
ORAL<br />
Evening Primrose Oil<br />
(Evening Primrose<br />
Oil)<br />
C<br />
Calcium Carbonate<br />
(Calcium Carbonate) C<br />
Ibuprofen<br />
(Ibuprofen)<br />
C<br />
Date:04/16/01ISR Number: 3720297-0Report Type:Expedited (15-DaCompany Report #EM2001-0474 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Rash Pruritic Study Proleukin
Injection Site Health (Aldesleukin)Injecti<br />
Inflammation Professional on PS Chiron Corporation SUBCUTANEOUS 4.5 MIU BID,<br />
Gait Disturbance<br />
SUBCUTAN<br />
Injection Site Pain<br />
Lariam (<strong>Mefloquine</strong>) SS<br />
Injection Site Reaction Ddi (Didanosine) C<br />
Hypersensitivity<br />
Hydrocortisone<br />
Urticaria (Hydrocortisone) C<br />
Nevirapine<br />
03-Apr-2012 09:37 AM Page: 323
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
(Nevirapine)<br />
Sudafed<br />
(Pseudoephedrine<br />
Hydrochloride)<br />
Tylenol<br />
(Paracetamol)<br />
Nizoral<br />
(Ketoconazole)<br />
Motrin (Ibuprofen)<br />
Zerit (Stavudine)<br />
Indinavir<br />
(Indinavir)<br />
Multivitamins<br />
(Ergocalciferol,<br />
Ascorbic Acid, Folic<br />
Acid, Thiamine<br />
Hydrochloide,<br />
C<br />
C<br />
C<br />
C<br />
C<br />
C<br />
C<br />
C<br />
Date:04/17/01ISR Number: 3705506-6Report Type:Expedited (15-DaCompany Report #235171 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delivery Lariam Tablets PS Roche THE PATIENT<br />
Abnormal Labour<br />
TOOK A TOTAL<br />
OF 7 TABLETS<br />
WHILE SHE WAS<br />
A FEW WEEKS 50 DAY<br />
Date:04/17/01ISR Number: 3705513-3Report Type:Expedited (15-DaCompany Report #246098 Age:22 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Impairment Lariam Tablets PS Roche 28 DAY<br />
Anxiety<br />
Aphasia<br />
Speech Disorder<br />
Paranoia<br />
Date:04/17/01ISR Number: 3705516-9Report Type:Expedited (15-DaCompany Report #257483 Age:50 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Lariam Tablets PS Roche<br />
Initial or Prolonged Rhabdomyolysis<br />
Malaise<br />
Diarrhoea<br />
Date:04/17/01ISR Number: 3705518-2Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Lariam Tablets PS Roche<br />
Abnormal Behaviour<br />
03-Apr-2012 09:37 AM Page: 324
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/17/01ISR Number: 3706802-9Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Larium PS ORAL ORAL<br />
Anxiety<br />
Arthralgia<br />
Depression<br />
Convulsion<br />
Suicidal Ideation<br />
Anger<br />
Thinking Abnormal<br />
Sleep Disorder<br />
Date:04/18/01ISR Number: 3707820-7Report Type:Expedited (15-DaCompany Report #235171 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Abnormal Labour Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Caesarean Section Health Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Professional WEEK ORAL<br />
Impairment/Damage<br />
Date:04/18/01ISR Number: 3707821-9Report Type:Expedited (15-DaCompany Report #246098 Age:22 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aphasia Foreign Lariam PS Hoffmann La Roche<br />
Anxiety Other Inc ORAL ORAL 28 DAY<br />
Visual Impairment<br />
Paranoia<br />
Speech Disorder<br />
Date:04/18/01ISR Number: 3707822-0Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Foreign Lariam PS Hoffmann La Roche<br />
Abnormal Behaviour Health Inc ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:04/18/01ISR Number: 3707823-2Report Type:Expedited (15-DaCompany Report #257483 Age:50 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Diarrhoea Health Inc ORAL 1 DOSE FORM 1<br />
Dialysis Professional PER WEEK ORAL<br />
Rhabdomyolysis<br />
Renal Failure
Epilepsy<br />
03-Apr-2012 09:37 AM Page: 325
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/23/01ISR Number: 3708595-8Report Type:Expedited (15-DaCompany Report #246098 Age:22 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Speech Disorder Lariam Tablets PS Roche 28 DAY<br />
Anxiety<br />
Paranoia<br />
Visual Impairment<br />
Date:04/24/01ISR Number: 3709086-0Report Type:Expedited (15-DaCompany Report #242380 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Platelet Count Decreased Lariam Tablets PS Roche 28 DAY<br />
Hyperuricaemia<br />
Intravascular Haemolysis<br />
Menometrorrhagia<br />
Liver Function Test<br />
Abnormal<br />
Date:04/24/01ISR Number: 3711028-9Report Type:Expedited (15-DaCompany Report #246098 Age:22 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Foreign Lariam PS Hoffmann La Roche<br />
Anxiety Other Inc ORAL ORAL 28 YR<br />
Aphasia<br />
Speech Disorder<br />
Visual Impairment<br />
Date:04/25/01ISR Number: 3710150-0Report Type:Expedited (15-DaCompany Report #258981 Age:53 YR Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Interaction Lariam Tablets PS Roche 9 DAY<br />
Hypersensitivity Multivitamins C<br />
Indinavir<br />
C<br />
Zerit<br />
C<br />
Ddi<br />
C<br />
Hydrocortisone C<br />
Nevirapine<br />
C<br />
Sudafed<br />
C<br />
Tylenol<br />
C<br />
Nizoral<br />
C<br />
Motrin<br />
C<br />
Proleukin I Roche 124 DAY<br />
Date:04/25/01ISR Number: 3710156-1Report Type:Expedited (15-DaCompany Report #258834 Age:56 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor <strong>Mefloquine</strong><br />
Depression Hydrochloride PS Roche 10 DAY<br />
Insomnia Avaxim C 1 DAY<br />
03-Apr-2012 09:37 AM Page: 326
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/25/01ISR Number: 3711519-0Report Type:Expedited (15-DaCompany Report #242380 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Hyperuricaemia Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Platelet Count Decreased Health Inc ORAL ORAL 28 DAY<br />
Prevent Permanent Menometrorrhagia Professional<br />
Impairment/Damage Intravascular Haemolysis<br />
Pregnancy<br />
Hepatic Function Abnormal<br />
Date:04/26/01ISR Number: 3711060-5Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Lariam Tablets PS Roche 8 DAY<br />
Delusion Toco C<br />
Xatral<br />
C<br />
Aricept<br />
C<br />
Tanakan<br />
C<br />
Date:04/26/01ISR Number: 3711849-2Report Type:Expedited (15-DaCompany Report #258834 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Foreign Lariam PS Hoffmann La Roche<br />
Tremor Other Inc ORAL 25 MG DAILY<br />
Insomnia<br />
Avaxim (Hepatitis A<br />
Vaccine)<br />
C<br />
ORAL<br />
Date:04/26/01ISR Number: 3711913-8Report Type:Expedited (15-DaCompany Report #258981 Age:53 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Erythema Health Lariam PS Hoffmann La Roche<br />
Swelling Professional Inc ORAL ORAL<br />
Injection Site Oedema Other Proleukin<br />
Dermatitis (Aldeslukin) SS SUBCUTANEOUS 4.5 MILLIONIU<br />
Injection Site Reaction<br />
2 PER 1 DAY<br />
Hypersensitivity<br />
SUBCUTANEOUS<br />
Injection Site Ddi (Didanosine) C<br />
Inflammation<br />
Hydrocortizone<br />
Rash Pruritic (Hydrocortizone) C<br />
Urticaria<br />
Nevirapine<br />
Drug Interaction (Nevirapine) C<br />
Injection Site Pain<br />
Sudafed<br />
(Pseudophedrine<br />
Hydrochloride) C<br />
Tylenol<br />
(Acetaminophen) C
Nizoral<br />
(Ketoconazole)<br />
Motrin (Ibuprofen)<br />
Zerit (Stavudine)<br />
Indinavir<br />
(Indinavir)<br />
Multivitamins<br />
(Multivitamin Nos)<br />
C<br />
C<br />
C<br />
C<br />
C<br />
03-Apr-2012 09:37 AM Page: 327
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/27/01ISR Number: 3713509-0Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Foreign Lariam PS Hoffmann La Roche<br />
Abnormal Behaviour Consumer Inc ORAL 1 DOSE FORM 1<br />
PER WEEK ORAL<br />
Toco (Vitamin E) C<br />
Xatral (Alfuzosin<br />
Hydrochloride) C<br />
Aricept (Donepezil) C<br />
Tanakan (Ginkgo) C<br />
Date:05/02/01ISR Number: 3714650-9Report Type:Expedited (15-DaCompany Report #220210 Age:17 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Accommodation Disorder Lariam Tablets PS Roche 47 DAY<br />
Initial or Prolonged<br />
Date:05/02/01ISR Number: 3714658-3Report Type:Expedited (15-DaCompany Report #259221 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Dysmorphism Lariam Tablets PS Roche 15 DAY<br />
Congenital Foot Arilvax SS 1 DAY<br />
Malformation Vivotif "Berna" SS 1 DAY<br />
Limb Reduction Defect<br />
Congenital Hand<br />
Malformation<br />
Congenital Central<br />
Nervous System Anomaly<br />
Thymus Disorder<br />
Date:05/03/01ISR Number: 3716049-8Report Type:Expedited (15-DaCompany Report #259450 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Urticaria Lariam Tablets PS Roche 30 DAY<br />
Initial or Prolonged Face Oedema<br />
Dyspnoea<br />
Laryngeal Oedema<br />
Date:05/03/01ISR Number: 3717035-4Report Type:Expedited (15-DaCompany Report #259221 Age: Gender:Female I/FU:I<br />
Outcome<br />
Congenital Anomaly<br />
PT<br />
Thymus Disorder<br />
Congenital Central<br />
Nervous System Anomaly
Dysmorphism<br />
Congenital Hand<br />
Malformation<br />
Localised Oedema<br />
Limb Reduction Defect<br />
Congenital Foot<br />
Malformation<br />
Tricuspid Valve Disease<br />
03-Apr-2012 09:37 AM Page: 328
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abortion Induced<br />
Maternal Drugs Affecting<br />
Foetus Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Cardiovascular Foreign Lariam PS Hoffmann La Roche<br />
Anomaly Health Inc ORAL 250 MG 1<br />
Professional<br />
PER WEEK<br />
Other<br />
ORAL<br />
Arilvax (Yellow<br />
Fever Vaccine) SS 1 PER ONE<br />
DOSE<br />
Vivotif "Berna"<br />
(Typhoid Vaccines) SS ORAL 1 PER ONE<br />
DOSE ORAL<br />
Date:05/03/01ISR Number: 3717038-XReport Type:Expedited (15-DaCompany Report #220210 Age:17 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Refraction Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Diplopia Consumer Inc ORAL 250 MG 1<br />
Hypertension Health PER WEEK<br />
Antinuclear Antibody Professional ORAL<br />
Positive<br />
Vision Blurred<br />
Platelet Count Decreased<br />
Herpes Zoster<br />
Weight Increased<br />
Neuritis<br />
Lipids Increased<br />
Headache<br />
Thrombocytosis<br />
Pupillary Disorder<br />
Binocular Eye Movement<br />
Disorder<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal<br />
Laboratory Test Abnormal<br />
Csf Test Abnormal<br />
Nervous System Disorder<br />
Infectious Mononucleosis<br />
Chlamydial Infection<br />
Visual Acuity Reduced<br />
Accommodation Disorder<br />
Herpes Simplex<br />
Folate Deficiency<br />
Measles<br />
Inflammation<br />
Date:05/04/01ISR Number: 3716893-7Report Type:Expedited (15-DaCompany Report #259699 Age:72 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Haemorrhagic Stroke Lariam Tablets PS Roche 37 DAY<br />
Drug Interaction Cozaar Comp C 285 DAY<br />
Circulatory Collapse Warfarin I<br />
International Normalised<br />
Ratio Increased<br />
03-Apr-2012 09:37 AM Page: 329
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/04/01ISR Number: 3717913-6Report Type:Expedited (15-DaCompany Report #259450 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Urticaria Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pruritus Other Inc ORAL 1 DOSE FORM 1<br />
Pain<br />
PER WEEK ORAL<br />
Face Oedema<br />
Dyspnoea<br />
Laryngeal Oedema<br />
Erythema<br />
Date:05/07/01ISR Number: 3717719-8Report Type:Direct Company Report # Age:68 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Lariam / 250 Mg. /<br />
Initial or Prolonged Blood Pressure Increased Laroche Laboritorys, PS 250 MG ONCE A<br />
Disability Dizziness WK 6 WKS<br />
Congenital Anomaly<br />
Other Serious<br />
Hyperventilation<br />
Disorientation<br />
Dyspnoea<br />
Tinnitus<br />
Date:05/07/01ISR Number: 3718783-2Report Type:Expedited (15-DaCompany Report #259699 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death International Normalised Foreign Lariam PS Hoffmann La Roche<br />
Ratio Increased Other Inc ORAL 250 MG 1 PER<br />
Haemorrhagic Stroke<br />
WEEK ORAL<br />
Circulatory Collapse<br />
Warfarin (Warfarin<br />
Drug Interaction Sodium) SS ORAL 4 MG 1 PER 2<br />
DAY ORAL<br />
Cozaar Comp<br />
(Hydrochlorothiazide<br />
/Losartan Potassium) C<br />
Date:05/08/01ISR Number: 3718309-3Report Type:Expedited (15-DaCompany Report #235171 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Labour Lariam Tablets PS Roche THE PATIENT<br />
Pregnancy<br />
TOOK A TOTAL<br />
Caesarean Section<br />
OF 7 TABLETS<br />
Maternal Drugs Affecting<br />
WHILE SHE WAS<br />
Foetus A FEW WEEKS 50 DAY<br />
Date:05/09/01ISR Number: 3719103-XReport Type:Expedited (15-DaCompany Report #251328 Age: Gender:Not SpecifiI/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Limb Reduction Defect Lariam Tablets PS Roche<br />
03-Apr-2012 09:37 AM Page: 330
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/09/01ISR Number: 3719121-1Report Type:Expedited (15-DaCompany Report #259776 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gamma-Glutamyltransferase Lariam Tablets PS Roche 1 DAY<br />
Increased<br />
Blood Thyroid Stimulating<br />
Hormone Increased<br />
Sleep Disorder<br />
Blood Cholesterol<br />
Increased<br />
Blood Triglycerides<br />
Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Blood Pressure Increased<br />
Malaise<br />
Muscle Spasms<br />
Thyroxine Decreased<br />
Xanthelasma<br />
Date:05/09/01ISR Number: 3719124-7Report Type:Expedited (15-DaCompany Report #259784 Age: Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Lariam Tablets PS Roche THE PATIENT<br />
Initial or Prolonged Nausea HAD RECEIVED<br />
Pyelonephritis<br />
THREE DOSE<br />
Vomiting<br />
FORMS IN<br />
Gastritis TOTAL. 14 DAY<br />
Date:05/09/01ISR Number: 3721171-6Report Type:Expedited (15-DaCompany Report #235171 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Caesarean Section Foreign Lariam PS Hoffmann La Roche<br />
Intervention to Abnormal Labour Health Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Professional WEEK ORAL<br />
Impairment/Damage<br />
Other<br />
Date:05/10/01ISR Number: 3720252-0Report Type:Expedited (15-DaCompany Report #259927 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Lariam Tablets PS Roche THE PATIENT<br />
Initial or Prolonged Haematochezia TOOK 3 DOSES<br />
OF LARIAM<br />
BEFORE DATE<br />
OF ONSET.<br />
Marcumar I Roche
Date:05/10/01ISR Number: 3721537-4Report Type:Expedited (15-DaCompany Report #259776 Age:49 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Thyroxine Decreased<br />
Muscle Spasms<br />
Blood Pressure Increased<br />
03-Apr-2012 09:37 AM Page: 331
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Triglycerides<br />
Increased<br />
Blood Thyroid Stimulating Report Source Product Role Manufacturer Route Dose Duration<br />
Hormone Increased Foreign Lariam PS Hoffmann La Roche<br />
Lipids Increased Health Inc ORAL ORAL<br />
Gamma-Glutamyltransferase Professional<br />
Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Malaise<br />
Sleep Disorder<br />
Blood Cholesterol<br />
Increased<br />
Xanthelasma<br />
Date:05/10/01ISR Number: 3721538-6Report Type:Expedited (15-DaCompany Report #259784 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyelonephritis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Vomiting Health Inc ORAL 1 DOSE FORM 1<br />
Gastritis Professional PER WEEK ORAL<br />
Asthenia<br />
Date:05/10/01ISR Number: 3721539-8Report Type:Expedited (15-DaCompany Report #251328 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Pregnancy Foreign Lariam PS Hoffmann La Roche<br />
Limb Malformation Health Inc ORAL 1 DOSE FORM 1<br />
Maternal Drugs Affecting Professional PER WEEK ORAL<br />
Foetus<br />
Limb Reduction Defect<br />
Date:05/11/01ISR Number: 3720799-7Report Type:Expedited (15-DaCompany Report #260010 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam PS Roche 46 DAY<br />
Initial or Prolonged Hallucination<br />
Suffocation Feeling<br />
Date:05/11/01ISR Number: 3720804-8Report Type:Expedited (15-DaCompany Report #259933 Age:62 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypertension Lariam PS Roche 60 DAY<br />
Panic Attack<br />
Fatigue
Asthenia<br />
03-Apr-2012 09:37 AM Page: 332
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/11/01ISR Number: 3720906-6Report Type:Direct Company Report # Age:68 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperventilation Lariam / 250 Mg. /<br />
Initial or Prolonged Asthenia Laroche Laboritorys, PS Laroche Laboritorys 250 MG. ONE<br />
Disability Disorientation A WK. 6 WKS<br />
Congenital Anomaly<br />
Other Serious<br />
Dyspnoea<br />
Blood Pressure Increased<br />
Tinnitus<br />
Dizziness<br />
Date:05/14/01ISR Number: 3721765-8Report Type:Expedited (15-DaCompany Report #259776 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thyroid Function Test Lariam Tablets PS Roche 1 DAY<br />
Abnormal<br />
Malaise<br />
Blood Pressure Increased<br />
Xanthelasma<br />
Sleep Disorder<br />
Alanine Aminotransferase<br />
Increased<br />
Lipids Increased<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Muscle Spasms<br />
Date:05/15/01ISR Number: 3723039-8Report Type:Expedited (15-DaCompany Report #259927 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haematochezia Lariam Tablets PS Roche THE PATIENT<br />
Initial or Prolonged<br />
TOOK 3 DOSES<br />
OF LARIAM<br />
BEFORE DATE<br />
OF ONSET.<br />
Marcumar<br />
C<br />
Date:05/16/01ISR Number: 3724398-2Report Type:Direct Company Report # Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Lariam 250 Mg Tablet<br />
Coordination Abnormal Roche Labs PS Roche Labs 1 TABLET PER<br />
Fatigue<br />
WEEK<br />
Photophobia I.G. Shot C<br />
Nausea<br />
Insomnia<br />
Disturbance In Attention
Hearing Impaired<br />
Sensation Of Pressure<br />
Headache<br />
Dizziness<br />
Sedation<br />
Memory Impairment<br />
Burning Sensation<br />
Speech Disorder<br />
03-Apr-2012 09:37 AM Page: 333
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/16/01ISR Number: 3724861-4Report Type:Expedited (15-DaCompany Report #259933 Age:62 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Asthenia Consumer Lariam PS Hoffmann La Roche<br />
Intervention to Dyspnoea Inc ORAL 1 PER WEEK<br />
Prevent Permanent Hypertension ORAL<br />
Impairment/Damage Disorientation<br />
Panic Attack<br />
Myocardial Infarction<br />
Dizziness<br />
Emotional Disorder<br />
Bronchitis<br />
Fatigue<br />
Pneumonia Mycoplasmal<br />
Chest Pain<br />
Anxiety<br />
Blood Pressure Increased<br />
Date:05/16/01ISR Number: 3725229-7Report Type:Expedited (15-DaCompany Report #260010 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Anxiety Other Inc ORAL 250 MG 1 PER<br />
Suffocation Feeling<br />
WEEK ORAL<br />
Date:05/17/01ISR Number: 3724379-9Report Type:Direct Company Report #USP 54068 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Medication Error Lariam PS Roche<br />
Monograph For<br />
<strong>Mefloquine</strong><br />
Hydrochloride SS Micromedex<br />
Date:05/17/01ISR Number: 3724772-4Report Type:Expedited (15-DaCompany Report #259927 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - International Normalised Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Ratio Increased Consumer Inc ORAL ORAL<br />
Haematochezia<br />
Marcumar<br />
(Phenprocoumon) C<br />
Date:05/18/01ISR Number: 3724986-3Report Type:Expedited (15-DaCompany Report #259784 Age: Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyelonephritis Lariam Tablets PS Roche THE PATIENT
Initial or Prolonged Vomiting HAD RECEIVED<br />
Nausea<br />
THREE DOSE<br />
Asthenia<br />
FORMS IN<br />
Gastritis TOTAL. 14 DAY<br />
03-Apr-2012 09:37 AM Page: 334
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/18/01ISR Number: 3725008-0Report Type:Expedited (15-DaCompany Report #260506 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Pain Lariam PS Roche THREE TABLETS<br />
Initial or Prolonged Gastritis TAKEN IN<br />
Asthenia TOTAL. 14 DAY<br />
Vomiting Stamaril Unidose C 1 DAY<br />
Pyelonephritis<br />
Nausea<br />
Date:05/18/01ISR Number: 3725014-6Report Type:Expedited (15-DaCompany Report #247738 Age: Gender:Not SpecifiI/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pregnancy Lariam PS Roche THREE TABLETS<br />
Uterine Hypertonus<br />
TAKEN IN<br />
TOTAL. 14 DAY<br />
Stamaril Unidose C 1 DAY<br />
Date:05/21/01ISR Number: 3725705-7Report Type:Expedited (15-DaCompany Report #203837 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Hallucinations, Mixed<br />
Flushing<br />
Nervous System Disorder<br />
Delirium<br />
Anxiety<br />
Musculoskeletal Stiffness<br />
Decreased Appetite<br />
Adjustment Disorder<br />
Amnesia<br />
Pyrexia<br />
Petit Mal Epilepsy<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Delusional Disorder,<br />
Persecutory Type<br />
Cognitive Disorder<br />
Chills<br />
Blood Cortisol Increased<br />
Depression<br />
Urinary Hesitation<br />
Headache<br />
Weight Decreased<br />
Photopsia<br />
Erythema<br />
Claustrophobia<br />
Hepatic Steatosis<br />
Hyperhidrosis<br />
Erectile Dysfunction
Completed Suicide<br />
Agitation<br />
Insomnia<br />
Fatigue<br />
Liver Function Test<br />
Abnormal<br />
Obsessive-Compulsive<br />
Disorder<br />
03-Apr-2012 09:37 AM Page: 335
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Polydipsia<br />
Abnormal Dreams<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche 49 DAY<br />
Date:05/21/01ISR Number: 3726835-6Report Type:Expedited (15-DaCompany Report #260506 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Pain Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Vomiting Health Inc ORAL 1 DOSE FORM 1<br />
Nausea Professional PER WEEK ORAL<br />
Pyelonephritis Stamaril Unidose C<br />
Asthenia<br />
Uterine Contractions<br />
During Pregnancy<br />
Gastritis<br />
Date:05/21/01ISR Number: 3726836-8Report Type:Expedited (15-DaCompany Report #247738 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Breech Presentation Foreign Lariam PS Hoffmann La Roche<br />
Pyelonephritis Health Inc ORAL 1 DOSE FORM<br />
Uterine Hypertonus Professional 1 PER WEEK<br />
ORAL<br />
Stamaril Unidose<br />
(Yellow Fever<br />
Vaccine)<br />
C<br />
Date:05/21/01ISR Number: 3726936-2Report Type:Expedited (15-DaCompany Report #259784 Age: Gender: I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyelonephritis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Gastritis Health Inc ORAL 1 DOSE FORM 1<br />
Breech Presentation Professional PER WEEK ORAL<br />
Vomiting<br />
Maternal Drugs Affecting<br />
Foetus<br />
Asthenia<br />
Date:05/22/01ISR Number: 3726521-2Report Type:Expedited (15-DaCompany Report #260569 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Weight Decreased Lariam PS Roche 8 DAY<br />
Initial or Prolonged Vomiting
Date:05/23/01ISR Number: 3727977-1Report Type:Expedited (15-DaCompany Report #203837 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Urinary Hesitation<br />
Hepatic Steatosis<br />
Photopsia<br />
03-Apr-2012 09:37 AM Page: 336
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Erythema<br />
Abnormal Dreams<br />
Polydipsia Report Source Product Role Manufacturer Route Dose Duration<br />
Petit Mal Epilepsy Consumer Lariam PS Hoffmann La Roche<br />
Hallucinations, Mixed Other Inc ORAL 250 MG 1<br />
Amnesia<br />
PER 1 WEEK<br />
Blood Cortisol Increased ORAL 49 DAY<br />
Delirium<br />
Stress<br />
Pyrexia<br />
Erectile Dysfunction<br />
Adjustment Disorder<br />
Agitation<br />
Nervous System Disorder<br />
Cognitive Disorder<br />
Claustrophobia<br />
Musculoskeletal Stiffness<br />
Hyperhidrosis<br />
Flushing<br />
Delusional Disorder,<br />
Persecutory Type<br />
Headache<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Completed Suicide<br />
Depression<br />
Obsessive-Compulsive<br />
Disorder<br />
Liver Function Test<br />
Abnormal<br />
Date:05/23/01ISR Number: 3728418-0Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Claustrophobia Larium Roche PS Roche ORAL 1 WEEKLY ORAL<br />
Anxiety<br />
Paranoia<br />
Hallucination<br />
Date:05/24/01ISR Number: 3728060-1Report Type:Direct Company Report # Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hypersensitivity Lariam PS ORAL 1 TABLET<br />
ORALLY<br />
Date:05/24/01ISR Number: 3728649-XReport Type:Expedited (15-DaCompany Report #260569 Age:29 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Weight Decreased Health Inc ORAL 250 MG 1 PER<br />
Professional<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 337
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/29/01ISR Number: 3728985-7Report Type:Expedited (15-DaCompany Report #259927 Age:73 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haematochezia Lariam Tablets PS Roche 36 DAY<br />
Initial or Prolonged Diverticulitis Aspirin Protect 100 C 584 DAY<br />
Marcumar<br />
C<br />
Date:05/29/01ISR Number: 3728990-0Report Type:Expedited (15-DaCompany Report #260569 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Lariam PS Roche 8 DAY<br />
Initial or Prolonged Weight Decreased<br />
Date:05/29/01ISR Number: 3728996-1Report Type:Expedited (15-DaCompany Report #260745 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nerve Root Lesion Lariam PS Roche FOUR TIMES IN<br />
TOTAL. 29 DAY<br />
Date:05/29/01ISR Number: 3729002-5Report Type:Expedited (15-DaCompany Report #260795 Age:65 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Extrasystoles Lariam PS Roche 1 DAY<br />
Transient Ischaemic Aspirin C<br />
Attack Mvi C<br />
Atrial Fibrillation<br />
Date:05/30/01ISR Number: 3729600-9Report Type:Expedited (15-DaCompany Report #257983 Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cystitis Lariam Tablets PS Roche 32 DAY<br />
Myalgia<br />
Influenza Like Illness<br />
Insomnia<br />
Arthralgia<br />
Prostatic Specific<br />
Antigen Increased<br />
Neuritis<br />
Back Pain<br />
Pyrexia<br />
Prostatic Disorder<br />
Anxiety<br />
Urinary Retention<br />
Fatigue
Date:05/31/01ISR Number: 3731355-9Report Type:Expedited (15-DaCompany Report #260795 Age:65 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Atrial Fibrillation<br />
Transient Ischaemic<br />
Attack<br />
03-Apr-2012 09:37 AM Page: 338
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Extrasystoles<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 250 MG 1 PER<br />
ONE DOSE;ORAL<br />
Aspirin (Aspirin) C<br />
Mvi (Multivitamin<br />
Nos)<br />
C<br />
Date:05/31/01ISR Number: 3731408-5Report Type:Expedited (15-DaCompany Report #259927 Age:73 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arthralgia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Faeces Discoloured Consumer Inc ORAL 1 DOSE FORM 1<br />
Required Haematochezia PER WEEK ORAL<br />
Intervention to Diverticulitis Aspirin Protect 100<br />
Prevent Permanent Haemoglobin Decreased (Aspirin) C<br />
Impairment/Damage<br />
Marcumar<br />
(Phenprocoumon) C<br />
Date:05/31/01ISR Number: 3731786-7Report Type:Expedited (15-DaCompany Report #257983 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Foreign Lariam PS Hoffmann La Roche<br />
Prostatic Disorder Health Inc ORAL 1 DOSE FORM<br />
Neuritis Professional 1 PER WEEK<br />
Bladder Disorder<br />
ORAL<br />
Urinary Incontinence<br />
Cystitis<br />
Arthralgia<br />
Insomnia<br />
Urinary Retention<br />
Influenza Like Illness<br />
Urinary Tract Infection<br />
Vestibular Disorder<br />
Prostatic Specific<br />
Antigen Increased<br />
Hyperhidrosis<br />
Back Pain<br />
Fatigue<br />
Myalgia<br />
Anxiety<br />
Date:05/31/01ISR Number: 3731789-2Report Type:Expedited (15-DaCompany Report #260745 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Paraesthesia Foreign Lariam PS Hoffmann La Roche<br />
Coordination Abnormal Other Inc ORAL 1 DOSE FORM<br />
Nerve Root Lesion<br />
1 PER WEEK<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 339
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/31/01ISR Number: 3731790-9Report Type:Expedited (15-DaCompany Report #260569 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Weight Decreased Health Inc ORAL 250 MG 1<br />
Professional<br />
PER WEEK<br />
ORAL<br />
Date:06/01/01ISR Number: 3731768-5Report Type:Expedited (15-DaCompany Report #260852 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Lariam PS Roche 46 DAY<br />
Tremor Diovan C<br />
Mental Disorder Roxibion C 10 DAY<br />
Insomnia<br />
Restlessness<br />
Pneumonia<br />
Anxiety<br />
Hyperhidrosis<br />
Date:06/04/01ISR Number: 3733157-6Report Type:Expedited (15-DaCompany Report #260852 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Foreign Lariam PS Hoffmann La Roche<br />
Hyperhidrosis Health Inc ORAL 250 MG 1 PER<br />
Pneumonia Professional WEEK ORAL<br />
Mental Disorder Diovan (Valsartan) C<br />
Tremor<br />
Roxibion<br />
Insomnia (Roxithromycin) C<br />
Restlessness<br />
Hypoaesthesia<br />
Date:06/04/01ISR Number: 3733741-XReport Type:Direct Company Report # Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Blood Pressure Increased Lariam PS OROPHARINGEAL 1 WEEK<br />
Dizziness<br />
OROPHARINGEAL<br />
Anxiety<br />
Date:06/05/01ISR Number: 3733342-3Report Type:Expedited (15-DaCompany Report #247738 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Uterine Hypertonus Lariam PS Roche THREE TABLETS<br />
Delivery<br />
TAKEN IN<br />
TOTAL. 14 DAY
Stamaril Unidose C 1 DAY<br />
03-Apr-2012 09:37 AM Page: 340
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3733356-3Report Type:Expedited (15-DaCompany Report #259776 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Xanthelasma Lariam Tablets PS Roche 1 DAY<br />
Sleep Disorder<br />
Malaise<br />
Blood Pressure Increased<br />
Muscle Spasms<br />
Thyroid Function Test<br />
Abnormal<br />
Alanine Aminotransferase<br />
Increased<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Lipids Increased<br />
Date:06/05/01ISR Number: 3733358-7Report Type:Expedited (15-DaCompany Report #260506 Age:33 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Lariam PS Roche THREE TABLETS<br />
Initial or Prolonged Asthenia TAKEN IN<br />
Sedation TOTAL. 14 DAY<br />
Abdominal Pain Stamaril Unidose I 1 DAY<br />
Balance Disorder<br />
Uterine Contractions<br />
During Pregnancy<br />
Gastritis<br />
Vertigo<br />
Vomiting In Pregnancy<br />
Caesarean Section<br />
Pyelonephritis<br />
Nausea<br />
Date:06/05/01ISR Number: 3733365-4Report Type:Expedited (15-DaCompany Report #261224 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arrhythmia Lariam PS Roche 2 DAY<br />
Initial or Prolonged Vertigo Nivaquine SS 50 DAY<br />
Tremor Paludrine C<br />
Dysarthria<br />
Myalgia<br />
Agitation<br />
Pyrexia<br />
Headache<br />
Arthralgia<br />
Date:06/05/01ISR Number: 3734384-4Report Type:Periodic Company Report #257975 Age:58 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chest Pain Consumer Lariam PS Hoffmann La Roche<br />
Feeling Hot Inc ORAL ORAL<br />
Calcium (Calcium<br />
Nos)<br />
C<br />
Prempro (Estrogens<br />
Conjugated/Medroxypr<br />
03-Apr-2012 09:37 AM Page: 341
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
ogesterone Acetate)<br />
C<br />
Date:06/05/01ISR Number: 3734385-6Report Type:Periodic Company Report #113216 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Asthenia Health Lariam PS Hoffmann La Roche<br />
Disorientation Professional Inc ORAL 250 MG DAILY,<br />
Nausea<br />
ORAL<br />
Fatigue<br />
Lamisil(Terbinafene) C<br />
Paraesthesia<br />
Depression<br />
Date:06/05/01ISR Number: 3734386-8Report Type:Periodic Company Report #222392 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Pressure Diastolic Consumer Lariam PS Hoffmann La Roche<br />
Increased Health Inc ORAL 250 MG 1 PER<br />
Tachycardia Professional WEEK ORAL<br />
Dizziness<br />
Pyrexia<br />
Sputum Discoloured<br />
Decreased Appetite<br />
Anxiety Disorder<br />
Body Temperature<br />
Increased<br />
Dyspnoea<br />
Nasopharyngitis<br />
Fatigue<br />
Oropharyngeal Pain<br />
Urinary Tract Infection<br />
Date:06/05/01ISR Number: 3734387-XReport Type:Periodic Company Report #910200285001 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Weight Decreased Health Lariam PS Hoffmann La Roche<br />
Insomnia Professional Inc ORAL 250 MG 1 PER<br />
Hallucination<br />
WEEK ORAL<br />
Depression<br />
Methotrexate<br />
Depersonalisation (Methotrexate) C<br />
Date:06/05/01ISR Number: 3734388-1Report Type:Periodic Company Report #231044 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Health Lariam PS Hoffmann La Roche<br />
Ear Infection Professional Inc ORAL 250MG 1 PER<br />
WEEK ORAL
Date:06/05/01ISR Number: 3734391-1Report Type:Periodic Company Report #232888 Age:21 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Restlessness<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 342
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paranoia<br />
Asthma<br />
Anxiety Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Health Lariam PS Hoffmann La Roche<br />
Feeling Abnormal Professional Inc ORAL 1 PER WEEK<br />
Vertigo<br />
ORAL<br />
Panic Attack<br />
Date:06/05/01ISR Number: 3734395-9Report Type:Periodic Company Report #246930 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Contractions Health Lariam PS Hoffmann La Roche<br />
Involuntary Professional Inc ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Date:06/05/01ISR Number: 3734397-2Report Type:Periodic Company Report #248729 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nasopharyngitis Health Lariam PS Hoffmann La Roche<br />
Oropharyngeal Pain Professional Inc ORAL 1 DOSE FORM 1<br />
Other<br />
PER WEEK ORAL<br />
Date:06/05/01ISR Number: 3734399-6Report Type:Periodic Company Report #249132 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chest Pain Other Lariam PS Hoffmann La Roche<br />
Sensory Disturbance Inc ORAL ORAL<br />
Insomnia<br />
Dizziness<br />
Date:06/05/01ISR Number: 3734400-XReport Type:Periodic Company Report #249387 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam PS Hoffmann La Roche<br />
Dyspepsia Inc ORAL 250 MG 1 PER<br />
Disturbance In Attention<br />
WEEK ORAL<br />
Dizziness<br />
Hearing Impaired<br />
Date:06/05/01ISR Number: 3734401-1Report Type:Periodic Company Report #253140 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vomiting Consumer Lariam PS Hoffmann La Roche
Nausea Inc ORAL 250 MG 1 PER<br />
Vaginal Haemorrhage<br />
WEEK ORAL<br />
Diarrhoea<br />
03-Apr-2012 09:37 AM Page: 343
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734402-3Report Type:Periodic Company Report #253766 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Health Lariam PS Hoffmann La Roche<br />
Electrocardiogram Professional Inc ORAL 500 MG DAILY,<br />
Abnormal ORAL 6 DAY<br />
Blood Potassium Decreased<br />
Hypoaesthesia<br />
Palpitations<br />
Dizziness<br />
Date:06/05/01ISR Number: 3734405-9Report Type:Periodic Company Report #255250 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blepharitis Health Lariam PS Hoffmann La Roche<br />
Eyelid Oedema Professional Inc ORAL 1 PER WEEK,<br />
Rash Erythematous<br />
ORAL<br />
Date:06/05/01ISR Number: 3734407-2Report Type:Periodic Company Report #255378 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Agitation Consumer Lariam PS Hoffmann La Roche<br />
Dysarthria Inc ORAL 250 MG ORAL<br />
Confusional State<br />
Anxiety<br />
Coordination Abnormal<br />
Feeling Jittery<br />
Dyspnoea<br />
Headache<br />
Date:06/05/01ISR Number: 3734408-4Report Type:Periodic Company Report #256761 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Health Lariam PS Hoffmann La Roche<br />
Vertigo Professional Inc ORAL ORAL<br />
Tremor<br />
Agitation<br />
Date:06/05/01ISR Number: 3734409-6Report Type:Periodic Company Report #257150 Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Health Lariam PS Hoffmann La Roche<br />
Abnormal Dreams Professional Inc ORAL 250 MG 1 PER<br />
Blood Triglycerides<br />
WEEK ORAL<br />
Increased
Date:06/05/01ISR Number: 3734410-2Report Type:Periodic Company Report #242997 Age: Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Constipation<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 344
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diarrhoea<br />
Malaise<br />
Dizziness Report Source Product Role Manufacturer Route Dose Duration<br />
Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734411-4Report Type:Periodic Company Report #243284 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viral Infection Health Lariam PS Hoffmann La Roche<br />
Pruritus Professional Inc ORAL 250MG, 1 PER<br />
Asthenia<br />
WEEK ORAL<br />
Fatigue<br />
Amnesia<br />
Date:06/05/01ISR Number: 3734412-6Report Type:Periodic Company Report #243594 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chest Pain Consumer Lariam PS Hoffmann La Roche<br />
Abnormal Dreams Other Inc ORAL ORAL 56 DAY<br />
Date:06/05/01ISR Number: 3734413-8Report Type:Periodic Company Report #243604 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Consumer Lariam PS Hoffmann La Roche<br />
Headache Other Inc ORAL ORAL<br />
Lethargy<br />
Migraine<br />
Nasopharyngitis<br />
Date:06/05/01ISR Number: 3734414-XReport Type:Periodic Company Report #243612 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Impairment Consumer Lariam PS Hoffmann La Roche<br />
Jaundice Other Inc ORAL 1 PER WEEK<br />
Abnormal Dreams<br />
ORAL<br />
Panic Attack<br />
Asthenia<br />
Anger<br />
Paranoia<br />
Date:06/05/01ISR Number: 3734415-1Report Type:Periodic Company Report #243613 Age: Gender: I/FU:I
Outcome<br />
Other Serious<br />
PT<br />
Headache<br />
Depression<br />
Lethargy<br />
Paraesthesia<br />
Feeling Abnormal<br />
Dizziness<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 345
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Epistaxis<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734416-3Report Type:Periodic Company Report #243638 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nervous System Disorder Consumer Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734417-5Report Type:Periodic Company Report #243640 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam PS Hoffmann La Roche<br />
Anxiety Other Inc ORAL ORAL<br />
Emotional Disorder<br />
Date:06/05/01ISR Number: 3734418-7Report Type:Periodic Company Report #243646 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lethargy Consumer Lariam PS Hoffmann La Roche<br />
Diarrhoea Other Inc ORAL ORAL<br />
Dizziness Tinidazole C<br />
Constipation<br />
1 Concomitant<br />
Pyrexia<br />
Drug(Generic<br />
Abnormal Faeces Components Unknown) C<br />
Headache<br />
Cipro (Ciprofloxacin<br />
Haematochezia Hydrochloride) C<br />
Abdominal Pain Upper<br />
Fatigue<br />
Visual Field Defect<br />
Hallucination<br />
Nausea<br />
Feeling Abnormal<br />
Decreased Appetite<br />
Date:06/05/01ISR Number: 3734419-9Report Type:Periodic Company Report #244422 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Jaundice Consumer Lariam PS Hoffmann La Roche<br />
Panic Attack Other Inc ORAL 1 PER WEEK<br />
Nausea<br />
ORAL<br />
Asthenia
Visual Impairment<br />
Abnormal Dreams<br />
Anger<br />
Paranoia<br />
03-Apr-2012 09:37 AM Page: 346
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734423-0Report Type:Periodic Company Report #236092 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hyperhidrosis Consumer Lariam PS Hoffmann La Roche<br />
Dermatitis Inc ORAL 1 DOSE FORM 1<br />
Diarrhoea<br />
PER WEEK ORAL<br />
Pruritus<br />
Sleep Disorder<br />
Nightmare<br />
Abnormal Faeces<br />
Date:06/05/01ISR Number: 3734429-1Report Type:Periodic Company Report #238527 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aspartate Health Lariam PS Hoffmann La Roche<br />
Aminotransferase Professional Inc ORAL ORAL<br />
Increased<br />
Blood Bilirubin Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Date:06/05/01ISR Number: 3734432-1Report Type:Periodic Company Report #238874 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Joint Swelling Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Oral Contraceptive<br />
Nos (Oral<br />
Contraceptive Nos) C<br />
Date:06/05/01ISR Number: 3734434-5Report Type:Periodic Company Report #239398 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Disorder Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 750MG 1 PER<br />
ONE DOSE ORAL<br />
Probenecid 500 Mg SS ORAL 500 MG 1 PER<br />
12 HOUR ORAL<br />
Amoxicillin 875mg SS ORAL 2625 MG 1 PER<br />
12 HOUR ORAL<br />
Date:06/05/01ISR Number: 3734437-0Report Type:Periodic Company Report #239624 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Rectal Haemorrhage Consumer Lariam PS Hoffmann La Roche<br />
Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Antibiotics Nos C<br />
03-Apr-2012 09:37 AM Page: 347
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734441-2Report Type:Periodic Company Report #240270 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Upper Respiratory Tract Consumer Lariam PS Hoffmann La Roche<br />
Infection Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734446-1Report Type:Periodic Company Report #240729 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Health Lariam PS Hoffmann La Roche<br />
Decreased Appetite Professional Inc ORAL 250 MG 1 PER<br />
Speech Disorder<br />
DAY ORAL<br />
Date:06/05/01ISR Number: 3734447-3Report Type:Periodic Company Report #242526 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pruritus Consumer Lariam PS Hoffmann La Roche<br />
Urticaria Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734448-5Report Type:Periodic Company Report #242575 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Influenza Like Illness Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734449-7Report Type:Periodic Company Report #242830 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hyperkeratosis Health Lariam PS Hoffmann La Roche<br />
Dermatitis Professional Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734460-6Report Type:Periodic Company Report #243644 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam PS Hoffmann La Roche<br />
Dissociation Other Inc ORAL ORAL<br />
Hallucination<br />
Headache<br />
Coordination Abnormal<br />
Abnormal Dreams<br />
Malaise<br />
Abdominal Pain<br />
Dermatitis
Flushing<br />
Dizziness<br />
Chills<br />
Decreased Appetite<br />
Disturbance In Attention<br />
Urticaria<br />
03-Apr-2012 09:37 AM Page: 348
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734461-8Report Type:Periodic Company Report #247060 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Health Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Headache Professional Inc ORAL 250 MG 1 PER<br />
Psychotic Disorder<br />
WEEK ORAL<br />
Dermatitis Dimetapp C<br />
Abdominal Pain Necon C<br />
Agitation Oral Contraceptions C<br />
White Blood Cell Count Pepto Bismol C<br />
Decreased<br />
Hepatic Enzyme Increased<br />
Sinusitis<br />
Hallucination<br />
Nasopharyngitis<br />
Confusional State<br />
Date:06/05/01ISR Number: 3734465-5Report Type:Periodic Company Report #248977 Age:71 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Health Lariam PS Hoffmann La Roche<br />
Nausea Professional Inc ORAL 250 MG 1 PER<br />
ONE DOSE ORAL<br />
Date:06/05/01ISR Number: 3734469-2Report Type:Periodic Company Report #249134 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Health Lariam PS Hoffmann La Roche<br />
Fear Professional Inc ORAL ORAL<br />
Insomnia<br />
Other<br />
Disturbance In Attention<br />
Date:06/05/01ISR Number: 3734473-4Report Type:Periodic Company Report #249872 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734475-8Report Type:Periodic Company Report #250139 Age:15 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hyperventilation Consumer Lariam PS Hoffmann La Roche<br />
Hospitalization - Chest Pain Other Inc ORAL 750 MG 1 PER<br />
Initial or Prolonged Suicidal Ideation ONE DOSE ORAL<br />
Hallucination
Insomnia<br />
Bipolar I Disorder<br />
Psychomotor Hyperactivity<br />
Emotional Disorder<br />
03-Apr-2012 09:37 AM Page: 349
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734478-3Report Type:Periodic Company Report #254302 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thinking Abnormal Health Lariam PS Hoffmann La Roche<br />
Hypomania Professional Inc ORAL 250 MG 1 PER<br />
Paranoia<br />
WEEK ORAL<br />
Inappropriate Affect<br />
Date:06/05/01ISR Number: 3734482-5Report Type:Periodic Company Report #255796 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Peripheral Nerve Injury Consumer Lariam PS Hoffmann La Roche<br />
Diarrhoea Inc ORAL 1 DOSE FORM 1<br />
Panic Attack<br />
PER WEEK ORAL<br />
Weight Decreased<br />
Muscular Weakness<br />
Balance Disorder<br />
Anxiety<br />
Nausea<br />
Date:06/05/01ISR Number: 3734485-0Report Type:Periodic Company Report #257836 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Health Lariam PS Hoffmann La Roche<br />
Psychiatric Symptom Professional Inc ORAL ORAL<br />
Other<br />
Date:06/05/01ISR Number: 3734488-6Report Type:Periodic Company Report #258349 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Consumer Lariam PS Hoffmann La Roche<br />
Drug Ineffective Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734490-4Report Type:Periodic Company Report #243098 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Motion Sickness Consumer Lariam PS Hoffmann La Roche<br />
Balance Disorder Other Inc ORAL ORAL<br />
Vision Blurred<br />
Date:06/05/01ISR Number: 3734493-XReport Type:Periodic Company Report #243433 Age:18 YR Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Delusion Health Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Paranoia Professional Inc ORAL ORAL<br />
Psychotic Disorder<br />
Agitation<br />
03-Apr-2012 09:37 AM Page: 350
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734495-3Report Type:Periodic Company Report #2433584 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Asthenia Consumer Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Circulatory Collapse Other Inc ORAL ORAL<br />
Headache<br />
Insomnia<br />
Tearfulness<br />
Feeling Abnormal<br />
Decreased Appetite<br />
Amnesia<br />
Paraesthesia<br />
Dizziness<br />
Date:06/05/01ISR Number: 3734497-7Report Type:Periodic Company Report #243588 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Consumer Lariam PS Hoffmann La Roche<br />
Other Inc ORAL ORAL 35 DAY<br />
Date:06/05/01ISR Number: 3734499-0Report Type:Periodic Company Report #243623 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Disorder Consumer Lariam PS Hoffmann La Roche<br />
Dizziness Other Inc ORAL ORAL<br />
Decreased Appetite<br />
Lethargy<br />
Weight Decreased<br />
Abnormal Dreams<br />
Hallucination<br />
Date:06/05/01ISR Number: 3734502-8Report Type:Periodic Company Report #243631 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Consumer Lariam PS Hoffmann La Roche<br />
Dyspnoea Other Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734504-1Report Type:Periodic Company Report #243632 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Consumer Lariam PS Hoffmann La Roche<br />
Paranoia Other Inc ORAL ORAL 91 DAY<br />
Diarrhoea<br />
Insomnia<br />
Hyperhidrosis
Movement Disorder<br />
Nightmare<br />
Loss Of Consciousness<br />
Depression<br />
Fatigue<br />
Gastroenteritis<br />
Visual Impairment<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 351
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3734507-7Report Type:Periodic Company Report #243636 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Consumer Lariam PS Hoffmann La Roche<br />
Malaria Other Inc ORAL ORAL<br />
Chills<br />
Hyperhidrosis<br />
Date:06/05/01ISR Number: 3734508-9Report Type:Periodic Company Report #243637 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Consumer Lariam PS Hoffmann La Roche<br />
Dizziness Other Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3734509-0Report Type:Periodic Company Report #243639 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Consumer Lariam PS Hoffmann La Roche<br />
Hallucination Other Inc ORAL ORAL<br />
Headache<br />
Paraesthesia<br />
Panic Attack<br />
Tremor<br />
Vomiting<br />
Diarrhoea<br />
Date:06/05/01ISR Number: 3735154-3Report Type:Periodic Company Report #236542 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Crying Consumer Lariam PS Hoffmann La Roche<br />
Hallucination, Auditory Health Inc ORAL 250 MG 1 PER<br />
Amnesia Professional WEEK ORAL<br />
Thinking Abnormal<br />
Hallucination, Tactile<br />
Hallucination, Visual<br />
Suicidal Ideation<br />
Hyperglycaemia<br />
Hyperventilation<br />
Jamais Vu<br />
Blood Urea Increased<br />
Anxiety<br />
Psychotic Disorder<br />
Tremor<br />
Date:06/05/01ISR Number: 3735158-0Report Type:Periodic Company Report #236861 Age:26 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Health Lariam PS Hoffmann La Roche<br />
Tachycardia Professional Inc ORAL 250 MG 1 PER<br />
Psychotic Disorder<br />
WEEK ORAL<br />
Sleep Terror<br />
Anxiety<br />
Ortho-Cyclen<br />
(Ethinyl<br />
Estradiol/Norgestima<br />
03-Apr-2012 09:37 AM Page: 352
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
te)<br />
C<br />
Date:06/05/01ISR Number: 3735160-9Report Type:Periodic Company Report #237292 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Confusional State Health Lariam PS Hoffmann La Roche<br />
Tremor Professional Inc ORAL 250 MG 1 PER<br />
Hallucination<br />
WEEK ORAL<br />
Depression<br />
Amnesia<br />
Abnormal Dreams<br />
Psychotic Disorder<br />
Crying<br />
Anxiety<br />
Date:06/05/01ISR Number: 3735161-0Report Type:Periodic Company Report #238170 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hypoaesthesia Consumer Lariam PS Hoffmann La Roche<br />
Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Vitamin E<br />
C<br />
Date:06/05/01ISR Number: 3735163-4Report Type:Periodic Company Report #240640 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam PS Hoffmann La Roche<br />
Paranoia Professional Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/05/01ISR Number: 3735166-XReport Type:Periodic Company Report #241120 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Asthenia Consumer Lariam PS Hoffmann La Roche<br />
Vomiting Inc ORAL 1250 MG<br />
Headache<br />
DAILY, ORAL<br />
Malaria<br />
Pyrexia<br />
Tremor<br />
Dysgraphia<br />
Diarrhoea<br />
Hallucination<br />
Nausea
Date:06/05/01ISR Number: 3735169-5Report Type:Periodic Company Report #241190 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Dissociation Consumer Lariam PS Hoffmann La Roche<br />
Intervention to Depression Inc ORAL 250 MG 1 PER<br />
Prevent Permanent Anxiety WEEK ORAL<br />
Impairment/Damage Hallucination<br />
03-Apr-2012 09:37 AM Page: 353
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/01ISR Number: 3735171-3Report Type:Periodic Company Report #241282 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Health Lariam PS Hoffmann La Roche<br />
Hallucination Professional Inc ORAL 250 MG 1 PER<br />
Confusional State<br />
ONE DOSE ORAL<br />
Diarrhoea<br />
Depression<br />
Insomnia<br />
Delirium<br />
Date:06/05/01ISR Number: 3735172-5Report Type:Periodic Company Report #241412 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypertension Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL ORAL<br />
Date:06/05/01ISR Number: 3735173-7Report Type:Periodic Company Report #243081 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Consumer Lariam PS Hoffmann La Roche<br />
Amnesia Other Inc ORAL 1 PER WEEK<br />
Fear<br />
ORAL<br />
Road Traffic Accident<br />
Feeling Of Despair<br />
Chest Discomfort<br />
Lethargy<br />
Palpitations<br />
Photophobia<br />
Skin Disorder<br />
Visual Acuity Reduced<br />
Depression<br />
Date:06/06/01ISR Number: 3733847-5Report Type:Expedited (15-DaCompany Report #261363 Age:10 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Onycholysis Lariam PS Roche 385 DAY<br />
Hepatitis<br />
Vaccination<br />
C<br />
Vivotif "Berna" C<br />
Date:06/08/01ISR Number: 3736478-6Report Type:Expedited (15-DaCompany Report #261224 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation Foreign Lariam PS Hoffmann La Roche
Initial or Prolonged Myalgia Other Inc ORAL 1.5 GRAM ORAL<br />
Vertigo<br />
Nivaquine<br />
Arrhythmia<br />
(Chloroquine<br />
Headache Sulfate) SS ORAL ORAL<br />
Pyrexia<br />
Paludrine<br />
Arthralgia<br />
(Chloroguanide<br />
Dysarthria Hydrochloride) C<br />
Tremor<br />
03-Apr-2012 09:37 AM Page: 354
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/01ISR Number: 3736491-9Report Type:Expedited (15-DaCompany Report #260506 Age:33 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Infectious Peritonitis Health Inc ORAL 250 MG 1 PER<br />
Asthenia Professional WEEK ORAL<br />
Abdominal Pain Other Stamaril Unidose<br />
Pregnancy<br />
(Yellow Fever<br />
Sedation Vaccination) SS<br />
Gastritis<br />
Nausea<br />
Vertigo<br />
Abscess<br />
Caesarean Section<br />
Arterial Occlusive<br />
Disease<br />
Pyelonephritis<br />
Balance Disorder<br />
Post Procedural<br />
Complication<br />
Uterine Hypertonus<br />
Date:06/08/01ISR Number: 3736492-0Report Type:Expedited (15-DaCompany Report #259776 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thyroid Function Test Foreign Lariam PS Hoffmann La Roche<br />
Abnormal Health Inc ORAL ORAL<br />
Xanthelasma<br />
Professional<br />
Sleep Disorder<br />
Other<br />
Muscle Spasms<br />
Alanine Aminotransferase<br />
Increased<br />
Lipids Increased<br />
Malaise<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Blood Pressure Increased<br />
Date:06/08/01ISR Number: 3736494-4Report Type:Expedited (15-DaCompany Report #247738 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyelonephritis Foreign Lariam PS Hoffmann La Roche<br />
Uterine Hypertonus Health Inc ORAL 250 MG 1 PER<br />
Breech Presentation Professional WEEK ORAL<br />
Premature Rupture Of Other Stamaril Unidose<br />
Membranes<br />
(Yellow Fever<br />
Maternal Drugs Affecting Vaccine) C<br />
Foetus<br />
Caesarean Section
Date:06/08/01ISR Number: 3736496-8Report Type:Expedited (15-DaCompany Report #261363 Age:10 YR Gender:Female I/FU:I<br />
Outcome PT Report Source<br />
Other Serious Onycholysis Foreign<br />
Health<br />
03-Apr-2012 09:37 AM Page: 355
Professional<br />
Other<br />
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Product Role Manufacturer Route Dose Duration<br />
Lariam PS Hoffmann La Roche<br />
Inc ORAL ORAL 385 DAY<br />
Hepatitis<br />
Vaccinatioin<br />
(Hepatitis Vaccine) C<br />
Vivotif "Berna"<br />
(Typhoid Vaccines) C<br />
Date:06/08/01ISR Number: 3736661-XReport Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Eyelid Oedema Larium PS<br />
Intervention to Burning Sensation Depakote SS<br />
Prevent Permanent<br />
Impairment/Damage<br />
Face Oedema<br />
Medication Error<br />
Liver Disorder<br />
Skin Disorder<br />
Date:06/11/01ISR Number: 3736292-1Report Type:Expedited (15-DaCompany Report #247340 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Motor Dysfunction Lariam Tablets PS Roche FOR MANY<br />
Nightmare<br />
YEARS.<br />
Vertigo Positional<br />
Dizziness<br />
Vomiting<br />
Date:06/11/01ISR Number: 3736334-3Report Type:Expedited (15-DaCompany Report #261664 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vaginal Haemorrhage Lariam PS Roche 8 DAY<br />
Date:06/11/01ISR Number: 3736338-0Report Type:Expedited (15-DaCompany Report #261561 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placenta Praevia Lariam PS Roche 30 DAY<br />
Initial or Prolonged<br />
Date:06/11/01ISR Number: 3736345-8Report Type:Expedited (15-DaCompany Report #261498 Age:72 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death International Normalised Lariam PS Roche 37 DAY<br />
Ratio Increased Cozaar Comp C 285 DAY<br />
Drug Interaction Warfarin I VARYING DOSES<br />
Haemorrhagic Stroke<br />
AS PER INR.<br />
03-Apr-2012 09:37 AM Page: 356
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/11/01ISR Number: 3736693-1Report Type:Direct Company Report # Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Palpitations Larium PS BUCCAL 1 TABLE<br />
Panic Attack<br />
WEEKLY BUCCAL<br />
Anxiety Loratadine C<br />
Muscle Spasms<br />
Nightmare<br />
Date:06/12/01ISR Number: 3738728-9Report Type:Expedited (15-DaCompany Report #261561 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placenta Praevia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Health Inc ORAL 1 DOSE FORM<br />
Professional<br />
1 PER WEEK<br />
ORAL<br />
Date:06/12/01ISR Number: 3738880-5Report Type:Expedited (15-DaCompany Report #261664 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vaginal Haemorrhage Health Lariam PS Hoffmann La Roche<br />
Professional Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/12/01ISR Number: 3738885-4Report Type:Expedited (15-DaCompany Report #247340 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nightmare Consumer Lariam PS Hoffmann La Roche<br />
Vomiting Inc ORAL ORAL<br />
Gait Disturbance<br />
Vertigo Positional<br />
Dizziness<br />
Motor Dysfunction<br />
Date:06/13/01ISR Number: 3738150-5Report Type:Direct Company Report # Age:62 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam 250mg Roche PS Roche ORAL 1 TABLET PER<br />
Fatigue<br />
WEEK BY MOUTH<br />
Disorientation Ambien C<br />
Hypertension Cardizem C<br />
Dizziness Biaxin C<br />
Panic Attack Premarin C<br />
Dyspnoea Provera C<br />
Chest Pain Captopril C
Cipro C<br />
03-Apr-2012 09:37 AM Page: 357
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/13/01ISR Number: 3738958-6Report Type:Expedited (15-DaCompany Report #261498 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Haemorrhagic Stroke Foreign Lariam PS Hoffmann La Roche<br />
Drug Interaction Other Inc ORAL 250 MG 1 PER<br />
International Normalised<br />
Ratio Increased<br />
Warfarin (Warfarin<br />
Sodium) SS ORAL ORAL<br />
Cozaar Comp<br />
(Hydrochlorothiazide<br />
/Losartan Potassium) C<br />
WEEK ORAL<br />
Date:06/15/01ISR Number: 3740168-3Report Type:Expedited (15-DaCompany Report #259927 Age:73 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diverticulitis Lariam Tablets PS Roche 36 DAY<br />
Initial or Prolonged Gastrointestinal Aspirin Protect 100 SS 372 DAY<br />
Haemorrhage Marcumar SS Roche<br />
Date:06/15/01ISR Number: 3740171-3Report Type:Expedited (15-DaCompany Report #260569 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Lariam PS Roche 8 DAY<br />
Initial or Prolonged Weight Decreased<br />
Date:06/18/01ISR Number: 3740645-5Report Type:Expedited (15-DaCompany Report #259750 Age:94 DY Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Staring Lariam Tablets PS Roche 3 DAY<br />
Inappropriate Affect<br />
Panic Reaction<br />
Date:06/18/01ISR Number: 3741496-8Report Type:Expedited (15-DaCompany Report #259927 Age:73 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haemoglobin Decreased Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Diverticulitis Consumer Inc ORAL 1 DOSE FORM 1<br />
Arthralgia Health PER WEEK ORAL<br />
Gastrointestinal Professional Aspirin Protect 100<br />
Haemorrhage (Aspirin) SS ORAL 100 MG DAILY<br />
ORAL<br />
Marcumar<br />
(Phenprocoumon) SS ORAL 0.5 DOSE FORM<br />
DAILY ORAL
Date:06/18/01ISR Number: 3741720-1Report Type:Expedited (15-DaCompany Report #260569 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Pregnancy Health Inc ORAL 250 MG 1 PER<br />
Weight Decreased Professional WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 358
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/20/01ISR Number: 3742310-7Report Type:Expedited (15-DaCompany Report #259933 Age:62 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Lariam PS Roche 74 DAY<br />
Hypertension Ambien C AT NIGHT.<br />
Fatigue Biaxin C TAKEN FOR 1<br />
Panic Attack WEEK. 7 DAY<br />
Premarin<br />
C<br />
Medroxyprogesterone C<br />
Cipro C TAKEN FOR 1<br />
WEEK 7 DAY<br />
Captopril C 34 DAY<br />
Cardizem<br />
C<br />
Date:06/20/01ISR Number: 3743711-3Report Type:Expedited (15-DaCompany Report #259750 Age:94 DY Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Inappropriate Affect Foreign Lariam PS Hoffmann La Roche<br />
Panic Reaction Health Inc ORAL 62.5 MG ORAL<br />
Staring<br />
Professional<br />
Hallucination<br />
Date:06/21/01ISR Number: 3743483-2Report Type:Expedited (15-DaCompany Report #261561 Age:30 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placenta Praevia Lariam PS Roche 30 DAY<br />
Initial or Prolonged<br />
Date:06/21/01ISR Number: 3743490-XReport Type:Expedited (15-DaCompany Report #260121 Age:51 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Creatine Lariam PS Roche 1056 DAY<br />
Phosphokinase Increased Voltarene C<br />
Blood Lactate Apranax C<br />
Dehydrogenase Increased<br />
Asthenia<br />
Date:06/21/01ISR Number: 3743545-XReport Type:Direct Company Report # Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paraesthesia Larium PS ORAL ORAL<br />
Insomnia<br />
Chest Pain<br />
Hypoaesthesia<br />
Anxiety
Date:06/22/01ISR Number: 3745665-2Report Type:Expedited (15-DaCompany Report #259933 Age:62 YR Gender:Female I/FU:F<br />
Outcome<br />
Required<br />
Intervention to<br />
03-Apr-2012 09:37 AM Page: 359
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Pneumonia Mycoplasmal Consumer Lariam PS Hoffmann La Roche<br />
Chest Pain Inc ORAL 1 PER WEEK<br />
Dizziness<br />
ORAL<br />
Asthenia<br />
Ambien (Zolpidem<br />
Disorientation Tartrate) C<br />
Emotional Disorder<br />
Biaxin<br />
Blood Pressure Increased (Clarithromycin) C<br />
Anxiety<br />
Premarin (Estrogens,<br />
Fatigue Conjugated) C<br />
Hypertension<br />
Medroxyprogesterone<br />
Bronchitis<br />
(Medroxyprogesterone<br />
Fear Acetate) C<br />
Panic Attack<br />
Cipro (Ciprofloxacin<br />
Dyspnoea Hydrochloride) C<br />
Captopril<br />
C<br />
Cardizem (Diltiazem<br />
Hydrochloride) C<br />
Date:06/25/01ISR Number: 3745221-6Report Type:Expedited (15-DaCompany Report #244451 Age: Gender:Not SpecifiI/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Intra-Uterine Death Lariam PS Roche 50 DAY<br />
Date:06/25/01ISR Number: 3745817-1Report Type:Direct Company Report # Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Sleep Disorder Larium PS ORAL 6/13/2001<br />
Intervention to Influenza Like Illness 10/4/2001<br />
Prevent Permanent Nervousness ORAL<br />
Impairment/Damage Oropharyngeal Pain<br />
Anxiety<br />
Nasal Congestion<br />
Ear Pain<br />
Malaise<br />
Date:06/25/01ISR Number: 3746822-1Report Type:Expedited (15-DaCompany Report #261561 Age:30 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placenta Praevia Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Caesarean Section Health Hydrochloride) 250<br />
Pregnancy Professional Mg PS Hoffmann La Roche<br />
Other Inc ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:06/25/01ISR Number: 3746823-3Report Type:Expedited (15-DaCompany Report #260121 Age:51 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Asthenia<br />
03-Apr-2012 09:37 AM Page: 360
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) 250<br />
Professional Mg PS Hoffmann La Roche<br />
Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Voltarene<br />
(Diclofenac Sodium) C<br />
Apranax (Naproxen<br />
Sodium)<br />
C<br />
Date:06/26/01ISR Number: 3746273-XReport Type:Expedited (15-DaCompany Report #262670 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Ineffective Lariam PS Roche 133 DAY<br />
Malaria<br />
Bacterial Infection<br />
Date:06/26/01ISR Number: 3748077-0Report Type:Expedited (15-DaCompany Report #244451 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam PS Hoffmann La Roche<br />
Foetus Health Inc ORAL 250 MG, 1 PER<br />
Pregnancy Professional WEEK ORAL<br />
Intra-Uterine Death<br />
Congenital Anomaly<br />
Abortion Missed<br />
Date:06/27/01ISR Number: 3748407-XReport Type:Direct Company Report # Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Psychotic Disorder Lariam -(<strong>Mefloquine</strong>) PS Hoffman-Laroche 1 X WEEK FOR<br />
Hospitalization - Anxiety 3 WKS (3<br />
Initial or Prolonged Hallucination PILLS) 3 WK<br />
Disability<br />
Depression<br />
Other Serious<br />
Nightmare<br />
Required<br />
Paraesthesia<br />
Intervention to<br />
Heart Rate Irregular<br />
Prevent Permanent<br />
Impairment/Damage<br />
Respiratory Arrest<br />
Heart Rate Increased<br />
Convulsion<br />
Hallucination, Auditory
Date:06/28/01ISR Number: 3748646-8Report Type:Expedited (15-DaCompany Report #261500 Age:30 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaria<br />
Asthenia<br />
Gastric Ulcer<br />
Irritability<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 361
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Insomnia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche THE PATIENT<br />
RECEIVED 3<br />
TABLETS, THEN<br />
TWO TABLETS<br />
AND LATER ONE 2 DAY<br />
Lariam SS Roche<br />
Quinine<br />
C<br />
Mopral<br />
C<br />
Date:06/28/01ISR Number: 3749664-6Report Type:Expedited (15-DaCompany Report #262670 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bacterial Infection Foreign Lariam PS Hoffmann La Roche<br />
Ill-Defined Disorder Consumer Inc ORAL 250 MG 1 PER<br />
Drug Ineffective WEEK ORAL 133 DAY<br />
Malaria<br />
Diarrhoea<br />
Date:07/02/01ISR Number: 3751059-6Report Type:Expedited (15-DaCompany Report #261500 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Irritability Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Drug Effect Decreased Health Inc ORAL ORAL<br />
Malaria Professional Lariam (<strong>Mefloquine</strong><br />
Hepatic Function Abnormal Hydrochloride) SS ORAL ORAL<br />
Gastric Ulcer Quinine C<br />
Ileus Paralytic Mopral (Oemprazole) C<br />
Insomnia<br />
Nausea<br />
Asthenia<br />
Date:07/05/01ISR Number: 3751625-8Report Type:Expedited (15-DaCompany Report #260745 Age:52 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Guillain-Barre Syndrome Lariam PS Roche FOUR TABLETS<br />
Initial or Prolonged IN TOTAL. 29 DAY<br />
Tetanus Vaccine C<br />
Yellow Fever Vaccine C<br />
Cholera Vaccine C<br />
Poliomyelitis<br />
Vaccine<br />
C<br />
Hepatitis Vaccine C
Date:07/05/01ISR Number: 3751630-1Report Type:Expedited (15-DaCompany Report #259147 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Polymyalgia Rheumatica Lariam PS Roche<br />
Nausea<br />
Erythema Infectiosum<br />
03-Apr-2012 09:37 AM Page: 362
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/05/01ISR Number: 3751640-4Report Type:Expedited (15-DaCompany Report #261224 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dysarthria Consumer Lariam PS Roche 2 DAY<br />
Initial or Prolonged Tremor Nivaquine SS 50 DAY<br />
Vertigo Paludrine C<br />
Asthenia<br />
Myalgia<br />
Arthralgia<br />
Headache<br />
Pyrexia<br />
Agitation<br />
Vestibular Disorder<br />
Arrhythmia<br />
Date:07/05/01ISR Number: 3751672-6Report Type:Expedited (15-DaCompany Report #259147 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Erythema Infectiosum Health Lariam PS Roche<br />
Polymyalgia Rheumatica Professional<br />
Nausea<br />
Date:07/05/01ISR Number: 3751687-8Report Type:Expedited (15-DaCompany Report #260745 Age:52 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Guillain-Barre Syndrome Consumer Lariam PS Roche FOUR TABLETS<br />
Initial or Prolonged IN TOTAL. 29 DAY<br />
Tetanus Vaccine C<br />
Yellow Fever Vaccine C<br />
Cholera Vaccine C<br />
Poliomyelitis<br />
Vaccine<br />
C<br />
Hepatitis Vaccine C<br />
Date:07/05/01ISR Number: 3751701-XReport Type:Expedited (15-DaCompany Report #261224 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dysarthria Consumer Lariam PS Roche 2 DAY<br />
Initial or Prolonged Asthenia Nivaquine SS 50 DAY<br />
Headache Paludrine C<br />
Arthralgia<br />
Vestibular Disorder<br />
Pyrexia<br />
Agitation<br />
Tremor<br />
Myalgia<br />
Vertigo
Arrhythmia<br />
03-Apr-2012 09:37 AM Page: 363
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/05/01ISR Number: 3751721-5Report Type:Expedited (15-DaCompany Report #263420 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia Lariam PS Roche 36 DAY<br />
Confusional State Edecrin C<br />
Naprosyn<br />
C<br />
Asa<br />
C<br />
Date:07/10/01ISR Number: 3754734-2Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Lariam Tablets PS Roche ESTIMATED 5<br />
Initial or Prolonged Vertigo DOSES IN<br />
Disability Headache TOTAL. 21 DAY<br />
Anxiety<br />
Panic Attack<br />
Date:07/10/01ISR Number: 3754747-0Report Type:Expedited (15-DaCompany Report #261469 Age:32 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Decreased Appetite Lariam PS Roche 43 DAY<br />
Anxiety Leios C<br />
Coordination Abnormal<br />
Night Sweats<br />
Hepatitis A Antibody<br />
Positive<br />
Depression<br />
Panic Attack<br />
Liver Function Test<br />
Abnormal<br />
Memory Impairment<br />
Flushing<br />
Dizziness<br />
Chills<br />
Nausea<br />
Diarrhoea<br />
Date:07/10/01ISR Number: 3755913-0Report Type:Expedited (15-DaCompany Report #263420 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Confusional State Consumer Lariam PS Hoffmann La Roche<br />
Amnesia Inc ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Edecrin (Ethacrynate<br />
Sodium Or Ethacrynic<br />
Acid)<br />
C<br />
Naprosyn (Naproxen) C
Asa (Aspirin) C<br />
03-Apr-2012 09:37 AM Page: 364
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/10/01ISR Number: 3756155-5Report Type:Expedited (15-DaCompany Report #261224 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Electroencephalogram Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Abnormal Other Inc ORAL 1.5 GRAM ORAL<br />
Malaria<br />
Nivaquine<br />
Agitation<br />
(Chloroquine<br />
Myalgia Sulfate) SS ORAL ORAL<br />
Headache<br />
Paludrine<br />
Dysarthria<br />
(Chloroguanide<br />
Asthenia Hydrochloride) C<br />
Arrhythmia<br />
Arthralgia<br />
Tremor<br />
Vestibular Disorder<br />
Vertigo<br />
Pyrexia<br />
Date:07/10/01ISR Number: 3756157-9Report Type:Expedited (15-DaCompany Report #260745 Age:52 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mumps Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Bacterial Infection Other Inc ORAL 1 DOSE FORM 1<br />
Guillain-Barre Syndrome<br />
PER WEEK ORAL<br />
Tetanus Vaccine<br />
(Tetanus Toxoid) C<br />
Yellow Fever Vaccine C<br />
Cholera Vaccine<br />
(Cholera Vaccine) C<br />
Poliomyelitis<br />
Vaccine<br />
(Poliomyelitis<br />
Vaccines)<br />
C<br />
Hepatitis Vaccine<br />
(Hepatitis Vaccine) C<br />
Date:07/10/01ISR Number: 3756283-4Report Type:Expedited (15-DaCompany Report #259147 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Polymyalgia Rheumatica Consumer Lariam PS Hoffmann La Roche<br />
Erythema Infectiosum Inc ORAL ORAL<br />
Nausea<br />
Date:07/11/01ISR Number: 3755609-5Report Type:Expedited (15-DaCompany Report #262670 Age:23 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Ineffective Lariam PS Roche 133 DAY
Initial or Prolonged<br />
Bacterial Infection<br />
Ill-Defined Disorder<br />
Diarrhoea<br />
Malaria<br />
03-Apr-2012 09:37 AM Page: 365
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/11/01ISR Number: 3755616-2Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Cardiac Arrest Lariam PS Roche 15 DAY<br />
Grand Mal Convulsion<br />
Date:07/12/01ISR Number: 3756377-3Report Type:Expedited (15-DaCompany Report #249874 Age:14 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Musculoskeletal Pain Lariam PS Roche TOTAL OF 5<br />
Aphasia WEEKLY DOSES. 29 DAY<br />
Nightmare Claritin-D C<br />
Headache Biaxin C<br />
Amnesia<br />
Insomnia<br />
Sensory Loss<br />
Dyspnoea<br />
Muscular Weakness<br />
Abnormal Dreams<br />
Weight Decreased<br />
Dizziness<br />
Palpitations<br />
Hyperventilation<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal<br />
Alcoholism<br />
Thirst<br />
Influenza Like Illness<br />
Injury<br />
Blood Pressure Decreased<br />
Anhedonia<br />
Encephalopathy<br />
Psychotic Disorder<br />
Emotional Distress<br />
Paraesthesia<br />
Mental Disorder<br />
Educational Problem<br />
Electroencephalogram<br />
Abnormal<br />
Agitation<br />
Visual Impairment<br />
Balance Disorder<br />
Sleep Disorder<br />
Tinnitus<br />
Hypophagia<br />
Cold Sweat<br />
Emotional Disorder
Date:07/12/01ISR Number: 3757198-8Report Type:Expedited (15-DaCompany Report #241365 Age:24 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Vertigo<br />
Panic Attack<br />
Depression<br />
Headache<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 366
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 250MG 1 PER<br />
Professional<br />
WEEK ORAL<br />
Date:07/12/01ISR Number: 3757293-3Report Type:Expedited (15-DaCompany Report #261469 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Foreign Lariam PS Hoffmann La Roche<br />
Panic Attack Consumer Inc ORAL 1 DOSE FORM 1<br />
Nausea Health PER WEEK ORAL<br />
Diarrhoea Professional Leios (Ethinyl<br />
Depression<br />
Estradiol/Levonorges<br />
Hepatitis A trel) C<br />
Chills<br />
Coordination Abnormal<br />
Agitation<br />
Memory Impairment<br />
Night Sweats<br />
Abnormal Faeces<br />
Anxiety<br />
Decreased Appetite<br />
Flushing<br />
Date:07/13/01ISR Number: 3757307-0Report Type:Expedited (15-DaCompany Report #263895 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rhabdomyolysis Lariam PS Roche<br />
Initial or Prolonged<br />
Date:07/13/01ISR Number: 3758782-8Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion Consumer Lariam PS Hoffmann La Roche<br />
Cardiac Arrest Inc ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Date:07/13/01ISR Number: 3758798-1Report Type:Expedited (15-DaCompany Report #262670 Age:23 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Ill-Defined Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Diarrhoea Consumer Inc ORAL 250 MG 1 PER<br />
Required Malaria Health WEEK ORAL 133 DAY
Intervention to Bacterial Infection Professional<br />
Prevent Permanent Drug Ineffective<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 367
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/16/01ISR Number: 3759497-2Report Type:Expedited (15-DaCompany Report #249874 Age:14 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Palpitations Other Lariam PS Hoffmann La Roche<br />
Headache Inc ORAL 250 MG 1 PER<br />
Encephalopathy<br />
WEEK ORAL<br />
Emotional Distress<br />
Claritin-D<br />
Educational Problem<br />
(Loratadine/Pseudoep<br />
Hyperventilation hedrine Sulfate) C<br />
Blood Pressure Decreased<br />
Biaxin<br />
Cognitive Disorder (Clarithromycin) C<br />
Influenza Like Illness<br />
Musculoskeletal Pain<br />
Insomnia<br />
Dyspnoea<br />
Weight Decreased<br />
Dizziness<br />
Visual Impairment<br />
Muscular Weakness<br />
Cold Sweat<br />
Hypophagia<br />
Nightmare<br />
Thirst<br />
Electroencephalogram<br />
Abnormal<br />
Paraesthesia<br />
Sensory Loss<br />
Hypoaesthesia<br />
Agitation<br />
Balance Disorder<br />
Dysarthria<br />
Amnesia<br />
Abnormal Dreams<br />
Psychotic Disorder<br />
Emotional Disorder<br />
Injury<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal<br />
Mental Disorder<br />
Coordination Abnormal<br />
Date:07/17/01ISR Number: 3760015-3Report Type:Direct Company Report # Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypophagia Lariam PS ORAL 1 PILL/ 4<br />
Other Serious Lethargy DAYS/ ORAL<br />
Anxiety<br />
Hallucination<br />
Fear
Date:07/17/01ISR Number: 3760208-5Report Type:Expedited (15-DaCompany Report #263895 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rhabdomyolysis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Consumer Inc ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 368
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/19/01ISR Number: 3760951-8Report Type:Direct Company Report # Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Amnesia Lariam (250 Mg)<br />
Intervention to Abnormal Dreams (<strong>Mefloquine</strong> Hcl)<br />
Prevent Permanent Sleep Disorder (Hoffmann-Laroche<br />
Impairment/Damage Hallucinations, Mixed Inc.) PS Hoffman-Laroche Inc ORAL 1 250 MG<br />
Chills<br />
TABLET TAKEN<br />
Depression<br />
OARLLY<br />
Anxiety<br />
Headache<br />
Disturbance In Attention<br />
Paraesthesia<br />
Gastrointestinal Disorder<br />
Confusional State<br />
Balance Disorder<br />
Dizziness<br />
Memory Impairment<br />
Date:07/19/01ISR Number: 3761240-8Report Type:Expedited (15-DaCompany Report #264097 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Retinal Pigmentation <strong>Mefloquine</strong><br />
Angiogram Abnormal Hydrochloride PS Roche 190 DAY<br />
Date:07/19/01ISR Number: 3764530-8Report Type:Periodic Company Report #1409764A Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicide Attempt Foreign Tylenol PS Mcneil Consumer<br />
Apathy Other Products Co Div<br />
Paranoia Mcneilab Inc UNKNOWN DOSE<br />
Intentional Overdose Pseudoephedrine SS<br />
Lariam (Meloquine)<br />
250 Mg SS ORAL UNKNOWN DOSE,<br />
PO<br />
Date:07/23/01ISR Number: 3762418-XReport Type:Direct Company Report # Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Larium/<strong>Mefloquine</strong><br />
Paraesthesia 250 Mg Roche PS Roche ORAL 250 MG ONCE A<br />
Syncope<br />
WEEK ORAL<br />
Dizziness Orthocept C<br />
Insomnia<br />
Anxiety<br />
Muscle Spasms<br />
Paranoia
Date:07/23/01ISR Number: 3763556-8Report Type:Expedited (15-DaCompany Report #264097 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source<br />
Other Serious Eye Disorder Foreign<br />
Health<br />
03-Apr-2012 09:37 AM Page: 369
Professional<br />
Other<br />
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Product Role Manufacturer Route Dose Duration<br />
Lariam PS Hoffmann La Roche<br />
Inc ORAL ORAL<br />
Date:07/23/01ISR Number: 3763734-8Report Type:Expedited (15-DaCompany Report #256731 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Drug Interaction Literature Lariam PS Hoffmann La Roche<br />
Malaria Health Inc ORAL ORAL<br />
Circulatory Collapse Professional Halofantrine<br />
Bronchopneumonia<br />
(Halofantrine<br />
Electrocardiogram Qt Hydrochloride) SS ORAL ORAL<br />
Prolonged<br />
Dirithromycin<br />
Sudden Death (Dirithromycin) C ORAL ORAL<br />
Arrhythmia Acetylcysteine C<br />
Hypertrophic<br />
Cardiomyopathy<br />
Date:07/23/01ISR Number: 3764087-1Report Type:Expedited (15-DaCompany Report #249748 Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Acute Psychosis Literature Inc<br />
Hypertonia<br />
Health<br />
Professional<br />
Date:07/24/01ISR Number: 3763645-8Report Type:Expedited (15-DaCompany Report #260795 Age:65 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Extrasystoles Health Lariam PS Roche 1 DAY<br />
Transient Ischaemic Professional Aspirin C<br />
Attack Mvi C<br />
Atrial Fibrillation<br />
Date:07/24/01ISR Number: 3763650-1Report Type:Expedited (15-DaCompany Report #263895 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rhabdomyolysis Health Lariam PS Roche 22 DAY<br />
Initial or Prolonged Headache Professional<br />
Dizziness
Date:07/24/01ISR Number: 3763651-3Report Type:Expedited (15-DaCompany Report #263420 Age:66 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Health Lariam PS Roche 36 DAY<br />
Confusional State Professional Edecrin C<br />
Amnesia Naprosyn C<br />
Depression Asa C<br />
03-Apr-2012 09:37 AM Page: 370
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/25/01ISR Number: 3764221-3Report Type:Expedited (15-DaCompany Report #264611 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Health Lariam PS Roche A TOTAL OF<br />
Initial or Prolonged Confusional State Professional THREE DOSES<br />
Automatism<br />
WERE<br />
Hypochondriasis RECEIVED. 1 DAY<br />
Agitation<br />
Persecutory Delusion<br />
Anxiety<br />
Aggression<br />
Amnesia<br />
Date:07/25/01ISR Number: 3764225-0Report Type:Expedited (15-DaCompany Report #264612 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia <strong>Mefloquine</strong><br />
Anxiety Hydrochloride PS Roche MEFLOQUINE<br />
Depression<br />
WAS GIVEN<br />
Suicidal Ideation<br />
DURING A<br />
Obsessive-Compulsive<br />
PREVIOUS TRIP<br />
Disorder<br />
ABOUT SIX<br />
Date:07/25/01ISR Number: 3765689-9Report Type:Expedited (15-DaCompany Report #264219 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death International Normalised Foreign Lariam PS Hoffmann La Roche<br />
Hospitalization - Ratio Increased Other Inc 1 PER WEEK<br />
Initial or Prolonged Pupils Unequal Warfarin Sodium<br />
Haemorrhagic Stroke (Warfarin Sodium) 5<br />
Hypothermia Mg SS 4 MG 1 PER 2<br />
Bradycardia<br />
DAY<br />
Precerebral Artery<br />
Cozaar (Losartan<br />
Occlusion Potassium) C<br />
Corneal Reflex Decreased<br />
Arteriosclerosis<br />
Hypotension<br />
Hyporeflexia<br />
Brain Oedema<br />
Brain Scan Abnormal<br />
Tachycardia<br />
Drug Interaction<br />
Respiratory Arrest<br />
Date:07/25/01ISR Number: 3765695-4Report Type:Expedited (15-DaCompany Report #264199 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Thyroiditis Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Arthralgia Health Inc ORAL 1 DOSE FORM 1<br />
Disability Psoriasis Professional PER ONE DOSE<br />
Drug Interaction<br />
ORAL<br />
Lexomil SS ORAL 2 PER DAY<br />
ORAL<br />
Effexor SS ORAL 2 DOSE FORM<br />
DAILY ORAL<br />
03-Apr-2012 09:37 AM Page: 371
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Teralithe SS ORAL 400 MG 3 PER<br />
DAY ORAL<br />
Seropram SS ORAL 2 DOSE FORM<br />
DAILY ORAL<br />
Havlane<br />
C<br />
Plaquenil<br />
C<br />
Date:07/26/01ISR Number: 3765431-1Report Type:Expedited (15-DaCompany Report #256317 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Sleep Disorder Lariam Tablets PS Roche 3288 DAY<br />
Mood Swings Unknown Medication C MEDICATIONS<br />
Diarrhoea<br />
TAKEN IN<br />
Mental Impairment<br />
1994,1995 AND<br />
Myalgia 1999.<br />
Nightmare Doxycycline C<br />
Anxiety<br />
Fatigue<br />
Depression<br />
Weight Decreased<br />
Alopecia<br />
Date:07/26/01ISR Number: 3765442-6Report Type:Expedited (15-DaCompany Report #264404 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Roche 2 DAY<br />
Initial or Prolonged Paranoia Proguanil C<br />
Flushing Chloroquine C<br />
Delusion<br />
Confusional State<br />
Date:07/26/01ISR Number: 3766229-0Report Type:Expedited (15-DaCompany Report #263420 Age:66 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Consumer Lariam PS Hoffmann La Roche<br />
Confusional State Inc ORAL 250 MG 1 PER<br />
Amnesia<br />
Fatigue<br />
Edecrin (Ethacrynate<br />
Sodium Or Ethacrynic<br />
Acid)<br />
Asa (Aspirin)<br />
C<br />
C<br />
WEEK ORAL<br />
Date:07/26/01ISR Number: 3766252-6Report Type:Expedited (15-DaCompany Report #260795 Age:65 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Atrial Fibrillation Health Lariam PS Hoffmann La Roche<br />
Heart Rate Irregular Professional Inc ORAL 250 MG, 1 PER<br />
Transient Ischaemic<br />
DOSE; ORAL<br />
Attack Aspirin C<br />
Extrasystoles<br />
Mvi (Multivitamin<br />
Speech Disorder Nos) C<br />
03-Apr-2012 09:37 AM Page: 372
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/26/01ISR Number: 3766523-3Report Type:Expedited (15-DaCompany Report #263895 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoaesthesia Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Chest Pain Consumer Inc ORAL<br />
Dizziness<br />
Health<br />
Rhabdomyolysis<br />
Professional<br />
Dyspnoea<br />
Headache<br />
Date:07/27/01ISR Number: 3767003-1Report Type:Expedited (15-DaCompany Report #264611 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Persecutory Delusion Literature Inc ORAL 250 MG 1 PER<br />
Automatism Health WEEK ORAL 1 DAY<br />
Amnesia<br />
Professional<br />
Agitation<br />
Other<br />
Aggression<br />
Hypochondriasis<br />
Insomnia<br />
Anxiety<br />
Drug Level Above<br />
Therapeutic<br />
Pyrexia<br />
Date:07/27/01ISR Number: 3767008-0Report Type:Expedited (15-DaCompany Report #264612 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Foreign Lariam PS Hoffmann La Roche<br />
Suicidal Ideation Literature Inc ORAL 250 MG 1 PER<br />
Insomnia Health WEEK ORAL<br />
Obsessive-Compulsive<br />
Professional<br />
Disorder<br />
Other<br />
Feeling Abnormal<br />
Depression<br />
Date:07/30/01ISR Number: 3766773-6Report Type:Expedited (15-DaCompany Report #264705 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bipolar Disorder <strong>Mefloquine</strong><br />
Hydrochloride PS Roche THE PATIENT<br />
TOOK A TOTAL<br />
OF 1750MG.
Date:07/30/01ISR Number: 3766776-1Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Delirium<br />
Agitation<br />
Depression<br />
Nervous System Disorder<br />
Abnormal Dreams<br />
03-Apr-2012 09:37 AM Page: 373
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Polydipsia<br />
Photopsia<br />
Delusional Disorder, Report Source Product Role Manufacturer Route Dose Duration<br />
Persecutory Type Lariam Tablets PS Roche THE PATIENT<br />
Hepatic Steatosis<br />
RECEIVED A<br />
Erectile Dysfunction<br />
TOTAL OF NINE<br />
Weight Decreased<br />
TABLETS OF<br />
Cognitive Disorder LARIAM. 67 DAY<br />
Chills<br />
Musculoskeletal Stiffness<br />
Erythema<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Blood Cortisol Increased<br />
Claustrophobia<br />
Obsessive-Compulsive<br />
Disorder<br />
Hallucinations, Mixed<br />
Hyperhidrosis<br />
Amnesia<br />
Anxiety<br />
Liver Function Test<br />
Abnormal<br />
Pyrexia<br />
Completed Suicide<br />
Flushing<br />
Urinary Hesitation<br />
Insomnia<br />
Headache<br />
Adjustment Disorder<br />
Fatigue<br />
Decreased Appetite<br />
Petit Mal Epilepsy<br />
Date:07/30/01ISR Number: 3766777-3Report Type:Expedited (15-DaCompany Report #264819 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaria Lariam PS Roche 93 DAY<br />
Initial or Prolonged Drug Ineffective<br />
Date:07/30/01ISR Number: 3768298-0Report Type:Expedited (15-DaCompany Report #256317 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Irritability<br />
Memory Impairment<br />
Diarrhoea<br />
Nightmare<br />
Alopecia<br />
Personality Disorder
Weight Decreased<br />
Tension<br />
Mood Swings<br />
Obsessive-Compulsive<br />
Disorder<br />
Sleep Disorder<br />
Mental Impairment<br />
Muscular Weakness<br />
03-Apr-2012 09:37 AM Page: 374
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Anxiety<br />
Stress<br />
Disturbance In Attention Report Source Product Role Manufacturer Route Dose Duration<br />
Myalgia Other Lariam PS Hoffmann La Roche<br />
Malaise<br />
Inc<br />
Fatigue<br />
Unknown Medication<br />
Depression<br />
(Generic<br />
Judgement Impaired<br />
Component(S)<br />
Unknown)<br />
C<br />
Doxycycline<br />
(Doxycycline)<br />
C<br />
Date:07/30/01ISR Number: 3769075-7Report Type:Expedited (15-DaCompany Report #264404 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Flushing Health Inc ORAL 250 MG ORAL<br />
Paranoia Professional Proguanil<br />
Delusion<br />
(Chloroguanide<br />
Confusional State Hydrochloride) C<br />
Chloroquine<br />
C<br />
Date:08/01/01ISR Number: 3769118-0Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Insomnia<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Arterial Injury<br />
Erythema<br />
Obsessive-Compulsive<br />
Disorder<br />
Cognitive Disorder<br />
Weight Decreased<br />
Adjustment Disorder<br />
Anxiety<br />
Petit Mal Epilepsy<br />
Nervous System Disorder<br />
Headache<br />
Chills<br />
Hepatic Fibrosis<br />
Spinal Cord Injury<br />
Cervical<br />
Subarachnoid Haemorrhage<br />
Hepatic Function Abnormal<br />
Fatigue<br />
Polydipsia<br />
Flushing<br />
Urinary Hesitation
Agitation<br />
Claustrophobia<br />
Delusional Disorder,<br />
Persecutory Type<br />
Hepatic Necrosis<br />
Completed Suicide<br />
Delirium<br />
Hallucinations, Mixed<br />
03-Apr-2012 09:37 AM Page: 375
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hepatitis<br />
Hepatic Steatosis<br />
Erectile Dysfunction Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Appetite Consumer Lariam PS Hoffmann La Roche<br />
Blood Cortisol Increased Other Inc ORAL 250 MG 1 PER<br />
Injury<br />
1 WEEK ORAL<br />
Cardiomegaly<br />
Amnesia<br />
Hyperhidrosis<br />
Abnormal Dreams<br />
Depression<br />
Respiratory Disorder<br />
Spinal Fracture<br />
Laceration<br />
Musculoskeletal Stiffness<br />
Pyrexia<br />
Photopsia<br />
Date:08/01/01ISR Number: 3769201-XReport Type:Expedited (15-DaCompany Report #264705 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Foreign Lariam PS Hoffmann La Roche<br />
Delusion Literature Inc 250 MG<br />
Bipolar Disorder<br />
Health<br />
Electrocardiogram Qt<br />
Professional<br />
Prolonged<br />
Date:08/01/01ISR Number: 3769641-9Report Type:Expedited (15-DaCompany Report #264819 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Foreign Lariam PS Hoffmann La Roche<br />
Initial or Prolonged Weight Decreased Consumer Inc ORAL 1 DOSE FORM 1<br />
Hyperhidrosis<br />
PER WEEK ORAL<br />
Malaise<br />
Spinal Fracture<br />
Fatigue<br />
Malaria<br />
Drug Ineffective<br />
Date:08/02/01ISR Number: 3769264-1Report Type:Expedited (15-DaCompany Report #265424 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Leukopenia Lariam PS Roche 37 DAY<br />
Date:08/03/01ISR Number: 3770284-1Report Type:Expedited (15-DaCompany Report #265284 Age:62 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Health Lariam PS Roche<br />
Dizziness Professional Malarone SS 4 DAY<br />
Tinnitus<br />
Cardiac Arrest<br />
03-Apr-2012 09:37 AM Page: 376
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/06/01ISR Number: 3770939-9Report Type:Expedited (15-DaCompany Report #265419 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Abnormal Behaviour Lariam PS Roche 411 DAY<br />
Emotional Disorder<br />
Irritability<br />
Hallucination<br />
Date:08/07/01ISR Number: 3771367-2Report Type:Expedited (15-DaCompany Report #265329 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Illusion Lariam PS Roche<br />
Dysarthria<br />
Hallucination<br />
Panic Attack<br />
Disturbance In Attention<br />
Date:08/07/01ISR Number: 3771784-0Report Type:Expedited (15-DaCompany Report #265284 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Health Lariam PS Hoffmann La Roche<br />
Dizziness Professional Inc ORAL ORAL<br />
Tinnitus Other Malarone<br />
Cardiac Arrest<br />
(Atovaquone/Chlorogu<br />
anide Hydrochloride) SS ORAL ORAL<br />
Date:08/07/01ISR Number: 3771899-7Report Type:Direct Company Report # Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Personality Change Lariam (<strong>Mefloquine</strong>)<br />
Initial or Prolonged Abnormal Behaviour 250mg PS ORAL 250 MG PO +<br />
Required Confusional State TAB QID<br />
Intervention to<br />
Insomnia<br />
Prevent Permanent Feeling Abnormal<br />
Impairment/Damage Disorientation<br />
Date:08/07/01ISR Number: 3772128-0Report Type:Expedited (15-DaCompany Report #265424 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dyspnoea Foreign Lariam PS Hoffmann La Roche<br />
Pneumococcal Infection Other Inc ORAL 250 MG 1 PER<br />
Leukopenia<br />
1 WEEK ORAL<br />
Pneumothorax<br />
Cough<br />
Blood Culture Positive
Pneumonia<br />
03-Apr-2012 09:37 AM Page: 377
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/08/01ISR Number: 3771798-0Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Mental Disorder Health Lariam Tablets PS Roche 4 TABLETS<br />
Hospitalization - Agitation Professional TAKEN IN<br />
Initial or Prolonged Pyrexia TOTAL. 21 DAY<br />
Disability<br />
Hyperventilation<br />
Coma<br />
Respiratory Failure<br />
Convulsion<br />
Influenza Like Illness<br />
Date:08/08/01ISR Number: 3772873-7Report Type:Expedited (15-DaCompany Report #265419 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hallucination Foreign Lariam (<strong>Mefloquine</strong><br />
Irritability Other Hydrochloride) PS Hoffmann La Roche<br />
Abnormal Behaviour Inc ORAL 250 MG ORAL<br />
Emotional Disorder<br />
Date:08/09/01ISR Number: 3773720-XReport Type:Direct Company Report # Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Neuropathy Peripheral Mefloquin PS 1 DAY<br />
Convulsion<br />
Visual Acuity Reduced<br />
Insomnia<br />
Disorientation<br />
Tremor<br />
Vertigo<br />
Fatigue<br />
Balance Disorder<br />
Headache<br />
Date:08/09/01ISR Number: 3775106-0Report Type:Expedited (15-DaCompany Report #252574 Age:21 YR Gender:Female I/FU:F<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Malaise<br />
Depressed Level Of<br />
Consciousness<br />
Autonomic Nervous System<br />
Imbalance<br />
Fear<br />
Muscle Spasms<br />
Hyperventilation<br />
Agitation<br />
Respiratory Arrest
Anxiety<br />
Dyspnoea<br />
Nausea<br />
Respiratory Disorder<br />
Convulsion<br />
Pyrexia<br />
Blood Ph Increased<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 378
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pco2 Increased<br />
Mental Disorder<br />
Cyanosis Report Source Product Role Manufacturer Route Dose Duration<br />
Respiratory Failure Foreign Lariam PS Hoffmann La Roche<br />
Fatigue Health Inc ORAL 250 MG 1 PER<br />
Apnoea Professional 1 WEEK ORAL<br />
Coma<br />
Other<br />
Loss Of Consciousness<br />
Stress<br />
Influenza Like Illness<br />
Restlessness<br />
Arthralgia<br />
Date:08/13/01ISR Number: 3774908-4Report Type:Expedited (15-DaCompany Report #64270 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Tremor Lariam PS Roche 154 DAY<br />
Anxiety<br />
Circulatory Collapse<br />
Chronic Fatigue Syndrome<br />
Hyperhidrosis<br />
Bradycardia<br />
Muscle Spasms<br />
Dizziness<br />
Headache<br />
Date:08/13/01ISR Number: 3774917-5Report Type:Expedited (15-DaCompany Report #264404 Age:22 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Lariam PS Roche 2 DAY<br />
Initial or Prolonged Psychotic Disorder Proguanil C 1 DAY<br />
Flushing Chloroquine C 1 DAY<br />
Confusional State<br />
Delusion<br />
Date:08/15/01ISR Number: 3778531-7Report Type:Expedited (15-DaCompany Report #64270 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Chronic Fatigue Syndrome Foreign Lariam PS Hoffmann La Roche<br />
Dizziness Literature Inc ORAL 250 MG ORAL<br />
Anxiety<br />
Other<br />
Headache<br />
Bradycardia<br />
Hyperhidrosis<br />
Tremor<br />
Muscle Spasms<br />
Circulatory Collapse
Date:08/15/01ISR Number: 3778546-9Report Type:Expedited (15-DaCompany Report #264404 Age:22 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Psychotic Disorder<br />
Delusion<br />
03-Apr-2012 09:37 AM Page: 379
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorientation<br />
Paranoia<br />
Confusional State Report Source Product Role Manufacturer Route Dose Duration<br />
Flushing Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 250 MG ORAL<br />
Professional<br />
Proguanil<br />
Other<br />
(Chloroguanide<br />
Hydrochloride) C<br />
Chloroquine<br />
(Chloroquine)<br />
C<br />
Date:08/17/01ISR Number: 3778887-5Report Type:Expedited (15-DaCompany Report #265854 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bronchospasm Lariam PS Roche 1 DAY<br />
Date:08/17/01ISR Number: 3779696-3Report Type:Direct Company Report # Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Larium 25o Mg Roche PS Roche ORAL PO<br />
Insomnia Typhoid Vi C<br />
Nausea Ipol C<br />
Date:08/21/01ISR Number: 3781466-7Report Type:Expedited (15-DaCompany Report #265854 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bronchospasm Foreign Lariam PS Hoffmann La Roche<br />
Health Inc ORAL 1 DOSE FORM<br />
Professional<br />
DAILY ORAL<br />
Date:08/21/01ISR Number: 3781711-8Report Type:Direct Company Report # Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Lariam / 250 / La<br />
Initial or Prolonged Depression Roche/Hoffman PS La Roche/Hoffman SUBLINGUAL 1 TABLE /<br />
Other Serious Abnormal Dreams ONCE A WEEK /<br />
Psychomotor Hyperactivity<br />
SUBLINGUAL<br />
Nervousness<br />
Nausea<br />
Panic Attack<br />
Anxiety<br />
Dizziness<br />
Flatulence<br />
Cold Sweat
Date:08/23/01ISR Number: 3782314-1Report Type:Direct Company Report # Age:18 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Required<br />
Intervention to<br />
03-Apr-2012 09:37 AM Page: 380
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Ecchymosis Lariam PS 1 TAB 250MG<br />
Grand Mal Convulsion<br />
EACH WK FOR<br />
Fall<br />
156 WKS<br />
PATIENT TOOK<br />
1 DOSE ON<br />
Date:08/23/01ISR Number: 3782498-5Report Type:Direct Company Report # Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Larium PS ORAL ONCE WEEK<br />
Paranoia<br />
ORAL<br />
Depression<br />
Date:08/28/01ISR Number: 3783549-4Report Type:Expedited (15-DaCompany Report #266589 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Feeling Jittery Lariam PS Roche 36 DAY<br />
Initial or Prolonged Paraesthesia Alcohol C<br />
Panic Attack<br />
Toxicity To Various<br />
Agents<br />
Anxiety<br />
Palpitations<br />
Confusional State<br />
Date:08/28/01ISR Number: 3783984-4Report Type:Direct Company Report # Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Panic Attack<br />
Larium 228.0 Mg Of<br />
Abnormal Dreams Free Base/ Roche PS Roche ORAL 1 TABLE/ PER<br />
Palpitations<br />
WEEK/ ORAL<br />
Muscle Spasms<br />
Abdominal Pain<br />
Fatigue<br />
Insomnia<br />
Convulsion<br />
Date:08/30/01ISR Number: 3785111-6Report Type:Expedited (15-DaCompany Report #264819 Age:31 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Ineffective Lariam PS Roche 46 DAY<br />
Initial or Prolonged Malaria
Date:08/30/01ISR Number: 3785996-3Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
03-Apr-2012 09:37 AM Page: 381
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disability<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety <strong>Mefloquine</strong> / Larium PS ONE PILL<br />
Panic Attack<br />
WEEKLY<br />
Dizziness<br />
Fatigue<br />
Paraesthesia<br />
Vision Blurred<br />
Movement Disorder<br />
Eye Pain<br />
Abnormal Dreams<br />
Dyspnoea<br />
Date:08/30/01ISR Number: 3786101-XReport Type:Expedited (15-DaCompany Report #266589 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Anxiety Hydrochloride) 250<br />
Toxicity To Various Mg PS ORAL 250 MG 1<br />
Agents<br />
PER WEEK ORAL<br />
Feeling Jittery Alcohol (Alcohol) C<br />
Paraesthesia<br />
Confusional State<br />
Panic Attack<br />
Date:09/03/01ISR Number: 3787527-0Report Type:Expedited (15-DaCompany Report #264819 Age:31 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperhidrosis Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Meningitis Viral Consumer Hydrochloride ) 250<br />
Back Injury Health Mg PS ORAL 250 MG 1 PER<br />
Spinal Fracture Professional WEEK ORAL<br />
Weight Decreased<br />
Malaria<br />
Drug Ineffective<br />
Date:09/04/01ISR Number: 3786713-3Report Type:Expedited (15-DaCompany Report #267025 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dementia Lariam PS Roche 1 DAY<br />
Chloroquine<br />
C<br />
Date:09/05/01ISR Number: 3787956-5Report Type:Direct Company Report # Age:28 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nightmare Lariam PS ORAL 1PILL WEEKLY<br />
Vertigo<br />
ORAL<br />
Mental Disorder Claritin C<br />
03-Apr-2012 09:37 AM Page: 382
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/05/01ISR Number: 3788705-7Report Type:Direct Company Report # Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Lariam 250 Mg<br />
Initial or Prolonged Affective Disorder (<strong>Mefloquine</strong> Hcl)<br />
Other Serious Psychotic Disorder (Roche Laboratories) PS Roche Laboratories 250 MG 1 X<br />
Disturbance In Attention<br />
WEEK<br />
Insomnia Halofentrine C<br />
Anxiety<br />
Hyperhidrosis<br />
Pyrexia<br />
Hallucination<br />
Neurotoxicity<br />
Diarrhoea<br />
Panic Attack<br />
Dizziness<br />
Tinnitus<br />
Date:09/06/01ISR Number: 3789407-3Report Type:Expedited (15-DaCompany Report #267025 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cerebral Atrophy Foreign Lariam PS<br />
Health<br />
Chloroquine<br />
Professional (Chloroquine) C<br />
Date:09/10/01ISR Number: 3789468-1Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Hyperhidrosis<br />
Hepatic Steatosis<br />
Abnormal Dreams<br />
Amnesia<br />
Photopsia<br />
Blood Cortisol Increased<br />
Nervous System Disorder<br />
Urinary Hesitation<br />
Agitation<br />
Weight Decreased<br />
Adjustment Disorder<br />
Erythema<br />
Delusional Disorder,<br />
Persecutory Type<br />
Flushing<br />
Insomnia<br />
Petit Mal Epilepsy<br />
Completed Suicide<br />
Polydipsia<br />
Delirium<br />
Fatigue
Musculoskeletal Stiffness<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Obsessive-Compulsive<br />
Disorder<br />
Depression<br />
Headache<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 383
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Decreased Appetite<br />
Liver Function Test<br />
Abnormal Report Source Product Role Manufacturer Route Dose Duration<br />
Pyrexia Lariam Tablets PS Roche THE PATIENT<br />
Chills<br />
RECEIVED A<br />
Hallucinations, Mixed<br />
TOTAL OF NINE<br />
Cognitive Disorder<br />
TABLETS OF<br />
Erectile Dysfunction LARIAM. 67 DAY<br />
Claustrophobia<br />
Date:09/10/01ISR Number: 3789481-4Report Type:Expedited (15-DaCompany Report #265469 Age:48 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Loss Of Libido Lariam PS Roche 1 DAY<br />
Sensory Loss Mirtazapine C<br />
Anorgasmia Prempak-C C DOSE<br />
DESCRIBED AS<br />
0.125 ONCE<br />
PER DAY. NO<br />
UNITS OF<br />
Fluoxetine C 1462 DAY<br />
Date:09/13/01ISR Number: 3792786-4Report Type:Expedited (15-DaCompany Report #265469 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Sexual Dysfunction Foreign Lariam (Melfloquine<br />
Loss Of Libido Health Hydrochloride) PS ORAL ORAL<br />
Sensory Loss Professional Mirtazapine C<br />
Anorgasmia<br />
Local Swelling<br />
Prempak-C<br />
(Estrogens,<br />
Conjugated/Norgestre<br />
l) C<br />
Fluoxetine<br />
C<br />
Date:09/13/01ISR Number: 3793046-8Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Adjustment Disorder<br />
Polydipsia<br />
Anxiety<br />
Delirium<br />
Musculoskeletal Stiffness<br />
Delusional Disorder,<br />
Persecutory Type<br />
Decreased Appetite<br />
Abnormal Dreams<br />
Dermatitis
Amnesia<br />
Hyperkeratosis<br />
Blood Cortisol Increased<br />
Petit Mal Epilepsy<br />
Weight Decreased<br />
Hallucinations, Mixed<br />
Hepatic Necrosis<br />
Erectile Dysfunction<br />
03-Apr-2012 09:37 AM Page: 384
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Agitation<br />
Fatigue<br />
Flushing Report Source Product Role Manufacturer Route Dose Duration<br />
Liver Disorder Consumer Lariam Tablets<br />
Urinary Hesitation Other (Melfloquine<br />
Hepatic Function Abnormal Hydrochloride) 250<br />
Headache Mg PS ORAL 250 MG 1 PER<br />
Insomnia<br />
1 WEEK ORAL<br />
Hyperhidrosis<br />
Depression<br />
Injury<br />
Claustrophobia<br />
Arteriosclerosis<br />
Pyrexia<br />
Hepatic Steatosis<br />
Photopsia<br />
Neoplasm<br />
Obsessive-Compulsive<br />
Disorder<br />
Cognitive Disorder<br />
Cardiomegaly<br />
Chills<br />
Completed Suicide<br />
Inflammation<br />
Nervous System Disorder<br />
Rash Erythematous<br />
Date:09/17/01ISR Number: 3793857-9Report Type:Direct Company Report # Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Lariam 250 Mg<br />
Panic Attack Hoffman-Laroche PS Hoffman-Laroche ORAL 1 250 WEEK<br />
Anxiety<br />
ORAL<br />
Paraesthesia Sudafed C<br />
Paranoia<br />
Suicidal Ideation<br />
Depression<br />
Heart Rate Increased<br />
Hallucination<br />
Hypoaesthesia<br />
Date:09/17/01ISR Number: 3793869-5Report Type:Direct Company Report # Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hyperhidrosis Larium PS Roche ORAL 250 MG/<br />
Tremor<br />
1X/WEEK/ ORAL<br />
Respiratory Disorder<br />
Tachycardia<br />
Panic Attack
Date:09/21/01ISR Number: 3796268-5Report Type:Expedited (15-DaCompany Report #252449 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Ventricular Septal Defect Lariam Tablets PS Roche 60 DAY<br />
Initial or Prolonged Congenital Pneumonia Typhoral C<br />
03-Apr-2012 09:37 AM Page: 385
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Yellow Fever Vaccine C 1 DAY<br />
Date:09/24/01ISR Number: 3797262-0Report Type:Expedited (15-DaCompany Report #267335 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tremor Lariam PS Roche 8 DAY<br />
Initial or Prolonged Hallucination<br />
Sleep Disorder<br />
Confusional State<br />
Anxiety<br />
Date:09/24/01ISR Number: 3797270-XReport Type:Expedited (15-DaCompany Report #268058 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Brain Oedema Lariam PS Roche<br />
Initial or Prolonged Depression Aspirin Soluble C<br />
Headache<br />
Pyrexia<br />
Influenza Like Illness<br />
Liver Function Test<br />
Abnormal<br />
Date:09/24/01ISR Number: 3797282-6Report Type:Expedited (15-DaCompany Report #268319 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brain Neoplasm Lariam PS Roche<br />
Date:09/24/01ISR Number: 3797771-4Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Throat Tightness Lariam PS Roche<br />
Initial or Prolonged Respiratory Rate Pharmaceuticals ORAL ONE PILL/ PER<br />
Disability Increased WEEK/ ORAL<br />
Panic Attack<br />
Ear Pain<br />
Emotional Disorder<br />
Paraesthesia<br />
Insomnia<br />
Heart Rate Increased<br />
Vertigo<br />
Dyspnoea<br />
Hypoaesthesia<br />
Tremor<br />
Burning Sensation<br />
Feeling Abnormal
Anxiety<br />
Palpitations<br />
03-Apr-2012 09:37 AM Page: 386
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/25/01ISR Number: 3797740-4Report Type:Expedited (15-DaCompany Report #259147 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Polymyalgia Rheumatica Lariam PS Roche<br />
Erythema Infectiosum<br />
Dengue Fever<br />
Nausea<br />
Date:09/25/01ISR Number: 3797747-7Report Type:Expedited (15-DaCompany Report #268381 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thrombocytopenia Lariam PS Roche<br />
Haematoma<br />
Date:09/26/01ISR Number: 3798430-4Report Type:Expedited (15-DaCompany Report #265854 Age:17 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bronchospasm Lariam PS Roche 1 DAY<br />
Date:09/26/01ISR Number: 3799276-3Report Type:Direct Company Report # Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Syncope <strong>Mefloquine</strong> PS ORAL WEEKLY ORAL<br />
Gastrooesophageal Reflux<br />
Disease<br />
Abdominal Distension<br />
Heart Rate Increased<br />
Discomfort<br />
Dizziness<br />
Myalgia<br />
Fatigue<br />
Anxiety<br />
Muscle Spasms<br />
Headache<br />
Date:09/27/01ISR Number: 3799042-9Report Type:Expedited (15-DaCompany Report #267025 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dementia Lariam PS Roche 43 DAY<br />
Chloroquine<br />
C<br />
Date:09/27/01ISR Number: 3801651-5Report Type:Expedited (15-DaCompany Report #252449 Age: Gender:Female I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Chlamydial Infection<br />
Pregnancy<br />
Cyanosis<br />
Ventricular Septal Defect<br />
Acquired<br />
Congenital Pneumonia<br />
Neonatal Respiratory<br />
03-Apr-2012 09:37 AM Page: 387
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Distress Syndrome<br />
Maternal Drugs Affecting<br />
Foetus<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam Tablets<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL ORAL<br />
Typhoral (Typhoid<br />
Vaccines)<br />
C<br />
Yellow Fever Vaccine<br />
(Yellow Feve<br />
Vaccine)<br />
C<br />
Date:09/28/01ISR Number: 3800344-8Report Type:Expedited (15-DaCompany Report #268717 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Asthenia Lariam PS Roche<br />
Palpitations<br />
Dry Mouth<br />
Agitation<br />
Anxiety<br />
Tinnitus<br />
Date:09/28/01ISR Number: 3800349-7Report Type:Expedited (15-DaCompany Report #268666 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Alkaline Lariam PS Roche<br />
Initial or Prolonged Phosphatase Increased<br />
Jaundice<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Date:09/28/01ISR Number: 3802046-0Report Type:Direct Company Report # Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Disturbance In Attention Lariam 250 Mg Tablet PS ORAL 250 MG ONCE A<br />
Other Serious Pain WEEK ORAL<br />
Influenza Like Illness<br />
Insomnia<br />
Dizziness<br />
Headache<br />
Arthralgia<br />
Date:10/01/01ISR Number: 3801850-2Report Type:Expedited (15-DaCompany Report #268622 Age:2 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Bilirubin Increased Lariam PS Roche<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 388
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/01/01ISR Number: 3803270-3Report Type:Expedited (15-DaCompany Report #267335 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Sleep Disorder Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Tremor Professional WEEK ORAL<br />
Hallucination<br />
Confusional State<br />
Date:10/01/01ISR Number: 3803271-5Report Type:Expedited (15-DaCompany Report #268058 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Liver Function Test Foreign Lariam<br />
Initial or Prolonged Abnormal Other (<strong>Mefloquine</strong><br />
Brain Oedema Hydrochloride) PS ORAL ORAL<br />
Influenza Like Illness<br />
Aspirin Soluble<br />
Pyrexia (Aspirin) C<br />
Hepatocellular Injury<br />
Headache<br />
Depression<br />
Date:10/01/01ISR Number: 3804291-7Report Type:Expedited (15-DaCompany Report #268319 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brain Neoplasm Consumer Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:10/02/01ISR Number: 3802203-3Report Type:Expedited (15-DaCompany Report #268857 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Diabetes Mellitus <strong>Mefloquine</strong><br />
Inadequate Control Hydrochloride PS Roche 2 DAY<br />
Insulin<br />
C<br />
Date:10/02/01ISR Number: 3802214-8Report Type:Expedited (15-DaCompany Report #268799 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania Lariam PS Roche THE PATIENT<br />
Initial or Prolonged Faecal Incontinence WAS REPORTED<br />
Nightmare<br />
TO HAVE TAKEN<br />
Insomnia TWO DOSES. 8 DAY<br />
Urinary Incontinence Prozac C<br />
Paranoia<br />
Tachycardia<br />
Hallucination
Date:10/02/01ISR Number: 3804021-9Report Type:Expedited (15-DaCompany Report #268381 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source<br />
Other Serious Haematoma Foreign<br />
Thrombocytopenia<br />
Health<br />
03-Apr-2012 09:37 AM Page: 389
Professional<br />
Other<br />
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Product Role Manufacturer Route Dose Duration<br />
Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) PS<br />
Date:10/02/01ISR Number: 3804064-5Report Type:Expedited (15-DaCompany Report #259147 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Polymyalgia Rheumatica Consumer Lariam (<strong>Mefloquine</strong><br />
Asthenia Hydrochloride) PS ORAL ORAL<br />
Joint Effusion<br />
Arthralgia<br />
Decreased Appetite<br />
Dengue Fever<br />
Erythema Infectiosum<br />
Bone Pain<br />
Date:10/03/01ISR Number: 3803283-1Report Type:Expedited (15-DaCompany Report #268895 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Melaena Lariam PS Roche<br />
Ziac<br />
C<br />
Lipitor<br />
C<br />
Date:10/03/01ISR Number: 3805396-7Report Type:Expedited (15-DaCompany Report #268622 Age:2 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Bilirubin Increased Foreign Lariam (<strong>Mefloquine</strong><br />
Aspartate Health Hydrochloride) PS ORAL ORAL<br />
Aminotransferase<br />
Professional<br />
Increased<br />
Date:10/03/01ISR Number: 3805674-1Report Type:Expedited (15-DaCompany Report #265854 Age:17 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bronchospasm Foreign Lariam (<strong>Mefloquine</strong><br />
Choking Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Professional<br />
DAILY ORAL<br />
Date:10/04/01ISR Number: 3804837-9Report Type:Expedited (15-DaCompany Report #268666 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Jaundice Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Blood Albumin Increased Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Aspartate Professional WEEK ORAL<br />
Aminotransferase<br />
Increased<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Blood Bilirubin Increased<br />
03-Apr-2012 09:37 AM Page: 390
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/04/01ISR Number: 3804838-0Report Type:Expedited (15-DaCompany Report #268717 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dry Mouth Foreign Lariam (<strong>Mefloquine</strong><br />
Anxiety Other Hydrochloride) 250<br />
Agitation Mg PS ORAL ORAL<br />
Tinnitus<br />
Palpitations<br />
Asthenia<br />
Date:10/04/01ISR Number: 3804840-9Report Type:Expedited (15-DaCompany Report #267025 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cerebral Atrophy Foreign Lariam (<strong>Mefloquine</strong><br />
Cognitive Disorder Health Hydrochloride) 250<br />
Professional Mg PS<br />
Other<br />
Chloroquine<br />
(Chloroquine)<br />
C<br />
Date:10/04/01ISR Number: 3805279-2Report Type:Expedited (15-DaCompany Report #268857 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Decreased Activity Foreign <strong>Mefloquine</strong><br />
Hypoglycaemia Health Hydrochloride PS ORAL 250 MG 1 PER<br />
Diabetes Mellitus Professional DAY ORAL<br />
Inadequate Control Other Insulin (Insulin) C<br />
Date:10/04/01ISR Number: 3805297-4Report Type:Expedited (15-DaCompany Report #268799 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hallucination Hydrochloride) PS ORAL 250 MG 1 PER<br />
Paranoia<br />
WEEK ORAL<br />
Faecal Incontinence<br />
Prozac (Fluoxetine<br />
Insomnia Hydrochloride) C<br />
Urinary Incontinence<br />
Mania<br />
Tachycardia<br />
Nightmare<br />
Date:10/05/01ISR Number: 3806709-2Report Type:Expedited (15-DaCompany Report #268895 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Melaena Consumer Lariam (<strong>Mefloquine</strong><br />
Faeces Discoloured Hydrochloride) PS ORAL ORAL
Ziac (Bisoprolol<br />
Fumarate/Hydrochloro<br />
thiazide)<br />
Lipitor<br />
(Atorvastatin<br />
Calcium)<br />
C<br />
C<br />
03-Apr-2012 09:37 AM Page: 391
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/09/01ISR Number: 3806216-7Report Type:Direct Company Report # Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Mefoquine 250mg La<br />
Decreased Appetite Roche PS La Roche ORAL 250MG WEEKLY<br />
Weight Decreased<br />
ORAL<br />
Tremor<br />
Vision Blurred<br />
Ill-Defined Disorder<br />
Disturbance In Attention<br />
Lethargy<br />
Frequent Bowel Movements<br />
Myalgia<br />
Date:10/11/01ISR Number: 3806934-0Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Polydipsia Consumer Lariam Tablets PS Roche THE PATIENT<br />
Hospitalization - Decreased Appetite RECEIVED A<br />
Initial or Prolonged Photopsia TOTAL OF NINE<br />
Claustrophobia<br />
TABLETS OF<br />
Hyperhidrosis LARIAM. 67 DAY<br />
Abnormal Dreams<br />
Cognitive Disorder<br />
Agitation<br />
Completed Suicide<br />
Anxiety<br />
Petit Mal Epilepsy<br />
Erythema<br />
Fatigue<br />
Liver Function Test<br />
Abnormal<br />
Obsessive-Compulsive<br />
Disorder<br />
Weight Decreased<br />
Headache<br />
Urinary Hesitation<br />
Amnesia<br />
Insomnia<br />
Hallucinations, Mixed<br />
Depression<br />
Flushing<br />
Delirium<br />
Adjustment Disorder<br />
Delusional Disorder,<br />
Persecutory Type<br />
Hepatic Steatosis<br />
Nervous System Disorder<br />
Chills<br />
Musculoskeletal Stiffness<br />
Pyrexia
Erectile Dysfunction<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Blood Cortisol Increased<br />
03-Apr-2012 09:37 AM Page: 392
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/11/01ISR Number: 3806935-2Report Type:Expedited (15-DaCompany Report #263895 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Health Lariam PS Roche 22 DAY<br />
Initial or Prolonged Headache Professional<br />
Rhabdomyolysis<br />
Date:10/12/01ISR Number: 3807758-0Report Type:Expedited (15-DaCompany Report #269202 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Haemorrhage Lariam PS Roche<br />
Skin Necrosis Metformin Hcl C<br />
Drug Interaction Marcumar I Roche<br />
Date:10/15/01ISR Number: 3809502-XReport Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Chills<br />
Anxiety<br />
Petit Mal Epilepsy<br />
Weight Decreased<br />
Pyrexia<br />
Cardiomegaly<br />
Spinal Cord Injury<br />
Hepatic Necrosis<br />
Amnesia<br />
Fatigue<br />
Delirium<br />
Musculoskeletal Stiffness<br />
Hepatic Steatosis<br />
Delusional Disorder,<br />
Persecutory Type<br />
Liver Function Test<br />
Abnormal<br />
Spinal Cord Injury<br />
Cervical<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Blood Cortisol Increased<br />
Depression<br />
Laceration<br />
Arterial Injury<br />
Injury<br />
Hyperhidrosis<br />
Decreased Appetite<br />
Flushing<br />
Headache<br />
Dermatitis<br />
Erectile Dysfunction
Hyperkeratosis<br />
Abnormal Dreams<br />
Agitation<br />
Completed Suicide<br />
Urinary Hesitation<br />
Adjustment Disorder<br />
Insomnia<br />
Cyst<br />
03-Apr-2012 09:37 AM Page: 393
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Subarachnoid Haemorrhage<br />
Claustrophobia<br />
Nervous System Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Spinal Fracture Consumer Lariam Tablets<br />
Arteriosclerosis Coronary Other (<strong>Mefloquine</strong><br />
Artery Hydrochloride) 250<br />
Polydipsia Mg PS Hoffman-La Roche ORAL 250 MG 1 PER<br />
Obsessive-Compulsive<br />
1 WEEK, ORAL<br />
Disorder<br />
Cognitive Disorder<br />
Hallucinations, Mixed<br />
Photopsia<br />
Erythema<br />
Hepatitis<br />
Brain Scan Abnormal<br />
Date:10/15/01ISR Number: 3809714-5Report Type:Expedited (15-DaCompany Report #263895 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Iris Adhesions Foreign Lariam (Melfloquine<br />
Initial or Prolonged Headache Consumer Hydrochloride) PS ORAL ORAL<br />
Rhabdomyolysis<br />
Health<br />
Antibody Test Positive Professional<br />
Chest Pain<br />
Dizziness<br />
Dyspnoea<br />
Astigmatism<br />
Muscle Atrophy<br />
Date:10/16/01ISR Number: 3809077-5Report Type:Expedited (15-DaCompany Report #263420 Age:66 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Lariam PS Roche 36 DAY<br />
Confusional State Edecrin C<br />
Depression Naprosyn C<br />
Amnesia Asa C<br />
Date:10/16/01ISR Number: 3809095-7Report Type:Expedited (15-DaCompany Report #269729 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Lariam PS Roche 1 DAY<br />
Paraesthesia<br />
Date:10/16/01ISR Number: 3810178-6Report Type:Expedited (15-DaCompany Report #269202 Age:63 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Ulcer Foreign Lariam (<strong>Mefloquine</strong><br />
Drug Interaction Other Hydrochloride) 250<br />
Skin Necrosis Mg PS ORAL 250 MG 1 PER<br />
Haemorrhage Subcutaneous<br />
WEEK ORAL<br />
Marcumar<br />
(Phenprocoumon) 3 Mg SS ORAL 1.5 MG DAILY<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 394
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Metformin Hcl<br />
(Metformin<br />
Hydrochloride)<br />
C<br />
Date:10/17/01ISR Number: 3809774-1Report Type:Expedited (15-DaCompany Report #268895 Age:60 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gastric Haemorrhage Lariam PS Roche 1584 DAY<br />
Ziac<br />
C<br />
Lipitor<br />
C<br />
Date:10/18/01ISR Number: 3811130-7Report Type:Direct Company Report # Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Mephaquin (Mepha<br />
Nightmare<br />
Ltd) Alsch-Basil,<br />
Switzerland PS Mepha Ltd ONE TABLET Q<br />
WEEK<br />
Date:10/18/01ISR Number: 3811971-6Report Type:Expedited (15-DaCompany Report #263420 Age:66 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hypoxia Consumer Lariam (<strong>Mefloquine</strong><br />
Depression Hydrochloride) 250<br />
Confusional State Mg PS ORAL 250 MG 1 PER<br />
Fatigue<br />
Amnesia<br />
Edecrin (Ethacrynate<br />
Sodium Or Ethacrynic<br />
Acid)<br />
Naprosyn (Naproxen)<br />
Asa (Aspirin)<br />
C<br />
C<br />
C<br />
WEEK ORAL<br />
Date:10/18/01ISR Number: 3812046-2Report Type:Expedited (15-DaCompany Report #269729 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eye Irritation Foreign Lariam PS ORAL 250 MG 1 PER<br />
Hypoaesthesia Other WEEK ORAL<br />
Paraesthesia<br />
Viith Nerve Paralysis<br />
Date:10/19/01ISR Number: 3812448-4Report Type:Expedited (15-DaCompany Report #268895 Age:60 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Melaena Consumer Lariam (<strong>Mefloquine</strong><br />
Haematemesis Hydrochloride) PS ORAL ORAL<br />
Gastrointestinal<br />
Ziac (Bisoprolol<br />
Haemorrhage<br />
Fumarate/Hydrochloro<br />
thiazide)<br />
C<br />
Lipitor<br />
(Atorvastatin<br />
Calcium)<br />
C<br />
03-Apr-2012 09:37 AM Page: 395
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/22/01ISR Number: 3813829-5Report Type:Expedited (15-DaCompany Report #253217 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placental Insufficiency Lariam Tablets PS Roche 3 DOSES TAKEN<br />
Initial or Prolonged Placental Disorder IN TOTAL ON<br />
Premature Labour 28 DEC 2000,<br />
Oligohydramnios 04 JAN 2001<br />
AND 11 JAN 15 DAY<br />
Ass-Ratiopharm C 5 DAY<br />
Codein C 1 DAY<br />
Gelsemium C 29 DAY<br />
Cimicifuga C 29 DAY<br />
Paracetamol C 29 DAY<br />
Acc Brausetabletten C 5 DAY<br />
Superpep C 1 DAY<br />
Date:10/22/01ISR Number: 3813843-XReport Type:Expedited (15-DaCompany Report #263895 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Lariam PS Roche 22 DAY<br />
Initial or Prolonged Rhabdomyolysis<br />
Dizziness<br />
Date:10/22/01ISR Number: 3813867-2Report Type:Expedited (15-DaCompany Report #268622 Age:2 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Bilirubin Increased Lariam PS Roche<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Date:10/23/01ISR Number: 3811978-9Report Type:Expedited (15-DaCompany Report #237587 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Intra-Uterine Death Lariam PS Roche 550 DAY<br />
Accidental Exposure<br />
Date:10/24/01ISR Number: 3812634-3Report Type:Expedited (15-DaCompany Report #242880 Age:42 YR Gender:Female I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Panic Reaction<br />
Alopecia<br />
Anxiety<br />
Depression<br />
Neurological Symptom
Thinking Abnormal<br />
Demyelination<br />
Hemiparesis<br />
Vision Blurred<br />
Nausea<br />
Dysgraphia<br />
Optic Neuritis<br />
Memory Impairment<br />
03-Apr-2012 09:37 AM Page: 396
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abdominal Pain<br />
Delusion<br />
Amnesia Report Source Product Role Manufacturer Route Dose Duration<br />
Diarrhoea Lariam PS Roche 15 DAY<br />
Abdominal Distension<br />
Nightmare<br />
Dizziness<br />
Date:10/24/01ISR Number: 3813739-3Report Type:Expedited (15-DaCompany Report #268622 Age:2 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Bilirubin Increased Foreign Lariam (<strong>Mefloquine</strong><br />
Aspartate Health Hydrochloride) 250<br />
Aminotransferase Professional Mg PS ORAL ORAL<br />
Increased<br />
Other<br />
Date:10/24/01ISR Number: 3813743-5Report Type:Expedited (15-DaCompany Report #253217 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placental Insufficiency Foreign Lariam Tablets<br />
Initial or Prolonged Foetal Growth Restriction Health (<strong>Mefloquine</strong><br />
Placental Disorder Professional Hydrochloride) PS ORAL 1 DOSE FORM<br />
Premature Labour<br />
1 PER WEEK<br />
Maternal Drugs Affecting<br />
ORAL<br />
Foetus<br />
Ass-Ratiopharm<br />
Oligohydramnios (Aspirin) C<br />
Codein (Codeine) C<br />
Gelsemium<br />
(Gelsemium)<br />
C<br />
Cimicifuga<br />
(Cimicifuga)<br />
C<br />
Paracetamol<br />
(Acetaminophen) C<br />
Acc Brausetabletten<br />
(Acetylcysteine) C<br />
Superpep<br />
(Dimenhydrinate) C<br />
Date:10/24/01ISR Number: 3813776-9Report Type:Expedited (15-DaCompany Report #263895 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Iris Adhesions Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Rhabdomyolysis Consumer Hydrochloride) PS ORAL ORAL<br />
Paraesthesia<br />
Health<br />
Dyspnoea<br />
Professional<br />
Chest Pain<br />
Dizziness
Eye Injury<br />
Astigmatism<br />
Headache<br />
Pulmonary Embolism<br />
Pupillary Reflex Impaired<br />
03-Apr-2012 09:37 AM Page: 397
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/25/01ISR Number: 3815531-2Report Type:Expedited (15-DaCompany Report #237587 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Accidental Exposure Foreign Lariam (<strong>Mefloquine</strong><br />
Maternal Drugs Affecting Consumer Hydrochloride) 250<br />
Foetus Health Mg PS ORAL ORAL<br />
Intra-Uterine Death<br />
Professional<br />
Pregnancy<br />
Other<br />
Date:10/26/01ISR Number: 3815722-0Report Type:Expedited (15-DaCompany Report #242880 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Health Lariam (<strong>Mefloquine</strong><br />
Toxicity To Various Professional Hydrochloride) PS ORAL 1 DOSE FORM<br />
Agents Other 1 PER WEEK<br />
Delusion<br />
ORAL<br />
Neuralgia<br />
Abdominal Pain<br />
Depression<br />
Amnesia<br />
Hypoaesthesia<br />
Thinking Abnormal<br />
Optic Neuritis<br />
Herpes Zoster<br />
Headache<br />
Hemiparesis<br />
Antinuclear Antibody<br />
Positive<br />
Panic Reaction<br />
Dizziness<br />
Paraesthesia<br />
Abdominal Distension<br />
Memory Impairment<br />
Demyelination<br />
Pain<br />
Alopecia<br />
Dysgraphia<br />
Vision Blurred<br />
Anxiety<br />
Diarrhoea<br />
Nightmare<br />
Date:10/30/01ISR Number: 3816587-3Report Type:Expedited (15-DaCompany Report #257525 Age:63 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depressed Level Of Lariam Tablets PS Roche ONLY ONE DOSE<br />
Initial or Prolonged Consciousness TAKEN. 1 DAY<br />
Aphasia<br />
Amnesia
Date:10/30/01ISR Number: 3816590-3Report Type:Expedited (15-DaCompany Report #253217 Age: Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Placental Disorder<br />
Premature Labour<br />
03-Apr-2012 09:37 AM Page: 398
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Placental Insufficiency<br />
Oligohydramnios<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Tablets PS Roche 3 DOSES TAKEN<br />
IN TOTAL ON<br />
28 DEC 2000,<br />
04 JAN 2001<br />
AND 11 JAN 15 DAY<br />
Ass-Ratiopharm C 5 DAY<br />
Codein C 1 DAY<br />
Gelsemium C 29 DAY<br />
Cimicifuga C 29 DAY<br />
Paracetamol C 29 DAY<br />
Acc Brausetabletten C 5 DAY<br />
Superpep C 1 DAY<br />
Date:10/30/01ISR Number: 3817332-8Report Type:Direct Company Report # Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Larium PS 1 TABLE<br />
Depression<br />
WEEKLY<br />
Date:10/31/01ISR Number: 3817244-XReport Type:Expedited (15-DaCompany Report #256317 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Weight Decreased Lariam PS Roche 3288 DAY<br />
Diarrhoea Unknown Medication C MEDICATIONS<br />
Nightmare<br />
TAKEN IN<br />
Fatigue<br />
1994,1995 AND<br />
Anxiety 1999.<br />
Myalgia Doxycycline C<br />
Bipolar Disorder<br />
Depression<br />
Alopecia<br />
Mental Impairment<br />
Mood Swings<br />
Sleep Disorder<br />
Date:11/01/01ISR Number: 3819874-8Report Type:Direct Company Report # Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Feeling Abnormal Lariam 250 Mg Roche PS Roche ORAL 1 TABLE WEEK<br />
Required Palpitations ORAL<br />
Intervention to<br />
Ventricular Extrasystoles<br />
Prevent Permanent<br />
Impairment/Damage<br />
Panic Attack<br />
Dissociation<br />
Hyperhidrosis
Dyspnoea<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 399
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/02/01ISR Number: 3818568-2Report Type:Expedited (15-DaCompany Report #300688 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Coordination Abnormal Lariam PS Roche<br />
Disturbance In Attention<br />
Dizziness<br />
Date:11/02/01ISR Number: 3819609-9Report Type:Expedited (15-DaCompany Report #253217 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Placental Disorder Foreign Lariam<br />
Initial or Prolonged Foetal Growth Restriction Health (<strong>Mefloquine</strong><br />
Maternal Drugs Affecting Professional Hydrochloride) PS TRANSPLACENTAL 1 DOSE FORM 1<br />
Foetus<br />
PER WEEK ORAL<br />
Oligohydramnios<br />
Ass-Ratiopharm<br />
Premature Labour (Aspirin) C<br />
Calcinosis Codein (Codeine) C<br />
Placental Insufficiency<br />
Gelsemium<br />
(Gelsemium)<br />
C<br />
Cimicifuga<br />
(Cimicifuga)<br />
C<br />
Paracetamol<br />
(Acetaminophen) C<br />
Acc Brausetabletten<br />
(Acetylcysteine) C<br />
Superpep<br />
(Dimenhydrinate) C<br />
Date:11/02/01ISR Number: 3819610-5Report Type:Expedited (15-DaCompany Report #257525 Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Aphasia Foreign Lariam<br />
Initial or Prolonged Speech Disorder Other (<strong>Mefloquine</strong><br />
Depressed Level Of Hydrochloride)250 Mg PS ORAL 250 MG 1 PER<br />
Consciousness<br />
ONE DOSE ORAL<br />
Amnesia<br />
Date:11/02/01ISR Number: 3819850-5Report Type:Expedited (15-DaCompany Report #256317 Age:54 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Disturbance In Attention<br />
Memory Impairment<br />
Cognitive Disorder<br />
Judgement Impaired<br />
Bipolar Disorder<br />
Mood Swings<br />
Alopecia
Depression<br />
Anxiety<br />
Diarrhoea<br />
Myalgia<br />
Fatigue<br />
Sleep Disorder<br />
Mental Impairment<br />
Muscular Weakness<br />
03-Apr-2012 09:37 AM Page: 400
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Weight Decreased<br />
Obsessive-Compulsive<br />
Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Nightmare Other Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) PS<br />
Doxycycline<br />
(Doxycycline)<br />
C<br />
Date:11/06/01ISR Number: 3820350-7Report Type:Direct Company Report # Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Lariam (<strong>Mefloquine</strong>)<br />
Fatigue 250 Mg Tablets PS ORAL ONCE A WEEK<br />
Disturbance In Attention<br />
ORAL<br />
Dizziness<br />
Feeling Abnormal<br />
Pyrexia<br />
Date:11/08/01ISR Number: 3821839-7Report Type:Direct Company Report # Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Larium PS<br />
Psychotic Disorder<br />
Depression<br />
Date:11/15/01ISR Number: 3824467-2Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion Lariam PS Roche 15 DAY<br />
Movement Disorder<br />
Tinnitus<br />
Vision Blurred<br />
Cardiac Arrest<br />
Hearing Impaired<br />
Dizziness<br />
Date:11/19/01ISR Number: 3825625-3Report Type:Expedited (15-DaCompany Report #251768 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Limb Malformation Lariam PS Roche<br />
Date:11/19/01ISR Number: 3827544-5Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F
Outcome<br />
Life-Threatening<br />
PT<br />
Dizziness<br />
Pulse Absent<br />
Tinnitus<br />
Thinking Abnormal<br />
Headache<br />
Muscle Twitching<br />
Vision Blurred<br />
03-Apr-2012 09:37 AM Page: 401
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Memory Impairment<br />
Cardiac Arrest<br />
Grand Mal Convulsion Report Source Product Role Manufacturer Route Dose Duration<br />
Deafness Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
Date:11/21/01ISR Number: 3829085-8Report Type:Expedited (15-DaCompany Report #251768 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Pregnancy Foreign Lariam (<strong>Mefloquine</strong><br />
Maternal Drugs Affecting Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Foetus Professional PER WEEK ORAL<br />
Limb Reduction Defect<br />
Abortion Induced<br />
Date:11/21/01ISR Number: 3829480-7Report Type:Direct Company Report # Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Coordination Abnormal Lariam 250 Mg PS ORAL 1 ORAL<br />
Dizziness<br />
Date:11/26/01ISR Number: 3828569-6Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Abnormal Dreams<br />
Fatigue<br />
Delusional Disorder,<br />
Persecutory Type<br />
Insomnia<br />
Erythema<br />
Depression<br />
Hepatic Steatosis<br />
Weight Decreased<br />
Cognitive Disorder<br />
Polydipsia<br />
Amnesia<br />
Chills<br />
Petit Mal Epilepsy<br />
Hyperhidrosis<br />
Flushing<br />
Nervous System Disorder<br />
Decreased Appetite<br />
Claustrophobia<br />
Headache<br />
Adjustment Disorder
Anxiety<br />
Musculoskeletal Stiffness<br />
Liver Function Test<br />
Abnormal<br />
Erectile Dysfunction<br />
Completed Suicide<br />
Pyrexia<br />
Agitation<br />
03-Apr-2012 09:37 AM Page: 402
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Blood Cortisol Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Obsessive-Compulsive Lariam Tablets PS Roche THE PATIENT<br />
Disorder<br />
RECEIVED A<br />
Hallucinations, Mixed<br />
TOTAL OF NINE<br />
Urinary Hesitation<br />
TABLETS OF<br />
Delirium LARIAM. 67 DAY<br />
Photopsia Cyproheptadine C AT BEDTIME.<br />
Date:11/26/01ISR Number: 3828570-2Report Type:Expedited (15-DaCompany Report #215428 Age:66 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Urinary Retention Lariam Tablets PS Roche 28 DAY<br />
Initial or Prolonged Panic Disorder Without Tamoxifen C<br />
Disability Agoraphobia Fosamax C<br />
Nightmare<br />
Urinary Tract Infection<br />
Dehydration<br />
Anxiety<br />
Schizophrenia<br />
Agitation<br />
Insomnia<br />
Date:11/26/01ISR Number: 3828571-4Report Type:Expedited (15-DaCompany Report #86887 Age:43 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Peripheral Sensory Lariam Tablets PS Roche 49 DAY<br />
Neuropathy<br />
Ill-Defined Disorder<br />
Suicidal Ideation<br />
Myalgia<br />
Tinnitus<br />
Epistaxis<br />
Sleep Disorder<br />
Csf Protein Abnormal<br />
Flushing<br />
Pain<br />
Asthenia<br />
Malaise<br />
Convulsion<br />
Nervousness<br />
Date:11/26/01ISR Number: 3828603-3Report Type:Expedited (15-DaCompany Report #301583 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nervous System Disorder Lariam PS Roche 8 TABLETS
Initial or Prolonged Gastrointestinal Disorder TAKEN OVER 2<br />
DAYS.<br />
03-Apr-2012 09:37 AM Page: 403
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/26/01ISR Number: 3828607-0Report Type:Expedited (15-DaCompany Report #302003 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS Roche 46 DAY<br />
Paraesthesia<br />
Demyelination<br />
Asthenia<br />
Date:11/28/01ISR Number: 3831370-0Report Type:Expedited (15-DaCompany Report #215428 Age:66 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Consumer Lariam Tablets<br />
Initial or Prolonged Panic Reaction Other (<strong>Mefloquine</strong><br />
Disability Schizophrenia Hydrochloride) 250<br />
Agitation Mg PS ORAL 250 MG 1 PER<br />
Anxiety<br />
WEEK ORAL<br />
Nightmare<br />
Tamoxifen (Tamoxifen<br />
Urinary Retention Citrate) C<br />
Emotional Disorder<br />
Fosamax (Alendronate<br />
Circadian Rhythm Sleep Sodium) C<br />
Disorder<br />
Oliguria<br />
Dehydration<br />
Date:11/28/01ISR Number: 3831775-8Report Type:Expedited (15-DaCompany Report #203837 Age:53 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Decreased Appetite<br />
Arterial Injury<br />
Erectile Dysfunction<br />
Insomnia<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Respiratory Disorder<br />
Hepatic Fibrosis<br />
Weight Decreased<br />
Hallucinations, Mixed<br />
Nervous System Disorder<br />
Subarachnoid Haemorrhage<br />
Photopsia<br />
Abnormal Dreams<br />
Obsessive-Compulsive<br />
Disorder<br />
Claustrophobia<br />
Ventricular Hypertrophy<br />
Musculoskeletal Stiffness<br />
Delusional Disorder,<br />
Persecutory Type<br />
Chills
Completed Suicide<br />
Blood Cortisol Increased<br />
Depression<br />
Arteriosclerosis<br />
Hepatic Function Abnormal<br />
Amnesia<br />
Anxiety<br />
Delirium<br />
03-Apr-2012 09:37 AM Page: 404
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Hepatic Steatosis<br />
Headache Report Source Product Role Manufacturer Route Dose Duration<br />
Hyperhidrosis Consumer Lariam Tablets<br />
Agitation Other (<strong>Mefloquine</strong><br />
Flushing Hydrochloride) 250<br />
Petit Mal Epilepsy Mg PS ORAL 250 MG 1 PER<br />
Urinary Hesitation<br />
1 WEEK ORAL<br />
Rash Erythematous<br />
Cyproheptadine<br />
Fatigue<br />
(Cyproheptadine<br />
Polydipsia Hydrochloride) C<br />
Injury<br />
Erythema<br />
Cognitive Disorder<br />
Spinal Fracture<br />
Adjustment Disorder<br />
Date:11/28/01ISR Number: 3831847-8Report Type:Expedited (15-DaCompany Report #86887 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Foreign Lariam Tablets<br />
Flushing Consumer (<strong>Mefloquine</strong><br />
Blood Immunoglobulin G Health Hydrochloride) PS ORAL ORAL 49 DAY<br />
Increased<br />
Professional<br />
Sleep Disorder<br />
Other<br />
Convulsion<br />
Herpes Simplex<br />
Myalgia<br />
Skin Atrophy<br />
Antinuclear Antibody<br />
Positive<br />
Malaise<br />
Laboratory Test Abnormal<br />
Tinnitus<br />
Suicidal Ideation<br />
Asthenia<br />
Muscular Weakness<br />
Norepinephrine Increased<br />
Peripheral Sensory<br />
Neuropathy<br />
Feeling Of Body<br />
Temperature Change<br />
Infectious Mononucleosis<br />
Nervousness<br />
Chest Pain<br />
Blood Albumin Decreased<br />
Epistaxis<br />
Herpes Zoster
Date:11/28/01ISR Number: 3831980-0Report Type:Expedited (15-DaCompany Report #301583 Age:37 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Abdominal Pain<br />
Affective Disorder<br />
Hallucination<br />
Paranoia<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 405
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Insomnia<br />
Dizziness<br />
Agitation Report Source Product Role Manufacturer Route Dose Duration<br />
Nausea Foreign Lariam (<strong>Mefloquine</strong><br />
Gastrointestinal Disorder Literature Hydrocloride) PS ORAL ORAL<br />
Anxiety<br />
Health<br />
Mental Disorder<br />
Professional<br />
Confusional State<br />
Constipation<br />
Date:11/28/01ISR Number: 3832228-3Report Type:Expedited (15-DaCompany Report #302003 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Electromyogram Abnormal Foreign Lariam (<strong>Mefloquine</strong><br />
Demyelination Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Neuropathy Peripheral Professional PER WEEK ORAL<br />
Asthenia<br />
Muscle Contractions<br />
Involuntary<br />
Anxiety<br />
Paraesthesia<br />
Date:12/04/01ISR Number: 3833485-XReport Type:Expedited (15-DaCompany Report #302763 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haematuria Lariam PS Roche<br />
Date:12/05/01ISR Number: 3836429-XReport Type:Direct Company Report # Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrioventricular Block <strong>Mefloquine</strong> PS<br />
Initial or Prolonged Second Degree Claritin C<br />
Motrin<br />
C<br />
Date:12/07/01ISR Number: 3836514-2Report Type:Direct Company Report # Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nervous System Disorder Lariam<br />
Initial or Prolonged Hoffman-Laroche PS Hoffman-Laroche 1 PILL 1 TIME<br />
Other Serious<br />
Date:12/07/01ISR Number: 3837755-0Report Type:Expedited (15-DaCompany Report #302763 Age:21 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haematuria Foreign Lariam (<strong>Mefloquine</strong><br />
Other Hydrochloride) 250<br />
Mg PS 1 DOSE FORM 1<br />
PER WEEK<br />
03-Apr-2012 09:37 AM Page: 406
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/10/01ISR Number: 3836508-7Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Lariam Tablets PS Roche 8 DAY<br />
Delusion Tanakan C<br />
Ludiomil C TAKEN AT<br />
BEDTIME.<br />
Toco<br />
C<br />
Aspegic C TAKEN AT<br />
BEDTIME.<br />
Aricept<br />
C<br />
Xatral<br />
C<br />
Date:12/12/01ISR Number: 3839260-4Report Type:Expedited (15-DaCompany Report #252788 Age:74 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Foreign Lariam Tablets<br />
Abnormal Behaviour Health (<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
PER WEEK ORAL<br />
Tanakan (Ginkgo) C<br />
Ludiomil<br />
(Maprotiline<br />
Hydrochloride) C<br />
Toco (Vitamin E) C<br />
Aspegic (Aspirin<br />
Dl-Lysine)<br />
C<br />
Aricept (Donepezil) C<br />
Xatral (Alfuzosin<br />
Hydrochloride) C<br />
Date:12/18/01ISR Number: 3839742-5Report Type:Expedited (15-DaCompany Report #303413 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsions Local Lariam PS Roche 3 DAY<br />
Initial or Prolonged Epilepsy Corticosteroids C<br />
Date:12/18/01ISR Number: 3839747-4Report Type:Expedited (15-DaCompany Report #303343 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Nausea Lariam PS Roche<br />
Hospitalization - Cardiac Arrest<br />
Initial or Prolonged Fatigue<br />
Date:12/18/01ISR Number: 3840025-8Report Type:Expedited (15-DaCompany Report #269729 Age:29 YR Gender:Female I/FU:F
Outcome<br />
Other Serious<br />
PT<br />
Eye Irritation<br />
Headache<br />
Fatigue<br />
Viith Nerve Paralysis<br />
Dizziness<br />
Paraesthesia<br />
03-Apr-2012 09:37 AM Page: 407
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diplopia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche 1 DAY<br />
Date:12/19/01ISR Number: 3842512-5Report Type:Expedited (15-DaCompany Report #303413 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Loss Of Consciousness Health Hydrochloride) PS ORAL ORAL 3 DAY<br />
Grand Mal Convulsion Professional Corticosteroids<br />
Depression<br />
(Corticosteroid Nos) C<br />
Date:12/19/01ISR Number: 3842514-9Report Type:Expedited (15-DaCompany Report #303343 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Fatigue Foreign Lariam (<strong>Mefloquine</strong><br />
Hospitalization - Meningism Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Initial or Prolonged Cerebral Ischaemia Professional WEEK ORAL<br />
Loss Of Consciousness<br />
Cardiac Arrest<br />
Date:12/20/01ISR Number: 3843382-1Report Type:Expedited (15-DaCompany Report #269729 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diplopia Foreign Lariam (<strong>Mefloquine</strong><br />
Dizziness Other Hydrochloride) 250<br />
Burning Sensation Mg PS ORAL 250 MG 1 PER<br />
Fatigue<br />
WEEK ORAL<br />
Headache<br />
Hypoaesthesia<br />
Viith Nerve Paralysis<br />
Paraesthesia<br />
Date:12/21/01ISR Number: 3842639-8Report Type:Expedited (15-DaCompany Report #303685 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Catatonia Lariam PS Roche 1 DAY<br />
Initial or Prolonged Psychomotor Retardation<br />
Confusional State<br />
Amnesia<br />
Date:12/21/01ISR Number: 3842640-4Report Type:Expedited (15-DaCompany Report #303614 Age:38 YR Gender:Male I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Alanine Aminotransferase<br />
Increased<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Blood Creatine<br />
03-Apr-2012 09:37 AM Page: 408
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Phosphokinase Increased<br />
Myalgia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche 44 DAY<br />
Date:12/27/01ISR Number: 3845437-4Report Type:Expedited (15-DaCompany Report #303614 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Alanine Aminotransferase Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Increased Other Hydrochloride) PS ORAL ORAL<br />
Oedema<br />
Muscle Disorder<br />
Inflammation<br />
Myalgia<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Back Pain<br />
Date:12/27/01ISR Number: 3845439-8Report Type:Expedited (15-DaCompany Report #303685 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Catatonia Health Hydrochloride) PS ORAL 5 DOSE FORM<br />
Amnesia Professional DAILY ORAL<br />
Psychomotor Retardation Other<br />
Date:12/31/01ISR Number: 3845986-9Report Type:Direct Company Report #CTU 157943 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Disorder <strong>Mefloquine</strong> PS 1 TAB Q WEEK<br />
Fatigue<br />
Dizziness<br />
Headache<br />
Chills<br />
Confusional State<br />
Depression<br />
Insomnia<br />
Nightmare<br />
Anxiety<br />
Date:01/02/02ISR Number: 3846227-9Report Type:Expedited (15-DaCompany Report #86887 Age:43 YR Gender:Female I/FU:F
Outcome<br />
Disability<br />
PT<br />
Convulsion<br />
Malaise<br />
Nervousness<br />
Sleep Disorder<br />
Pain<br />
Asthenia<br />
Tinnitus<br />
03-Apr-2012 09:37 AM Page: 409
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Muscular Weakness<br />
Peripheral Sensory<br />
Neuropathy Report Source Product Role Manufacturer Route Dose Duration<br />
Suicidal Ideation Lariam Tablets PS Roche 49 DAY<br />
Csf Protein Abnormal<br />
Flushing<br />
Epistaxis<br />
Myalgia<br />
Ill-Defined Disorder<br />
Date:01/04/02ISR Number: 3849007-3Report Type:Expedited (15-DaCompany Report #86887 Age:43 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Feeling Hot Foreign Lariam Tablets<br />
Loss Of Consciousness Consumer (<strong>Mefloquine</strong><br />
Peripheral Sensory Health Hydrochloride) 250<br />
Neuropathy Professional Mg PS ORAL ORAL 49 DAY<br />
Decreased Activity<br />
Other<br />
Epistaxis<br />
Nervousness<br />
Tinnitus<br />
Hepatitis B Surface<br />
Antigen Positive<br />
Asthenia<br />
Pain<br />
Arthropod Bite<br />
Back Pain<br />
Lipoatrophy<br />
Convulsion<br />
Csf Oligoclonal Band<br />
Present<br />
Neck Pain<br />
Sleep Disorder<br />
Angina Pectoris<br />
Antibody Test Positive<br />
Fatigue<br />
Malaise<br />
Myalgia<br />
Suicidal Ideation<br />
Csf Protein Abnormal<br />
Muscular Weakness<br />
Paraesthesia<br />
Epstein-Barr Virus<br />
Antibody Positive<br />
Feeling Cold<br />
Hypoaesthesia<br />
Date:01/07/02ISR Number: 3848563-9Report Type:Expedited (15-DaCompany Report #303413 Age:25 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsions Local Lariam PS Roche 11 DAY<br />
Initial or Prolonged Epilepsy Corticosteroids C<br />
03-Apr-2012 09:37 AM Page: 410
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/07/02ISR Number: 3848564-0Report Type:Expedited (15-DaCompany Report #303976 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Headache Lariam PS Roche 21 DAY<br />
Extrasystoles<br />
Convulsion<br />
Cardiac Arrest<br />
Bradycardia<br />
Visual Impairment<br />
Date:01/07/02ISR Number: 3849817-2Report Type:Direct Company Report #CTU 158576 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Arrhythmia Lariam Roche PS Roche ORAL 1 TABLE WEEK<br />
Hospitalization - Ventricular Tachycardia ORAL<br />
Initial or Prolonged<br />
Date:01/08/02ISR Number: 3849126-1Report Type:Expedited (15-DaCompany Report #304340 Age:15 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depressed Level Of Lariam PS Roche 32 DAY<br />
Initial or Prolonged Consciousness<br />
Disability<br />
Visual Acuity Reduced<br />
Suicidal Ideation<br />
Macular Oedema<br />
Myalgia<br />
Hemiparesis<br />
Hypoaesthesia<br />
Hallucination<br />
Headache<br />
Date:01/09/02ISR Number: 3849476-9Report Type:Expedited (15-DaCompany Report #302003 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS Roche 59 DAY<br />
Asthenia<br />
Date:01/09/02ISR Number: 3849479-4Report Type:Expedited (15-DaCompany Report #304145 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Guillain-Barre Syndrome Lariam PS Roche<br />
Date:01/09/02ISR Number: 3850223-5Report Type:Expedited (15-DaCompany Report #303976 Age:48 YR Gender:Male I/FU:I
Outcome<br />
Life-Threatening<br />
PT<br />
Cardiac Arrest<br />
Extrasystoles<br />
Eye Pain<br />
Grand Mal Convulsion<br />
Bradycardia<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 411
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Visual Impairment<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam (<strong>Mefloquine</strong><br />
Other Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
PER WEEK ORAL<br />
Date:01/09/02ISR Number: 3850225-9Report Type:Expedited (15-DaCompany Report #303413 Age:25 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Epilepsy Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Grand Mal Convulsion Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Depression Professional PER WEEK ORAL<br />
Corticosteroids<br />
(Corticosteroids<br />
Nos)<br />
C<br />
Date:01/10/02ISR Number: 3850029-7Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion Lariam PS Roche 15 DAY<br />
Hearing Impaired<br />
Movement Disorder<br />
Tinnitus<br />
Vision Blurred<br />
Cardiac Arrest<br />
Date:01/10/02ISR Number: 3851407-2Report Type:Expedited (15-DaCompany Report #304340 Age:15 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depressed Level Of Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Consciousness Hydrochloride) 250<br />
Disability Myalgia Mg PS ORAL 250 MG 1 PER<br />
Hypoaesthesia<br />
WEEK ORAL<br />
Paraesthesia<br />
Hemiparesis<br />
Hemiplegia<br />
Hallucination<br />
Headache<br />
Suicidal Ideation<br />
Depression<br />
Eye Swelling<br />
Blindness<br />
Date:01/11/02ISR Number: 3850921-3Report Type:Expedited (15-DaCompany Report #259087 Age: Gender:Not SpecifiI/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Abortion Induced Lariam Tablets PS Roche 8 DAY<br />
Anencephaly<br />
03-Apr-2012 09:37 AM Page: 412
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/11/02ISR Number: 3852084-7Report Type:Expedited (15-DaCompany Report #304145 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Guillain-Barre Syndrome Foreign Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:01/11/02ISR Number: 3852090-2Report Type:Expedited (15-DaCompany Report #302003 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Electromyogram Abnormal Foreign Lariam (<strong>Mefloquine</strong><br />
Affective Disorder Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Dysaesthesia Professional PER WEEK ORAL<br />
Muscle Contractions<br />
Involuntary<br />
Anxiety<br />
Demyelinating<br />
Polyneuropathy<br />
Asthenia<br />
Date:01/14/02ISR Number: 3852358-XReport Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Cardiac Arrest Health Lariam (<strong>Mefloquine</strong><br />
Grand Mal Convulsion Professional Hydrochloride) 250<br />
Headache Mg PS ORAL 250 MG 1 PER<br />
Memory Impairment<br />
1 WEEK ORAL<br />
Deafness<br />
Obstructive Airways<br />
Disorder<br />
Pulse Absent<br />
Dizziness<br />
Thinking Abnormal<br />
Movement Disorder<br />
Vision Blurred<br />
Tinnitus<br />
Date:01/14/02ISR Number: 3852926-5Report Type:Expedited (15-DaCompany Report #259087 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Abortion Induced Foreign Lariam Tablets<br />
Maternal Drugs Affecting Health (<strong>Mefloquine</strong><br />
Foetus Professional Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Anencephaly<br />
PER WEEK ORAL
Date:01/17/02ISR Number: 3854155-8Report Type:Expedited (15-DaCompany Report #304615 Age: Gender:Male I/FU:I<br />
Outcome<br />
Congenital Anomaly<br />
PT<br />
Ear Malformation<br />
Hydrocephalus<br />
Dysmorphism<br />
Maternal Exposure During<br />
03-Apr-2012 09:37 AM Page: 413
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pregnancy<br />
Abortion Induced<br />
Pregnancy<br />
Maternal Drugs Affecting Report Source Product Role Manufacturer Route Dose Duration<br />
Foetus Foreign Lariam (<strong>Mefloquine</strong><br />
Haemorrhage Other Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Deformity Of Orbit<br />
PER WEEK ORAL<br />
Thrombocytopenia<br />
Date:01/21/02ISR Number: 3854428-9Report Type:Expedited (15-DaCompany Report #304615 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Multiple Congenital Lariam PS Roche 21 DAY<br />
Abnormalities<br />
Maternal Drugs Affecting<br />
Foetus<br />
Hydrocephalus<br />
Chondromatosis<br />
Ear Malformation<br />
Dysmorphism<br />
Date:01/21/02ISR Number: 3854429-0Report Type:Expedited (15-DaCompany Report #305025 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Withdrawal Syndrome <strong>Mefloquine</strong><br />
Panic Disorder Hydrochloride PS Roche 430 DAY<br />
Date:01/21/02ISR Number: 3854469-1Report Type:Expedited (15-DaCompany Report #305014 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chronic Myeloid Leukaemia Lariam PS Roche 265 DAY<br />
Initial or Prolonged<br />
Date:01/22/02ISR Number: 3855316-4Report Type:Expedited (15-DaCompany Report #US-BRISTOL-MYERS SQUIBB COMPANY-11660594 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaria Aprovel Tabs PS Bristol-Myers Squibb<br />
Initial or Prolonged Liver Function Test Company ORAL 3 MON<br />
Abnormal Lariam SS ORAL 6 WK<br />
Pyrexia<br />
Hepatitis B Virus<br />
Vaccine<br />
C<br />
Hepatitis A Virus<br />
Vaccine<br />
C<br />
Typhoid Vaccine C<br />
Yellow Fever Vaccine C
Date:01/22/02ISR Number: 3857526-9Report Type:Expedited (15-DaCompany Report #305025 Age:54 YR Gender:Male I/FU:I<br />
Outcome<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
03-Apr-2012 09:37 AM Page: 414
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Impairment/Damage<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Drug Withdrawal Syndrome Foreign <strong>Mefloquine</strong><br />
Panic Disorder Other Hydrochloride<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 1 PER DAY<br />
ORAL<br />
Date:01/22/02ISR Number: 3857527-0Report Type:Expedited (15-DaCompany Report #304615 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Amniotic Cavity Disorder Foreign Lariam (Melfloquine<br />
Abortion Induced Other Hydrochloride) PS ORAL DOSE FORM 1<br />
Dysmorphism PER WEEK ORAL 21 DAY<br />
Foetal Disorder<br />
Deformity Of Orbit<br />
Hydrocephalus<br />
Thrombocytopenia<br />
Maternal Drugs Affecting<br />
Foetus<br />
Cerebral Haemorrhage<br />
Foetal<br />
Date:01/23/02ISR Number: 3856157-4Report Type:Expedited (15-DaCompany Report #301407 Age:23 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Lariam PS Roche 36 DAY<br />
Disturbance In Attention Zyban SS 7 DAY<br />
Vertigo Trinordiol C<br />
Insomnia<br />
Tremor<br />
Dry Mouth<br />
Hyperhidrosis<br />
Energy Increased<br />
Date:01/23/02ISR Number: 3859719-3Report Type:Expedited (15-DaCompany Report #305014 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chronic Myeloid Leukaemia Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Other Hydrochloride) PS ORAL 250 MG DAILY<br />
ORAL<br />
Date:01/23/02ISR Number: 3860005-6Report Type:Direct Company Report #CTU 159881 Age:59 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Hearing Impaired Lariam -<strong>Mefloquine</strong>-<br />
Intervention to 250 Mg Roch PS Roch ORAL 250 MG<br />
Prevent Permanent<br />
WEEKLY ORAL<br />
Impairment/Damage Tri-Estrogen +<br />
Progesterone<br />
C<br />
03-Apr-2012 09:37 AM Page: 415
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/23/02ISR Number: 3860023-8Report Type:Direct Company Report #CTU 159878 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Lariam 250mg Roche PS Roche ORAL 1 TABLE WEEK<br />
Insomnia<br />
ORAL<br />
Memory Impairment<br />
Panic Attack<br />
Decreased Appetite<br />
Anxiety<br />
Depression<br />
Disturbance In Attention<br />
Nervousness<br />
Fatigue<br />
Date:01/24/02ISR Number: 3860046-9Report Type:Direct Company Report #CTU 159858 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Lariam (250 Mg)<br />
Initial or Prolonged Paranoia (Tablets (Ud),Roche<br />
Required Panic Disorder Labs) PS Roche Labs ORAL 250MG 1<br />
Intervention to Dizziness TABLET ORALLY<br />
Prevent Permanent Fatigue WEEKLY<br />
Impairment/Damage Hallucination<br />
Abnormal Dreams<br />
Heart Rate Increased<br />
Nausea<br />
Date:01/25/02ISR Number: 3860071-8Report Type:Expedited (15-DaCompany Report #301407 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Foreign Lariam<br />
Dry Mouth Health (<strong>Mefloquine</strong><br />
Hyperhidrosis Professional Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Vertigo<br />
PER WEEK ORAL<br />
Vision Blurred<br />
Zyban (Bupropion<br />
Energy Increased Hydrochloride) SS ORAL 1 DOSE FORM<br />
Insomnia<br />
Disturbance In Attention<br />
Trinordiol (Ethinyl<br />
Estradiol/Levonorges<br />
trel)<br />
C<br />
DAILY ORAL<br />
Date:01/28/02ISR Number: 3861035-0Report Type:Expedited (15-DaCompany Report #305169 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Foreign Lariam (<strong>Mefloquine</strong><br />
Shock Other Hydrochloride) 250<br />
Weight Decreased Mg PS ORAL 1 DOSE FORM
Confusional State DAILY ORAL<br />
Nausea<br />
Decreased Appetite<br />
Fatigue<br />
Apathy<br />
03-Apr-2012 09:37 AM Page: 416
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/31/02ISR Number: 3860653-3Report Type:Expedited (15-DaCompany Report #256017 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Kidney Malformation Lariam Tablets PS Roche<br />
Date:01/31/02ISR Number: 3861555-9Report Type:Direct Company Report #CTU 160493 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Disturbance In Attention Lariam<br />
Malaise<br />
(<strong>Mefloquine</strong>)(250 Mg<br />
Night Sweats<br />
/ Roche<br />
Palpitations Laboratories) PS Roche Laboratories ORAL 250MG PO Q<br />
Headache WEEK X 7<br />
Fatigue<br />
WEEKS<br />
Date:01/31/02ISR Number: 3862951-6Report Type:Expedited (15-DaCompany Report #256017 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Drugs Affecting Foreign Lariam Tablets<br />
Foetus Health (<strong>Mefloquine</strong><br />
Pregnancy Professional Hydrochloride) PS ORAL 1 DOSEFORM 1<br />
Congenital Genitourinary<br />
PER WEEK ORAL<br />
Abnormality<br />
Date:02/04/02ISR Number: 3862342-8Report Type:Expedited (15-DaCompany Report #302003 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Lariam PS Roche 59 DAY<br />
Anxiety<br />
Date:02/04/02ISR Number: 3862356-8Report Type:Expedited (15-DaCompany Report #305855 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mouth Ulceration <strong>Mefloquine</strong><br />
Erythema Multiforme Hydrochloride PS Roche 25 DAY<br />
Date:02/05/02ISR Number: 3863086-9Report Type:Expedited (15-DaCompany Report #305877 Age:42 YR Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pulmonary Congestion Lariam PS Roche DESCRIBED AS<br />
Dyspnoea<br />
3 TABLETS IN<br />
THE MORNING,<br />
2 IN THE
MIDDLE OF THE 1 DAY<br />
03-Apr-2012 09:37 AM Page: 417
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/05/02ISR Number: 3865088-5Report Type:Direct Company Report #CTU 160910 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Thinking Abnormal Lariam PS ORAL 1 PILL<br />
Initial or Prolonged Dissociation EVERYDAY/2MN<br />
Fear<br />
ORAL<br />
Anxiety<br />
Hypothyroidism<br />
Dysentery<br />
Feeling Abnormal<br />
Paranoia<br />
Stress<br />
Date:02/06/02ISR Number: 3866167-9Report Type:Expedited (15-DaCompany Report #305855 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mouth Ulceration Foreign <strong>Mefloquine</strong><br />
Weight Decreased Other Hydrochloride<br />
Erythema Multiforme<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL 250 MG ORAL<br />
Date:02/07/02ISR Number: 3865776-0Report Type:Direct Company Report #CTU 161186 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aphasia Larium PS ORAL ONE PILL<br />
ONCE A WEEK<br />
ORAL<br />
Date:02/07/02ISR Number: 3866397-6Report Type:Direct Company Report #CTU 161175 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Memory Impairment <strong>Mefloquine</strong> (Larium) PS ORAL 1 TABLE WEEK<br />
Hallucination<br />
ORAL<br />
Disorientation<br />
Confusional State<br />
Date:02/07/02ISR Number: 3866467-2Report Type:Direct Company Report #CTU 161208 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fear Lariam Hoffmann-La<br />
Anxiety Roche PS Hoffmann-La Roche ORAL ONE 250 MG<br />
ONCE A WEEK<br />
ORAL
Date:02/07/02ISR Number: 3867764-7Report Type:Expedited (15-DaCompany Report #305877 Age:42 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lung Infiltration Foreign Lariam (<strong>Mefloquine</strong><br />
Dyspnoea Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 418
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/08/02ISR Number: 3866762-7Report Type:Direct Company Report #CTU 161353 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Larium (?Standard<br />
Headache For Malaria) PS ORAL 1 TABLE<br />
Diarrhoea<br />
WEEKLY ORAL<br />
Vertigo<br />
Abdominal Pain<br />
Decreased Appetite<br />
Nausea<br />
Visual Impairment<br />
Date:02/08/02ISR Number: 3866871-2Report Type:Direct Company Report #CTU 161327 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia Larium 250mg Roche<br />
Required Anxiety Pharacuticals PS Roche Pharacuticals ORAL 250MG ONE A<br />
Intervention to Suicidal Ideation WEEK ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Delusion<br />
Depression<br />
Nightmare<br />
Date:02/08/02ISR Number: 3866875-XReport Type:Direct Company Report #CTU 161331 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Initial Insomnia Lariam 250mg Roche PS Roche 1/WEEK<br />
Nervousness Echinacea C<br />
Fear<br />
Topical Insect Spray C<br />
Agitation Multi-Vitamin C<br />
Date:02/12/02ISR Number: 3866799-8Report Type:Expedited (15-DaCompany Report #261498 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death International Normalised Consumer Lariam PS Roche 37 DAY<br />
Ratio Increased Cozaar Comp C 285 DAY<br />
Haemorrhagic Stroke Warfarin I VARYING DOSES<br />
Drug Interaction<br />
AS PER INR.<br />
Date:02/12/02ISR Number: 3866804-9Report Type:Expedited (15-DaCompany Report #251768 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Limb Reduction Defect Health Lariam PS Roche<br />
Pregnancy<br />
Professional<br />
Abortion Induced<br />
Maternal Drugs Affecting
Foetus<br />
Maternal Exposure During<br />
Pregnancy<br />
03-Apr-2012 09:37 AM Page: 419
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/12/02ISR Number: 3867634-4Report Type:Direct Company Report #CTU 161443 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Larium PS ONCE A TABLET<br />
Panic Attack<br />
Date:02/12/02ISR Number: 3867716-7Report Type:Direct Company Report #CTU 161424 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Larium PS ORAL 1 WEEK ORAL<br />
Initial or Prolonged Malaise Xanax C<br />
Other Serious<br />
Chest Pain<br />
Paranoia<br />
Chest Discomfort<br />
Panic Attack<br />
Date:02/12/02ISR Number: 3867740-4Report Type:Direct Company Report #CTU 161427 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Paranoia <strong>Mefloquine</strong> Hci 250<br />
Hospitalization - Blood Pressure Increased Mg Hoffman-Laroche PS Hoffman-Laroche ORAL 250 MG DAILY<br />
Initial or Prolonged Pyrexia ORAL<br />
Heart Rate Increased Cephalexin C<br />
Tongue Disorder Valium C<br />
Hallucination<br />
Hypersensitivity<br />
Date:02/12/02ISR Number: 3868199-3Report Type:Direct Company Report #CTU 161515 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Menstrual Disorder Lariam PS 1 WEEK<br />
Paranoia<br />
Depression<br />
Nightmare<br />
Insomnia<br />
Personality Change<br />
Hallucination<br />
Dizziness<br />
Date:02/14/02ISR Number: 3869224-6Report Type:Direct Company Report #CTU 161586 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia Lariam PS ORAL ONE WEEK ORAL<br />
Nightmare<br />
Panic Attack
Phobia Of Flying<br />
03-Apr-2012 09:37 AM Page: 420
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/14/02ISR Number: 3869307-0Report Type:Direct Company Report #CTU 161585 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Lariam PS ORAL 1 PILL ORAL<br />
Malaise<br />
Conversion Disorder<br />
Confusional State<br />
Depression<br />
Palpitations<br />
Panic Attack<br />
Anxiety<br />
Claustrophobia<br />
Crying<br />
Mental Disorder<br />
Date:02/14/02ISR Number: 3870022-8Report Type:Direct Company Report #CTU 161642 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Larium PS ONE TABLE<br />
Obsessive-Compulsive<br />
ONCE A WEEK<br />
Disorder<br />
Nightmare<br />
Date:02/15/02ISR Number: 3869297-0Report Type:Expedited (15-DaCompany Report #264219 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Drug Interaction Lariam PS Roche<br />
Hospitalization - Speech Disorder Cozaar C<br />
Initial or Prolonged Brain Oedema Warfarin Sodium I<br />
Brain Death<br />
Pupils Unequal<br />
Bradycardia<br />
Aphasia<br />
Hemiplegia<br />
International Normalised<br />
Ratio Increased<br />
Tachycardia<br />
Haemorrhagic Stroke<br />
Blood Pressure Systolic<br />
Increased<br />
Hypotension<br />
Date:02/15/02ISR Number: 3869736-5Report Type:Direct Company Report #CTU 161716 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Lariam PS ORAL STANDARD ONCE<br />
Initial or Prolonged Dehydration A WEEK ORAL
Dizziness<br />
Pain<br />
Depression<br />
Photosensitivity Reaction<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 421
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/15/02ISR Number: 3871175-8Report Type:Expedited (15-DaCompany Report #259699 Age:72 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Drug Interaction Foreign Lariam (<strong>Mefloquine</strong><br />
International Normalised Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Ratio Increased<br />
WEEK ORAL<br />
Circulatory Collapse<br />
Warfarin (Warfarin<br />
Cerebral Haemorrhage Sodium) SS ORAL 4 MG 1 PER 2<br />
DAY ORAL<br />
Cozaar Comp<br />
(Hydrochlorothiazide<br />
/Losartan Potassium) C<br />
Date:02/15/02ISR Number: 3871379-4Report Type:Expedited (15-DaCompany Report #261498 Age:72 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Potentiating Drug Foreign Lariam (<strong>Mefloquine</strong><br />
Interaction Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Cerebral Haemorrhage<br />
WEEK ORAL<br />
International Normalised<br />
Warfarin (Warfarin<br />
Ratio Increased Sodium) SS ORAL ORAL<br />
Cozaar Comp<br />
(Hydrochlorothiazide<br />
/Losartan Potassium) C<br />
Date:02/15/02ISR Number: 3871385-XReport Type:Expedited (15-DaCompany Report #261498 Age:72 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Cerebral Haemorrhage Foreign Lariam (<strong>Mefloquine</strong><br />
International Normalised Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Ratio Increased<br />
WEEK ORAL<br />
Potentiating Drug<br />
Warfarin (Warfarin<br />
Interaction Sodium) SS ORAL ORAL<br />
Cozaar Comp<br />
(Hydrochlorothiazide<br />
/Losartan Potassium) C<br />
Date:02/18/02ISR Number: 3869837-1Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion Lariam PS Roche 15 DAY<br />
Movement Disorder Malarone SS 4 DAY<br />
Tinnitus<br />
Vision Blurred<br />
Cardiac Arrest<br />
Hearing Impaired
Date:02/18/02ISR Number: 3869840-1Report Type:Expedited (15-DaCompany Report #303413 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Epilepsy Lariam PS Roche 12 DAY<br />
Initial or Prolonged Convulsions Local Epitomax C 1590 DAY<br />
Diane<br />
C<br />
03-Apr-2012 09:37 AM Page: 422
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Corticosteroids<br />
Erythromycine<br />
C<br />
C<br />
Date:02/18/02ISR Number: 3869843-7Report Type:Expedited (15-DaCompany Report #265284 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Arrest Lariam PS Roche<br />
Grand Mal Convulsion Malarone SS 4 DAY<br />
Tinnitus<br />
Date:02/19/02ISR Number: 3870399-3Report Type:Expedited (15-DaCompany Report #259699 Age:72 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Drug Interaction Lariam PS Roche 37 DAY<br />
Hospitalization - Haemorrhagic Stroke Cozaar Comp C 285 DAY<br />
Initial or Prolonged International Normalised Warfarin I<br />
Ratio Increased<br />
Date:02/19/02ISR Number: 3871276-4Report Type:Direct Company Report #CTU 161854 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Larium 250mg<br />
Vomiting Hoffmann-La Roche PS Hoffmann-La Roche ORAL 1 TABLE PER<br />
Malaise<br />
WEEK ORAL<br />
Panic Attack<br />
Date:02/19/02ISR Number: 3872467-9Report Type:Expedited (15-DaCompany Report #264219 Age:72 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Brain Oedema Foreign Lariam (Melfoquine<br />
Hospitalization - Drug Interaction Other Hydrochloride) 250<br />
Initial or Prolonged Aphasia Mg PS 1 PER WEEK<br />
Facial Paresis<br />
Warfarin Sodium<br />
Haemorrhagic Stroke (Warfarin Sodium) 5<br />
Hypotension Mg SS 4 MG 1 PER 2<br />
Blood Pressure Systolic<br />
DAY<br />
Increased<br />
Cozaar (Losartan<br />
Bradycardia Potassium) C<br />
Hemiparesis<br />
Hypothermia<br />
Papilloedema<br />
International Normalised<br />
Ratio Increased<br />
Hyporeflexia<br />
Pupils Unequal
Tachycardia<br />
03-Apr-2012 09:37 AM Page: 423
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/20/02ISR Number: 3873797-7Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion Health Lariam (<strong>Mefloquine</strong><br />
Dizziness Professional Hydrochloride)250 Mg PS ORAL 250 MG 1 PER<br />
Deafness Other 1 WEEK ORAL<br />
Memory Impairment<br />
Malarone<br />
Muscle Twitching<br />
(Atovawuone/Chlorogu<br />
Tinnitus ande Hydrochloride) SS ORAL ORAL<br />
Obstructive Airways<br />
Disorder<br />
Headache<br />
Cardiac Arrest<br />
Confusional State<br />
Vision Blurred<br />
Date:02/20/02ISR Number: 3873798-9Report Type:Expedited (15-DaCompany Report #265284 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Health Lariam (<strong>Mefloquine</strong><br />
Dizziness Professional Hydrochloride) PS ORAL ORAL<br />
Tinnitus Other Malarone<br />
Cardiac Arrest<br />
(Atovaquone/Chlorogu<br />
anide Hydrochloride) SS ORAL ORAL<br />
Date:02/20/02ISR Number: 3873922-8Report Type:Expedited (15-DaCompany Report #303413 Age:24 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Nervous System Disorder<br />
Convulsion<br />
Weight Increased<br />
Antinuclear Antibody<br />
Positive<br />
Dystonia<br />
Epilepsy<br />
Loss Of Consciousness<br />
Rash Erythematous<br />
Tremor<br />
Aphthous Stomatitis<br />
Arthralgia<br />
Depression<br />
Electroencephalogram<br />
Abnormal<br />
Joint Ankylosis<br />
Carpal Tunnel Syndrome<br />
Hypersensitivity<br />
Inflammation<br />
Sinusitis<br />
Sensation Of Heaviness
Feeling Abnormal<br />
Drug Tolerance Decreased<br />
Anaemia<br />
Gastrointestinal Disorder<br />
Peroneal Nerve Palsy<br />
Skin Disorder<br />
Bronchitis<br />
Nuclear Magnetic<br />
03-Apr-2012 09:37 AM Page: 424
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Resonance Imaging Brain<br />
Abnormal<br />
Paraesthesia<br />
Partial Seizures Report Source Product Role Manufacturer Route Dose Duration<br />
Sensory Disturbance Foreign Lariam (<strong>Mefloquine</strong><br />
Clonic Convulsion Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Epitomax<br />
(Topiramate)<br />
C<br />
Diane (Cyproterone<br />
Acetate/Ethinyl<br />
Estradiol)<br />
C<br />
Corticosteriods Nos<br />
(Corticosteroid Nos) C<br />
Erythromycine<br />
(Erythromycin)<br />
C<br />
Date:02/21/02ISR Number: 3872711-8Report Type:Direct Company Report #CTU 161966 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Lariam PS ONE PER<br />
Dizziness<br />
Influenza Like Illness<br />
Pyrexia<br />
Nausea<br />
Pain<br />
Hyperhidrosis<br />
Malaise<br />
Oropharyngeal Pain<br />
Chills<br />
Date:02/21/02ISR Number: 3873492-4Report Type:Expedited (15-DaCompany Report #259699 Age:72 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hemiplegia Foreign Lariam (<strong>Mefloquine</strong><br />
Hospitalization - Brain Oedema Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Initial or Prolonged Pupils Unequal WEEK ORAL<br />
Cerebral Haemorrhage<br />
Warfarin (Warfarin<br />
Extensor Plantar Response Sodium) SS ORAL 4 MG 1 PER 2<br />
Hemiparesis<br />
DAY ORAL<br />
Tachycardia<br />
Cozaar Comp<br />
Bradycardia<br />
(Hydrochlorothiazide<br />
Drug Interaction<br />
/Losartan Potassium) C<br />
Aphasia<br />
Coma<br />
Papilloedema<br />
Hypertension<br />
Hyporeflexia<br />
Hypotension<br />
International Normalised
Ratio Increased<br />
03-Apr-2012 09:37 AM Page: 425
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/22/02ISR Number: 3871938-9Report Type:Expedited (15-DaCompany Report #307365 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Headache Lariam PS Roche 8 DAY<br />
Fear<br />
Depression<br />
Dry Mouth<br />
Paraesthesia<br />
Dizziness<br />
Anxiety<br />
Date:02/26/02ISR Number: 3875863-9Report Type:Expedited (15-DaCompany Report #307365 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dry Mouth Foreign Lariam (<strong>Mefloquine</strong><br />
Dizziness Consumer Hydrochloride) 250<br />
Paraesthesia Mg PS ORAL 1 DOSE FORM<br />
Anxiety DAILY ORAL<br />
Depression<br />
Headache<br />
Hypoaesthesia<br />
Date:02/27/02ISR Number: 3874072-7Report Type:Expedited (15-DaCompany Report #303413 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypersensitivity Lariam PS Roche 12 DAY<br />
Initial or Prolonged Feeling Abnormal Epitomax C 1590 DAY<br />
Partial Seizures Diane C<br />
Epilepsy Corticosteroids C<br />
Brain Scan Abnormal Erythromycine C<br />
Clonic Convulsion<br />
Mouth Ulceration<br />
Bronchitis<br />
Convulsions Local<br />
Sinusitis<br />
Carpal Tunnel Syndrome<br />
Date:02/27/02ISR Number: 3874950-9Report Type:Direct Company Report #CTU 162354 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Lariam 250mg Roche PS Roche<br />
Abnormal Dreams<br />
Dizziness<br />
Mood Swings
Date:02/28/02ISR Number: 3877137-9Report Type:Direct Company Report #CTU 162536 Age:36 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Disturbance In Attention<br />
Myalgia<br />
Dizziness<br />
Sensation Of Heaviness<br />
Decreased Appetite<br />
03-Apr-2012 09:37 AM Page: 426
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Neuralgia<br />
Sleep Disorder<br />
Vision Blurred Report Source Product Role Manufacturer Route Dose Duration<br />
Asthenia <strong>Mefloquine</strong> (Lariam) PS ORAL 250 MG PO Q<br />
Weight Decreased<br />
WEEK<br />
Medication Error Vit C C<br />
Diarrhoea Vit E C<br />
Ventricular Extrasystoles Calcium Carbonate C<br />
Levothyroxin<br />
C<br />
B Vits<br />
C<br />
Date:02/28/02ISR Number: 3878070-9Report Type:Expedited (15-DaCompany Report #303413 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arrhythmia Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Depression Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Joint Ankylosis Professional PER WEEK ORAL<br />
Sensory Loss<br />
Epitomax<br />
Sinusitis (Topiramate) C<br />
Clonic Convulsion<br />
Diane (Cyproterone<br />
Dystonia<br />
Acetate/Ethinyl<br />
Paraesthesia Estradiol) C<br />
Partial Seizures<br />
Corticosteroids<br />
Systemic Inflammatory<br />
(Corticosteroid Nos) C<br />
Response Syndrome<br />
Erythromycine<br />
Bronchitis (Erythromycin) C<br />
Condition Aggravated<br />
Hypersensitivity<br />
Tremor<br />
Feeling Abnormal<br />
Rash Erythematous<br />
Epigastric Discomfort<br />
Aphthous Stomatitis<br />
Carpal Tunnel Syndrome<br />
Loss Of Consciousness<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal<br />
Sensation Of Heaviness<br />
Convulsion<br />
Epilepsy<br />
Skin Disorder<br />
Date:03/01/02ISR Number: 3877283-XReport Type:Direct Company Report #CTU 162584 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Larium PS ORAL ONE PILL<br />
Mental Disorder<br />
WEEKLY ORAL<br />
Memory Impairment
Date:03/01/02ISR Number: 3877309-3Report Type:Direct Company Report #CTU 162608 Age:30 YR Gender:Female I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Tremor<br />
Anxiety Disorder<br />
03-Apr-2012 09:37 AM Page: 427
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hyperventilation<br />
Panic Attack<br />
Depression Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong> PS ORAL ORAL<br />
Date:03/01/02ISR Number: 3878265-4Report Type:Direct Company Report #CTU 162642 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Larium PS ORAL DON''T ONCE A<br />
Hallucination<br />
WEEK ORAL<br />
Irritability<br />
Insomnia<br />
Nightmare<br />
Date:03/01/02ISR Number: 3878287-3Report Type:Direct Company Report #CTU 162674 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Lariam PS ORAL 1 PILL WEEK<br />
Decreased Appetite<br />
ORAL<br />
Psychotic Disorder Xalatan C<br />
Muscle Spasms Allese C<br />
Pyrexia<br />
Night Sweats<br />
Vomiting<br />
Weight Decreased<br />
Date:03/04/02ISR Number: 3877211-7Report Type:Direct Company Report #CTU 162600 Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Lariam 4oz Took For<br />
Initial or Prolonged Heart Rate Increased 5 Weeks PS 1 PER WEEK<br />
FOR 6 WEEKS<br />
Date:03/04/02ISR Number: 3878268-XReport Type:Direct Company Report #CTU 162647 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS Hoffman La Roche ORAL 250 MG WEEKLY<br />
Psychotic Disorder<br />
ORAL<br />
Toothache Erythromycin C<br />
Nightmare Flagyl C<br />
Panic Disorder Ponstan Forte -<br />
Paranoia Mefanamic Acid C<br />
Screaming Ibuprofen C<br />
Convulsion<br />
Crying
Date:03/06/02ISR Number: 3879593-9Report Type:Direct Company Report #CTU 162890 Age:30 YR Gender:Male I/FU:I<br />
Outcome<br />
PT<br />
Anxiety<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 428
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Irritability<br />
Nightmare<br />
Panic Reaction Report Source Product Role Manufacturer Route Dose Duration<br />
Memory Impairment Lariam 250mg Roche PS Roche BUCCAL 1 WEEKS<br />
Mental Disorder<br />
BUCCAL<br />
Disturbance In Attention<br />
Feeling Of Body<br />
Temperature Change<br />
Nervousness<br />
Date:03/07/02ISR Number: 3881201-8Report Type:Direct Company Report #CTU 162987 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Feeling Abnormal<br />
<strong>Mefloquine</strong> -Lariam-<br />
Vomiting 250 Mg PS `<br />
Eyelid Ptosis<br />
Lid Lag<br />
Headache<br />
Eyelid Function Disorder<br />
Speech Disorder<br />
Fall<br />
Vertigo<br />
Date:03/08/02ISR Number: 3879919-6Report Type:Expedited (15-DaCompany Report #308528 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Prothrombin Time <strong>Mefloquine</strong><br />
Hospitalization - Prolonged Hydrochloride PS Roche<br />
Initial or Prolonged Drug Interaction Carvedilol C<br />
Betoptic<br />
C<br />
Captopril<br />
C<br />
Digoxin<br />
C<br />
Warfarin<br />
I<br />
Date:03/11/02ISR Number: 3880623-9Report Type:Expedited (15-DaCompany Report #308514 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Moderate Mental Lariam PS Roche<br />
Retardation<br />
Date:03/12/02ISR Number: 3881606-5Report Type:Direct Company Report #CTU 163184 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam 1 Pill Per<br />
Sinusitis Week PS ORAL 1 WEEKLY
Hyperventilation ORAL<br />
Disorientation Vit E C<br />
Claustrophobia Anti-Biotic C<br />
Multi-Herbal Med C<br />
03-Apr-2012 09:37 AM Page: 429
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/12/02ISR Number: 3882986-7Report Type:Expedited (15-DaCompany Report #308514 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Neurological Foreign Lariam (<strong>Mefloquine</strong><br />
Degeneration Other Hydrochloride) PS 250 MG 1 PER<br />
Maternal Drugs Affecting<br />
WEEK<br />
Foetus<br />
Mental Retardation<br />
Pregnancy<br />
Neonatal Disorder<br />
Date:03/13/02ISR Number: 3882388-3Report Type:Direct Company Report #CTU 163302 Age:71 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Confusional State Lariam 250mg PS 1 PILL ONCE A<br />
Mental Impairment<br />
WEEK<br />
Convulsion<br />
Abnormal Behaviour<br />
Date:03/13/02ISR Number: 3882791-1Report Type:Expedited (15-DaCompany Report #308528 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Abdominal Distension Foreign <strong>Mefloquine</strong><br />
Hospitalization - Cardiomegaly Health Hydrochloride<br />
Initial or Prolonged Gallop Rhythm Present Professional (<strong>Mefloquine</strong><br />
Blood Pressure Decreased Hydrochloride) PS 250 MG 1 PER<br />
Computerised Tomogram<br />
WEEK<br />
Abnormal<br />
Warfarin (Warfarin<br />
Anaemia Sodium) SS<br />
Drug Interaction Digoxin (Digoxin) C<br />
Haemorrhage<br />
Captopril<br />
Infectious Peritonitis (Captopril) C<br />
Congestive Cardiomyopathy<br />
Betoptic (Betaxolol<br />
Melaena Hydrochloride) C<br />
Tachycardia<br />
Carvedilol<br />
Ejection Fraction (Carvedilol) C<br />
Decreased<br />
Ascites<br />
Ileus Paralytic<br />
Abdominal Tenderness<br />
Prothrombin Time<br />
Prolonged<br />
Date:03/14/02ISR Number: 3882692-9Report Type:Direct Company Report #CTU 163449 Age:7 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hallucination Lariam PS
Hospitalization -<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Dyspnoea<br />
03-Apr-2012 09:37 AM Page: 430
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/14/02ISR Number: 3882716-9Report Type:Direct Company Report #CTU 163401 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Myalgia Lariam PS ORAL 1 TABLE<br />
Anxiety<br />
WEEKLY ORAL<br />
Depression<br />
Headache<br />
Palpitations<br />
Insomnia<br />
Dizziness<br />
Paranoia<br />
Vision Blurred<br />
Angina Pectoris<br />
Arthralgia<br />
Date:03/15/02ISR Number: 3883529-4Report Type:Direct Company Report #CTU 163518 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Lariam PS ORAL ONCE WEEK<br />
Disorientation<br />
ORAL<br />
Panic Disorder<br />
Flashback<br />
Delusion<br />
Fear<br />
Date:03/19/02ISR Number: 3884395-3Report Type:Expedited (15-DaCompany Report #308974 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pulmonary Toxicity <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche<br />
Date:03/19/02ISR Number: 3886232-XReport Type:Direct Company Report #CTU 163734 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam -Mefloquin<br />
Palpitations<br />
Hci- 250 Mg Roche<br />
Chest Pain Laboratories PS Roche Laboratories ORAL 1 PER WEEK<br />
Confusional State<br />
ORAL<br />
Nightmare<br />
Insomnia<br />
Date:03/19/02ISR Number: 3886246-XReport Type:Direct Company Report #CTU 163742 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam
Dizziness Hoffmann-Laroche PS Hoffmann-Laroche ORAL 1 TABLE ONCE<br />
Palpitations<br />
A WEEK ORAL<br />
Anxiety<br />
Nightmare<br />
03-Apr-2012 09:37 AM Page: 431
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/21/02ISR Number: 3886508-6Report Type:Direct Company Report #CTU 163886 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoaesthesia Lariam 250mg Roche PS Roche ORAL ONE TABLET WK<br />
Initial or Prolonged Chest Pain / ORAL<br />
Other Serious<br />
Arrhythmia<br />
Date:03/21/02ISR Number: 3886963-1Report Type:Expedited (15-DaCompany Report #308974 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoxia Foreign <strong>Mefloquine</strong><br />
Initial or Prolonged Lung Disorder Literature Hydrochloride<br />
Cough Health (<strong>Mefloquine</strong><br />
White Blood Cell Count Professional Hydrochloride) PS<br />
Increased<br />
Tachypnoea<br />
Biopsy Lung Abnormal<br />
Dyspnoea<br />
Rales<br />
C-Reactive Protein<br />
Increased<br />
Lung Infiltration<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Haematocrit Decreased<br />
Blood Pressure Diastolic<br />
Decreased<br />
Diarrhoea<br />
Haemoglobin Decreased<br />
Pulmonary Toxicity<br />
Date:03/22/02ISR Number: 3886649-3Report Type:Expedited (15-DaCompany Report #262856 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Multiple Congenital Lariam PS Roche 1 DAY<br />
Abnormalities<br />
Date:03/22/02ISR Number: 3886657-2Report Type:Expedited (15-DaCompany Report #308521 Age:12 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Pain Lariam PS Roche<br />
Initial or Prolonged Sleep Disorder<br />
Headache<br />
Muscle Twitching<br />
Malaise<br />
Vomiting<br />
Balance Disorder
Date:03/26/02ISR Number: 3888125-0Report Type:Expedited (15-DaCompany Report #308521 Age:12 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Malaise<br />
Sleep Disorder<br />
03-Apr-2012 09:37 AM Page: 432
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Headache<br />
Abdominal Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Balance Disorder Lariam PS Roche 23 DAY<br />
Muscle Twitching<br />
Vomiting<br />
Date:03/26/02ISR Number: 3889699-6Report Type:Expedited (15-DaCompany Report #262856 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Breech Presentation Foreign Lariam (<strong>Mefloquine</strong><br />
Pregnancy Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Caesarean Section Professional WEEK ORAL<br />
Low Set Ears<br />
Premature Baby<br />
Maternal Drugs Affecting<br />
Foetus<br />
Congenital Hand<br />
Malformation<br />
Skull Malformation<br />
Date:03/26/02ISR Number: 3889700-XReport Type:Expedited (15-DaCompany Report #308521 Age:12 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Muscle Twitching Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Sleep Disorder Health Hydrochloride) PS<br />
Abdominal Pain<br />
Professional<br />
Headache<br />
Vomiting<br />
Asthenia<br />
Gait Disturbance<br />
Malaise<br />
Date:03/27/02ISR Number: 3890784-3Report Type:Direct Company Report #CTU 164380 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Depression Lariam PS 250MG WEEKLY<br />
Sleep Disorder<br />
Headache<br />
Abnormal Dreams<br />
Paraesthesia<br />
Anxiety<br />
Date:03/28/02ISR Number: 3891877-7Report Type:Expedited (15-DaCompany Report #308521 Age:12 YR Gender:Male I/FU:F
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Balance Disorder<br />
Headache<br />
Sleep Disorder<br />
Malaise<br />
Pyrexia<br />
Vomiting<br />
Muscle Twitching<br />
03-Apr-2012 09:37 AM Page: 433
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Asthenia<br />
Abdominal Pain<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam<br />
Health<br />
(<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL ORAL<br />
Other<br />
Date:03/29/02ISR Number: 3891942-4Report Type:Direct Company Report #CTU 164490 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nervousness Lariam PS ORAL ONE CAPSUL<br />
Hospitalization - Overdose WEEKLY ORAL<br />
Initial or Prolonged Anxiety<br />
Disability<br />
Other Serious<br />
Depression<br />
Suicidal Ideation<br />
Suicide Attempt<br />
Condition Aggravated<br />
Mania<br />
Agitation<br />
Bipolar Disorder<br />
Date:03/29/02ISR Number: 3891958-8Report Type:Direct Company Report #CTU 164480 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Larium PS ORAL ONE PILL<br />
Fear<br />
WEEKLY ORAL<br />
Estrogen<br />
C<br />
Progesterone<br />
C<br />
Synthroid<br />
C<br />
Date:03/29/02ISR Number: 3891961-8Report Type:Direct Company Report #CTU 164482 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fibromyalgia Lariam/Mefloquin PS ORAL WEEKLY ORAL<br />
Other Serious<br />
Depression<br />
Suicidal Ideation<br />
Date:04/01/02ISR Number: 3892044-3Report Type:Expedited (15-DaCompany Report #309830 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Ataxia Lariam PS Roche<br />
Initial or Prolonged Nivaquine SS<br />
Paludrine<br />
SS<br />
Artemether C PO. 4 DAY
Date:04/03/02ISR Number: 3895454-3Report Type:Expedited (15-DaCompany Report #309830 Age:52 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Muscular Weakness<br />
Plasmodium Falciparum<br />
03-Apr-2012 09:37 AM Page: 434
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Infection<br />
Tachypnoea<br />
Blood Pressure Decreased<br />
Heart Rate Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Confusional State Foreign Lariam (<strong>Mefloquine</strong><br />
Aggression Other Hydrochloride) PS ORAL ARTEMETHER<br />
Dehydration<br />
(ARTEMETHER)<br />
Loss Of Consciousness<br />
Paludrine<br />
Asthenia<br />
(Chloroguanide<br />
Cerebellar Ataxia Hydrochloride) 100<br />
Diarrhoea Mg SS ORAL 1 DOSE FORM<br />
DAILY ORAL<br />
Artemether<br />
(Artemether) SS 200 MG<br />
MULTIPLE<br />
Date:04/08/02ISR Number: 3895865-6Report Type:Expedited (15-DaCompany Report #309830 Age:52 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Ataxia Lariam PS Roche<br />
Initial or Prolonged Paludrine SS<br />
Nivaquine<br />
SS<br />
Artemether SS PO. 4 DAY<br />
Date:04/08/02ISR Number: 3898379-2Report Type:Direct Company Report #CTU 165199 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Lariam PS ORAL 1 PER WK/5<br />
ORAL<br />
Date:04/09/02ISR Number: 3896411-3Report Type:Expedited (15-DaCompany Report #310770 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ankle Fracture Lariam PS Roche<br />
Palpitations Fragmin C DAILY DOSE<br />
Chest Pain<br />
REPORTED AS<br />
Ventricular Extrasystoles<br />
2500 IE.<br />
Anxiety<br />
Electrocardiogram Qt<br />
Prolonged<br />
Date:04/10/02ISR Number: 3897120-7Report Type:Expedited (15-DaCompany Report #310741 Age: Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche 22 DAY
Date:04/10/02ISR Number: 3898007-6Report Type:Expedited (15-DaCompany Report #309830 Age:52 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Diarrhoea<br />
Muscular Weakness<br />
Cerebellar Ataxia<br />
03-Apr-2012 09:37 AM Page: 435
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Loss Of Consciousness<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam (<strong>Mefloquine</strong><br />
Other Hydrochloride) PS ORAL ORAL<br />
Paludrine<br />
(Chloroguanide<br />
Hydrochloride) SS ORAL 1 DOSE FORM<br />
DAILY ORAL<br />
Nivaquine<br />
(Chloroquine<br />
Sulfate) SS ORAL ORAL<br />
Artemether<br />
(Artemether) SS 200 MG<br />
MULTIPLE<br />
Date:04/10/02ISR Number: 3899376-3Report Type:Direct Company Report #CTU 165430 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Lariam 250 Mg PS<br />
Discomfort Flagyl C<br />
Dizziness Zithromax C<br />
Nausea Prilosec C<br />
Anxiety Supplements C<br />
Eye Pain Librax C<br />
Mood Swings Elavil C<br />
Biaxin<br />
C<br />
Vitamins,<br />
C<br />
Date:04/11/02ISR Number: 3899398-2Report Type:Expedited (15-DaCompany Report #310770 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Electrocardiogram Qt Foreign Lariam (<strong>Mefloquine</strong><br />
Prolonged Other Hydrochloride) 250<br />
Anxiety Mg PS ORAL<br />
Chest Pain<br />
Fragmin (Dalteparin<br />
Palpitations Sodium) C<br />
Ventricular Extrasystoles<br />
Date:04/12/02ISR Number: 3899745-1Report Type:Expedited (15-DaCompany Report #310741 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pregnancy Foreign Lariam (<strong>Mefloquine</strong><br />
Maternal Drugs Affecting Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Foetus<br />
WEEK ORAL<br />
Abortion Spontaneous
Date:04/12/02ISR Number: 3900290-5Report Type:Direct Company Report #CTU 165624 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nightmare Larium PS 1 PER<br />
Disorientation<br />
Fear<br />
03-Apr-2012 09:37 AM Page: 436
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/15/02ISR Number: 3900029-3Report Type:Direct Company Report #CTU 165643 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia Lariam PS 1 DOSE PER<br />
Nervousness<br />
WEEK X 6 WKS<br />
Panic Attack<br />
Flushing<br />
Bladder Disorder<br />
Date:04/17/02ISR Number: 3900592-2Report Type:Expedited (15-DaCompany Report #310850 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Neutropenia Lariam PS Roche<br />
Initial or Prolonged Haemoglobin Decreased Kinin C 10 DAY<br />
Pyrexia<br />
Leukopenia<br />
Date:04/17/02ISR Number: 3900596-XReport Type:Expedited (15-DaCompany Report #310913 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pollakiuria Lariam PS Roche 1 DAY<br />
Abnormal Dreams<br />
Date:04/22/02ISR Number: 3904484-4Report Type:Direct Company Report #CTU 166347 Age:67 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia <strong>Mefloquine</strong> 250mg PS ORAL 250MG WEEKLY<br />
Initial or Prolonged Delusion ORAL<br />
Confusional State Bisoprolol C<br />
Agitation Estradiol C<br />
Pressure Of Speech<br />
Date:04/23/02ISR Number: 3904592-8Report Type:Expedited (15-DaCompany Report #311417 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thrombocytopenia Lariam PS Roche 1 DAY<br />
Neutropenia Aspirine C<br />
Efferalgan<br />
C<br />
Date:04/24/02ISR Number: 3905788-1Report Type:Expedited (15-DaCompany Report #311387 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coordination Abnormal Lariam PS Roche 24 DAY
Initial or Prolonged<br />
Dizziness<br />
Diplopia<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 437
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/24/02ISR Number: 3906283-6Report Type:Direct Company Report #CTU 166528 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Influenza Lariam PS ONCE A WEEK,<br />
Depression<br />
8 DOSES<br />
Decreased Activity<br />
Date:04/25/02ISR Number: 3906132-6Report Type:Expedited (15-DaCompany Report #310913 Age:28 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pollakiuria Lariam PS Roche 1 DAY<br />
Abnormal Dreams<br />
Date:04/26/02ISR Number: 3906880-8Report Type:Expedited (15-DaCompany Report #269045 Age:25 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Rash Lariam PS Roche 32 DAY<br />
Abdominal Pain<br />
Hyperaesthesia<br />
Arthralgia<br />
Diarrhoea<br />
Liver Function Test<br />
Abnormal<br />
Vomiting<br />
Date:04/26/02ISR Number: 3907842-7Report Type:Expedited (15-DaCompany Report #311417 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Febrile Convulsion Foreign Lariam (<strong>Mefloquine</strong><br />
Thrombocytopenia Health Hydrochloride) PS ORAL 6 DOSE FORM<br />
Neutropenia Professional DAILY ORAL<br />
Aspirine (Aspirin) C<br />
Efferalgan<br />
(Acetaminophen) C<br />
Date:04/26/02ISR Number: 3907914-7Report Type:Expedited (15-DaCompany Report #311387 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Diplopia Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Encephalitis<br />
WEEK ORAL<br />
Coordination Abnormal<br />
Vomiting
Date:04/29/02ISR Number: 3907812-9Report Type:Expedited (15-DaCompany Report #309533 Age:33 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Failure Lariam PS Roche 1 DAY<br />
Ventricular Hypokinesia<br />
03-Apr-2012 09:37 AM Page: 438
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/29/02ISR Number: 3907824-5Report Type:Expedited (15-DaCompany Report #311417 Age:29 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Neutropenia Lariam PS Roche 1 DAY<br />
Thrombocytopenia Aspirine C<br />
Efferalgan<br />
C<br />
Date:05/01/02ISR Number: 3909145-3Report Type:Expedited (15-DaCompany Report #310913 Age:28 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pollakiuria Lariam PS Roche 1 DAY<br />
Abnormal Dreams<br />
Date:05/01/02ISR Number: 3911255-1Report Type:Expedited (15-DaCompany Report #269045 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Diarrhoea Foreign Lariam(<strong>Mefloquine</strong><br />
Rash Health Hydrochloride) 250<br />
Arthralgia Professional Mg PS ORAL 250 MG 1 PER<br />
Liver Function Test Other WEEK ORAL<br />
Abnormal<br />
Vomiting<br />
Abdominal Pain<br />
Hyperaesthesia<br />
Date:05/01/02ISR Number: 3911290-3Report Type:Expedited (15-DaCompany Report #311417 Age:29 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thrombocytopenia Foreign Lariam (<strong>Mefloquine</strong><br />
Neutropenia Health Hydrochloride) PS ORAL 6 DOSE FORM<br />
Febrile Convulsion Professional DAILY ORAL<br />
Aspirine (Aspirin) C<br />
Efferalgan<br />
(Acetaminophen) C<br />
Date:05/01/02ISR Number: 3911292-7Report Type:Expedited (15-DaCompany Report #309533 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Failure Foreign Lariam (<strong>Mefloquine</strong><br />
Dilatation Ventricular Health Hydrochloride) PS ORAL ORAL<br />
Professional<br />
Date:05/01/02ISR Number: 3911529-4Report Type:Direct Company Report #CTU 167187 Age:50 YR Gender:Female I/FU:I
Outcome<br />
Other Serious<br />
PT<br />
Burning Sensation<br />
Anxiety<br />
Insomnia<br />
Feeling Cold<br />
Dizziness<br />
Dyspnoea<br />
03-Apr-2012 09:37 AM Page: 439
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Feeling Jittery<br />
Pain<br />
Chills Report Source Product Role Manufacturer Route Dose Duration<br />
Tremor<br />
Lariam 250 -Mg<br />
Paraesthesia Tablets Roche PS Roche ORAL 1 TABLE 1<br />
Palpitations<br />
PER WEEK<br />
ORAL<br />
Date:05/02/02ISR Number: 3909837-6Report Type:Expedited (15-DaCompany Report #311387 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Dizziness Consumer Lariam PS Roche 24 DAY<br />
Hospitalization - Coordination Abnormal<br />
Initial or Prolonged Vomiting<br />
Diplopia<br />
Encephalitis Viral<br />
Immune System Disorder<br />
Date:05/02/02ISR Number: 3909838-8Report Type:Expedited (15-DaCompany Report #312383 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vith Nerve Paralysis Lariam PS Roche 37 DAY<br />
Date:05/03/02ISR Number: 3910667-XReport Type:Expedited (15-DaCompany Report #312383 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vith Nerve Paralysis Lariam PS Roche 37 DAY<br />
Date:05/03/02ISR Number: 3912167-XReport Type:Direct Company Report #CTU 167303 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dyspnoea Larium PS ORAL 1 PILL 1X<br />
Other Serious Heart Rate Decreased WEEK ORAL<br />
Palpitations Aspirin C<br />
Arrhythmia Birth Control Pills C<br />
Mental Disorder<br />
Nightmare<br />
Panic Attack<br />
Dizziness<br />
Paranoia<br />
Hyperhidrosis<br />
Anxiety<br />
Nervousness
Date:05/06/02ISR Number: 3912508-3Report Type:Direct Company Report #CTU 167374 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Suicidal Ideation <strong>Mefloquine</strong> (Lariam) PS ORAL 1 DAY ORAL<br />
Anxiety<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 440
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/07/02ISR Number: 3912580-0Report Type:Expedited (15-DaCompany Report #311387 Age:48 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Coordination Abnormal Foreign Lariam (<strong>Mefloquine</strong><br />
Hospitalization - Vomiting Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Initial or Prolonged Diplopia WEEK ORAL<br />
Encephalitis Viral<br />
Dizziness<br />
Date:05/07/02ISR Number: 3912581-2Report Type:Expedited (15-DaCompany Report #312383 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vith Nerve Paralysis Foreign Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Professional<br />
1 PER WEEK<br />
ORAL<br />
Date:05/07/02ISR Number: 3912886-5Report Type:Expedited (15-DaCompany Report #312383 Age:51 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vith Nerve Paralysis Foreign Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:05/10/02ISR Number: 3913789-2Report Type:Expedited (15-DaCompany Report #312621 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Oral Lariam PS Roche 15 DAY<br />
Viith Nerve Paralysis<br />
Paraesthesia Oral<br />
Date:05/14/02ISR Number: 3915583-5Report Type:Expedited (15-DaCompany Report #264418 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Premature Baby Lariam PS Roche<br />
Premature Separation Of Quinine Sulfate C 24 HOUR<br />
Placenta<br />
PERIODS EVERY<br />
8 HOURS, 10<br />
MG/KG.<br />
TREATED 03 TO 25 DAY<br />
Date:05/14/02ISR Number: 3915591-4Report Type:Expedited (15-DaCompany Report #311132 Age:49 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Deafness Lariam PS Roche 36 DAY<br />
Tinnitus<br />
Ear Discomfort<br />
03-Apr-2012 09:37 AM Page: 441
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/14/02ISR Number: 3915609-9Report Type:Expedited (15-DaCompany Report #311320 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Field Defect Lariam PS Roche 363 DAY<br />
Date:05/14/02ISR Number: 3916964-6Report Type:Expedited (15-DaCompany Report #312621 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nuclear Magnetic Health Lariam (<strong>Mefloquine</strong><br />
Resonance Imaging Professional Hydrochloride) 250<br />
Abnormal Mg PS ORAL 250 MG 1<br />
Hypoaesthesia Oral<br />
PER WEEK<br />
Paraesthesia Oral<br />
ORAL<br />
Viith Nerve Paralysis<br />
Date:05/15/02ISR Number: 3916379-0Report Type:Direct Company Report #CTU 168079 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour <strong>Mefloquine</strong> PS ONE TABLET A<br />
Anger<br />
WEEK<br />
Date:05/16/02ISR Number: 3916689-7Report Type:Expedited (15-DaCompany Report #312628 Age:25 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Influenza Like Illness Lariam PS Roche<br />
Date:05/16/02ISR Number: 3917735-7Report Type:Direct Company Report #CTU 168204 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Arthralgia Lariam PS ONCE A<br />
Nausea<br />
Neck Pain<br />
Pyrexia<br />
Myalgia<br />
Headache<br />
Muscle Spasms<br />
Pain<br />
Movement Disorder<br />
Back Pain<br />
Gait Disturbance<br />
Chills<br />
Date:05/16/02ISR Number: 3917737-0Report Type:Direct Company Report #CTU 168205 Age: Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Lariam PS ONCE A<br />
Dysstasia<br />
Gait Disturbance<br />
Feeling Abnormal<br />
03-Apr-2012 09:37 AM Page: 442
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/17/02ISR Number: 3917198-1Report Type:Expedited (15-DaCompany Report #313299 Age:70 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gastrointestinal Lariam PS Roche 77 DAY<br />
Haemorrhage<br />
Date:05/17/02ISR Number: 3917205-6Report Type:Expedited (15-DaCompany Report #313197 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation <strong>Mefloquine</strong><br />
Initial or Prolonged Psychotic Disorder Hydrochloride PS Roche 1 DAY<br />
Disability Hallucination, Auditory Cannabis Sativa C<br />
Dependent Personality<br />
Disorder<br />
Paranoia<br />
Date:05/20/02ISR Number: 3919404-6Report Type:Expedited (15-DaCompany Report #312628 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chills Foreign Lariam (<strong>Mefloquine</strong><br />
Pain Health Hydrochloride) PS 250 MG 1 PER<br />
Influenza Like Illness Professional WEEK<br />
Date:05/20/02ISR Number: 3919494-0Report Type:Expedited (15-DaCompany Report #311320 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Visual Field Defect Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:05/20/02ISR Number: 3919569-6Report Type:Expedited (15-DaCompany Report #264418 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Apgar Score Low Consumer Lariam (<strong>Mefloquine</strong><br />
Intervention to Pregnancy Hydrochloride) 250<br />
Prevent Permanent Caesarean Section Mg PS ORAL 250 MG 1 PER<br />
Impairment/Damage Maternal Drugs Affecting WEEK ORAL<br />
Foetus<br />
Premature Baby<br />
Premature Separation Of<br />
Placenta
Date:05/20/02ISR Number: 3919652-5Report Type:Expedited (15-DaCompany Report #311132 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Ear Discomfort Foreign Lariam (<strong>Mefloquine</strong><br />
Deafness Health Hydrochloride) 250<br />
Tinnitus Professional Mg PS ORAL 250 MG 1 PER<br />
Other<br />
WEEK; ORAL<br />
03-Apr-2012 09:37 AM Page: 443
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/21/02ISR Number: 3920502-1Report Type:Direct Company Report #CTU 168520 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Major Depression Lariam PS 250 MG/WK<br />
Suicidal Ideation<br />
Date:05/22/02ISR Number: 3922870-3Report Type:Expedited (15-DaCompany Report #313197 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Foreign <strong>Mefloquine</strong><br />
Initial or Prolonged Agitation Other Hydrochloride(Mefloq<br />
Disability Dependent Personality uine Hydrochloride) PS ORAL DOSE FORM E<br />
Disorder<br />
DAILY ORAL<br />
Hallucination, Auditory<br />
Cannabis<br />
Paranoia Sativa(Cannabis) C<br />
Date:05/22/02ISR Number: 3922872-7Report Type:Expedited (15-DaCompany Report #313299 Age:70 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gastrointestinal Foreign Lariam (<strong>Mefloquine</strong><br />
Haemorrhage Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:05/23/02ISR Number: 3920961-4Report Type:Direct Company Report #CTU 168739 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Depression Lariam PS Roche ORAL 1 PILL WEEK<br />
Disability Palpitations ORAL<br />
Required Paranoia Ortho-Tricyclen C<br />
Intervention to<br />
Pyrexia<br />
Prevent Permanent<br />
Impairment/Damage<br />
Vertigo<br />
Suicidal Ideation<br />
Heart Rate Increased<br />
Panic Reaction<br />
Fatigue<br />
Sleep Disorder<br />
Headache<br />
Mood Swings<br />
Date:05/23/02ISR Number: 3920972-9Report Type:Direct Company Report #CTU 168747 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Mefloguine Roche PS Roche ORAL NOT SURE 1<br />
Feeling Abnormal<br />
WEEKLY ORAL
Dizziness Cipro C<br />
Palpitations<br />
Visual Impairment<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 444
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/29/02ISR Number: 3924251-5Report Type:Expedited (15-DaCompany Report #304145 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Health Lariam PS Roche<br />
Respiratory Disorder<br />
Professional<br />
Rash<br />
Guillain-Barre Syndrome<br />
Date:05/29/02ISR Number: 3924280-1Report Type:Expedited (15-DaCompany Report #313979 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Drug Withdrawal Syndrome Lariam PS Roche 121 DAY<br />
Mood Altered<br />
Depression<br />
Anxiety<br />
Date:05/29/02ISR Number: 3924281-3Report Type:Expedited (15-DaCompany Report #313825 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Paranoia Lariam PS Roche<br />
Death<br />
Mood Altered<br />
Date:05/31/02ISR Number: 3927273-3Report Type:Expedited (15-DaCompany Report #313825 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Personality Change Foreign Lariam (<strong>Mefloquine</strong><br />
Asphyxia Other Hydrochloride) PS<br />
Fear<br />
Feeling Abnormal<br />
Paranoia<br />
Date:05/31/02ISR Number: 3927275-7Report Type:Expedited (15-DaCompany Report #304145 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Rash Foreign Lariam (<strong>Mefloquine</strong><br />
Guillain-Barre Syndrome Health Hydrochloride)250mg PS ORAL 250 MG 1 PER<br />
Pyrexia Professional WEEK ORAL<br />
Date:05/31/02ISR Number: 3927276-9Report Type:Expedited (15-DaCompany Report #313979 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Foreign Lariam (<strong>Mefloquine</strong>
Mood Altered Other Hydrochloride) PS ORAL 250 MG DAILY<br />
Drug Withdrawal Syndrome<br />
ORAL<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 445
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/04/02ISR Number: 3932755-4Report Type:Periodic Company Report #258485 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 1 DOSE FORM 1<br />
PER WEEK ORAL<br />
Date:06/04/02ISR Number: 3932756-6Report Type:Periodic Company Report #258606 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam Tablets<br />
Hallucination<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/04/02ISR Number: 3932757-8Report Type:Periodic Company Report #258950 Age:62 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Consumer Lariam Tablets<br />
Initial or Prolonged Diplopia (<strong>Mefloquine</strong><br />
Hallucination Hydrochloride) 250<br />
Balance Disorder Mg PS ORAL 250 MG 1 PER<br />
Coordination Abnormal<br />
WEEK ORAL<br />
Nightmare<br />
Doxycycline<br />
Insomnia (Doxycyline) C<br />
Mental Disorder<br />
Prempro (Estrogens,<br />
Conjugated/Medroypro<br />
gesterone Acetate) C<br />
Cardizem (Diltiazem<br />
Hydrochloride) C<br />
Cipro (Ciprofloxacin<br />
Hydrochloride) C<br />
Lasix (Furosemide) C<br />
Date:06/04/02ISR Number: 3932758-XReport Type:Periodic Company Report #259310 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Anxiety Health Lariam Tablets<br />
Intervention to Decreased Appetite Professional (<strong>Mefloquine</strong><br />
Prevent Permanent Depression Hydrochloride) 250<br />
Impairment/Damage Hypomania Mg PS ORAL 250 MG 1 PER<br />
Agitation<br />
WEEK ORAL<br />
Insomnia<br />
Disturbance In Attention<br />
Hyperaesthesia
Date:06/04/02ISR Number: 3932759-1Report Type:Periodic Company Report #259529 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Consumer Lariam Tablets<br />
Initial or Prolonged Haemoglobin Decreased (<strong>Mefloquine</strong><br />
Malaria Hydrochloride) 250<br />
03-Apr-2012 09:37 AM Page: 446
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Centrum<br />
(Ascorbic/Cholecalci<br />
ferol/Cr./Cu./Folic<br />
Acid/Iodine/Iron/Iv SS ORAL 1 DOSE FORM 1<br />
PER DAY ORAL<br />
Date:06/04/02ISR Number: 3932760-8Report Type:Periodic Company Report #262967 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Bradycardia Health Lariam Tablets<br />
Initial or Prolonged Dysarthria Professional (<strong>Mefloquine</strong><br />
Arrhythmia Hydrochloride) 250<br />
Insomnia Mg PS ORAL 250 MG 1 PER<br />
DAY ORAL<br />
Date:06/04/02ISR Number: 3932761-XReport Type:Periodic Company Report #263260 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aggression Health Lariam Tablets<br />
Lethargy Professional (<strong>Mefloquine</strong><br />
Psychotic Disorder Hydrochloride) 250<br />
Hallucination Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/04/02ISR Number: 3932762-1Report Type:Periodic Company Report #266436 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/04/02ISR Number: 3932763-3Report Type:Periodic Company Report #266437 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL
Date:06/04/02ISR Number: 3932764-5Report Type:Periodic Company Report #266439 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Health Lariam Tablets<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 447
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/04/02ISR Number: 3943960-5Report Type:Periodic Company Report #266440 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/04/02ISR Number: 3943961-7Report Type:Periodic Company Report #266772 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Dizziness Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
DAY ORAL<br />
Allegra (Fexofenadne<br />
Hydrochloride) C<br />
Premarin (Estrogens) C<br />
Date:06/04/02ISR Number: 3943962-9Report Type:Periodic Company Report #269411 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination, Visual Literature <strong>Mefloquine</strong><br />
Initial or Prolonged Paranoia Other Hydrochloride<br />
Depression<br />
(<strong>Mefloquine</strong><br />
Confusional State Hydrochloride) PS ORAL 250 MG 1 PER<br />
Anxiety WEEK ORAL 21 DAY<br />
Cognitive Disorder<br />
Date:06/04/02ISR Number: 3943963-0Report Type:Periodic Company Report #304961 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Heart Rate Increased Professional Hydrochloride) PS ORAL 1 PER WEEK<br />
Anxiety<br />
ORAL<br />
Malaria<br />
Birth Control Pill<br />
Tremor<br />
(Oral Contractptive<br />
Nos)<br />
C<br />
Luvox (Fluvoxamine) C<br />
Date:06/04/02ISR Number: 3943964-2Report Type:Periodic Company Report #237292 Age:25 YR Gender:Female I/FU:F<br />
Outcome<br />
Required<br />
Intervention to<br />
PT<br />
Hyperventilation<br />
Psychotic Disorder
Prevent Permanent<br />
Impairment/Damage<br />
Anxiety<br />
Hallucination, Auditory<br />
Crying<br />
Hallucination, Visual<br />
Jamais Vu<br />
Thinking Abnormal<br />
Tremor<br />
Dissociation<br />
03-Apr-2012 09:37 AM Page: 448
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fatigue<br />
Hyperglycaemia<br />
Suicidal Ideation Report Source Product Role Manufacturer Route Dose Duration<br />
Depression Consumer Lariam (<strong>Mefloquine</strong><br />
Hallucination Health Hydrochloride) 250<br />
Abnormal Dreams Professional Mg PS ORAL 250 MG 1 PER<br />
Confusional State<br />
WEEK ORAL<br />
Feeling Abnormal<br />
Risperdal<br />
(Risperidone)<br />
SS<br />
Zyprexa (Olanzapine) SS<br />
Valium (Diazepam) SS<br />
Date:06/04/02ISR Number: 3943965-4Report Type:Periodic Company Report #255796 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Muscular Weakness Consumer Lariam Tablets<br />
Diarrhoea<br />
(<strong>Mefloquine</strong><br />
Weight Decreased Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Balance Disorder<br />
PER WEEK ORAL<br />
Panic Attack<br />
Dizziness<br />
Nerve Injury<br />
Anxiety<br />
Nausea<br />
Date:06/04/02ISR Number: 3946386-3Report Type:Periodic Company Report #268911 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Consumer Lariam (<strong>Mefloquine</strong><br />
Hallucination Hydrochloride) PS ORAL 250 MG 1 PER<br />
Dysgeusia<br />
WEEK ORAL<br />
Lethargy<br />
Prempro (Estrogens<br />
Insomnia<br />
Conjugagted/Medroxyp<br />
Depression<br />
rogesterone<br />
Suicidal Ideation Acetate) C<br />
Fosamax (Alendronate<br />
Sodium)<br />
C<br />
Date:06/10/02ISR Number: 3930038-XReport Type:Expedited (15-DaCompany Report #304145 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Guillain-Barre Syndrome Health Lariam PS Roche 4 TABLETS<br />
Dyspnoea Professional TAKEN IN<br />
TOTAL. 31 DAY
Date:06/10/02ISR Number: 3930229-8Report Type:Direct Company Report #CTU 169778 Age:47 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Lariam 250mg Roche<br />
Mania Laboratories PS Roche Laboratories ORAL 62.5MG WEEK<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 449
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/11/02ISR Number: 3931023-4Report Type:Expedited (15-DaCompany Report #314691 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nightmare <strong>Mefloquine</strong><br />
Bipolar Disorder Hydrochloride PS Roche WEEKLY 8 DAY<br />
Date:06/12/02ISR Number: 3931889-8Report Type:Expedited (15-DaCompany Report #269729 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Lariam PS Roche 1 DAY<br />
Paraesthesia<br />
Date:06/13/02ISR Number: 3934018-XReport Type:Expedited (15-DaCompany Report #314691 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Paranoia Foreign <strong>Mefloquine</strong><br />
Mania Other Hydrochloride<br />
Nightmare<br />
(<strong>Mefloquine</strong><br />
Bipolar Disorder Hydrochloride) PS ORAL ORAL<br />
Date:06/13/02ISR Number: 3934164-0Report Type:Expedited (15-DaCompany Report #304145 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Respiratory Disorder Foreign Lariam (<strong>Mefloquine</strong><br />
Pyrexia Health Hydrochloride) 250<br />
Guillain-Barre Syndrome Professional Mg PS ORAL 250 MG 1 PER<br />
Rash<br />
WEEK ORAL<br />
Date:06/17/02ISR Number: 3934814-9Report Type:Direct Company Report #CTU 170287 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia Lariam/<strong>Mefloquine</strong><br />
Hcl 250 Milligram<br />
Tablets Hoffman La<br />
Roche PS Hoffman La Roche ORAL 1 TABLE WEEK<br />
ORAL<br />
Date:06/17/02ISR Number: 3935025-3Report Type:Expedited (15-DaCompany Report #269729 Age:29 YR Gender:Female I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Fatigue<br />
Dizziness<br />
Facial Nerve Disorder
Eye Irritation<br />
Hypoaesthesia<br />
Arteriosclerosis Coronary<br />
Artery<br />
Paraesthesia<br />
Viith Nerve Paralysis<br />
Diplopia<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 450
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Trigeminal Nerve Disorder<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG 1 PER<br />
Other<br />
WEEK ORAL<br />
Date:06/18/02ISR Number: 3934117-2Report Type:Expedited (15-DaCompany Report #314691 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nightmare <strong>Mefloquine</strong><br />
Bipolar Disorder Hydrochloride PS Roche WEEKLY 8 DAY<br />
Date:06/18/02ISR Number: 3934122-6Report Type:Expedited (15-DaCompany Report #314899 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disorientation Lariam PS Roche EVERY WEEK. 9 DAY<br />
Initial or Prolonged Speech Disorder Valium SS Roche 1 DAY<br />
Disability<br />
Incontinence<br />
Convulsion<br />
Agitation<br />
Drug Ineffective<br />
Hallucination<br />
Depression<br />
Headache<br />
Fatigue<br />
Memory Impairment<br />
Psychotic Disorder<br />
Loss Of Libido<br />
Paranoia<br />
Hostility<br />
Aggression<br />
Gait Disturbance<br />
Antisocial Behaviour<br />
Date:06/20/02ISR Number: 3935868-6Report Type:Expedited (15-DaCompany Report #313825 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Respiratory Failure Consumer Lariam PS Roche<br />
Abnormal Behaviour<br />
Fear<br />
Asphyxia<br />
Date:06/20/02ISR Number: 3937360-1Report Type:Expedited (15-DaCompany Report #314691 Age:38 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bipolar Disorder Foreign <strong>Mefloquine</strong><br />
Nightmare Other Hydrochloride<br />
Paranoia<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 451
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/20/02ISR Number: 3937925-7Report Type:Expedited (15-DaCompany Report #314899 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Gait Disturbance Hydrochloride) 250<br />
Disability Incoherent Mg PS ORAL 250 MG ORAL<br />
Abnormal Behaviour Valium (Diazepam) SS INTRAVENOUS 2 PER ONE<br />
Drug Ineffective<br />
DOSE<br />
Memory Impairment<br />
INTRAVENOUS<br />
Speech Disorder<br />
Circulatory Collapse<br />
Hallucination<br />
Incontinence<br />
Fatigue<br />
Psychotic Disorder<br />
Aggression<br />
Antisocial Behaviour<br />
Hypersomnia<br />
Headache<br />
Loss Of Libido<br />
Paranoia<br />
Agitation<br />
Depression<br />
Disorientation<br />
Hostility<br />
Date:06/20/02ISR Number: 3938627-3Report Type:Expedited (15-DaCompany Report #314899 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disorientation Consumer Lariam PS Roche EVERY WEEK. 9 DAY<br />
Initial or Prolonged Drug Ineffective Valium SS Roche 1 DAY<br />
Disability<br />
Headache<br />
Psychotic Disorder<br />
Agitation<br />
Speech Disorder<br />
Memory Impairment<br />
Hostility<br />
Loss Of Libido<br />
Circulatory Collapse<br />
Hypersomnia<br />
Paranoia<br />
Gait Disturbance<br />
Convulsion<br />
Antisocial Behaviour<br />
Incontinence<br />
Hallucination<br />
Fatigue<br />
Depression<br />
Aggression<br />
Incoherent
Date:06/24/02ISR Number: 3937485-0Report Type:Expedited (15-DaCompany Report #315549 Age:52 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Anxiety<br />
Delirium<br />
Aggression<br />
03-Apr-2012 09:37 AM Page: 452
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Agitation<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche 2 DAY<br />
Date:06/24/02ISR Number: 3938733-3Report Type:Expedited (15-DaCompany Report #314899 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gait Disturbance Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Fatigue Hydrochloride) 250<br />
Disability Loss Of Libido Mg PS ORAL 250 MG ORAL<br />
Psychotic Disorder Valium (Diazepam) SS INTRAVENOUS 2 PER ONE<br />
Incontinence<br />
DOSE<br />
Cognitive Disorder<br />
INTRAVENOUS<br />
Depression<br />
Aggression<br />
Speech Disorder<br />
Antisocial Behaviour<br />
Hypersomnia<br />
Abnormal Behaviour<br />
Drug Ineffective<br />
Paranoia<br />
Agitation<br />
Communication Disorder<br />
Disorientation<br />
Hallucination<br />
Headache<br />
Hostility<br />
Memory Impairment<br />
Convulsion<br />
Date:06/24/02ISR Number: 3939023-5Report Type:Expedited (15-DaCompany Report #313825 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Fear Foreign Lariam (<strong>Mefloquine</strong><br />
Abnormal Behaviour Health Hydrochloride) PS<br />
Respiratory Failure<br />
Professional<br />
Asphyxia<br />
Other<br />
Personality Change<br />
Date:06/26/02ISR Number: 3940478-0Report Type:Expedited (15-DaCompany Report #315549 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delirium Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Anxiety Other Hydrochloride) PS ORAL 1 DOSE FORM 3<br />
Agitation PER DAY ;<br />
Aggression<br />
ORAL
Date:07/01/02ISR Number: 3941764-0Report Type:Expedited (15-DaCompany Report #312685 Age:52 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nervous System Disorder Lariam PS Roche<br />
Initial or Prolonged Loss Of Consciousness<br />
03-Apr-2012 09:37 AM Page: 453
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/02/02ISR Number: 3943540-1Report Type:Direct Company Report #CTU 171531 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Lariam, 250 Mg,<br />
Thinking Abnormal Roche PS Roche 1 TABLET/WEEK<br />
Hallucinations, Mixed<br />
Diarrhoea<br />
Decreased Appetite<br />
Insomnia<br />
Anxiety<br />
Panic Attack<br />
Tinnitus<br />
Nightmare<br />
Paraesthesia<br />
Date:07/03/02ISR Number: 3942894-XReport Type:Expedited (15-DaCompany Report #316199 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Papilloedema Lariam PS Roche<br />
Date:07/03/02ISR Number: 3942896-3Report Type:Expedited (15-DaCompany Report #316127 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oedema Lariam PS Roche 32 DAY<br />
Prozac C 1/2 TABLET Q.<br />
Date:07/03/02ISR Number: 3944277-5Report Type:Expedited (15-DaCompany Report #312685 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nervous System Disorder Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hunger Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Loss Of Consciousness Professional PER WEEK ORAL<br />
Malaise<br />
Date:07/08/02ISR Number: 3944365-3Report Type:Expedited (15-DaCompany Report #316149 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Optic Ischaemic Lariam PS Roche 48 DAY<br />
Neuropathy<br />
Date:07/08/02ISR Number: 3945803-2Report Type:Direct Company Report #CTU 171760 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Fatigue Lariam 250 Mg<br />
Panic Attack<br />
Roche Laboratories<br />
Confusional State Inc. PS Roche Laboratories<br />
Abnormal Dreams Inc. ORAL 1 WEEK ORAL<br />
Paranoia<br />
Anxiety<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 454
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/10/02ISR Number: 3945975-XReport Type:Expedited (15-DaCompany Report #313891 Age: Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche 1 DAY<br />
Date:07/11/02ISR Number: 3946728-9Report Type:Expedited (15-DaCompany Report #316885 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Transient Ischaemic Lariam PS Roche<br />
Attack<br />
Date:07/11/02ISR Number: 3946741-1Report Type:Direct Company Report #CTU 172130 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Lariam 250mg Roche PS Roche ORAL 250MG WEEKLY<br />
Dizziness<br />
ORAL<br />
Date:07/12/02ISR Number: 3947476-1Report Type:Expedited (15-DaCompany Report #313825 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Asphyxia Lariam PS Roche<br />
Mood Altered<br />
Fear<br />
Death<br />
Date:07/17/02ISR Number: 3949407-7Report Type:Direct Company Report #CTU 172441 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Headache Mefloquin 250mg PS ORAL 250MG ONE PER<br />
Intervention to Feeling Of Body ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Temperature Change<br />
Palpitations<br />
Influenza Like Illness<br />
Paraesthesia<br />
Hyperhidrosis<br />
Fatigue<br />
Disorientation<br />
Eye Pain<br />
Neck Pain<br />
Tinnitus<br />
Date:07/22/02ISR Number: 3951636-3Report Type:Direct Company Report #CTU 172653 Age:23 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Lariam 250mg Hoffman<br />
Initial or Prolonged Nightmare Roche PS Hoffman Roche 2TAB ONCE PER<br />
Sleep Disorder<br />
Pain<br />
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 455
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/24/02ISR Number: 3952731-5Report Type:Expedited (15-DaCompany Report #317246 Age:21 YR Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Obsessive-Compulsive Lariam PS Roche WEEKLY<br />
Disorder DOSING. 15 DAY<br />
Restlessness<br />
Anxiety<br />
Delusional Disorder,<br />
Persecutory Type<br />
Date:07/25/02ISR Number: 3953195-8Report Type:Expedited (15-DaCompany Report #317246 Age:21 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusional Disorder, Lariam PS Roche WEEKLY<br />
Persecutory Type DOSING. 15 DAY<br />
Obsessive-Compulsive<br />
Disorder<br />
Restlessness<br />
Anxiety<br />
Date:07/26/02ISR Number: 3953701-3Report Type:Expedited (15-DaCompany Report #312621 Age:30 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Lariam PS Roche 15 DAY<br />
Paraesthesia Oral<br />
Hypoaesthesia Oral<br />
Neuritis<br />
Date:07/26/02ISR Number: 3953703-7Report Type:Expedited (15-DaCompany Report #316035 Age:17 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam PS Roche 52 DAY<br />
Irritability Contraceptive Pill C<br />
Fatigue<br />
Hallucination, Visual<br />
Flashback<br />
Nightmare<br />
Date:07/30/02ISR Number: 3955307-9Report Type:Expedited (15-DaCompany Report #316127 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Oedema Lariam PS Roche 32 DAY<br />
Prozac C 1/2 TABLET Q.
Date:07/30/02ISR Number: 3955636-9Report Type:Direct Company Report #CTU 173213 Age:22 YR Gender:Female I/FU:I<br />
Outcome<br />
PT<br />
Fear<br />
Anxiety<br />
Insomnia<br />
Paranoia<br />
03-Apr-2012 09:37 AM Page: 456
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Euphoric Mood<br />
Energy Increased<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong> 250 Mg PS Geneva Pharm Tech<br />
Corp 250 MG X 1<br />
Hepatitis A Vaccine C<br />
Yellow Fever Vaccine C<br />
Date:07/31/02ISR Number: 3955740-5Report Type:Expedited (15-DaCompany Report #263822 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Vision Blurred Health Lariam PS Roche 15 DAY<br />
Cardiac Arrest Professional Malarone SS 4 DAY<br />
Tinnitus<br />
Movement Disorder<br />
Grand Mal Convulsion<br />
Dizziness<br />
Deafness<br />
Date:07/31/02ISR Number: 3956338-5Report Type:Direct Company Report #CTU 173279 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion <strong>Mefloquine</strong> ( Lariam) PS ORAL WEEKLY ORAL 3 YR<br />
Epilepsy Acetaminophen C<br />
Aspirin<br />
C<br />
Date:08/02/02ISR Number: 3956916-3Report Type:Expedited (15-DaCompany Report #316127 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pruritus Lariam PS Roche 25 DAY<br />
Oedema Peripheral Prozac C 1/2 TABLET Q.<br />
Date:08/05/02ISR Number: 3957894-3Report Type:Direct Company Report #CTU 173530 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Lariam PS<br />
Date:08/07/02ISR Number: 3959487-0Report Type:Direct Company Report #CTU 173717 Age: Gender:Male I/FU:I<br />
Outcome<br />
PT<br />
Blood Pressure Increased<br />
Depression<br />
Suicidal Ideation
Thinking Abnormal<br />
Anxiety<br />
Chest Pain<br />
Confusional State<br />
Heart Rate Increased<br />
Panic Attack<br />
Fatigue<br />
Feeling Abnormal<br />
03-Apr-2012 09:37 AM Page: 457
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mood Swings<br />
Agitation<br />
Cold Sweat Report Source Product Role Manufacturer Route Dose Duration<br />
Disorientation Lariam PS 1 SERIES<br />
Feeling Hot<br />
Headache<br />
Paranoia<br />
Disturbance In Attention<br />
Insomnia<br />
Memory Impairment<br />
Psychotic Disorder<br />
Date:08/08/02ISR Number: 3959678-9Report Type:Expedited (15-DaCompany Report #318504 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Lariam PS Roche PATIENT<br />
Initial or Prolonged Haemorrhage RECEIVED 6 OR<br />
7 TABLETS IN<br />
TOTAL.<br />
Date:08/12/02ISR Number: 3960866-6Report Type:Expedited (15-DaCompany Report #316035 Age:17 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Irritability Lariam PS Roche 52 DAY<br />
Depression Contraceptive Pill C<br />
Nightmare<br />
Flashback<br />
Hallucination, Visual<br />
Fatigue<br />
Date:08/19/02ISR Number: 3963568-5Report Type:Expedited (15-DaCompany Report #319090 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicide Attempt Lariam PS Roche<br />
Date:08/19/02ISR Number: 3963569-7Report Type:Expedited (15-DaCompany Report #319098 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chills Lariam PS Roche<br />
Initial or Prolonged Crying<br />
Vomiting<br />
Hallucination<br />
Asthenia<br />
Chest Pain<br />
Anxiety
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 458
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/19/02ISR Number: 3963579-XReport Type:Expedited (15-DaCompany Report #319480 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Homicide Lariam PS Roche<br />
Suicide Attempt<br />
Physical Assault<br />
Hypoxic-Ischaemic<br />
Encephalopathy<br />
Date:08/20/02ISR Number: 3964267-6Report Type:Direct Company Report #CTU 174573 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Distress Larium/Mefloquin PS ORAL 1 PER FOR TWO<br />
Anxiety<br />
ORAL<br />
Mental Disorder<br />
Date:08/20/02ISR Number: 3964533-4Report Type:Direct Company Report #CTU 174624 Age:70 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Lariam Tab 250 Mg Ud PS 250 MG Q WK 5 WK<br />
Nitrostat<br />
C<br />
Date:08/21/02ISR Number: 3964552-8Report Type:Expedited (15-DaCompany Report #319597 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asphyxia Lariam PS Roche<br />
Homicide<br />
Feeling Abnormal<br />
Aggression<br />
Date:08/23/02ISR Number: 3965918-2Report Type:Direct Company Report #CTU 174896 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety <strong>Mefloquine</strong> 250mg<br />
Depression Roche PS Roche ORAL 250MG QWEEK<br />
Panic Attack<br />
ORAL<br />
Malaria<br />
Mental Disorder<br />
Date:08/23/02ISR Number: 3965956-XReport Type:Direct Company Report #CTU 174911 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam Standard PS
Bipolar Disorder<br />
Psychomotor Hyperactivity<br />
Insomnia<br />
Mania<br />
03-Apr-2012 09:37 AM Page: 459
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/26/02ISR Number: 3966695-1Report Type:Direct Company Report #CTU 175006 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hypersensitivity Lariam Roche PS Roche ORAL 1 Q WEEK ORAL<br />
Disability<br />
Muscle Spasticity<br />
Required<br />
Tremor<br />
Intervention to<br />
Anaphylactic Reaction<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:08/26/02ISR Number: 3966696-3Report Type:Direct Company Report #CTU 175007 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Asthenia Lariam PS AURICULAR<br />
Intervention to Disorientation (OTIC) 1 WEEKLY<br />
Prevent Permanent Insomnia AURICULAR<br />
Impairment/Damage Dyspnoea<br />
Headache<br />
Vertigo<br />
Vision Blurred<br />
Anxiety<br />
Dizziness<br />
Date:08/26/02ISR Number: 3966794-4Report Type:Direct Company Report #CTU 175027 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Emotional Disorder Lariam PS 1X/WK<br />
Confusional State<br />
Crying<br />
Mania<br />
Panic Attack<br />
Stress<br />
Suicidal Ideation<br />
Anxiety<br />
Depression<br />
Paranoia<br />
Date:08/26/02ISR Number: 3966896-2Report Type:Direct Company Report #CTU 174984 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation Lariam PS ORAL ORAL<br />
Paranoia<br />
Hypoaesthesia<br />
Panic Attack<br />
Confusional State
Date:08/26/02ISR Number: 3966913-XReport Type:Direct Company Report #CTU 174991 Age: Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Psychomotor Skills<br />
Impaired<br />
Memory Impairment<br />
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 460
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Disorientation<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Larium<br />
PS<br />
Date:08/27/02ISR Number: 3967147-5Report Type:Direct Company Report #CTU 175112 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Screaming Lariam 250 Mg Roche PS Roche ORAL 1 TABLE WEEK<br />
Initial or Prolonged Fear ORAL<br />
Hallucination<br />
Nervous System Disorder<br />
Hyperhidrosis<br />
Feeling Cold<br />
Nervousness<br />
Poisoning<br />
Depression<br />
Crying<br />
Anxiety<br />
Flatulence<br />
Panic Attack<br />
Feeling Abnormal<br />
Date:08/27/02ISR Number: 3967173-6Report Type:Direct Company Report #CTU 175102 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam<br />
Anxiety Hoffman-Laroche PS Hoffman-Laroche ORAL 250MG WEEKLY<br />
Paranoia<br />
ORAL<br />
Date:08/28/02ISR Number: 3967043-3Report Type:Expedited (15-DaCompany Report #264418 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pregnancy Lariam PS Roche<br />
Apgar Score Low Quinine Sulfate C 24 HOUR<br />
Premature Separation Of<br />
PERIODS EVERY<br />
Placenta 8 HOURS, 10<br />
Caesarean Section<br />
MG/KG.<br />
Premature Baby TREATED 03 TO 25 DAY<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:08/30/02ISR Number: 3969670-6Report Type:Direct Company Report #CTU 175355 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Paranoia Lariam Roche PS Roche ORAL 1/WEEK ORAL<br />
Panic Reaction<br />
03-Apr-2012 09:37 AM Page: 461
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/03/02ISR Number: 3970033-8Report Type:Direct Company Report #CTU 175541 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression <strong>Mefloquine</strong> Hoffman<br />
Paranoia Laroche PS Hoffman Laroche BUCCAL 250 MG ONCE A<br />
Insomnia<br />
WEEK BUCCAL<br />
Hallucination<br />
Activities Of Daily<br />
Living Impaired<br />
Date:09/05/02ISR Number: 3970808-5Report Type:Expedited (15-DaCompany Report #320492 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Depression Lariam PS Roche<br />
Overdose<br />
Completed Suicide<br />
Date:09/05/02ISR Number: 3970809-7Report Type:Expedited (15-DaCompany Report #311320 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blindness Health Lariam PS Roche 363 DAY<br />
Visual Field Defect<br />
Professional<br />
Date:09/05/02ISR Number: 3970849-8Report Type:Direct Company Report #CTU 175737 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Abnormal Dreams Mefloquone PS ORAL STANDARD<br />
Other Serious Suicidal Ideation WEEKLY ORAL<br />
Date:09/05/02ISR Number: 3971375-2Report Type:Direct Company Report #CTU 175736 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disturbance In Attention Larium 250 Mg PS<br />
Abnormal Behaviour<br />
Fatigue<br />
Insomnia<br />
Psychomotor Hyperactivity<br />
Nightmare<br />
Date:09/06/02ISR Number: 3971323-5Report Type:Expedited (15-DaCompany Report #A0379543A Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Health Malarone PS Glaxo Wellcome ORAL 18 DAY
Tinnitus Professional <strong>Mefloquine</strong> SS ORAL 1 DAY<br />
03-Apr-2012 09:37 AM Page: 462
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/06/02ISR Number: 3971398-3Report Type:Expedited (15-DaCompany Report #316149 Age:50 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Optic Ischaemic Health Lariam PS Roche 48 DAY<br />
Neuropathy<br />
Professional<br />
Date:09/06/02ISR Number: 3971399-5Report Type:Expedited (15-DaCompany Report #319597 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anger Consumer Lariam PS Roche<br />
Social Avoidant Behaviour<br />
Nervousness<br />
Homicide<br />
Communication Disorder<br />
Tremor<br />
Incoherent<br />
Aggression<br />
Feeling Abnormal<br />
Date:09/06/02ISR Number: 3971407-1Report Type:Expedited (15-DaCompany Report #320484 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Lariam PS Roche<br />
Anger<br />
Date:09/09/02ISR Number: 3974309-XReport Type:Expedited (15-DaCompany Report #02P-056-0198998-00 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myalgia Foreign Isoptine (Verapamil)<br />
Initial or Prolonged Amyotrophy Health (Verapamil) PS ORAL 240 MG, PER<br />
Red Blood Cell Professional ORAL<br />
Sedimentation Rate Insuline Humalog SS<br />
Increased <strong>Mefloquine</strong> SS ORAL 250 MG, 1 IN<br />
Pain<br />
1 WK, PER<br />
Systemic Inflammatory<br />
ORAL<br />
Response Syndrome Perindopril SS ORAL 2 MG, PER<br />
ORAL<br />
Simvastatin SS ORAL PER ORAL<br />
Insuline "Umuline" SS SUBCUTANEOUS SUBCUTANEOUS<br />
Acetylsalicylic Acid SS ORAL PER ORAL<br />
Date:09/10/02ISR Number: 3974021-7Report Type:Direct Company Report #CTU 176057 Age:37 YR Gender:Male I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Paranoia
Fatigue<br />
Hyperventilation<br />
Agitation<br />
Dizziness<br />
Hallucination<br />
Insomnia<br />
Fear<br />
Nightmare<br />
03-Apr-2012 09:37 AM Page: 463
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Panic Disorder<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam 250mg Roche PS Roche ORAL 1 TABLE ONCE<br />
PER ORAL<br />
Date:09/10/02ISR Number: 3974116-8Report Type:Direct Company Report #CTU 176039 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Unevaluable Event Lariam 250mg F<br />
Hoffman-Laroche PS F Hoffman-Laroche ORAL 1 TABLE ONCE<br />
A WEEK ORAL<br />
Date:09/11/02ISR Number: 3974266-6Report Type:Expedited (15-DaCompany Report #PERI00202002217 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Red Blood Cell Foreign Coversyl<br />
Initial or Prolonged Sedimentation Rate Health (Perindopril) PS ORAL 2 MG QD PO<br />
Increased Professional Lariam (<strong>Mefloquine</strong>) SS ORAL 250 MG DAILY<br />
Myalgia Other PO<br />
Pain In Extremity<br />
Kardegic<br />
(Acetylsalicylate<br />
Lysine) SS ORAL DAILY PO<br />
Umuline (Insulin<br />
Human Zinc<br />
Suspension) SS SUBCUTANEOUS DAILY SC<br />
Humalog (Insulin<br />
Lispro) SS SUBCUTANEOUS DAILY SC<br />
Lodales<br />
(Simvastatin) SS DAILY<br />
Isoptin (Verapamil<br />
Hydrochloride) SS ORAL DAILY PO<br />
Date:09/12/02ISR Number: 3975274-1Report Type:Direct Company Report #CTU 176271 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Lariam -Mefloaquin-<br />
Fatigue 250 Mg. Roche PS Roche ORAL 250MG WEEKLY<br />
Vertigo<br />
ORAL<br />
Tinnitus<br />
Dysgraphia<br />
Date:09/13/02ISR Number: 3975636-2Report Type:Direct Company Report #CTU 176356 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Insomnia Larium PS ORAL ONCE WEEK<br />
Required Anxiety ORAL<br />
Intervention to Paranoia Plaquenil C<br />
Prevent Permanent Nervousness Ambien C<br />
Impairment/Damage Biaxin C<br />
03-Apr-2012 09:37 AM Page: 464
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/16/02ISR Number: 3975606-4Report Type:Expedited (15-DaCompany Report #321059 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Inflammation Lariam PS Roche 29 DAY<br />
Initial or Prolonged Amyotrophy Lodales SS<br />
Pain Coversyl SS<br />
Pain In Extremity Kardegic SS FORMULATION<br />
Myalgia<br />
LYOPHILISED.<br />
Red Blood Cell Umuline SS<br />
Sedimentation Rate Isoptine SS<br />
Increased Humalog C<br />
Date:09/16/02ISR Number: 3976184-6Report Type:Direct Company Report #CTU 176452 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Aggression <strong>Mefloquine</strong> Hoffman<br />
Hospitalization - Suicidal Ideation La Roche PS Hoffman La Roche ORAL BI-WEEKLY<br />
Initial or Prolonged Homicidal Ideation ORAL<br />
Other Serious<br />
Hallucination<br />
Required<br />
Toxicity To Various<br />
Intervention to<br />
Agents<br />
Prevent Permanent Psychotic Disorder<br />
Impairment/Damage<br />
Date:09/17/02ISR Number: 3977060-5Report Type:Direct Company Report #CTU 176592 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Alopecia Lariam PS ORAL ONCE WEEKLY<br />
Dry Skin<br />
ORAL<br />
Vision Blurred<br />
Feeling Abnormal<br />
Headache<br />
Dizziness<br />
Swelling<br />
Date:09/17/02ISR Number: 3977073-3Report Type:Direct Company Report #CTU 176590 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Lariam 250mg Roche<br />
Initial or Prolonged Illusion Lab PS Roche Lab ORAL ONCE WEEKLY<br />
Other Serious Dyspnoea ORAL<br />
Vertigo<br />
Dizziness<br />
Disorientation<br />
Visual Impairment<br />
Dysphagia<br />
Headache
Anxiety<br />
03-Apr-2012 09:37 AM Page: 465
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/18/02ISR Number: 3977706-1Report Type:Direct Company Report #CTU 176752 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hallucination Lariam PS Roche<br />
Hospitalization - Neurosis Pharmaceuticals ONE PER<br />
Initial or Prolonged Panic Attack<br />
Suicidal Ideation<br />
Night Sweats<br />
Tachycardia<br />
Insomnia<br />
Muscle Spasms<br />
Paranoia<br />
Decreased Appetite<br />
Psychotic Disorder<br />
Thirst<br />
Date:09/23/02ISR Number: 3978040-6Report Type:Expedited (15-DaCompany Report #320927 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Lariam PS Roche 25 DAY<br />
Nausea Amlodipine C 143 DAY<br />
Headache Bendrofluazide C 24 DAY<br />
Vision Blurred<br />
Sedation<br />
Date:09/23/02ISR Number: 3978044-3Report Type:Expedited (15-DaCompany Report #320902 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Feeling Abnormal Lariam PS Roche<br />
Initial or Prolonged Cold Sweat<br />
Hallucination<br />
Aggression<br />
Disorientation<br />
Date:09/23/02ISR Number: 3979340-6Report Type:Direct Company Report #CTU 176999 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delusion Lariam Laroche PS Laroche<br />
Initial or Prolonged Performance Status<br />
Disability<br />
Decreased<br />
Other Serious<br />
Anxiety<br />
Mental Disorder<br />
Blood Pressure Increased<br />
Suicidal Ideation<br />
Abnormal Behaviour<br />
Aggression<br />
Anger
Divorced<br />
Hallucination<br />
Depression<br />
Palpitations<br />
03-Apr-2012 09:37 AM Page: 466
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/24/02ISR Number: 3978154-0Report Type:Expedited (15-DaCompany Report #321488 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Night Sweats Lariam PS Roche 43 DAY<br />
Delirium<br />
Nightmare<br />
Heart Rate Increased<br />
Date:09/26/02ISR Number: 3981417-6Report Type:Direct Company Report #CTU 177370 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Agitation Larium - Roche PS Roche 1 TABLET<br />
Initial or Prolonged Depression WEEKLY<br />
Other Serious<br />
Paraesthesia<br />
Palpitations<br />
Toxicity To Various<br />
Agents<br />
Anxiety<br />
Hypertension<br />
Nausea<br />
Date:09/27/02ISR Number: 3982056-3Report Type:Direct Company Report #CTU 177430 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Chills Lariam PS Roche ORAL 250MG WEEK<br />
Intervention to Paraesthesia ORAL<br />
Prevent Permanent Renal Pain Doxycycline C<br />
Impairment/Damage Pollakiuria Bactrim C<br />
Morbid Thoughts Ciprofloxacin C<br />
Feeling Of Despair<br />
Panic Attack<br />
Insomnia<br />
Sleep Disorder<br />
Date:09/27/02ISR Number: 3985386-4Report Type:Expedited (15-DaCompany Report #321488 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Health Lariam (<strong>Mefloquine</strong><br />
Heart Rate Increased Professional Hydrochloride) PS ORAL ORAL<br />
Night Sweats<br />
Delirium<br />
Date:10/01/02ISR Number: 3985463-8Report Type:Direct Company Report #CTU 177643 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Facial Pain Lariam PS ORAL WEEKLY ORAL<br />
Dizziness<br />
Microgestin-Birth<br />
Nightmare Control- C<br />
Disorientation<br />
03-Apr-2012 09:37 AM Page: 467
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/03/02ISR Number: 3985611-XReport Type:Direct Company Report #CTU 177838 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation <strong>Mefloquine</strong> PS 1 PILL WEEK<br />
Depression<br />
Mood Altered<br />
Date:10/07/02ISR Number: 3984410-2Report Type:Expedited (15-DaCompany Report #A0379543A Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Malarone PS Glaxo Wellcome ORAL 18 DAY<br />
Diarrhoea <strong>Mefloquine</strong> SS ORAL 1 DAY<br />
Irritability Mvi C<br />
Date:10/07/02ISR Number: 3984911-7Report Type:Expedited (15-DaCompany Report #305877 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lung Infiltration Health Lariam PS Roche DESCRIBED AS<br />
Dyspnoea Professional 3 TABLETS IN<br />
THE MORNING,<br />
2 IN THE<br />
MIDDLE OF THE 1 DAY<br />
Date:10/07/02ISR Number: 3984914-2Report Type:Expedited (15-DaCompany Report #321938 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vomiting <strong>Mefloquine</strong><br />
Nausea Hydrochloride PS Roche<br />
Disturbance In Attention<br />
Abnormal Dreams<br />
Date:10/07/02ISR Number: 3984952-XReport Type:Expedited (15-DaCompany Report #316199 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Optic Neuritis Health Lariam PS Roche 366 DAY<br />
Professional<br />
Date:10/07/02ISR Number: 3984973-7Report Type:Expedited (15-DaCompany Report #319597 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Consumer Lariam PS Roche<br />
Social Avoidant Behaviour<br />
Dysphemia
Tremor<br />
Aggression<br />
Feeling Abnormal<br />
Nervousness<br />
Abnormal Behaviour<br />
Incoherent<br />
Anger<br />
03-Apr-2012 09:37 AM Page: 468
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/07/02ISR Number: 3987080-2Report Type:Direct Company Report #CTU 178124 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Lariam- Roche Labs PS Roche Labs ORAL WEEKLY- 136<br />
Myalgia<br />
WKS ORAL<br />
Fibromyalgia<br />
Date:10/07/02ISR Number: 3987117-0Report Type:Direct Company Report #CTU 178129 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Lariam -Roche Labs PS Roche Labs ORAL WEEKLY<br />
Tinnitus<br />
-150WKS ORAL<br />
Depression<br />
Headache<br />
Abnormal Dreams<br />
Anxiety<br />
Pruritus<br />
Date:10/07/02ISR Number: 3990430-4Report Type:Direct Company Report #CTU 178059 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Headache <strong>Mefloquine</strong><br />
Panic Disorder<br />
Hydrochloride 250mg<br />
Abnormal Behaviour Geneva Pharm Tech PS Geneva Pharm Tech ONCE WEEKL<br />
Abdominal Pain<br />
Confusional State<br />
Mood Swings<br />
Emotional Disorder<br />
Insomnia<br />
Anxiety<br />
Decreased Appetite<br />
Date:10/08/02ISR Number: 3990489-4Report Type:Direct Company Report #CTU 178231 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aggression Larium PS 2 TABLETS 10<br />
Vomiting<br />
DAYS<br />
Abnormal Behaviour<br />
Malaise<br />
Mood Swings<br />
Date:10/09/02ISR Number: 3987337-5Report Type:Direct Company Report #CTU 178319 Age:41 YR Gender:Female I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Mood Swings
Pulmonary Embolism<br />
Back Pain<br />
Chest Pain<br />
Dyspepsia<br />
Aggression<br />
Dizziness<br />
Stress<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 469
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Feeling Abnormal<br />
Headache<br />
Sleep Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Panic Attack Larium PS ORAL 1 WEEKLY ORAL<br />
Oral Ulcer-Type<br />
Products<br />
C<br />
Cimetidine<br />
C<br />
Warfarin<br />
C<br />
Date:10/10/02ISR Number: 3987531-3Report Type:Expedited (15-DaCompany Report #320484 Age:52 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Extrasystoles Consumer Lariam PS Roche 61 DAY<br />
Initial or Prolonged Panic Attack<br />
Insomnia<br />
Vestibular Disorder<br />
Hallucination<br />
Suicidal Ideation<br />
Cognitive Disorder<br />
Depression<br />
Toxicity To Various<br />
Agents<br />
Brain Injury<br />
Tremor<br />
Illusion<br />
Nervous System Disorder<br />
Hyperacusis<br />
Anger<br />
Date:10/11/02ISR Number: 3988282-1Report Type:Expedited (15-DaCompany Report #322910 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Lariam PS Roche<br />
Gastrointestinal<br />
Haemorrhage<br />
Date:10/14/02ISR Number: 3989477-3Report Type:Expedited (15-DaCompany Report #265329 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Illusion Lariam PS Roche<br />
Hallucination<br />
Disturbance In Attention<br />
Dysarthria<br />
Panic Attack
Date:10/15/02ISR Number: 3989917-XReport Type:Expedited (15-DaCompany Report #323150 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Health Lariam PS Roche<br />
Initial or Prolonged Psoas Sign Professional Previscan I<br />
Coagulopathy<br />
Haematoma<br />
03-Apr-2012 09:37 AM Page: 470
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/15/02ISR Number: 3991717-1Report Type:Direct Company Report #CTU 178703 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Contusion Lariam Hoffman La<br />
Required Decreased Appetite Roche PS Hoffman La Roche ORAL STANDARD ONCE<br />
Intervention to Heart Rate Increased A WEEK ORAL<br />
Prevent Permanent Tremor Cardizem Cd C<br />
Impairment/Damage Chills Vioxx C<br />
Syncope Centrum Silver C<br />
Nausea<br />
Hypertension<br />
Hyperhidrosis<br />
Date:10/16/02ISR Number: 3990823-5Report Type:Expedited (15-DaCompany Report #86887 Age:43 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Pain Consumer Lariam Tablets PS Roche 49 DAY<br />
Suicidal Ideation<br />
Myalgia<br />
Arthropod Bite<br />
Csf Protein Abnormal<br />
Malaise<br />
Angina Pectoris<br />
Atrophy<br />
Feeling Of Body<br />
Temperature Change<br />
Asthenia<br />
Flushing<br />
Convulsion<br />
Tinnitus<br />
Nervousness<br />
Muscular Weakness<br />
Sleep Disorder<br />
Neck Pain<br />
Peripheral Sensory<br />
Neuropathy<br />
Epistaxis<br />
Fatigue<br />
Date:10/17/02ISR Number: 3991243-XReport Type:Expedited (15-DaCompany Report #323291 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Respiratory Failure Lariam PS Roche 63 DAY<br />
Maternal Drugs Affecting<br />
Foetus<br />
Microcephaly<br />
Pregnancy
Date:10/17/02ISR Number: 3997443-7Report Type:Expedited (15-DaCompany Report #323150 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coagulopathy Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Drug Interaction Health Hydrochloride) PS ORAL ORAL<br />
Muscle Haemorrhage Professional Previscan<br />
(Fluindione) SS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 471
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/18/02ISR Number: 3992136-4Report Type:Expedited (15-DaCompany Report #200841 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Accommodation Disorder Health Lariam PS Roche CUMULATIVE<br />
Initial or Prolonged Speech Disorder Professional DOSAGE= 750<br />
Disability Dry Mouth MG 380 DAY<br />
Dysphagia<br />
Meningococcal<br />
Paraesthesia Vaccine C 1 DAY<br />
Emotional Disorder Flu Vaccine C 1 DAY<br />
Panic Attack<br />
Asthenia<br />
Movement Disorder<br />
Tinnitus<br />
Vision Blurred<br />
Gait Disturbance<br />
Feeling Drunk<br />
Malaise<br />
Fall<br />
Heterophoria<br />
Tremor<br />
Balance Disorder<br />
Hallucination, Visual<br />
Diarrhoea<br />
Diplopia<br />
Hydrocephalus<br />
Arrhythmia<br />
Somnolence<br />
Fatigue<br />
Hyperventilation<br />
Nausea<br />
Dyspnoea<br />
Disturbance In Attention<br />
Date:10/21/02ISR Number: 3993095-0Report Type:Expedited (15-DaCompany Report #305230 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Congenital Tongue Anomaly Lariam PS Roche 36 DAY<br />
Cryptorchism<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:10/22/02ISR Number: 3994346-9Report Type:Expedited (15-DaCompany Report #314899 Age: Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Incontinence<br />
Paranoia<br />
Speech Disorder<br />
Antisocial Behaviour<br />
Loss Of Libido
Communication Disorder<br />
Reading Disorder<br />
Depression<br />
Memory Impairment<br />
Incoherent<br />
Circulatory Collapse<br />
Hallucination<br />
Disorientation<br />
03-Apr-2012 09:37 AM Page: 472
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Psychotic Disorder<br />
Gait Disturbance<br />
Agitation Report Source Product Role Manufacturer Route Dose Duration<br />
Convulsion Lariam PS Roche EVERY WEEK. 9 DAY<br />
Hostility Valium SS Roche 1 DAY<br />
Drug Ineffective<br />
Aggression<br />
Headache<br />
Delusion<br />
Fatigue<br />
Mania<br />
Confusional State<br />
Date:10/22/02ISR Number: 3994559-6Report Type:Expedited (15-DaCompany Report #323614 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lung Infection Lariam PS Roche<br />
Initial or Prolonged Hypoxia<br />
Lung Disorder<br />
Date:10/22/02ISR Number: 3994562-6Report Type:Expedited (15-DaCompany Report #323551 Age:13 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Haemorrhagic Lariam PS Roche<br />
Date:10/23/02ISR Number: 3995061-8Report Type:Expedited (15-DaCompany Report #265329 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Lariam PS Roche<br />
Hallucinations, Mixed<br />
Panic Attack<br />
Illusion<br />
Disturbance In Attention<br />
Hallucination, Olfactory<br />
Dysarthria<br />
Date:10/24/02ISR Number: 3996343-6Report Type:Direct Company Report #CTU 179445 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Lariam 250 Mg<br />
Required Insomnia Mephaquin PS ORAL 1 PILL ONCE A<br />
Intervention to Amnesia WEEK ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Crying<br />
Anger<br />
Vertigo
Mood Swings<br />
03-Apr-2012 09:37 AM Page: 473
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/25/02ISR Number: 3998338-5Report Type:Direct Company Report #CTU 179642 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspepsia Lariam 250 Mg<br />
Depression<br />
Mefloquin Mfr<br />
-Hoffman La Roche PS Hoffman La Roche 250 MG EVERY<br />
WEEK (ONE'S<br />
A WEEK) 6 WK<br />
Date:10/25/02ISR Number: 4000382-9Report Type:Direct Company Report #CTU 179617 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Feeling Abnormal <strong>Mefloquine</strong> (Lariam) PS ORAL 250 MG ONCE A<br />
Other Serious Stress WEEK ORAL<br />
Depression<br />
Anxiety<br />
Date:10/29/02ISR Number: 3998705-XReport Type:Expedited (15-DaCompany Report #323614 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoxia Lariam PS Roche<br />
Initial or Prolonged Infection<br />
Lung Disorder<br />
Date:10/31/02ISR Number: 4000596-8Report Type:Expedited (15-DaCompany Report #323900 Age: Gender:Not SpecifiI/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Lariam PS Roche 549 DAY<br />
Foetus<br />
Abortion Spontaneous<br />
Paternal Drugs Affecting<br />
Foetus<br />
Date:11/01/02ISR Number: 4003334-8Report Type:Direct Company Report #CTU 180174 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nervousness<br />
Lariam (<strong>Mefloquine</strong><br />
Nightmare Hydrochloride) PS ONCE A WEEK<br />
Weight Decreased<br />
Anxiety<br />
Date:11/01/02ISR Number: 4003598-0Report Type:Direct Company Report #CTU 180150 Age:36 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Larium PS ORAL 1 PILL WEEK<br />
Nausea<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 474
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/04/02ISR Number: 4002692-8Report Type:Expedited (15-DaCompany Report #324511 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haemolytic Anaemia Lariam PS Roche<br />
Initial or Prolonged<br />
Date:11/06/02ISR Number: 4004677-4Report Type:Expedited (15-DaCompany Report #324735 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Gait Disturbance Lariam PS Roche 213 DAY<br />
Visual Impairment<br />
Tinnitus<br />
Date:11/07/02ISR Number: 4005549-1Report Type:Direct Company Report #CTU 180546 Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Lariam La Roche PS La Roche ORAL 1 TABLE ONCE<br />
Initial or Prolonged Psychotic Disorder A WEEK ORAL<br />
Disability<br />
Delusion<br />
Required<br />
Hallucination<br />
Intervention to<br />
Thinking Abnormal<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:11/13/02ISR Number: 4012180-0Report Type:Expedited (15-DaCompany Report #321428 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Foreign Lariam (<strong>Mefloquine</strong><br />
Foetus Health Hydrochloride) 250<br />
Abortion Spontaneous Professional Mg PS ORAL 250 MG 1 PER<br />
Pregnancy<br />
ONE DOSE ORAL<br />
Date:11/13/02ISR Number: 4012181-2Report Type:Expedited (15-DaCompany Report #324511 Age:25 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haemorrhage Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Haemolytic Anaemia Health Hydrochloride) PS<br />
Professional<br />
Date:11/13/02ISR Number: 4012182-4Report Type:Expedited (15-DaCompany Report #323150 Age:48 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Muscle Haemorrhage Foreign Lariam (<strong>Mefloquine</strong>
Initial or Prolonged Pain Health Hydrochloride) PS ORAL ORAL<br />
Flatulence Professional Previscan<br />
Constipation (Fluindione) SS ORAL 1 DOSE FORM<br />
Drug Interaction<br />
Hypocoagulable State<br />
Lovenox (Enoxaparin<br />
Sodium)<br />
C<br />
DAILY ORAL<br />
03-Apr-2012 09:37 AM Page: 475
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/13/02ISR Number: 4012183-6Report Type:Expedited (15-DaCompany Report #324949 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Foreign Lariam(<strong>Mefloquine</strong><br />
Initial or Prolonged Multiple Sclerosis Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Optic Neuritis Professional PER WEEK ORAL<br />
Date:11/15/02ISR Number: 4011394-3Report Type:Direct Company Report #CTU 181033 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Asthenia Lariam (Mefloquin) PS ORAL 1X WK 1 WK<br />
Hospitalization - Anxiety BEFOR ORAL; 1<br />
Initial or Prolonged Blood Pressure Decreased X WK 4WKS AFT<br />
Disability Psychotic Disorder ORAL<br />
Congenital Anomaly Dizziness Prozac C<br />
Required Chills Fludrocortizone C<br />
Intervention to<br />
Headache<br />
Prevent Permanent Suicidal Ideation<br />
Impairment/Damage<br />
Date:11/18/02ISR Number: 4013350-8Report Type:Expedited (15-DaCompany Report #324949 Age:29 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Optic Neuritis Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:11/18/02ISR Number: 4013366-1Report Type:Expedited (15-DaCompany Report #321428 Age: Gender:Unknown I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Maternal Drugs Affecting Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Foetus Health Hydrochloride) 250<br />
Abortion Spontaneous Professional Mg PS ORAL 250 MG 1 PER<br />
Pregnancy<br />
ONE DOSE ORAL<br />
Date:11/19/02ISR Number: 4014667-3Report Type:Expedited (15-DaCompany Report #325394 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicide Attempt Other Lariam PS ORAL ORAL<br />
Suicidal Ideation<br />
Homicidal Ideation<br />
Paranoia<br />
Confusional State<br />
Depression<br />
Hallucination
Date:11/19/02ISR Number: 4014668-5Report Type:Expedited (15-DaCompany Report #325416 Age:27 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Hallucination<br />
Confusional State<br />
03-Apr-2012 09:37 AM Page: 476
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Delusion<br />
Anger<br />
Loss Of Consciousness Report Source Product Role Manufacturer Route Dose Duration<br />
Tremor Other Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) PS<br />
Date:11/20/02ISR Number: 4012638-4Report Type:Direct Company Report #CTU 181421 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hallucination, Auditory <strong>Mefloquine</strong> (Lariam)<br />
Other Serious Homicidal Ideation (Roche Pharm) PS Roche Pharm 2 DOSES OVER<br />
Hypoaesthesia<br />
2 WEEKS<br />
Anxiety<br />
Schizophrenia, Paranoid<br />
Type<br />
Suicidal Ideation<br />
Depression<br />
Psychotic Disorder<br />
Restlessness<br />
Depersonalisation<br />
Date:11/20/02ISR Number: 4012681-5Report Type:Direct Company Report #CTU 181369 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams Larium PS<br />
Fear<br />
Disorientation<br />
Hallucination<br />
Date:11/26/02ISR Number: 4017612-XReport Type:Expedited (15-DaCompany Report #324855 Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Aspartate Foreign Lariam (<strong>Mefloquine</strong><br />
Aminotransferase Other Hydrochloride) PS ORAL ORAL<br />
Increased<br />
Pneumonitis<br />
Cerebral Atrophy<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Prothrombin Time<br />
Prolonged<br />
Alcohol Withdrawal<br />
Syndrome<br />
Haemoglobin Abnormal<br />
Platelet Count Abnormal<br />
Alanine Aminotransferase<br />
Increased
Hyperhidrosis<br />
Blood Potassium Abnormal<br />
Blood Sodium Abnormal<br />
Livedo Reticularis<br />
Nosocomial Infection<br />
Encephalopathy<br />
Respiratory Alkalosis<br />
Peripheral Coldness<br />
03-Apr-2012 09:37 AM Page: 477
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/26/02ISR Number: 4018662-XReport Type:Expedited (15-DaCompany Report #325725 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Multiple Sclerosis Foreign Lariam (Meloquine<br />
Optic Neuritis Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Professional<br />
Yellow Fever Vaccine<br />
(Yellow Fever<br />
Vaccine)<br />
C<br />
PER WEEK ORAL<br />
Date:11/26/02ISR Number: 4018664-3Report Type:Expedited (15-DaCompany Report #323150 Age:48 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Motility Foreign Lariam(<strong>Mefloquine</strong><br />
Initial or Prolonged Disorder Health Hydrochloride) PS ORAL ORAL<br />
Hypocoagulable State Professional Previsan<br />
Flatulence (Fluindione) SS ORAL DOSE FORM<br />
Pain<br />
DAILY ORAL<br />
Drug Interaction<br />
Muscle Haemorrhage<br />
Date:11/27/02ISR Number: 4019178-7Report Type:Expedited (15-DaCompany Report #304340 Age:15 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Bipolar Disorder Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hallucination Hydrochloride) 250<br />
Disability Hemiparesis Mg PS ORAL 250 MG 1 PER<br />
Myalgia<br />
WEEK ORAL<br />
Blindness<br />
Depression<br />
Paraesthesia<br />
Depressed Level Of<br />
Consciousness<br />
Headache<br />
Paranoia<br />
Eye Swelling<br />
Hypoaesthesia<br />
Suicidal Ideation<br />
Date:11/27/02ISR Number: 4019228-8Report Type:Expedited (15-DaCompany Report #325881 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Jaundice Foreign Lariam (<strong>Mefloquine</strong><br />
Erythema Multiforme Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
Arthralgia<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
WEEK ORAL
Date:12/06/02ISR Number: 4022746-XReport Type:Expedited (15-DaCompany Report #324949 Age:29 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Multiple Sclerosis<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 478
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Optic Neuritis<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:12/09/02ISR Number: 4020858-8Report Type:Direct Company Report #CTU 182353 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Personality Change Lariam - <strong>Mefloquine</strong>- PS<br />
Pain<br />
Abnormal Behaviour<br />
Date:12/09/02ISR Number: 4020882-5Report Type:Direct Company Report #CTU 182327 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Lariam PS ORAL 250MG WEEKLY<br />
ORAL<br />
Date:12/16/02ISR Number: 4027039-2Report Type:Expedited (15-DaCompany Report #319597 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aggression Other Lariam (<strong>Mefloquine</strong><br />
Homicide Hydrochloride) 250<br />
Anger Mg PS ORAL ORAL<br />
Social Avoidant Behaviour<br />
Tremor<br />
Speech Disorder<br />
Feeling Abnormal<br />
Nervousness<br />
Date:12/16/02ISR Number: 4027400-6Report Type:Expedited (15-DaCompany Report #324949 Age:29 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Multiple Sclerosis Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Optic Neuritis Professional PER WEEK ORAL<br />
Date:12/23/02ISR Number: 4028551-2Report Type:Direct Company Report #CTU 183235 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam 250mg Roche
Panic Reaction Labs. PS Roche Labs 1 TABLE<br />
Toxicity To Various<br />
WEEKLY<br />
Agents<br />
Confusional State<br />
Irritability<br />
03-Apr-2012 09:37 AM Page: 479
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/23/02ISR Number: 4028552-4Report Type:Direct Company Report #CTU 183236 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam (Mefloqune)<br />
Anxiety 250gm Roche Labs. PS Roche Labs ONE TABLE PER<br />
Dizziness<br />
WEEK<br />
Nausea<br />
Paranoia<br />
Tinnitus<br />
Vision Blurred<br />
Toxicity To Various<br />
Agents<br />
Muscle Twitching<br />
Muscle Spasms<br />
Date:12/23/02ISR Number: 4028553-6Report Type:Direct Company Report #CTU 183237 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Cerebral Atrophy Larium 250 Mg<br />
Required Nausea Roche Lab PS Roche Lab ORAL 1 PER 7 WEEKS<br />
Intervention to Balance Disorder ORAL<br />
Prevent Permanent Dizziness<br />
Impairment/Damage Syncope<br />
Date:12/23/02ISR Number: 4033084-3Report Type:Expedited (15-DaCompany Report #327602 Age:47 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Anticholinergic Syndrome Literature <strong>Mefloquine</strong><br />
Intervention to Health Hydrochloride PS<br />
Prevent Permanent<br />
Professional<br />
Impairment/Damage<br />
Date:12/23/02ISR Number: 4033392-6Report Type:Expedited (15-DaCompany Report #324949 Age:29 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Optic Neuritis Health Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
Professional<br />
PER WEEK ORAL<br />
Date:12/26/02ISR Number: 4030737-8Report Type:Direct Company Report #CTU 183365 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Disorder Larium<br />
Required Visual Impairment Unsure-Gov.'T Issue PS 1 PILL PER<br />
Intervention to Diarrhoea WEEK
Prevent Permanent Vomiting Furamide C<br />
Impairment/Damage Dizziness Metronidozol C<br />
Insomnia Bactrim C<br />
Nausea Fazygen C<br />
Panic Attack<br />
Anxiety<br />
Hallucination<br />
Skin Ulcer<br />
03-Apr-2012 09:37 AM Page: 480
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/06/03ISR Number: 4039907-6Report Type:Expedited (15-DaCompany Report #323965 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nightmare Consumer Lariam (<strong>Mefloquine</strong><br />
Paraesthesia Hydrochloride) 250<br />
Electrocardiogram T Wave Mg PS ORAL 250 MG 1 PER<br />
Abnormal<br />
WEEK ORAL<br />
Agitation<br />
Electrocardiogram<br />
Abnormal<br />
Fear<br />
Hypotension<br />
Panic Attack<br />
Gastrointestinal Disorder<br />
Abnormal Dreams<br />
Convulsion<br />
Diarrhoea<br />
Anxiety<br />
Conduction Disorder<br />
Supraventricular<br />
Tachycardia<br />
Dyspepsia<br />
Constipation<br />
Dizziness<br />
Gastrooesophageal Reflux<br />
Disease<br />
Insomnia<br />
Restlessness<br />
Chills<br />
Tremor<br />
Mental Disorder<br />
Atrioventricular Block<br />
First Degree<br />
Choking Sensation<br />
Nausea<br />
Sensory Loss<br />
Hypertension<br />
Night Sweats<br />
Vision Blurred<br />
Eructation<br />
Flushing<br />
Palpitations<br />
Supraventricular<br />
Extrasystoles<br />
Weight Decreased<br />
Dysphagia<br />
Date:01/08/03ISR Number: 4040684-3Report Type:Expedited (15-DaCompany Report #328238 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paresis Foreign Lariam (<strong>Mefloquine</strong>
Health Hydrochloride) 250<br />
Professional Mg PS<br />
Other<br />
03-Apr-2012 09:37 AM Page: 481
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/14/03ISR Number: 4042880-8Report Type:Expedited (15-DaCompany Report #328748 Age:54 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Labyrinthitis Foreign <strong>Mefloquine</strong><br />
Bradyphrenia Literature Hydrochloride<br />
Headache Health (<strong>Mefloquine</strong><br />
Hypercholesterolaemia Professional Hydrochloride) PS 250 MG 1<br />
Clonic Convulsion<br />
Cerivastatin<br />
(Cerivastatin<br />
Sodium)<br />
C<br />
PER WEEK<br />
Date:01/14/03ISR Number: 4043507-1Report Type:Expedited (15-DaCompany Report #303888 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Panic Attack Foreign Lariam (<strong>Mefloquine</strong><br />
Required Visual Impairment Health Hydrochloride) PS ORAL 250 MG DAILY<br />
Intervention to Agoraphobia Professional ORAL<br />
Prevent Permanent Psychotic Disorder Other Chloroquine<br />
Impairment/Damage Nervousness (Chloroquine) C<br />
Date:01/16/03ISR Number: 4042779-7Report Type:Direct Company Report #CTU 184663 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Psychotic Disorder Lariam PS 2 TABLE<br />
Hospitalization - Aggression<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:01/17/03ISR Number: 4043730-6Report Type:Direct Company Report #CTU 184788 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Panic Attack Lariam 250mg.#7<br />
Paraesthesia<br />
Roche? Not On Label<br />
Eustachian Tube Disorder I Have Left PS Roche? ORAL 1-250 WEEK<br />
Feeling Abnormal<br />
ORAL<br />
Hearing Impaired<br />
Insomnia<br />
Decreased Appetite<br />
Dissociation<br />
Tinnitus<br />
Disturbance In Attention<br />
Loss Of Consciousness<br />
Anxiety
Fatigue<br />
Suicidal Ideation<br />
Asthenia<br />
03-Apr-2012 09:37 AM Page: 482
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/21/03ISR Number: 4045508-6Report Type:Expedited (15-DaCompany Report #328238 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Foreign Lariam (<strong>Mefloquine</strong><br />
Paresis Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG ORAL<br />
Other<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
Date:01/21/03ISR Number: 4045971-0Report Type:Expedited (15-DaCompany Report #328238 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paresis Foreign Lariam (<strong>Mefloquine</strong><br />
Viith Nerve Paralysis Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG ORAL<br />
Other<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
Date:01/23/03ISR Number: 4046538-0Report Type:Expedited (15-DaCompany Report #329521 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Thrombocytopenia Foreign Lariam<br />
Somnolence Health (<strong>Mefloquine</strong><br />
Haemolysis Professional Hydrochloride) PS ORAL 3 DOSE FORM<br />
Renal Disorder<br />
DAILY ORAL<br />
Date:01/23/03ISR Number: 4046936-5Report Type:Expedited (15-DaCompany Report #325725 Age:43 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Optic Neuritis Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Multiple Sclerosis Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Professional<br />
Yellow Fever Vaccine<br />
(Yellow Fever<br />
Vaccine)<br />
C<br />
1 PER WEEK<br />
ORAL<br />
Date:01/23/03ISR Number: 4046940-7Report Type:Expedited (15-DaCompany Report #312331 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Foreign Lariam (<strong>Mefloquine</strong><br />
Foetus Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Intra-Uterine Death Professional 1 PER WEEK<br />
Antepartum Haemorrhage<br />
ORAL<br />
Umbilical Cord<br />
Trandate (Labetalol
Abnormality Hydrochloride) C<br />
03-Apr-2012 09:37 AM Page: 483
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/23/03ISR Number: 4046951-1Report Type:Expedited (15-DaCompany Report #329423 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hypertension Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Professional<br />
Trandate (Labetalol<br />
Hydrochloride)<br />
C<br />
1 PER WEEK<br />
ORAL<br />
Date:01/23/03ISR Number: 4046976-6Report Type:Expedited (15-DaCompany Report #329220 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Carpal Tunnel Syndrome Consumer Lariam (<strong>Mefloquine</strong><br />
Intervention to Diarrhoea Hydrochloride) PS ORAL 1 PER WEEK<br />
Prevent Permanent Insomnia ORAL<br />
Impairment/Damage Depression Adderall<br />
Post-Traumatic Stress<br />
(Amphetamine<br />
Disorder<br />
Aspartate/Amphetamin<br />
Disturbance In Social<br />
e<br />
Behaviour<br />
Sulfate/Dextroamphet C<br />
Muscle Twitching<br />
Chills<br />
Vomiting<br />
Hyperacusis<br />
Disturbance In Attention<br />
Fatigue<br />
Memory Impairment<br />
Nervousness<br />
Restless Legs Syndrome<br />
Paranoia<br />
Date:01/26/03ISR Number: 4050195-7Report Type:Expedited (15-DaCompany Report #328612 Age:18 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cyanosis Foreign Lariam (<strong>Mefloquine</strong><br />
Fatigue Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Convulsion Professional WEEK ORAL<br />
Urinary Incontinence<br />
Bcg Vaccine (Bcg<br />
Disease Recurrence Vaccine) C<br />
Muscle Rigidity<br />
Date:01/27/03ISR Number: 4047938-5Report Type:Expedited (15-DaCompany Report #329220 Age:47 YR Gender:Male I/FU:F<br />
Outcome<br />
Required<br />
Intervention to<br />
PT<br />
Diarrhoea<br />
Vomiting
Prevent Permanent<br />
Impairment/Damage<br />
Fatigue<br />
Paranoia<br />
Depression<br />
Disturbance In Attention<br />
Hyperacusis<br />
Muscle Twitching<br />
Memory Impairment<br />
Post-Traumatic Stress<br />
03-Apr-2012 09:37 AM Page: 484
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorder<br />
Restless Legs Syndrome<br />
Chills<br />
Insomnia Report Source Product Role Manufacturer Route Dose Duration<br />
Nervousness Consumer Lariam (<strong>Mefloquine</strong><br />
Carpal Tunnel Syndrome Hydrochloride) PS ORAL 1 PER WEEK<br />
ORAL<br />
(Amphetamine<br />
Aspartate/Amphetamin<br />
e<br />
Sulfate/Dextroamphet<br />
amine<br />
C<br />
Adderall<br />
C<br />
Date:01/27/03ISR Number: 4048136-1Report Type:Expedited (15-DaCompany Report #328612 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urinary Incontinence Foreign Lariam (<strong>Mefloquine</strong><br />
Convulsion Health Hydrochloride) 250<br />
Syncope Professional Mg PS ORAL 250 MG 1 PER<br />
Muscle Rigidity<br />
Bcg Vaccine (Bcg<br />
Vaccine)<br />
C<br />
WEEK ORAL<br />
Date:01/27/03ISR Number: 4048137-3Report Type:Expedited (15-DaCompany Report #329947 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myoglobin Blood Increased Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Heat Stroke Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Hepatocellular Injury Professional 1 PER ONE<br />
Blood Creatine<br />
DOSE ORAL<br />
Phosphokinase Increased<br />
Date:01/27/03ISR Number: 4048138-5Report Type:Expedited (15-DaCompany Report #329796 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Erythema Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Burning Sensation Health Hydrochloride) PS<br />
Facial Pain<br />
Professional<br />
Mental Disorder<br />
Tenderness<br />
Emotional Disorder<br />
Date:01/29/03ISR Number: 4050367-1Report Type:Expedited (15-DaCompany Report #324511 Age:25 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Convulsion Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Headache Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Transfusion Reaction Professional WEEK ORAL<br />
Anaemia Haemolytic<br />
Propecia<br />
Autoimmune (Finasteride) C<br />
Splenomegaly<br />
03-Apr-2012 09:37 AM Page: 485
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/29/03ISR Number: 4050369-5Report Type:Expedited (15-DaCompany Report #309389 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Maternal Drugs Affecting Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Foetus Health Hydrochloride) PS ORAL 250 MG 1 PER<br />
Pregnancy Professional WEEK ORAL<br />
Abortion Spontaneous Other Zinnat (Cefuroxime<br />
Axetil Or Cefuroxime<br />
Sodium/*Lidocaine<br />
Hydrochloride) C<br />
Date:01/30/03ISR Number: 4049529-9Report Type:Direct Company Report #CTU 185653 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paranoia Larium PS<br />
Psychotic Disorder<br />
Date:02/03/03ISR Number: 4053696-0Report Type:Expedited (15-DaCompany Report #2003-102650-NL B0006-2003 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Syncope Bcg Vaccine PS<br />
Convulsion <strong>Mefloquine</strong> SS 250 MG<br />
Date:02/05/03ISR Number: 4052084-0Report Type:Direct Company Report #CTU 185987 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Syncope <strong>Mefloquine</strong> PS<br />
Convulsion<br />
Bacillus Calmette<br />
Urinary Incontinence Guerin Vaccine C<br />
Date:02/06/03ISR Number: 4055962-1Report Type:Expedited (15-DaCompany Report #329796 Age:19 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Emotional Disorder Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Burning Sensation Health Hydrochloride) PS<br />
Facial Pain<br />
Professional<br />
Mental Disorder<br />
Erythema<br />
Sensation Of Pressure<br />
Tenderness<br />
Date:02/07/03ISR Number: 4054267-2Report Type:Expedited (15-DaCompany Report #324949 Age:29 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Multiple Sclerosis Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Blood Immunoglobulin G Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Increased Professional 1 PER WEEK<br />
Headache<br />
ORAL<br />
Optic Neuritis<br />
Measles<br />
03-Apr-2012 09:37 AM Page: 486
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/13/03ISR Number: 4056506-0Report Type:Direct Company Report #CTU 186611 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt <strong>Mefloquine</strong> 250 Mg<br />
Hospitalization - Overdose Geneva PS Geneva ORAL ONE WEEKLY<br />
Initial or Prolonged<br />
ORAL<br />
Required Doxycycline C<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:02/13/03ISR Number: 4057400-1Report Type:Expedited (15-DaCompany Report #328238 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Viith Nerve Paralysis Foreign Lariam (<strong>Mefloquine</strong><br />
Paresis Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG ORAL<br />
Other<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
Date:02/13/03ISR Number: 4057959-4Report Type:Expedited (15-DaCompany Report #329220 Age:47 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Depression Consumer Lariam (<strong>Mefloquine</strong><br />
Intervention to Fatigue Hydrochloride) PS 1 PER WEEK<br />
Prevent Permanent Insomnia Tramadol (Tramadol<br />
Impairment/Damage Skin Discolouration Hydrochloride) C<br />
Carpal Tunnel Syndrome<br />
Indomethacin<br />
Disturbance In Attention (Indomethacine) C<br />
Emotional Disorder<br />
Adderall<br />
Stress<br />
(Amphetamine<br />
Suicidal Ideation<br />
Aspartate/Amphetamin<br />
Nightmare<br />
e<br />
Pollakiuria<br />
Sulfate/Dextroamphen C<br />
Weight Decreased<br />
Hypersensitivity<br />
Blood Cholesterol<br />
Increased<br />
Nausea<br />
Nervousness<br />
Pain In Extremity<br />
Chills<br />
Vomiting<br />
Diarrhoea<br />
Paranoia<br />
Post-Traumatic Stress<br />
Disorder<br />
Vision Blurred<br />
Anxiety
Headache<br />
Memory Impairment<br />
Muscle Twitching<br />
Restless Legs Syndrome<br />
03-Apr-2012 09:37 AM Page: 487
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/14/03ISR Number: 4056675-2Report Type:Direct Company Report #CTU 186682 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaise Larium PS ORAL 1/2 TABLE<br />
Treatment Noncompliance<br />
TWICE A WEEK<br />
Depression<br />
ORAL<br />
Crying<br />
Nightmare<br />
Date:02/14/03ISR Number: 4060284-9Report Type:Expedited (15-DaCompany Report #329220 Age:46 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Stress Consumer Lariam (<strong>Mefloquine</strong><br />
Intervention to Vision Blurred Hydrochloride) PS ORAL 1 PER WEEK<br />
Prevent Permanent Insomnia ORAL<br />
Impairment/Damage Depression Tramadol (Tramadol<br />
Headache Hydrochloride) C<br />
Nausea<br />
Indomethacin<br />
Nervousness (Indomethacin) C<br />
Chills<br />
Adderall<br />
Delusional Disorder,<br />
(Amphetamine<br />
Persecutory Type<br />
Aspartate/Amphetamin<br />
Post-Traumatic Stress<br />
e<br />
Disorder<br />
Sulfate/Dextroamphet C<br />
Suicidal Ideation<br />
Emotional Disorder<br />
Restless Legs Syndrome<br />
Skin Disorder<br />
Skin Papilloma<br />
Blood Cholesterol<br />
Increased<br />
Diarrhoea<br />
Paranoia<br />
Memory Impairment<br />
Pollakiuria<br />
Disturbance In Attention<br />
Anxiety<br />
Carpal Tunnel Syndrome<br />
Hyperacusis<br />
Muscle Twitching<br />
Nightmare<br />
Pain In Extremity<br />
Vomiting<br />
Weight Decreased<br />
Date:02/19/03ISR Number: 4059353-9Report Type:Expedited (15-DaCompany Report #329799 Age:16 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
PT<br />
Headache
Initial or Prolonged<br />
Disability<br />
Nausea<br />
Abdominal Distension<br />
Middle Insomnia<br />
Visual Impairment<br />
Diarrhoea<br />
Disorientation<br />
Disturbance In Attention<br />
Haemorrhage<br />
03-Apr-2012 09:37 AM Page: 488
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Influenza Like Illness<br />
Initial Insomnia<br />
Oropharyngeal Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Pyrexia Consumer Lariam (<strong>Mefloquine</strong><br />
Constipation Health Hydrochloride) 250<br />
Musculoskeletal Disorder Professional Mg PS ORAL 1 DOSE FORM<br />
Dizziness<br />
ORAL<br />
Thinking Abnormal<br />
Hepatitis A Vaccine<br />
Dry Mouth<br />
(Hepatitis A<br />
Dry Skin Vaccine) C<br />
Insomnia<br />
Amnesia<br />
Tinnitus<br />
Abdominal Pain<br />
Depression<br />
Nervous System Disorder<br />
Pruritus<br />
Skin Depigmentation<br />
Chronic Fatigue Syndrome<br />
Orthostatic Hypotension<br />
Date:02/20/03ISR Number: 4061246-8Report Type:Expedited (15-DaCompany Report #2003005986 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Drug Interaction Foreign Atorvastatin<br />
Arrhythmia Health (Atorvastatin) PS<br />
Myocardial Infarction Professional <strong>Mefloquine</strong> SS<br />
Company<br />
Representative<br />
Date:02/24/03ISR Number: 4059792-6Report Type:Direct Company Report #CTU 187215 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea <strong>Mefloquine</strong> Larium PS ORAL 1 PILL<br />
Chapped Lips<br />
ONCE A<br />
Abnormal Dreams<br />
ORAL<br />
Feeling Of Body<br />
Temperature Change<br />
Influenza Like Illness<br />
Rash<br />
Flushing<br />
Nightmare<br />
Date:02/24/03ISR Number: 4064761-6Report Type:Expedited (15-DaCompany Report #2003005986 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Arrhythmia Foreign Atorvastatin
Drug Interaction Health (Atorvastatin) PS<br />
Myocardial Infarction Professional <strong>Mefloquine</strong> SS<br />
Company<br />
Representative<br />
03-Apr-2012 09:37 AM Page: 489
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/25/03ISR Number: 4062009-XReport Type:Direct Company Report #CTU 187388 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Bipolar Disorder Lariam PS<br />
Depression<br />
Mental Impairment<br />
Abnormal Behaviour<br />
Anxiety<br />
Date:02/25/03ISR Number: 4065847-2Report Type:Expedited (15-DaCompany Report #267379 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Foreign Lariam (<strong>Mefloquine</strong><br />
Maternal Drugs Affecting Health Hydrochloride) PS ORAL 250 MG ORAL<br />
Foetus<br />
Professional<br />
Other<br />
Date:02/25/03ISR Number: 4066502-5Report Type:Expedited (15-DaCompany Report #329799 Age:16 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chronic Fatigue Syndrome Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Depression Health Hydrochloride) 250<br />
Disability Dry Skin Professional Mg PS ORAL 1 DOSE FORM<br />
Tinnitus<br />
ORAL<br />
Exercise Tolerance<br />
Hepatitis A Vaccine<br />
Decreased<br />
(Hepatitis A<br />
Abdominal Pain Vaccine) C<br />
Disorientation<br />
Dry Mouth<br />
Skin Depigmentation<br />
Nervous System Disorder<br />
Amnesia<br />
Disturbance In Attention<br />
Nausea<br />
Abdominal Distension<br />
Oropharyngeal Pain<br />
Constipation<br />
Fatigue<br />
Pyrexia<br />
Syncope<br />
Mental Impairment<br />
Middle Insomnia<br />
Diarrhoea<br />
Headache<br />
Insomnia<br />
Orthostatic Hypotension<br />
Pruritus<br />
Heart Rate Decreased<br />
Influenza Like Illness
Initial Insomnia<br />
Haemorrhage<br />
03-Apr-2012 09:37 AM Page: 490
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/26/03ISR Number: 4061186-4Report Type:Direct Company Report #CTU 187477 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam Roche PS Roche ORAL WEEK ORAL<br />
Dyspepsia<br />
Influenza Like Illness<br />
Suicidal Ideation<br />
Insomnia<br />
Anxiety<br />
Date:02/27/03ISR Number: 4067619-1Report Type:Expedited (15-DaCompany Report #329799 Age:16 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Memory Impairment Health Hydrochloride) 250<br />
Disability Nausea Professional Mg PS ORAL 1 DOSE FORM<br />
Skin Depigmentation<br />
ORAL<br />
Dry Mouth<br />
Hepatitis A Vaccine<br />
Dry Skin<br />
(Hepatitis A<br />
Insomnia Vaccine) C<br />
Orthostatic Hypotension<br />
Oropharyngeal Pain<br />
Fatigue<br />
Constipation<br />
Diarrhoea<br />
Influenza Like Illness<br />
Abdominal Pain<br />
Dizziness<br />
Syncope<br />
Tinnitus<br />
Nervous System Disorder<br />
Sleep Disorder<br />
Chronic Fatigue Syndrome<br />
Depression<br />
Abdominal Distension<br />
Disorientation<br />
Disturbance In Attention<br />
Menorrhagia<br />
Pruritus<br />
Date:03/06/03ISR Number: 4071806-6Report Type:Expedited (15-DaCompany Report #332282 Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Memory Impairment Consumer Lariam (<strong>Mefloquine</strong><br />
Viith Nerve Paralysis Hydrochloride) 240<br />
Visual Impairment Mg PS ORAL ORAL<br />
Hypoaesthesia
Date:03/11/03ISR Number: 4069555-3Report Type:Expedited (15-DaCompany Report #US-GLAXOSMITHKLINE-A0398888A Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Consumer Paxil PS Glaxosmithkline<br />
Lariam<br />
SS<br />
03-Apr-2012 09:37 AM Page: 491
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/11/03ISR Number: 4074039-2Report Type:Expedited (15-DaCompany Report #324511 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Chromaturia Foreign Lariam (<strong>Mefloquine</strong><br />
Hospitalization - Decreased Appetite Health Hydrochloride) PS<br />
Initial or Prolonged Heart Rate Increased Professional Propecia<br />
Pallor Other (Finasteride) C<br />
Asthenia<br />
Anaemia Haemolytic<br />
Autoimmune<br />
Coombs Positive<br />
Haemolytic Anaemia<br />
Headache<br />
Convulsion<br />
Fatigue<br />
Jaundice<br />
Pyrexia<br />
Splenomegaly<br />
Date:03/11/03ISR Number: 4074044-6Report Type:Expedited (15-DaCompany Report #328238 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Viith Nerve Paralysis Foreign Lariam (<strong>Mefloquine</strong><br />
Paresis Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG ORAL<br />
Other<br />
Havrix (Hepatitis A<br />
Vaccine)<br />
C<br />
Date:03/13/03ISR Number: 4074065-3Report Type:Direct Company Report #CTU 188642 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Lariam 250 Mg Roche PS Roche ORAL 250 MG WEEK<br />
Initial or Prolonged Toxicity To Various ORAL<br />
Disability<br />
Agents<br />
Suicidal Ideation<br />
Aggression<br />
Abnormal Behaviour<br />
Paranoia<br />
Hallucination<br />
Psychotic Disorder<br />
Confusional State<br />
Date:03/14/03ISR Number: 4076565-9Report Type:Expedited (15-DaCompany Report #304340 Age:15 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Crying<br />
Depression
Disability<br />
Diplopia<br />
Eye Swelling<br />
Hemiplegia<br />
Encephalitis<br />
Mental Disorder Due To A<br />
General Medical Condition<br />
Sleep Disorder<br />
Visual Field Defect<br />
03-Apr-2012 09:37 AM Page: 492
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucination<br />
Memory Impairment<br />
Tinnitus Report Source Product Role Manufacturer Route Dose Duration<br />
Bipolar I Disorder Other Lariam (<strong>Mefloquine</strong><br />
Blindness Hydrochloride) 250<br />
Paranoia Mg PS ORAL 250 MG 1 PER<br />
Intentional Self-Injury<br />
WEEK ORAL<br />
Vitreous Floaters<br />
Papilloedema<br />
Suicidal Ideation<br />
Suicide Attempt<br />
Arthralgia<br />
Cognitive Disorder<br />
Depressed Level Of<br />
Consciousness<br />
Mood Swings<br />
Nervous System Disorder<br />
Obsessive-Compulsive<br />
Disorder<br />
Date:03/14/03ISR Number: 4076957-8Report Type:Expedited (15-DaCompany Report #333205 Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Insomnia Health Hydrochloride) PS ORAL ORAL<br />
Bipolar I Disorder<br />
Professional<br />
Agitation<br />
Crying<br />
Date:03/17/03ISR Number: 4074480-8Report Type:Direct Company Report #CTU 188872 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eye Disorder Lariam Peace Corps<br />
Panic Attack Standard In PS ORAL 1 PILL WEEK<br />
Fear<br />
ORAL<br />
Muscle Twitching Doxycyline C<br />
Blepharospasm<br />
Food Allergy<br />
Hyperventilation<br />
Dyskinesia<br />
Fatigue<br />
Feeling Abnormal<br />
Grimacing<br />
Vertigo<br />
Date:03/18/03ISR Number: 4074965-4Report Type:Direct Company Report #CTU 188951 Age: Gender:Female I/FU:I
Outcome<br />
Other Serious<br />
PT<br />
Dizziness<br />
Myalgia<br />
Fatigue<br />
Hallucination<br />
Tinnitus<br />
Tremor<br />
Sleep Terror<br />
03-Apr-2012 09:37 AM Page: 493
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Suicidal Ideation<br />
Depression<br />
Nausea Report Source Product Role Manufacturer Route Dose Duration<br />
Paranoia Mefloquin Generic<br />
Nightmare For Hoffman-La<br />
Hyperhidrosis Roche PS Hoffmann- La Roche ORAL 1 TABLE PER<br />
WEEK ORAL<br />
Date:03/19/03ISR Number: 4078912-0Report Type:Expedited (15-DaCompany Report #329521 Age:60 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Somnolence Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged General Physical Health Health Hydrochloride) PS ORAL 3 DOSE FORM<br />
Deterioration Professional DAILY ORAL<br />
Renal Disorder<br />
Riamet (Coartem)<br />
Haemolytic Anaemia<br />
(Artemether/Lumefant<br />
Thrombocytopenia rine) SS ORAL 6 DOSE FORM 1<br />
PER 8 HOUR<br />
ORAL<br />
Date:03/21/03ISR Number: 4082093-7Report Type:Expedited (15-DaCompany Report #328748 Age:54 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness Foreign <strong>Mefloquine</strong><br />
Dyskinesia Literature Hydrochloride<br />
Headache Health (<strong>Mefloquine</strong><br />
Hypercholesterolaemia Professional Hydrochloride) PS 250 MG 1 PER<br />
Clonic Convulsion<br />
WEEK<br />
Bradyphrenia<br />
Shock<br />
Cerivastatin<br />
(Cerivastatin<br />
Sodium)<br />
C<br />
Date:04/01/03ISR Number: 4082569-2Report Type:Expedited (15-DaCompany Report #CA-ROCHE-334734 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Electrocardiogram Qt Lariam PS Roche UNKNOWN THREE DOSES<br />
Prolonged<br />
WERE TAKEN BY<br />
Hallucination<br />
THE PATIENT.<br />
Suicidal Ideation<br />
Chloroquine<br />
Paranoia Phosphate SS UNKNOWN<br />
Toxicity To Various<br />
Agents<br />
Coordination Abnormal<br />
Decreased Appetite
Date:04/01/03ISR Number: 4082624-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-328127 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche 30 DAY<br />
03-Apr-2012 09:37 AM Page: 494
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/02/03ISR Number: 4088082-0Report Type:Expedited (15-DaCompany Report #328127 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pregnancy Consumer Lariam (<strong>Mefloquine</strong><br />
Abortion Spontaneous Hydrochloride) PS ORAL 250 MG 1 PER<br />
Maternal Drugs Affecting<br />
WEEK ORAL<br />
Foetus<br />
Date:04/04/03ISR Number: 4085178-4Report Type:Expedited (15-DaCompany Report #GB-ROCHE-320492 Age:22 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Lariam PS Roche UNKNOWN<br />
Personality Change<br />
Loss Of Consciousness<br />
Intentional Overdose<br />
Depression<br />
Date:04/07/03ISR Number: 4088101-1Report Type:Direct Company Report #CTU 190308 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain Larium 250mg<br />
Initial or Prolonged Anxiety Tablets Roche<br />
Disability Palpitations Products PS Roche Products ORAL 1 TABLE<br />
Dizziness<br />
WEEKLY ORAL<br />
Malaise Ventolin C<br />
Chills Becotide C<br />
Date:04/14/03ISR Number: 4090106-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-305025 Age:54 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Disorder <strong>Mefloquine</strong><br />
Drug Withdrawal Syndrome Hydrochloride PS Roche ORAL 430 DAY<br />
Drug Dependence<br />
Date:04/14/03ISR Number: 4090111-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-313979 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam PS Roche ORAL<br />
Anxiety<br />
Drug Withdrawal Syndrome<br />
Mood Altered<br />
Date:04/15/03ISR Number: 4093685-3Report Type:Direct Company Report #CTU 190928 Age:24 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Larium PS WEEKLY<br />
Hospitalization - Depression<br />
Initial or Prolonged<br />
Disability<br />
03-Apr-2012 09:37 AM Page: 495
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/18/03ISR Number: 4092995-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-336048 Age:6 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Lariam PS Roche ORAL<br />
Initial or Prolonged Gastrointestinal<br />
Infection<br />
Weight Decreased<br />
Diarrhoea<br />
Date:04/22/03ISR Number: 4094866-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-333205 Age:50 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Lariam PS Roche ORAL 9 DAY<br />
Initial or Prolonged Agitation<br />
Bipolar I Disorder<br />
Hallucination<br />
Crying<br />
Date:04/28/03ISR Number: 4098493-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-329220 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Lariam PS Roche ORAL<br />
Post-Traumatic Stress Tramadol C ORAL<br />
Disorder Indomethacin C ORAL<br />
Tenosynovitis Adderall C ORAL<br />
Insomnia<br />
Surgical Procedure<br />
Repeated<br />
Paranoia<br />
Disturbance In Attention<br />
Memory Impairment<br />
Vomiting<br />
Muscle Twitching<br />
Blood Cholesterol<br />
Increased<br />
Suicidal Ideation<br />
Nausea<br />
Restless Legs Syndrome<br />
Libido Decreased<br />
Skin Papilloma<br />
Carpal Tunnel Syndrome<br />
Nervousness<br />
Chills<br />
Nightmare<br />
Hyperacusis<br />
Headache<br />
Pollakiuria<br />
Migraine<br />
Pain In Extremity
Loss Of Employment<br />
Fatigue<br />
Weight Decreased<br />
Skin Discolouration<br />
Visual Impairment<br />
Stress<br />
Depression<br />
Vision Blurred<br />
03-Apr-2012 09:37 AM Page: 496
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/01/03ISR Number: 4105493-5Report Type:Direct Company Report #CTU 192075 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression <strong>Mefloquine</strong> 250 Mg<br />
Palpitations<br />
Geneva Pharm Tech<br />
Supraventricular Corporation PS Geneva Pharm Tech<br />
Extrasystoles Corporation PARENTERAL 250 MG Q<br />
Mood Altered<br />
WEEK<br />
PARENTERAL<br />
Date:05/02/03ISR Number: 4102404-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-336087 Age:71 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anosmia Lariam PS Roche ORAL<br />
Ageusia<br />
Date:05/06/03ISR Number: 4106524-9Report Type:Direct Company Report #CTU 192353 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Sinus Disorder <strong>Mefloquine</strong> PS<br />
Dysphagia<br />
Oropharyngeal Pain<br />
Date:05/06/03ISR Number: 4106525-0Report Type:Direct Company Report #CTU 192354 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression <strong>Mefloquine</strong> 250mg PS ORAL 250 MG PO<br />
Loss Of Consciousness<br />
WEEKLY<br />
Affect Lability<br />
Date:05/08/03ISR Number: 4105846-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-336087 Age:71 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anosmia Lariam PS Roche ORAL<br />
Ageusia<br />
Date:05/12/03ISR Number: 4107655-XReport Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Lariam PS Roche UNKNOWN<br />
Paranoia<br />
Hallucination, Auditory<br />
Condition Aggravated
Hallucination, Olfactory<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 497
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/12/03ISR Number: 4110899-4Report Type:Direct Company Report #CTU 192740 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety Lariam 250 Mg Per<br />
Hospitalization - Fatigue Week Roche Usa PS Roche Usa ORAL 250 MG PER<br />
Initial or Prolonged Suicidal Ideation WEEK ORAL<br />
Disability<br />
Other Serious<br />
Night Sweats<br />
Nightmare<br />
Depression<br />
Hallucination<br />
Date:05/19/03ISR Number: 4111056-8Report Type:Expedited (15-DaCompany Report #SE-ROCHE-328238 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Viith Nerve Paralysis Lariam PS Roche ORAL 1 TABLET ONLY<br />
TAKEN. 1 DAY<br />
Havrix C INTRAMUSCULAR<br />
Date:05/19/03ISR Number: 4111080-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-337777 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Tachycardia Lariam PS Roche ORAL<br />
Insomnia<br />
Drowning<br />
Anxiety<br />
Date:05/19/03ISR Number: 4114514-5Report Type:Direct Company Report #CTU 193224 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Lariam <strong>Mefloquine</strong><br />
Required Panic Attack Hydrochloride 250mg<br />
Intervention to Anxiety Tabs PS SEE IMAGE<br />
Prevent Permanent Paranoia Omeprazole Magnesium C<br />
Impairment/Damage Hallucination Losec C<br />
Decreased Appetite<br />
Emotional Disorder<br />
Date:05/21/03ISR Number: 4113122-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-337388 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hepatic Enzyme Increased Consumer Lariam PS Roche ORAL 2 DAY<br />
Hospitalization - Psychotic Disorder Lariam SS Roche ORAL 1 DAY<br />
Initial or Prolonged Clont SS ORAL 11 DAY<br />
Lariam SS Roche ORAL 1 DAY
Date:05/28/03ISR Number: 4117382-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-329799 Age:16 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Psychotic Disorder<br />
Abdominal Pain<br />
Cognitive Disorder<br />
03-Apr-2012 09:37 AM Page: 498
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Diarrhoea<br />
Dry Mouth Report Source Product Role Manufacturer Route Dose Duration<br />
Pruritus Lariam PS Roche ORAL 1 TABLET<br />
Mania<br />
WEEKLY<br />
Influenza Like Illness Hepatitis A Vaccine C<br />
Nausea<br />
Constipation<br />
Headache<br />
Disorientation<br />
Initial Insomnia<br />
Nervous System Disorder<br />
Chronic Fatigue Syndrome<br />
Menstruation Irregular<br />
Dry Skin<br />
Disturbance In Attention<br />
Orthostatic Hypotension<br />
Abdominal Distension<br />
Amnesia<br />
Heart Rate Decreased<br />
Tinnitus<br />
Skin Depigmentation<br />
Exercise Tolerance<br />
Decreased<br />
Date:05/30/03ISR Number: 4187487-7Report Type:Periodic Company Report #255796 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nerve Injury Consumer Lariam (<strong>Mefloquine</strong><br />
Dizziness Health Hydrochloride) 250<br />
Weight Decreased Professional Mg PS ORAL 1 DOSE FORM 1<br />
Nausea<br />
PER WEEK ORAL<br />
Anxiety<br />
Balance Disorder<br />
Diarrhoea<br />
Panic Attack<br />
Vertigo<br />
Muscular Weakness<br />
Date:05/30/03ISR Number: 4187488-9Report Type:Periodic Company Report #312535 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Pressure Abnormal Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Blood Pressure Increased Hydrochloride) 250<br />
Weight Decreased Mg PS ORAL 250 MG 1 PER<br />
Decreased Appetite<br />
WEEK ORAL<br />
Depression<br />
Trandate (Labetalol<br />
Tachycardia Hydrochloride) C<br />
Anxiety
Suicidal Ideation<br />
Bradycardia<br />
Syncope<br />
03-Apr-2012 09:37 AM Page: 499
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/30/03ISR Number: 4187489-0Report Type:Periodic Company Report #317591 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:05/30/03ISR Number: 4187490-7Report Type:Periodic Company Report #322558 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Delusion Other Lariam (<strong>Mefloquine</strong><br />
Disability Depression Hydrochloride) 250<br />
Anxiety Mg PS ORAL ORAL<br />
Delusional Disorder,<br />
Persecutory Type<br />
Completed Suicide<br />
Mood Swings<br />
Confusional State<br />
Headache<br />
Date:05/30/03ISR Number: 4187491-9Report Type:Periodic Company Report #322574 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Feeling Cold Health Lariam (<strong>Mefloquine</strong><br />
Intervention to Confusional State Professional Hydrochloride) 250<br />
Prevent Permanent Nausea Mg PS ORAL 250 MG 1 PER<br />
Impairment/Damage Diarrhoea DAY ORAL<br />
Medication Error<br />
Date:05/30/03ISR Number: 4187492-0Report Type:Periodic Company Report #324697 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Consumer Lariam (<strong>Mefloquine</strong><br />
Weight Decreased Hydrochloride) 240<br />
Suicidal Ideation Mg PS ORAL 240 MG 1 PER<br />
1 WEEK ORAL<br />
Date:05/30/03ISR Number: 4187493-2Report Type:Periodic Company Report #325385 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hallucination Hydrochloride) PS ORAL ORAL<br />
Depression
Date:05/30/03ISR Number: 4187494-4Report Type:Periodic Company Report #327591 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Asthenia Literature <strong>Mefloquine</strong><br />
Health<br />
Hydrochloride<br />
Professional<br />
(<strong>Mefloquine</strong><br />
03-Apr-2012 09:37 AM Page: 500
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hydrochloride)<br />
Pyridostigmine<br />
(Pyridostigmine<br />
Bromide)<br />
PS<br />
C<br />
Date:05/30/03ISR Number: 4187495-6Report Type:Periodic Company Report #327601 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Asthenia Literature <strong>Mefloquine</strong><br />
Health<br />
Hydrochloride<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS<br />
Date:05/30/03ISR Number: 4187496-8Report Type:Periodic Company Report #329345 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS<br />
Date:05/30/03ISR Number: 4188986-4Report Type:Periodic Company Report #329978 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Eye Irritation Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hallucination Health Hydrochloride) PS ORAL 1 DOSE FORM<br />
Heart Rate Increased Professional 1 PER WEEK<br />
Insomnia<br />
ORAL<br />
Nervousness<br />
Ultran<br />
Nightmare (Phenaglycodol) C<br />
Dizziness<br />
Lorazepam<br />
Dry Mouth (Lorazepam) C<br />
Dyspnoea<br />
Oxycontin<br />
Headache (Oxycodone) C<br />
Psychotic Disorder<br />
Unspecified<br />
Tremor<br />
Immunizations<br />
Claustrophobia (Immunisation) C<br />
Panic Attack<br />
Effexor (Venlafaxine<br />
Depression Hydrochloride) C<br />
Agitation<br />
Date:05/30/03ISR Number: 4188987-6Report Type:Periodic Company Report #332290 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Self Esteem Decreased Consumer Lariam (<strong>Mefloquine</strong><br />
Anxiety Hydrochloride) 250<br />
Insomnia Mg PS ORAL ORAL 152 DAY<br />
Dyspnoea
Chest Pain<br />
Mood Swings<br />
Suicide Attempt<br />
Nervousness<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 501
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/30/03ISR Number: 4188988-8Report Type:Periodic Company Report #332637 Age:2 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Petit Mal Epilepsy Health Lariam (<strong>Mefloquine</strong><br />
Convulsion Professional Hydrochloride) 250<br />
Mg PS ORAL 0.25 DOSE<br />
FORM 1 PER<br />
WEEK ORAL<br />
Date:05/30/03ISR Number: 4188989-XReport Type:Periodic Company Report #334655 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Acute Psychosis Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Professional Hydrochloride) PS<br />
Date:05/30/03ISR Number: 4188990-6Report Type:Periodic Company Report #335354 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaria Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Abnormal Dreams Hydrochloride) 250<br />
Hypotension Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/02/03ISR Number: 4120310-5Report Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Health Lariam PS Roche UNKNOWN 3 MON<br />
Hallucination, Olfactory Professional Antidepressants Nos C<br />
Flashback<br />
Paranoia<br />
Hallucination, Auditory<br />
Date:06/03/03ISR Number: 4121733-0Report Type:Direct Company Report #CTU 194609 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nausea <strong>Mefloquine</strong> 250 Mg<br />
Hospitalization - Aggression Geneva PS Geneva 1 TABLET PER<br />
Initial or Prolonged Paranoia WEEK<br />
Disability Headache BEGINNING 1<br />
Required Anxiety WK B/F<br />
Intervention to Hallucination DEPARTURE AND<br />
Prevent Permanent<br />
Impairment/Damage<br />
Oropharyngeal Pain<br />
Tinnitus<br />
Fatigue<br />
Syncope
Skin Disorder<br />
Confusional State<br />
Dizziness<br />
Mood Altered<br />
Orthostatic Hypotension<br />
Depression<br />
Psychotic Disorder<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 502
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/03/03ISR Number: 4122087-6Report Type:Expedited (15-DaCompany Report #DE-GLAXOSMITHKLINE-D0041072A Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Intentional Overdose Halfan PS Glaxosmithkline ORAL 3000MG Single<br />
Initial or Prolonged Alcohol Poisoning dose 1 DAY<br />
Suicide Attempt Alcohol SS ORAL 1 DAY<br />
Vomiting Lariam SS ORAL 1500MG Single<br />
dose 1 DAY<br />
Date:06/04/03ISR Number: 4122521-1Report Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination, Auditory Lariam PS Roche UNKNOWN 3 MON<br />
Hallucination, Olfactory Antidepressants Nos C<br />
Paranoia<br />
Flashback<br />
Homicide<br />
Date:06/04/03ISR Number: 4122561-2Report Type:Expedited (15-DaCompany Report #FR-GLAXOSMITHKLINE-B0299754A Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Condition Aggravated Deroxat PS Glaxosmithkline ORAL 20MG Twice<br />
Initial or Prolonged Overdose per day<br />
Anxiety Lariam SS ORAL 250MG Unknown<br />
Fear Prozac SS ORAL 36 DAY<br />
Hallucination Lysanxia SS ORAL 36 DAY<br />
Insomnia Xanax SS ORAL<br />
Suicide Attempt<br />
Depression<br />
Nightmare<br />
Date:06/04/03ISR Number: 4123059-8Report Type:Direct Company Report #CTU 194940 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Larium Roche PS Roche ORAL 250MG WEEKLY<br />
Initial or Prolonged Depression ORAL<br />
Required<br />
Disturbance In Attention<br />
Intervention to<br />
Memory Impairment<br />
Prevent Permanent<br />
Impairment/Damage<br />
Night Sweats<br />
Suicidal Ideation<br />
Insomnia<br />
Date:06/05/03ISR Number: 4123097-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-339395 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Atrioventricular Block Lariam PS Roche ORAL<br />
Complete Beta Blocker Nos C ORAL<br />
03-Apr-2012 09:37 AM Page: 503
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/05/03ISR Number: 4123733-3Report Type:Direct Company Report #CTU 195063 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Disorder With Larium 250mg Roche PS Roche ORAL 250MG 1 PER<br />
Initial or Prolonged Agoraphobia WEEK ORAL<br />
Depression<br />
Date:06/06/03ISR Number: 4123954-XReport Type:Expedited (15-DaCompany Report #CA-ROCHE-339135 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Kawasaki'S Disease Lariam PS Roche ORAL<br />
Cholestasis<br />
Date:06/10/03ISR Number: 4125466-6Report Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination, Auditory Health Lariam PS Roche ORAL 3 MON<br />
Paranoia Professional Flagyl C ORAL 8 DAY<br />
Homicide Unknown Medication C ORAL<br />
Hallucination, Olfactory Antidepressants Nos C<br />
Flashback<br />
Date:06/10/03ISR Number: 4125480-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-339481 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Meningism Consumer Lariam PS Roche ORAL 53 DAY<br />
Initial or Prolonged Hepatitis Ibuprofene SS ORAL 7 DAY<br />
Doliprane SS ORAL 7 DAY<br />
Adepal C RECEIVED<br />
TREATMENT FOR<br />
SEVERAL<br />
MONTHS<br />
Date:06/10/03ISR Number: 4126187-6Report Type:Direct Company Report #CTU 195411 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Reaction Larium Laroche PS Laroche ORAL 1 TABLE<br />
Anxiety<br />
WEEKLY ORAL<br />
Paraesthesia<br />
Depression<br />
Dizziness<br />
Distractibility<br />
Neurotoxicity<br />
Fatigue
Date:06/11/03ISR Number: 4126537-0Report Type:Expedited (15-DaCompany Report #SE-ROCHE-328238 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Viith Nerve Paralysis Consumer Lariam PS Roche ORAL 1 TABLET ONLY<br />
TAKEN. 1 DAY<br />
Havrix SS INTRAMUSCULAR 1 DAY<br />
03-Apr-2012 09:37 AM Page: 504
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/13/03ISR Number: 4128467-7Report Type:Direct Company Report #CTU 195725 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Depression Lariam 250 Mg<br />
Life-Threatening Asthenia Hoffman-Laroche,<br />
Disability Sleep Apnoea Syndrome Inc. PS Hoffman-Laroche,<br />
Other Serious Weight Increased Inc. ORAL 250 MG OVER 5<br />
Required Panic Disorder ORAL<br />
Intervention to Completed Suicide Phen/Fen SS ORAL OVER 9 ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:06/23/03ISR Number: 4134248-0Report Type:Direct Company Report #CTU 196344 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Feeling Abnormal Lariam Roche PS Roche ORAL 1 TABLE ONCE<br />
Hospitalization - Mental Disorder A WEEK ORAL<br />
Initial or Prolonged Activities Of Daily<br />
Disability<br />
Living Impaired<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:06/24/03ISR Number: 4133560-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-336087 Age:71 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Ageusia Lariam PS Roche ORAL<br />
Viral Infection<br />
Anosmia<br />
Date:06/26/03ISR Number: 4135803-4Report Type:Expedited (15-DaCompany Report #DE-GLAXOSMITHKLINE-D0041072A Age:39 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicide Attempt Health Halfan PS Glaxosmithkline ORAL 3000MG Single<br />
Initial or Prolonged Alcohol Poisoning Professional dose 1 DAY<br />
Intentional Overdose Alcohol SS ORAL 1 DAY<br />
Vomiting Lariam SS ORAL 1500MG Single<br />
dose 1 DAY<br />
Date:06/27/03ISR Number: 4136538-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-329799 Age:16 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Fatigue<br />
Tinnitus
Disability<br />
Diarrhoea<br />
Dry Skin<br />
Skin Depigmentation<br />
Insomnia<br />
Abdominal Distension<br />
Nausea<br />
Laboratory Test Abnormal<br />
Disturbance In Attention<br />
03-Apr-2012 09:37 AM Page: 505
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorientation<br />
Malaise<br />
Haemorrhage Report Source Product Role Manufacturer Route Dose Duration<br />
Psychotic Disorder Lariam PS Roche ORAL 1 TABLET<br />
Depression<br />
WEEKLY<br />
Headache Hepatitis A Vaccine C<br />
Red Blood Cell<br />
Sedimentation Rate<br />
Increased<br />
Amnesia<br />
Exercise Tolerance<br />
Decreased<br />
Mania<br />
C-Reactive Protein<br />
Increased<br />
Immunology Test Abnormal<br />
Bipolar Disorder<br />
Constipation<br />
Abdominal Pain<br />
Nervous System Disorder<br />
Influenza Like Illness<br />
Syncope<br />
Heart Rate Decreased<br />
Brain Scan Normal<br />
Orthostatic Hypotension<br />
Dry Mouth<br />
Pyrexia<br />
Initial Insomnia<br />
Date:06/27/03ISR Number: 4137636-1Report Type:Direct Company Report #CTU 196820 Age:66 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Speech Disorder Lariam 250 Mg PS ORAL 1 TABLET BY<br />
Initial or Prolonged Ear Pain MOUTH WEEKLY<br />
Disability<br />
Weight Decreased<br />
Feeling Abnormal<br />
Diplopia<br />
Cerebrovascular Accident<br />
Balance Disorder<br />
Headache<br />
Migraine<br />
Nausea<br />
Pain In Extremity<br />
Vertigo<br />
Vision Blurred<br />
Lupus-Like Syndrome<br />
Decreased Appetite<br />
Asthenia<br />
Confusional State<br />
Gait Disturbance<br />
Dizziness
Eye Disorder<br />
Vomiting<br />
Dysphemia<br />
Arterial Occlusive<br />
Disease<br />
Arthralgia<br />
03-Apr-2012 09:37 AM Page: 506
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/01/03ISR Number: 4138763-5Report Type:Expedited (15-DaCompany Report #KE-ROCHE-340706 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Prothrombin Time <strong>Mefloquine</strong><br />
Initial or Prolonged Prolonged Hydrochloride PS Roche UNKNOWN DOSE REPORTED<br />
Abdominal Distension<br />
AS 250MG PER<br />
Ascites<br />
WEEK.<br />
Blood Pressure Decreased Carvedilol C<br />
Drug Interaction Betoptic Eye Drops C<br />
Tachycardia Captoril C<br />
Infectious Peritonitis Digoxin C<br />
Cardiomegaly Warfarin I UNKNOWN<br />
Abdominal Rebound<br />
Tenderness<br />
Anaemia<br />
Gallop Rhythm Present<br />
Date:07/01/03ISR Number: 4138771-4Report Type:Expedited (15-DaCompany Report #KE-ROCHE-340709 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Prothrombin Time <strong>Mefloquine</strong><br />
Initial or Prolonged Prolonged Hydrochloride PS Roche UNKNOWN DOSE REPORTED<br />
Oedema Peripheral<br />
AS 250MG<br />
Injury<br />
WEEKLY.<br />
Muscle Twitching Sotalol C<br />
Confusional State Frusemide C REPORTED AS<br />
Hypoglycaemia<br />
GIVEN<br />
Aggression<br />
OCASIONALLY.<br />
Haemorrhage Digoxin C<br />
Blood Glucose Fluctuation Glibenclamide I Roche UNKNOWN<br />
Cellulitis Chlorpropamide I UNKNOWN<br />
Drug Interaction<br />
Dermatitis Exfoliative<br />
Blister<br />
Retrograde Amnesia<br />
Date:07/03/03ISR Number: 4140824-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-339743 Age:41 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Platelet Disorder Lariam PS Roche ORAL THE PATIENT<br />
Contusion<br />
WAS REPORTED<br />
Menorrhagia<br />
TO HAVE<br />
RECEIVED<br />
MEFLOQUINE<br />
Lariam SS Roche ORAL<br />
Lariam SS Roche ORAL
Date:07/07/03ISR Number: 4142181-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-341198 Age:75 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability C-Reactive Protein Lariam PS Roche ORAL<br />
Increased<br />
Arthropathy<br />
Muscle Disorder<br />
03-Apr-2012 09:37 AM Page: 507
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/07/03ISR Number: 4142198-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-341603 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Retinitis Pigmentosa Lariam PS Roche UNKNOWN<br />
Date:07/07/03ISR Number: 4142200-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-341571 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Groin Pain Lariam PS Roche UNKNOWN 5 WK<br />
Quadriplegia<br />
Date:07/07/03ISR Number: 4142985-7Report Type:Direct Company Report #CTU 197286 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion <strong>Mefloquine</strong> 250<br />
Initial or Prolonged Road Traffic Accident Lariam PS ORAL 1 WEEK ORAL<br />
Lethargy<br />
Depression<br />
Date:07/07/03ISR Number: 4144312-8Report Type:Expedited (15-DaCompany Report #FRWYE193930JUN03 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatitis Health Adepal<br />
Initial or Prolonged Cytolytic Hepatitis Professional (Levonorgestrel/Ethi<br />
Antinuclear Antibody Other nyl Estradiol,<br />
Positive Tablet, O) PS ORAL ORAL/SEVERAL<br />
MONTHS<br />
Lariam (<strong>Mefloquine</strong>,<br />
0) SS ORAL 250 MG 1X PER<br />
1 WK, ORAL 53 DAY<br />
Doliprane<br />
(Paracetamol)<br />
C<br />
Ibuprofen<br />
(Ibuprofen)<br />
C<br />
Date:07/10/03ISR Number: 4144762-XReport Type:Direct Company Report #CTU 197662 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Viith Nerve Paralysis <strong>Mefloquine</strong> PS ORAL 250MG ORAL<br />
Hepatitis A Vaccine C<br />
Date:07/10/03ISR Number: 4147062-7Report Type:Expedited (15-DaCompany Report #FRWYE193930JUN03 Age:42 YR Gender:Female I/FU:F
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Diarrhoea<br />
Myalgia<br />
Arthralgia<br />
Hepatitis<br />
Headache<br />
Cytolytic Hepatitis<br />
Antinuclear Antibody<br />
03-Apr-2012 09:37 AM Page: 508
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Positive<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Health<br />
Adepal<br />
Professional<br />
(Levonorgestrel/Ethi<br />
Other<br />
nyl Estradiol,<br />
Tablet, 0) PS ORAL<br />
Lariam (<strong>Mefloquine</strong>,<br />
, 0) SS ORAL 250 MG 1X PER<br />
1 WK 53 DAY<br />
Date:07/11/03ISR Number: 4145359-8Report Type:Expedited (15-DaCompany Report #KE-ROCHE-340706 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Peritoneal Haemorrhage <strong>Mefloquine</strong><br />
Initial or Prolonged Ascites Hydrochloride PS Roche UNKNOWN DOSE REPORTED<br />
Infectious Peritonitis<br />
AS 250MG PER<br />
Blood Pressure Decreased<br />
WEEK.<br />
Prothrombin Time Carvedilol C<br />
Prolonged Betoptic Eye Drops C<br />
Cardiomegaly Captoril C<br />
Anaemia Digoxin C<br />
Gallop Rhythm Present Warfarin I UNKNOWN<br />
Gastrointestinal<br />
Haemorrhage<br />
Drug Interaction<br />
Tachycardia<br />
Melaena<br />
Date:07/14/03ISR Number: 4146493-9Report Type:Direct Company Report #CTU 197817 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delusion Larium PS<br />
Memory Impairment<br />
Social Avoidant Behaviour<br />
Sensory Loss<br />
Panic Attack<br />
Visual Impairment<br />
Speech Disorder<br />
Crying<br />
Depression<br />
Poisoning<br />
Hearing Impaired<br />
Flat Affect<br />
Emotional Disorder<br />
Bipolar Disorder
Date:07/14/03ISR Number: 4148525-0Report Type:Expedited (15-DaCompany Report #200316652GDDC Age:63 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Drug Interaction<br />
Aggression<br />
Hyperhidrosis<br />
Confusional State<br />
Depressed Level Of<br />
03-Apr-2012 09:37 AM Page: 509
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Consciousness<br />
Hunger<br />
Hypoglycaemia<br />
Cellulitis Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Foreign Glibenclamide PS<br />
Haemorrhage Literature <strong>Mefloquine</strong><br />
Irritability Health Hydrochloride SS 250 MG<br />
Oedema Peripheral Professional Frusemide C<br />
Muscle Twitching Other Sotalol C<br />
Prothrombin Time Digoxin C<br />
Prolonged<br />
Mental Disorder<br />
Blister<br />
Retrograde Amnesia<br />
Blood Glucose Fluctuation<br />
Skin Graft<br />
Injury<br />
Dermatitis Exfoliative<br />
Date:07/15/03ISR Number: 4147230-4Report Type:Direct Company Report #CTU 197900 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Chest Pain <strong>Mefloquine</strong> PS ORAL 250MG Q7D<br />
Dyspnoea<br />
ORAL<br />
Hypoaesthesia<br />
Conversion Disorder<br />
Speech Disorder<br />
Anxiety<br />
Costochondritis<br />
Date:07/15/03ISR Number: 4147241-9Report Type:Direct Company Report #CTU 197894 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Lariam PS ORAL 1 WEEKLY ORAL<br />
Initial or Prolonged Eyelid Oedema<br />
Local Swelling<br />
Chest Discomfort<br />
Nausea<br />
Feeling Abnormal<br />
Vision Blurred<br />
Abdominal Discomfort<br />
Abdominal Pain<br />
Oedema Peripheral<br />
Decreased Appetite<br />
Rash<br />
Swollen Tongue<br />
Swelling Face
Date:07/15/03ISR Number: 4147253-5Report Type:Expedited (15-DaCompany Report #KE-ROCHE-308528 Age:66 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Drug Interaction<br />
Prothrombin Time<br />
Prolonged<br />
Gallop Rhythm Present<br />
Abdominal Rebound<br />
03-Apr-2012 09:37 AM Page: 510
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tenderness<br />
Blood Pressure Decreased<br />
Gastrointestinal<br />
Haemorrhage Report Source Product Role Manufacturer Route Dose Duration<br />
Ejection Fraction<br />
<strong>Mefloquine</strong><br />
Decreased Hydrochloride PS Roche UNKNOWN<br />
Infectious Peritonitis Carvedilol C<br />
Anaemia Betoptic Eye Drops C<br />
Congestive Cardiomyopathy Captopril C<br />
Peritoneal Haemorrhage Digoxin C<br />
Tachycardia Warfarin I UNKNOWN<br />
Gastrointestinal Sounds<br />
Abnormal<br />
Abdominal Distension<br />
Ascites<br />
Cardiomegaly<br />
Ileus<br />
Date:07/16/03ISR Number: 4148165-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-339481 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Antinuclear Antibody Consumer Lariam PS Roche ORAL 53 DAY<br />
Initial or Prolonged Positive Doliprane C ORAL 7 DAY<br />
Hepatitis Adepal C RECEIVED<br />
TREATMENT FOR<br />
SEVERAL<br />
MONTHS<br />
Ibuprofene C ORAL 7 DAY<br />
Date:07/17/03ISR Number: 4148786-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-342147 Age:9 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebrovascular Accident Lariam PS Roche UNKNOWN REPORTED AS<br />
Initial or Prolonged<br />
THREE-QUARTER<br />
S OF A<br />
TABLET.<br />
Date:07/21/03ISR Number: 4150523-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-342003 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Pregnancy Lariam PS Roche UNKNOWN 40 DAY<br />
Maternal Drugs Affecting Stamaril Unidose SS UNKNOWN 1 DAY<br />
Foetus Librax SS Roche UNKNOWN TAKEN<br />
Abortion Induced<br />
OCCASIONALLY.<br />
Foetal Disorder<br />
Congenital Hydrocephalus
Date:07/22/03ISR Number: 4151451-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-336087 Age:71 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Cardiac Failure<br />
Hyperlipidaemia<br />
Viral Infection<br />
Anosmia<br />
03-Apr-2012 09:37 AM Page: 511
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Ageusia<br />
Hypothyroidism<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Date:07/22/03ISR Number: 4151460-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-342003 Age:29 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Foetal Disorder Lariam PS Roche UNKNOWN 40 DAY<br />
Lung Disorder Stamaril Unidose SS UNKNOWN 1 DAY<br />
Maternal Drugs Affecting Librax SS Roche UNKNOWN TAKEN<br />
Foetus<br />
OCCASIONALLY.<br />
Nervous System Disorder<br />
Abortion Induced<br />
Congenital Hydrocephalus<br />
Metrorrhagia<br />
Pregnancy<br />
Dysplasia<br />
Date:07/23/03ISR Number: 4152252-3Report Type:Direct Company Report #CTU 198498 Age:62 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Impaired Work Ability <strong>Mefloquine</strong> 250 Mg<br />
Required Confusional State Roche PS Roche 1@ WEEK 10<br />
Intervention to Arthritis WEEKS<br />
Prevent Permanent<br />
Impairment/Damage<br />
Anxiety<br />
Night Sweats<br />
Hyperhidrosis<br />
Insomnia<br />
Depression<br />
Hallucination, Auditory<br />
Impaired Driving Ability<br />
Date:07/24/03ISR Number: 4153323-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-339481 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatitis Lariam PS Roche ORAL 53 DAY<br />
Initial or Prolonged Adepal SS ORAL RECEIVED<br />
TREATMENT FOR<br />
SEVERAL<br />
MONTHS<br />
Ibuprofene C ORAL 7 DAY<br />
Doliprane C ORAL 7 DAY<br />
Date:07/25/03ISR Number: 4154030-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-341571 Age:30 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paralysis Lariam PS Roche ORAL 36 DAY<br />
Groin Pain<br />
03-Apr-2012 09:37 AM Page: 512
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/25/03ISR Number: 4154884-5Report Type:Direct Company Report #CTU 198688 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Restlessness Lariam = <strong>Mefloquine</strong><br />
Initial or Prolonged Rash 250 Mg PS 250MG 7 WEEKS<br />
Heart Rate Increased<br />
Dizziness<br />
Anxiety<br />
Confusional State<br />
Date:07/28/03ISR Number: 4155347-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-324511 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Headache Lariam PS Roche ORAL THERAPY<br />
Hospitalization - Convulsion DURATION<br />
Initial or Prolonged Pyrexia REPORTED AS 5<br />
Heart Rate Increased<br />
MONTHS.<br />
Splenomegaly Propecia C<br />
Jaundice<br />
Circulatory Collapse<br />
Anaemia Haemolytic<br />
Autoimmune<br />
Date:07/28/03ISR Number: 4155373-4Report Type:Expedited (15-DaCompany Report #GB-ROCHE-342738 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pain In Extremity Lariam PS Roche ORAL<br />
Headache<br />
Date:07/30/03ISR Number: 4158822-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-329796 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Flushing Lariam PS Roche ORAL<br />
Initial or Prolonged Emotional Distress<br />
Disability<br />
Facial Pain<br />
Burning Sensation<br />
Tenderness<br />
Erythema<br />
Date:07/31/03ISR Number: 4158590-2Report Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Flashback Lariam PS Roche ORAL 3 MON<br />
Paranoia Flagyl C ORAL 8 DAY<br />
Homicide Unknown Medication C ORAL
Hallucination, Auditory Antidepressants Nos C<br />
Hallucination, Olfactory<br />
03-Apr-2012 09:37 AM Page: 513
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/31/03ISR Number: 4158597-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-342869 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lymphocyte Morphology Lariam PS Roche ORAL 198 DAY<br />
Initial or Prolonged Abnormal<br />
Dermatitis Exfoliative<br />
Pseudolymphoma<br />
Lymphadenopathy<br />
Date:07/31/03ISR Number: 4158599-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-343049 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatic Enzyme Increased Lariam PS Roche ORAL REPORTED AS 3<br />
Initial or Prolonged Diarrhoea TABLETS EVERY<br />
Hepatitis Cholestatic<br />
8 HOURS, THEN<br />
2 EVERY 8<br />
HOURS, THEN 1 3 DAY<br />
Di Antalvic C ORAL 1 DAY<br />
Perfalgan C INTRAVENOUS 2 DAY<br />
Date:08/04/03ISR Number: 4160446-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam PS Roche ORAL<br />
Hallucination, Visual<br />
Vestibular Disorder<br />
Headache<br />
Hypertension<br />
Confusional State<br />
Depression<br />
Memory Impairment<br />
Dyspnoea<br />
Skin Disorder<br />
Gastrointestinal Disorder<br />
Chronic Fatigue Syndrome<br />
Mitral Valve Prolapse<br />
Date:08/07/03ISR Number: 4163376-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-343702 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Spasms Lariam PS Roche ORAL<br />
Date:08/11/03ISR Number: 4166680-3Report Type:Direct Company Report #CTU 199689 Age:31 YR Gender:Male I/FU:I<br />
Outcome<br />
PT
Other Serious<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Escherichia Infection<br />
Anxiety<br />
Disturbance In Attention<br />
Paranoia<br />
Thinking Abnormal<br />
Diarrhoea<br />
Insomnia<br />
Vision Blurred<br />
03-Apr-2012 09:37 AM Page: 514
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Toxicity To Various<br />
Agents Report Source Product Role Manufacturer Route Dose Duration<br />
Headache Lariam 250 Mg<br />
Vomiting Roche Labs Inc. Nj PS Roche Labs Inc. Nj ORAL 1 PILL 1 A<br />
Abdominal Pain Upper<br />
WEEK ORAL<br />
Depression<br />
Date:08/13/03ISR Number: 4166562-7Report Type:Expedited (15-DaCompany Report #AU-ROCHE-344285 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arrhythmia Lariam PS Roche ORAL 22 DAY<br />
Initial or Prolonged Supraventricular<br />
Sinus Bradycardia<br />
Date:08/13/03ISR Number: 4166567-6Report Type:Expedited (15-DaCompany Report #DE-ROCHE-341571 Age:30 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Lariam PS Roche ORAL 36 DAY<br />
Paraesthesia<br />
Groin Pain<br />
Paralysis<br />
Date:08/15/03ISR Number: 4169049-0Report Type:Direct Company Report #CTU 200038 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety <strong>Mefloquine</strong> PS ORAL 250 MG Q7D<br />
Costochondritis<br />
ORAL<br />
Dyspnoea<br />
Speech Disorder<br />
Hypoaesthesia<br />
Conversion Disorder<br />
Chest Pain<br />
Date:08/15/03ISR Number: 4169053-2Report Type:Direct Company Report #CTU 200041 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Oedema Peripheral Lariam PS ORAL 1 WEEKLY ORAL<br />
Initial or Prolonged Abdominal Discomfort<br />
Diarrhoea<br />
Swollen Tongue<br />
Eyelid Oedema<br />
Feeling Abnormal<br />
Nausea<br />
Swelling Face
Rash<br />
Local Swelling<br />
Pain<br />
Decreased Appetite<br />
Chest Discomfort<br />
Vision Blurred<br />
03-Apr-2012 09:37 AM Page: 515
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/18/03ISR Number: 4171983-2Report Type:Direct Company Report #CTU 200182 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Larium - Mefloquin<br />
Mental Disorder Roche PS Roche ORAL 250 MG, ORAL<br />
Nervousness<br />
WEEKLY<br />
Abnormal Behaviour<br />
Disturbance In Attention<br />
Swollen Tongue<br />
Irritability<br />
Date:08/19/03ISR Number: 4169987-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-339743 Age:41 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haemorrhage Lariam PS Roche ORAL THE PATIENT<br />
Von Willebrand'S Disease<br />
WAS REPORTED<br />
TO HAVE<br />
RECEIVED<br />
MEFLOQUINE<br />
Lariam SS Roche ORAL<br />
Lariam SS Roche ORAL<br />
Date:08/25/03ISR Number: 4177287-6Report Type:Expedited (15-DaCompany Report #2003034182 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Ineffective Foreign Doxycycline<br />
Initial or Prolonged Dyspnoea Literature (Doxycycline) PS 100 MG<br />
Other Serious Nausea Health (DAILY),<br />
Paraesthesia Professional UNKNOWN<br />
Productive Cough<br />
<strong>Mefloquine</strong><br />
Hyperhidrosis (<strong>Mefloquine</strong>) SS<br />
Medication Error<br />
Fatigue<br />
Pyrexia<br />
Myalgia<br />
Self-Medication<br />
Insomnia<br />
Oropharyngeal Pain<br />
Sputum Discoloured<br />
Malaria<br />
Pharyngitis<br />
Chills<br />
Sleep Disorder<br />
Decreased Appetite<br />
Arthralgia<br />
Headache<br />
Lymphadenopathy
Date:08/27/03ISR Number: 4175954-1Report Type:Direct Company Report #CTU 200820 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Lariam PS ORAL 250 MG ORAL<br />
Drug Abuser Marijuana C<br />
03-Apr-2012 09:37 AM Page: 516
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/29/03ISR Number: 4176059-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-345390 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Fall Lariam PS Roche ORAL<br />
Hallucination<br />
Coordination Abnormal<br />
Abnormal Behaviour<br />
Balance Disorder<br />
Confusional State<br />
Date:09/02/03ISR Number: 4178742-5Report Type:Direct Company Report #CTU 201022 Age:16 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Emotional Disorder Mefloquin PS ORAL AS DIREC ORAL<br />
Initial or Prolonged Anxiety Accutane C<br />
Disturbance In Attention<br />
Thinking Abnormal<br />
Suicidal Ideation<br />
Obsessive-Compulsive<br />
Disorder<br />
Confusional State<br />
Dysgraphia<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Binge Eating<br />
Abnormal Dreams<br />
Educational Problem<br />
Bipolar Disorder<br />
Date:09/04/03ISR Number: 4178977-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-343702 Age:17 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Overdose Lariam PS Roche ORAL<br />
Muscle Spasms<br />
Poisoning<br />
Date:09/05/03ISR Number: 4180293-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-340171 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Coordination Abnormal Lariam PS Roche ORAL 40 DAY<br />
Initial or Prolonged Anxiety<br />
Transient Ischaemic<br />
Attack<br />
Myalgia<br />
Paraesthesia<br />
Viith Nerve Paralysis
Neuromyopathy<br />
03-Apr-2012 09:37 AM Page: 517
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/15/03ISR Number: 4185949-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-344428 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Lariam PS Roche ORAL 48 DAY<br />
Nausea Trinordiol C ORAL<br />
Gastroenteritis<br />
Bacterial Infection<br />
C-Reactive Protein<br />
Increased<br />
Decreased Appetite<br />
Date:09/22/03ISR Number: 4190417-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-346816 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged Condition Aggravated<br />
Date:09/22/03ISR Number: 4193952-9Report Type:Direct Company Report #CTU 202092 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Fear Lariam 250 Mg Roche<br />
Disability Abnormal Dreams Labs PS Roche Labs ORAL 1 WEEK ORAL<br />
Ear Disorder Yellow Fever Shot C<br />
Gastrointestinal Pain<br />
Muscle Twitching<br />
Panic Attack<br />
Balance Disorder<br />
Dyspepsia<br />
Vestibular Disorder<br />
Depressed Mood<br />
Insomnia<br />
Muscle Spasms<br />
Muscular Weakness<br />
Nausea<br />
Depression<br />
Dizziness<br />
Motion Sickness<br />
Pruritus<br />
Somnambulism<br />
Heart Rate Increased<br />
Anxiety<br />
Hypersensitivity<br />
Irritable Bowel Syndrome<br />
Date:09/23/03ISR Number: 4195373-1Report Type:Direct Company Report #CTU 2002273 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Life-Threatening Fear Lariam 250 Mg<br />
Panic Attack Hoffmann-Laroche PS Hoffmann- Loroche ORAL 1 PILL WEEK<br />
Dyspnoea<br />
ORAL<br />
Thinking Abnormal<br />
Diamox-Acetazolamide C<br />
03-Apr-2012 09:37 AM Page: 518
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/25/03ISR Number: 4196918-8Report Type:Direct Company Report #CTU 202631 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Selective Iga Larium (<strong>Mefloquine</strong>) PS Hoffman-La Roche ORAL 1 TAB WEEK<br />
Immunodeficiency<br />
ORAL<br />
Sleep Disorder<br />
Balance Disorder<br />
Mood Swings<br />
Vestibular Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
Activities Of Daily<br />
Living Impaired<br />
Coordination Abnormal<br />
Date:09/26/03ISR Number: 4197035-3Report Type:Direct Company Report #CTU 202661 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Lariam -<strong>Mefloquine</strong>-<br />
Activities Of Daily Hoffman-La Roche PS Hoffman-La Roche ORAL 1 TAB WEEK<br />
Living Impaired<br />
ORAL<br />
Sleep Disorder<br />
Coordination Abnormal<br />
Vestibular Neuronitis<br />
Mood Swings<br />
Vestibular Disorder<br />
Date:10/01/03ISR Number: 4199107-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-346816 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged<br />
Date:10/02/03ISR Number: 4200052-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-347539 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Feeling Abnormal <strong>Mefloquine</strong><br />
Panic Reaction Hydrochloride PS Roche ORAL 1 DAY<br />
Tremor<br />
Hypoaesthesia<br />
Vitreous Floaters<br />
Date:10/02/03ISR Number: 4200058-9Report Type:Expedited (15-DaCompany Report #BE-ROCHE-347367 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Disability Dizziness <strong>Mefloquine</strong><br />
Speech Disorder Hydrochloride PS Roche ORAL<br />
Coordination Abnormal<br />
Nystagmus<br />
03-Apr-2012 09:37 AM Page: 519
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/02/03ISR Number: 4200060-7Report Type:Expedited (15-DaCompany Report #CA-ROCHE-348024 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Creatine Lariam PS Roche ORAL<br />
Initial or Prolonged Phosphokinase Echinacea C ORAL<br />
Loss Of Consciousness<br />
Vomiting<br />
Nausea<br />
Diarrhoea<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Dehydration<br />
Hepatic Enzyme Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Myalgia<br />
Date:10/03/03ISR Number: 4200906-2Report Type:Expedited (15-DaCompany Report #KR-ROCHE-347870 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Lariam PS Roche ORAL 4 DAY<br />
Initial or Prolonged Visual Impairment<br />
Nasal Congestion<br />
Syncope<br />
Date:10/08/03ISR Number: 4203837-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-347843 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dehydration Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Blood Calcium Decreased<br />
Blood Magnesium Decreased<br />
Blood Parathyroid Hormone<br />
Decreased<br />
Oligodipsia<br />
Date:10/10/03ISR Number: 4205951-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-348532 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pyrexia Lariam PS Roche ORAL<br />
Nausea Lariam SS Roche ORAL<br />
Metrorrhagia Microgynon C ORAL 17 DAY<br />
Date:10/20/03ISR Number: 4212657-9Report Type:Direct Company Report #CTU 204129 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Death Completed Suicide Lariam Roche PS Roche ORAL ORAL<br />
Physical Assault Prozak C<br />
Abnormal Behaviour<br />
Aggression<br />
Thinking Abnormal<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 520
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/21/03ISR Number: 4215201-5Report Type:Expedited (15-DaCompany Report #JP-ROCHE-348824 Age:71 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haemolytic Anaemia Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Jaundice<br />
Systemic Lupus<br />
Erythematosus<br />
Date:10/21/03ISR Number: 4215206-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-349152 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Rhabdomyolysis Lariam PS Roche UNKNOWN<br />
Date:10/22/03ISR Number: 4215411-7Report Type:Direct Company Report #CTU 204278 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Diarrhoea<br />
Mephaquin 275 Mg<br />
Abnormal Dreams<br />
<strong>Mefloquine</strong><br />
Psychiatric Symptom<br />
Hydrichloride Mepha<br />
Thinking Abnormal Ltd PS Mepha Ltd ORAL 1 TABLET 1/<br />
Morbid Thoughts<br />
WEEK ORAL<br />
Anxiety<br />
Date:10/23/03ISR Number: 4215803-6Report Type:Direct Company Report #CTU 204499 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Balance Disorder Lariam 250 Mg PS ORAL 250 MG Q WEEK<br />
Nightmare<br />
ORAL<br />
Date:10/27/03ISR Number: 4218198-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-320492 Age:22 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hypersomnia Lariam PS Roche UNKNOWN<br />
Depression<br />
Feeling Abnormal<br />
Personality Change<br />
Loss Of Consciousness<br />
Overdose<br />
Completed Suicide<br />
Weight Increased<br />
Sensory Disturbance<br />
Tearfulness
Date:10/27/03ISR Number: 4218616-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-349710 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Drugs Affecting Lariam PS Roche ORAL<br />
Foetus<br />
Abortion Spontaneous<br />
03-Apr-2012 09:37 AM Page: 521
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/28/03ISR Number: 4219696-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Antisocial Behaviour Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Initial or Prolonged Convulsion Valium SS Roche INTRAVENOUS 1 DAY<br />
Disability Headache Prozac C ORAL<br />
Mania<br />
Psychotic Disorder<br />
Speech Disorder<br />
Drug Ineffective<br />
Blood Potassium Decreased<br />
Incontinence<br />
Memory Impairment<br />
Delusion<br />
Hostility<br />
Confusional State<br />
Hallucination<br />
Disorientation<br />
Aggression<br />
Paranoia<br />
Depression<br />
Gait Disturbance<br />
Agitation<br />
Fatigue<br />
Loss Of Libido<br />
Date:10/30/03ISR Number: 4222386-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-257719 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Memory Impairment Lariam Tablets PS Roche ORAL SEVEN TABLETS<br />
Suicidal Ideation<br />
TAKEN IN<br />
Hypertension<br />
TOTAL.<br />
Hallucination<br />
Dissociative Disorder<br />
Anxiety<br />
Paranoia<br />
Psychotic Disorder<br />
Agoraphobia<br />
Dizziness<br />
Confusional State<br />
Balance Disorder<br />
Headache<br />
Depression<br />
Date:11/05/03ISR Number: 4237482-4Report Type:Expedited (15-DaCompany Report #12424925 Age:66 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Pressure Decreased Foreign Warfarin Sodium PS ORAL ORAL<br />
Initial or Prolonged Infectious Peritonitis Literature <strong>Mefloquine</strong>
Tachycardia Health (<strong>Mefloquine</strong> Hcl) SS 250<br />
Anaemia Professional MILLIGRAM, 1<br />
Drug Interaction Other WEEK<br />
Prothrombin Time<br />
Captopril Tabs<br />
Prolonged (Captopril) C<br />
Ascites Digoxin (Digoxin) C<br />
Carvedilol<br />
(Carvedilol)<br />
03-Apr-2012 09:37 AM Page: 522<br />
C
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/10/03ISR Number: 4233982-1Report Type:Expedited (15-DaCompany Report #IE-ROCHE-350696 Age:23 MON Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Lariam PS Roche UNKNOWN 0.25 OF A<br />
Initial or Prolonged Lower Respiratory Tract TABLET.<br />
Infection Folic Acid C<br />
Date:11/10/03ISR Number: 4233988-2Report Type:Expedited (15-DaCompany Report #IE-ROCHE-350774 Age:23 MON Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Lariam PS Roche UNKNOWN 0.25 OF A<br />
Initial or Prolonged Lower Respiratory Tract TABLET.<br />
Infection Folic Acid C<br />
Date:11/12/03ISR Number: 4233947-XReport Type:Expedited (15-DaCompany Report #CA-ROCHE-348024 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dehydration Lariam PS Roche ORAL<br />
Initial or Prolonged Blood Creatine Echinacea C ORAL<br />
Phosphokinase<br />
Diarrhoea<br />
Nausea<br />
Vomiting<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Loss Of Consciousness<br />
Myalgia<br />
Hepatitis<br />
Hepatitis Viral<br />
Date:11/14/03ISR Number: 4235630-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-351227 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Lariam PS Roche ORAL 1 DAY<br />
Vision Blurred<br />
Dysgeusia<br />
Product Quality Issue<br />
Corneal Reflex Decreased<br />
Eating Disorder<br />
Date:11/17/03ISR Number: 4236524-XReport Type:Direct Company Report #CTU 206203 Age:35 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
PT<br />
Abdominal Pain<br />
Night Sweats
Depression<br />
Suicidal Ideation<br />
Nightmare<br />
Homicidal Ideation<br />
Fatigue<br />
Insomnia<br />
Overwork<br />
Diarrhoea<br />
03-Apr-2012 09:37 AM Page: 523
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Circadian Rhythm Sleep<br />
Disorder<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam Roche 250 Mg<br />
Tablets PS Roche (1) 250 MG<br />
TABLET 1X<br />
WEEKLY X 5<br />
WKS<br />
Date:11/19/03ISR Number: 4237499-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-351387 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche ORAL<br />
Maternal Drugs Affecting Stamaril Unidose C SUBCUTANEOUS 1 DAY<br />
Foetus<br />
Intra-Uterine Death<br />
Date:11/21/03ISR Number: 4239347-0Report Type:Direct Company Report #CTU 206548 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Abasia Lariam 250 Mg Roche PS Roche ORAL 250 MG WEEKLY<br />
Hospitalization - Photophobia ORAL<br />
Initial or Prolonged Visual Field Defect<br />
Disability<br />
Bladder Disorder<br />
Required<br />
Feeling Abnormal<br />
Intervention to<br />
Hallucination, Auditory<br />
Prevent Permanent<br />
Impairment/Damage<br />
Fall<br />
Suicidal Ideation<br />
Tremor<br />
Anger<br />
Anxiety<br />
Visual Impairment<br />
Insomnia<br />
Aphasia<br />
Panic Attack<br />
Date:12/01/03ISR Number: 4243237-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-350389 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Lariam PS Roche UNKNOWN 3 DOSES TAKEN<br />
Disorientation IN TOTAL. 24 DAY<br />
Panic Attack<br />
Date:12/01/03ISR Number: 4243240-7Report Type:Expedited (15-DaCompany Report #IE-ROCHE-350696 Age:23 MON Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Lariam PS Roche UNKNOWN 0.25 OF A<br />
Initial or Prolonged Malaise TABLET.<br />
Lower Respiratory Tract Penicillin V C ORAL DOSE<br />
Infection<br />
INCREASED TO<br />
White Blood Cell Count<br />
250MG BD.<br />
Increased Cefotaxim C INTRAVENOUS 6 DAY<br />
Dexamethasone C INTRAVENOUS 4MG TO 1 MG,<br />
03-Apr-2012 09:37 AM Page: 524
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
QDS 2 DAY<br />
Folic Acid C ORAL<br />
Clarithromycin C ORAL 8 DAY<br />
Date:12/01/03ISR Number: 4243244-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-350389 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disorientation Lariam PS Roche UNKNOWN 3 DOSES TAKEN<br />
Panic Attack IN TOTAL. 24 DAY<br />
Amnesia<br />
Date:12/01/03ISR Number: 4243580-1Report Type:Expedited (15-DaCompany Report #IE-ROCHE-350774 Age:23 MON Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lower Respiratory Tract Lariam PS Roche ORAL 0.25 OF A<br />
Initial or Prolonged Infection TABLET.<br />
Malaise Penicillin V C ORAL<br />
Pyrexia Clarithromycin C ORAL 8 DAY<br />
White Blood Cell Count Folic Acid C ORAL<br />
Increased Cefotaxime C INTRAVENOUS 8 DAY<br />
Date:12/01/03ISR Number: 4243635-1Report Type:Expedited (15-DaCompany Report #IE-ROCHE-350774 Age:23 MON Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lower Respiratory Tract Lariam PS Roche ORAL 0.25 OF A<br />
Initial or Prolonged Infection TABLET.<br />
Pyrexia Penicillin V C ORAL<br />
Malaise Clarithromycin C ORAL 8 DAY<br />
White Blood Cell Count Folic Acid C ORAL<br />
Increased Cefotaxime C INTRAVENOUS 8 DAY<br />
Date:12/02/03ISR Number: 4244263-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-352392 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicidal Ideation Lariam PS Roche ORAL<br />
Suicidal Ideation<br />
Hallucination<br />
Date:12/04/03ISR Number: 4245784-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-331974 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche UNKNOWN PATIENT TOOK<br />
Maternal Drugs Affecting<br />
3 TABLETS IN<br />
Foetus TOTAL 3 WK
Alcohol Use Oral Contraceptive<br />
Pregnancy On Oral Pill C ORAL<br />
Contraceptive<br />
03-Apr-2012 09:37 AM Page: 525
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/04/03ISR Number: 4245788-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-352436 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Lariam PS Roche UNKNOWN<br />
Iiird Nerve Disorder<br />
Hypoaesthesia<br />
Paraesthesia<br />
Date:12/08/03ISR Number: 4248397-XReport Type:Direct Company Report #CTU 207633 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Decreased Appetite Larium PS ORAL DAILY ORAL<br />
Hyperhidrosis<br />
Night Sweats<br />
Asthenia<br />
Heat Rash<br />
Depression<br />
Insomnia<br />
Lethargy<br />
Date:12/15/03ISR Number: 4251655-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-353732 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche ORAL 50 DAY<br />
Pregnancy<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:12/15/03ISR Number: 4252145-7Report Type:Direct Company Report #CTU 208025 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Attack <strong>Mefloquine</strong> PS ORAL ONCE WEEK<br />
Anxiety<br />
ORAL<br />
Birth Control Pill C<br />
Cipro<br />
C<br />
Zovia<br />
C<br />
Date:12/18/03ISR Number: 4253973-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Paranoia<br />
Disorientation<br />
Blood Potassium Decreased<br />
Vision Blurred<br />
Memory Impairment
Hallucination<br />
Psychotic Disorder<br />
Incontinence<br />
Disturbance In Attention<br />
Hostility<br />
Disability<br />
Somnolence<br />
Gait Disturbance<br />
03-Apr-2012 09:37 AM Page: 526
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fatigue<br />
Circulatory Collapse<br />
Delusion Report Source Product Role Manufacturer Route Dose Duration<br />
Decreased Activity Consumer Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Drug Ineffective Valium SS Roche INTRAVENOUS 1 DAY<br />
Aggression Prozac C ORAL<br />
Depression<br />
Speech Disorder<br />
Agitation<br />
Headache<br />
Antisocial Behaviour<br />
Loss Of Libido<br />
Confusional State<br />
Mania<br />
Convulsion<br />
Date:12/18/03ISR Number: 4253974-6Report Type:Expedited (15-DaCompany Report #CA-ROCHE-348024 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myalgia Health Lariam PS Roche ORAL<br />
Initial or Prolonged Blood Creatine Professional Echinacea C ORAL<br />
Phosphokinase<br />
Dehydration<br />
Hepatitis Viral<br />
Loss Of Consciousness<br />
Vomiting<br />
Nausea<br />
Hepatitis<br />
Syncope<br />
Diarrhoea<br />
Date:12/22/03ISR Number: 4256133-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-354080 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion <strong>Mefloquine</strong><br />
Loss Of Consciousness Hydrochloride PS Roche ORAL<br />
Respiratory Arrest<br />
Date:12/22/03ISR Number: 4256138-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-354423 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Biliary Colic Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Cholecystitis<br />
Date:12/23/03ISR Number: 4257040-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-352436 Age:30 YR Gender:Female I/FU:F
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Feeling Cold<br />
Muscle Rigidity<br />
Protein C Increased<br />
Iiird Nerve Disorder<br />
Nervous System Disorder<br />
Gait Disturbance<br />
03-Apr-2012 09:37 AM Page: 527
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Injection Site<br />
Anaesthesia<br />
Dyspnoea Report Source Product Role Manufacturer Route Dose Duration<br />
Paraesthesia Health Lariam PS Roche UNKNOWN<br />
Chest Discomfort<br />
Professional<br />
Tremor<br />
Diplopia<br />
Painful Respiration<br />
Hypoaesthesia<br />
Cerebellar Syndrome<br />
Date:01/05/04ISR Number: 4263391-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paranoia Lariam PS Roche ORAL 23 TABLETS<br />
Balance Disorder<br />
DISPENSED.<br />
Suicidal Ideation<br />
Hallucination<br />
Neuropathy Peripheral<br />
Viiith Nerve Lesion<br />
Visual Impairment<br />
Tremor<br />
Photophobia<br />
Hepatic Failure<br />
Panic Attack<br />
Date:01/07/04ISR Number: 4265742-XReport Type:Expedited (15-DaCompany Report #GB-ROCHE-354080 Age:21 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Consumer <strong>Mefloquine</strong><br />
Respiratory Arrest Hydrochloride PS Roche ORAL<br />
Loss Of Consciousness<br />
Date:01/09/04ISR Number: 4268113-5Report Type:Direct Company Report #CTU 209610 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nightmare Mefloquin 250 Mg<br />
Initial or Prolonged Tremor Lariam PS ORAL 1 WEEK ORAL<br />
Disability<br />
Depression<br />
Anxiety<br />
Convulsion<br />
Date:01/12/04ISR Number: 4268779-XReport Type:Direct Company Report #CTU 209704 Age:54 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Depression Lariam PS ORAL ONE TIME ORAL<br />
Initial or Prolonged Convulsion Antibiotic C<br />
Anxiety Levaquen C<br />
Reglan<br />
C<br />
Prevacid<br />
C<br />
03-Apr-2012 09:37 AM Page: 528
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/14/04ISR Number: 4271467-7Report Type:Direct Company Report #CTU 210092 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Feeling Abnormal Lariam <strong>Mefloquine</strong><br />
Sleep Disorder Roche PS Roche ORAL 1 WEEK ORAL<br />
Hyperacusis<br />
Stress<br />
Depression<br />
Anxiety<br />
Emotional Disorder<br />
Date:01/20/04ISR Number: 4274670-5Report Type:Direct Company Report #CTU 210400 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS WEEKLY<br />
Panic Attack<br />
Nightmare<br />
Vertigo<br />
Date:01/29/04ISR Number: 4281325-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-351227 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dysgeusia Consumer Lariam PS Roche ORAL 1 DAY<br />
Viith Nerve Paralysis<br />
Product Quality Issue<br />
Vision Blurred<br />
Lacrimation Increased<br />
Date:01/30/04ISR Number: 4282938-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-357211 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Amnesia <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL PATIENT TOOK<br />
ONLY ONE DOSE<br />
OF<br />
MEFLOQUINE. 1 DAY<br />
Date:02/02/04ISR Number: 4283667-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-357080 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dysphagia Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Urticaria
Date:02/04/04ISR Number: 4285179-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Hallucination<br />
Mania<br />
Psychotic Disorder<br />
Speech Disorder<br />
Confusional State<br />
03-Apr-2012 09:37 AM Page: 529
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Potassium Decreased<br />
Hostility<br />
Decreased Activity Report Source Product Role Manufacturer Route Dose Duration<br />
Gait Disturbance Health Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Antisocial Behaviour Professional Valium SS Roche INTRAVENOUS 1 DAY<br />
Loss Of Libido Prozac C ORAL<br />
Disturbance In Attention<br />
Paranoia<br />
Disorientation<br />
Convulsion<br />
Agitation<br />
Fatigue<br />
Aggression<br />
Vision Blurred<br />
Incontinence<br />
Drug Ineffective<br />
Headache<br />
Depression<br />
Memory Impairment<br />
Delusion<br />
Date:02/05/04ISR Number: 4286243-9Report Type:Direct Company Report #CTU 211605 Age:10 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Lariam 125 Mg/5ml. PS 1 TSP ONE<br />
TIME PER WK<br />
Methlaquine Tabs 250<br />
Mg - Compounded SS<br />
Date:02/06/04ISR Number: 4286779-0Report Type:Expedited (15-DaCompany Report #NL-ROCHE-348532 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Metrorrhagia Health Lariam PS Roche ORAL<br />
Pyrexia Professional Lariam SS Roche ORAL<br />
Nausea Microgynon 30 C ORAL 17 DAY<br />
Date:02/06/04ISR Number: 4286781-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-355117 Age:21 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Spasms Lariam PS Roche ORAL 1 DAY<br />
Mucosal Inflammation<br />
Throat Irritation<br />
Oropharyngeal Blistering<br />
Palpitations<br />
Dizziness
Date:02/12/04ISR Number: 4293681-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Hallucination<br />
Depression<br />
Gait Disturbance<br />
Blood Potassium Decreased<br />
03-Apr-2012 09:37 AM Page: 530
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paranoia<br />
Speech Disorder<br />
Fatigue Report Source Product Role Manufacturer Route Dose Duration<br />
Mania Health Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Confusional State Professional Valium SS Roche INTRAVENOUS 1 DAY<br />
Delusion Prozac C ORAL<br />
Pyrexia<br />
Muscle Rigidity<br />
Disorientation<br />
Hostility<br />
Memory Impairment<br />
Aggression<br />
Headache<br />
Psychotic Disorder<br />
Convulsion<br />
Loss Of Libido<br />
Incontinence<br />
Agitation<br />
Antisocial Behaviour<br />
Drug Ineffective<br />
Date:02/12/04ISR Number: 4293695-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-354080 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Consumer <strong>Mefloquine</strong><br />
Loss Of Consciousness Hydrochloride PS Roche ORAL<br />
Date:02/12/04ISR Number: 4294092-0Report Type:Direct Company Report #CTU 212123 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Anger Larium 250 Mg Roache<br />
Aggression Pharmaceuticals PS Roache<br />
Psychotic Disorder Pharmaceuticals ORAL 250 MG ,<br />
Imprisonment<br />
WEEKLY ORAL<br />
Date:02/16/04ISR Number: 4295449-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-351227 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Viith Nerve Paralysis Consumer Lariam PS Roche ORAL 1 DAY<br />
Dysgeusia<br />
Date:02/16/04ISR Number: 4295459-7Report Type:Expedited (15-DaCompany Report #CA-ROCHE-358463 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Schizoaffective Disorder Lariam PS Roche ORAL
Mania<br />
Psychotic Disorder<br />
03-Apr-2012 09:37 AM Page: 531
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/24/04ISR Number: 4303509-4Report Type:Direct Company Report #CTU 213006 Age:69 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Abnormal Dreams <strong>Mefloquine</strong> Hcl 250<br />
Intervention to Hallucination Mg Tab Geneva Pharm.<br />
Prevent Permanent Anxiety Tech. Corp PS Geneva Pharm Tech.<br />
Impairment/Damage Sleep Disorder Corp ORAL 1 A WEEK ORAL<br />
Affect Lability Flomax C<br />
Insomnia Proscar C<br />
Decreased Appetite<br />
Depression<br />
Panic Attack<br />
Date:02/26/04ISR Number: 4307492-7Report Type:Expedited (15-DaCompany Report #2003IC000217 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Intra-Uterine Death Foreign Librax<br />
Congenital Anomaly Pregnancy Health (Chlordiazepoxide<br />
Abortion Induced Professional Hydrochloride/Clidin<br />
Foetal Malformation ium Bromide) PS TRANSPLACENTAL TRANSPLACENTA<br />
Maternal Drugs Affecting<br />
L<br />
Foetus<br />
Stamaril Amaril<br />
Hydrocephalus<br />
Vaccin (Generic<br />
Foetal Disorder Unknown) SS TRANSPLACENTAL 1 FD;<br />
Haemorrhage<br />
TRANSPLACENTA<br />
Foetal Malposition<br />
L<br />
Lariam (<strong>Mefloquine</strong><br />
Chlorhydrate)<br />
(Generic Unknown) SS TRANSPLACENTAL TRANSPLANCENT<br />
AL<br />
Date:03/01/04ISR Number: 4308476-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-359452 Age:53 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Lymphadenopathy Consumer Lariam PS Roche ORAL DOSAGE<br />
Initial or Prolonged Pulmonary Fibrosis REPORTED AS<br />
Interstitial Lung Disease 1.1 AND 1.2<br />
MG/L.<br />
Unknown Medication C INTRAVENOUS DRUG REPORTED<br />
AS CLAMOXIL. 3 DAY<br />
Date:03/03/04ISR Number: 4310349-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-355117 Age:21 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Spasms Health Lariam PS Roche ORAL 1 DAY<br />
Palpitations<br />
Professional<br />
Product Quality Issue
Medication Error<br />
Mucosal Inflammation<br />
Throat Irritation<br />
Oropharyngeal Blistering<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 532
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/03/04ISR Number: 4310471-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-351227 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dysgeusia Health Lariam PS Roche ORAL 1 DAY<br />
Vision Blurred<br />
Professional<br />
Face Oedema<br />
Corneal Reflex Decreased<br />
Viith Nerve Paralysis<br />
Date:03/08/04ISR Number: 4313479-0Report Type:Expedited (15-DaCompany Report #CA-ROCHE-358463 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mania Health Lariam PS Roche ORAL<br />
Psychotic Disorder<br />
Professional<br />
Schizoaffective Disorder<br />
Treatment Noncompliance<br />
Date:03/09/04ISR Number: 4314313-5Report Type:Direct Company Report #CTU 214091 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety Lariam 250 Mg Roche PS Roche ORAL 500 MG WEEKLY<br />
Hospitalization - Panic Attack ORAL, 1500 MG<br />
Initial or Prolonged Malaria ONE TIME ORAL<br />
Required Drug Ineffective Cipro C<br />
Intervention to Major Depression Flagyl C<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:03/11/04ISR Number: 4315538-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-351227 Age:55 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vision Blurred Lariam PS Roche ORAL 1 DAY<br />
Dysgeusia<br />
Viith Nerve Paralysis<br />
Date:03/15/04ISR Number: 4317682-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Mitral Valve Prolapse<br />
Depression<br />
Viral Infection<br />
Arthralgia<br />
Constipation<br />
Upper-Airway Cough<br />
Syndrome
Hypertension<br />
Angina Pectoris<br />
Total Lung Capacity<br />
Decreased<br />
Somnolence<br />
Bronchospasm<br />
Low Density Lipoprotein<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 533
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Exfoliative Rash<br />
Dry Skin<br />
Chest Discomfort Report Source Product Role Manufacturer Route Dose Duration<br />
Vestibular Disorder Lariam PS Roche ORAL<br />
Skin Disorder Zoloft C UNKNOWN<br />
Bronchitis<br />
Sinus Tachycardia<br />
Blood Triglycerides<br />
Increased<br />
Dehydration<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Hallucination, Visual<br />
Gastrointestinal Disorder<br />
Diastolic Dysfunction<br />
Cardiac Murmur<br />
Dry Eye<br />
Vitiligo<br />
Blood Glucose Increased<br />
Gastrooesophageal Reflux<br />
Disease<br />
Amnesia<br />
Fatigue<br />
Rash<br />
Dry Mouth<br />
Snoring<br />
Anxiety<br />
Headache<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Rhinitis<br />
Dyspnoea<br />
Weight Increased<br />
Blood Cholesterol<br />
Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Diarrhoea<br />
Oral Disorder<br />
Insomnia<br />
Confusional State<br />
Ventricular Hypertrophy<br />
Restrictive Pulmonary<br />
Disease<br />
Nocturia<br />
Acrochordon<br />
Date:03/16/04ISR Number: 4317812-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Anxiety
Ventricular Hypertrophy<br />
Hypertension<br />
Bronchitis<br />
Cardiac Murmur<br />
Dry Eye<br />
Gastrooesophageal Reflux<br />
Disease<br />
Upper-Airway Cough<br />
03-Apr-2012 09:37 AM Page: 534
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Syndrome<br />
Amnesia<br />
Skin Disorder<br />
Fatigue Report Source Product Role Manufacturer Route Dose Duration<br />
Somnolence Lariam PS Roche ORAL<br />
Rash Zoloft C UNKNOWN<br />
Weight Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Dehydration<br />
Oral Disorder<br />
Vestibular Disorder<br />
Angina Pectoris<br />
Dry Mouth<br />
Constipation<br />
Rhinitis<br />
Hallucination, Visual<br />
Headache<br />
Total Lung Capacity<br />
Decreased<br />
Sinus Tachycardia<br />
Low Density Lipoprotein<br />
Increased<br />
Exfoliative Rash<br />
Confusional State<br />
Restrictive Pulmonary<br />
Disease<br />
Nocturia<br />
Vitiligo<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Blood Glucose Increased<br />
Hypersomnia<br />
Mitral Valve Prolapse<br />
Depression<br />
Diastolic Dysfunction<br />
Bronchospasm<br />
Blood Cholesterol<br />
Increased<br />
Blood Triglycerides<br />
Increased<br />
Dry Skin<br />
Chest Discomfort<br />
Insomnia<br />
Dyspnoea<br />
Diarrhoea<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Snoring<br />
Gastrointestinal Disorder<br />
Viral Infection<br />
Arthralgia<br />
Acrochordon
Date:03/16/04ISR Number: 4317891-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Skin Disorder<br />
Viral Infection<br />
Bronchitis<br />
03-Apr-2012 09:37 AM Page: 535
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Chest Discomfort<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Fundoscopy Abnormal<br />
Body Temperature<br />
Increased<br />
Dyspnoea<br />
Diastolic Dysfunction<br />
Weight Increased<br />
Blood Cholesterol<br />
Increased<br />
Dry Mouth<br />
Vitiligo<br />
Exercise Tolerance<br />
Decreased<br />
Nausea<br />
Anxiety<br />
Hypertension<br />
Blood Triglycerides<br />
Increased<br />
Dehydration<br />
Constipation<br />
Rhinitis<br />
Myalgia<br />
Heart Sounds Abnormal<br />
Occult Blood Positive<br />
Exfoliative Rash<br />
Dry Eye<br />
Chronic Fatigue Syndrome<br />
Hypersomnia<br />
Rash<br />
Low Density Lipoprotein<br />
Increased<br />
Upper-Airway Cough<br />
Syndrome<br />
Pain In Jaw<br />
Chills<br />
Mitral Valve Prolapse<br />
Bronchospasm<br />
Cardiac Murmur<br />
Blood Glucose Increased<br />
Insomnia<br />
Tongue Coated<br />
Listless<br />
Neck Pain<br />
Amnesia<br />
Hallucination, Visual<br />
Gastrointestinal Disorder<br />
Fatigue<br />
Angina Pectoris<br />
Total Lung Capacity<br />
Decreased<br />
Somnolence
Diarrhoea<br />
Dry Skin<br />
Snoring<br />
Nocturia<br />
Oral Disorder<br />
Upper Respiratory Tract<br />
Infection<br />
Impaired Work Ability<br />
03-Apr-2012 09:37 AM Page: 536
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abdominal Tenderness<br />
Confusional State<br />
Vestibular Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Depression Lariam PS Roche ORAL<br />
Headache Zoloft C UNKNOWN<br />
Ventricular Hypertrophy<br />
Restrictive Pulmonary<br />
Disease<br />
Arthralgia<br />
Sinus Tachycardia<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Acrochordon<br />
Gastrooesophageal Reflux<br />
Disease<br />
Drug Tolerance Decreased<br />
Pyrexia<br />
Injury<br />
Date:03/18/04ISR Number: 4318870-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-321435 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Maternal Exposure During Consumer Lariam PS Roche 37 DAY<br />
Initial or Prolonged Pregnancy Nivaquine SS 14 DAY<br />
Premature Baby Quinine SS 6 DAY<br />
Paludrine SS 14 DAY<br />
Date:03/18/04ISR Number: 4318889-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-361697 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Premature Baby Consumer Lariam PS Roche 37 DAY<br />
Initial or Prolonged Twin Pregnancy Nivaquine SS 14 DAY<br />
Paludrine SS 14 DAY<br />
Quinine SS 6 DAY<br />
Date:03/23/04ISR Number: 4322410-3Report Type:Direct Company Report #CTU 215048 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Impaired Work Ability Larium Standard For<br />
Activities Of Daily Malaria PS<br />
Living Impaired<br />
Depression
Date:03/29/04ISR Number: 4325789-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Fatigue<br />
Rash<br />
Constipation<br />
Oral Disorder<br />
Dyspnoea<br />
03-Apr-2012 09:37 AM Page: 537
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mitral Valve Prolapse<br />
Total Lung Capacity<br />
Decreased<br />
Bronchospasm<br />
Weight Increased<br />
Chest Discomfort<br />
Major Depression<br />
Scratch<br />
Chest X-Ray Abnormal<br />
Vestibular Disorder<br />
Blood Cholesterol<br />
Increased<br />
Dehydration<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Exfoliative Rash<br />
Abnormal Dreams<br />
Bipolar Disorder<br />
Haemorrhoids<br />
Balance Disorder<br />
Affective Disorder<br />
Confusional State<br />
Restrictive Pulmonary<br />
Disease<br />
Blood Triglycerides<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Diarrhoea<br />
Dry Skin<br />
Dry Eye<br />
Vitiligo<br />
Gastrooesophageal Reflux<br />
Disease<br />
Rhinitis<br />
Clumsiness<br />
Coordination Abnormal<br />
Adjustment Disorder<br />
Neck Pain<br />
Ventricular Hypertrophy<br />
Somnolence<br />
Arthralgia<br />
Cardiac Murmur<br />
Blood Glucose Increased<br />
Visual Impairment<br />
Asthma<br />
Hypersomnia<br />
Wheezing<br />
Sputum Abnormal<br />
Oedema Peripheral<br />
Skin Disorder<br />
Diastolic Dysfunction<br />
Acrochordon
Contusion<br />
Upper Respiratory Tract<br />
Infection<br />
Dyspnoea Exertional<br />
Depression<br />
Hypertension<br />
Angina Pectoris<br />
Bronchitis<br />
03-Apr-2012 09:37 AM Page: 538
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sinus Tachycardia<br />
Total Cholesterol/Hdl<br />
Ratio Decreased Report Source Product Role Manufacturer Route Dose Duration<br />
Snoring Consumer Lariam PS Roche ORAL<br />
Angiotensin Converting Zoloft C UNKNOWN<br />
Enzyme Increased<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Body Temperature<br />
Increased<br />
Amnesia<br />
Hallucination, Visual<br />
Anxiety<br />
Gastrointestinal Disorder<br />
Headache<br />
Viral Infection<br />
Dry Mouth<br />
Nocturia<br />
Upper-Airway Cough<br />
Syndrome<br />
Insomnia<br />
Memory Impairment<br />
Date:04/05/04ISR Number: 4332092-2Report Type:Expedited (15-DaCompany Report #ZA-ROCHE-362924 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psoriasis Lariam PS Roche ORAL 15 DAY<br />
Date:04/08/04ISR Number: 4334985-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-360920 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Muscular Weakness Lariam PS Roche ORAL 2 DAY<br />
Myalgia Lariam SS Roche ORAL<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Balance Disorder<br />
Insomnia<br />
Vertigo<br />
Hypertension<br />
Fall<br />
Date:04/21/04ISR Number: 4343035-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Dyspnoea
Mitral Valve Prolapse<br />
Hypertension<br />
Somnolence<br />
Cardiac Murmur<br />
Blood Cholesterol<br />
Increased<br />
Blood Triglycerides<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 539
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Upper-Airway Cough<br />
Syndrome<br />
Insomnia<br />
Affective Disorder<br />
Depression<br />
Headache<br />
Ventricular Hypertrophy<br />
Diastolic Dysfunction<br />
Bronchitis<br />
Dehydration<br />
Dry Skin<br />
Bipolar Disorder<br />
Major Depression<br />
Restrictive Pulmonary<br />
Disease<br />
Viral Infection<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Exfoliative Rash<br />
Nocturia<br />
Vitiligo<br />
Visual Impairment<br />
Abnormal Dreams<br />
Coordination Abnormal<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Adjustment Disorder<br />
Hallucination, Visual<br />
Gastrointestinal Disorder<br />
Fatigue<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Gastrooesophageal Reflux<br />
Disease<br />
Balance Disorder<br />
Hypersomnia<br />
Anxiety<br />
Bronchospasm<br />
Diarrhoea<br />
Memory Impairment<br />
Angina Pectoris<br />
Sleep Apnoea Syndrome<br />
Rash<br />
Weight Increased<br />
Dry Eye<br />
Oral Disorder<br />
Blood Glucose Increased<br />
Rhinitis<br />
Contusion<br />
Amnesia<br />
Vestibular Disorder<br />
Arthralgia<br />
Low Density Lipoprotein
Increased<br />
Constipation<br />
Acrochordon<br />
Clumsiness<br />
Haemorrhoids<br />
Scratch<br />
Confusional State<br />
Skin Disorder<br />
03-Apr-2012 09:37 AM Page: 540
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Total Lung Capacity<br />
Decreased<br />
Sinus Tachycardia Report Source Product Role Manufacturer Route Dose Duration<br />
Dry Mouth Consumer Lariam PS Roche ORAL<br />
Snoring Zoloft C UNKNOWN<br />
Chest Discomfort<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Asthma<br />
Date:04/21/04ISR Number: 4344421-4Report Type:Direct Company Report #CTU 217021 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anger <strong>Mefloquine</strong> 250 Mg PS ORAL 250 MG Q WK<br />
Headache PO 2 MON<br />
Myalgia<br />
Mood Swings<br />
Date:04/26/04ISR Number: 4347305-0Report Type:Direct Company Report #CTU 217365 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Tachycardia <strong>Mefloquine</strong> Lariam PS ORAL ONE TAB<br />
Intervention to Heart Rate Irregular WEEKLY ORAL<br />
Prevent Permanent Extrasystoles<br />
Impairment/Damage<br />
Date:04/27/04ISR Number: 4348517-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-365129 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania Lariam PS Roche ORAL<br />
Initial or Prolonged Psychotic Disorder<br />
Date:04/27/04ISR Number: 4349105-4Report Type:Direct Company Report #CTU 217446 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gait Disturbance Lariam 250 Mg<br />
Eating Disorder Hoffmann-La Roche PS Hoffmann-La Roche ORAL 1 TABLET<br />
Muscle Spasms<br />
WEEKLY ORAL<br />
Dizziness<br />
Date:04/30/04ISR Number: 4351518-1Report Type:Direct Company Report #CTU 217725 Age:60 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Loss Of Consciousness <strong>Mefloquine</strong> 250mg<br />
Initial or Prolonged Geneva PS Geneva 1 PER WEEK<br />
Minocycline 200mg SS 200MG FIRST<br />
DAY, 100MG<br />
AFTER<br />
03-Apr-2012 09:37 AM Page: 541
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/03/04ISR Number: 4351449-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-328967 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Anomaly Lariam PS Roche 1 DAY<br />
Hypospadias<br />
Testicular Atrophy<br />
Testicular Disorder<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:05/12/04ISR Number: 4357189-2Report Type:Direct Company Report #CTU 218492 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Lariam 250 Mg Roche PS Roche ORAL 1 PILL PER WK<br />
Required Fear ORAL<br />
Intervention to<br />
Thinking Abnormal<br />
Prevent Permanent<br />
Impairment/Damage<br />
Mental Disorder<br />
Malaise<br />
Feeling Abnormal<br />
Confusional State<br />
Date:05/14/04ISR Number: 4358415-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vestibular Disorder<br />
Rash<br />
Weight Increased<br />
Diarrhoea<br />
Chest Discomfort<br />
Blood Glucose Increased<br />
Clumsiness<br />
Haemorrhoids<br />
Schizoaffective Disorder<br />
Sleep Apnoea Syndrome<br />
Bronchospasm<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Exfoliative Rash<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Rhinitis<br />
Balance Disorder<br />
Psychotic Disorder<br />
Anxiety<br />
Headache<br />
Restrictive Pulmonary<br />
Disease
Viral Infection<br />
Arthralgia<br />
Bronchitis<br />
Sinus Tachycardia<br />
Constipation<br />
Upper-Airway Cough<br />
Syndrome<br />
Asthma<br />
03-Apr-2012 09:37 AM Page: 542
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Fatigue Report Source Product Role Manufacturer Route Dose Duration<br />
Total Lung Capacity Consumer Lariam PS Roche ORAL<br />
Decreased Zoloft C UNKNOWN<br />
Cardiac Murmur<br />
Blood Cholesterol<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Oral Disorder<br />
Vitiligo<br />
Contusion<br />
Memory Impairment<br />
Confusional State<br />
Gastrointestinal Disorder<br />
Depression<br />
Snoring<br />
Bipolar Disorder<br />
Major Depression<br />
Hypertension<br />
Dry Mouth<br />
Gastrooesophageal Reflux<br />
Disease<br />
Visual Impairment<br />
Abnormal Dreams<br />
Scratch<br />
Amnesia<br />
Dyspnoea<br />
Mitral Valve Prolapse<br />
Skin Disorder<br />
Angina Pectoris<br />
Blood Triglycerides<br />
Increased<br />
Dry Skin<br />
Coordination Abnormal<br />
Affective Disorder<br />
Ventricular Hypertrophy<br />
Diastolic Dysfunction<br />
Somnolence<br />
Dehydration<br />
Dry Eye<br />
Nocturia<br />
Acrochordon<br />
Insomnia<br />
Date:05/14/04ISR Number: 4358421-1Report Type:Expedited (15-DaCompany Report #CH-ROCHE-357220 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blindness Unilateral Lariam PS Roche ORAL DOSAGE<br />
Initial or Prolonged Vitreous Haemorrhage REGIMEN
Lariam SS Roche ORAL<br />
REPORTED AS<br />
250 MG.<br />
03-Apr-2012 09:37 AM Page: 543
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/14/04ISR Number: 4358430-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-367059 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Glaucoma Lariam PS Roche ORAL<br />
Arrhythmia<br />
Date:05/14/04ISR Number: 4358440-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-367058 Age:81 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Glaucoma Lariam PS Roche ORAL<br />
Silent Myocardial<br />
Infarction<br />
Date:05/24/04ISR Number: 4363710-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Disorientation Lariam PS Roche ORAL<br />
Insomnia<br />
Vertigo<br />
Diarrhoea<br />
Vestibular Disorder<br />
Chest Pain<br />
Night Sweats<br />
Agitation<br />
Anxiety Disorder<br />
Fatigue<br />
Psychotic Disorder<br />
Nightmare<br />
Amnesia<br />
Tinnitus<br />
Vomiting<br />
Memory Impairment<br />
Nausea<br />
Mood Swings<br />
Date:05/27/04ISR Number: 4366518-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Paranoia<br />
Depression<br />
Delusion<br />
Speech Disorder<br />
Confusional State<br />
Aggression<br />
Blood Potassium Decreased<br />
Agitation<br />
Antisocial Behaviour
Drug Ineffective<br />
Muscle Rigidity<br />
Disorientation<br />
Convulsion<br />
Loss Of Libido<br />
Psychotic Disorder<br />
Gait Disturbance<br />
Incontinence<br />
03-Apr-2012 09:37 AM Page: 544
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Memory Impairment<br />
Mania<br />
Pyrexia Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination Health Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Hostility Professional Valium SS Roche INTRAVENOUS 1 DAY<br />
Fatigue Prozac C ORAL<br />
Headache<br />
Date:05/27/04ISR Number: 4366535-5Report Type:Expedited (15-DaCompany Report #CA-ROCHE-368340 Age:17 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urinary Incontinence Lariam PS Roche ORAL 58 DAY<br />
Panic Reaction<br />
Dizziness<br />
Date:05/27/04ISR Number: 4367724-6Report Type:Periodic Company Report #262840 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cold Sweat Health Lariam (<strong>Mefloquine</strong><br />
Heart Rate Irregular Professional Hydrochloride) 250<br />
Depression Other Mg PS ORAL 1 DOSE FROM 1<br />
Tinnitus<br />
PER WEEK ORAL<br />
Abdominal Distension<br />
Weight Decreased<br />
Acrophobia<br />
Heart Rate Increased<br />
Diarrhoea<br />
Dyspnoea<br />
Disturbance In Attention<br />
Anxiety<br />
Panic Attack<br />
Motion Sickness<br />
Hypersensitivity<br />
Phobia Of Flying<br />
Suicidal Ideation<br />
Dizziness<br />
Self Esteem Decreased<br />
Photophobia<br />
Burning Sensation<br />
Paraesthesia<br />
Nausea<br />
Abdominal Pain<br />
Rash<br />
Vestibular Disorder<br />
Faeces Discoloured<br />
Phobia<br />
Claustrophobia<br />
Disorientation<br />
Balance Disorder
Emotional Distress<br />
Flatulence<br />
Constipation<br />
Insomnia<br />
Headache<br />
Irritable Bowel Syndrome<br />
03-Apr-2012 09:37 AM Page: 545
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/27/04ISR Number: 4367725-8Report Type:Periodic Company Report #350185 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Tinnitus Health Lariam (<strong>Mefloquine</strong><br />
Anxiety Professional Hydrochloride) 250<br />
Dizziness Mg PS ORAL 250 MG, 1 PER<br />
Psychotic Disorder<br />
WEEK ORAL<br />
Insomnia<br />
Tremor<br />
Confusional State<br />
Abnormal Behaviour<br />
Head Discomfort<br />
Date:05/27/04ISR Number: 4367728-3Report Type:Periodic Company Report #354441 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Arrhythmia Consumer Lariam (<strong>Mefloquine</strong><br />
Death Other Hydrochloride) 250<br />
Adverse Event Mg PS ORAL ORAL<br />
Date:05/27/04ISR Number: 4367729-5Report Type:Periodic Company Report #358251 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Professional Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Propranolol<br />
(Propranolol<br />
Hydrochloride) C<br />
Date:05/27/04ISR Number: 4367731-3Report Type:Periodic Company Report #358470 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Health Lariam (<strong>Mefloquine</strong><br />
Obsessive Thoughts Professional Hydrochloride) 250<br />
Panic Attack Mg PS ORAL ORAL<br />
Anxiety<br />
Date:05/27/04ISR Number: 4367732-5Report Type:Periodic Company Report #360727 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Health Lariam (<strong>Mefloquine</strong><br />
Anxiety Professional Hydrochloride) 250<br />
Mg PS ORAL ORAL
Date:05/27/04ISR Number: 4367733-7Report Type:Periodic Company Report #361035 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Social Avoidant Behaviour Health Lariam (<strong>Mefloquine</strong><br />
Anger Professional Hydrochloride) 250<br />
Paranoia Mg PS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 546
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/27/04ISR Number: 4367734-9Report Type:Periodic Company Report #361644 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Reactive Psychosis Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG ORAL<br />
Malarone<br />
(Atovaquone/Chlorogu<br />
anide Hydrochloride) SS ORAL ORAL<br />
Date:05/27/04ISR Number: 4367735-0Report Type:Periodic Company Report #361850 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL 1 PER WEEK<br />
ORAL<br />
Date:05/27/04ISR Number: 4367736-2Report Type:Periodic Company Report #362309 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Adverse Event Health Lariam (<strong>Mefloquine</strong><br />
Completed Suicide Professional Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:05/27/04ISR Number: 4367737-4Report Type:Periodic Company Report #336177 Age:40 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Oral Disorder<br />
Toxicity To Various<br />
Agents<br />
Hyperventilation<br />
Dizziness<br />
Vision Blurred<br />
Insomnia<br />
Mood Altered<br />
Dyspnoea<br />
Abnormal Dreams<br />
Heart Rate Increased<br />
Panic Attack<br />
Hyperhidrosis<br />
Fatigue<br />
Blood Pressure Increased<br />
Adverse Event<br />
Headache<br />
Paraesthesia<br />
Agitation
Vertigo<br />
Decreased Activity<br />
Erythema<br />
Chest Discomfort<br />
Balance Disorder<br />
Lichen Planus<br />
Disturbance In Attention<br />
Nightmare<br />
03-Apr-2012 09:37 AM Page: 547
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Anxiety<br />
Panic Disorder<br />
Depression Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer<br />
Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) 250<br />
Professional Mg PS ORAL 250 MG ORAL<br />
Other<br />
Date:05/27/04ISR Number: 4367738-6Report Type:Periodic Company Report #339621 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Confusional State Hydrochloride) 250<br />
Disability Night Sweats Mg PS ORAL 250 MG 1 PER<br />
Adverse Event<br />
WEEK ORAL<br />
Anxiety<br />
Disturbance In Attention<br />
Depression<br />
Palpitations<br />
Date:05/27/04ISR Number: 4367739-8Report Type:Periodic Company Report #339943 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Consumer Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) PS<br />
Date:05/27/04ISR Number: 4367740-4Report Type:Periodic Company Report #342838 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Tinnitus Other Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 1 DOSE FORM 1<br />
PER WEEK ORAL<br />
Date:05/27/04ISR Number: 4367741-6Report Type:Periodic Company Report #343641 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Adverse Event Literature <strong>Mefloquine</strong><br />
Initial or Prolonged Psychotic Disorder Health Hydrochloride<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Hydrochloride) PS 250 MG 1 PER<br />
WEEK<br />
Oral Contraceptive<br />
Nos (Oral<br />
Contraceptive Nos)<br />
C
Date:05/27/04ISR Number: 4367744-1Report Type:Periodic Company Report #344143 Age:42 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Anxiety<br />
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 548
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Emotional Disorder<br />
Insomnia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Consumer<br />
Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL 1 DOSE FOR 1<br />
PER WEEK ORAL<br />
Liquibid<br />
(Guaifenesin)<br />
C<br />
Flonase (Fluticasone<br />
Propionate)<br />
C<br />
Zyrtec (Cetirizine<br />
Hydrochloride) C<br />
Date:05/27/04ISR Number: 4367745-3Report Type:Periodic Company Report #346923 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chills Consumer Lariam (<strong>Mefloquine</strong><br />
Suicidal Ideation Health Hydrochloride) PS ORAL ORAL<br />
Weight Decreased<br />
Professional<br />
Depression<br />
Decreased Appetite<br />
Fatigue<br />
Hyperhidrosis<br />
Adverse Event<br />
Date:05/27/04ISR Number: 4367746-5Report Type:Periodic Company Report #347625 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Consumer Lariam (<strong>Mefloquine</strong><br />
Vomiting Hydrochloride) PS ORAL ORAL<br />
Neuropathy Peripheral<br />
Chloroquine<br />
Dizziness (Chloroquine) C<br />
Headache<br />
Nitroglycerine<br />
(Nitroglycerin) C<br />
Premarin (Estrogens,<br />
Conjugated)<br />
C<br />
Fansidar<br />
(Pyrimethamine/Sulfa<br />
doxine)<br />
C<br />
Date:05/27/04ISR Number: 4367747-7Report Type:Periodic Company Report #348770 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Discomfort Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Insomnia Hydrochloride) PS ORAL ORAL<br />
Hallucination
Fatigue<br />
Dyspepsia<br />
Myalgia<br />
03-Apr-2012 09:37 AM Page: 549
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/27/04ISR Number: 4367748-9Report Type:Periodic Company Report #350183 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Health Lariam (<strong>Mefloquine</strong><br />
Fatigue Professional Hydrochloride) PS ORAL ORAL<br />
Convulsion<br />
Muscle Twitching<br />
Dizziness<br />
Adverse Event<br />
Date:06/01/04ISR Number: 4367871-9Report Type:Direct Company Report #CTU 219805 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Lariam 250 Mg Roche<br />
Disability Abnormal Behaviour Pharmaceuticals PS Roche<br />
Required Depression Pharmaceuticals ORAL ONE-1- PER<br />
Intervention to Fall DAY ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Vision Blurred<br />
Suicidal Ideation<br />
Amnesia<br />
Balance Disorder<br />
Date:06/08/04ISR Number: 4374191-5Report Type:Direct Company Report #CTU 220327 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Larium PS 2 TAB<br />
Vertigo<br />
Date:06/08/04ISR Number: 4374192-7Report Type:Direct Company Report #CTU 220326 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Larium PS 2 TAB<br />
Vertigo<br />
Date:06/08/04ISR Number: 4374195-2Report Type:Direct Company Report #CTU 220324 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Balance Disorder Larium PS 2 TAB<br />
Vertigo<br />
Date:06/08/04ISR Number: 4374199-XReport Type:Direct Company Report #CTU 220323 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Disability Balance Disorder Larium PS 2 TAB<br />
Vertigo<br />
03-Apr-2012 09:37 AM Page: 550
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/08/04ISR Number: 4374203-9Report Type:Direct Company Report #CTU 220322 Age:45 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Vertigo Larium PS 2 TAB<br />
Balance Disorder<br />
Date:06/08/04ISR Number: 4374205-2Report Type:Direct Company Report #CTU 220321 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Vertigo Larium PS 2 TABS<br />
Balance Disorder<br />
Date:06/09/04ISR Number: 4375988-8Report Type:Direct Company Report #CTU 220405 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Rash Larium-Mefloquin-<br />
Standard Pills PS ORAL 1 PILL DAILY<br />
ORAL<br />
Date:06/09/04ISR Number: 4376006-8Report Type:Direct Company Report #CTU 220398 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Aggression Lariam PS WEEKLY 2/03-><br />
Physical Assault 12/03<br />
Incoherent<br />
Psychotic Disorder<br />
Completed Suicide<br />
Date:06/15/04ISR Number: 4377670-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-369567 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Heat Stroke Lariam PS Roche ORAL<br />
Disorientation Omeprazole C ORAL<br />
Tremor Risperdal C ORAL<br />
Dehydration<br />
Cerebral Ventricle<br />
Dilatation<br />
Normal Pressure<br />
Hydrocephalus<br />
Date:06/21/04ISR Number: 4381191-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age: Gender:Male I/FU:F<br />
Outcome<br />
PT
Disability<br />
Hepatic Failure<br />
Cranial Nerve Disorder<br />
Balance Disorder<br />
Nausea<br />
Amnesia<br />
Incontinence<br />
Aggression<br />
Disturbance In Attention<br />
03-Apr-2012 09:37 AM Page: 551
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Photosensitivity Reaction<br />
Paranoia<br />
Visual Field Defect Report Source Product Role Manufacturer Route Dose Duration<br />
Depression Consumer Lariam PS Roche ORAL 23 TABLETS<br />
Photophobia<br />
DISPENSED.<br />
Panic Attack<br />
Suicidal Ideation<br />
Visual Impairment<br />
Vestibular Disorder<br />
Hallucination<br />
Insomnia<br />
Diarrhoea<br />
Anger<br />
Tremor<br />
Vomiting<br />
Toxicity To Various<br />
Agents<br />
Date:06/29/04ISR Number: 4386462-7Report Type:Expedited (15-DaCompany Report #CH-ROCHE-371304 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Lariam PS Roche ORAL 29 DAY<br />
Initial or Prolonged Urinary Incontinence<br />
Disability<br />
Asthenia<br />
Eosinophilia<br />
Date:06/29/04ISR Number: 4386466-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-371475 Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dementia Lariam PS Roche ORAL<br />
Date:07/07/04ISR Number: 4391114-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-372586 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Lariam PS Roche ORAL<br />
Disability<br />
Homicide<br />
Brain Injury<br />
Aggression<br />
Date:07/12/04ISR Number: 4394587-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-372726 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sarcoidosis Lariam PS Roche ORAL<br />
Initial or Prolonged Pulmonary Oedema<br />
Left Ventricular Failure
Myocarditis<br />
03-Apr-2012 09:37 AM Page: 552
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/13/04ISR Number: 4395807-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-371475 Age:58 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Health Lariam PS Roche ORAL<br />
Cognitive Disorder<br />
Professional<br />
Cerebral Disorder<br />
Delusion<br />
Dementia<br />
Date:08/02/04ISR Number: 4413078-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Suicidal Ideation<br />
Heart Rate Irregular<br />
Disorientation<br />
Balance Disorder<br />
Phobia<br />
Generalised Anxiety<br />
Disorder<br />
Abdominal Distension<br />
Constipation<br />
Anxiety<br />
Panic Attack<br />
Vestibular Disorder<br />
Fatigue<br />
Abdominal Discomfort<br />
House Dust Allergy<br />
Hyperventilation<br />
Depression<br />
Rash<br />
Dyspnoea<br />
Headache<br />
Irritable Bowel Syndrome<br />
Acrophobia<br />
Claustrophobia<br />
Migraine<br />
Heart Rate Increased<br />
Nausea<br />
Diarrhoea<br />
Hypersensitivity<br />
Tinnitus<br />
Self Esteem Decreased<br />
Photophobia<br />
Melanocytic Naevus<br />
Body Tinea<br />
Bronchospasm<br />
Emotional Distress<br />
Paraesthesia<br />
Phobia Of Flying<br />
Abdominal Pain<br />
Disturbance In Attention
Weight Decreased<br />
Dizziness<br />
Insomnia<br />
Motion Sickness<br />
Sensory Integrative<br />
Dysfunction<br />
Flatulence<br />
Lipoma<br />
03-Apr-2012 09:37 AM Page: 553
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Injury<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL THE PATIENT<br />
HAD TAKEN 4-5<br />
TABLETS OF<br />
MEFLOQUINE IN<br />
TOTAL. 5 WK<br />
Date:08/03/04ISR Number: 4414927-8Report Type:Direct Company Report #CTU 224121 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Convulsion <strong>Mefloquine</strong> 250 Mg<br />
Hospitalization - Bradycardia Geneva Pharm Tech /<br />
Initial or Prolonged Atrioventricular Block Sandoz PS Geneva Pharm Tech /<br />
Required Complete Sandoz ORAL 1 PILL WEEK<br />
Intervention to<br />
ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:08/05/04ISR Number: 4416339-XReport Type:Expedited (15-DaCompany Report #CH-ROCHE-371304 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Urinary Incontinence Lariam PS Roche ORAL 29 DAY<br />
Initial or Prolonged Fatigue<br />
Disability<br />
Eosinophilia<br />
Asthenia<br />
Date:08/10/04ISR Number: 4422188-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-376379 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Syndrome Lariam PS Roche ORAL 60 DAY<br />
Initial or Prolonged Coordination Abnormal<br />
Date:08/10/04ISR Number: 4422192-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-319597 Age:36 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Lariam PS Roche ORAL TAKEN WEEKLY 99 DAY<br />
Anger<br />
Social Avoidant Behaviour<br />
Communication Disorder<br />
Tremor<br />
Mood Altered<br />
Disorientation<br />
Nervousness
Feeling Abnormal<br />
Psychotic Disorder<br />
Aggression<br />
Abnormal Behaviour<br />
03-Apr-2012 09:37 AM Page: 554
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/10/04ISR Number: 4424148-0Report Type:Direct Company Report #CTU 224675 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Lariam 250 Mg PS 1 A WK<br />
<strong>Mefloquine</strong> (Hci)<br />
Roche Labs SS Roche Labs<br />
Date:08/16/04ISR Number: 4426723-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-308521 Age:12 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Lariam PS Roche ORAL 23 DAY<br />
Initial or Prolonged Vomiting<br />
Malaise<br />
Sleep Disorder<br />
Muscle Twitching<br />
Headache<br />
Abdominal Pain<br />
Balance Disorder<br />
Date:08/16/04ISR Number: 4426725-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-369567 Age:54 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Cerebral Ventricle Health Lariam PS Roche ORAL 21 DAY<br />
Dilatation Professional Omeprazole C ORAL<br />
Fatigue L-Thyroxine C ORAL<br />
Disorientation Risperdal C ORAL INCREASED TO<br />
Affect Lability<br />
1 MG DAILY<br />
Heat Stroke FROM 07-15<br />
Tremor MAY 2004.<br />
Hydrocephalus<br />
Dehydration<br />
Date:08/20/04ISR Number: 4429680-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377653 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Brain Oedema Lariam PS Roche UNKNOWN<br />
Initial or Prolonged<br />
Date:08/20/04ISR Number: 4430060-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-377087 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anger Lariam PS Roche ORAL<br />
Homicide
Date:08/20/04ISR Number: 4430095-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-377090 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Lariam PS Roche ORAL<br />
Anger<br />
03-Apr-2012 09:37 AM Page: 555
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/20/04ISR Number: 4430108-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-377096 Age:33 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anger Lariam PS Roche ORAL<br />
Homicide<br />
Date:08/23/04ISR Number: 4430901-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-377100 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Injury Lariam PS Roche ORAL<br />
Anger<br />
Physical Assault<br />
Date:08/23/04ISR Number: 4430909-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377775 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Macular Degeneration Lariam PS Roche ORAL<br />
Initial or Prolonged Befizal C ORAL<br />
Date:08/27/04ISR Number: 4435139-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Sleep Disorder Due To<br />
General Medical<br />
Condition, Hypersomnia<br />
Type<br />
Suicidal Ideation<br />
Disorientation<br />
Headache<br />
Weight Decreased<br />
Fear<br />
Injury<br />
Nausea<br />
Balance Disorder<br />
Vestibular Disorder<br />
Cold Sweat<br />
Body Tinea<br />
Emotional Distress<br />
Anxiety<br />
Abdominal Pain<br />
Hypersensitivity<br />
Self Esteem Decreased<br />
Disturbance In Attention<br />
Photophobia<br />
Gastrointestinal Pain<br />
Adjustment Disorder With<br />
Anxiety
Motion Sickness<br />
Diarrhoea<br />
Paraesthesia<br />
Dyspnoea<br />
Bronchospasm<br />
Dizziness<br />
Heart Rate Irregular<br />
Acrophobia<br />
03-Apr-2012 09:37 AM Page: 556
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Claustrophobia<br />
Adjustment Disorder<br />
Self Esteem Inflated Report Source Product Role Manufacturer Route Dose Duration<br />
Impetigo Health Lariam PS Roche ORAL THE PATIENT<br />
Adjustment Disorder With Professional HAD TAKEN 4-5<br />
Depressed Mood<br />
TABLETS OF<br />
Insomnia<br />
MEFLOQUINE IN<br />
Panic Attack TOTAL. 5 WK<br />
Tinnitus<br />
Phobia<br />
Rash<br />
Irritable Bowel Syndrome<br />
Generalised Anxiety<br />
Disorder<br />
Sensory Integrative<br />
Dysfunction<br />
Fatigue<br />
Sleep Disorder<br />
Melanocytic Naevus<br />
Lipoma<br />
Mood Disorder Due To A<br />
General Medical Condition<br />
Heart Rate Increased<br />
Depression<br />
Phobia Of Flying<br />
Migraine<br />
Lactose Intolerance<br />
Cognitive Disorder<br />
Date:08/27/04ISR Number: 4435146-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-369567 Age:54 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Fatigue Health Lariam PS Roche ORAL 21 DAY<br />
Dehydration Professional Omeprazole C ORAL<br />
Tremor L-Thyroxine C ORAL<br />
Hydrocephalus Risperdal C ORAL INCREASED TO<br />
Affect Lability<br />
1 MG DAILY<br />
Disorientation FROM 07-15<br />
Cerebral Ventricle MAY 2004.<br />
Dilatation<br />
Date:09/02/04ISR Number: 4441050-9Report Type:Direct Company Report #CTU 226240 Age: Gender:Female I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Disability<br />
PT<br />
Nausea<br />
Fear<br />
Thinking Abnormal<br />
Blood Pressure Decreased<br />
Suicidal Ideation
Dizziness<br />
Sleep Terror<br />
Heart Rate Increased<br />
Stress<br />
Fatigue<br />
Nightmare<br />
Decreased Appetite<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 557
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Panic Reaction<br />
Chest Pain<br />
Hypoaesthesia Report Source Product Role Manufacturer Route Dose Duration<br />
Carpal Tunnel Syndrome Lariam PS 250 MG ONCE<br />
Cold Sweat WEEKLY 2<br />
Anxiety<br />
DOSES<br />
Dyspnoea Antibiotic C<br />
Feeling Abnormal<br />
Confusional State<br />
Vertigo<br />
Date:09/03/04ISR Number: 4443505-XReport Type:Direct Company Report #CTU 226320 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Feeling Abnormal Lariam PS ORAL ONE WEEK ORAL<br />
Intervention to<br />
Tremor<br />
Prevent Permanent Nightmare<br />
Impairment/Damage Dizziness<br />
Disorientation<br />
Date:09/07/04ISR Number: 4441862-1Report Type:Expedited (15-DaCompany Report #CA-ROCHE-378987 Age:73 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Disorientation Lariam PS Roche ORAL TAKEN<br />
Malaise<br />
INTERMITTENTL<br />
Vomiting<br />
Y<br />
Cardiac Failure<br />
Diarrhoea<br />
Headache<br />
Asthenia<br />
Dyspnoea<br />
Date:09/08/04ISR Number: 4447144-6Report Type:Direct Company Report #CTU 226602 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Hallucination <strong>Mefloquine</strong> PS ORAL PO 250 MG QWK<br />
Social Avoidant Behaviour<br />
Fall<br />
Date:09/08/04ISR Number: 4447208-7Report Type:Direct Company Report #CTU 226584 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicide Attempt Lariam PS ORAL ONCE A WEEK<br />
Depression<br />
ORAL
Date:09/09/04ISR Number: 4444958-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-379100 Age: Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Insomnia<br />
Eating Disorder<br />
Schizophrenia, Paranoid<br />
03-Apr-2012 09:37 AM Page: 558
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Type<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Date:09/09/04ISR Number: 4444960-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377653 Age:22 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Health Lariam PS Roche ORAL 15 DAY<br />
Initial or Prolonged Brain Oedema Professional<br />
Headache<br />
Vomiting<br />
Vertigo<br />
Date:09/09/04ISR Number: 4448491-4Report Type:Direct Company Report #CTU 226761 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Larium PS ORAL 1 A DAY ORAL<br />
Required<br />
Feeling Abnormal<br />
Intervention to<br />
Insomnia<br />
Prevent Permanent Headache<br />
Impairment/Damage Suicidal Ideation<br />
Anxiety<br />
Anger<br />
Date:09/13/04ISR Number: 4449499-5Report Type:Direct Company Report #CTU 226923 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia Lariam PS ORAL TWICE A WEEK<br />
Hospitalization - Anxiety ORAL<br />
Initial or Prolonged Hypertension Doxycycline C<br />
Nightmare<br />
Panic Attack<br />
Suicidal Ideation<br />
Tinnitus<br />
Date:09/14/04ISR Number: 4449511-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377992 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Lactate Lariam PS Roche UNKNOWN 4 DAY<br />
Initial or Prolonged Dehydrogenase Increased<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Blood Creatine
Phosphokinase Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Confusional State<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 559
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/15/04ISR Number: 4449981-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377992 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delirium Health Lariam PS Roche UNKNOWN 4 DAY<br />
Initial or Prolonged Confusional State Professional<br />
Nausea<br />
Alanine Aminotransferase<br />
Increased<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Date:09/15/04ISR Number: 4450070-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-377992 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State Lariam PS Roche UNKNOWN 4 DAY<br />
Initial or Prolonged Blood Lactate<br />
Dehydrogenase Increased<br />
Delirium<br />
Pyrexia<br />
Alanine Aminotransferase<br />
Increased<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Gastroenteritis<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Nausea<br />
Date:09/16/04ISR Number: 4451065-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377992 Age:42 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Lactate Health Lariam PS Roche UNKNOWN 4 DAY<br />
Initial or Prolonged Dehydrogenase Increased Professional<br />
Alanine Aminotransferase<br />
Increased<br />
Confusional State<br />
Aspartate<br />
Aminotransferase<br />
Increased<br />
Gastroenteritis<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Delirium
Date:09/16/04ISR Number: 4451425-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-377992 Age:42 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Nausea<br />
Delirium<br />
03-Apr-2012 09:37 AM Page: 560
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Aspartate<br />
Aminotransferase<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Confusional State Health Lariam PS Roche UNKNOWN 4 DAY<br />
Blood Creatine<br />
Professional<br />
Phosphokinase Increased<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Alanine Aminotransferase<br />
Increased<br />
Date:09/16/04ISR Number: 4451447-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-380221 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychiatric Symptom Lariam PS Roche ORAL<br />
Brain Stem Syndrome<br />
Suicidal Ideation<br />
Date:09/17/04ISR Number: 4452279-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Bronchospasm<br />
Dizziness<br />
Heart Rate Irregular<br />
Rash<br />
Disturbance In Attention<br />
Headache<br />
Acrophobia<br />
Lactose Intolerance<br />
Burning Sensation<br />
Anxiety<br />
Heart Rate Increased<br />
Motion Sickness<br />
Hypersensitivity<br />
Phobia Of Flying<br />
Claustrophobia<br />
Migraine<br />
Back Pain<br />
Nasal Congestion<br />
Injury<br />
Sleep Disorder Due To<br />
General Medical<br />
Condition, Hypersomnia<br />
Type<br />
Insomnia<br />
Panic Attack<br />
Amnesia<br />
Body Tinea<br />
Emotional Distress
Paraesthesia<br />
Irritable Bowel Syndrome<br />
Weight Decreased<br />
Haemorrhoids<br />
Vitreous Floaters<br />
Mood Disorder Due To A<br />
General Medical Condition<br />
Disorientation<br />
03-Apr-2012 09:37 AM Page: 561
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Vestibular Disorder<br />
Throat Irritation<br />
Hypoaesthesia Report Source Product Role Manufacturer Route Dose Duration<br />
Constipation Lariam PS Roche ORAL THE PATIENT<br />
Cold Sweat HAD TAKEN 4-5<br />
Impetigo<br />
TABLETS OF<br />
Lipoma<br />
MEFLOQUINE IN<br />
Hyperventilation TOTAL. 5 WK<br />
Adjustment Disorder With Vaccine C OTHER 1 DAY<br />
Depressed Mood<br />
Nausea<br />
Dyspnoea<br />
Self Esteem Decreased<br />
Photophobia<br />
Flatulence<br />
Adjustment Disorder With<br />
Anxiety<br />
Suicidal Ideation<br />
Tinnitus<br />
Balance Disorder<br />
Phobia<br />
Generalised Anxiety<br />
Disorder<br />
Sensory Integrative<br />
Dysfunction<br />
Fatigue<br />
Musculoskeletal Stiffness<br />
Abdominal Distension<br />
Abdominal Pain Lower<br />
Gait Disturbance<br />
Abdominal Discomfort<br />
Abdominal Symptom<br />
Sinus Bradycardia<br />
Abdominal Pain<br />
Diarrhoea<br />
Depression<br />
Sleep Disorder<br />
Neck Pain<br />
Condition Aggravated<br />
Abdominal Pain Upper<br />
Melanocytic Naevus<br />
Panic Disorder<br />
Date:09/17/04ISR Number: 4452299-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-376379 Age:37 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Syndrome Lariam PS Roche ORAL 60 DAY<br />
Initial or Prolonged Coordination Abnormal
Date:09/17/04ISR Number: 4452312-3Report Type:Expedited (15-DaCompany Report #IE-ROCHE-379920 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Lariam PS Roche ORAL<br />
Dyskinesia<br />
Grand Mal Convulsion<br />
Muscle Twitching<br />
03-Apr-2012 09:37 AM Page: 562
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/20/04ISR Number: 4453827-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-377423 Age:63 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cardiac Failure Lariam PS Roche ORAL<br />
Initial or Prolonged Electrocardiogram Toprol Xl C<br />
Abnormal<br />
Cerebrovascular Accident<br />
Date:09/20/04ISR Number: 4453925-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-377423 Age:63 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Electrocardiogram Health Lariam PS Roche ORAL<br />
Initial or Prolonged Abnormal Professional Toprol Xl C<br />
Cerebrovascular Accident<br />
Cardiac Failure<br />
Date:09/20/04ISR Number: 4454571-XReport Type:Direct Company Report #CTU 227503 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Fatigue Lariam (<strong>Mefloquine</strong><br />
Intervention to Headache Hydrochloride) PS Roche 1 TABLET PER<br />
Prevent Permanent Insomnia WEEK FOR 7<br />
Impairment/Damage Anxiety WEEKS<br />
Feeling Of Body<br />
Temperature Change<br />
Disturbance In Attention<br />
Paraesthesia<br />
Paranoia<br />
Balance Disorder<br />
Heart Rate Irregular<br />
Hyperhidrosis<br />
Chills<br />
Depression<br />
Panic Reaction<br />
Alanine Aminotransferase<br />
Increased<br />
Date:09/21/04ISR Number: 4454933-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Mitral Valve Prolapse<br />
Arthralgia<br />
Diarrhoea<br />
Dry Eye<br />
Constipation<br />
Upper-Airway Cough<br />
Syndrome
Rhinitis<br />
Visual Impairment<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Schizoaffective Disorder<br />
Confusional State<br />
Angina Pectoris<br />
Sleep Apnoea Syndrome<br />
03-Apr-2012 09:37 AM Page: 563
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Rash<br />
Bronchitis<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Dry Skin<br />
Blood Glucose Increased<br />
Scratch<br />
Headache<br />
Weight Increased<br />
Cardiac Murmur<br />
Abnormal Dreams<br />
Skin Disorder<br />
Fatigue<br />
Hypertension<br />
Bronchospasm<br />
Nocturia<br />
Vitiligo<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Gastrooesophageal Reflux<br />
Disease<br />
Asthma<br />
Affective Disorder<br />
Depression<br />
Sinus Tachycardia<br />
Blood Triglycerides<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Dehydration<br />
Dry Mouth<br />
Chest Discomfort<br />
Balance Disorder<br />
Amnesia<br />
Diastolic Dysfunction<br />
Somnolence<br />
Blood Cholesterol<br />
Increased<br />
Exfoliative Rash<br />
Snoring<br />
Acrochordon<br />
Major Depression<br />
Psychotic Disorder<br />
Ventricular Hypertrophy<br />
Viral Infection<br />
Oral Disorder<br />
Insomnia<br />
Bipolar Disorder<br />
Haemorrhoids<br />
Coordination Abnormal<br />
Memory Impairment
Anxiety<br />
Dyspnoea<br />
Vestibular Disorder<br />
Gastrointestinal Disorder<br />
Total Lung Capacity<br />
Decreased<br />
Restrictive Pulmonary<br />
Disease<br />
03-Apr-2012 09:37 AM Page: 564
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Clumsiness<br />
Contusion<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Zoloft C UNKNOWN<br />
Date:09/22/04ISR Number: 4459307-4Report Type:Direct Company Report #CTU 227806 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Paranoia <strong>Mefloquine</strong> PS ORAL 250 MG Q7D<br />
Intervention to Toxicity To Various ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Agents<br />
Vertigo<br />
Tremor<br />
Vision Blurred<br />
Headache<br />
Medication Error<br />
Date:09/29/04ISR Number: 4462210-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-380927 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache <strong>Mefloquine</strong><br />
Neurotoxicity Hydrochloride PS Roche ORAL SINGLE DOSE<br />
Diplopia<br />
TAKEN.<br />
Beclomethasone C RESPIRATORY<br />
(INHALATION)<br />
Typhim Vi C INTRAMUSCULAR 1 DAY<br />
Date:09/30/04ISR Number: 4463201-2Report Type:Expedited (15-DaCompany Report #IE-ROCHE-379920 Age:25 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Twitching Health Lariam PS Roche ORAL<br />
Grand Mal Convulsion<br />
Professional<br />
Visual Impairment<br />
Dyskinesia<br />
Fall<br />
Date:10/01/04ISR Number: 4464169-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-381273 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Disorder Lariam PS Roche ORAL 2 DAY<br />
Initial or Prolonged Diarrhoea Aspirine C ORAL<br />
Confusional State<br />
Psychotic Disorder<br />
Disorientation
Delirium<br />
03-Apr-2012 09:37 AM Page: 565
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/04/04ISR Number: 4465047-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-381273 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Disorder Lariam PS Roche ORAL 2 DAY<br />
Initial or Prolonged Fatigue Aspirine C ORAL<br />
Diarrhoea<br />
Psychotic Disorder<br />
Delirium<br />
Disorientation<br />
Confusional State<br />
Date:10/08/04ISR Number: 4469780-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-381674 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Foetal Malformation Lariam PS Roche UNKNOWN<br />
Abortion Induced<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:10/08/04ISR Number: 4469794-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-382257 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Complex Regional Pain Lariam PS Roche ORAL<br />
Syndrome Lariam SS Roche ORAL<br />
Date:10/12/04ISR Number: 4471713-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Bronchospasm<br />
Gastrooesophageal Reflux<br />
Disease<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Depression<br />
Bronchitis<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Snoring<br />
Nocturia<br />
Clumsiness<br />
Tremor<br />
Confusional State<br />
Dyspnoea<br />
Headache<br />
Rash<br />
Blood Cholesterol<br />
Increased
Blood Triglycerides<br />
Increased<br />
Chest Discomfort<br />
Oral Disorder<br />
Insomnia<br />
Abnormal Dreams<br />
Coordination Abnormal<br />
Amnesia<br />
03-Apr-2012 09:37 AM Page: 566
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diastolic Dysfunction<br />
Arthralgia<br />
Sinus Tachycardia Report Source Product Role Manufacturer Route Dose Duration<br />
Low Density Lipoprotein Consumer Lariam PS Roche ORAL<br />
Increased Zoloft C UNKNOWN<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Vitiligo<br />
Bipolar Disorder<br />
Contusion<br />
Aggression<br />
Anxiety<br />
Skin Disorder<br />
Gastrointestinal Disorder<br />
Angina Pectoris<br />
Sleep Apnoea Syndrome<br />
Dry Mouth<br />
Constipation<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Major Depression<br />
Affective Disorder<br />
Psychotic Disorder<br />
Hypertension<br />
Viral Infection<br />
Dry Eye<br />
Acrochordon<br />
Upper-Airway Cough<br />
Syndrome<br />
Scratch<br />
Vestibular Disorder<br />
Mitral Valve Prolapse<br />
Restrictive Pulmonary<br />
Disease<br />
Somnolence<br />
Weight Increased<br />
Cardiac Murmur<br />
Dehydration<br />
Exfoliative Rash<br />
Dry Skin<br />
Blood Glucose Increased<br />
Balance Disorder<br />
Schizoaffective Disorder<br />
Fatigue<br />
Ventricular Hypertrophy<br />
Total Lung Capacity<br />
Decreased<br />
Diarrhoea<br />
Rhinitis<br />
Visual Impairment<br />
Asthma<br />
Haemorrhoids<br />
Memory Impairment
Date:10/12/04ISR Number: 4471726-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-377775 Age:49 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Macular Degeneration Health Lariam PS Roche ORAL 4 MON<br />
Professional Befizal C ORAL<br />
03-Apr-2012 09:37 AM Page: 567
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/12/04ISR Number: 4471734-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-382418 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Upper Lariam PS Roche UNKNOWN<br />
Nausea<br />
Full Blood Count Abnormal<br />
Oropharyngeal Pain<br />
Lymphocyte Count<br />
Increased<br />
Fatigue<br />
Date:10/12/04ISR Number: 4472797-6Report Type:Direct Company Report #CTU 229406 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Palpitations Lariam 750mg<br />
Tremor <strong>Mefloquine</strong> PS ORAL 1 WEEK ORAL<br />
Date:10/15/04ISR Number: 4475448-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-382481 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Balance Disorder Lariam PS Roche ORAL<br />
Anxiety<br />
Malaria<br />
Hallucination, Auditory<br />
Psychotic Disorder<br />
Homicide<br />
Hallucination<br />
Date:10/19/04ISR Number: 4478306-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-383127 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Toxicity To Various Lariam PS Roche ORAL<br />
Agents<br />
Aphasia<br />
Date:10/19/04ISR Number: 4480777-XReport Type:Direct Company Report #CTU 229949 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Medication Error <strong>Mefloquine</strong> (Lariam)<br />
- 250 Mgs -Sandoz PS Sandoz 1 DAILY W/<br />
SUPPER - X 23<br />
Date:10/26/04ISR Number: 4486473-7Report Type:Direct Company Report #CTU 230455 Age: Gender:Female I/FU:I
Outcome<br />
Disability<br />
PT<br />
Asthenia<br />
Dyspnoea<br />
Flatulence<br />
Eructation<br />
Chest Pain<br />
Morbid Thoughts<br />
03-Apr-2012 09:37 AM Page: 568
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Palpitations<br />
Nausea<br />
Dyspepsia Report Source Product Role Manufacturer Route Dose Duration<br />
Dizziness Larium 250 Mg La<br />
Tremor Roche PS La Roche ORAL 1 TABLET<br />
Condition Aggravated<br />
ONCE A WEEK<br />
Panic Attack<br />
ORAL<br />
Eye Disorder Protonix C<br />
Hiatus Hernia<br />
Feeling Abnormal<br />
Date:10/27/04ISR Number: 4487799-3Report Type:Direct Company Report #CTU 230523 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety Lariam PS<br />
Hospitalization - Depression<br />
Initial or Prolonged Convulsion<br />
Mental Disorder<br />
Date:10/28/04ISR Number: 4488116-5Report Type:Expedited (15-DaCompany Report #IE-ROCHE-379920 Age:25 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Muscle Twitching Health Lariam PS Roche ORAL<br />
Visual Impairment<br />
Professional<br />
Grand Mal Convulsion<br />
Date:10/28/04ISR Number: 4491873-5Report Type:Direct Company Report #CTU 230702 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia Larium One Tablet<br />
Impaired Work Ability Per Week Roche PS Roche ORAL 1 TABLET<br />
Headache WEEK ORAL<br />
Dizziness<br />
Depression<br />
Vision Blurred<br />
Suicidal Ideation<br />
Date:10/29/04ISR Number: 4489101-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-384026 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Lariam PS Roche INTRAMUSCULAR<br />
Medication Error
Date:11/02/04ISR Number: 4490702-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-372726 Age:32 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Sarcoidosis<br />
Pulmonary Oedema<br />
Pericardial Disease<br />
Myocarditis<br />
Myocardial Infarction<br />
03-Apr-2012 09:37 AM Page: 569
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Left Ventricular Failure<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL 41 DAY<br />
Date:11/04/04ISR Number: 4494141-0Report Type:Direct Company Report #CTU 231293 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Mood Swings Lariam Probably 250<br />
Intervention to Fatigue Mg Tablet PS ORAL 250 MG WEEKLY<br />
Prevent Permanent Feeling Abnormal ORAL<br />
Impairment/Damage Tension<br />
Crying<br />
Impaired Work Ability<br />
Depression<br />
Hypersomnia<br />
Date:11/04/04ISR Number: 4494371-8Report Type:Direct Company Report #CTU 231346 Age:14 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Aphasia <strong>Mefloquine</strong> (Lariam) PS ORAL 250 MG PO<br />
Other Serious Catatonia EVERY WEEK<br />
Required<br />
Toxicity To Various<br />
Intervention to<br />
Agents<br />
Prevent Permanent<br />
Impairment/Damage<br />
Panic Attack<br />
Amnesia<br />
Thinking Abnormal<br />
Anxiety<br />
Psychotic Disorder<br />
Antisocial Behaviour<br />
Pyrexia<br />
Date:11/11/04ISR Number: 4499163-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-381674 Age:31 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Maternal Exposure During Lariam PS Roche UNKNOWN<br />
Pregnancy<br />
Abortion Induced<br />
Foetal Malformation<br />
Date:11/12/04ISR Number: 4500146-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age: Gender:Male I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Insomnia<br />
Mood Swings
Balance Disorder<br />
Toxicity To Various<br />
Agents<br />
Aggression<br />
Abnormal Behaviour<br />
Agitation<br />
Disturbance In Attention<br />
Memory Impairment<br />
03-Apr-2012 09:37 AM Page: 570
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Suicidal Ideation<br />
Coordination Abnormal<br />
Visual Impairment Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination, Visual Lariam PS Roche ORAL<br />
Depression<br />
Anxiety<br />
Vertigo<br />
Date:11/15/04ISR Number: 4501752-2Report Type:Direct Company Report #CTU 232000 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Coordination Abnormal Lariam - <strong>Mefloquine</strong><br />
Disability Weight Decreased - 1 Tablet 250mg<br />
Required Paranoia Roche PS Roche ORAL 250MG ONCE<br />
Intervention to Self Esteem Decreased WEEKLY ORAL<br />
Prevent Permanent<br />
Impairment/Damage<br />
Hypoaesthesia<br />
Tinnitus<br />
Nausea<br />
Panic Attack<br />
Thinking Abnormal<br />
Decreased Interest<br />
Erectile Dysfunction<br />
Fear<br />
Headache<br />
Tremor<br />
Pain<br />
Disturbance In Attention<br />
Loss Of Libido<br />
Suicidal Ideation<br />
Muscle Twitching<br />
Decreased Appetite<br />
Hyperhidrosis<br />
Amnesia<br />
Feeling Abnormal<br />
Sensory Disturbance<br />
Major Depression<br />
Temperature Regulation<br />
Disorder<br />
Nightmare<br />
Functional<br />
Gastrointestinal Disorder<br />
Chills<br />
Autism<br />
Emotional Disorder<br />
Date:11/16/04ISR Number: 4502163-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-361839 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Exposure During Lariam PS Roche ORAL
Pregnancy<br />
Ectopic Pregnancy<br />
Abortion Spontaneous<br />
03-Apr-2012 09:37 AM Page: 571
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/19/04ISR Number: 4512858-6Report Type:Direct Company Report #CTU 232457 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tremor Larium 250 Mg Tabs PS 1 Q WEEK<br />
Palpitations<br />
Feeling Abnormal<br />
Feeling Jittery<br />
Anxiety<br />
Date:11/22/04ISR Number: 4506772-XReport Type:Expedited (15-DaCompany Report #GB-ROCHE-385961 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - General Physical Health Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Deterioration<br />
Joint Dislocation<br />
Balance Disorder<br />
Upper Limb Fracture<br />
Fall<br />
Decreased Appetite<br />
Insomnia<br />
Date:11/23/04ISR Number: 4507897-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-377423 Age:63 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebrovascular Accident Lariam PS Roche ORAL<br />
Initial or Prolonged Electrocardiogram Toprol Xl C<br />
Abnormal<br />
Cardiac Failure<br />
Date:11/23/04ISR Number: 4507908-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-386256 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Photosensitivity Reaction Lariam PS Roche ORAL<br />
Initial or Prolonged Malaise<br />
Feeling Abnormal<br />
Suicidal Ideation<br />
Depression<br />
Date:11/29/04ISR Number: 4512071-2Report Type:Expedited (15-DaCompany Report #DE-ROCHE-382481 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide Lariam PS Roche ORAL TAKEN WEEKLY. 316 DAY<br />
Malaria<br />
Psychotic Disorder<br />
Anxiety
Hallucination, Auditory<br />
Hallucination<br />
Balance Disorder<br />
03-Apr-2012 09:37 AM Page: 572
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/29/04ISR Number: 4512170-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-386651 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Blindness Lariam PS Roche ORAL<br />
Date:11/29/04ISR Number: 4512180-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-386791 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Retinopathy Lariam PS Roche ORAL 1 MON<br />
Initial or Prolonged Lariam SS Roche ORAL 1 MON<br />
Lariam SS Roche ORAL UNKNOWN<br />
DURATION.<br />
Lariam SS Roche ORAL 1 MON<br />
Date:12/08/04ISR Number: 4521121-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-386256 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Toxicity To Various Lariam PS Roche ORAL<br />
Initial or Prolonged Agents<br />
Suicidal Ideation<br />
Depression<br />
Feeling Abnormal<br />
Anxiety<br />
Photosensitivity Reaction<br />
Malaise<br />
Fear<br />
Negative Thoughts<br />
Date:12/13/04ISR Number: 4525226-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Tremor<br />
Vomiting<br />
Vestibular Disorder<br />
Tinnitus<br />
Muscle Twitching<br />
Photophobia<br />
Visual Field Defect<br />
Nausea<br />
Aggression<br />
Disturbance In Attention<br />
Anxiety<br />
Insomnia<br />
Jaundice<br />
Hallucination<br />
Depression<br />
Amnesia
Photosensitivity Reaction<br />
Toxicity To Various<br />
Agents<br />
Irritability<br />
Endolymphatic Hydrops<br />
Hepatic Failure<br />
Cranial Nerve Disorder<br />
Visual Impairment<br />
03-Apr-2012 09:37 AM Page: 573
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Diarrhoea<br />
Suicidal Ideation<br />
Asthenia Report Source Product Role Manufacturer Route Dose Duration<br />
Obsessive Thoughts Lariam PS Roche ORAL 23 TABLETS<br />
Depressed Level Of<br />
DISPENSED.<br />
Consciousness Sudafed C<br />
Paranoia<br />
Incontinence<br />
General Symptom<br />
Cough<br />
Mood Swings<br />
Balance Disorder<br />
Panic Attack<br />
Anger<br />
Post-Traumatic Stress<br />
Disorder<br />
Migraine<br />
Date:12/15/04ISR Number: 4530035-XReport Type:Direct Company Report #CTU 234381 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia <strong>Mefloquine</strong> 250mg<br />
Disability Panic Attack Sandoz PS Sandoz ORAL 250MG PER<br />
Anxiety WEEK ORAL<br />
Hallucination<br />
Suicidal Ideation<br />
Night Sweats<br />
Depression<br />
Delusion<br />
Date:12/22/04ISR Number: 4535059-4Report Type:Expedited (15-DaCompany Report #SE-ROCHE-388966 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged Tardive Dyskinesia Zyprexa SS ORAL<br />
Date:12/23/04ISR Number: 4536778-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-384854 Age:37 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Insomnia<br />
Paraesthesia<br />
Skin Disorder<br />
Hyperhidrosis<br />
Fatigue<br />
Disorientation<br />
Liver Function Test<br />
Abnormal
Disturbance In Attention<br />
Agitation<br />
Vertigo<br />
Diarrhoea<br />
Paranoia<br />
Chills<br />
Back Pain<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 574
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Panic Attack<br />
Depression<br />
Heart Rate Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Heart Rate Irregular Lariam PS Roche ORAL 3-4 PILLS<br />
Mood Swings WERE TAKEN. 43 DAY<br />
Balance Disorder<br />
Date:01/03/05ISR Number: 4544470-7Report Type:Direct Company Report #CTU 235515 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Headache Lariam 250 Mg PS ORAL 1 PILL WEEK<br />
Disability Hallucination ORAL<br />
Balance Disorder<br />
Disturbance In Attention<br />
Sleep Disorder<br />
Impaired Work Ability<br />
Dizziness<br />
Confusional State<br />
Memory Impairment<br />
Homicidal Ideation<br />
Thinking Abnormal<br />
Mania<br />
Motion Sickness<br />
Suicidal Ideation<br />
Heart Rate Irregular<br />
Depression<br />
Nightmare<br />
Panic Attack<br />
Night Sweats<br />
Self Esteem Decreased<br />
Date:01/05/05ISR Number: 4544789-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Diarrhoea<br />
Hepatic Failure<br />
Nausea<br />
Disturbance In Attention<br />
Photosensitivity Reaction<br />
Jaundice<br />
Cranial Nerve Disorder<br />
Visual Field Defect<br />
Insomnia<br />
General Symptom<br />
Obsessive Thoughts<br />
Depressed Level Of<br />
Consciousness<br />
Post-Traumatic Stress<br />
Disorder
Migraine<br />
Paranoia<br />
Depression<br />
Amnesia<br />
Vestibular Disorder<br />
Anxiety<br />
Tinnitus<br />
Irritability<br />
03-Apr-2012 09:37 AM Page: 575
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Muscle Twitching<br />
Photophobia<br />
Tremor Report Source Product Role Manufacturer Route Dose Duration<br />
Vomiting Lariam PS Roche ORAL 23 TABLETS<br />
Anger<br />
DISPENSED.<br />
Asthenia Sudafed C<br />
Endolymphatic Hydrops<br />
Panic Attack<br />
Aggression<br />
Mood Swings<br />
Hallucination<br />
Balance Disorder<br />
Incontinence<br />
Toxicity To Various<br />
Agents<br />
Visual Impairment<br />
Suicidal Ideation<br />
Cough<br />
Date:01/06/05ISR Number: 4546225-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-390293 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pneumonitis Lariam PS Roche UNKNOWN<br />
Date:01/07/05ISR Number: 4547388-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-386651 Age:23 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Blindness Lariam PS Roche ORAL<br />
Date:01/07/05ISR Number: 4547393-2Report Type:Expedited (15-DaCompany Report #KR-ROCHE-389876 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatitis Acute Lariam PS Roche ORAL DOSAGE<br />
Initial or Prolonged<br />
REGIMEN:<br />
1T/WEEK<br />
Lariam SS Roche ORAL DOSAGE<br />
REGIMEN:<br />
1T/WEEK<br />
Date:01/10/05ISR Number: 4549486-2Report Type:Direct Company Report #CTU 236012 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Multiple Allergies Lariam 250 Mg Roche PS Roche ORAL 1 WEEK ORAL<br />
Hallucination<br />
Nightmare
Headache<br />
Diarrhoea<br />
03-Apr-2012 09:37 AM Page: 576
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/11/05ISR Number: 4549536-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-390682 Age:59 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Panic Attack Lariam PS Roche UNKNOWN<br />
Hospitalization - Anxiety<br />
Initial or Prolonged Hyperhidrosis<br />
Atrial Fibrillation<br />
Heart Rate Increased<br />
Fluid Retention<br />
Palpitations<br />
Date:01/12/05ISR Number: 4550163-2Report Type:Expedited (15-DaCompany Report #KR-ROCHE-389876 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Product Quality Issue Consumer Lariam PS Roche ORAL DOSAGE<br />
Initial or Prolonged Hepatitis Acute REGIMEN:<br />
1T/WEEK<br />
Lariam SS Roche ORAL DOSAGE<br />
REGIMEN:<br />
1T/WEEK<br />
Date:01/14/05ISR Number: 4552774-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-319597 Age:33 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Disorientation Lariam PS Roche ORAL TAKEN WEEKLY 99 DAY<br />
Abnormal Behaviour Primaquine Phosphate C ORAL 15 DAY<br />
Social Avoidant Behaviour<br />
Psychotic Disorder<br />
Feeling Abnormal<br />
Anger<br />
Communication Disorder<br />
Tremor<br />
Mood Altered<br />
Completed Suicide<br />
Aggression<br />
Nervousness<br />
Date:01/14/05ISR Number: 4552794-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-391105 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Oligohydramnios Lariam PS Roche UNKNOWN 38 DAY<br />
Initial or Prolonged Abortion Induced Lysine<br />
Congenital Anomaly Premature Rupture Of Acetylsalicylate SS UNKNOWN 36 DAY<br />
Membranes<br />
Talipes<br />
Maternal Exposure During<br />
Pregnancy
Date:01/14/05ISR Number: 4552795-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-391660 Age: Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
03-Apr-2012 09:37 AM Page: 577
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disability<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Self Esteem Decreased Lariam PS Roche UNKNOWN<br />
Depression<br />
Anxiety Disorder<br />
Panic Attack<br />
Mood Swings<br />
Memory Impairment<br />
Psychotic Disorder<br />
Suicidal Ideation<br />
Amnesia<br />
Mental Disorder<br />
Date:01/14/05ISR Number: 4553803-7Report Type:Direct Company Report #CTU 236752 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare <strong>Mefloquine</strong> (Larium) PS 1 TIME A WEEK 10 WK<br />
Pregnancy<br />
Maternal Drugs Affecting<br />
Foetus<br />
Hypervigilance<br />
Amnesia<br />
Hallucination<br />
Date:01/19/05ISR Number: 4555880-6Report Type:Expedited (15-DaCompany Report #FR-ROCHE-386791 Age:58 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Retinopathy Lariam PS Roche ORAL 1 MON<br />
Initial or Prolonged Lariam SS Roche ORAL 1 MON<br />
Lariam SS Roche ORAL UNKNOWN<br />
DURATION.<br />
Lariam SS Roche ORAL 1 MON<br />
Date:01/24/05ISR Number: 4559263-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Cough<br />
Mood Swings<br />
Migraine<br />
Endolymphatic Hydrops<br />
Jaw Disorder<br />
Gilbert'S Syndrome<br />
Hyperacusis<br />
Visual Impairment<br />
Tremor<br />
Obsessive Thoughts
Lacrimation Increased<br />
Mania<br />
Visual Field Defect<br />
Balance Disorder<br />
Diarrhoea<br />
Aggression<br />
Anger<br />
Jaundice<br />
03-Apr-2012 09:37 AM Page: 578
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Post-Traumatic Stress<br />
Disorder<br />
Incontinence Report Source Product Role Manufacturer Route Dose Duration<br />
Vestibular Disorder Lariam PS Roche ORAL 23 TABLETS<br />
General Symptom<br />
DISPENSED.<br />
Depressed Level Of Sudafed C<br />
Consciousness<br />
Confusional State<br />
Dysarthria<br />
Abnormal Dreams<br />
Hallucination<br />
Cranial Nerve Disorder<br />
Photosensitivity Reaction<br />
Toxicity To Various<br />
Agents<br />
Decreased Appetite<br />
Paranoia<br />
Nausea<br />
Disturbance In Attention<br />
Suicidal Ideation<br />
Irritability<br />
Thirst<br />
Cognitive Disorder<br />
Gait Disturbance<br />
Dysphemia<br />
Hepatic Failure<br />
Photophobia<br />
Panic Attack<br />
Insomnia<br />
Depression<br />
Amnesia<br />
Anxiety<br />
Tinnitus<br />
Tardive Dyskinesia<br />
Vomiting<br />
Asthenia<br />
Muscle Twitching<br />
Motor Dysfunction<br />
Conduction Disorder<br />
Date:01/24/05ISR Number: 4560679-0Report Type:Direct Company Report #CTU 237716 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrial Fibrillation Lariam PS ORAL 1 PER WEEK<br />
Initial or Prolonged Nausea FIVE WEEKS<br />
Panic Attack<br />
ORAL<br />
Chest Pain<br />
Disturbance In Attention<br />
Cardiac Disorder<br />
Anxiety<br />
Arrhythmia
Heart Rate Increased<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 579
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/25/05ISR Number: 4560377-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Motion Sickness<br />
Hypersensitivity<br />
Irritable Bowel Syndrome<br />
Phobia Of Flying<br />
Claustrophobia<br />
Bronchospasm<br />
Mood Disorder Due To A<br />
General Medical Condition<br />
Balance Disorder<br />
Migraine<br />
Musculoskeletal Stiffness<br />
Adjustment Disorder With<br />
Anxiety<br />
Suicidal Ideation<br />
Sinus Bradycardia<br />
Dyspnoea<br />
Disturbance In Attention<br />
Vestibular Disorder<br />
Photophobia<br />
Sensory Integrative<br />
Dysfunction<br />
Throat Irritation<br />
Nasal Congestion<br />
Vertigo<br />
Dizziness<br />
Heart Rate Increased<br />
Tinnitus<br />
Self Esteem Decreased<br />
Acrophobia<br />
Fatigue<br />
Sleep Disorder<br />
Neck Pain<br />
Hypoaesthesia<br />
Amnesia<br />
Insomnia<br />
Diarrhoea<br />
Paraesthesia<br />
Disorientation<br />
Headache<br />
Abdominal Pain Lower<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Abdominal Pain<br />
Generalised Anxiety<br />
Disorder<br />
Lactose Intolerance<br />
Abdominal Distension<br />
Emotional Distress<br />
Anxiety<br />
Nausea
Panic Attack<br />
Depression<br />
Weight Decreased<br />
Back Pain<br />
Sleep Disorder Due To<br />
General Medical<br />
Condition, Hypersomnia<br />
Type<br />
03-Apr-2012 09:37 AM Page: 580
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Heart Rate Irregular<br />
Rash<br />
Phobia Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL THE PATIENT<br />
HAD TAKEN 4-5<br />
TABLETS OF<br />
MEFLOQUINE IN<br />
TOTAL. 21 DAY<br />
Vaccine C OTHER 1 DAY<br />
Date:01/27/05ISR Number: 4562580-5Report Type:Expedited (15-DaCompany Report #CH-ROCHE-392759 Age:62 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatocellular Injury Lariam PS Roche ORAL 6 WK<br />
Initial or Prolonged Decreased Appetite Activelle C ORAL DOSE DURATION<br />
Gamma-Glutamyltransferase<br />
WAS REPORTED<br />
Increased<br />
AS LONG TERM.<br />
Nausea Alcacyl C ORAL<br />
Abdominal Pain Upper Selipran C ORAL DOSE DURATION<br />
Jaundice<br />
WAS REPORTED<br />
Pyrexia<br />
AS LONG TERM.<br />
Date:01/27/05ISR Number: 4562583-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-386791 Age:58 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Retinopathy Lariam PS Roche ORAL 1 MON<br />
Initial or Prolonged Lariam SS Roche ORAL 1 MON<br />
Lariam SS Roche ORAL UNKNOWN<br />
DURATION.<br />
Lariam SS Roche ORAL 1 MON<br />
Date:01/27/05ISR Number: 4562585-4Report Type:Expedited (15-DaCompany Report #SE-ROCHE-392506 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disturbance In Attention Lariam PS Roche ORAL<br />
Initial or Prolonged Grand Mal Convulsion<br />
Amnesia<br />
Date:01/27/05ISR Number: 4570256-3Report Type:Direct Company Report #CTU 238338 Age:45 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam 250 Mg Nsn<br />
Depression 6505-00-Foh-3872 PS F.Hoffman-La Roche<br />
Abnormal Dreams Ltd WEEKLY<br />
Stress
Date:01/28/05ISR Number: 4564224-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-392922 Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Lariam PS Roche ORAL<br />
Initial or Prolonged Thrombocytopenia<br />
03-Apr-2012 09:37 AM Page: 581
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/31/05ISR Number: 4575489-8Report Type:Direct Company Report #CTU 238692 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam PS ORAL WEEKLY<br />
Feeling Abnormal<br />
ORAL<br />
Anxiety<br />
Date:02/01/05ISR Number: 4567071-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-393160 Age:6 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Respiratory Arrest Lariam PS Roche ORAL<br />
Initial or Prolonged Convulsion<br />
Date:02/01/05ISR Number: 4567072-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-393292 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypothyroidism Lariam PS Roche ORAL<br />
Autoimmune Thyroiditis<br />
Date:02/04/05ISR Number: 4570324-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Incontinence<br />
Amnesia<br />
Endolymphatic Hydrops<br />
Abnormal Dreams<br />
Hyperacusis<br />
Tremor<br />
Insomnia<br />
Photosensitivity Reaction<br />
General Symptom<br />
Depressed Level Of<br />
Consciousness<br />
Dysarthria<br />
Motor Dysfunction<br />
Hallucination<br />
Cranial Nerve Disorder<br />
Anxiety<br />
Obsessive Thoughts<br />
Gait Disturbance<br />
Visual Field Defect<br />
Balance Disorder<br />
Panic Attack<br />
Vestibular Disorder<br />
Migraine<br />
Thirst<br />
Tardive Dyskinesia
Conduction Disorder<br />
Paranoia<br />
Photophobia<br />
Nausea<br />
Aggression<br />
Suicidal Ideation<br />
Tinnitus<br />
Post-Traumatic Stress<br />
03-Apr-2012 09:37 AM Page: 582
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorder<br />
Decreased Appetite<br />
Lacrimation Increased<br />
Hepatic Failure Report Source Product Role Manufacturer Route Dose Duration<br />
Diarrhoea Lariam PS Roche ORAL 23 TABLETS<br />
Disturbance In Attention<br />
DISPENSED.<br />
Toxicity To Various Sudafed C<br />
Agents<br />
Mood Swings<br />
Dysphemia<br />
Visual Impairment<br />
Vomiting<br />
Anger<br />
Asthenia<br />
Cough<br />
Jaundice<br />
Confusional State<br />
Mania<br />
Gilbert'S Syndrome<br />
Depression<br />
Irritability<br />
Muscle Twitching<br />
Jaw Disorder<br />
Cognitive Disorder<br />
Date:02/04/05ISR Number: 4570348-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-393292 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Autoimmune Thyroiditis Lariam PS Roche ORAL<br />
Hypothyroidism<br />
Date:02/04/05ISR Number: 4570355-6Report Type:Expedited (15-DaCompany Report #CA-ROCHE-393893 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypotrichosis Lariam PS Roche ORAL 15 DAY<br />
Hallucination Vivotif "Berna" C ORAL<br />
White Blood Cell Count Dukoral C ORAL<br />
Decreased<br />
Vomiting<br />
Loss Of Consciousness<br />
Fatigue<br />
Somnolence<br />
Panic Attack<br />
Dizziness<br />
Date:02/07/05ISR Number: 4571642-8Report Type:Expedited (15-DaCompany Report #CH-BRISTOL-MYERS SQUIBB COMPANY-12841631 Age:59 YR Gender:Female I/FU:I<br />
Outcome<br />
PT
Hospitalization -<br />
Initial or Prolonged<br />
Nausea<br />
Transaminases Increased<br />
Prothrombin Time<br />
Prolonged<br />
Chromaturia<br />
Jaundice Cholestatic<br />
Hepatitis A Antibody<br />
Positive<br />
03-Apr-2012 09:37 AM Page: 583
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abdominal Pain<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Selipran Tabs PS Bristol-Myers Squibb<br />
Company<br />
ORAL<br />
Activella SS ORAL<br />
Lariam SS ORAL<br />
Date:02/07/05ISR Number: 4571701-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-384854 Age:37 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Consumer Lariam PS Roche ORAL 3-4 PILLS<br />
Mood Swings WERE TAKEN. 43 DAY<br />
Fatigue<br />
Panic Attack<br />
Chills<br />
Depression<br />
Agitation<br />
Liver Function Test<br />
Abnormal<br />
Heart Rate Increased<br />
Anxiety<br />
Paranoia<br />
Hyperhidrosis<br />
Disorientation<br />
Disturbance In Attention<br />
Back Pain<br />
Balance Disorder<br />
Vertigo<br />
Paraesthesia<br />
Cold Sweat<br />
Skin Disorder<br />
Feeling Abnormal<br />
Heart Rate Irregular<br />
Insomnia<br />
Alanine Aminotransferase<br />
Increased<br />
Date:02/09/05ISR Number: 4578673-2Report Type:Direct Company Report #CTU 239959 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disturbance In Attention Lariam 250mg<br />
Dizziness Roxane PS Roxane ORAL 1 WEEKLY<br />
Memory Impairment<br />
ORAL<br />
Anxiety<br />
Date:02/10/05ISR Number: 4577110-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-319597 Age:33 YR Gender:Male I/FU:F
Outcome<br />
Death<br />
PT<br />
Completed Suicide<br />
Tremor<br />
Brain Oedema<br />
Aggression<br />
Nervousness<br />
Abnormal Behaviour<br />
03-Apr-2012 09:37 AM Page: 584
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Psychotic Disorder<br />
Asphyxia<br />
Stress Report Source Product Role Manufacturer Route Dose Duration<br />
Mood Altered Lariam PS Roche ORAL TAKEN WEEKLY 99 DAY<br />
Homicide Primaquine Phosphate C ORAL 15 DAY<br />
Disorientation<br />
Incoherent<br />
Communication Disorder<br />
Dysphemia<br />
Social Avoidant Behaviour<br />
Pulmonary Congestion<br />
Feeling Abnormal<br />
Anger<br />
Marital Problem<br />
Date:02/10/05ISR Number: 4577145-9Report Type:Expedited (15-DaCompany Report #DK-ROCHE-394266 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nervousness Lariam PS Roche ORAL<br />
Mental Impairment<br />
Petit Mal Epilepsy<br />
Date:02/10/05ISR Number: 4577147-2Report Type:Expedited (15-DaCompany Report #CH-ROCHE-394176 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Transaminases Increased Lariam PS Roche ORAL 45 DAY<br />
Initial or Prolonged Abdominal Pain Selipran SS ORAL<br />
Jaundice Cholestatic Activelle SS ORAL FREQUENCY<br />
Nausea<br />
REPORTED AS<br />
Chromaturia<br />
"AS<br />
Prothrombin Time<br />
NECESSARY"<br />
Prolonged<br />
Date:02/10/05ISR Number: 4577157-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-394348 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Disturbance In Attention Lariam PS Roche ORAL DOSAGE<br />
Palpitations<br />
REGIMEN<br />
Paraesthesia REPORTED AS 1<br />
Sleep Disorder TABLET WEEKLY 15 DAY<br />
Dizziness<br />
Restlessness<br />
Date:02/15/05ISR Number: 4586918-8Report Type:Direct Company Report #CTU 240367 Age:27 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Lariam<br />
Nausea Roche PS Roche<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 585
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/16/05ISR Number: 4584087-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Balance Disorder<br />
Diarrhoea<br />
Muscle Twitching<br />
Endolymphatic Hydrops<br />
Tardive Dyskinesia<br />
Lacrimation Increased<br />
Dysphemia<br />
Conduction Disorder<br />
Hallucination<br />
Cranial Nerve Disorder<br />
Tremor<br />
Toxicity To Various<br />
Agents<br />
Obsessive Thoughts<br />
Post-Traumatic Stress<br />
Disorder<br />
Gait Disturbance<br />
Abnormal Dreams<br />
Neuropathy Peripheral<br />
Hepatic Failure<br />
Visual Field Defect<br />
Insomnia<br />
Anger<br />
Depression<br />
Vestibular Disorder<br />
Photosensitivity Reaction<br />
Depressed Level Of<br />
Consciousness<br />
Visual Impairment<br />
Amnesia<br />
Hyperacusis<br />
Paranoia<br />
Panic Attack<br />
Incontinence<br />
Asthenia<br />
Cough<br />
Tinnitus<br />
Jaundice<br />
Dysarthria<br />
Motor Dysfunction<br />
Photophobia<br />
Nausea<br />
Aggression<br />
Anxiety<br />
General Symptom<br />
Migraine<br />
Jaw Disorder<br />
Cognitive Disorder<br />
Mania<br />
Viiith Nerve Lesion
Vomiting<br />
Suicidal Ideation<br />
Irritability<br />
Disturbance In Attention<br />
Mood Swings<br />
Decreased Appetite<br />
Thirst<br />
Confusional State<br />
03-Apr-2012 09:37 AM Page: 586
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Gilbert'S Syndrome<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL 23 TABLETS<br />
DISPENSED.<br />
Sudafed<br />
C<br />
Date:02/17/05ISR Number: 4586792-XReport Type:Direct Company Report #CTU 240711 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia Lariam PS 1 TABLET 10<br />
Phobia<br />
DAYS??<br />
Panic Attack<br />
Suicidal Ideation<br />
Personality Change<br />
Gastric Disorder<br />
Crying<br />
Anxiety<br />
Paranoia<br />
Date:02/23/05ISR Number: 4589655-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-390682 Age:59 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety Lariam PS Roche UNKNOWN<br />
Hospitalization - Palpitations<br />
Initial or Prolonged Weight Increased<br />
Foot Fracture<br />
Panic Attack<br />
Heart Rate Increased<br />
Fluid Retention<br />
Balance Disorder<br />
Atrial Fibrillation<br />
Hyperhidrosis<br />
Date:03/04/05ISR Number: 4598556-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-381724 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche ORAL<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:03/04/05ISR Number: 4599949-9Report Type:Direct Company Report #CTU 242097 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delirium <strong>Mefloquine</strong> 250 Mg PS ORAL 250 MG QWEEK
Initial or Prolonged Thinking Abnormal ORAL<br />
Hallucination<br />
03-Apr-2012 09:37 AM Page: 587
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/09/05ISR Number: 4603011-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-397065 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Toxicity To Various Lariam PS Roche ORAL FREQUENCY<br />
Agents<br />
STATED AS<br />
Dizziness WEEKLY. 47 DAY<br />
Psychotic Disorder<br />
Hearing Impaired<br />
Visual Impairment<br />
Tinnitus<br />
Panic Attack<br />
Anxiety<br />
Depression<br />
Confusional State<br />
Mood Swings<br />
Paranoia<br />
Insomnia<br />
Date:03/10/05ISR Number: 4603890-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Sinus Tachycardia<br />
Blood Cholesterol<br />
Increased<br />
Snoring<br />
Constipation<br />
Blood Glucose Increased<br />
Upper-Airway Cough<br />
Syndrome<br />
Visual Impairment<br />
Psychotic Disorder<br />
Amnesia<br />
Dry Mouth<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Bipolar Disorder<br />
Scratch<br />
Schizoaffective Disorder<br />
Depression<br />
Fatigue<br />
Headache<br />
Restrictive Pulmonary<br />
Disease<br />
Somnolence<br />
Viral Infection<br />
Bronchospasm<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Diarrhoea<br />
Dehydration<br />
Nocturia
Gastrooesophageal Reflux<br />
Disease<br />
Confusional State<br />
Anxiety<br />
Bronchitis<br />
Weight Increased<br />
Exfoliative Rash<br />
Acrochordon<br />
03-Apr-2012 09:37 AM Page: 588
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Asthma<br />
Major Depression<br />
Aggression Report Source Product Role Manufacturer Route Dose Duration<br />
Skin Disorder Lariam PS Roche ORAL<br />
Gastrointestinal Disorder Zoloft C UNKNOWN<br />
Diastolic Dysfunction<br />
Total Lung Capacity<br />
Decreased<br />
Dry Eye<br />
Balance Disorder<br />
Vestibular Disorder<br />
Hypertension<br />
Angina Pectoris<br />
Arthralgia<br />
Low Density Lipoprotein<br />
Increased<br />
Dry Skin<br />
Chest Discomfort<br />
Vitiligo<br />
Haemorrhoids<br />
Contusion<br />
Affective Disorder<br />
Dyspnoea<br />
Sleep Apnoea Syndrome<br />
Rash<br />
Cardiac Murmur<br />
Blood Triglycerides<br />
Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Rhinitis<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Memory Impairment<br />
Tremor<br />
Mitral Valve Prolapse<br />
Ventricular Hypertrophy<br />
Oral Disorder<br />
Insomnia<br />
Clumsiness<br />
Abnormal Dreams<br />
Coordination Abnormal<br />
Date:03/10/05ISR Number: 4603962-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-397276 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Furuncle Lariam PS Roche ORAL<br />
Initial or Prolonged
Date:03/11/05ISR Number: 4606008-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Hepatic Failure<br />
Hallucination<br />
Tremor<br />
Insomnia<br />
Thirst<br />
03-Apr-2012 09:37 AM Page: 589
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dysphemia<br />
Abnormal Dreams<br />
Tooth Discolouration<br />
Gastroenteritis<br />
Incontinence<br />
Aggression<br />
Suicidal Ideation<br />
Irritability<br />
Atrial Fibrillation<br />
Antisocial Behaviour<br />
Amnesia<br />
Asthenia<br />
Mood Swings<br />
Depressed Level Of<br />
Consciousness<br />
Decreased Appetite<br />
Cognitive Disorder<br />
Lacrimation Increased<br />
Viiith Nerve Lesion<br />
Nervous System Disorder<br />
Cranial Nerve Disorder<br />
Balance Disorder<br />
Nausea<br />
Cough<br />
Tinnitus<br />
Jaw Disorder<br />
Confusional State<br />
Gilbert'S Syndrome<br />
Memory Impairment<br />
Visual Impairment<br />
Depression<br />
General Symptom<br />
Obsessive Thoughts<br />
Post-Traumatic Stress<br />
Disorder<br />
Endolymphatic Hydrops<br />
Dysarthria<br />
Photophobia<br />
Panic Attack<br />
Toxicity To Various<br />
Agents<br />
Muscle Twitching<br />
Tardive Dyskinesia<br />
Gait Disturbance<br />
Hyperacusis<br />
Creutzfeldt-Jakob Disease<br />
Paranoia<br />
Diarrhoea<br />
Vestibular Disorder<br />
Anxiety<br />
Jaundice<br />
Migraine<br />
Motor Dysfunction
Conduction Disorder<br />
Hepato-Lenticular<br />
Degeneration<br />
Metal Poisoning<br />
Visual Field Defect<br />
Vomiting<br />
Anger<br />
Disturbance In Attention<br />
03-Apr-2012 09:37 AM Page: 590
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Photosensitivity Reaction<br />
Mania<br />
Alcohol Poisoning Report Source Product Role Manufacturer Route Dose Duration<br />
Rash Pruritic Lariam PS Roche ORAL 23 TABLETS<br />
DISPENSED.<br />
Sudafed<br />
C<br />
Date:03/16/05ISR Number: 4610613-XReport Type:Expedited (15-DaCompany Report #004337 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Consumer <strong>Mefloquine</strong><br />
Dizziness<br />
Hydrochloride<br />
Myalgia<br />
(<strong>Mefloquine</strong><br />
Pyrexia<br />
Hydrochloride)<br />
Gait Disturbance Tablet, 250 Mg PS ORAL SEE IMAGE 30 DAY<br />
Aphasia<br />
Toprol Xl<br />
(Metoprolol<br />
Succinate)<br />
C<br />
Zoloft (Sertraline<br />
Hydrochloride) C<br />
Date:03/16/05ISR Number: 4612641-7Report Type:Direct Company Report #CTU 243413 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Aggression <strong>Mefloquine</strong>-Lariam- PS ORAL 1 WEEK ORAL<br />
Hospitalization - Self Injurious Behaviour<br />
Initial or Prolonged Feeling Of Despair<br />
Disturbance In Social<br />
Behaviour<br />
Thinking Abnormal<br />
Anxiety<br />
Mental Disorder<br />
Emotional Disorder<br />
Psychotic Disorder<br />
Abnormal Behaviour<br />
Suicidal Ideation<br />
Multiple Injuries<br />
Date:03/21/05ISR Number: 4613947-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Fatigue<br />
Total Lung Capacity<br />
Decreased<br />
Abnormal Dreams<br />
Asthma<br />
Haemorrhoids
Contusion<br />
Psychotic Disorder<br />
Amnesia<br />
Vestibular Disorder<br />
Gastrointestinal Disorder<br />
Headache<br />
Somnolence<br />
Chest Discomfort<br />
03-Apr-2012 09:37 AM Page: 591
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Rhinitis<br />
Insomnia<br />
Balance Disorder<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Major Depression<br />
Dyspnoea<br />
Mitral Valve Prolapse<br />
Hypertension<br />
Arthralgia<br />
Weight Increased<br />
Cardiac Murmur<br />
Sinus Tachycardia<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Bipolar Disorder<br />
Memory Impairment<br />
Tremor<br />
Depression<br />
Restrictive Pulmonary<br />
Disease<br />
Bronchitis<br />
Bronchospasm<br />
Dry Eye<br />
Blood Glucose Increased<br />
Schizoaffective Disorder<br />
Angina Pectoris<br />
Blood Cholesterol<br />
Increased<br />
Blood Triglycerides<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Diarrhoea<br />
Constipation<br />
Acrochordon<br />
Coordination Abnormal<br />
Affective Disorder<br />
Aggression<br />
Rash<br />
Dehydration<br />
Dry Skin<br />
Oral Disorder<br />
Vitiligo<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Gastrooesophageal Reflux<br />
Disease<br />
Scratch<br />
Anxiety<br />
Skin Disorder
Diastolic Dysfunction<br />
Exfoliative Rash<br />
Nocturia<br />
Clumsiness<br />
Visual Impairment<br />
Confusional State<br />
Ventricular Hypertrophy<br />
Sleep Apnoea Syndrome<br />
03-Apr-2012 09:37 AM Page: 592
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Viral Infection<br />
Dry Mouth<br />
Snoring Report Source Product Role Manufacturer Route Dose Duration<br />
Upper-Airway Cough Lariam PS Roche ORAL<br />
Syndrome Zoloft C UNKNOWN<br />
Date:03/21/05ISR Number: 4613968-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-392506 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Grand Mal Convulsion Lariam PS Roche ORAL<br />
Initial or Prolonged Disturbance In Attention<br />
Amnesia<br />
Date:03/22/05ISR Number: 4620246-7Report Type:Direct Company Report #CTU 243980 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness <strong>Mefloquine</strong> PS ORAL 1 WEEK ORAL<br />
Anxiety<br />
Depressed Mood<br />
Nausea<br />
Date:03/23/05ISR Number: 4617079-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Dyspnoea<br />
Hypertension<br />
Restrictive Pulmonary<br />
Disease<br />
Somnolence<br />
Bronchitis<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Exfoliative Rash<br />
Snoring<br />
Vitiligo<br />
Rhinitis<br />
Insomnia<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Major Depression<br />
Psychotic Disorder<br />
Body Temperature<br />
Increased<br />
Wheezing<br />
Rectal Haemorrhage<br />
Depression<br />
Angina Pectoris
Total Lung Capacity<br />
Decreased<br />
Sleep Apnoea Syndrome<br />
Dry Mouth<br />
Oral Disorder<br />
Aggression<br />
Cough<br />
Sarcoidosis<br />
03-Apr-2012 09:37 AM Page: 593
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Amnesia<br />
Vestibular Disorder<br />
Fatigue<br />
Arthralgia<br />
Rash<br />
Low Density Lipoprotein<br />
Increased<br />
Dehydration<br />
Upper-Airway Cough<br />
Syndrome<br />
Asthma<br />
Scratch<br />
Tremor<br />
Productive Cough<br />
Photosensitivity Reaction<br />
Anxiety<br />
Weight Increased<br />
Blood Glucose Increased<br />
Visual Impairment<br />
Peripheral Sensory<br />
Neuropathy<br />
Exercise Tolerance<br />
Decreased<br />
Sinusitis<br />
Confusional State<br />
Mitral Valve Prolapse<br />
Blood Cholesterol<br />
Increased<br />
Diarrhoea<br />
Chest Discomfort<br />
Constipation<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Nervous System Disorder<br />
Skin Disorder<br />
Headache<br />
Ventricular Hypertrophy<br />
Diastolic Dysfunction<br />
Bronchospasm<br />
Blood Triglycerides<br />
Increased<br />
Dry Skin<br />
Acrochordon<br />
Contusion<br />
Schizoaffective Disorder<br />
Listless<br />
Upper Respiratory Tract<br />
Infection<br />
Oedema<br />
Jaw Disorder<br />
Gastrointestinal Disorder<br />
Cardiac Murmur<br />
Blood Creatine
Phosphokinase Increased<br />
Dry Eye<br />
Nocturia<br />
Gastrooesophageal Reflux<br />
Disease<br />
Haemorrhoids<br />
Affective Disorder<br />
Eye Pruritus<br />
03-Apr-2012 09:37 AM Page: 594
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Lacrimation Increased<br />
Viral Infection<br />
Sinus Tachycardia Report Source Product Role Manufacturer Route Dose Duration<br />
Clumsiness Lariam PS Roche ORAL<br />
Abnormal Dreams Zoloft C UNKNOWN<br />
Bipolar Disorder<br />
Balance Disorder<br />
Coordination Abnormal<br />
Memory Impairment<br />
Sputum Discoloured<br />
Myalgia<br />
Sjogren'S Syndrome<br />
Embolism Arterial<br />
Discomfort<br />
Nasal Congestion<br />
Date:03/23/05ISR Number: 4617081-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Cranial Nerve Disorder<br />
Insomnia<br />
Disturbance In Attention<br />
Vestibular Disorder<br />
Cough<br />
Irritability<br />
Cognitive Disorder<br />
Hyperacusis<br />
Dizziness<br />
Weight Decreased<br />
Psychiatric Symptom<br />
Nightmare<br />
Antisocial Behaviour<br />
Incontinence<br />
Aggression<br />
Obsessive Thoughts<br />
Muscle Twitching<br />
Thirst<br />
Gait Disturbance<br />
Motor Dysfunction<br />
Gilbert'S Syndrome<br />
Electrocardiogram St-T<br />
Change<br />
Tooth Discolouration<br />
Stress<br />
Upper Respiratory Tract<br />
Infection<br />
Tremor<br />
Anger<br />
Depressed Level Of<br />
Consciousness<br />
Dysarthria
Fatigue<br />
Electroencephalogram<br />
Abnormal<br />
Hepatic Failure<br />
Hallucination<br />
Visual Field Defect<br />
Nausea<br />
Jaundice<br />
03-Apr-2012 09:37 AM Page: 595
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tearfulness<br />
Acoustic Neuritis<br />
Vomiting Report Source Product Role Manufacturer Route Dose Duration<br />
Suicidal Ideation Lariam PS Roche ORAL 23 TABLETS<br />
Anxiety<br />
DISPENSED.<br />
General Symptom Sudafed C<br />
Tinnitus<br />
Post-Traumatic Stress<br />
Disorder<br />
Endolymphatic Hydrops<br />
Decreased Appetite<br />
Abnormal Dreams<br />
Reading Disorder<br />
Tic<br />
Panic Attack<br />
Toxicity To Various<br />
Agents<br />
Mania<br />
Vertigo<br />
Narcissistic Personality<br />
Disorder<br />
Paranoia<br />
Photophobia<br />
Diarrhoea<br />
Depression<br />
Mood Swings<br />
Jaw Disorder<br />
Tardive Dyskinesia<br />
Lacrimation Increased<br />
Confusional State<br />
Dysphemia<br />
Visual Impairment<br />
Balance Disorder<br />
Amnesia<br />
Photosensitivity Reaction<br />
Asthenia<br />
Migraine<br />
Conduction Disorder<br />
Bipolar Disorder<br />
Gastroenteritis<br />
Hyporeflexia<br />
Feeling Abnormal<br />
Date:03/24/05ISR Number: 4617721-8Report Type:Expedited (15-DaCompany Report #CA-ROCHE-398664 Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pancreatitis Lariam PS Roche ORAL 3 DOSES TAKEN<br />
Date:03/24/05ISR Number: 4617730-9Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398996 Age:30 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche ORAL TOTAL OF 7-8<br />
Maternal Exposure During<br />
250 MG<br />
Pregnancy<br />
TABLETS WERE<br />
TAKEN<br />
03-Apr-2012 09:37 AM Page: 596
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/24/05ISR Number: 4617734-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-399038 Age:61 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Lariam PS Roche UNKNOWN<br />
Dermatitis Exfoliative<br />
Vision Blurred<br />
Dry Skin<br />
Vomiting<br />
Date:03/30/05ISR Number: 4622495-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-257719 Age:32 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Lariam Tablets PS Roche ORAL SEVEN TABLETS<br />
Visual Impairment<br />
TAKEN IN<br />
Panic Attack<br />
TOTAL.<br />
Muscle Twitching<br />
Insomnia<br />
Anxiety<br />
Agoraphobia<br />
Confusional State<br />
Hepatocellular Injury<br />
Hallucination<br />
Aggression<br />
Nervous System Disorder<br />
Suicidal Ideation<br />
Paranoia<br />
Psychotic Disorder<br />
Hypertension<br />
Dissociative Disorder<br />
Tremor<br />
Mood Swings<br />
Fatigue<br />
Pyrexia<br />
Headache<br />
Feeling Abnormal<br />
Immune System Disorder<br />
Amnesia<br />
Balance Disorder<br />
Depression<br />
Myalgia<br />
Date:03/31/05ISR Number: 4623912-2Report Type:Expedited (15-DaCompany Report #CA-ROCHE-393893 Age:22 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Loss Of Consciousness Lariam PS Roche ORAL 15 DAY<br />
Dizziness Vivotif "Berna" C ORAL<br />
Vomiting Dukoral C ORAL<br />
Somnolence<br />
Panic Attack
Hallucination<br />
Fatigue<br />
White Blood Cell Count<br />
Decreased<br />
Hypotrichosis<br />
03-Apr-2012 09:37 AM Page: 597
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/01/05ISR Number: 4625199-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Mitral Valve Prolapse<br />
Diastolic Dysfunction<br />
Bronchitis<br />
Blood Triglycerides<br />
Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Exfoliative Rash<br />
Balance Disorder<br />
Sinusitis<br />
Amnesia<br />
Anxiety<br />
Sinus Tachycardia<br />
Diarrhoea<br />
Nocturia<br />
Vitiligo<br />
Gastrooesophageal Reflux<br />
Disease<br />
Haemorrhoids<br />
Psychotic Disorder<br />
Gastrointestinal Disorder<br />
Ventricular Hypertrophy<br />
Hypertension<br />
Restrictive Pulmonary<br />
Disease<br />
Dry Skin<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Bipolar Disorder<br />
Major Depression<br />
Contusion<br />
Aggression<br />
Depression<br />
Oral Disorder<br />
Fatigue<br />
Angina Pectoris<br />
Total Lung Capacity<br />
Decreased<br />
Viral Infection<br />
Rash<br />
Blood Cholesterol<br />
Increased<br />
Dehydration<br />
Dry Mouth<br />
Insomnia<br />
Visual Impairment<br />
Scratch<br />
Memory Impairment<br />
Affective Disorder<br />
Tremor
Confusional State<br />
Dyspnoea<br />
Somnolence<br />
Bronchospasm<br />
Cardiac Murmur<br />
Snoring<br />
Chest Discomfort<br />
Constipation<br />
03-Apr-2012 09:37 AM Page: 598
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Acrochordon<br />
Blood Glucose Increased<br />
Headache Report Source Product Role Manufacturer Route Dose Duration<br />
Sleep Apnoea Syndrome Lariam PS Roche ORAL<br />
Arthralgia Zoloft C UNKNOWN<br />
Weight Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Clumsiness<br />
Asthma<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Vestibular Disorder<br />
Skin Disorder<br />
Dry Eye<br />
Upper-Airway Cough<br />
Syndrome<br />
Rhinitis<br />
Abnormal Dreams<br />
Coordination Abnormal<br />
Schizoaffective Disorder<br />
Date:04/01/05ISR Number: 4625201-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Paranoia<br />
Hallucination<br />
Depression<br />
Anxiety<br />
Obsessive Thoughts<br />
Jaw Disorder<br />
Gait Disturbance<br />
Abnormal Dreams<br />
Cranial Nerve Disorder<br />
Panic Attack<br />
Incontinence<br />
Cough<br />
Irritability<br />
Depressed Level Of<br />
Consciousness<br />
Tardive Dyskinesia<br />
Motor Dysfunction<br />
Gilbert'S Syndrome<br />
Amnesia<br />
General Symptom<br />
Muscle Twitching<br />
Mood Swings<br />
Migraine<br />
Hepatic Failure
Tremor<br />
Diarrhoea<br />
Aggression<br />
Vestibular Disorder<br />
Photosensitivity Reaction<br />
Tinnitus<br />
Lacrimation Increased<br />
Dysarthria<br />
03-Apr-2012 09:37 AM Page: 599
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Conduction Disorder<br />
Hyperacusis<br />
Photophobia Report Source Product Role Manufacturer Route Dose Duration<br />
Disturbance In Attention Lariam PS Roche ORAL 23 TABLETS<br />
Visual Impairment<br />
DISPENSED.<br />
Visual Field Defect Sudafed C<br />
Balance Disorder<br />
Anger<br />
Endolymphatic Hydrops<br />
Decreased Appetite<br />
Mania<br />
Insomnia<br />
Jaundice<br />
Cognitive Disorder<br />
Nausea<br />
Vomiting<br />
Toxicity To Various<br />
Agents<br />
Suicidal Ideation<br />
Asthenia<br />
Post-Traumatic Stress<br />
Disorder<br />
Thirst<br />
Confusional State<br />
Dysphemia<br />
Date:04/04/05ISR Number: 4626282-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-399129 Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Contusion Lariam PS Roche ORAL 250 MG<br />
International Normalised<br />
RECEIVED<br />
Ratio Increased<br />
WEEKLY.<br />
Cefaclor SS ORAL 5 DAY<br />
Warfarin SS ORAL<br />
Date:04/08/05ISR Number: 4630752-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-399038 Age:61 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dermatitis Exfoliative Lariam PS Roche ORAL 20 DAY<br />
Dry Skin<br />
Feeling Abnormal<br />
Vision Blurred<br />
Vomiting<br />
Sensory Disturbance<br />
Date:04/11/05ISR Number: 4632263-1Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398996 Age:29 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche ORAL TOTAL OF 7-8<br />
Maternal Exposure During<br />
250 MG<br />
Pregnancy<br />
TABLETS WERE<br />
Ultrasound Antenatal<br />
TAKEN<br />
Screen Abnormal Trinordiol C ORAL<br />
Vaginal Haemorrhage<br />
03-Apr-2012 09:37 AM Page: 600
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/11/05ISR Number: 4657678-7Report Type:Periodic Company Report #GXBR2005US00443 Age:51 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nausea Consumer <strong>Mefloquine</strong> Hcl<br />
Initial or Prolonged Tinnitus Tablets<br />
Convulsion<br />
(Ngx)(<strong>Mefloquine</strong>)<br />
Dizziness Tablet PS ORAL 250 MG, QW,<br />
ORAL; 500<br />
MG, QW<br />
Valium (Diazepam) C<br />
Vicodin (Hydrocodone<br />
Bitartrate,<br />
Paracetamol)<br />
C<br />
Date:04/11/05ISR Number: 4657680-5Report Type:Periodic Company Report #GXBR2005US00448 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Postural Consumer <strong>Mefloquine</strong> Hcl<br />
Asthenia<br />
Tablets<br />
Loss Of Consciousness<br />
(Ngx)(<strong>Mefloquine</strong>)<br />
Palpitations Tablet PS ORAL 250 MG, QW,<br />
Disturbance In Attention<br />
ORAL<br />
Tremor<br />
Cipro (Ciprofloxacin<br />
Hydrochloride) C<br />
Date:04/18/05ISR Number: 4637881-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Confusional State<br />
Depression<br />
Headache<br />
Bronchospasm<br />
Diarrhoea<br />
Chest Discomfort<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Bronchitis<br />
Weight Increased<br />
Cardiac Murmur<br />
Sinus Tachycardia<br />
Blood Triglycerides<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Nocturia<br />
Vitiligo<br />
Upper-Airway Cough<br />
Syndrome<br />
Abnormal Dreams
Coordination Abnormal<br />
Major Depression<br />
Aggression<br />
Amnesia<br />
Gastrointestinal Disorder<br />
Hypertension<br />
Dry Skin<br />
Snoring<br />
03-Apr-2012 09:37 AM Page: 601
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Acrochordon<br />
Insomnia<br />
Anxiety Report Source Product Role Manufacturer Route Dose Duration<br />
Dyspnoea Lariam PS Roche ORAL<br />
Vestibular Disorder Zoloft C UNKNOWN<br />
Mitral Valve Prolapse<br />
Skin Disorder<br />
Angina Pectoris<br />
Restrictive Pulmonary<br />
Disease<br />
Rhinitis<br />
Bipolar Disorder<br />
Contusion<br />
Schizoaffective Disorder<br />
Somnolence<br />
Rash<br />
Dehydration<br />
Dry Eye<br />
Blood Glucose Increased<br />
Asthma<br />
Haemorrhoids<br />
Affective Disorder<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Exfoliative Rash<br />
Dry Mouth<br />
Scratch<br />
Tremor<br />
Ventricular Hypertrophy<br />
Sleep Apnoea Syndrome<br />
Viral Infection<br />
Arthralgia<br />
Oral Disorder<br />
Gastrooesophageal Reflux<br />
Disease<br />
Clumsiness<br />
Balance Disorder<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Psychotic Disorder<br />
Fatigue<br />
Diastolic Dysfunction<br />
Total Lung Capacity<br />
Decreased<br />
Blood Cholesterol<br />
Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Constipation<br />
Visual Impairment<br />
Memory Impairment
Date:04/18/05ISR Number: 4638885-6Report Type:Direct Company Report #CTU 246445 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sleep Talking <strong>Mefloquine</strong> PS<br />
Abnormal Behaviour<br />
03-Apr-2012 09:37 AM Page: 602
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/25/05ISR Number: 4644025-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Diarrhoea Lariam PS Roche ORAL 23 TABLETS<br />
Toxicity To Various<br />
DISPENSED.<br />
Agents Sudafed C<br />
Irritability<br />
Obsessive Thoughts<br />
Migraine<br />
Endolymphatic Hydrops<br />
Thirst<br />
Hepatic Failure<br />
Gilbert'S Syndrome<br />
Hyperacusis<br />
Visual Field Defect<br />
Balance Disorder<br />
Vomiting<br />
Asthenia<br />
Muscle Twitching<br />
Post-Traumatic Stress<br />
Disorder<br />
Dysphemia<br />
Mania<br />
Cranial Nerve Disorder<br />
Tremor<br />
Nausea<br />
Suicidal Ideation<br />
Motor Dysfunction<br />
Photophobia<br />
Anger<br />
Depressed Level Of<br />
Consciousness<br />
Tardive Dyskinesia<br />
Gait Disturbance<br />
Hallucination<br />
Insomnia<br />
Photosensitivity Reaction<br />
Anxiety<br />
Tinnitus<br />
Decreased Appetite<br />
Jaw Disorder<br />
Confusional State<br />
Dysarthria<br />
Abnormal Dreams<br />
Incontinence<br />
Aggression<br />
Amnesia<br />
Vestibular Disorder<br />
General Symptom<br />
Cough<br />
Jaundice<br />
Mood Swings<br />
Cognitive Disorder
Conduction Disorder<br />
Paranoia<br />
Visual Impairment<br />
Panic Attack<br />
Disturbance In Attention<br />
Depression<br />
Lacrimation Increased<br />
03-Apr-2012 09:37 AM Page: 603
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/26/05ISR Number: 4645005-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Vomiting Lariam PS Roche ORAL<br />
Blood Pressure Increased Imodium C<br />
Chest Pain Compazine C ON 18 APR<br />
Depression<br />
2001, THERAPY<br />
Rash<br />
REPORTED AS<br />
Photophobia<br />
TWICE DAILY.<br />
Phonophobia Antibiotics Nos C<br />
Insomnia Ambien C<br />
Nightmare Prednisone C BY TAPER.<br />
Vertigo Valium C ON 18 APR<br />
Tinnitus<br />
2001, THERAPY<br />
Inner Ear Disorder<br />
REPORTED AS:<br />
Vision Blurred<br />
AS NEEDED.<br />
Disturbance In Attention Phenergan C<br />
Hypoaesthesia Biaxin C<br />
Blood Urea Increased Advil C<br />
Gait Disturbance Bactrim C<br />
Amnesia Antivert C<br />
Dizziness<br />
Eye Pain<br />
Pain Of Skin<br />
Abnormal Dreams<br />
Anxiety Disorder<br />
Memory Impairment<br />
Headache<br />
Nystagmus<br />
Irritable Bowel Syndrome<br />
Neutrophil Count<br />
Increased<br />
Lymphocyte Count<br />
Decreased<br />
Epididymitis<br />
Nausea<br />
Fatigue<br />
Psychotic Disorder<br />
Testicular Pain<br />
Diarrhoea<br />
Disorientation<br />
Vestibular Disorder<br />
Colitis<br />
Monocyte Count Increased<br />
Night Sweats<br />
Agitation<br />
Mood Swings<br />
Abdominal Pain<br />
Nervousness<br />
Pain<br />
Enteritis
Date:04/28/05ISR Number: 4647479-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-319597 Age:33 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
PT<br />
Completed Suicide<br />
Incoherent<br />
Feeling Abnormal<br />
Aggression<br />
03-Apr-2012 09:37 AM Page: 604
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abnormal Behaviour<br />
Homicide<br />
Psychotic Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Social Avoidant Behaviour Lariam PS Roche ORAL TAKEN WEEKLY 99 DAY<br />
Disorientation Primaquine Phosphate C ORAL 15 DAY<br />
Pulmonary Congestion<br />
Asphyxia<br />
Communication Disorder<br />
Mood Altered<br />
Dysphemia<br />
Anger<br />
Tremor<br />
Nervousness<br />
Brain Oedema<br />
Date:04/28/05ISR Number: 4647505-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-385961 Age:70 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Insomnia Lariam PS Roche ORAL<br />
Initial or Prolonged Neuropathy Peripheral<br />
Disability<br />
Limb Deformity<br />
General Physical Health<br />
Deterioration<br />
Joint Dislocation<br />
Upper Limb Fracture<br />
Fall<br />
Balance Disorder<br />
Decreased Appetite<br />
Date:04/28/05ISR Number: 4647549-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-402425 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatocellular Injury Lariam PS Roche ORAL 3 YR<br />
Liver Function Test Alcohol SS UNKNOWN<br />
Abnormal<br />
Date:04/28/05ISR Number: 4647735-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-402089 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Epilepsy Lariam PS Roche UNKNOWN 5 MON<br />
Anxiety<br />
Nightmare<br />
Date:04/28/05ISR Number: 4649734-4Report Type:Direct Company Report #CTU 247330 Age: Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nervous System Disorder Lariam Standard<br />
Disability Cognitive Disorder Hoffman Laroche PS Hoffman Laroche ORAL 1 PILL<br />
Required Toxicity To Various WEEKLY ORAL<br />
Intervention to Agents Namenda C<br />
Prevent Permanent Vestibular Disorder Paxil C<br />
Impairment/Damage Depakote C<br />
Benzodiazapines C<br />
03-Apr-2012 09:37 AM Page: 605
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sleeping Pills<br />
C<br />
Date:05/02/05ISR Number: 4649996-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Mitral Valve Prolapse<br />
Viral Infection<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Exfoliative Rash<br />
Oral Disorder<br />
Gastrooesophageal Reflux<br />
Disease<br />
Insomnia<br />
Schizoaffective Disorder<br />
Confusional State<br />
Headache<br />
Hypertension<br />
Rash<br />
Dry Mouth<br />
Clumsiness<br />
Coordination Abnormal<br />
Memory Impairment<br />
Restrictive Pulmonary<br />
Disease<br />
Bronchitis<br />
Cardiac Murmur<br />
Sinus Tachycardia<br />
Dry Eye<br />
Blood Glucose Increased<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Depression<br />
Diastolic Dysfunction<br />
Somnolence<br />
Blood Triglycerides<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Dehydration<br />
Rhinitis<br />
Haemorrhoids<br />
Balance Disorder<br />
Aggression<br />
Angina Pectoris<br />
Total Lung Capacity<br />
Decreased<br />
Diarrhoea<br />
Dry Skin<br />
Visual Impairment<br />
Abnormal Dreams
Scratch<br />
Amnesia<br />
Vestibular Disorder<br />
Gastrointestinal Disorder<br />
Fatigue<br />
Sleep Apnoea Syndrome<br />
Arthralgia<br />
Weight Increased<br />
03-Apr-2012 09:37 AM Page: 606
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Cholesterol<br />
Increased<br />
Vitiligo Report Source Product Role Manufacturer Route Dose Duration<br />
Acrochordon Lariam PS Roche ORAL<br />
Angiotensin Converting Zoloft C UNKNOWN<br />
Enzyme Increased<br />
Upper-Airway Cough<br />
Syndrome<br />
Asthma<br />
Skin Disorder<br />
Ventricular Hypertrophy<br />
Bronchospasm<br />
Nocturia<br />
Bipolar Disorder<br />
Major Depression<br />
Psychotic Disorder<br />
Anxiety<br />
Dyspnoea<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Snoring<br />
Chest Discomfort<br />
Constipation<br />
Contusion<br />
Affective Disorder<br />
Tremor<br />
Date:05/10/05ISR Number: 4657892-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Hepatic Failure<br />
Visual Impairment<br />
Panic Attack<br />
Insomnia<br />
Nausea<br />
Depression<br />
Photosensitivity Reaction<br />
Anxiety<br />
Cough<br />
Depressed Level Of<br />
Consciousness<br />
Cognitive Disorder<br />
Dysarthria<br />
Dysphemia<br />
Tearfulness<br />
Hallucination<br />
Photophobia<br />
Vomiting<br />
Muscle Twitching<br />
Jaw Disorder<br />
Lacrimation Increased
Tinnitus<br />
Irritability<br />
Endolymphatic Hydrops<br />
Gilbert'S Syndrome<br />
Nervous System Disorder<br />
Weight Decreased<br />
Deafness<br />
Toxicity To Various<br />
03-Apr-2012 09:37 AM Page: 607
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Agents<br />
General Symptom<br />
Post-Traumatic Stress<br />
Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Thirst Lariam PS Roche ORAL 23 TABLETS<br />
Confusional State<br />
DISPENSED.<br />
Gait Disturbance Sudafed C<br />
Gastroenteritis<br />
Electroencephalogram<br />
Abnormal<br />
Migraine<br />
Decreased Appetite<br />
Conduction Disorder<br />
Dizziness<br />
Sensory Disturbance<br />
Paranoia<br />
Tremor<br />
Incontinence<br />
Disturbance In Attention<br />
Amnesia<br />
Tardive Dyskinesia<br />
Abnormal Dreams<br />
Water Pollution<br />
Stress<br />
Chest Pain<br />
Balance Disorder<br />
Aggression<br />
Anger<br />
Vestibular Disorder<br />
Asthenia<br />
Mania<br />
Viiith Nerve Lesion<br />
Nystagmus<br />
Hepato-Lenticular<br />
Degeneration<br />
Creutzfeldt-Jakob Disease<br />
Sinus Tachycardia<br />
Cranial Nerve Disorder<br />
Visual Field Defect<br />
Diarrhoea<br />
Suicidal Ideation<br />
Obsessive Thoughts<br />
Jaundice<br />
Mood Swings<br />
Motor Dysfunction<br />
Hyperacusis<br />
Respiratory Disorder<br />
Blood Bilirubin Increased<br />
Tooth Discolouration<br />
Accidental Exposure<br />
Binocular Eye Movement<br />
Disorder<br />
Disorientation<br />
Metal Poisoning
Date:05/13/05ISR Number: 4661718-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Anxiety<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 608
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Headache<br />
Arthralgia<br />
Bronchitis<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Dry Eye<br />
Insomnia<br />
Asthma<br />
Ventricular Hypertrophy<br />
Diastolic Dysfunction<br />
Restrictive Pulmonary<br />
Disease<br />
Sleep Apnoea Syndrome<br />
Diarrhoea<br />
Dry Skin<br />
Constipation<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Balance Disorder<br />
Memory Impairment<br />
Psychotic Disorder<br />
Mitral Valve Prolapse<br />
Somnolence<br />
Viral Infection<br />
Gastrooesophageal Reflux<br />
Disease<br />
Upper-Airway Cough<br />
Syndrome<br />
Abnormal Dreams<br />
Bipolar Disorder<br />
Aggression<br />
Dyspnoea<br />
Snoring<br />
Acrochordon<br />
Blood Glucose Increased<br />
Scratch<br />
Amnesia<br />
Vestibular Disorder<br />
Skin Disorder<br />
Angina Pectoris<br />
Weight Increased<br />
Exfoliative Rash<br />
Dry Mouth<br />
Rhinitis<br />
Haemorrhoids<br />
Rash<br />
Sinus Tachycardia<br />
Visual Impairment<br />
Affective Disorder<br />
Confusional State<br />
Total Lung Capacity<br />
Decreased<br />
Cardiac Murmur
Blood Triglycerides<br />
Increased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Coordination Abnormal<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Tremor<br />
03-Apr-2012 09:37 AM Page: 609
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Gastrointestinal Disorder<br />
Fatigue<br />
Hypertension Report Source Product Role Manufacturer Route Dose Duration<br />
Bronchospasm Lariam PS Roche ORAL<br />
Blood Cholesterol Zoloft C UNKNOWN<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Dehydration<br />
Nocturia<br />
Chest Discomfort<br />
Oral Disorder<br />
Vitiligo<br />
Clumsiness<br />
Major Depression<br />
Contusion<br />
Schizoaffective Disorder<br />
Date:05/13/05ISR Number: 4662475-2Report Type:Direct Company Report #CTU 248485 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Idiopathic Larium 250 Mg PS ORAL 250 MG<br />
Hospitalization - Thrombocytopenic Purpura WEEKLY ORAL<br />
Initial or Prolonged<br />
Disability<br />
Date:05/16/05ISR Number: 4662466-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Agitation<br />
Vertigo<br />
Disorientation<br />
Dizziness<br />
Nervousness<br />
Paraesthesia<br />
Migraine Without Aura<br />
Diarrhoea<br />
Mood Swings<br />
Chest Pain<br />
Nystagmus<br />
Testicular Pain<br />
Sinus Disorder<br />
Low Density Lipoprotein<br />
Increased<br />
Night Sweats<br />
Anxiety Disorder<br />
Memory Impairment<br />
Amnesia<br />
Asthenia
Cluster Headache<br />
Weight Increased<br />
Vomiting<br />
Insomnia<br />
Psychotic Disorder<br />
Inner Ear Disorder<br />
Abdominal Pain<br />
Rash<br />
03-Apr-2012 09:37 AM Page: 610
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pain Of Skin<br />
Blood Pressure Increased<br />
Balance Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Upper Respiratory Tract Lariam PS Roche ORAL<br />
Infection Imodium C<br />
Nightmare Compazine C ON 18 APR<br />
Irritable Bowel Syndrome<br />
2001, THERAPY<br />
Disturbance In Attention<br />
REPORTED AS<br />
Eye Pain<br />
TWICE DAILY.<br />
Phonophobia Antibiotics Nos C<br />
Blood Cholesterol Ambien C<br />
Increased Prednisone C BY TAPER.<br />
Pharyngitis Streptococcal Valium C ON 18 APR<br />
Fatigue<br />
2001, THERAPY<br />
Headache<br />
REPORTED AS:<br />
Photophobia<br />
AS NEEDED.<br />
Abnormal Dreams Phenergan C<br />
Blood Urea Increased Biaxin C<br />
Nausea Advil C<br />
Vestibular Disorder Bactrim C<br />
Vision Blurred Antivert C<br />
Depression<br />
Colitis<br />
Hypoaesthesia<br />
Viral Labyrinthitis<br />
Blood Triglycerides<br />
Increased<br />
Toxicity To Various<br />
Agents<br />
Tinnitus<br />
Epididymitis<br />
Date:05/17/05ISR Number: 4663595-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-403695 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Ineffective Lariam PS Roche ORAL<br />
Malaria<br />
Date:05/18/05ISR Number: 4664622-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-384854 Age:37 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Feeling Abnormal<br />
Cold Sweat<br />
Liver Function Test<br />
Abnormal<br />
Chills<br />
Disorientation<br />
Agitation<br />
Fatigue
Skin Disorder<br />
Vertigo<br />
Heart Rate Increased<br />
Hyperhidrosis<br />
Anxiety<br />
Depression<br />
Back Pain<br />
Heart Rate Irregular<br />
03-Apr-2012 09:37 AM Page: 611
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paranoia<br />
Diarrhoea<br />
Insomnia Report Source Product Role Manufacturer Route Dose Duration<br />
Mood Swings Lariam PS Roche ORAL 3-4 PILLS<br />
Paraesthesia WERE TAKEN. 43 DAY<br />
Panic Attack<br />
Disturbance In Attention<br />
Balance Disorder<br />
Date:05/19/05ISR Number: 4666042-6Report Type:Expedited (15-DaCompany Report #CH-ROCHE-334363 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dyspnoea Lariam PS Roche ORAL<br />
Headache<br />
Memory Impairment<br />
Fatigue<br />
Asthenia<br />
Tachycardia<br />
Apathy<br />
Dizziness<br />
Date:05/19/05ISR Number: 4666059-1Report Type:Expedited (15-DaCompany Report #NO-ROCHE-404116 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fatigue Lariam PS Roche ORAL 22 DAY<br />
Dyspnoea<br />
Atrioventricular Block<br />
Complete<br />
Dizziness<br />
Atrioventricular Block<br />
Date:05/19/05ISR Number: 4667441-9Report Type:Direct Company Report #CTU 249136 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Mental Disorder <strong>Mefloquine</strong><br />
Disability Injury Unknown Unknown PS ORAL<br />
Other Serious Ill-Defined Disorder 2OR3XDAILY<br />
ORAL<br />
<strong>Mefloquine</strong><br />
Stronger Than First<br />
Dose Unknown SS ORAL STRONGER<br />
SAME ORAL<br />
Date:05/27/05ISR Number: 4676616-4Report Type:Expedited (15-DaCompany Report #GB-ROCHE-405127 Age: Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Speech Disorder Lariam PS Roche ORAL STARTED AFTER<br />
Initial or Prolonged Loss Of Consciousness VERAPAMIL.<br />
Blood Pressure Increased Lariam SS Roche ORAL<br />
Paraesthesia Verapamil SS Roche UNKNOWN STARTED<br />
BEFORE<br />
MEFLOQUINE<br />
(LARIAM).<br />
03-Apr-2012 09:37 AM Page: 612
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Verapamil SS Roche UNKNOWN<br />
Aspirin<br />
C<br />
Simvastatin<br />
C<br />
Date:06/02/05ISR Number: 4682080-1Report Type:Periodic Company Report #379157 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Weight Decreased Consumer Lariam (<strong>Mefloquine</strong><br />
Anxiety Hydrochloride) PS ORAL ORAL<br />
Paranoia<br />
Fear<br />
Date:06/02/05ISR Number: 4682082-5Report Type:Periodic Company Report #379218 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion Consumer Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) PS ORAL ORAL<br />
Date:06/02/05ISR Number: 4682085-0Report Type:Periodic Company Report #380190 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Diarrhoea Other Lariam (<strong>Mefloquine</strong><br />
Paranoia Hydrochloride) 250<br />
Fatigue Mg PS ORAL 1 DOSE FORM 1<br />
Depression<br />
PER WEEK ORAL<br />
Aggression<br />
Agitation<br />
Delusion<br />
Headache<br />
Completed Suicide<br />
Night Sweats<br />
Mood Swings<br />
Insomnia<br />
Date:06/02/05ISR Number: 4682086-2Report Type:Periodic Company Report #380226 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anger Other Lariam (<strong>Mefloquine</strong><br />
Nightmare Hydrochloride) 250<br />
Abdominal Pain Upper Mg PS ORAL 250 MG 1 PER<br />
Dizziness<br />
WEEK ORAL<br />
Fear<br />
Panic Attack<br />
Diarrhoea<br />
Physical Assault
Nausea<br />
03-Apr-2012 09:37 AM Page: 613
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/02/05ISR Number: 4682088-6Report Type:Periodic Company Report #380311 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Anger Other Lariam (<strong>Mefloquine</strong><br />
Psychotic Disorder Hydrochloride) 250<br />
Panic Attack Mg PS ORAL ORAL<br />
Completed Suicide<br />
Diarrhoea<br />
Physical Abuse<br />
Rash<br />
Date:06/02/05ISR Number: 4682089-8Report Type:Periodic Company Report #384553 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia Health Lariam (<strong>Mefloquine</strong><br />
Headache Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/02/05ISR Number: 4682090-4Report Type:Periodic Company Report #386934 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Health Lariam (<strong>Mefloquine</strong><br />
Hallucination Professional Hydrochloride) 250<br />
Anxiety Mg PS ORAL ORAL<br />
Date:06/02/05ISR Number: 4682091-6Report Type:Periodic Company Report #395642 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Other Lariam (<strong>Mefloquine</strong><br />
Imprisonment Hydrochloride) PS ORAL ORAL 213 DAY<br />
Paranoia<br />
Anxiety<br />
Theft<br />
Date:06/02/05ISR Number: 4682156-9Report Type:Periodic Company Report #369650 Age:49 YR Gender:Male I/FU:I<br />
Outcome<br />
Death<br />
PT<br />
Agitation<br />
Mood Swings<br />
Tinnitus<br />
Disorientation<br />
Self Esteem Decreased<br />
Hostility<br />
Fear
Completed Suicide<br />
Wound<br />
Fatigue<br />
Abnormal Dreams<br />
Memory Impairment<br />
Speech Disorder<br />
Communication Disorder<br />
Emotional Distress<br />
03-Apr-2012 09:37 AM Page: 614
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Sleep Apnoea Syndrome<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Other<br />
Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:06/02/05ISR Number: 4682159-4Report Type:Periodic Company Report #370361 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:06/02/05ISR Number: 4682161-2Report Type:Periodic Company Report #370897 Age:63 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypersensitivity Health Lariam (<strong>Mefloquine</strong><br />
Urticaria Professional Hydrochloride) PS ORAL 1 PER WEEK<br />
Rash<br />
ORAL<br />
Pruritus<br />
Date:06/02/05ISR Number: 4682163-6Report Type:Periodic Company Report #372533 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:06/02/05ISR Number: 4682166-1Report Type:Periodic Company Report #372536 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam (<strong>Mefloquine</strong><br />
Physical Assault Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
Date:06/02/05ISR Number: 4682167-3Report Type:Periodic Company Report #372585 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg PS ORAL ORAL
Date:06/02/05ISR Number: 4682176-4Report Type:Periodic Company Report #374396 Age: Gender:Unknown I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Deafness Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) 250<br />
Mg PS ORAL ORAL<br />
03-Apr-2012 09:37 AM Page: 615
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/02/05ISR Number: 4682196-XReport Type:Periodic Company Report #378593 Age:15 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Urticaria Health Lariam (<strong>Mefloquine</strong><br />
Professional Hydrochloride) PS ORAL 1 DOSE FORM 1<br />
PER 1 WEEK<br />
ORAL<br />
Date:06/02/05ISR Number: 4684026-9Report Type:Periodic Company Report #395651 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Mood Swings Other Lariam (<strong>Mefloquine</strong><br />
Deafness Hydrochloride) 250<br />
Tinnitus Mg PS ORAL 250 MG 1 PER<br />
Fatigue<br />
WEEK ORAL<br />
Completed Suicide<br />
Depression<br />
Psychotic Disorder<br />
Agitation<br />
Date:06/02/05ISR Number: 4684027-0Report Type:Periodic Company Report #398558 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Consumer Lariam (<strong>Mefloquine</strong><br />
Disturbance In Attention Hydrochloride) PS ORAL ORAL<br />
Amnesia<br />
Anxiety<br />
Date:06/02/05ISR Number: 4684029-4Report Type:Periodic Company Report #400091 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Nightmare Other Lariam (<strong>Mefloquine</strong><br />
Injury Hydrochloride) PS ORAL ORAL<br />
Sleep Disorder<br />
Personality Change<br />
Completed Suicide<br />
Weight Decreased<br />
Depression<br />
Date:06/03/05ISR Number: 4681183-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Nausea<br />
Diarrhoea<br />
Mood Swings
Vision Blurred<br />
Enteritis Infectious<br />
Lymphocyte Count<br />
Decreased<br />
Blood Triglycerides<br />
Increased<br />
Streptococcal Infection<br />
Mucous Membrane Disorder<br />
03-Apr-2012 09:37 AM Page: 616
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Night Sweats<br />
Photophobia<br />
Blood Pressure Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Epididymitis Lariam PS Roche ORAL<br />
Amnesia Imodium C<br />
Headache Compazine C ON 18 APR<br />
Psychotic Disorder<br />
2001, THERAPY<br />
Nystagmus<br />
REPORTED AS<br />
Testicular Pain<br />
TWICE DAILY.<br />
Dizziness Antibiotics Nos C<br />
Eye Pain Ambien C<br />
Colitis Prednisone C BY TAPER.<br />
Rash Valium C ON 18 APR<br />
Abnormal Dreams<br />
2001, THERAPY<br />
Vertigo<br />
REPORTED AS:<br />
Chest Pain<br />
AS NEEDED.<br />
Depression Phenergan C<br />
Agitation Biaxin C<br />
Insomnia Advil C<br />
Anxiety Disorder Bactrim C<br />
Memory Impairment Antivert C<br />
Vestibular Disorder<br />
Inner Ear Disorder<br />
Nervousness<br />
Pain Of Skin<br />
Blood Cholesterol<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Vomiting<br />
Fatigue<br />
Abdominal Pain<br />
Disturbance In Attention<br />
Phonophobia<br />
Blood Urea Increased<br />
Viral Labyrinthitis<br />
Balance Disorder<br />
Posture Abnormal<br />
Nightmare<br />
Disorientation<br />
Hypoaesthesia<br />
Monocyte Count Increased<br />
Upper Respiratory Tract<br />
Infection<br />
Tinnitus<br />
Irritable Bowel Syndrome<br />
Paraesthesia<br />
Burning Sensation<br />
Neutrophil Count<br />
Increased<br />
Nuclear Magnetic<br />
Resonance Imaging<br />
Abnormal
Activities Of Daily<br />
Living Impaired<br />
03-Apr-2012 09:37 AM Page: 617
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/03/05ISR Number: 4681185-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vestibular Disorder<br />
Hypertension<br />
Sinus Tachycardia<br />
Low Density Lipoprotein<br />
Increased<br />
Dry Mouth<br />
Constipation<br />
Upper-Airway Cough<br />
Syndrome<br />
Asthma<br />
Haemorrhoids<br />
Affective Disorder<br />
Productive Cough<br />
Cough<br />
Electrocardiogram T Wave<br />
Abnormal<br />
Pharyngeal Disorder<br />
Weight Decreased<br />
Rectal Haemorrhage<br />
Anal Fissure<br />
Road Traffic Accident<br />
Bronchitis<br />
Blood Cholesterol<br />
Increased<br />
Exfoliative Rash<br />
Major Depression<br />
Musculoskeletal<br />
Discomfort<br />
Mental Status Changes<br />
Hallucination, Auditory<br />
Sinusitis<br />
Amnesia<br />
Anxiety<br />
Dyspnoea<br />
Restrictive Pulmonary<br />
Disease<br />
Sleep Apnoea Syndrome<br />
Bronchospasm<br />
Insomnia<br />
Abnormal Dreams<br />
Bipolar Disorder<br />
Coordination Abnormal<br />
Body Temperature<br />
Increased<br />
Sputum Discoloured<br />
Myalgia<br />
Collagen-Vascular Disease<br />
Fatigue<br />
Total Lung Capacity<br />
Decreased
Arthralgia<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Oral Disorder<br />
Gastrooesophageal Reflux<br />
Disease<br />
Scratch<br />
Contusion<br />
03-Apr-2012 09:37 AM Page: 618
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Exercise Tolerance<br />
Decreased<br />
Sleep Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Jaw Disorder Lariam PS Roche ORAL<br />
Rectal Neoplasm Zoloft C UNKNOWN<br />
Anhedonia<br />
Psychomotor Hyperactivity<br />
Cognitive Disorder<br />
Post-Traumatic Neck<br />
Syndrome<br />
Skin Disorder<br />
Gastrointestinal Disorder<br />
Angina Pectoris<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Diarrhoea<br />
Snoring<br />
Acrochordon<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Blood Glucose Increased<br />
Memory Impairment<br />
Aggression<br />
Erythema<br />
Mitral Valve Prolapse<br />
Diastolic Dysfunction<br />
Dry Skin<br />
Chest Discomfort<br />
Vitiligo<br />
Visual Impairment<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Oedema<br />
Ventricular Hypertrophy<br />
Rash<br />
Weight Increased<br />
Cardiac Murmur<br />
Blood Triglycerides<br />
Increased<br />
Rhinitis<br />
Balance Disorder<br />
Schizoaffective Disorder<br />
Upper Respiratory Tract<br />
Infection<br />
Colitis<br />
Feelings Of Worthlessness<br />
Confusional State<br />
Depression<br />
Headache<br />
Somnolence<br />
Viral Infection<br />
Dehydration<br />
Dry Eye
Nocturia<br />
Clumsiness<br />
Psychotic Disorder<br />
Tremor<br />
Wheezing<br />
Blood Pressure Diastolic<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 619
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/03/05ISR Number: 4681187-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Visual Field Defect<br />
Suicidal Ideation<br />
Cough<br />
Muscle Twitching<br />
Mood Swings<br />
Confusional State<br />
Hyperacusis<br />
Mental Disorder<br />
Eye Movement Disorder<br />
Dizziness<br />
Cranial Nerve Disorder<br />
Anger<br />
General Symptom<br />
Endolymphatic Hydrops<br />
Thirst<br />
Gilbert'S Syndrome<br />
Conduction Disorder<br />
Tearfulness<br />
Sensory Disturbance<br />
Creutzfeldt-Jakob Disease<br />
Anxiety Disorder<br />
Energy Increased<br />
Paranoia<br />
Insomnia<br />
Depression<br />
Vestibular Disorder<br />
Photosensitivity Reaction<br />
Depressed Level Of<br />
Consciousness<br />
Migraine<br />
Gait Disturbance<br />
Dysphemia<br />
Nervous System Disorder<br />
Antisocial Behaviour<br />
Narcissistic Personality<br />
Disorder<br />
Hepatic Failure<br />
Panic Attack<br />
Nausea<br />
Disturbance In Attention<br />
Tardive Dyskinesia<br />
Respiratory Disorder<br />
Blood Bilirubin Increased<br />
Hepato-Lenticular<br />
Degeneration<br />
Abnormal Behaviour<br />
Photophobia<br />
Vomiting<br />
Aggression<br />
Tinnitus
Mania<br />
Water Pollution<br />
Binocular Eye Movement<br />
Disorder<br />
Rapid Eye Movements Sleep<br />
Abnormal<br />
Electrocardiogram St-T<br />
Change<br />
03-Apr-2012 09:37 AM Page: 620
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucination<br />
Balance Disorder<br />
Toxicity To Various Report Source Product Role Manufacturer Route Dose Duration<br />
Agents Lariam PS Roche ORAL 23 TABLETS<br />
Asthenia<br />
DISPENSED.<br />
Obsessive Thoughts Sudafed C<br />
Jaundice<br />
Post-Traumatic Stress<br />
Disorder<br />
Amyotrophic Lateral<br />
Sclerosis<br />
Electroencephalogram<br />
Abnormal<br />
Hyporeflexia<br />
Electrocardiogram<br />
Abnormal<br />
Tic<br />
Incontinence<br />
Amnesia<br />
Anxiety<br />
Irritability<br />
Lacrimation Increased<br />
Motor Dysfunction<br />
Abnormal Dreams<br />
Tooth Discolouration<br />
Affective Disorder<br />
Metal Poisoning<br />
Visual Impairment<br />
Tremor<br />
Diarrhoea<br />
Decreased Appetite<br />
Jaw Disorder<br />
Cognitive Disorder<br />
Dysarthria<br />
Weight Decreased<br />
Gastroenteritis<br />
Date:06/08/05ISR Number: 4687515-6Report Type:Direct Company Report #CTU 250678 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Affect Lability Lariam PS ORAL ONE TABLET<br />
Aggression WEEK ORAL<br />
Date:06/17/05ISR Number: 4692525-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-407146 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Lariam PS Roche ORAL 29 DAY<br />
Initial or Prolonged Fall Xalacom SS TOPICAL<br />
Glycosylated Haemoglobin Fenofibrate SS ORAL
Increased Captopril SS Roche ORAL<br />
Vision Blurred<br />
Atrioventricular Block<br />
Bradycardia<br />
03-Apr-2012 09:37 AM Page: 621
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/17/05ISR Number: 4693517-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Abdominal Pain<br />
Dyspnoea<br />
Headache<br />
Acrophobia<br />
Sensory Integrative<br />
Dysfunction<br />
Neck Pain<br />
Vertigo<br />
Insomnia<br />
Panic Attack<br />
Generalised Anxiety<br />
Disorder<br />
Sleep Disorder<br />
Dizziness<br />
Motion Sickness<br />
Vestibular Disorder<br />
Photophobia<br />
Weight Decreased<br />
Adjustment Disorder With<br />
Anxiety<br />
Sinus Bradycardia<br />
Migraine<br />
Fatigue<br />
Lactose Intolerance<br />
Abdominal Distension<br />
Hypoaesthesia<br />
Emotional Distress<br />
Diarrhoea<br />
Disorientation<br />
Self Esteem Decreased<br />
Disturbance In Attention<br />
Phobia<br />
Claustrophobia<br />
Back Pain<br />
Nasal Congestion<br />
Abdominal Pain Lower<br />
Bronchospasm<br />
Anxiety<br />
Heart Rate Increased<br />
Nausea<br />
Rash<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Heart Rate Irregular<br />
Irritable Bowel Syndrome<br />
Phobia Of Flying<br />
Musculoskeletal Stiffness<br />
Amnesia<br />
Sleep Disorder Due To<br />
General Medical
Condition, Hypersomnia<br />
Type<br />
Mood Disorder Due To A<br />
General Medical Condition<br />
Suicidal Ideation<br />
Depression<br />
Paraesthesia<br />
Hypersensitivity<br />
03-Apr-2012 09:37 AM Page: 622
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tinnitus<br />
Balance Disorder<br />
Throat Irritation Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL THE PATIENT<br />
HAD TAKEN 4-5<br />
TABLETS OF<br />
MEFLOQUINE IN<br />
TOTAL. 21 DAY<br />
Vaccine C OTHER 1 DAY<br />
Date:06/17/05ISR Number: 4693518-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Incontinence<br />
Diarrhoea<br />
Post-Traumatic Stress<br />
Disorder<br />
Endolymphatic Hydrops<br />
Thirst<br />
Dysarthria<br />
Hyperacusis<br />
Hepatic Failure<br />
Paranoia<br />
Amnesia<br />
Muscle Twitching<br />
Lacrimation Increased<br />
Gait Disturbance<br />
Visual Impairment<br />
Tremor<br />
Obsessive Thoughts<br />
Motor Dysfunction<br />
Photophobia<br />
Insomnia<br />
Vomiting<br />
Vestibular Disorder<br />
Photosensitivity Reaction<br />
Suicidal Ideation<br />
Depressed Level Of<br />
Consciousness<br />
Confusional State<br />
Nausea<br />
Anxiety<br />
Tardive Dyskinesia<br />
Abnormal Dreams<br />
Conduction Disorder<br />
Hallucination<br />
Panic Attack<br />
Aggression<br />
Cough<br />
Cognitive Disorder<br />
Cranial Nerve Disorder
Visual Field Defect<br />
Balance Disorder<br />
Anger<br />
Disturbance In Attention<br />
Asthenia<br />
General Symptom<br />
Tinnitus<br />
Jaundice<br />
03-Apr-2012 09:37 AM Page: 623
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dysphemia<br />
Gilbert'S Syndrome<br />
Depression Report Source Product Role Manufacturer Route Dose Duration<br />
Toxicity To Various Lariam PS Roche ORAL 23 TABLETS<br />
Agents<br />
DISPENSED.<br />
Irritability Sudafed C<br />
Mood Swings<br />
Migraine<br />
Decreased Appetite<br />
Jaw Disorder<br />
Mania<br />
Date:06/17/05ISR Number: 4693539-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-389470 Age:12 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Personality Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged Psychomotor Retardation<br />
Adjustment Disorder<br />
Tinnitus<br />
Bipolar I Disorder<br />
Psychotic Disorder<br />
Hearing Impaired<br />
Hallucination, Auditory<br />
Depression<br />
Suicidal Ideation<br />
Major Depression<br />
Insomnia<br />
Anxiety<br />
Headache<br />
Decreased Appetite<br />
Suicide Attempt<br />
Autonomic Nervous System<br />
Imbalance<br />
Agitation<br />
Date:06/20/05ISR Number: 4694445-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Abnormal Behaviour<br />
Vertigo<br />
Bronchospasm<br />
Productive Cough<br />
Chest Discomfort<br />
Headache<br />
Hyperhidrosis<br />
Cognitive Disorder<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder
Respiratory Rate<br />
Increased<br />
Convulsion<br />
Nocturia<br />
Myalgia<br />
Eosinophilia<br />
Arthralgia<br />
Disturbance In Attention<br />
03-Apr-2012 09:37 AM Page: 624
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucination, Visual<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder<br />
Fall<br />
Decreased Activity<br />
Dehydration<br />
Musculoskeletal Stiffness<br />
Tinnitus<br />
Agitation<br />
Mood Swings<br />
Depression<br />
Fatigue<br />
Joint Dislocation<br />
Psychiatric Symptom<br />
Hypoaesthesia<br />
Ventricular Extrasystoles<br />
Bacterial Infection<br />
Dizziness<br />
Visual Impairment<br />
Asthenia<br />
Respiratory Disorder<br />
Meniscus Lesion<br />
Asthma<br />
Oxygen Saturation<br />
Decreased<br />
Coronary Artery Disease<br />
Homicidal Ideation<br />
Hiatus Hernia<br />
Panic Attack<br />
Haemophilus Infection<br />
Palpitations<br />
Anxiety<br />
Balance Disorder<br />
Suicidal Ideation<br />
Selective Iga<br />
Immunodeficiency<br />
Cranial Nerve Disorder<br />
Dysthymic Disorder<br />
Bundle Branch Block Right<br />
Eye Movement Disorder<br />
Contusion<br />
Rhinitis Allergic<br />
Insomnia<br />
Memory Impairment<br />
Panic Disorder<br />
Hypertension<br />
Psychotic Disorder<br />
Self-Injurious Ideation<br />
Musculoskeletal Pain<br />
Mitral Valve<br />
Calcification<br />
Humerus Fracture
Gait Disturbance<br />
Ulna Fracture<br />
Aggression<br />
Dyspnoea<br />
Sleep Apnoea Syndrome<br />
Tremor<br />
Mitral Valve Incompetence<br />
Exostosis<br />
03-Apr-2012 09:37 AM Page: 625
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Weight Decreased<br />
Nystagmus<br />
Ligament Sprain Report Source Product Role Manufacturer Route Dose Duration<br />
Pyrexia Lariam PS Roche ORAL<br />
Rash Papular Primaquine C<br />
Weight Increased Cipro C<br />
Deafness<br />
Coordination Abnormal<br />
Upper Respiratory Tract<br />
Infection<br />
Blood Pressure Increased<br />
Chest Pain<br />
Post-Traumatic Stress<br />
Disorder<br />
Thinking Abnormal<br />
Toxicity To Various<br />
Agents<br />
Antinuclear Antibody<br />
Positive<br />
Rheumatoid Factor<br />
Increased<br />
Ill-Defined Disorder<br />
Amnesia<br />
Snoring<br />
Nightmare<br />
Date:06/23/05ISR Number: 4698813-4Report Type:Expedited (15-DaCompany Report #05-06-1067 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Speech Disorder Foreign Verapamil Hcl Er<br />
Initial or Prolonged Blood Pressure Increased Other -Ipi PS Ipi<br />
Paraesthesia Lariam SS ORAL ORAL<br />
Aspirin<br />
C<br />
Simvastatin<br />
C<br />
Date:06/24/05ISR Number: 4699090-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-405127 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paraesthesia Lariam PS Roche ORAL STARTED AFTER<br />
Initial or Prolonged Speech Disorder VERAPAMIL.<br />
Loss Of Consciousness Lariam SS Roche ORAL<br />
Blood Pressure Increased Verapamil SS Roche UNKNOWN STARTED<br />
BEFORE<br />
MEFLOQUINE<br />
(LARIAM).<br />
Verapamil SS Roche UNKNOWN<br />
Aspirin<br />
C<br />
Simvastatin<br />
C
Date:06/27/05ISR Number: 4700390-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Sleep Apnoea Syndrome<br />
Dehydration<br />
Rhinitis<br />
03-Apr-2012 09:37 AM Page: 626
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Clumsiness<br />
Upper Respiratory Tract<br />
Infection<br />
Listless<br />
Dyspnoea Exertional<br />
Adjustment Disorder<br />
Anxiety<br />
Dyspnoea<br />
Vestibular Disorder<br />
Somnolence<br />
Nocturia<br />
Blood Glucose Increased<br />
Memory Impairment<br />
Psychotic Disorder<br />
Gastrointestinal Disorder<br />
Ventricular Hypertrophy<br />
Arthralgia<br />
Weight Increased<br />
Blood Triglycerides<br />
Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Upper-Airway Cough<br />
Syndrome<br />
Aggression<br />
Psychomotor Hyperactivity<br />
Confusional State<br />
Diastolic Dysfunction<br />
Viral Infection<br />
Insomnia<br />
Visual Impairment<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Contusion<br />
Schizoaffective Disorder<br />
Condition Aggravated<br />
Rectal Haemorrhage<br />
Skin Disorder<br />
Fatigue<br />
Total Lung Capacity<br />
Decreased<br />
Blood Cholesterol<br />
Increased<br />
Dry Skin<br />
Snoring<br />
Acrochordon<br />
Abnormal Dreams<br />
Affective Disorder<br />
Tremor<br />
Multiple Allergies<br />
Depression<br />
Headache<br />
Bronchospasm
Diarrhoea<br />
Exfoliative Rash<br />
Bipolar Disorder<br />
Scratch<br />
Neck Pain<br />
Drug Intolerance<br />
Amnesia<br />
Mitral Valve Prolapse<br />
03-Apr-2012 09:37 AM Page: 627
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Cardiac Murmur<br />
Sinus Tachycardia<br />
Low Density Lipoprotein Report Source Product Role Manufacturer Route Dose Duration<br />
Increased Lariam PS Roche ORAL<br />
Dry Mouth Zoloft C UNKNOWN<br />
Chest Discomfort<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Balance Disorder<br />
Nausea<br />
Hypertension<br />
Angina Pectoris<br />
Restrictive Pulmonary<br />
Disease<br />
Rash<br />
Bronchitis<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Dry Eye<br />
Constipation<br />
Oral Disorder<br />
Vitiligo<br />
Gastrooesophageal Reflux<br />
Disease<br />
Asthma<br />
Haemorrhoids<br />
Coordination Abnormal<br />
Major Depression<br />
Date:06/28/05ISR Number: 4701483-XReport Type:Expedited (15-DaCompany Report #SE-ROCHE-407993 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Lariam PS Roche ORAL 9 DAY<br />
Visual Impairment Lexinor C<br />
Amnesia<br />
Date:06/28/05ISR Number: 4702207-2Report Type:Direct Company Report #CTU 252039 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam Roche PS Roche<br />
Memory Impairment<br />
Headache<br />
Dizziness<br />
Date:06/29/05ISR Number: 4702966-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-408409 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Asthenia Lariam PS Roche ORAL THE PATIENT<br />
Initial or Prolonged Hyponatraemia TOOK JUST ONE<br />
Feeling Drunk<br />
DOSE OF THREE<br />
Psychotic Disorder<br />
TABLETS AT<br />
Fatigue<br />
ONCE.<br />
03-Apr-2012 09:37 AM Page: 628
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/05/05ISR Number: 4706612-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Photophobia<br />
Vestibular Disorder<br />
Suicidal Ideation<br />
Confusional State<br />
Motor Dysfunction<br />
Abnormal Dreams<br />
Gastroenteritis<br />
Impulsive Behaviour<br />
Hallucination<br />
Tinnitus<br />
Irritability<br />
Thirst<br />
Hyperacusis<br />
Paranoia<br />
Tremor<br />
Nausea<br />
Vomiting<br />
Anger<br />
Muscle Twitching<br />
Amnesia<br />
Photosensitivity Reaction<br />
Toxicity To Various<br />
Agents<br />
Cranial Nerve Disorder<br />
Balance Disorder<br />
Panic Attack<br />
Mood Swings<br />
Migraine<br />
Dysarthria<br />
Hepatic Failure<br />
Aggression<br />
Anxiety<br />
Asthenia<br />
Gait Disturbance<br />
Tooth Discolouration<br />
Ear Infection<br />
Visual Impairment<br />
Diarrhoea<br />
Depression<br />
General Symptom<br />
Cough<br />
Obsessive Thoughts<br />
Depressed Level Of<br />
Consciousness<br />
Jaw Disorder<br />
Tardive Dyskinesia<br />
Lacrimation Increased<br />
Dysphemia<br />
Mania<br />
Conduction Disorder
Visual Field Defect<br />
Insomnia<br />
Incontinence<br />
Disturbance In Attention<br />
Jaundice<br />
Post-Traumatic Stress<br />
Disorder<br />
Endolymphatic Hydrops<br />
03-Apr-2012 09:37 AM Page: 629
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Decreased Appetite<br />
Cognitive Disorder<br />
Gilbert'S Syndrome Report Source Product Role Manufacturer Route Dose Duration<br />
Electrocardiogram St-T Lariam PS Roche ORAL 23 TABLETS<br />
Change<br />
DISPENSED.<br />
Attention Sudafed C<br />
Deficit/Hyperactivity<br />
Disorder<br />
Daydreaming<br />
Date:07/08/05ISR Number: 4711571-XReport Type:Direct Company Report #CTU 252820 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion <strong>Mefloquine</strong> PS 250 MG Q WEEK<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:07/12/05ISR Number: 4712969-6Report Type:Direct Company Report #CTU 253019 Age:7 MON Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Petechiae <strong>Mefloquine</strong> 250 Mg<br />
Haematochezia Tab PS ORAL 1/8 TAB WEEK<br />
Platelet Count Decreased<br />
ORAL<br />
Bactrim<br />
C<br />
Date:07/13/05ISR Number: 4713176-3Report Type:Direct Company Report #CTU 253105 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety <strong>Mefloquine</strong> PS 250 MG Q<br />
Depression<br />
WEEKLY<br />
Tri-Sprintec<br />
C<br />
Luvox<br />
C<br />
Date:07/13/05ISR Number: 4713437-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-402425 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Liver Function Test Lariam PS Roche ORAL 3 YR<br />
Abnormal Alcohol SS UNKNOWN<br />
Hepatocellular Injury<br />
Alcohol Use
Date:07/14/05ISR Number: 4714346-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-391660 Age: Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Initial Insomnia<br />
Disturbance In Attention<br />
Amoebiasis<br />
Coordination Abnormal<br />
Hypersomnia<br />
03-Apr-2012 09:37 AM Page: 630
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Erectile Dysfunction<br />
Social Avoidant Behaviour<br />
Judgement Impaired Report Source Product Role Manufacturer Route Dose Duration<br />
Bradycardia Lariam PS Roche UNKNOWN<br />
Self Esteem Decreased Diazepam C<br />
Generalised Anxiety Lorazepam C<br />
Disorder Seroquel C<br />
Mood Swings Gabapentin C<br />
Suicidal Ideation Effexor Xr C<br />
Fatigue<br />
Crying<br />
Somatisation Disorder<br />
Varicocele<br />
Hypotension<br />
Mitral Valve Prolapse<br />
Libido Decreased<br />
Amnesia<br />
Inadequate Diet<br />
Psychomotor Retardation<br />
Scrotal Varicose Veins<br />
Memory Impairment<br />
Mental Disorder<br />
Palpitations<br />
Restlessness<br />
Aphasia<br />
Dysarthria<br />
Blood Pressure Increased<br />
Viral Infection<br />
Major Depression<br />
Psychotic Disorder<br />
Terminal Insomnia<br />
Adjustment Disorder<br />
Panic Attack<br />
Self Injurious Behaviour<br />
Anhedonia<br />
Feeling Jittery<br />
Flat Affect<br />
Date:07/14/05ISR Number: 4714347-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Hepatic Failure<br />
Cranial Nerve Disorder<br />
Visual Impairment<br />
Tremor<br />
Photosensitivity Reaction<br />
Asthenia<br />
Endolymphatic Hydrops<br />
Tardive Dyskinesia<br />
Motor Dysfunction<br />
Hyperacusis
Hallucination<br />
Insomnia<br />
Vomiting<br />
Anxiety<br />
Cough<br />
Confusional State<br />
Panic Attack<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 631
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Post-Traumatic Stress<br />
Disorder<br />
Dysphemia Report Source Product Role Manufacturer Route Dose Duration<br />
Gilbert'S Syndrome Lariam PS Roche ORAL 23 TABLETS<br />
Balance Disorder<br />
DISPENSED.<br />
Aggression Sudafed C<br />
Amnesia<br />
Vestibular Disorder<br />
Jaw Disorder<br />
Gait Disturbance<br />
Paranoia<br />
Nausea<br />
Anger<br />
Muscle Twitching<br />
Mood Swings<br />
Migraine<br />
Lacrimation Increased<br />
Irritability<br />
Depressed Level Of<br />
Consciousness<br />
Thirst<br />
Photophobia<br />
Disturbance In Attention<br />
Toxicity To Various<br />
Agents<br />
Suicidal Ideation<br />
General Symptom<br />
Obsessive Thoughts<br />
Cognitive Disorder<br />
Dysarthria<br />
Conduction Disorder<br />
Visual Field Defect<br />
Incontinence<br />
Diarrhoea<br />
Tinnitus<br />
Jaundice<br />
Decreased Appetite<br />
Mania<br />
Abnormal Dreams<br />
Bipolar Disorder<br />
Date:07/15/05ISR Number: 4715449-7Report Type:Direct Company Report #CTU 253311 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Lariam PS ORAL ORAL<br />
Initial or Prolonged Screaming<br />
Required<br />
Depression<br />
Intervention to<br />
Aggression<br />
Prevent Permanent Abnormal Behaviour<br />
Impairment/Damage
Date:07/18/05ISR Number: 4718297-7Report Type:Direct Company Report #CTU 253463 Age:28 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
03-Apr-2012 09:37 AM Page: 632
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Other Serious<br />
Required<br />
Intervention to PT Report Source Product Role Manufacturer Route Dose Duration<br />
Prevent Permanent Depression Larium La Roche PS La Roche ORAL 1<br />
Impairment/Damage Sensory Loss WEEKLY ORAL<br />
Anxiety<br />
Brain Injury<br />
Muscle Twitching<br />
Monoplegia<br />
Tremor<br />
Date:07/18/05ISR Number: 4718303-XReport Type:Direct Company Report #CTU 253466 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Tinnitus <strong>Mefloquine</strong> Unknown<br />
Influenza Like Illness Generic PS ORAL 1 PER WEEK<br />
Fatigue<br />
9 WEEKS<br />
Diarrhoea<br />
ORAL<br />
Abdominal Pain Upper Gingko Biloba C<br />
Emotional Disorder Lexapro C<br />
Reading Disorder<br />
Pain<br />
Anxiety<br />
Weight Decreased<br />
Abdominal Distension<br />
Pyrexia<br />
Flatulence<br />
Disturbance In Attention<br />
Amnesia<br />
Myoclonus<br />
Dyspepsia<br />
Malaise<br />
Visual Impairment<br />
Dizziness<br />
Feeling Abnormal<br />
Educational Problem<br />
Stress<br />
Depression<br />
Cognitive Disorder<br />
Date:07/21/05ISR Number: 4723398-3Report Type:Direct Company Report #CTU 253857 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Drug Dependence Lariam Weekly Sr<br />
Other Serious Eye Pain Roche PS Roche ORAL 1 WEEKLY ORAL<br />
Mood Swings Verapamil C<br />
Headache
Date:07/22/05ISR Number: 4722019-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Insomnia<br />
Fatigue<br />
Tinnitus<br />
Psychotic Disorder<br />
03-Apr-2012 09:37 AM Page: 633
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Testicular Pain<br />
Irritable Bowel Syndrome<br />
Eye Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Mood Swings Lariam PS Roche ORAL<br />
Anxiety Disorder Imodium C<br />
Abdominal Pain Compazine C ON 18 APR<br />
Photophobia<br />
2001, THERAPY<br />
Nightmare<br />
REPORTED AS<br />
Nystagmus<br />
TWICE DAILY.<br />
Inner Ear Disorder Antibiotics Nos C<br />
Pain Of Skin Ambien C<br />
Vomiting Prednisone C BY TAPER.<br />
Rash Valium C ON 18 APR<br />
Nervousness<br />
2001, THERAPY<br />
Phonophobia<br />
REPORTED AS:<br />
Night Sweats<br />
AS NEEDED.<br />
Agitation Phenergan C<br />
Nausea Biaxin C<br />
Vertigo Advil C<br />
Headache Bactrim C<br />
Hypoaesthesia Antivert C<br />
Blood Pressure Increased<br />
Memory Impairment<br />
Vestibular Disorder<br />
Chest Pain<br />
Colitis<br />
Disorientation<br />
Dizziness<br />
Diarrhoea<br />
Amnesia<br />
Vision Blurred<br />
Depression<br />
Disturbance In Attention<br />
Date:07/28/05ISR Number: 4727555-1Report Type:Direct Company Report #CTU 254655 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam Unsure<br />
Required Lethargy Roche PS Roche ORAL DAILY ORAL<br />
Intervention to Hallucination Birth Control Pills C<br />
Prevent Permanent<br />
Impairment/Damage<br />
Depression<br />
Paranoia<br />
Nightmare<br />
Date:08/02/05ISR Number: 4731828-6Report Type:Direct Company Report #CTU 255169 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Lariam PS ORAL 1 WEEK<br />
Motion Sickness<br />
ORAL
Dizziness<br />
Mood Altered<br />
Circadian Rhythm Sleep<br />
Disorder<br />
Abnormal Dreams<br />
Anger<br />
Insomnia<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 634
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/03/05ISR Number: 4734187-8Report Type:Direct Company Report #CTU 255326 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Anomaly <strong>Mefloquine</strong> 250mg<br />
Other Serious Pregnancy Qweek PS 250MG Q WEEK<br />
Anencephaly<br />
X 100 WKS<br />
Hallucination<br />
<strong>Mefloquine</strong> 1250mg<br />
Maternal Drugs Affecting Stat Dose SS 1250 MG STAT<br />
Foetus<br />
X 1 DOSE<br />
Date:08/04/05ISR Number: 4734436-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-397065 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Panic Disorder STATED AS<br />
Disability Paraesthesia WEEKLY. 47 DAY<br />
Anxiety Disorder<br />
Ear Pain<br />
Decreased Appetite<br />
Tinnitus<br />
Chills<br />
Headache<br />
Mood Swings<br />
Depression<br />
Diarrhoea<br />
Skin Disorder<br />
Paranoia<br />
Dizziness<br />
Hearing Impaired<br />
Panic Attack<br />
Abdominal Pain<br />
Hyperhidrosis<br />
Fatigue<br />
Toxicity To Various<br />
Agents<br />
Confusional State<br />
Memory Impairment<br />
Arthralgia<br />
Insomnia<br />
Visual Impairment<br />
Chest Pain<br />
Agitation<br />
Myalgia<br />
Date:08/17/05ISR Number: 4746842-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Insomnia<br />
Disorientation
Vestibular Disorder<br />
Nystagmus<br />
Dizziness<br />
Pain Of Skin<br />
Agitation<br />
Fatigue<br />
Amnesia<br />
Eye Pain<br />
03-Apr-2012 09:37 AM Page: 635
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Rash<br />
Headache<br />
Chest Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Irritable Bowel Syndrome Lariam PS Roche ORAL<br />
Nightmare Imodium C<br />
Tinnitus Compazine C ON 18 APR<br />
Disturbance In Attention<br />
2001, THERAPY<br />
Hypoaesthesia<br />
REPORTED AS<br />
Memory Impairment<br />
TWICE DAILY.<br />
Vertigo Antibiotics Nos C<br />
Psychotic Disorder Ambien C<br />
Inner Ear Disorder Prednisone C BY TAPER.<br />
Abdominal Pain Valium C ON 18 APR<br />
Enteritis Infectious<br />
2001, THERAPY<br />
Night Sweats<br />
REPORTED AS:<br />
Nausea<br />
AS NEEDED.<br />
Diarrhoea Phenergan C<br />
Vision Blurred Biaxin C<br />
Nervousness Advil C<br />
Vomiting Bactrim C<br />
Mood Swings Antivert C<br />
Testicular Pain<br />
Depression<br />
Colitis<br />
Phonophobia<br />
Blood Pressure Increased<br />
Anxiety Disorder<br />
Photophobia<br />
Date:08/22/05ISR Number: 4749682-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-414147 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Lariam PS Roche ORAL 1 DAY<br />
Disturbance In Attention Nitrofurantoin C ORAL<br />
Vision Blurred<br />
Dizziness<br />
Paraesthesia<br />
Disorientation<br />
Date:08/23/05ISR Number: 4750604-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-408409 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyponatraemia Lariam PS Roche ORAL THE PATIENT<br />
Initial or Prolonged Asthenia TOOK JUST ONE<br />
Psychotic Disorder<br />
DOSE OF THREE<br />
Fatigue<br />
TABLETS AT<br />
Blood Creatine<br />
ONCE.<br />
Phosphokinase Increased<br />
Feeling Drunk
Date:08/26/05ISR Number: 4754048-8Report Type:Expedited (15-DaCompany Report #2005-BP-13941RO Age:20 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Nausea<br />
Electrocardiogram Qt<br />
03-Apr-2012 09:37 AM Page: 636
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Prolonged<br />
Paraesthesia<br />
Headache<br />
Dizziness Report Source Product Role Manufacturer Route Dose Duration<br />
Depressed Level Of Literature <strong>Mefloquine</strong><br />
Consciousness Health (<strong>Mefloquine</strong>) PS ORAL 240 MG ONCE<br />
Hypotension Professional WEEKLY (250<br />
MG, 1 IN 1<br />
WK), PO<br />
Date:08/29/05ISR Number: 4754920-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Lariam PS Roche ORAL<br />
Memory Impairment Imodium C<br />
Nystagmus Compazine C ON 18 APR<br />
Irritable Bowel Syndrome<br />
2001, THERAPY<br />
Rash<br />
REPORTED AS<br />
Vomiting<br />
TWICE DAILY.<br />
Headache Antibiotics Nos C<br />
Abdominal Pain Ambien C<br />
Night Sweats Prednisone C BY TAPER.<br />
Disorientation Valium C ON 18 APR<br />
Vestibular Disorder<br />
2001, THERAPY<br />
Disturbance In Attention<br />
REPORTED AS:<br />
Eye Pain<br />
AS NEEDED.<br />
Colitis Phenergan C<br />
Blood Pressure Increased Biaxin C<br />
Mood Swings Advil C<br />
Insomnia Bactrim C<br />
Nightmare Antivert C<br />
Agitation<br />
Anxiety Disorder<br />
Depression<br />
Amnesia<br />
Tinnitus<br />
Inner Ear Disorder<br />
Testicular Pain<br />
Hypoaesthesia<br />
Nervousness<br />
Phonophobia<br />
Diarrhoea<br />
Fatigue<br />
Chest Pain<br />
Vision Blurred<br />
Photophobia<br />
Pain Of Skin<br />
Vertigo<br />
Psychotic Disorder<br />
Dizziness
Date:08/29/05ISR Number: 4754954-4Report Type:Expedited (15-DaCompany Report #SE-ROCHE-392506 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Amnesia<br />
Stress<br />
Grand Mal Convulsion<br />
Fatigue<br />
03-Apr-2012 09:37 AM Page: 637
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Headache<br />
Disturbance In Attention<br />
Palpitations Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Lexinor C ORAL<br />
Malarone C ORAL<br />
Date:08/30/05ISR Number: 4756595-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-415094 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Fall Lariam PS Roche ORAL THE PATIENT<br />
Hospitalization - Foaming At Mouth DID NOT TAKE<br />
Initial or Prolonged Coma ANY OTHER<br />
Postictal State DRUGS. 1 DAY<br />
Loss Of Consciousness<br />
Date:08/31/05ISR Number: 4759812-7Report Type:Direct Company Report #CTU 257313 Age:21 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Legal Problem Lariam 250 Mg<br />
Other Serious Abnormal Behaviour Hoffman Laroche PS Hoffman Laroche BUCCAL 250 MG 1/WEEK<br />
Stress BUCCAL 6 WK<br />
Treatment Noncompliance<br />
Mania<br />
Nervousness<br />
Economic Problem<br />
Insomnia<br />
Heart Rate Increased<br />
Bipolar Disorder<br />
Loss Of Employment<br />
Educational Problem<br />
Depression<br />
Date:09/02/05ISR Number: 4763450-XReport Type:Expedited (15-DaCompany Report #2005-BP-14576RO Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insulinoma Literature <strong>Mefloquine</strong><br />
Hypoglycaemia Health (<strong>Mefloquine</strong>) PS ONCE/WEEK<br />
Insulin C-Peptide<br />
Professional<br />
Increased<br />
Neuroglycopenia<br />
Blood Insulin Increased<br />
Date:09/06/05ISR Number: 4762148-1Report Type:Direct Company Report #CTU 257623 Age: Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety <strong>Mefloquine</strong> 250 Mg<br />
Hospitalization - Feeling Abnormal Sandoz PS Sandoz ORAL 1 TABLET -250<br />
Initial or Prolonged Abnormal Behaviour MG ONCE A<br />
Required Homicidal Ideation WEEK PO<br />
Intervention to<br />
Creutzfeldt-Jakob Disease<br />
Prevent Permanent Hallucination, Visual<br />
Impairment/Damage Self Injurious Behaviour<br />
03-Apr-2012 09:37 AM Page: 638
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/08/05ISR Number: 4763724-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-415971 Age:61 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Amyotrophy Lariam PS Roche ORAL<br />
Respiratory Failure Engerix B SS INTRAMUSCULAR THE PATIENT<br />
Amyotrophic Lateral<br />
RECEIVED<br />
Sclerosis<br />
THREE<br />
Upper Motor Neurone<br />
INJECTIONS IN<br />
Lesion 1995 (MARCH, 189 DAY<br />
Date:09/09/05ISR Number: 4764962-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-415763 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Panic Reaction Lariam PS Roche ORAL<br />
Disturbance In Attention Dpt C<br />
Psychotic Disorder<br />
Hepatitis<br />
Depression Vaccination C<br />
Date:09/14/05ISR Number: 4768435-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Gastrointestinal Disorder<br />
Arthralgia<br />
Blood Triglycerides<br />
Increased<br />
Diarrhoea<br />
Dry Mouth<br />
Nocturia<br />
Gastrooesophageal Reflux<br />
Disease<br />
Visual Impairment<br />
Abnormal Dreams<br />
Major Depression<br />
Scratch<br />
Anxiety<br />
Hypertension<br />
Diastolic Dysfunction<br />
Angina Pectoris<br />
Somnolence<br />
Viral Infection<br />
Cardiac Murmur<br />
Haemorrhoids<br />
Memory Impairment<br />
Vestibular Disorder<br />
Depression<br />
Headache<br />
Sleep Apnoea Syndrome<br />
Bronchospasm<br />
Ventricular Hypertrophy
Exfoliative Rash<br />
Dry Skin<br />
Contusion<br />
Schizoaffective Disorder<br />
Tremor<br />
Mitral Valve Prolapse<br />
Fatigue<br />
Restrictive Pulmonary<br />
03-Apr-2012 09:37 AM Page: 639
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disease<br />
Snoring<br />
Constipation<br />
Oral Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Bipolar Disorder Lariam PS Roche ORAL<br />
Skin Disorder Zoloft C UNKNOWN<br />
Sinus Tachycardia<br />
Low Density Lipoprotein<br />
Increased<br />
Vitiligo<br />
Rhinitis<br />
Balance Disorder<br />
Coordination Abnormal<br />
Affective Disorder<br />
Amnesia<br />
Confusional State<br />
Blood Cholesterol<br />
Increased<br />
Dehydration<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Insomnia<br />
Clumsiness<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Psychotic Disorder<br />
Aggression<br />
Dyspnoea<br />
Total Lung Capacity<br />
Decreased<br />
Rash<br />
Bronchitis<br />
Weight Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Dry Eye<br />
Chest Discomfort<br />
Acrochordon<br />
Blood Glucose Increased<br />
Upper-Airway Cough<br />
Syndrome<br />
Asthma<br />
Date:09/14/05ISR Number: 4770717-8Report Type:Direct Company Report #CTU 258158 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Nightmare <strong>Mefloquine</strong> PS ONCE A WEEK<br />
Completed Suicide<br />
Depression
Date:09/15/05ISR Number: 4770906-2Report Type:Expedited (15-DaCompany Report #SE-ROCHE-407993 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Visual Impairment<br />
Convulsion<br />
Disturbance In Attention<br />
03-Apr-2012 09:37 AM Page: 640
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Amnesia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL 9 DAY<br />
Lexinor<br />
C<br />
Date:09/22/05ISR Number: 4778888-4Report Type:Expedited (15-DaCompany Report #343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vestibular Disorder<br />
Bipolar Disorder<br />
Major Depression<br />
Schizoaffective Disorder<br />
Arthralgia<br />
Blood Triglycerides<br />
Increased<br />
Diarrhoea<br />
Acrochordon<br />
Peripheral Sensory<br />
Neuropathy<br />
Exercise Tolerance<br />
Decreased<br />
Hypersensitivity<br />
Upper Respiratory Tract<br />
Infection<br />
Somnolence<br />
Contusion<br />
Viral Infection<br />
Bronchospasm<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Exfoliative Rash<br />
Productive Cough<br />
Cough<br />
Blood Pressure Systolic<br />
Increased<br />
Allergy To Animal<br />
Neck Pain<br />
Drug Tolerance Decreased<br />
Dyspnoea Exertional<br />
Dyspnoea<br />
Fatigue<br />
Diastolic Dysfunction<br />
Affective Disorder<br />
Blood Cholesterol<br />
Increased<br />
Snoring<br />
Vitiligo<br />
Rhinitis<br />
Sleep Apnoea Syndrome<br />
Spirometry Abnormal
Adjustment Disorder<br />
Scratch<br />
Bronchitis<br />
Weight Increased<br />
Dry Mouth<br />
Insomnia<br />
Pyrexia<br />
Listless<br />
03-Apr-2012 09:37 AM Page: 641
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Electrocardiogram T Wave<br />
Abnormal<br />
Amnesia Report Source Product Role Manufacturer Route Dose Duration<br />
Depression Consumer Lariam (<strong>Mefloquine</strong><br />
Ventricular Hypertrophy Other Hydrochloride) 250<br />
Abnormal Dreams Mg PS ORAL 250 MG 1 PER<br />
Sinus Tachycardia<br />
1 WEEK ORAL<br />
Dry Skin<br />
Zoloft (Sertraline<br />
Nocturia Hydrochloride) C<br />
Sputum Discoloured<br />
Total Cholesterol/Hdl<br />
Ratio Increased<br />
Restlessness<br />
Confusional State<br />
Skin Disorder<br />
Gastrointestinal Disorder<br />
Headache<br />
Clumsiness<br />
Asthma<br />
Coordination Abnormal<br />
Depressive Symptom<br />
Low Density Lipoprotein<br />
Increased<br />
Dry Eye<br />
Blood Glucose Increased<br />
Upper-Airway Cough<br />
Syndrome<br />
Haemorrhoids<br />
Aggression<br />
Wheezing<br />
Allergy Test Positive<br />
Mitral Valve Prolapse<br />
Hypertension<br />
Angina Pectoris<br />
Visual Impairment<br />
Balance Disorder<br />
Psychotic Disorder<br />
Rash<br />
Dehydration<br />
Chest Discomfort<br />
Myalgia<br />
Chest X-Ray Abnormal<br />
Mycotic Allergy<br />
Anxiety<br />
Lung Disorder<br />
Memory Impairment<br />
Cardiac Murmur<br />
Constipation<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Gastrooesophageal Reflux<br />
Disease<br />
Tremor
Nervous System Disorder<br />
03-Apr-2012 09:37 AM Page: 642
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/26/05ISR Number: 4778694-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-384854 Age:37 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Lariam PS Roche ORAL 3-4 PILLS<br />
Panic Attack WERE TAKEN. 43 DAY<br />
Balance Disorder<br />
Paranoia<br />
Insomnia<br />
Disorientation<br />
Feeling Abnormal<br />
Fatigue<br />
Liver Function Test<br />
Abnormal<br />
Heart Rate Increased<br />
Hyperhidrosis<br />
Depression<br />
Disturbance In Attention<br />
Cold Sweat<br />
Vertigo<br />
Alcohol Use<br />
Mood Swings<br />
Alanine Aminotransferase<br />
Increased<br />
Heart Rate Irregular<br />
Diarrhoea<br />
Chills<br />
Anxiety<br />
Skin Disorder<br />
Back Pain<br />
Agitation<br />
Date:09/26/05ISR Number: 4778714-3Report Type:Expedited (15-DaCompany Report #SE-ROCHE-407993 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Lariam PS Roche ORAL 9 DAY<br />
Cerebral Disorder Lexinor C<br />
Visual Impairment<br />
Disturbance In Attention<br />
Amnesia<br />
Fatigue<br />
Date:09/30/05ISR Number: 4784739-4Report Type:Expedited (15-DaCompany Report #IT-ROCHE-418516 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety Lariam PS Roche ORAL 29 DAY<br />
Panic Attack<br />
Dyspnoea<br />
Crying
Date:09/30/05ISR Number: 4784747-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-418484 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lung Disorder Lariam PS Roche ORAL<br />
Hypoxia<br />
03-Apr-2012 09:37 AM Page: 643
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/05/05ISR Number: 4795220-0Report Type:Direct Company Report #CTU 259799 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nightmare Lariam PS 1 PILL /WK<br />
Congenital Anomaly Paranoia<br />
Required<br />
Anxiety<br />
Intervention to<br />
Suicide Attempt<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:10/07/05ISR Number: 4791872-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-391660 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Generalised Anxiety Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Disorder<br />
Disability<br />
Panic Attack<br />
Suicidal Ideation<br />
Mood Swings<br />
Memory Impairment<br />
Mental Disorder<br />
Amnesia<br />
Self-Medication<br />
Self Esteem Decreased<br />
Major Depression<br />
Date:10/11/05ISR Number: 4794427-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-419662 Age:4 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Cardiac Arrest Lariam PS Roche ORAL FREQUENCY:<br />
Convulsion<br />
WEEKLY.<br />
Haemolytic Uraemic Immunizations C SUBCUTANEOUS FIRST AND<br />
Syndrome<br />
SECOND SERIES<br />
Vomiting<br />
OF CHILDHOOD<br />
IMMUNIZATIONS<br />
NOS.<br />
Date:10/11/05ISR Number: 4794488-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Vertigo<br />
Visual Impairment<br />
Panic Disorder<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder<br />
Meniscus Lesion<br />
Tremor
Cognitive Disorder<br />
Dysthymic Disorder<br />
Exostosis<br />
Nocturia<br />
Agitation<br />
Balance Disorder<br />
Ill-Defined Disorder<br />
Fall<br />
03-Apr-2012 09:37 AM Page: 644
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Insomnia<br />
Memory Impairment<br />
Fatigue<br />
Dyspnoea<br />
Cranial Nerve Disorder<br />
Toxicity To Various<br />
Agents<br />
Bundle Branch Block Right<br />
Respiratory Rate<br />
Increased<br />
Contusion<br />
Antinuclear Antibody<br />
Positive<br />
Eosinophilia<br />
Aggression<br />
Mitral Valve Incompetence<br />
Eye Movement Disorder<br />
Ligament Sprain<br />
Pyrexia<br />
Suicidal Ideation<br />
Asthenia<br />
Chest Pain<br />
Hypertension<br />
Psychiatric Symptom<br />
Musculoskeletal Pain<br />
Decreased Activity<br />
Humerus Fracture<br />
Weight Decreased<br />
Rhinitis Allergic<br />
Ventricular Extrasystoles<br />
Abnormal Behaviour<br />
Mood Swings<br />
Depression<br />
Hallucination, Visual<br />
Headache<br />
Sleep Apnoea Syndrome<br />
Self-Injurious Ideation<br />
Dehydration<br />
Myalgia<br />
Disturbance In Attention<br />
Selective Iga<br />
Immunodeficiency<br />
Bronchospasm<br />
Chest Discomfort<br />
Joint Dislocation<br />
Post-Traumatic Stress<br />
Disorder<br />
Hyperhidrosis<br />
Homicidal Ideation<br />
Oxygen Saturation
Decreased<br />
Convulsion<br />
Rheumatoid Factor<br />
Increased<br />
Anxiety<br />
Productive Cough<br />
Psychotic Disorder<br />
Asthma<br />
03-Apr-2012 09:37 AM Page: 645
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Thinking Abnormal<br />
Mitral Valve<br />
Calcification Report Source Product Role Manufacturer Route Dose Duration<br />
Hypoaesthesia Lariam PS Roche ORAL<br />
Nystagmus Primaquine C<br />
Rash Papular Cipro C<br />
Date:10/11/05ISR Number: 4794512-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Visual Impairment<br />
Dyspnoea<br />
Generalised Anxiety<br />
Disorder<br />
Decreased Activity<br />
Respiratory Rate<br />
Increased<br />
Rhinitis Allergic<br />
Mood Swings<br />
Bronchospasm<br />
Chest Discomfort<br />
Chest Pain<br />
Self-Injurious Ideation<br />
Cognitive Disorder<br />
Humerus Fracture<br />
Hypoaesthesia<br />
Vertigo<br />
Hypertension<br />
Fall<br />
Thinking Abnormal<br />
Bundle Branch Block Right<br />
Oxygen Saturation<br />
Decreased<br />
Rash Papular<br />
Ventricular Extrasystoles<br />
Asthenia<br />
Cranial Nerve Disorder<br />
Tremor<br />
Homicidal Ideation<br />
Toxicity To Various<br />
Agents<br />
Nocturia<br />
Eosinophilia<br />
Anxiety<br />
Suicidal Ideation<br />
Selective Iga<br />
Immunodeficiency<br />
Major Depression<br />
Mitral Valve<br />
Calcification<br />
Antinuclear Antibody
Positive<br />
Pyrexia<br />
Insomnia<br />
Memory Impairment<br />
Balance Disorder<br />
Fatigue<br />
Ill-Defined Disorder<br />
Sleep Apnoea Syndrome<br />
03-Apr-2012 09:37 AM Page: 646
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Psychotic Disorder<br />
Post-Traumatic Stress<br />
Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Musculoskeletal Pain Lariam PS Roche ORAL<br />
Dysthymic Disorder Primaquine C<br />
Dehydration Cipro C<br />
Weight Decreased<br />
Nystagmus<br />
Eye Movement Disorder<br />
Rheumatoid Factor<br />
Increased<br />
Abnormal Behaviour<br />
Aggression<br />
Agitation<br />
Disturbance In Attention<br />
Productive Cough<br />
Headache<br />
Panic Disorder<br />
Joint Dislocation<br />
Psychiatric Symptom<br />
Hyperhidrosis<br />
Mitral Valve Incompetence<br />
Exostosis<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Convulsion<br />
Contusion<br />
Myalgia<br />
Depression<br />
Hallucination, Visual<br />
Meniscus Lesion<br />
Asthma<br />
Ligament Sprain<br />
Date:10/13/05ISR Number: 4797132-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Hepatic Failure<br />
Cranial Nerve Disorder<br />
Tremor<br />
Insomnia<br />
Disturbance In Attention<br />
Tinnitus<br />
Migraine<br />
Dysarthria<br />
Dysphemia<br />
Syncope<br />
Self-Injurious Ideation<br />
Visual Impairment<br />
Photophobia
Visual Field Defect<br />
Aggression<br />
Amnesia<br />
Photosensitivity Reaction<br />
Post-Traumatic Stress<br />
Disorder<br />
Cognitive Disorder<br />
Gilbert'S Syndrome<br />
03-Apr-2012 09:37 AM Page: 647
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abnormal Dreams<br />
Dizziness<br />
Sleep Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Psychomotor Hyperactivity Lariam PS Roche ORAL 23 TABLETS<br />
Panic Attack<br />
DISPENSED.<br />
Nausea Sudafed C<br />
Vestibular Disorder<br />
Toxicity To Various<br />
Agents<br />
Muscle Twitching<br />
Mood Swings<br />
Thirst<br />
Neck Pain<br />
Homicidal Ideation<br />
Abnormal Behaviour<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Vomiting<br />
Depression<br />
Suicidal Ideation<br />
Asthenia<br />
Cough<br />
Confusional State<br />
Mania<br />
Fatigue<br />
Dehydration<br />
Obsessive Thoughts<br />
Depressed Level Of<br />
Consciousness<br />
Endolymphatic Hydrops<br />
Jaw Disorder<br />
Tardive Dyskinesia<br />
Gait Disturbance<br />
Road Traffic Accident<br />
Stress<br />
Atrial Fibrillation<br />
Anger<br />
General Symptom<br />
Bipolar Disorder<br />
Hypoaesthesia<br />
Memory Impairment<br />
Alcohol Poisoning<br />
Hallucination<br />
Balance Disorder<br />
Incontinence<br />
Anxiety<br />
Jaundice<br />
Conduction Disorder<br />
Hyperacusis<br />
Pain In Extremity<br />
Paranoia<br />
Diarrhoea
Irritability<br />
Decreased Appetite<br />
Lacrimation Increased<br />
Motor Dysfunction<br />
Feeling Abnormal<br />
03-Apr-2012 09:37 AM Page: 648
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/13/05ISR Number: 4799799-4Report Type:Direct Company Report #CTU 260561 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination <strong>Mefloquine</strong><br />
Required Feeling Hot Lariam PS ORAL PO<br />
Intervention to<br />
Pruritus<br />
Prevent Permanent<br />
Impairment/Damage<br />
Pyrexia<br />
Headache<br />
Loss Of Consciousness<br />
Feeling Abnormal<br />
Burning Sensation<br />
Date:10/18/05ISR Number: 4801794-3Report Type:Expedited (15-DaCompany Report #DE-JNJFOC-20051002297 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Imodium PS ORAL<br />
Initial or Prolonged Overdose Gelonida SS ORAL<br />
Talvosilen SS ORAL<br />
Lariam SS ORAL 16 tablets.<br />
Talvosilen SS ORAL<br />
Gelonida SS ORAL<br />
Ibuprofen SS ORAL 20 tablets.<br />
Date:10/18/05ISR Number: 4804112-XReport Type:Direct Company Report #CTU 260897 Age:55 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Paranoia <strong>Mefloquine</strong> 250mg<br />
Required Hallucination Geneva PS Geneva 1 EVERY WEEK<br />
Intervention to Anxiety White Sidel: Gp 118 C<br />
Prevent Permanent Suicidal Ideation<br />
Impairment/Damage<br />
Date:10/21/05ISR Number: 4805756-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-407993 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion Lariam PS Roche ORAL 9 DAY<br />
Amnesia Lexinor C<br />
Visual Impairment<br />
Date:10/21/05ISR Number: 4805757-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-420600 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chills Lariam PS Roche ORAL<br />
Joint Swelling<br />
Erythema
Dyspnoea<br />
Arrhythmia<br />
03-Apr-2012 09:37 AM Page: 649
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/21/05ISR Number: 4805773-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-420097 Age:4 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hypothyroidism Lariam PS Roche UNKNOWN<br />
Congenital Anomaly Delayed Fontanelle<br />
Closure<br />
Haemolytic Uraemic<br />
Syndrome<br />
Skin Discolouration<br />
Pallor<br />
Plagiocephaly<br />
Penis Disorder<br />
Developmental Delay<br />
Vomiting<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:10/21/05ISR Number: 4805981-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Disturbance In Attention<br />
Amnesia<br />
Endolymphatic Hydrops<br />
Conduction Disorder<br />
Paranoia<br />
Hallucination<br />
Cranial Nerve Disorder<br />
Tremor<br />
Aggression<br />
Anger<br />
Vestibular Disorder<br />
Irritability<br />
Dysphemia<br />
Ototoxicity<br />
Panic Attack<br />
Toxicity To Various<br />
Agents<br />
Suicidal Ideation<br />
Anxiety<br />
Mood Swings<br />
Post-Traumatic Stress<br />
Disorder<br />
Gait Disturbance<br />
Bipolar Disorder<br />
Tinnitus<br />
Obsessive Thoughts<br />
Jaundice<br />
Muscle Twitching<br />
Cognitive Disorder<br />
Syncope<br />
Hepatic Failure
Vomiting<br />
Depression<br />
Depressed Level Of<br />
Consciousness<br />
Thirst<br />
Confusional State<br />
Dysarthria<br />
Motor Dysfunction<br />
03-Apr-2012 09:37 AM Page: 650
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abnormal Dreams<br />
Photophobia<br />
Visual Field Defect Report Source Product Role Manufacturer Route Dose Duration<br />
Balance Disorder Lariam PS Roche ORAL 23 TABLETS<br />
Insomnia<br />
DISPENSED.<br />
Asthenia Sudafed C<br />
Cough<br />
Jaw Disorder<br />
Tardive Dyskinesia<br />
Hyperacusis<br />
Incontinence<br />
Diarrhoea<br />
Photosensitivity Reaction<br />
General Symptom<br />
Migraine<br />
Gilbert'S Syndrome<br />
Visual Impairment<br />
Nausea<br />
Decreased Appetite<br />
Lacrimation Increased<br />
Mania<br />
Homicidal Ideation<br />
Date:10/21/05ISR Number: 4806101-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-419686 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Leukopenia Lariam PS Roche ORAL DOSING<br />
Initial or Prolonged Neutropenia REGIMEN<br />
Thrombocytopenia REPORTED AS 3<br />
Hepatic Enzyme Increased TABLETS, 2<br />
TABLETS THEN 2 DAY<br />
Date:10/21/05ISR Number: 4806107-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-419662 Age:4 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Haemolytic Uraemic Lariam PS Roche ORAL FREQUENCY:<br />
Syndrome<br />
WEEKLY.<br />
Convulsion Immunizations C SUBCUTANEOUS FIRST AND<br />
Vomiting<br />
SECOND SERIES<br />
Cardiac Arrest<br />
OF CHILDHOOD<br />
IMMUNIZATIONS<br />
NOS.<br />
Date:10/25/05ISR Number: 4811091-8Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
PT<br />
Aggression
Initial or Prolonged<br />
Disability<br />
Toxicity To Various<br />
Agents<br />
Chest Discomfort<br />
Wheezing<br />
Rhinitis Allergic<br />
Abnormal Behaviour<br />
Hallucination, Visual<br />
Snoring<br />
03-Apr-2012 09:37 AM Page: 651
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bundle Branch Block Right<br />
Generalised Anxiety<br />
Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Amnesia Other Lariam (<strong>Mefloquine</strong><br />
Coordination Abnormal Hydrochloride) PS ORAL ORAL<br />
Palpitations<br />
Primaquine<br />
Nocturia<br />
(Primaquine<br />
Chest Pain Phosphate) C<br />
Asthma<br />
Cipro (Ciprofloxacin<br />
Joint Stiffness Hydrochloride) C<br />
Mood Swings<br />
Weight Increased<br />
Agitation<br />
Vertigo<br />
Balance Disorder<br />
Dyspnoea<br />
Pulmonary Function<br />
Challenge Test Abnormal<br />
Troponin Increased<br />
Arthralgia<br />
Decreased Activity<br />
Chondroplasty<br />
Occupational Exposure To<br />
Air Contaminants<br />
Insomnia<br />
Disturbance In Attention<br />
Suicidal Ideation<br />
Headache<br />
Fatigue<br />
Hyperhidrosis<br />
Tremor<br />
Morbid Thoughts<br />
Deafness<br />
Anxiety<br />
Poor Quality Drug<br />
Administered<br />
Cough<br />
Blood Potassium Decreased<br />
Loose Body In Joint<br />
Rash Papular<br />
Musculoskeletal Pain<br />
Tinnitus<br />
Depression<br />
Visual Impairment<br />
Myalgia<br />
Acute Coronary Syndrome<br />
Coronary Artery Disease<br />
Meniscus Removal<br />
Pyrexia<br />
Hypervigilance
Date:10/28/05ISR Number: 4814893-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Abnormal Behaviour<br />
Mood Swings<br />
Cranial Nerve Disorder<br />
Joint Dislocation<br />
Post-Traumatic Stress<br />
03-Apr-2012 09:37 AM Page: 652
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorder<br />
Asthma<br />
Nystagmus<br />
Suicidal Ideation<br />
Selective Iga<br />
Immunodeficiency<br />
Headache<br />
Panic Disorder<br />
Hypertension<br />
Generalised Anxiety<br />
Disorder<br />
Self-Injurious Ideation<br />
Fall<br />
Mitral Valve Incompetence<br />
Dehydration<br />
Weight Decreased<br />
Convulsion<br />
Eye Movement Disorder<br />
Disturbance In Attention<br />
Vertigo<br />
Hallucination, Visual<br />
Asthenia<br />
Bronchospasm<br />
Chest Discomfort<br />
Major Depression<br />
Decreased Activity<br />
Humerus Fracture<br />
Hypoaesthesia<br />
Ligament Sprain<br />
Rheumatoid Factor<br />
Increased<br />
Memory Impairment<br />
Depression<br />
Productive Cough<br />
Chest Pain<br />
Toxicity To Various<br />
Agents<br />
Exostosis<br />
Antinuclear Antibody<br />
Positive<br />
Pyrexia<br />
Balance Disorder<br />
Ill-Defined Disorder<br />
Sleep Apnoea Syndrome<br />
Psychiatric Symptom<br />
Cognitive Disorder<br />
Bundle Branch Block Right<br />
Contusion<br />
Agitation<br />
Anxiety<br />
Psychotic Disorder<br />
Meniscus Lesion<br />
Thinking Abnormal<br />
Homicidal Ideation
Mitral Valve<br />
Calcification<br />
Respiratory Rate<br />
Increased<br />
Myalgia<br />
Rash Papular<br />
Insomnia<br />
Fatigue<br />
03-Apr-2012 09:37 AM Page: 653
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dyspnoea<br />
Hyperhidrosis<br />
Tremor Report Source Product Role Manufacturer Route Dose Duration<br />
Dysthymic Disorder Lariam PS Roche ORAL<br />
Oxygen Saturation Primaquine C<br />
Decreased Cipro C<br />
Rhinitis Allergic<br />
Eosinophilia<br />
Ventricular Extrasystoles<br />
Aggression<br />
Visual Impairment<br />
Musculoskeletal Pain<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Nocturia<br />
Date:11/02/05ISR Number: 4818003-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-392506 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Grand Mal Convulsion Lariam PS Roche ORAL 9 DAY<br />
Initial or Prolonged Palpitations Lexinor C ORAL<br />
Amnesia Malarone C ORAL<br />
Visual Impairment<br />
Fatigue<br />
Disturbance In Attention<br />
Headache<br />
Date:11/02/05ISR Number: 4818006-7Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398212 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Balance Disorder Lariam PS Roche ORAL 15 DAY<br />
Initial or Prolonged Tinnitus<br />
Disability<br />
Nystagmus<br />
Hearing Impaired<br />
Dizziness<br />
Vertigo<br />
Diarrhoea<br />
Nausea<br />
Date:11/03/05ISR Number: 4821792-3Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Disturbance In Attention<br />
Memory Impairment<br />
Toxicity To Various<br />
Agents
Mood Swings<br />
Bronchopneumonia<br />
Nocturia<br />
Arthralgia<br />
Musculoskeletal Stiffness<br />
Sputum Culture<br />
Joint Dislocation<br />
Vestibular Disorder<br />
03-Apr-2012 09:37 AM Page: 654
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Cognitive Disorder<br />
Agitation<br />
Depression<br />
Vertigo<br />
Visual Impairment<br />
Hallucination, Visual<br />
Asthenia<br />
Dizziness<br />
Loss Of Libido<br />
Convulsion<br />
Dissociation<br />
Meniscus Lesion<br />
Respiratory Fume<br />
Inhalation Disorder<br />
Selective Iga<br />
Immunodeficiency<br />
Haemarthrosis<br />
Homicidal Ideation<br />
Insomnia<br />
Stress<br />
Hiatus Hernia<br />
Dysthymic Disorder<br />
Dehydration<br />
Palpitations<br />
Rash Papular<br />
Bronchial Hyperreactivity<br />
Exostosis<br />
Amnesia<br />
Coordination Abnormal<br />
Tinnitus<br />
Gait Disturbance<br />
Mucosal Inflammation<br />
Hypervigilance<br />
Eye Movement Disorder<br />
Sleep Apnoea Syndrome<br />
Mitral Valve<br />
Calcification<br />
Electroencephalogram<br />
Abnormal<br />
Nightmare<br />
Balance Disorder<br />
Electrocardiogram St<br />
Segment Depression<br />
Panic Attack<br />
Accident<br />
Decreased Appetite<br />
Intentional Self-Injury<br />
Nystagmus<br />
Derealisation<br />
Hypertension<br />
Rheumatoid Factor<br />
Increased<br />
Rhinitis Allergic
Myalgia<br />
Marital Problem<br />
Medication Error<br />
Chondropathy<br />
Fatigue<br />
Self-Medication<br />
Electrocardiogram St<br />
Segment Abnormal<br />
03-Apr-2012 09:37 AM Page: 655
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Headache<br />
Blood Pressure Increased<br />
Paranoia Report Source Product Role Manufacturer Route Dose Duration<br />
Contusion Other Lariam<br />
Blood Potassium Decreased<br />
(<strong>Mefloquine</strong><br />
Acute Coronary Syndrome Hydrochloride) PS ORAL ORAL<br />
Asthma<br />
Primaquine<br />
Nodule<br />
(Primaquine<br />
Bacterial Infection Phosphate) C<br />
Haemophilus Infection<br />
Cipro<br />
Mitral Valve Incompetence<br />
(Ciprofloxacin<br />
Tremor Hydrochloride) C<br />
Eosinophilia<br />
Hallucination<br />
Suicidal Ideation<br />
Product Quality Issue<br />
Fall<br />
Deafness<br />
Chronic Sinusitis<br />
Physical Assault<br />
Weight Decreased<br />
Respiratory Rate<br />
Increased<br />
Oxygen Saturation<br />
Decreased<br />
Ventricular Extrasystoles<br />
Joint Lock<br />
Humerus Fracture<br />
Ototoxicity<br />
Aggression<br />
Anxiety<br />
Poor Quality Drug<br />
Administered<br />
Productive Cough<br />
Hypoaesthesia<br />
Ligament Sprain<br />
Antinuclear Antibody<br />
Positive<br />
Weight Increased<br />
Bundle Branch Block Right<br />
Post-Traumatic Stress<br />
Disorder<br />
Hyperhidrosis<br />
Emotional Disorder<br />
Impulsive Behaviour<br />
Abnormal Dreams<br />
Date:11/04/05ISR Number: 4820968-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
PT<br />
Disturbance In Attention
Initial or Prolonged<br />
Disability<br />
Vertigo<br />
Asthenia<br />
Chest Pain<br />
Major Depression<br />
Toxicity To Various<br />
Agents<br />
Convulsion<br />
Nocturia<br />
03-Apr-2012 09:37 AM Page: 656
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Coordination Abnormal<br />
Electrocardiogram St<br />
Segment Depression<br />
Panic Attack<br />
Skin Mass<br />
Productive Cough<br />
Chest Discomfort<br />
Hypertension<br />
Psychotic Disorder<br />
Homicidal Ideation<br />
Oxygen Saturation<br />
Decreased<br />
Dizziness<br />
Haemarthrosis<br />
Decreased Appetite<br />
Priapism<br />
Weight Increased<br />
Snoring<br />
Agitation<br />
Selective Iga<br />
Immunodeficiency<br />
Meniscus Lesion<br />
Mitral Valve Incompetence<br />
Ventricular Extrasystoles<br />
Electrocardiogram Qrs<br />
Complex Prolonged<br />
Disease Recurrence<br />
Partner Stress<br />
Libido Decreased<br />
Paranoia<br />
Dissociation<br />
Derealisation<br />
Acute Coronary Syndrome<br />
Insomnia<br />
Sleep Apnoea Syndrome<br />
Joint Dislocation<br />
Generalised Anxiety<br />
Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
Decreased Activity<br />
Asthma<br />
Dehydration<br />
Nystagmus<br />
Eye Movement Disorder<br />
Ligament Sprain<br />
Hyperkeratosis<br />
Tinnitus<br />
Gait Disturbance<br />
Sputum Culture Positive<br />
Bronchial Hyperreactivity<br />
Nightmare<br />
Loss Of Consciousness
Arthralgia<br />
Musculoskeletal Stiffness<br />
Abnormal Behaviour<br />
Visual Impairment<br />
Hallucination, Visual<br />
Balance Disorder<br />
Suicidal Ideation<br />
Cranial Nerve Disorder<br />
03-Apr-2012 09:37 AM Page: 657
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tremor<br />
Mitral Valve<br />
Calcification<br />
Respiratory Rate<br />
Increased<br />
Antinuclear Antibody<br />
Positive<br />
Rhinitis Allergic<br />
Rash Papular<br />
Eosinophilia<br />
Electrocardiogram T Wave<br />
Abnormal<br />
Bronchopneumonia<br />
Mood Altered<br />
Deafness<br />
Emotional Disorder<br />
Blood Pressure Increased<br />
Bronchospasm<br />
Headache<br />
Panic Disorder<br />
Self-Injurious Ideation<br />
Fall<br />
Musculoskeletal Pain<br />
Hyperhidrosis<br />
Thinking Abnormal<br />
Bundle Branch Block Right<br />
Exostosis<br />
Hypoaesthesia<br />
Contusion<br />
Pyrexia<br />
Bacterial Infection<br />
Amnesia<br />
Apathy<br />
Aggression<br />
Anxiety<br />
Memory Impairment<br />
Fatigue<br />
Ill-Defined Disorder<br />
Dyspnoea<br />
Psychiatric Symptom<br />
Cognitive Disorder<br />
Dysthymic Disorder<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Rheumatoid Factor<br />
Increased<br />
Myalgia<br />
Chronic Sinusitis<br />
Upper Respiratory Tract<br />
Infection<br />
Vestibular Disorder<br />
Injury
Palpitations<br />
Wheezing<br />
Blood Potassium Decreased<br />
Tongue Disorder<br />
Mood Swings<br />
Depression<br />
Humerus Fracture<br />
Weight Decreased<br />
03-Apr-2012 09:37 AM Page: 658
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hiatus Hernia<br />
Mucous Membrane Disorder<br />
Haemophilus Infection Report Source Product Role Manufacturer Route Dose Duration<br />
Hypervigilance Lariam PS Roche ORAL<br />
Respiratory Fume Primaquine C<br />
Inhalation Disorder Cipro C<br />
Feeling Abnormal<br />
Date:11/09/05ISR Number: 4823715-XReport Type:Expedited (15-DaCompany Report #NO-ROCHE-398212 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tinnitus Lariam PS Roche ORAL TAKEN ON 22<br />
Initial or Prolonged Diarrhoea FEB, 01 MAR<br />
Hearing Impaired<br />
AND 08 MAR<br />
Nausea 2005. 15 DAY<br />
Vertigo<br />
Dizziness<br />
Nystagmus<br />
Balance Disorder<br />
Date:11/11/05ISR Number: 4826347-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-354748 Age:44 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Hepatic Failure<br />
Tremor<br />
Diarrhoea<br />
Disturbance In Attention<br />
Toxicity To Various<br />
Agents<br />
Post-Traumatic Stress<br />
Disorder<br />
Decreased Appetite<br />
Mania<br />
Hallucination<br />
Visual Field Defect<br />
Nausea<br />
Depressed Level Of<br />
Consciousness<br />
Ototoxicity<br />
Conduction Disorder<br />
Hyperacusis<br />
Balance Disorder<br />
Incontinence<br />
General Symptom<br />
Cough<br />
Migraine<br />
Tardive Dyskinesia<br />
Vestibular Disorder<br />
Suicidal Ideation
Tinnitus<br />
Irritability<br />
Muscle Twitching<br />
Confusional State<br />
Cranial Nerve Disorder<br />
Insomnia<br />
Photosensitivity Reaction<br />
Anxiety<br />
03-Apr-2012 09:37 AM Page: 659
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Asthenia<br />
Obsessive Thoughts<br />
Yellow Skin Report Source Product Role Manufacturer Route Dose Duration<br />
Lacrimation Increased Lariam PS Roche ORAL 23 TABLETS<br />
Paranoia<br />
DISPENSED.<br />
Photophobia Sudafed C<br />
Aggression<br />
Amnesia<br />
Jaw Disorder<br />
Cognitive Disorder<br />
Motor Dysfunction<br />
Dysphemia<br />
Visual Impairment<br />
Panic Attack<br />
Anger<br />
Depression<br />
Thirst<br />
Gait Disturbance<br />
Gilbert'S Syndrome<br />
Abnormal Dreams<br />
Syncope<br />
Vomiting<br />
Mood Swings<br />
Endolymphatic Hydrops<br />
Dysarthria<br />
Bipolar Disorder<br />
Date:11/11/05ISR Number: 4826351-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Insomnia<br />
Aggression<br />
Sleep Apnoea Syndrome<br />
Fall<br />
Decreased Activity<br />
Dysthymic Disorder<br />
Humerus Fracture<br />
Respiratory Rate<br />
Increased<br />
Rash Papular<br />
Memory Impairment<br />
Mood Swings<br />
Visual Impairment<br />
Selective Iga<br />
Immunodeficiency<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
Musculoskeletal Pain
Antinuclear Antibody<br />
Positive<br />
Myalgia<br />
Abnormal Behaviour<br />
Asthenia<br />
Hypoaesthesia<br />
Rhinitis Allergic<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 660
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bronchial Hyperreactivity<br />
Chest Pain<br />
Asthma Report Source Product Role Manufacturer Route Dose Duration<br />
Homicidal Ideation Lariam PS Roche ORAL<br />
Dehydration Primaquine C<br />
Ligament Sprain Cipro C<br />
Contusion<br />
Ventricular Extrasystoles<br />
Balance Disorder<br />
Ill-Defined Disorder<br />
Chest Discomfort<br />
Cranial Nerve Disorder<br />
Hypertension<br />
Psychotic Disorder<br />
Convulsion<br />
Nystagmus<br />
Anxiety<br />
Disturbance In Attention<br />
Hallucination, Visual<br />
Suicidal Ideation<br />
Productive Cough<br />
Dyspnoea<br />
Psychiatric Symptom<br />
Tremor<br />
Thinking Abnormal<br />
Cognitive Disorder<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Nocturia<br />
Vertigo<br />
Fatigue<br />
Headache<br />
Panic Disorder<br />
Meniscus Lesion<br />
Hyperhidrosis<br />
Mitral Valve<br />
Calcification<br />
Weight Decreased<br />
Eye Movement Disorder<br />
Pyrexia<br />
Agitation<br />
Joint Dislocation<br />
Self-Injurious Ideation<br />
Toxicity To Various<br />
Agents<br />
Bundle Branch Block Right<br />
Mitral Valve Incompetence<br />
Exostosis<br />
Oxygen Saturation<br />
Decreased<br />
Rheumatoid Factor<br />
Increased
Eosinophilia<br />
03-Apr-2012 09:37 AM Page: 661
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/14/05ISR Number: 4827825-2Report Type:Direct Company Report #CTU 262994 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Lariam-<strong>Mefloquine</strong>-<br />
Disturbance In Attention Roche PS Roche ORAL 1 PILL PER<br />
Impaired Driving Ability<br />
WEEK 11 WEEKS<br />
Visual Impairment<br />
PO<br />
Raynaud'S Phenomenon<br />
Generalised Anxiety<br />
Disorder<br />
Fatigue<br />
Suicidal Ideation<br />
Panic Attack<br />
Tremor<br />
Abdominal Pain Upper<br />
Nervousness<br />
Thinking Abnormal<br />
Date:11/14/05ISR Number: 4827832-XReport Type:Direct Company Report #CTU 262979 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nausea Lariam PS ORAL ONE PILL ONCE<br />
Required Weight Decreased WEEKLY PO<br />
Intervention to<br />
Depression<br />
Prevent Permanent<br />
Impairment/Damage<br />
Anxiety<br />
Nightmare<br />
Hallucination<br />
Skin Discomfort<br />
Social Avoidant Behaviour<br />
Paranoia<br />
Date:11/14/05ISR Number: 4832250-4Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Abnormal Behaviour<br />
Agitation<br />
Anxiety<br />
Disturbance In Attention<br />
Hallucination, Visual<br />
Bronchopneumonia<br />
Acquired Diaphragmatic<br />
Eventration<br />
Electrocardiogram<br />
Repolarisation<br />
Abnormality<br />
Tinnitus<br />
Mitral Valve Incompetence<br />
Electroencephalogram<br />
Abnormal
Nightmare<br />
Dysthymic Disorder<br />
Priapism<br />
Paranoia<br />
Nystagmus<br />
Rheumatoid Factor<br />
Increased<br />
Snoring<br />
03-Apr-2012 09:37 AM Page: 662
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bundle Branch Block Right<br />
Aggression<br />
Mood Swings<br />
Depression<br />
Electrocardiogram St<br />
Segment Abnormal<br />
Cardiac Stress Test<br />
Abnormal<br />
Deafness<br />
Inflammation<br />
Sputum Culture Positive<br />
Marital Problem<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Visual Field Defect<br />
Impulsive Behaviour<br />
Abnormal Dreams<br />
Weight Increased<br />
Chest Pain<br />
Coronary Artery Disease<br />
Generalised Anxiety<br />
Disorder<br />
Pyrexia<br />
Rash<br />
Joint Crepitation<br />
Arthroscopy<br />
Gait Disturbance<br />
Major Depression<br />
Apathy<br />
Respiratory Rate<br />
Increased<br />
Convulsion<br />
Dissociation<br />
Cognitive Disorder<br />
Vital Functions Abnormal<br />
Tongue Disorder<br />
Musculoskeletal Pain<br />
Musculoskeletal Stiffness<br />
Insomnia<br />
Chondroplasty<br />
Fatigue<br />
Pain<br />
Headache<br />
Panic Attack<br />
Rapid Eye Movements Sleep<br />
Abnormal<br />
Hyperhidrosis<br />
Accident<br />
Libido Decreased<br />
Eye Movement Disorder<br />
Hypertension<br />
Memory Impairment
Chest Discomfort<br />
Self-Medication<br />
Stress<br />
Dizziness<br />
Emotional Disorder<br />
Joint Dislocation<br />
Exostosis<br />
Humerus Fracture<br />
03-Apr-2012 09:37 AM Page: 663
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Eosinophilia<br />
Psychomotor Skills<br />
Impaired<br />
Loss Of Consciousness<br />
Palpitations<br />
Wheezing<br />
Rhinitis Allergic<br />
Acute Coronary Syndrome<br />
Thinking Abnormal<br />
Speech Disorder<br />
Asthma<br />
Poor Quality Drug<br />
Administered<br />
Vertigo<br />
Coordination Abnormal<br />
Fall<br />
Bronchial Hyperreactivity<br />
Haemarthrosis<br />
Post-Traumatic Stress<br />
Disorder<br />
Tremor<br />
Divorced<br />
Physical Assault<br />
Weight Decreased<br />
Hypervigilance<br />
Derealisation<br />
Ventricular Extrasystoles<br />
Contusion<br />
Antinuclear Antibody<br />
Positive<br />
Cough<br />
Nocturia<br />
Myalgia<br />
Visual Impairment<br />
Suicidal Ideation<br />
Mental Disorder<br />
Dyspnoea<br />
Mood Altered<br />
Hiatus Hernia<br />
Chronic Sinusitis<br />
Haemophilus Infection<br />
Bacterial Infection<br />
Selective Iga<br />
Immunodeficiency<br />
Vestibular Disorder<br />
Amnesia<br />
Decreased Appetite<br />
Feeling Abnormal<br />
Psychiatric Symptom<br />
Heart Rate Decreased<br />
Hypoaesthesia<br />
Meniscus Lesion<br />
Blood Potassium Increased
Rash Papular<br />
Balance Disorder<br />
Toxicity To Various<br />
Agents<br />
Productive Cough<br />
Respiratory Tract<br />
Infection<br />
Sleep Apnoea Syndrome<br />
03-Apr-2012 09:37 AM Page: 664
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mitral Valve<br />
Calcification<br />
Mental Retardation Report Source Product Role Manufacturer Route Dose Duration<br />
Dehydration Other Lariam (<strong>Mefloquine</strong><br />
Self-Injurious Ideation Hydrochloride) 250<br />
Ligament Sprain Mg PS ORAL ORAL<br />
Movement Disorder<br />
Primaquine<br />
Eye Disorder<br />
(Primaquine<br />
Drug Ineffective Phosphate) C<br />
Arthralgia<br />
Cipro (Ciprofloxacin<br />
Joint Lock Hydrochloride) C<br />
Date:11/15/05ISR Number: 4828508-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-391660 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Generalised Anxiety Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Disorder<br />
Disability<br />
Mental Disorder<br />
Major Depression<br />
Self Esteem Decreased<br />
Panic Attack<br />
Amnesia<br />
Suicidal Ideation<br />
Mood Swings<br />
Memory Impairment<br />
Date:11/17/05ISR Number: 4832517-XReport Type:Direct Company Report #CTU 263333 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Lariam Laroche PS Laroche ORAL 1 PILL WEEKLY<br />
Required Insomnia PO<br />
Intervention to<br />
Panic Attack<br />
Prevent Permanent<br />
Impairment/Damage<br />
Heart Rate Increased<br />
Depression<br />
Feeling Of Despair<br />
Suicidal Ideation<br />
Date:11/21/05ISR Number: 4833061-6Report Type:Expedited (15-DaCompany Report #FR-ROCHE-415971 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Upper Motor Neurone Lariam PS Roche ORAL<br />
Lesion Engerix B SS INTRAMUSCULAR THE PATIENT<br />
Amyotrophy<br />
RECEIVED<br />
THREE<br />
INJECTIONS IN<br />
1995 (MARCH, 189 DAY
Date:11/21/05ISR Number: 4835576-3Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Abnormal Behaviour<br />
Major Depression<br />
Weight Increased<br />
03-Apr-2012 09:37 AM Page: 665
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Snoring<br />
Musculoskeletal Stiffness<br />
Sputum Culture Positive<br />
Exostosis<br />
Insomnia<br />
Disturbance In Attention<br />
Vertigo<br />
Affective Disorder<br />
Bronchial Hyperreactivity<br />
Hyperhidrosis<br />
Dehydration<br />
Hypoaesthesia<br />
Hypertension<br />
Electrocardiogram St<br />
Segment Depression<br />
Marital Problem<br />
Occupational Exposure To<br />
Dust<br />
Occupational Exposure To<br />
Toxic Agent<br />
Agitation<br />
Memory Impairment<br />
Visual Impairment<br />
Tinnitus<br />
Chronic Sinusitis<br />
Nightmare<br />
Chest Discomfort<br />
Blood Pressure Increased<br />
Heart Rate Decreased<br />
Posterior Capsule Rupture<br />
Rheumatoid Factor<br />
Increased<br />
Bronchopneumonia<br />
Accident At Work<br />
Mood Swings<br />
Depression<br />
Mental Disorder<br />
Disease Recurrence<br />
Haemorrhage<br />
Homicidal Ideation<br />
Loss Of Consciousness<br />
Myalgia<br />
Asthma<br />
Rash<br />
Decreased Activity<br />
Poor Quality Drug<br />
Administered<br />
Haemophilus Infection<br />
Suicidal Ideation<br />
Dizziness<br />
Fall<br />
Gait Disturbance<br />
Sleep Apnoea Syndrome
Mitral Valve<br />
Calcification<br />
Electroencephalogram<br />
Abnormal<br />
Post-Traumatic Stress<br />
Disorder<br />
Vision Blurred<br />
Ventricular Extrasystoles<br />
03-Apr-2012 09:37 AM Page: 666
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tongue Injury<br />
Hiatus Hernia<br />
Product Quality Issue Report Source Product Role Manufacturer Route Dose Duration<br />
Electrocardiogram Other Lariam (<strong>Mefloquine</strong><br />
Repolarisation Hydrochloride) 250<br />
Abnormality Mg PS ORAL ORAL<br />
Generalised Anxiety<br />
Primaquine<br />
Disorder<br />
(Primaquine<br />
Deafness Phosphate) C<br />
Joint Dislocation<br />
Cipro (Ciprofloxacin<br />
Cognitive Disorder Hydrochloride) C<br />
Amnesia<br />
Convulsion<br />
Contusion<br />
Coronary Artery Disease<br />
Self-Medication<br />
Stress<br />
Electrocardiogram St<br />
Segment Abnormal<br />
Humerus Fracture<br />
Hallucination, Visual<br />
Balance Disorder<br />
Coordination Abnormal<br />
Toxicity To Various<br />
Agents<br />
Headache<br />
Selective Iga<br />
Immunodeficiency<br />
Mitral Valve Incompetence<br />
Vestibular Disorder<br />
Dysthymic Disorder<br />
Pain<br />
Fatigue<br />
Acute Coronary Syndrome<br />
Aggression<br />
Panic Attack<br />
Upper Respiratory Tract<br />
Infection<br />
Emotional Disorder<br />
Tremor<br />
Eosinophilia<br />
Weight Decreased<br />
Antinuclear Antibody<br />
Positive<br />
Rhinitis Allergic<br />
Bundle Branch Block Right<br />
Haemarthrosis<br />
Date:11/28/05ISR Number: 4837942-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-424819 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Psychotic Disorder Lariam PS Roche ORAL<br />
03-Apr-2012 09:37 AM Page: 667
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/28/05ISR Number: 4837946-6Report Type:Expedited (15-DaCompany Report #HU-ROCHE-424837 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety <strong>Mefloquine</strong><br />
Initial or Prolonged Persecutory Delusion Hydrochloride PS Roche UNKNOWN TOTAL DOSE<br />
Hallucination, Auditory REPORTED AS 2<br />
TABLETS ( 1<br />
TABLET = 250<br />
MG OF ACTIVE<br />
Date:11/28/05ISR Number: 4837983-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-411818 Age:15 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Creatine Lariam PS Roche ORAL 28 DAY<br />
Phosphokinase Increased Ritonavir C ORAL<br />
Epzicom C ORAL<br />
Fosamprenavir C ORAL<br />
Date:11/29/05ISR Number: 4839920-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-425462 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aphasia Lariam PS Roche UNKNOWN<br />
Catatonia<br />
Psychotic Disorder<br />
Date:12/02/05ISR Number: 4844064-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Selective Iga<br />
Immunodeficiency<br />
Chest Discomfort<br />
Sleep Apnoea Syndrome<br />
Meniscus Lesion<br />
Decreased Activity<br />
Mitral Valve<br />
Calcification<br />
Mitral Valve Incompetence<br />
Dehydration<br />
Contusion<br />
Rash Papular<br />
Bronchopneumonia<br />
Visual Acuity Reduced<br />
Haemarthrosis<br />
Aggression<br />
Anxiety<br />
Depression<br />
Suicidal Ideation
Productive Cough<br />
Ill-Defined Disorder<br />
Headache<br />
Joint Dislocation<br />
Hyperhidrosis<br />
Nystagmus<br />
Stress<br />
Dizziness<br />
03-Apr-2012 09:37 AM Page: 668
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bacterial Infection<br />
Emotional Disorder<br />
Amnesia<br />
Hypervigilance<br />
Snoring<br />
Somnolence<br />
Agitation<br />
Hallucination, Visual<br />
Dyspnoea<br />
Chest Pain<br />
Weight Decreased<br />
Ventricular Extrasystoles<br />
Deafness<br />
Gait Disturbance<br />
Electroencephalogram<br />
Abnormal<br />
Mental Impairment<br />
Vision Blurred<br />
Loss Of Consciousness<br />
Paranoia<br />
Hypertension<br />
Tremor<br />
Exostosis<br />
Convulsion<br />
Hypoaesthesia<br />
Electrocardiogram St<br />
Segment Abnormal<br />
Vestibular Disorder<br />
Impulsive Behaviour<br />
Drug Effect Decreased<br />
Memory Impairment<br />
Mood Swings<br />
Visual Impairment<br />
Balance Disorder<br />
Asthenia<br />
Self-Injurious Ideation<br />
Musculoskeletal Pain<br />
Asthma<br />
Bundle Branch Block Right<br />
Humerus Fracture<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Conduction Disorder<br />
Upper Respiratory Tract<br />
Infection<br />
Rapid Eye Movements Sleep<br />
Abnormal<br />
Priapism<br />
Palpitations<br />
Acute Coronary Syndrome<br />
Abnormal Behaviour<br />
Insomnia
Panic Disorder<br />
Psychotic Disorder<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder<br />
Fall<br />
Respiratory Rate<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 669
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Rheumatoid Factor<br />
Increased<br />
Myalgia Report Source Product Role Manufacturer Route Dose Duration<br />
Injury Lariam PS Roche ORAL<br />
Marital Problem Primaquine C<br />
Derealisation Cipro C<br />
Rash<br />
Bronchial Hyperreactivity<br />
Cranial Nerve Disorder<br />
Psychiatric Symptom<br />
Thinking Abnormal<br />
Cognitive Disorder<br />
Homicidal Ideation<br />
Dysthymic Disorder<br />
Toxicity To Various<br />
Agents<br />
Antinuclear Antibody<br />
Positive<br />
Rhinitis Allergic<br />
Eosinophilia<br />
Acquired Diaphragmatic<br />
Eventration<br />
Cardiac Stress Test<br />
Abnormal<br />
Panic Attack<br />
Chronic Sinusitis<br />
Dissociative Disorder<br />
Disturbance In Attention<br />
Vertigo<br />
Fatigue<br />
Post-Traumatic Stress<br />
Disorder<br />
Oxygen Saturation<br />
Decreased<br />
Eye Movement Disorder<br />
Ligament Sprain<br />
Nocturia<br />
Pyrexia<br />
Hyperkeratosis<br />
Hiatus Hernia<br />
Tinnitus<br />
Haemophilus Infection<br />
Nightmare<br />
Musculoskeletal Stiffness<br />
Date:12/02/05ISR Number: 4844102-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vestibular Disorder<br />
Dry Eye<br />
Dry Mouth
Gastrooesophageal Reflux<br />
Disease<br />
Abnormal Dreams<br />
Bipolar Disorder<br />
Balance Disorder<br />
Confusional State<br />
Anxiety<br />
Diastolic Dysfunction<br />
03-Apr-2012 09:37 AM Page: 670
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Cholesterol<br />
Increased<br />
Blood Triglycerides<br />
Increased<br />
Memory Impairment<br />
Upper Respiratory Tract<br />
Infection<br />
Bursitis<br />
Gastrointestinal Disorder<br />
Angina Pectoris<br />
Dehydration<br />
Snoring<br />
Oral Disorder<br />
Acrochordon<br />
Visual Impairment<br />
Scratch<br />
Contusion<br />
Joint Dislocation<br />
Colitis<br />
Mitral Valve Prolapse<br />
Restrictive Pulmonary<br />
Disease<br />
Bronchitis<br />
Diarrhoea<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Nocturia<br />
Major Depression<br />
Amnesia<br />
Skin Disorder<br />
Ventricular Hypertrophy<br />
Dry Skin<br />
Clumsiness<br />
Tinnitus<br />
Dyspnoea<br />
Depression<br />
Total Lung Capacity<br />
Decreased<br />
Bronchospasm<br />
Cardiac Murmur<br />
Sinus Tachycardia<br />
Chest Discomfort<br />
Constipation<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Blood Glucose Increased<br />
Upper-Airway Cough<br />
Syndrome<br />
Haemorrhoids<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Affective Disorder<br />
Psychotic Disorder
Schizoaffective Disorder<br />
Tremor<br />
Aggression<br />
Headache<br />
Hypertension<br />
Rash<br />
Low Density Lipoprotein<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 671
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Rhinitis Report Source Product Role Manufacturer Route Dose Duration<br />
Asthma Lariam PS Roche ORAL A TOTAL OF 6<br />
Coordination Abnormal<br />
TABLETS WERE<br />
Adjustment Disorder<br />
INGESTED.<br />
Anhedonia Zoloft C UNKNOWN<br />
Intervertebral Disc<br />
Degeneration<br />
Fatigue<br />
Somnolence<br />
Sleep Apnoea Syndrome<br />
Viral Infection<br />
Arthralgia<br />
Weight Increased<br />
Exfoliative Rash<br />
Vitiligo<br />
Insomnia<br />
Date:12/02/05ISR Number: 4844105-XReport Type:Expedited (15-DaCompany Report #GB-ROCHE-425626 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Confusional State Lariam PS Roche ORAL 56 DAY<br />
Asthma<br />
Dizziness<br />
Date:12/12/05ISR Number: 4851571-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-419686 Age:38 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Thrombocytopenia Lariam PS Roche ORAL DOSING<br />
Initial or Prolonged Leukopenia REGIMEN<br />
Neutropenia REPORTED AS 3<br />
Hepatic Enzyme Increased TABLETS, 2<br />
Hyperthermia TABLETS THEN 2 DAY<br />
Imovane<br />
C<br />
Date:12/12/05ISR Number: 4851573-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Somnolence<br />
Sleep Apnoea Syndrome<br />
Viral Infection<br />
Bronchitis<br />
Vitiligo<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Coordination Abnormal
Memory Impairment<br />
Schizoaffective Disorder<br />
Vision Blurred<br />
Myalgia<br />
Anxiety<br />
Ventricular Hypertrophy<br />
Total Lung Capacity<br />
Decreased<br />
03-Apr-2012 09:37 AM Page: 672
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bronchospasm<br />
Chest Discomfort<br />
Insomnia<br />
Major Depression<br />
Paraesthesia<br />
Confusional State<br />
Skin Disorder<br />
Upper-Airway Cough<br />
Syndrome<br />
Asthma<br />
Bipolar Disorder<br />
Haemorrhoids<br />
Psychotic Disorder<br />
Mitral Valve Prolapse<br />
Angina Pectoris<br />
Restrictive Pulmonary<br />
Disease<br />
Arthralgia<br />
Rash<br />
Weight Increased<br />
Sinus Tachycardia<br />
Low Density Lipoprotein<br />
Increased<br />
Snoring<br />
Oral Disorder<br />
Abnormal Dreams<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Amnesia<br />
Fatigue<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Diarrhoea<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Constipation<br />
Scratch<br />
Affective Disorder<br />
Aggression<br />
Tinnitus<br />
Self Esteem Decreased<br />
Gastrointestinal Disorder<br />
Depression<br />
Diastolic Dysfunction<br />
Blood Triglycerides<br />
Increased<br />
Exfoliative Rash<br />
Dry Eye<br />
Acrochordon<br />
Gastrooesophageal Reflux<br />
Disease<br />
Hypersensitivity<br />
Dyspnoea
Headache<br />
Dehydration<br />
Dry Mouth<br />
Nocturia<br />
Rhinitis<br />
Contusion<br />
Cold Sweat<br />
Vestibular Disorder<br />
03-Apr-2012 09:37 AM Page: 673
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hypertension<br />
Cardiac Murmur<br />
Blood Cholesterol Report Source Product Role Manufacturer Route Dose Duration<br />
Increased Lariam PS Roche ORAL A TOTAL OF 6<br />
Dry Skin<br />
TABLETS WERE<br />
Blood Glucose Increased<br />
INGESTED.<br />
Clumsiness Zoloft C UNKNOWN<br />
Visual Impairment<br />
Balance Disorder<br />
Tremor<br />
Dizziness<br />
Date:12/12/05ISR Number: 4851582-7Report Type:Expedited (15-DaCompany Report #DE-ROCHE-427130 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paralysis Lariam PS Roche UNKNOWN<br />
Date:12/12/05ISR Number: 4853723-4Report Type:Expedited (15-DaCompany Report #343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Angina Pectoris<br />
Somnolence<br />
Bipolar Disorder<br />
Weight Increased<br />
Sinus Tachycardia<br />
Dry Skin<br />
Chest Discomfort<br />
Blood Glucose Increased<br />
Myalgia<br />
Feelings Of Worthlessness<br />
Hallucination, Visual<br />
Sinusitis<br />
Amnesia<br />
Dyspnoea<br />
Headache<br />
Balance Disorder<br />
Scratch<br />
Contusion<br />
Constipation<br />
Tremor<br />
Peripheral Sensory<br />
Neuropathy<br />
Sjogren'S Syndrome<br />
Confusional State<br />
Skin Disorder<br />
Hypertension<br />
Clumsiness<br />
Asthma<br />
Psychotic Disorder
Bronchitis<br />
Blood Cholesterol<br />
Increased<br />
Blood Triglycerides<br />
Increased<br />
Low Density Lipoprotein<br />
Increased<br />
Exfoliative Rash<br />
03-Apr-2012 09:37 AM Page: 674
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dry Mouth<br />
Snoring<br />
Aggression<br />
Pharyngeal Disorder<br />
Chronic Fatigue Syndrome<br />
Obesity<br />
Restrictive Pulmonary<br />
Disease<br />
Colitis<br />
Mental Status Changes<br />
Intervertebral Disc<br />
Degeneration<br />
Bursitis<br />
Cold Sweat<br />
Dizziness<br />
Anxiety<br />
Gastrointestinal Disorder<br />
Ventricular Hypertrophy<br />
Lung Disorder<br />
Adjustment Disorder<br />
Depressive Symptom<br />
Schizoaffective Disorder<br />
Rash<br />
Cardiac Murmur<br />
Diarrhoea<br />
Upper-Airway Cough<br />
Syndrome<br />
Listless<br />
Sinus Disorder<br />
Anal Neoplasm<br />
Anhedonia<br />
Tendonitis<br />
Fatigue<br />
Visual Impairment<br />
Coordination Abnormal<br />
Arthralgia<br />
Acrochordon<br />
Anal Fissure<br />
Psychomotor Hyperactivity<br />
Nightmare<br />
Photosensitivity Reaction<br />
Asthenia<br />
Diplopia<br />
Disorientation<br />
Vestibular Disorder<br />
Mitral Valve Prolapse<br />
Abnormal Dreams<br />
Affective Disorder<br />
Total Cholesterol/Hdl<br />
Ratio Increased<br />
Dry Eye<br />
Nocturia<br />
Insomnia
Sleep Apnoea Syndrome<br />
Haemorrhoids<br />
Dyspnoea Exertional<br />
Rectal Haemorrhage<br />
Feeling Guilty<br />
Disturbance In Attention<br />
Hallucination, Auditory<br />
Paraesthesia<br />
03-Apr-2012 09:37 AM Page: 675
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Vision Blurred<br />
Self Esteem Decreased<br />
Major Depression Report Source Product Role Manufacturer Route Dose Duration<br />
Bronchospasm Consumer Lariam (<strong>Mefloquine</strong><br />
Dehydration Other Hydrochloride) 250<br />
Blood Creatine Mg PS ORAL 250 MG 1 PER<br />
Phosphokinase Increased<br />
1 WEEK ORAL<br />
Vitiligo<br />
Zoloft (Sertraline<br />
Gastrooesophageal Reflux Hydrochloride) C<br />
Disease<br />
Neck Pain<br />
Bronchial Hyperreactivity<br />
Collagen-Vascular Disease<br />
Road Traffic Accident<br />
Musculoskeletal Stiffness<br />
Post-Traumatic Neck<br />
Syndrome<br />
Cognitive Disorder<br />
Depression<br />
Diastolic Dysfunction<br />
Memory Impairment<br />
Viral Infection<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Rhinitis<br />
Upper Respiratory Tract<br />
Infection<br />
Pain In Jaw<br />
Weight Decreased<br />
Tinnitus<br />
Hyperhidrosis<br />
Date:12/12/05ISR Number: 4853725-8Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Amnesia<br />
Coordination Abnormal<br />
Productive Cough<br />
Tinnitus<br />
Sputum Culture Positive<br />
Mitral Valve<br />
Calcification<br />
Dysthymic Disorder<br />
Weight Decreased<br />
Social Avoidant Behaviour<br />
Self-Injurious Ideation<br />
Paranoia<br />
Derealisation<br />
Contusion<br />
Wheezing<br />
Rash
Arthralgia<br />
Agitation<br />
Disturbance In Attention<br />
Loose Body In Joint<br />
Bronchopneumonia<br />
Self-Medication<br />
Electrocardiogram St<br />
Segment Abnormal<br />
03-Apr-2012 09:37 AM Page: 676
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fall<br />
Selective Iga<br />
Immunodeficiency<br />
Electroencephalogram<br />
Abnormal<br />
Homicidal Ideation<br />
Apathy<br />
Psychiatric Symptom<br />
Ligament Sprain<br />
Coronary Artery Disease<br />
Vertigo<br />
Injury<br />
Hiatus Hernia<br />
Gait Disturbance<br />
Condition Aggravated<br />
Chronic Sinusitis<br />
Mental Impairment<br />
Emotional Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
Tremor<br />
Eosinophilia<br />
Thinking Abnormal<br />
Hypertension<br />
Physical Assault<br />
Decreased Appetite<br />
Eye Movement Disorder<br />
Meniscus Lesion<br />
Weight Increased<br />
Rhinitis Allergic<br />
Chest Pain<br />
Blood Potassium Decreased<br />
Acute Coronary Syndrome<br />
Speech Disorder<br />
Asthma<br />
Arthropathy<br />
Insomnia<br />
Toxicity To Various<br />
Agents<br />
Mood Altered<br />
Dizziness<br />
Sleep Apnoea Syndrome<br />
Vestibular Disorder<br />
Occupational Exposure To<br />
Noise<br />
Visual Field Defect<br />
Cognitive Disorder<br />
Accident<br />
Convulsion<br />
Nystagmus<br />
Dissociation<br />
Anxiety Disorder<br />
Nocturia
Non-Cardiac Chest Pain<br />
Generalised Anxiety<br />
Disorder<br />
Suicidal Ideation<br />
Poor Quality Drug<br />
Administered<br />
Mental Disorder<br />
Unevaluable Event<br />
03-Apr-2012 09:37 AM Page: 677
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Chest Discomfort<br />
Bronchial Hyperreactivity<br />
Mitral Valve Incompetence<br />
Humerus Fracture<br />
Hyperhidrosis<br />
Hypoaesthesia<br />
Eye Disorder<br />
No Therapeutic Response<br />
Cough<br />
Myalgia<br />
Joint Lock<br />
Decreased Activity<br />
Aggression<br />
Visual Impairment<br />
Hallucination, Visual<br />
Product Quality Issue<br />
Stress<br />
Acquired Diaphragmatic<br />
Eventration<br />
Headache<br />
Haemophilus Infection<br />
Haemarthrosis<br />
Occupational Exposure To<br />
Toxic Agent<br />
Loss Of Consciousness<br />
Occupational Exposure To<br />
Dust<br />
Psychotic Disorder<br />
Impaired Work Ability<br />
Oxygen Saturation<br />
Decreased<br />
Rheumatoid Factor<br />
Increased<br />
Hallucination, Auditory<br />
Blood Creatine<br />
Phosphokinase Mb Normal<br />
Pyrexia<br />
Musculoskeletal Pain<br />
Abnormal Behaviour<br />
Anxiety<br />
Mood Swings<br />
Depression<br />
Balance Disorder<br />
Fatigue<br />
Electrocardiogram St<br />
Segment Depression<br />
Exercise Test Abnormal<br />
Deafness<br />
Bacterial Test Positive<br />
Exostosis<br />
Divorced<br />
Nightmare<br />
Loss Of Libido
Priapism<br />
Respiratory Rate<br />
Increased<br />
Panic Disorder<br />
Snoring<br />
Tongue Disorder<br />
Chondropathy<br />
Dyspnoea<br />
03-Apr-2012 09:37 AM Page: 678
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Panic Attack<br />
Joint Dislocation<br />
Marital Problem Report Source Product Role Manufacturer Route Dose Duration<br />
Occupational Exposure To Other Lariam (<strong>Mefloquine</strong><br />
Air Contaminants Hydrochloride) 250<br />
Dehydration Mg PS ORAL ORAL<br />
Major Depression<br />
Primaquine<br />
Palpitations<br />
(Primaquine<br />
Hypervigilance Phosphate) C<br />
Ventricular Extrasystoles<br />
Cipro (Ciprofloxacin<br />
Antinuclear Antibody Hydrochloride) C<br />
Positive<br />
Pulmonary Function<br />
Challenge Test Abnormal<br />
Bundle Branch Block Right<br />
Rash Papular<br />
Feeling Abnormal<br />
Musculoskeletal Stiffness<br />
Upper Respiratory Tract<br />
Infection<br />
Date:12/13/05ISR Number: 4852051-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-427130 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Lariam PS Roche UNKNOWN<br />
Paresis<br />
Hypokinesia<br />
Date:12/14/05ISR Number: 4853197-3Report Type:Expedited (15-DaCompany Report #JP-ROCHE-426965 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cyanosis Lariam PS Roche UNKNOWN PATIENT TOOK<br />
Initial or Prolonged Dyspnoea FOUR DOSES. 21 DAY<br />
Pneumonia<br />
Red Blood Cell<br />
Sedimentation Rate<br />
Increased<br />
C-Reactive Protein<br />
Increased<br />
Sinus Tachycardia<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Respiratory Failure<br />
Date:12/16/05ISR Number: 4855695-5Report Type:Expedited (15-DaCompany Report #CA-ROCHE-427697 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Irritability <strong>Mefloquine</strong><br />
Personality Disorder Hydrochloride PS Roche ORAL 3 WK<br />
Logorrhoea Doxycycline C ORAL<br />
Aggression<br />
Insomnia<br />
Thinking Abnormal<br />
Neurosis<br />
Cognitive Disorder<br />
03-Apr-2012 09:37 AM Page: 679
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/16/05ISR Number: 4855703-1Report Type:Expedited (15-DaCompany Report #CA-ROCHE-427620 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaise Lariam PS Roche UNKNOWN 6 WK<br />
Chest Pain Malarone C<br />
Dizziness Typhoid Vaccine C ORAL<br />
Tinnitus<br />
Diphtheria And<br />
Insomnia Tetanus Vaccine C<br />
Fatigue Hepatitis A Vaccine C<br />
Nausea Hepatitis B Vaccine C<br />
Paraesthesia<br />
Hypoaesthesia<br />
Palpitations<br />
Upper Respiratory Tract<br />
Infection<br />
Hypertension<br />
Date:12/19/05ISR Number: 4864051-5Report Type:Direct Company Report #CTU 265337 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Insomnia <strong>Mefloquine</strong> (Lariam) PS ONCE PER WEEK<br />
Agitation<br />
Thinking Abnormal<br />
Memory Impairment<br />
Arthralgia<br />
Neck Pain<br />
Rash<br />
Pruritus<br />
Confusional State<br />
Headache<br />
Fatigue<br />
Myalgia<br />
Skin Disorder<br />
Skin Lesion<br />
Anxiety<br />
Date:12/22/05ISR Number: 4864524-5Report Type:Expedited (15-DaCompany Report #IT-ROCHE-426078 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Acute Lariam PS Roche ORAL 62 DAY<br />
Date:12/22/05ISR Number: 4867674-2Report Type:Direct Company Report #CTU 265598 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Mental Disorder Lariam 150 Mg.<br />
Other Serious Stress Not Sure Hoffman<br />
Required Feeling Abnormal Laroche, Inc. PS Hoffman Laroche, Inc ORAL 1 PILL EVERY
Intervention to Self Injurious Behaviour WEEK PO<br />
Prevent Permanent Paranoia<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 680
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/23/05ISR Number: 4865446-6Report Type:Expedited (15-DaCompany Report #AU-ROCHE-428550 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia <strong>Mefloquine</strong><br />
Initial or Prolonged Agitation Hydrochloride PS Roche ORAL ONE DAY<br />
Illusion TREATMENT. 1 DAY<br />
Date:12/23/05ISR Number: 4865465-XReport Type:Expedited (15-DaCompany Report #CH-ROCHE-428290 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - General Physical Health <strong>Mefloquine</strong><br />
Initial or Prolonged Deterioration Hydrochloride PS Roche UNKNOWN RECEIVED A<br />
Pulmonary Toxicity<br />
TOTAL DOSE OF<br />
Blood Pressure Decreased<br />
Diffuse Alveolar Damage<br />
1500MG OF<br />
MEFLOQUINE<br />
OVER 24 HOURS<br />
Paracetamol<br />
C<br />
Halofantrine C FOR 48 HOURS<br />
Date:12/23/05ISR Number: 4865509-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-420600 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Inflammation Lariam PS Roche ORAL<br />
Joint Swelling<br />
Arrhythmia<br />
Erythema<br />
Chills<br />
Dyspnoea<br />
Date:12/28/05ISR Number: 4867307-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-407146 Age:64 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vision Blurred Lariam PS Roche ORAL 29 DAY<br />
Initial or Prolonged Vertigo Xalacom SS TOPICAL<br />
Atrioventricular Block Fenofibrate SS ORAL<br />
Complete Captopril SS Roche ORAL<br />
Fall<br />
Atrioventricular Block<br />
Date:12/28/05ISR Number: 4867316-6Report Type:Expedited (15-DaCompany Report #AU-ROCHE-429059 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Exposure During Lariam PS Roche ORAL 29 DAY<br />
Pregnancy<br />
Abortion Spontaneous
Date:12/28/05ISR Number: 4867318-XReport Type:Expedited (15-DaCompany Report #AU-ROCHE-429052 Age:42 YR Gender: I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Oropharyngeal Pain<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 681
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dizziness<br />
Hallucination<br />
Vomiting Report Source Product Role Manufacturer Route Dose Duration<br />
Dysphagia Lariam PS Roche ORAL 41 DAY<br />
Mouth Ulceration Unspecified Drug C REPORTED AS<br />
Pruritus<br />
ADT.<br />
Gastrooesophageal Reflux Havrix C<br />
Disease<br />
Date:12/28/05ISR Number: 4870956-1Report Type:Expedited (15-DaCompany Report #343474 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Chest X-Ray Abnormal<br />
Exercise Tolerance<br />
Decreased<br />
Impaired Work Ability<br />
Palatal Disorder<br />
Feelings Of Worthlessness<br />
Sinusitis<br />
Asthenia<br />
Treatment Noncompliance<br />
Hyperhidrosis<br />
Amnesia<br />
Gastrointestinal Disorder<br />
Hypertension<br />
Diastolic Dysfunction<br />
Psychotic Disorder<br />
Bronchospasm<br />
Cardiac Murmur<br />
Sinus Tachycardia<br />
Aggression<br />
Drug Intolerance<br />
Collagen-Vascular Disease<br />
Blood Triglycerides<br />
Abnormal<br />
Low Density Lipoprotein<br />
Abnormal<br />
Weight Decreased<br />
Feeling Guilty<br />
Cognitive Disorder<br />
Dislocation Of Vertebra<br />
Intervertebral Disc<br />
Degeneration<br />
Spinal X-Ray Abnormal<br />
Vision Blurred<br />
Injury<br />
Restrictive Pulmonary<br />
Disease<br />
Bipolar Disorder<br />
Blood Cholesterol<br />
Increased
Rhinitis<br />
Blood Cholesterol<br />
Abnormal<br />
Total Cholesterol/Hdl<br />
Ratio Abnormal<br />
Pharyngeal Disorder<br />
Oxygen Saturation<br />
Decreased<br />
03-Apr-2012 09:37 AM Page: 682
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Weight Loss Poor<br />
Drug Effect Decreased<br />
Paraesthesia<br />
Diplopia<br />
Vestibular Disorder<br />
Clumsiness<br />
Asthma<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Schizoaffective Disorder<br />
Diarrhoea<br />
Insomnia<br />
Hyperacusis<br />
Nocturia<br />
Jaw Disorder<br />
Neoplasm<br />
Musculoskeletal Pain<br />
Post-Traumatic Neck<br />
Syndrome<br />
Rhinitis Perennial<br />
Self Esteem Decreased<br />
Headache<br />
Angina Pectoris<br />
Hypersomnia<br />
Coordination Abnormal<br />
Arthralgia<br />
Bronchitis<br />
Dehydration<br />
Dry Skin<br />
Dry Mouth<br />
Upper-Airway Cough<br />
Syndrome<br />
Neck Pain<br />
Listless<br />
Disturbance In Attention<br />
Cervical Spine Flattening<br />
Tinnitus<br />
Confusional State<br />
Fatigue<br />
Visual Impairment<br />
Affective Disorder<br />
Memory Impairment<br />
Viral Infection<br />
Rash<br />
Vitiligo<br />
Acrochordon<br />
Angiotensin Converting<br />
Enzyme Increased<br />
Blood Glucose Increased<br />
Gastrooesophageal Reflux<br />
Disease<br />
Sleep Apnoea Syndrome<br />
Haemorrhoids
Oedema<br />
Obesity<br />
Myalgia<br />
Rectal Haemorrhage<br />
Colitis<br />
Psychomotor Hyperactivity<br />
Mental Status Changes<br />
Nightmare<br />
03-Apr-2012 09:37 AM Page: 683
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Musculoskeletal Stiffness<br />
Brain Stem Auditory<br />
Evoked Response Abnormal Report Source Product Role Manufacturer Route Dose Duration<br />
Photosensitivity Reaction Consumer Lariam (<strong>Mefloquine</strong><br />
Cold Sweat Other Hydrochloride) 250<br />
Multiple Allergies Mg PS ORAL 250 MG, 1 PER<br />
Ventricular Hypertrophy<br />
1 WEEK, ORAL<br />
Scratch<br />
Zoloft (Sertraline<br />
Low Density Lipoprotein Hydrochloride) C<br />
Increased<br />
Chest Discomfort<br />
Upper Respiratory Tract<br />
Infection<br />
Exercise Lack Of<br />
Lung Neoplasm<br />
Pulmonary Granuloma<br />
Nausea<br />
Anhedonia<br />
Dizziness<br />
Anxiety<br />
Mitral Valve Prolapse<br />
Depression<br />
Abnormal Dreams<br />
Condition Aggravated<br />
Balance Disorder<br />
Major Depression<br />
Contusion<br />
Snoring<br />
Constipation<br />
Tremor<br />
Embolism Arterial<br />
Musculoskeletal<br />
Discomfort<br />
Hallucinations, Mixed<br />
Road Traffic Accident<br />
Disorientation<br />
Skin Disorder<br />
Weight Increased<br />
Blood Triglycerides<br />
Increased<br />
Total Cholesterol/Hdl<br />
Ratio Decreased<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Exfoliative Rash<br />
Dry Eye<br />
Gastrointestinal Tract<br />
Mucosal Discolouration<br />
Date:01/04/06ISR Number: 4871979-9Report Type:Expedited (15-DaCompany Report #ZM-ROCHE-430119 Age:28 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nephrotic Syndrome <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL 3 DAY<br />
03-Apr-2012 09:37 AM Page: 684
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/05/06ISR Number: 4873359-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-429867 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pericardial Effusion Lariam PS Roche ORAL<br />
Hypersensitivity<br />
Arrhythmia<br />
Myocarditis<br />
Asthenia<br />
Date:01/10/06ISR Number: 4876923-6Report Type:Expedited (15-DaCompany Report #DE-ROCHE-430544 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Lactate Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Dehydrogenase Increased Valette C ORAL<br />
Jaundice<br />
Blood Alkaline<br />
Phosphatase Increased<br />
Pyrexia<br />
Epigastric Discomfort<br />
Gallbladder Oedema<br />
Hiatus Hernia<br />
Cholecystitis Acute<br />
Transaminases Increased<br />
Ocular Icterus<br />
Date:01/11/06ISR Number: 4879688-7Report Type:Expedited (15-DaCompany Report #2005-BP-21825RO Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dysphoria Foreign <strong>Mefloquine</strong><br />
Initial or Prolonged Nausea Literature (<strong>Mefloquine</strong>) PS ORAL 250 MG/WEEK X<br />
Treatment Noncompliance Health 8 WEEKS (250<br />
Dry Mouth Professional MG), PO<br />
Weight Decreased<br />
Alprazolam<br />
Anxiety (Alprazolam) C<br />
Asthenia<br />
Fluoxetine<br />
Tremor (Fluoxetine) C<br />
Sleep Disorder<br />
Depression<br />
Agitation<br />
Date:01/16/06ISR Number: 4881711-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-386256 Age:23 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Depression<br />
Fear<br />
Nervousness<br />
Suicidal Ideation
Malaise<br />
Paranoia<br />
Toxicity To Various<br />
Agents<br />
Anxiety<br />
Skin Odour Abnormal<br />
Photosensitivity Reaction<br />
Photophobia<br />
03-Apr-2012 09:37 AM Page: 685
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Epistaxis<br />
Abnormal Dreams<br />
Feeling Abnormal Report Source Product Role Manufacturer Route Dose Duration<br />
Lethargy Lariam PS Roche ORAL<br />
Marijuana SS ORAL<br />
Date:01/20/06ISR Number: 4889556-2Report Type:Direct Company Report #CTU 267348 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Disorder Lariam 250 Mg Barr<br />
Anxiety Laboratories, Inc. PS Barr Laboratories,<br />
Inc. ORAL ONE 250 MG<br />
PILL ONCE A<br />
WEEK PO<br />
Date:01/23/06ISR Number: 4891871-3Report Type:Expedited (15-DaCompany Report #430544 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hiatus Hernia Foreign Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Cholecystitis Acute Literature Hydrochloride) PS<br />
Health<br />
Professional<br />
Valette<br />
(Dienogest/Ethinyl<br />
Estradiol)<br />
C<br />
Date:01/24/06ISR Number: 4890014-XReport Type:Expedited (15-DaCompany Report #DK-ROCHE-430861 Age:34 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Lariam PS Roche ORAL ORDINARY<br />
White Blood Cell Count<br />
GROWN-UP<br />
Decreased<br />
PROPHYLACTIC<br />
Blood Thyroid Stimulating<br />
LARIAM DOSIS.<br />
Hormone Increased<br />
Leukopenia<br />
Date:01/24/06ISR Number: 4891009-2Report Type:Direct Company Report #CTU 267616 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Lariam PS ORAL 1 PER WEEK<br />
Initial or Prolonged Heart Rate Irregular FIVE WEEKS PO<br />
Panic Attack<br />
Atrial Fibrillation<br />
Chest Pain<br />
Nausea<br />
Anxiety
Date:01/26/06ISR Number: 4892620-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-392506 Age:42 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Illogical Thinking<br />
Grand Mal Convulsion<br />
Amnesia<br />
03-Apr-2012 09:37 AM Page: 686
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disturbance In Attention<br />
Headache<br />
Visual Impairment Report Source Product Role Manufacturer Route Dose Duration<br />
Palpitations Lariam PS Roche ORAL 9 DAY<br />
Brain Injury Lexinor C ORAL<br />
Fatigue Malarone C ORAL<br />
Date:01/27/06ISR Number: 4894727-5Report Type:Direct Company Report #CTU 267976 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS 1 PILL PER<br />
Required Paranoia WEEK<br />
Intervention to<br />
Weight Increased<br />
Prevent Permanent Heart Rate Increased<br />
Impairment/Damage Hypertension<br />
Date:01/30/06ISR Number: 4895159-6Report Type:Expedited (15-DaCompany Report #DE-JNJFOC-20051002297 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Imodium PS ORAL<br />
Initial or Prolonged Overdose Gelonida SS ORAL<br />
Liver Function Test Talvosilen SS ORAL<br />
Abnormal Lariam SS ORAL 16 tablets.<br />
Somnolence Talvosilen SS ORAL<br />
Gelonida SS ORAL<br />
Ibuprofen SS ORAL 20 tablets.<br />
Date:01/30/06ISR Number: 4895544-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-426242 Age:10 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nightmare Lariam PS Roche UNKNOWN 25 DAY<br />
Initial or Prolonged Syncope<br />
Malaise<br />
Urinary Incontinence<br />
Visual Impairment<br />
Date:01/31/06ISR Number: 4896641-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-418484 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lung Disorder Lariam PS Roche ORAL<br />
Date:02/01/06ISR Number: 4897778-XReport Type:Expedited (15-DaCompany Report #DE-JNJFOC-20051002297 Age: Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Suicidal Ideation Imodium PS ORAL<br />
Initial or Prolonged Liver Function Test Gelonida SS ORAL<br />
Abnormal Talvosilen SS ORAL<br />
Somnolence Lariam SS ORAL 16 tablets.<br />
Overdose Talvosilen SS ORAL<br />
Gelonida SS ORAL<br />
Ibuprofen SS ORAL 20 tablets.<br />
03-Apr-2012 09:37 AM Page: 687
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/01/06ISR Number: 4898042-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-391660 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mood Swings Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Amnesia<br />
Disability<br />
Self-Medication<br />
Feeling Guilty<br />
Memory Impairment<br />
Suicidal Ideation<br />
Fatigue<br />
Mental Disorder<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder<br />
Anxiety<br />
Nervousness<br />
Self Esteem Decreased<br />
Decreased Interest<br />
Panic Attack<br />
Social Phobia<br />
Disturbance In Attention<br />
Date:02/02/06ISR Number: 4899221-3Report Type:Expedited (15-DaCompany Report #AU-ROCHE-433139 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Lariam PS Roche ORAL 15 DAY<br />
Initial or Prolonged Dizziness<br />
Haemolytic Anaemia<br />
Headache<br />
Nausea<br />
Myalgia<br />
Cardiac Murmur<br />
Alopecia<br />
Date:02/03/06ISR Number: 4900280-XReport Type:Expedited (15-DaCompany Report #SE-ROCHE-392506 Age:42 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Palpitations Lariam PS Roche ORAL 9 DAY<br />
Initial or Prolonged Visual Impairment Lexinor C ORAL<br />
Grand Mal Convulsion Malarone C ORAL<br />
Amnesia<br />
Disturbance In Attention<br />
Headache<br />
Feeling Abnormal<br />
Fatigue<br />
Cerebral Disorder
Date:02/09/06ISR Number: 4909064-XReport Type:Direct Company Report #CTU 269215 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Deafness <strong>Mefloquine</strong> 250 Mg<br />
Intervention to Generic PS ORAL 250 MG 3 X<br />
Prevent Permanent WITHIN' 5<br />
Impairment/Damage<br />
DAYS PO<br />
03-Apr-2012 09:37 AM Page: 688
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/10/06ISR Number: 4910665-3Report Type:Direct Company Report #USP 57779 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Drug Dispensing Error Lariam PS Roche TABLET<br />
Medication Error Zaroxolyn SS Cell Tech TABLET<br />
Wrong Drug Administered<br />
Date:02/15/06ISR Number: 4912780-7Report Type:Expedited (15-DaCompany Report #SE-ROCHE-433848 Age:73 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nephropathy Toxic Lariam PS Roche ORAL 8 WK<br />
Initial or Prolonged<br />
Date:02/15/06ISR Number: 4912790-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Insomnia<br />
Nightmare<br />
Irritable Bowel Syndrome<br />
Disturbance In Attention<br />
Pharyngitis Streptococcal<br />
Rash<br />
Phonophobia<br />
Blood Pressure Increased<br />
Epididymitis<br />
Agitation<br />
Dizziness<br />
Mood Swings<br />
Anxiety Disorder<br />
Fatigue<br />
Vertigo<br />
Nystagmus<br />
Inner Ear Disorder<br />
Eye Pain<br />
Pain Of Skin<br />
Night Sweats<br />
Nausea<br />
Diarrhoea<br />
Amnesia<br />
Abdominal Pain<br />
Testicular Pain<br />
Depression<br />
Colitis<br />
Hypoaesthesia<br />
Nervousness<br />
Activities Of Daily<br />
Living Impaired<br />
Vomiting<br />
Disorientation
Psychotic Disorder<br />
Memory Impairment<br />
Headache<br />
Vestibular Disorder<br />
Chest Pain<br />
Tinnitus<br />
Vision Blurred<br />
Photophobia<br />
03-Apr-2012 09:37 AM Page: 689
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Upper Respiratory Tract<br />
Infection<br />
Weight Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Imodium<br />
C<br />
Compazine C ON 18 APR<br />
2001, THERAPY<br />
REPORTED AS<br />
TWICE DAILY.<br />
Antibiotics Nos C<br />
Ambien<br />
C<br />
Prednisone C BY TAPER.<br />
Valium C ON 18 APR<br />
2001, THERAPY<br />
REPORTED AS:<br />
AS NEEDED.<br />
Phenergan<br />
C<br />
Biaxin<br />
C<br />
Advil<br />
C<br />
Bactrim<br />
C<br />
Antivert<br />
C<br />
Date:02/16/06ISR Number: 4914041-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-359452 Age:53 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Respiratory Failure Lariam PS Roche ORAL DOSAGE<br />
Initial or Prolonged Pulmonary Fibrosis REPORTED AS<br />
Lung Disorder 1.1 AND 1.2<br />
Toxicity To Various<br />
MG/L.<br />
Agents Unknown Medication C INTRAVENOUS DRUG REPORTED<br />
AS CLAMOXIL. 3 DAY<br />
Date:02/17/06ISR Number: 4915831-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-435505 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Restlessness Lariam PS Roche ORAL 22 DAY<br />
Anxiety<br />
Chest Discomfort<br />
Depression<br />
Panic Attack<br />
Date:02/17/06ISR Number: 4920328-6Report Type:Expedited (15-DaCompany Report #GXKR2006IT00866 Age:56 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Vision Blurred<br />
Leukocytosis<br />
Thrombotic
Thrombocytopenic Purpura<br />
Anaemia<br />
Neutrophil Count<br />
Increased<br />
Confusional State<br />
Ocular Icterus<br />
Anisocytosis<br />
Myalgia<br />
03-Apr-2012 09:37 AM Page: 690
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Lethargy<br />
Decreased Appetite<br />
Low Density Lipoprotein Report Source Product Role Manufacturer Route Dose Duration<br />
Increased Foreign <strong>Mefloquine</strong><br />
Plasmapheresis Literature (Ngx)(<strong>Mefloquine</strong>)<br />
Asthenia Health Tablet PS 250 MG,QW<br />
Professional<br />
Date:02/20/06ISR Number: 4916768-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-424819 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Lariam PS Roche ORAL<br />
Alcohol Use<br />
Stress<br />
Anxiety<br />
Persecutory Delusion<br />
Date:02/23/06ISR Number: 4920624-2Report Type:Expedited (15-DaCompany Report #SE-ROCHE-436796 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Trisomy 21 Lariam PS Roche ORAL<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:02/24/06ISR Number: 4922524-0Report Type:Expedited (15-DaCompany Report #IT-ROCHE-436175 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Gastrointestinal Lariam PS Roche ORAL 36 DAY<br />
Haemorrhage<br />
Thrombocytopenia<br />
Headache<br />
Vomiting<br />
Cerebral Haemorrhage<br />
Coma<br />
Date:02/27/06ISR Number: 4924436-5Report Type:Expedited (15-DaCompany Report #ZM-ROCHE-430119 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nephrotic Syndrome <strong>Mefloquine</strong><br />
Initial or Prolonged Abdominal Distension Hydrochloride PS Roche ORAL 3 DAY<br />
Glucose Urine Present<br />
Respiratory Rate<br />
Increased
Date:02/28/06ISR Number: 4928936-3Report Type:Direct Company Report #CTU 270859 Age:22 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Anxiety<br />
Mental Disorder<br />
Panic Attack<br />
Hyperhidrosis<br />
03-Apr-2012 09:37 AM Page: 691
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Fear<br />
Palpitations<br />
Dyspnoea Report Source Product Role Manufacturer Route Dose Duration<br />
Thinking Abnormal<br />
Larium/<strong>Mefloquine</strong><br />
Hallucination Roche PS Roche ORAL ONE PILL<br />
Diarrhoea<br />
ONCE PER WEEK<br />
Feeling Abnormal<br />
PO<br />
Date:03/06/06ISR Number: 4933631-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-432400 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sleep Disorder Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Dyspnoea Exertional Lariam SS Roche UNKNOWN 25 DAY<br />
Tachycardia<br />
Anxiety<br />
Hyperventilation<br />
Neurosis<br />
Anaemia<br />
Malaise<br />
Histrionic Personality<br />
Disorder<br />
Asthenia<br />
Vertigo<br />
Conversion Disorder<br />
Date:03/06/06ISR Number: 4933637-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-436796 Age:38 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Trisomy 21 Lariam PS Roche ORAL<br />
Abortion<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:03/06/06ISR Number: 4933650-4Report Type:Expedited (15-DaCompany Report #DK-ROCHE-430861 Age:34 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sensory Disturbance Lariam PS Roche ORAL ORDINARY<br />
Monocyte Count Abnormal<br />
GROWN-UP<br />
Neutrophil Count Abnormal<br />
PROPHYLACTIC<br />
White Blood Cell Count<br />
LARIAM DOSIS.<br />
Decreased<br />
Hypoaesthesia<br />
Blood Thyroid Stimulating<br />
Hormone Increased<br />
Abortion<br />
Alanine Aminotransferase<br />
Abnormal
Lymphocyte Count Abnormal<br />
Leukopenia<br />
03-Apr-2012 09:37 AM Page: 692
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/09/06ISR Number: 4937024-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Anxiety<br />
Panic Disorder<br />
Major Depression<br />
Psychiatric Symptom<br />
Fall<br />
Toxicity To Various<br />
Agents<br />
Convulsion<br />
Nocturia<br />
Insomnia<br />
Disturbance In Attention<br />
Selective Iga<br />
Immunodeficiency<br />
Fatigue<br />
Ill-Defined Disorder<br />
Chest Discomfort<br />
Joint Dislocation<br />
Respiratory Rate<br />
Increased<br />
Rheumatoid Factor<br />
Increased<br />
Dizziness<br />
Abnormal Behaviour<br />
Memory Impairment<br />
Hyperhidrosis<br />
Tremor<br />
Bundle Branch Block Right<br />
Exostosis<br />
Hypoaesthesia<br />
Myalgia<br />
Mood Swings<br />
Visual Impairment<br />
Hallucination, Visual<br />
Dyspnoea<br />
Post-Traumatic Stress<br />
Disorder<br />
Decreased Activity<br />
Antinuclear Antibody<br />
Positive<br />
Rhinitis Allergic<br />
Ventricular Extrasystoles<br />
Balance Disorder<br />
Asthenia<br />
Chest Pain<br />
Psychotic Disorder<br />
Generalised Anxiety<br />
Disorder<br />
Mitral Valve<br />
Calcification<br />
Dehydration
Weight Decreased<br />
Oxygen Saturation<br />
Decreased<br />
Eye Movement Disorder<br />
Eosinophilia<br />
Coordination Abnormal<br />
Mood Altered<br />
Aggression<br />
03-Apr-2012 09:37 AM Page: 693
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Suicidal Ideation<br />
Sleep Apnoea Syndrome<br />
Meniscus Lesion Report Source Product Role Manufacturer Route Dose Duration<br />
Thinking Abnormal Lariam PS Roche ORAL<br />
Dysthymic Disorder Primaquine C<br />
Attention Cipro C<br />
Deficit/Hyperactivity<br />
Disorder<br />
Ligament Sprain<br />
Rash Papular<br />
Cervical Vertebral<br />
Fracture<br />
Bronchopneumonia<br />
Agitation<br />
Depression<br />
Bronchial Hyperreactivity<br />
Productive Cough<br />
Cranial Nerve Disorder<br />
Hypertension<br />
Self-Injurious Ideation<br />
Cognitive Disorder<br />
Mitral Valve Incompetence<br />
Humerus Fracture<br />
Contusion<br />
Pyrexia<br />
Tinnitus<br />
Vertigo<br />
Headache<br />
Musculoskeletal Pain<br />
Asthma<br />
Homicidal Ideation<br />
Nystagmus<br />
Upper Respiratory Tract<br />
Infection<br />
Date:03/09/06ISR Number: 4988015-6Report Type:Periodic Company Report #GXBR2005US01407 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Back Pain Consumer <strong>Mefloquine</strong> Hcl<br />
Initial or Prolonged Panic Attack Tablets<br />
Hypoaesthesia<br />
(Ngx)(<strong>Mefloquine</strong>)<br />
Neck Pain Tablet PS ORAL 250 MG, QW,<br />
Asthenia<br />
ORAL<br />
Headache Motrin (Ibuprofen) C<br />
Dysphemia<br />
Muscle Spasms<br />
Tremor<br />
Disturbance In Attention<br />
Anxiety
Date:03/09/06ISR Number: 4988019-3Report Type:Periodic Company Report #GXBR2005US00770 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Platelet Count Decreased Consumer <strong>Mefloquine</strong> Hcl<br />
Initial or Prolonged<br />
Tablets<br />
(Ngx)(<strong>Mefloquine</strong>)<br />
Tablet PS OPHTHALMIC 250 MG QW,<br />
03-Apr-2012 09:37 AM Page: 694
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Z-Pak (Azithromycin) C<br />
Prednisone<br />
(Prednisone)<br />
C<br />
Albuterol<br />
(Salbutamol)<br />
C<br />
Advair Diskus<br />
(Fluticasone<br />
Propionate,<br />
Salmeterol<br />
Xinafoate)<br />
C<br />
ORAL<br />
Date:03/13/06ISR Number: 4941688-6Report Type:Expedited (15-DaCompany Report #SE-ROCHE-435563 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Arthropod Bite Lariam PS Roche ORAL 56 DAY<br />
Initial or Prolonged Headache<br />
Depressed Mood<br />
Sleep Disorder<br />
Hyperaesthesia<br />
Date:03/14/06ISR Number: 4943232-6Report Type:Expedited (15-DaCompany Report #DK-ROCHE-430861 Age:34 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Blood Thyroid Stimulating Lariam PS Roche ORAL ORDINARY<br />
Hormone Increased<br />
GROWN-UP<br />
Leukopenia<br />
PROPHYLACTIC<br />
Hypoaesthesia<br />
LARIAM DOSIS.<br />
White Blood Cell Count<br />
Decreased<br />
Hypothyroidism<br />
Date:03/14/06ISR Number: 4943235-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-424819 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Lariam PS Roche ORAL<br />
Delusion<br />
Confusional State<br />
Persecutory Delusion<br />
Anxiety<br />
Date:03/14/06ISR Number: 4943292-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-391660 Age: Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Suicidal Ideation<br />
Mental Disorder
Disability<br />
Generalised Anxiety<br />
Disorder<br />
Major Depression<br />
Feeling Guilty<br />
Panic Attack<br />
Amnesia<br />
Self Esteem Decreased<br />
Mood Swings<br />
03-Apr-2012 09:37 AM Page: 695
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Memory Impairment<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche UNKNOWN<br />
Date:03/15/06ISR Number: 4944593-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-439267 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anhedonia Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Pain<br />
Disability<br />
Cognitive Disorder<br />
Injury<br />
Mental Disorder<br />
Date:03/20/06ISR Number: 4948316-4Report Type:Expedited (15-DaCompany Report #IT-ROCHE-439289 Age:56 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Thrombotic <strong>Mefloquine</strong><br />
Initial or Prolonged Thrombocytopenic Purpura Hydrochloride PS Roche ORAL<br />
Asthenia<br />
Pyrexia<br />
Vision Blurred<br />
Confusional State<br />
Decreased Appetite<br />
Myalgia<br />
Lethargy<br />
Date:03/20/06ISR Number: 4948344-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Agitation<br />
Mood Swings<br />
Depression<br />
Asthenia<br />
Headache<br />
Post-Traumatic Stress<br />
Disorder<br />
Meniscus Lesion<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Suicidal Ideation<br />
Productive Cough<br />
Decreased Activity<br />
Dysthymic Disorder<br />
Bundle Branch Block Right<br />
Eye Movement Disorder
Contusion<br />
Anxiety<br />
Bronchial Hyperreactivity<br />
Thinking Abnormal<br />
Brain Stem Syndrome<br />
Memory Impairment<br />
Ill-Defined Disorder<br />
Dyspnoea<br />
03-Apr-2012 09:37 AM Page: 696
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Chest Discomfort<br />
Hypertension<br />
Psychiatric Symptom Report Source Product Role Manufacturer Route Dose Duration<br />
Musculoskeletal Pain Lariam PS Roche ORAL<br />
Homicidal Ideation Primaquine C<br />
Convulsion Cipro C<br />
Rheumatoid Factor<br />
Increased<br />
Nocturia<br />
Pyrexia<br />
Rash Papular<br />
Abnormal Behaviour<br />
Aggression<br />
Selective Iga<br />
Immunodeficiency<br />
Cranial Nerve Disorder<br />
Joint Dislocation<br />
Generalised Anxiety<br />
Disorder<br />
Asthma<br />
Cognitive Disorder<br />
Mitral Valve Incompetence<br />
Humerus Fracture<br />
Dehydration<br />
Weight Decreased<br />
Respiratory Rate<br />
Increased<br />
Antinuclear Antibody<br />
Positive<br />
Rhinitis Allergic<br />
Hallucination, Visual<br />
Balance Disorder<br />
Fatigue<br />
Panic Disorder<br />
Self-Injurious Ideation<br />
Oxygen Saturation<br />
Decreased<br />
Nystagmus<br />
Eosinophilia<br />
Insomnia<br />
Visual Impairment<br />
Chest Pain<br />
Hyperhidrosis<br />
Tremor<br />
Craniocerebral Injury<br />
Toxicity To Various<br />
Agents<br />
Hypoaesthesia<br />
Disturbance In Attention<br />
Vertigo<br />
Sleep Apnoea Syndrome<br />
Psychotic Disorder<br />
Major Depression
Fall<br />
Mitral Valve<br />
Calcification<br />
Exostosis<br />
Ligament Sprain<br />
Myalgia<br />
Ventricular Extrasystoles<br />
03-Apr-2012 09:37 AM Page: 697
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/24/06ISR Number: 4958844-3Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Hypoaesthesia<br />
Humerus Fracture<br />
Mitral Valve Incompetence<br />
Mitral Valve<br />
Calcification<br />
Aggression<br />
Meniscus Lesion<br />
Major Depression<br />
Panic Disorder<br />
Chest Pain<br />
Visual Impairment<br />
Memory Impairment<br />
Abnormal Behaviour<br />
Cough<br />
Angina Pectoris<br />
Joint Lock<br />
Deafness<br />
Anger<br />
Upper Respiratory Tract<br />
Infection<br />
Eructation<br />
Abdominal Pain Upper<br />
Mucous Stools<br />
Exostosis<br />
Homicidal Ideation<br />
Post-Traumatic Stress<br />
Disorder<br />
Joint Dislocation<br />
Vestibular Function Test<br />
Abnormal<br />
Headache<br />
Balance Disorder<br />
Snoring<br />
Speech Disorder<br />
Joint Crepitation<br />
Diarrhoea<br />
Rash Macular<br />
Pyrexia<br />
Myalgia<br />
Rhinitis Allergic<br />
Ligament Sprain<br />
Convulsion<br />
Hypertension<br />
Sleep Apnoea Syndrome<br />
Productive Cough<br />
Poor Quality Drug<br />
Administered<br />
Weight Increased<br />
Thinking Abnormal<br />
Sedation
Tongue Eruption<br />
Arthralgia<br />
Rectal Haemorrhage<br />
Eosinophilia<br />
Nystagmus<br />
Hyperhidrosis<br />
Fatigue<br />
Selective Iga<br />
03-Apr-2012 09:37 AM Page: 698
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Immunodeficiency<br />
Suicidal Ideation<br />
Agitation<br />
Insomnia<br />
Family Stress<br />
Nausea<br />
Ventricular Extrasystoles<br />
Tremor<br />
Nocturia<br />
Antinuclear Antibody<br />
Positive<br />
Bundle Branch Block Right<br />
Dysthymic Disorder<br />
Dyspnoea<br />
Asthenia<br />
Coordination Abnormal<br />
Vertigo<br />
Derealisation<br />
Exposure To Toxic Agent<br />
Wheezing<br />
Occupational Problem<br />
Environmental<br />
Chondroplasty<br />
Musculoskeletal Stiffness<br />
Breath Odour<br />
Injury<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Weight Decreased<br />
Generalised Anxiety<br />
Disorder<br />
Chest Discomfort<br />
Pulmonary Congestion<br />
Bronchial Hyperreactivity<br />
Hallucination, Visual<br />
Disturbance In Attention<br />
Acute Coronary Syndrome<br />
Psychiatric Symptom<br />
Tinnitus<br />
Hypervigilance<br />
Impaired Work Ability<br />
Constipation<br />
Decreased Activity<br />
Rash Papular<br />
Oxygen Saturation<br />
Decreased<br />
Respiratory Rate<br />
Increased<br />
Dehydration<br />
Toxicity To Various<br />
Agents<br />
Impulsive Behaviour<br />
Self-Injurious Ideation
Anxiety Disorder<br />
Depression<br />
Mood Swings<br />
Anxiety<br />
Dissociative Disorder<br />
Palpitations<br />
Dizziness<br />
Brain Stem Syndrome<br />
03-Apr-2012 09:37 AM Page: 699
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Visual Field Defect<br />
Rheumatoid Factor<br />
Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Contusion Other Lariam (<strong>Mefloquine</strong><br />
Eye Movement Disorder Hydrochloride) 250mg PS ORAL ORAL<br />
Cognitive Disorder<br />
Primaquine<br />
Asthma<br />
(Primaquine<br />
Musculoskeletal Pain Phosphate) C<br />
Fall<br />
Cirpo (Ciprofloxacin<br />
Psychotic Disorder Hydrochloride) C<br />
Priapism<br />
Paranoia<br />
Blood Potassium Decreased<br />
Emotional Distress<br />
Dyspepsia<br />
Abdominal Distension<br />
Date:03/24/06ISR Number: 4958848-0Report Type:Expedited (15-DaCompany Report #385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Agitation<br />
Product Quality Issue<br />
Hyperkeratosis<br />
Coronary Artery Disease<br />
Headache<br />
Dizziness<br />
Joint Dislocation<br />
Choking<br />
Weight Decreased<br />
Meniscus Lesion<br />
Respiratory Fume<br />
Inhalation Disorder<br />
Acute Coronary Syndrome<br />
Disturbance In Attention<br />
Visual Impairment<br />
Loose Body In Joint<br />
Fall<br />
Humerus Fracture<br />
Divorced<br />
Accident<br />
Hypertension<br />
Weight Increased<br />
Rhinitis Allergic<br />
Bundle Branch Block Right<br />
Arthralgia<br />
Anxiety<br />
Suicidal Ideation<br />
Chondroplasty<br />
Electrocardiogram T Wave<br />
Abnormal<br />
Chest X-Ray Abnormal
Haemophilus Infection<br />
Sleep Apnoea Syndrome<br />
Visual Field Defect<br />
Eosinophilia<br />
Cognitive Disorder<br />
Exposure To Noise<br />
Priapism<br />
Ventricular Extrasystoles<br />
03-Apr-2012 09:37 AM Page: 700
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Palpitations<br />
Snoring<br />
Abnormal Behaviour<br />
Aggression<br />
Vertigo<br />
Hallucination, Visual<br />
Toxicity To Various<br />
Agents<br />
Poor Quality Drug<br />
Administered<br />
Stress<br />
Hiatus Hernia<br />
Electrocardiogram St<br />
Segment Depression<br />
Upper Respiratory Tract<br />
Infection<br />
Mitral Valve<br />
Calcification<br />
Nightmare<br />
Ligament Sprain<br />
Nocturia<br />
Blood Creatine<br />
Phosphokinase Increased<br />
Psychiatric Symptom<br />
Insomnia<br />
Mood Swings<br />
Depression<br />
Cardiac Stress Test<br />
Abnormal<br />
Panic Attack<br />
Tinnitus<br />
Bacteraemia<br />
Selective Iga<br />
Immunodeficiency<br />
Marital Problem<br />
Exostosis<br />
Post-Traumatic Stress<br />
Disorder<br />
Vestibular Disorder<br />
Hyperhidrosis<br />
Abnormal Dreams<br />
Nystagmus<br />
Blood Potassium Decreased<br />
Asthma<br />
Rash<br />
Fatigue<br />
Mood Altered<br />
Chronic Sinusitis<br />
Rapid Eye Movements Sleep<br />
Abnormal<br />
Mitral Valve Incompetence<br />
Electroencephalogram<br />
Abnormal
Emotional Disorder<br />
Homicidal Ideation<br />
Performance Fear<br />
Loss Of Consciousness<br />
Eye Movement Disorder<br />
Memory Impairment<br />
Balance Disorder<br />
Coordination Abnormal<br />
03-Apr-2012 09:37 AM Page: 701
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Haemarthrosis<br />
Impulsive Behaviour<br />
Dysthymic Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Dehydration Other Lariam (<strong>Mefloquine</strong><br />
Paranoia Hydrochloride) 250<br />
Derealisation Mg PS ORAL ORAL<br />
Contusion<br />
Primaquine<br />
Antinuclear Antibody<br />
(Primaquine<br />
Positive Phosphate) C<br />
Rheumatoid Factor<br />
Cipro (Ciprofloxacin<br />
Increased Hydrochloride) C<br />
Chest Pain<br />
Musculoskeletal Stiffness<br />
Hypervigilance<br />
Meniscus Removal<br />
Conduction Disorder<br />
Pneumonia<br />
Deafness<br />
Bronchial Hyperreactivity<br />
Tremor<br />
Blood Pressure Increased<br />
Physical Assault<br />
Convulsion<br />
Hypoaesthesia<br />
Myalgia<br />
Date:04/03/06ISR Number: 4960823-7Report Type:Expedited (15-DaCompany Report #DE-ROCHE-427130 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Neuralgic Amyotrophy Lariam PS Roche UNKNOWN FOR FIVE TO<br />
Initial or Prolonged Paresis SIX WEEKS,<br />
BEGINNING THE<br />
END OF AUGUST<br />
2005.<br />
Date:04/10/06ISR Number: 4968399-5Report Type:Expedited (15-DaCompany Report #CH-ABBOTT-06P-151-0329756-00 Age:59 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paresis Klaricid Film-Coated<br />
Hepatic Cyst Tablets PS ORAL 5 DAY<br />
Paraesthesia <strong>Mefloquine</strong> SS<br />
<strong>Mefloquine</strong><br />
SS<br />
<strong>Mefloquine</strong> SS ORAL<br />
Klaricid Film-Coated<br />
Tablets SS 5 DAY<br />
Melisec<br />
C<br />
Thyorsian<br />
C<br />
Yecueine<br />
C<br />
Pepermint<br />
C
Salvia C<br />
Clavulin C ORAL 500 MG/125 MG<br />
03-Apr-2012 09:37 AM Page: 702
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/11/06ISR Number: 4969270-5Report Type:Expedited (15-DaCompany Report #CH-ROCHE-442702 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Paresis Lariam PS Roche ORAL<br />
Hepatic Cyst Lariam SS Roche ORAL TAKEN 6<br />
Drug Interaction<br />
CAPSULES OF<br />
Paraesthesia 250 MG IN 3<br />
Confusional State<br />
DAYS<br />
Drug Level Decreased (SELF-MEDICAT 4 DAY<br />
Fatigue Lariam SS Roche ORAL<br />
Augmentin C ORAL STRENGHTH<br />
REPORTED AS<br />
500 MG/ 125<br />
MG. FILM<br />
COATED 6 DAY<br />
Klacid I ORAL FILM COATED<br />
TABLETS. 6 DAY<br />
Date:04/13/06ISR Number: 4973397-1Report Type:Direct Company Report #CTU 274136 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack <strong>Mefloquine</strong> (Unknown<br />
Initial or Prolonged Manuf.) PS ORAL 250 MG<br />
WEEKLY P.O.<br />
Date:04/14/06ISR Number: 4973077-2Report Type:Expedited (15-DaCompany Report #CA-ROCHE-443261 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety <strong>Mefloquine</strong><br />
Delusion Hydrochloride PS Roche UNKNOWN 3 WK<br />
Paranoia Cipralex C UNKNOWN<br />
Depression Clonazepam C UNKNOWN<br />
Risperdal C UNKNOWN<br />
Date:04/18/06ISR Number: 4976519-1Report Type:Direct Company Report #CTU 274487 Age:41 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety <strong>Mefloquine</strong> 250mg<br />
Other Serious Dyspnoea Barr Laboratories, PS Barr Laboratories BUCCAL 250MG ONCE A<br />
Insomnia<br />
WEEK BUCCAL<br />
Chest Discomfort<br />
Date:04/18/06ISR Number: 4976726-8Report Type:Direct Company Report #CTU 274491 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Panic Attack <strong>Mefloquine</strong><br />
Hallucination<br />
Hydrochloride Tablet<br />
Tinnitus<br />
250 Mg<br />
Nightmare Sandoz, Inc PS Sandoz, Inc ORAL 1/4 TABLET<br />
Anxiety<br />
ONCE WEEKLY<br />
PO<br />
03-Apr-2012 09:37 AM Page: 703
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/21/06ISR Number: 4979539-6Report Type:Expedited (15-DaCompany Report #NG-ROCHE-444178 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Hypersensitivity<br />
Disorientation<br />
Incoherent<br />
Date:04/21/06ISR Number: 4979606-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-444160 Age:76 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Retinal Artery Occlusion Lariam PS Roche ORAL 29 DAY<br />
Unspecified<br />
Medication C ORAL<br />
Norflex C ORAL<br />
Voltarol C INTRAMUSCULAR<br />
Simvastatin C ORAL<br />
Date:04/24/06ISR Number: 4980521-3Report Type:Expedited (15-DaCompany Report #SE-ROCHE-444010 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Oedema Peripheral<br />
Nausea<br />
Cold Sweat<br />
Fatigue<br />
Date:04/25/06ISR Number: 4982322-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Abnormal Behaviour<br />
Visual Impairment<br />
Suicidal Ideation<br />
Selective Iga<br />
Immunodeficiency<br />
Cranial Nerve Disorder<br />
Joint Dislocation<br />
Hyperhidrosis<br />
Thinking Abnormal<br />
Eosinophilia<br />
Ventricular Extrasystoles<br />
Hypervigilance<br />
Ill-Defined Disorder<br />
Psychiatric Symptom<br />
Cognitive Disorder<br />
Dysthymic Disorder<br />
Oxygen Saturation
Decreased<br />
Antinuclear Antibody<br />
Positive<br />
Mood Swings<br />
Depression<br />
Hallucination, Visual<br />
Chest Pain<br />
Decreased Activity<br />
03-Apr-2012 09:37 AM Page: 704
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Respiratory Rate<br />
Increased<br />
Ligament Sprain Report Source Product Role Manufacturer Route Dose Duration<br />
Nocturia Lariam PS Roche ORAL<br />
Anxiety Primaquine C<br />
Fatigue Cipro C<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder<br />
Toxicity To Various<br />
Agents<br />
Bundle Branch Block Right<br />
Exostosis<br />
Dehydration<br />
Convulsion<br />
Eye Movement Disorder<br />
Insomnia<br />
Memory Impairment<br />
Vertigo<br />
Productive Cough<br />
Mitral Valve<br />
Calcification<br />
Nystagmus<br />
Disturbance In Attention<br />
Dyspnoea<br />
Hypertension<br />
Meniscus Lesion<br />
Fall<br />
Tremor<br />
Homicidal Ideation<br />
Craniocerebral Injury<br />
Mitral Valve Incompetence<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Contusion<br />
Myalgia<br />
Agitation<br />
Balance Disorder<br />
Chest Discomfort<br />
Headache<br />
Panic Disorder<br />
Sleep Apnoea Syndrome<br />
Psychotic Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
Musculoskeletal Pain<br />
Asthma<br />
Humerus Fracture<br />
Weight Decreased<br />
Aggression<br />
Asthenia<br />
Bronchial Hyperreactivity
Self-Injurious Ideation<br />
Hypoaesthesia<br />
Rheumatoid Factor<br />
Increased<br />
Rhinitis Allergic<br />
Pyrexia<br />
Rash Papular<br />
03-Apr-2012 09:37 AM Page: 705
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/28/06ISR Number: 4990843-8Report Type:Expedited (15-DaCompany Report #GXKR2006IN02239 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Areflexia Foreign <strong>Mefloquine</strong> (Ngx)<br />
Other Serious Abasia Literature (<strong>Mefloquine</strong>) PS ORAL SEE IMAGE 1 DAY<br />
Dysgraphia Health Chloroquine C<br />
Polyneuropathy<br />
Professional<br />
Rash Erythematous<br />
Other<br />
Hypokinesia<br />
Hypotonia<br />
Neuropathy Peripheral<br />
Date:04/28/06ISR Number: 4991115-8Report Type:Expedited (15-DaCompany Report #GXKR2006IN01102 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Asthenia Foreign <strong>Mefloquine</strong> (Ngx)<br />
Polyneuropathy Literature (<strong>Mefloquine</strong>) PS 1000 MG<br />
Dermatitis<br />
Infected Skin Ulcer<br />
Health<br />
Professional<br />
Other<br />
Date:05/01/06ISR Number: 4989706-3Report Type:Expedited (15-DaCompany Report #007178 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Confusional State Consumer <strong>Mefloquine</strong><br />
Heart Rate Increased<br />
Hydrochloride<br />
Decreased Appetite<br />
(<strong>Mefloquine</strong><br />
Suicidal Ideation<br />
Hydrochloride)<br />
Disturbance In Attention Tablet, 250mg PS ORAL 1 TABLET,<br />
Anxiety<br />
WEEKLY, ORAL<br />
Prenatal Vitamins<br />
(Ascorbic Acid,<br />
Vitamin D Nos,<br />
Minerals Nos,<br />
Vitamins B Nos, C<br />
Date:05/08/06ISR Number: 4995548-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-329897 Age:30 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Suicide Attempt<br />
Depression<br />
Paraesthesia<br />
Respiratory Disorder<br />
Panic Attack<br />
Dysthymic Disorder<br />
Heart Rate Increased<br />
Intentional Self-Injury
Fatigue<br />
Fear<br />
Claustrophobia<br />
Chest Pain<br />
Nightmare<br />
Headache<br />
Mood Swings<br />
Mental Impairment<br />
03-Apr-2012 09:37 AM Page: 706
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Gastrointestinal Disorder<br />
Overdose<br />
Anxiety Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia Lariam PS Roche ORAL TAKEN TWO<br />
DOSES IN<br />
TOTAL. 8 DAY<br />
Prozac C ORAL<br />
Trazodone C ORAL<br />
Date:05/11/06ISR Number: 4999408-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-446876 Age:65 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Acute Lariam PS Roche ORAL 1 DOSE<br />
Initial or Prolonged Dialysis WEEKLY. 6 DAY<br />
Lasilix C ORAL<br />
Solupred C ORAL 3 DOSES<br />
DAILY.<br />
Renitec C ORAL<br />
Neoral C ORAL<br />
Mopral C ORAL ONE DOSE<br />
DAILY.<br />
Date:05/11/06ISR Number: 4999413-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-446907 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Chest Pain Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Syncope Cilest C ORAL<br />
Blood Pressure Decreased<br />
Myalgia<br />
Musculoskeletal Stiffness<br />
Loss Of Consciousness<br />
Date:05/16/06ISR Number: 5004582-0Report Type:Direct Company Report #CTU 276678 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams <strong>Mefloquine</strong><br />
Anxiety<br />
Hydrochloride 250 Mg<br />
Mood Altered Roche Laboratories PS Roche Laboratories ORAL 1 TABLET ONCE<br />
Paranoia<br />
A WEEK PO<br />
Nausea<br />
Date:05/18/06ISR Number: 5005520-7Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398212 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nystagmus Lariam PS Roche ORAL TAKEN ON 22
Initial or Prolonged Balance Disorder FEB, 01 MAR<br />
Disability Diarrhoea AND 08 MAR<br />
Tinnitus 2005. 15 DAY<br />
Hearing Impaired<br />
Vertigo<br />
Dizziness<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 707
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/24/06ISR Number: 5009780-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-444178 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam PS Roche ORAL 15 DAY<br />
Initial or Prolonged Confusional State<br />
Disability<br />
Incoherent<br />
Hypersensitivity<br />
Depression<br />
Disorientation<br />
Date:05/24/06ISR Number: 5011972-9Report Type:Expedited (15-DaCompany Report #2006-BP-05361RO Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Infected Skin Ulcer Foreign <strong>Mefloquine</strong><br />
Polyneuropathy Literature (<strong>Mefloquine</strong>) PS ORAL 1000 MG<br />
Dermatitis Health COURSE, PO<br />
Skin Ulcer<br />
Professional<br />
Date:05/24/06ISR Number: 5011974-2Report Type:Expedited (15-DaCompany Report #2006-BP-05360RO Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Polyneuropathy Foreign <strong>Mefloquine</strong><br />
Hypotonia Literature (<strong>Mefloquine</strong>) PS ORAL 1000 MG DAY<br />
Hypoaesthesia Health 1, 500 MG DAY<br />
Rash Erythematous Professional 2, PO<br />
Dysstasia<br />
Abasia<br />
Burning Sensation<br />
Muscular Weakness<br />
Asthenia<br />
Activities Of Daily<br />
Living Impaired<br />
Paraesthesia<br />
Arthropathy<br />
Areflexia<br />
Date:05/26/06ISR Number: 5013961-7Report Type:Direct Company Report #CTU 277654 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam -<strong>Mefloquine</strong>- PS ORAL ONCE A WEEK<br />
Nightmare<br />
PO<br />
Feeling Abnormal<br />
Fear<br />
Mental Disorder<br />
Depression<br />
Suicidal Ideation<br />
Mental Impairment
Thinking Abnormal<br />
Malaise<br />
03-Apr-2012 09:37 AM Page: 708
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/30/06ISR Number: 5013837-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-448428 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Umbilical Malformation Lariam PS Roche ORAL<br />
Initial or Prolonged Maternal Exposure During<br />
Congenital Anomaly Pregnancy<br />
Abortion Induced<br />
Date:05/31/06ISR Number: 5014899-1Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398212 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nystagmus Lariam PS Roche ORAL TAKEN ON 22<br />
Initial or Prolonged Tinnitus FEB, 01 MAR<br />
Disability Balance Disorder AND 08 MAR<br />
Diarrhoea 2005. 15 DAY<br />
Deafness Unilateral<br />
Nausea<br />
Hearing Impaired<br />
Dizziness<br />
Vertigo<br />
Vestibular Neuronitis<br />
Date:05/31/06ISR Number: 5014941-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-441981 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Irritable Bowel Syndrome Lariam PS Roche UNKNOWN<br />
Personality Change<br />
Depression<br />
Date:06/01/06ISR Number: 5015586-6Report Type:Expedited (15-DaCompany Report #SE-ROCHE-449547 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Umbilical Malformation Lariam PS Roche ORAL<br />
Initial or Prolonged Abortion<br />
Maternal Exposure During<br />
Pregnancy<br />
Pregnancy<br />
Date:06/01/06ISR Number: 5036335-1Report Type:Periodic Company Report #402101 Age:36 YR Gender:Male I/FU:I<br />
Outcome<br />
Death<br />
PT<br />
Headache<br />
Decreased Appetite<br />
Chills<br />
Abdominal Pain Upper
Pyrexia<br />
Diarrhoea<br />
Anuria<br />
Respiratory Rate<br />
Increased<br />
Psychiatric Symptom<br />
Eye Disorder<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 709
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Local Swelling<br />
Nausea<br />
Oropharyngeal Pain Report Source Product Role Manufacturer Route Dose Duration<br />
Ear Congestion Consumer Lariam (<strong>Mefloquine</strong><br />
Balance Disorder Hydrochloride) PS ORAL ORAL<br />
Ear Disorder<br />
Dizziness<br />
Malaria<br />
Cough<br />
Swelling Face<br />
Date:06/01/06ISR Number: 5036336-3Report Type:Periodic Company Report #409218 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Loss Of Consciousness Consumer Lariam (<strong>Mefloquine</strong><br />
Insomnia Hydrochloride) 250<br />
Vision Blurred Mg PS ORAL ORAL<br />
Amnesia<br />
Depression<br />
Mental Impairment<br />
Disturbance In Attention<br />
Suicidal Ideation<br />
Balance Disorder<br />
Date:06/01/06ISR Number: 5036337-5Report Type:Periodic Company Report #411025 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Anxiety Hydrochloride) 250<br />
Mg PS ORAL ORAL 30 DAY<br />
Date:06/01/06ISR Number: 5036338-7Report Type:Periodic Company Report #412629 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Attack Health Lariam (<strong>Mefloquine</strong><br />
Depression Professional Hydrochloride) 250<br />
Paranoia Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Birth Control Pills<br />
Nos (Oral<br />
Contraceptive Nos) C<br />
Albuterol<br />
(Albuterol)<br />
C<br />
Lexapro<br />
(Escitalopram) C
Date:06/01/06ISR Number: 5036339-9Report Type:Periodic Company Report #425065 Age:22 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Suicidal Ideation<br />
Memory Impairment<br />
Enuresis<br />
Emotional Disorder<br />
03-Apr-2012 09:37 AM Page: 710
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Joint Injury<br />
Disorientation<br />
Alcohol Intolerance Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety Consumer Lariam (<strong>Mefloquine</strong><br />
Depression Hydrochloride) 250<br />
Neurological Examination Mg PS ORAL 250 MG 1 PER<br />
Abnormal<br />
WEEK ORAL<br />
Panic Attack<br />
Coordination Abnormal<br />
Abnormal Dreams<br />
Paranoia<br />
Date:06/01/06ISR Number: 5036340-5Report Type:Periodic Company Report #426777 Age:49 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/01/06ISR Number: 5036341-7Report Type:Periodic Company Report #427138 Age:69 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Health Lariam (<strong>Mefloquine</strong><br />
Required Depression Professional Hydrochloride) 250<br />
Intervention to Mg PS ORAL 1 PER WEEK<br />
Prevent Permanent<br />
Impairment/Damage<br />
Antidepressant Nos<br />
(Antidepressant Nos) C<br />
ORAL<br />
Date:06/01/06ISR Number: 5036342-9Report Type:Periodic Company Report #428956 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Health Hydrochloride) PS ORAL ORAL<br />
Professional<br />
Birth Control Pills<br />
Nos (Oral<br />
Contraceptive Nos) C<br />
Date:06/01/06ISR Number: 5036343-0Report Type:Periodic Company Report #336177 Age:40 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Lichen Planus<br />
Headache<br />
Suicidal Ideation
Hyperhidrosis<br />
Anhedonia<br />
Tinnitus<br />
Groin Pain<br />
Haematuria<br />
Vestibular Disorder<br />
Oral Disorder<br />
Heart Rate Increased<br />
03-Apr-2012 09:37 AM Page: 711
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paraesthesia<br />
Nightmare<br />
Blood Pressure Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Hyperventilation Consumer Lariam (<strong>Mefloquine</strong><br />
Depression Health Hydrochloride) 250<br />
Major Depression Professional Mg PS ORAL 250 MG ORAL<br />
Dysuria<br />
Other<br />
Rash Papular<br />
Vision Blurred<br />
Insomnia<br />
Mood Altered<br />
Nystagmus<br />
Urethritis<br />
Dyspnoea<br />
Toxicity To Various<br />
Agents<br />
Chest Discomfort<br />
Depressed Mood<br />
Agoraphobia<br />
Pain In Extremity<br />
Agitation<br />
Panic Disorder<br />
Epicondylitis<br />
Disturbance In Attention<br />
Vertigo<br />
Balance Disorder<br />
Panic Attack<br />
Anxiety<br />
Erythema<br />
Dizziness<br />
Fatigue<br />
Decreased Activity<br />
Decreased Interest<br />
Vitreous Floaters<br />
Activities Of Daily<br />
Living Impaired<br />
Date:06/01/06ISR Number: 5036344-2Report Type:Periodic Company Report #369650 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam (<strong>Mefloquine</strong><br />
Fatigue Hydrochloride) 250<br />
Hostility Mg PS ORAL ORAL<br />
Sleep Apnoea Syndrome<br />
Inderal<br />
Abnormal Dreams (Propranolol) C<br />
Memory Impairment<br />
Cimetidine<br />
Disorientation (Cimetidine) C<br />
Communication Disorder Xanax (Alprazolam) C<br />
Emotional Distress<br />
Dysarthria<br />
Gun Shot Wound
Mood Swings<br />
Agitation<br />
Self Esteem Decreased<br />
Tinnitus<br />
Fear<br />
03-Apr-2012 09:37 AM Page: 712
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/01/06ISR Number: 5036345-4Report Type:Periodic Company Report #380190 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Paranoia Other Lariam (<strong>Mefloquine</strong><br />
Night Sweats Hydrochloride) 250<br />
Fatigue Mg PS ORAL 1 DOSE FORM 1<br />
Gun Shot Wound<br />
PER WEEK ORAL<br />
Post-Traumatic Stress<br />
Disorder<br />
Social Avoidant Behaviour<br />
Delusion<br />
Diarrhoea<br />
Sleep Disorder<br />
Emotional Disorder<br />
Poor Quality Sleep<br />
Completed Suicide<br />
Insomnia<br />
Agitation<br />
Depression<br />
Mood Swings<br />
Headache<br />
Aggression<br />
Date:06/01/06ISR Number: 5036346-6Report Type:Periodic Company Report #384553 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Amnesia Health Lariam (<strong>Mefloquine</strong><br />
Headache Professional Hydrochloride) 250<br />
Mg PS ORAL 250 MG 1 PER<br />
WEEK ORAL<br />
Date:06/01/06ISR Number: 5036347-8Report Type:Periodic Company Report #395651 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Psychotic Disorder Other Lariam (<strong>Mefloquine</strong><br />
Back Pain Hydrochloride) 250<br />
Depression Mg PS ORAL 250 MG 1 PER<br />
Agitation<br />
WEEK ORAL<br />
Rash<br />
Glucosamine<br />
Pyrexia (Glucosamine) C<br />
Paraesthesia<br />
Chondroitin<br />
Hypoaesthesia<br />
(Chondroitin<br />
Headache Sulfate) C<br />
Tinnitus<br />
Celebrex (Celecoxib) C<br />
Fatigue<br />
Cough<br />
Myalgia<br />
Diarrhoea<br />
Mood Swings
Anger<br />
Completed Suicide<br />
Deafness<br />
Arthralgia<br />
Asthenia<br />
Spousal Abuse<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 713
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/01/06ISR Number: 5036348-XReport Type:Periodic Company Report #398411 Age:10 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Sleep Disorder Health Lariam (<strong>Mefloquine</strong><br />
Mood Swings Professional Hydrochloride) 250<br />
Insomnia Mg PS ORAL 1 PER WEEK<br />
ORAL<br />
Date:06/06/06ISR Number: 5019015-8Report Type:Expedited (15-DaCompany Report #FR-GLAXOSMITHKLINE-B0426071A Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Ineffective Malarone PS Glaxosmithkline ORAL 3 DAY<br />
Initial or Prolonged Hallucination Lariam SS ORAL 2250MG per<br />
Major Depression<br />
day<br />
Date:06/06/06ISR Number: 5019336-9Report Type:Expedited (15-DaCompany Report #SE-ROCHE-448488 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Angioedema Lariam PS Roche ORAL 2 MON<br />
Date:06/09/06ISR Number: 5022485-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-450157 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pericarditis Lariam PS Roche ORAL REPORTED AS<br />
Initial or Prolonged Ventricular Extrasystoles 250 MG AT<br />
Myocardial Infarction TOTAL. 1 DAY<br />
Sinus Tachycardia Mediator 150 SS ORAL 726 DAY<br />
Myocarditis Havrix C INTRAMUSCULAR 1 DOSE. 1 DAY<br />
Extrasystoles Rabipur C INTRAMUSCULAR 1 DOSE. 1 DAY<br />
Date:06/15/06ISR Number: 5026560-8Report Type:Expedited (15-DaCompany Report #CH-ROCHE-308974 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pulmonary Toxicity <strong>Mefloquine</strong><br />
Initial or Prolonged Toxicity To Various Hydrochloride PS Roche UNKNOWN<br />
Agents<br />
Diffuse Alveolar Damage<br />
Date:06/16/06ISR Number: 5027852-9Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398212 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Lariam PS Roche ORAL TAKEN ON 22<br />
Initial or Prolonged Vertigo FEB, 01 MAR
Disability Balance Disorder AND 08 MAR<br />
Nausea 2005. 15 DAY<br />
Tinnitus<br />
Diarrhoea<br />
Nystagmus<br />
Hearing Impaired<br />
03-Apr-2012 09:37 AM Page: 714
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/16/06ISR Number: 5027885-2Report Type:Expedited (15-DaCompany Report #SE-ROCHE-449547 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Maternal Exposure During Lariam PS Roche ORAL<br />
Initial or Prolonged Pregnancy<br />
Abortion<br />
Umbilical Malformation<br />
Date:06/20/06ISR Number: 5030269-4Report Type:Expedited (15-DaCompany Report #CH-ROCHE-428290 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypoxia <strong>Mefloquine</strong><br />
Initial or Prolonged Haemoglobin Decreased Hydrochloride PS Roche UNKNOWN RECEIVED A<br />
Diarrhoea<br />
TOTAL DOSE OF<br />
Lung Infiltration<br />
1500MG OF<br />
Pulmonary Toxicity<br />
MEFLOQUINE<br />
Blood Pressure Decreased<br />
OVER 24 HOURS<br />
General Physical Health Paracetamol C<br />
Deterioration Halofantrine C FOR 48 HOURS<br />
Diffuse Alveolar Damage<br />
Date:06/20/06ISR Number: 5032579-3Report Type:Direct Company Report #CTU 279190 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Simple Partial Seizures <strong>Mefloquine</strong> (Lariam) PS Sandoz ORAL 250 MG ONE<br />
Muscle Spasms<br />
TIME DOSE PO<br />
Sleep Disorder<br />
Paraesthesia<br />
Sensory Loss<br />
Depression<br />
Abnormal Sensation In Eye<br />
Dizziness<br />
Auricular Swelling<br />
Hypoaesthesia<br />
Disorientation<br />
Paranoia<br />
Anxiety<br />
Asthenia<br />
Fatigue<br />
Menstrual Disorder<br />
Oropharyngeal Pain<br />
Nervous System Disorder<br />
Mood Swings<br />
Confusional State<br />
Intracranial Pressure<br />
Increased<br />
Feeling Cold<br />
Anhidrosis<br />
Blindness
Date:06/20/06ISR Number: 5032742-1Report Type:Direct Company Report #CTU 279122 Age:25 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
03-Apr-2012 09:37 AM Page: 715
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent PT Report Source Product Role Manufacturer Route Dose Duration<br />
Impairment/Damage Dysarthria Lariam Roche PS Roche 250 MG WEEKLY<br />
Hallucination<br />
Confusional State<br />
Toxicity To Various<br />
Agents<br />
Disturbance In Attention<br />
Fatigue<br />
Insomnia<br />
Dizziness<br />
Suicidal Ideation<br />
Date:06/22/06ISR Number: 5033028-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Abnormal Behaviour<br />
Disturbance In Attention<br />
Productive Cough<br />
Ill-Defined Disorder<br />
Tremor<br />
Oxygen Saturation<br />
Decreased<br />
Hypoaesthesia<br />
Mucous Stools<br />
Rectal Haemorrhage<br />
Balance Disorder<br />
Bronchial Hyperreactivity<br />
Headache<br />
Sleep Apnoea Syndrome<br />
Musculoskeletal Pain<br />
Hyperhidrosis<br />
Dysthymic Disorder<br />
Rheumatoid Factor<br />
Increased<br />
Rash Papular<br />
Eosinophilia<br />
Paranoia<br />
Hypervigilance<br />
Breath Odour<br />
Nausea<br />
Aggression<br />
Psychiatric Symptom<br />
Decreased Activity<br />
Convulsion<br />
Dissociative Disorder<br />
Hypoacusis<br />
Dyspepsia<br />
Insomnia<br />
Depression
Selective Iga<br />
Immunodeficiency<br />
Fatigue<br />
Asthenia<br />
Chest Discomfort<br />
Chest Pain<br />
Psychotic Disorder<br />
Self-Injurious Ideation<br />
03-Apr-2012 09:37 AM Page: 716
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Meniscus Lesion<br />
Thinking Abnormal<br />
Homicidal Ideation<br />
Mitral Valve Incompetence<br />
Dehydration<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Contusion<br />
Antinuclear Antibody<br />
Positive<br />
Derealisation<br />
Product Contamination<br />
Palpitations<br />
Somnolence<br />
Dizziness<br />
Tinnitus<br />
Emotional Disorder<br />
Eructation<br />
Anxiety<br />
Memory Impairment<br />
Hallucination, Visual<br />
Hypertension<br />
Fall<br />
Asthma<br />
Weight Decreased<br />
Nystagmus<br />
Eye Movement Disorder<br />
Ligament Sprain<br />
Coordination Abnormal<br />
Vestibular Disorder<br />
Weight Increased<br />
Snoring<br />
Coronary Artery Disease<br />
Non-Cardiac Chest Pain<br />
Angina Pectoris<br />
Visual Impairment<br />
Dyspnoea<br />
Cranial Nerve Disorder<br />
Joint Dislocation<br />
Cognitive Disorder<br />
Craniocerebral Injury<br />
Humerus Fracture<br />
Pyrexia<br />
Product Quality Issue<br />
Abdominal Pain Upper<br />
Photophobia<br />
Agitation<br />
Mood Swings<br />
Generalised Anxiety<br />
Disorder<br />
Post-Traumatic Stress<br />
Disorder
Bundle Branch Block Right<br />
Exostosis<br />
Rhinitis Allergic<br />
Nocturia<br />
Heart Rate Decreased<br />
Rash<br />
Vertigo<br />
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 717
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Panic Disorder<br />
Major Depression<br />
Toxicity To Various Report Source Product Role Manufacturer Route Dose Duration<br />
Agents Lariam PS Roche ORAL<br />
Mitral Valve Primaquine C<br />
Calcification Cipro C<br />
Myalgia<br />
Ventricular Extrasystoles<br />
Eye Disorder<br />
Merycism<br />
Upper Respiratory Tract<br />
Infection<br />
Abdominal Distension<br />
Gastrointestinal Motility<br />
Disorder<br />
Date:06/30/06ISR Number: 5040526-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-452577 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam PS Roche UNKNOWN<br />
Suicidal Ideation<br />
Date:07/10/06ISR Number: 5046420-6Report Type:Expedited (15-DaCompany Report #ZM-ROCHE-453365 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Blood Creatinine <strong>Mefloquine</strong><br />
Hospitalization - Increased Hydrochloride PS Roche ORAL 1 DAY<br />
Initial or Prolonged Nephrotic Syndrome<br />
Date:07/10/06ISR Number: 5048049-2Report Type:Direct Company Report #CTU 280300 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Generalised Anxiety Larium Don'T<br />
Disorder<br />
Know<br />
Malaise Hoffman-Laroche PS Hoffman-Laroche 1 PILL<br />
Emotional Disorder<br />
WEEKLY<br />
Palpitations<br />
Feeling Abnormal<br />
Heart Rate Increased<br />
Date:07/17/06ISR Number: 5052750-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-450157 Age:43 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myocardial Infarction Lariam PS Roche ORAL REPORTED AS<br />
Initial or Prolonged Supraventricular 250 MG AS
Extrasystoles TOTAL. 1 DAY<br />
Sinus Tachycardia Mediator 150 SS ORAL 726 DAY<br />
Pericarditis Havrix C INTRAMUSCULAR FORM:<br />
Ventricular Extrasystoles<br />
INJECTABLE<br />
SOLUTION. 1 DAY<br />
Rabipur C INTRAMUSCULAR 1 DAY<br />
03-Apr-2012 09:37 AM Page: 718
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/18/06ISR Number: 5053702-0Report Type:Expedited (15-DaCompany Report #SE-ROCHE-454148 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Lariam PS Roche ORAL 8 DAY<br />
Angioedema<br />
Anxiety<br />
Date:07/18/06ISR Number: 5054159-6Report Type:Expedited (15-DaCompany Report #CA-ROCHE-454857 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nodular Vasculitis Lariam PS Roche ORAL<br />
Synthroid<br />
C<br />
Date:07/24/06ISR Number: 5059676-0Report Type:Expedited (15-DaCompany Report #SE-ROCHE-454148 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS Roche ORAL 8 DAY<br />
Angioedema<br />
Dyspnoea<br />
Sensation Of Foreign Body<br />
Date:07/31/06ISR Number: 5065746-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-451414 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Asthenia Lariam PS Roche ORAL<br />
Nausea Lariam SS Roche ORAL<br />
Oropharyngeal Pain Lariam SS Roche ORAL<br />
Mood Swings Artesunate C ORAL<br />
Anxiety Chloroquine C ORAL<br />
Abdominal Discomfort Doxycycline C 3 DAY<br />
Depression<br />
Confusional State<br />
Thinking Abnormal<br />
Abdominal Pain<br />
Diarrhoea<br />
Musculoskeletal Pain<br />
Tremor<br />
Feeling Abnormal<br />
Amnesia<br />
Disease Recurrence<br />
Fatigue<br />
Myalgia<br />
Bedridden<br />
Date:08/11/06ISR Number: 5078369-7Report Type:Expedited (15-DaCompany Report #FR-GLAXOSMITHKLINE-B0426071A Age:28 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Ineffective Malarone PS Glaxosmithkline ORAL 2TAB Three<br />
Initial or Prolonged Hallucination times per day 3 DAY<br />
Disability Insomnia Lariam SS Other ORAL 1250MG per<br />
Major Depression day 1 DAY<br />
Rohypnol SS Other ORAL 5MG Per day 3 WK<br />
Ciflox C Other UNKNOWN 8 DAY<br />
03-Apr-2012 09:37 AM Page: 719
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/14/06ISR Number: 5079426-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-452577 Age:62 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Psychiatric Symptom Lariam PS Roche ORAL 29 DAY<br />
Anxiety Teoptic C OPHTHALMIC<br />
Suicidal Ideation<br />
Depression<br />
Panic Attack<br />
Date:08/17/06ISR Number: 5082526-3Report Type:Expedited (15-DaCompany Report #SE-ROCHE-454148 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Lariam PS Roche ORAL 8 DAY<br />
Angioedema<br />
Sensation Of Foreign Body<br />
Anxiety<br />
Date:08/22/06ISR Number: 5087940-8Report Type:Direct Company Report #CTU 283391 Age:70 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rash <strong>Mefloquine</strong> PS ORAL PO<br />
Initial or Prolonged Duodenitis Chloroquine SS ORAL PO<br />
Decreased Appetite Pravachol C<br />
Leukocytoclastic Loratidine C<br />
Vasculitis Plavix C<br />
Infection Lotrel C<br />
Nonspecific Reaction Toprol C<br />
Asthenia Nitroglycerin Patch C<br />
Gastritis Aspirin C<br />
Date:08/23/06ISR Number: 5088480-2Report Type:Direct Company Report #CTU 283589 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Lariam (<strong>Mefloquine</strong>) PS 250 MG 1<br />
Vertigo<br />
TABLET WEEKLY<br />
Sleep Disorder<br />
Abnormal Dreams<br />
Suicidal Ideation<br />
Insomnia<br />
Paranoia<br />
Anger<br />
Affect Lability<br />
Panic Attack<br />
Tinnitus<br />
Anxiety<br />
Depression<br />
Headache
Dizziness<br />
03-Apr-2012 09:37 AM Page: 720
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/25/06ISR Number: 5088864-2Report Type:Expedited (15-DaCompany Report #BE-ROCHE-460045 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pneumonitis <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche UNKNOWN<br />
<strong>Mefloquine</strong><br />
Hydrochloride SS Roche UNKNOWN<br />
Bisoprolol Fumarate C REPORTED AS<br />
CHRONIC<br />
MEDICAITON.<br />
Ciprofibratum C REPORTED AS<br />
CHRONIC<br />
MEDICAITON.<br />
Aspirin C REPORTED AS:<br />
CHRONIC<br />
MEDICATION -<br />
LOW DOSE<br />
ASPIRIN.<br />
Date:08/28/06ISR Number: 5093449-8Report Type:Expedited (15-DaCompany Report #2006-BP-09661RO Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disinhibition Foreign <strong>Mefloquine</strong><br />
Initial or Prolonged Hallucination, Visual Literature (<strong>Mefloquine</strong>) PS 250 MG PER<br />
Other Serious Delusion Health WEEK (250<br />
Delusion Of Grandeur Professional MG), PO 2 WK<br />
Insomnia<br />
Persecutory Delusion<br />
Delusion Of Reference<br />
Mania<br />
Psychotic Disorder<br />
Date:08/31/06ISR Number: 5094068-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-459650 Age:39 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Parkinson'S Disease Lariam PS Roche ORAL<br />
Date:09/05/06ISR Number: 5096297-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-461700 Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Lariam PS Roche ORAL<br />
Initial or Prolonged Anxiety<br />
Confusional State<br />
Psychotic Disorder<br />
Insomnia<br />
Abnormal Behaviour<br />
Liver Disorder
Date:09/06/06ISR Number: 5097232-9Report Type:Expedited (15-DaCompany Report #SE-ROCHE-461385 Age:11 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Iron Deficiency Lariam PS Roche ORAL DOSAGE<br />
Leukopenia<br />
REGIMEN<br />
03-Apr-2012 09:37 AM Page: 721
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
REPORTED AS<br />
3/4 TABLET.<br />
Date:09/07/06ISR Number: 5098825-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-461020 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Thrombosis Lariam PS Roche ORAL 24 DAY<br />
Initial or Prolonged<br />
Date:09/08/06ISR Number: 5099741-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-461579 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delusion <strong>Mefloquine</strong><br />
Initial or Prolonged Hypomania Hydrochloride PS Roche ORAL 57 DAY<br />
Insomnia Salbutamol C<br />
Agitation Betnovate C TOPICAL<br />
Tearfulness<br />
Delusional Disorder,<br />
Persecutory Type<br />
Date:09/08/06ISR Number: 5099758-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-461827 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Acute Lariam PS Roche ORAL 8 DAY<br />
Initial or Prolonged Ecchymosis Ciprofloxacine SS ORAL 15 DAY<br />
Headache Flagyl SS ORAL 15 DAY<br />
Tendon Disorder Paracetamol C<br />
Urinary Tract Infection Polygynax C<br />
Vaginal Infection Solubacter C<br />
Vertigo<br />
Rash<br />
Date:09/08/06ISR Number: 5099759-2Report Type:Expedited (15-DaCompany Report #GB-ROCHE-461998 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Renal Dysplasia Lariam PS Roche TRANSPLACENTAL<br />
Pregnancy Alcohol C<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:09/11/06ISR Number: 5104007-0Report Type:Expedited (15-DaCompany Report #GXKR2006BE05415 Age:60 YR Gender:Female I/FU:I<br />
Outcome<br />
Hospitalization -<br />
PT<br />
Respiratory Distress
Initial or Prolonged<br />
Interstitial Lung Disease<br />
Leukocytosis<br />
Hypoxia<br />
Cyanosis<br />
Myalgia<br />
Headache<br />
Rales<br />
Blood Lactate<br />
03-Apr-2012 09:37 AM Page: 722
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Dehydrogenase Increased<br />
Tachycardia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Foreign<br />
<strong>Mefloquine</strong><br />
Literature (Ngx)(<strong>Mefloquine</strong>) PS 250 MG, QW<br />
Health Bisoprolol Fumarate C<br />
Professional Ciprofibrate C<br />
Other<br />
Aspirine<br />
(Acetylsalicylic<br />
Acid)<br />
C<br />
Date:09/12/06ISR Number: 5102387-3Report Type:Expedited (15-DaCompany Report #FR-AVENTIS-200613012FR Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Acute Flagyl "Aventis" PS Aventis<br />
Initial or Prolonged Ecchymosis Pharmaceuticals Inc. ORAL<br />
Rash Papular Lariam SS ORAL<br />
Pruritus Ciprofloxacine SS ORAL<br />
Vertigo Morphine C<br />
Chills Motilium C<br />
Tendon Disorder Di-Antalvic C<br />
Abdominal Pain Lower Spasfon C<br />
Urinary Tract Infection Polygynax C<br />
Vaginal Infection Solubacter C<br />
Headache<br />
Date:09/15/06ISR Number: 5105797-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-462646 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Exposure During Lariam PS Roche ORAL<br />
Pregnancy<br />
Abortion Spontaneous<br />
Date:09/19/06ISR Number: 5108165-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-443462 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Lariam PS Roche UNKNOWN 4 TABLETS IN<br />
Initial or Prolonged Vomiting TOTAL.<br />
Fatigue<br />
Disturbance In Attention<br />
Vertigo<br />
Visual Impairment<br />
Date:09/20/06ISR Number: 5109127-2Report Type:Expedited (15-DaCompany Report #ZM-ROCHE-453365 Age:28 YR Gender:Male I/FU:I
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Blood Creatinine<br />
Increased<br />
Nephrotic Syndrome<br />
Oedema Peripheral<br />
Blood Albumin Decreased<br />
Blood Sodium Decreased<br />
Abdominal Distension<br />
03-Apr-2012 09:37 AM Page: 723
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Blood Potassium Decreased<br />
Body Temperature<br />
Decreased Report Source Product Role Manufacturer Route Dose Duration<br />
Respiratory Rate<br />
<strong>Mefloquine</strong><br />
Increased Hydrochloride PS Roche ORAL 1 DAY<br />
Date:09/20/06ISR Number: 5109459-8Report Type:Expedited (15-DaCompany Report #BE-ROXANE LABORATORIES, INC-2006-BP-10708RO Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Leukocytosis Inc. ORAL 250 mg x 1<br />
Headache<br />
dose<br />
Hypoxia Bisoprolol Fumarate C<br />
Pneumonitis Ciprofibratum C<br />
Dyspnoea Aspirin C<br />
C-Reactive Protein<br />
Increased<br />
Cough<br />
Chills<br />
Tachycardia<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Cyanosis<br />
Myalgia<br />
Date:09/20/06ISR Number: 5109460-4Report Type:Expedited (15-DaCompany Report #FR-ROXANE LABORATORIES, INC-2006-BP-10993RO Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Contusion <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Malaria Inc. 48 HR<br />
Mental Disorder<br />
Proguanil-Chloroquin<br />
Wound e SS 3 MON<br />
Drug Ineffective<br />
Completed Suicide<br />
Haemothorax<br />
Date:09/20/06ISR Number: 5112513-8Report Type:Direct Company Report #CTU 285351 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Incontinence Lariam PS<br />
Grand Mal Convulsion<br />
Tongue Biting<br />
Date:09/28/06ISR Number: 5118014-5Report Type:Direct Company Report #CTU 285895 Age:28 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder <strong>Mefloquine</strong> Roche PS Roche ORAL ONE TABLET<br />
Initial or Prolonged<br />
ONCE A WEEK<br />
PO<br />
03-Apr-2012 09:37 AM Page: 724
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/29/06ISR Number: 5117526-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-447408 Age:44 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vomiting Lariam PS Roche ORAL<br />
Coordination Abnormal<br />
Anaesthetic<br />
Asthenia Medication C REPORTED AS<br />
Tachycardia<br />
ANAESTHESIA,<br />
Dizziness<br />
LOCAL.<br />
Nausea<br />
Hypertension<br />
Hot Flush<br />
Date:10/09/06ISR Number: 5122975-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-465214 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Myalgia Lariam PS Roche ORAL<br />
Monoplegia<br />
Pain<br />
Date:10/10/06ISR Number: 5123787-1Report Type:Expedited (15-DaCompany Report #FR-AVENTIS-200613012FR Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Flagyl "Aventis" PS Aventis<br />
Initial or Prolonged Rash Papular Pharmaceuticals Inc. ORAL<br />
Headache Lariam SS ORAL<br />
Leukocytosis Ciprofloxacine SS ORAL<br />
Ecchymosis Morphine C<br />
Chills Motilium C<br />
Urinary Tract Infection Diantalvic C<br />
Renal Failure Acute Spasfon C<br />
Tendon Disorder Polygynax C<br />
Gamma-Glutamyltransferase Solubacter C<br />
Increased<br />
Muscle Spasms<br />
Pruritus<br />
Date:10/16/06ISR Number: 5127772-5Report Type:Expedited (15-DaCompany Report #ZM-ROCHE-430119 Age:28 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Nephrotic Syndrome <strong>Mefloquine</strong><br />
Hospitalization - Blood Creatinine Hydrochloride PS Roche ORAL 3 DAY<br />
Initial or Prolonged Increased<br />
Date:10/16/06ISR Number: 5127780-4Report Type:Expedited (15-DaCompany Report #ZM-ROCHE-453365 Age:28 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Blood Creatinine <strong>Mefloquine</strong><br />
Hospitalization - Increased Hydrochloride PS Roche ORAL 1 DAY<br />
Initial or Prolonged Nephrotic Syndrome<br />
Respiratory Rate<br />
Increased<br />
03-Apr-2012 09:37 AM Page: 725
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/17/06ISR Number: 5128708-3Report Type:Expedited (15-DaCompany Report #FR-AVENTIS-200613012FR Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Constipation Flagyl PS Aventis<br />
Initial or Prolonged Chills Pharmaceuticals Inc. ORAL<br />
Gamma-Glutamyltransferase Lariam SS ORAL<br />
Increased Ciprofloxacine SS ORAL<br />
Muscle Spasms Morphine C<br />
Rash Papular Motilium C<br />
Vertigo Diantalvic C<br />
Headache Spasfon C<br />
Pain Polygynax C<br />
Renal Failure Acute Solubacter C<br />
Pruritus<br />
Abdominal Pain<br />
Tendon Disorder<br />
Ecchymosis<br />
Leukocytosis<br />
Date:10/18/06ISR Number: 5129717-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-466784 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tongue Ulceration Lariam PS Roche ORAL<br />
Pharyngeal Ulceration<br />
Oropharyngeal Pain<br />
Inflammation<br />
Oral Administration<br />
Complication<br />
Date:10/23/06ISR Number: 5132543-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-461827 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Prurigo Lariam PS Roche ORAL FIRST TABLET<br />
Initial or Prolonged Urinary Tract Infection ON 17 JUL<br />
Rash Papular<br />
2006, SECOND<br />
Constipation TABLET ON 24<br />
Rash Erythematous JUL 2006. 8 DAY<br />
Renal Failure Acute Ciprofloxacine SS ORAL 15 DAY<br />
Vertigo Flagyl SS ORAL 15 DAY<br />
Abdominal Pain Paracetamol C<br />
Headache Polygynax C<br />
Ecchymosis Solubacter C<br />
Date:10/24/06ISR Number: 5133707-1Report Type:Expedited (15-DaCompany Report #IT-ROCHE-467032 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatic Failure <strong>Mefloquine</strong>
Initial or Prolonged Hepatomegaly Hydrochloride PS Roche UNKNOWN TREATMENT<br />
Hepatic Steatosis<br />
INTERRUPTED<br />
AFTER THE 5TH<br />
WEEK<br />
03-Apr-2012 09:37 AM Page: 726
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/30/06ISR Number: 5138679-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-459650 Age:39 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Parkinsonism Lariam PS Roche ORAL<br />
Date:10/30/06ISR Number: 5138717-6Report Type:Expedited (15-DaCompany Report #NL-ROCHE-467834 Age:59 YR Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypertensive Crisis Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Metoprolol Tartrate C ORAL<br />
Simvastatin C ORAL<br />
Coversyl Plus C ORAL STRENGTH<br />
REPORTED AS<br />
4/1,25 MG.<br />
Date:10/30/06ISR Number: 5138723-1Report Type:Expedited (15-DaCompany Report #RU-ROCHE-467843 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gamma-Glutamyltransferase Lariam PS Roche ORAL ONCE ONE<br />
Increased TABLET OF 250<br />
MG. 1 DAY<br />
Date:11/06/06ISR Number: 5144153-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457439 Age:3 MON Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Haematocrit Decreased<br />
Respiratory Disorder<br />
Vomiting<br />
Salivary Hypersecretion<br />
Haemoglobin Decreased<br />
Date:11/14/06ISR Number: 5158996-9Report Type:Expedited (15-DaCompany Report #D0045516A Age:43 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Drug Ineffective Foreign Malarone PS ORAL ORAL<br />
Hospitalization - Depressed Level Of Literature Sodium Artesunate<br />
Initial or Prolonged Consciousness Health (Sodium Artesunate) SS ORAL PER DAY ORAL<br />
Cerebral Malaria<br />
Professional<br />
Psychiatric Symptom<br />
Other<br />
Myoclonic Epilepsy<br />
Date:11/22/06ISR Number: 5156058-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-447408 Age:43 YR Gender:Female I/FU:F
Outcome<br />
Other Serious<br />
PT<br />
Tachycardia<br />
Asthenia<br />
Dizziness<br />
Hypertension<br />
Hot Flush<br />
Vomiting<br />
03-Apr-2012 09:37 AM Page: 727
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Coordination Abnormal<br />
Nausea<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Femoston C TAKEN FOR<br />
YEARS.<br />
Jodetten<br />
C<br />
Anaesthetic<br />
Medication C REPORTED AS<br />
ANAESTHESIA,<br />
LOCAL.<br />
Date:11/22/06ISR Number: 5157901-9Report Type:Direct Company Report #CTU 289911 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam 250 Mg<br />
Panic Attack (Weekly) Roche PS Roche ORAL 250 MG ONCE<br />
Insomnia<br />
WEEKLY ORALLY<br />
Depression<br />
Date:11/24/06ISR Number: 5156951-6Report Type:Expedited (15-DaCompany Report #NL-ROCHE-467834 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypertensive Crisis Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Malignant Hypertension Cosopt C OTHER ROUTE<br />
REPORTED AS<br />
OCULAR AND<br />
FORM REPORTED<br />
AS EYEDROPS.<br />
Simvastatin C ORAL<br />
Coversyl Plus C ORAL STRENGTH<br />
REPORTED AS<br />
4/1,25 MG.<br />
Selokeen C ORAL<br />
Date:11/27/06ISR Number: 5165169-2Report Type:Expedited (15-DaCompany Report #GXKR2006SG07387 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Contusion Foreign <strong>Mefloquine</strong> (Ngx)<br />
Rectal Haemorrhage Literature (<strong>Mefloquine</strong>) Unknown PS QW<br />
Platelet Count Increased Health<br />
Professional<br />
Other<br />
Date:11/28/06ISR Number: 5159052-6Report Type:Expedited (15-DaCompany Report #SG-ROXANE LABORATORIES, INC-2006-BP-13605RO Age:46 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Platelet Count Increased <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Contusion Inc. once weekly x<br />
Rectal Haemorrhage 3 weeks 3 WK<br />
03-Apr-2012 09:37 AM Page: 728
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/01/06ISR Number: 5162883-XReport Type:Expedited (15-DaCompany Report #113217 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatomegaly Lariam Tablets PS Roche ORAL<br />
Initial or Prolonged Toxicity To Various<br />
Agents<br />
Metabolic Disorder<br />
Nausea<br />
Nervousness<br />
Diarrhoea<br />
Abdominal Distension<br />
Depression<br />
Vomiting<br />
Alcohol Use<br />
Weight Decreased<br />
Dehydration<br />
Hepatic Steatosis<br />
Decreased Appetite<br />
Gastroenteritis<br />
Gravitational Oedema<br />
Hepatic Failure<br />
Date:12/04/06ISR Number: 5163420-6Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457439 Age:3 MON Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Haematocrit Decreased Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Platelet Count Increased<br />
Fatigue<br />
Convulsion<br />
Respiratory Disorder<br />
Haemoglobin Decreased<br />
Vomiting<br />
Dyspnoea<br />
Salivary Hypersecretion<br />
Date:12/08/06ISR Number: 5168390-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-472987 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Torticollis Lariam PS Roche UNKNOWN<br />
Laryngeal Disorder<br />
Date:12/11/06ISR Number: 5175373-5Report Type:Direct Company Report #CTU 291283 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Restlessness Lariam Roche PS Roche THREE PILLS<br />
Required Tinnitus OF 250 MG ONE<br />
Intervention to Fatigue A WEEK 3 WK
Prevent Permanent<br />
Impairment/Damage<br />
Mood Swings<br />
03-Apr-2012 09:37 AM Page: 729
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/14/06ISR Number: 5174215-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-474014 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vasculitis Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Human Ehrlichiosis<br />
Creatinine Renal<br />
Clearance Increased<br />
Nephritis<br />
Date:12/18/06ISR Number: 5177195-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-474927 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Aplasia Lariam PS Roche ORAL 72 DAY<br />
Maternal Exposure During<br />
Pregnancy<br />
Abortion Induced<br />
Foetal Malformation<br />
Date:12/19/06ISR Number: 5179370-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-98097 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Depression Lariam Tablets PS Roche ORAL<br />
Femur Fracture<br />
Completed Suicide<br />
Skull Fracture<br />
Abnormal Behaviour<br />
Fall<br />
Multiple Injuries<br />
Facial Bones Fracture<br />
Aortic Rupture<br />
Date:12/19/06ISR Number: 5179424-3Report Type:Expedited (15-DaCompany Report #CA-ROCHE-940804124001 Age:23 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hallucination Lariam PS Roche ORAL<br />
Life-Threatening Completed Suicide Alcohol C ORAL<br />
Date:12/19/06ISR Number: 5191295-8Report Type:Direct Company Report #CTU 291816 Age:42 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Hallucination<br />
Insomnia<br />
Rapid Eye Movements Sleep<br />
Abnormal<br />
Hyperhidrosis
Crying<br />
Nausea<br />
Nightmare<br />
Emotional Disorder<br />
Hypoaesthesia<br />
Myocardial Infarction<br />
Decreased Appetite<br />
Paranoia<br />
03-Apr-2012 09:37 AM Page: 730
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paraesthesia<br />
Dizziness<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong> 250 Mg<br />
Sandoz PS Sandoz ORAL 1 TABLET<br />
WEEKLY PO<br />
Date:12/21/06ISR Number: 5188158-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-447408 Age:43 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypertension Lariam PS Roche ORAL<br />
Hot Flush Femoston C ORAL DRUG NAME<br />
Coordination Abnormal<br />
REPORTED AS<br />
Nausea<br />
FEMOSTON<br />
Vomiting<br />
2/10. TAKEN<br />
Tachycardia<br />
FOR YEARS.<br />
Asthenia Jodetten C ORAL DRUG NAME<br />
Dizziness<br />
REPORTED AS<br />
JODETTEN<br />
DEPOT. DOSAGE<br />
REGIMEN<br />
Anaesthetic<br />
Medication C REPORTED AS<br />
ANAESTHESIA,<br />
LOCAL.<br />
Date:01/02/07ISR Number: 5195733-6Report Type:Expedited (15-DaCompany Report #FR-ROCHE-472987 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Laryngeal Disorder Lariam PS Roche UNKNOWN<br />
Dystonia<br />
Torticollis<br />
Speech Disorder<br />
Date:01/03/07ISR Number: 5199854-3Report Type:Direct Company Report #CTU 292560 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Asthenia Larium PS<br />
Intervention to<br />
Depression<br />
Prevent Permanent Hypothyroidism<br />
Impairment/Damage Lethargy<br />
Fatigue<br />
Mental Disorder<br />
Date:01/09/07ISR Number: 5202168-6Report Type:Expedited (15-DaCompany Report #IT-ROCHE-476898 Age:44 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Angioedema Lariam PS Roche ORAL 52 DAY<br />
Initial or Prolonged Urticaria Amoxicilline SS ORAL 7 DAY<br />
03-Apr-2012 09:37 AM Page: 731
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/09/07ISR Number: 5203930-6Report Type:Direct Company Report #CTU 292996 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Larium PS ORAL 1 TABLET ONCE<br />
Initial or Prolonged Paranoia DAILY ORALLY 8 DAY<br />
Other Serious<br />
Delusion<br />
Convulsion<br />
Loss Of Proprioception<br />
Heart Rate Increased<br />
Panic Attack<br />
Psychotic Disorder<br />
Date:01/11/07ISR Number: 5208350-6Report Type:Direct Company Report #CTU 293144 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Balance Disorder Lariam 250mg Roche PS Roche ORAL 250 MG WEEKLY<br />
Hospitalization - Anxiety PO 3 WK<br />
Initial or Prolonged Fatigue Travel C<br />
Other Serious<br />
Brain Injury<br />
Required<br />
Hallucination<br />
Intervention to<br />
Panic Attack<br />
Prevent Permanent<br />
Impairment/Damage<br />
Insomnia<br />
Impaired Work Ability<br />
Aphasia<br />
Suicidal Ideation<br />
Psychiatric Symptom<br />
Feeling Of Body<br />
Temperature Change<br />
Cold Sweat<br />
Psychotic Disorder<br />
Vestibular Neuronitis<br />
Dizziness<br />
Chills<br />
Headache<br />
Disturbance In Attention<br />
Depression<br />
Neurotoxicity<br />
Convulsion<br />
Date:01/17/07ISR Number: 5210481-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-477655 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vestibular Disorder Lariam PS Roche UNKNOWN THE PATIENT<br />
Initial or Prolonged Convulsion REPORTED<br />
Psychotic Disorder<br />
TAKING THREE<br />
Nightmare<br />
LARIAM<br />
Hallucination<br />
"PILLS" IN<br />
Suicidal Ideation<br />
Thinking Abnormal
Aggression<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 732
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/17/07ISR Number: 5211569-1Report Type:Direct Company Report #CTU 293643 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Night Blindness <strong>Mefloquine</strong> 250 Mg<br />
Maculopathy Roche PS Roche ORAL 250 MG PO QWK<br />
Visual Acuity Reduced<br />
PO<br />
Colour Blindness Erythromycin C<br />
Scotoma<br />
Date:01/19/07ISR Number: 5213113-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-478024 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rectal Haemorrhage Lariam PS Roche ORAL<br />
Initial or Prolonged Diarrhoea Lariam SS Roche ORAL<br />
Anisocytosis Tenstaten C ORAL<br />
Poikilocytosis<br />
Microcytic Anaemia<br />
Blood Potassium Decreased<br />
Diarrhoea Infectious<br />
Date:01/25/07ISR Number: 5219034-2Report Type:Expedited (15-DaCompany Report #NO-ROCHE-398212 Age:38 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Lariam PS Roche ORAL TAKEN ON 22<br />
Initial or Prolonged Tinnitus FEB, 01 MAR<br />
Disability Hearing Impaired AND 08 MAR<br />
Vertigo 2005. 15 DAY<br />
Balance Disorder<br />
Nausea<br />
Dizziness<br />
Nystagmus<br />
Date:01/26/07ISR Number: 5221301-3Report Type:Expedited (15-DaCompany Report #ZA-ROCHE-477672 Age:34 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dry Eye Lariam PS Roche ORAL<br />
Hospitalization - Corneal Scar<br />
Initial or Prolonged Stevens-Johnson Syndrome<br />
Disability<br />
Date:01/29/07ISR Number: 5224770-8Report Type:Direct Company Report #CTU 294509 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Wound Secretion Larium PS ORAL 1 TABLET ONCE<br />
Initial or Prolonged Rash DAILY ORALLY 9 DAY
Other Serious<br />
Menstruation Irregular<br />
03-Apr-2012 09:37 AM Page: 733
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/30/07ISR Number: 5223920-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Nausea<br />
Depression<br />
Paraesthesia<br />
Disorientation<br />
Dyspnoea<br />
Irritable Bowel Syndrome<br />
Lactose Intolerance<br />
Lipoma<br />
Haemorrhoids<br />
Vitreous Floaters<br />
Bronchial Hyperreactivity<br />
Anxiety<br />
Panic Attack<br />
Rash<br />
Hypersensitivity<br />
Generalised Anxiety<br />
Disorder<br />
Musculoskeletal Stiffness<br />
Amnesia<br />
Eye Movement Disorder<br />
Treatment Noncompliance<br />
Food Allergy<br />
Melanocytic Naevus<br />
Dizziness<br />
Motion Sickness<br />
Tinnitus<br />
Self Esteem Decreased<br />
Disturbance In Attention<br />
Headache<br />
Neck Pain<br />
Abdominal Pain Lower<br />
Vertigo<br />
Sleep Disorder Due To<br />
General Medical<br />
Condition, Hypersomnia<br />
Type<br />
Heart Rate Increased<br />
Abdominal Pain<br />
Diarrhoea<br />
Photophobia<br />
Acrophobia<br />
Throat Irritation<br />
Rhinitis Allergic<br />
Mental Disorder<br />
Adjustment Disorder With<br />
Anxiety<br />
Mood Disorder Due To A<br />
General Medical Condition<br />
Insomnia<br />
Balance Disorder
Abdominal Distension<br />
Condition Aggravated<br />
Gilbert'S Syndrome<br />
Infection Parasitic<br />
Emotional Distress<br />
Suicidal Ideation<br />
Heart Rate Irregular<br />
Burning Sensation<br />
03-Apr-2012 09:37 AM Page: 734
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Injury<br />
Palpitations<br />
Sinus Bradycardia Report Source Product Role Manufacturer Route Dose Duration<br />
Vestibular Disorder Lariam PS Roche ORAL THE PATIENT<br />
Weight Decreased HAD TAKEN 4-5<br />
Phobia Of Flying<br />
TABLETS OF<br />
Phobia<br />
MEFLOQUINE IN<br />
Migraine TOTAL. 21 DAY<br />
Blood Bilirubin Increased Vaccine C OTHER 1 DAY<br />
Impetigo<br />
Abdominal Pain Upper<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Claustrophobia<br />
Sensory Integrative<br />
Dysfunction<br />
Fatigue<br />
Sleep Disorder<br />
Back Pain<br />
Nasal Congestion<br />
Hypoaesthesia<br />
Gait Disturbance<br />
Visual Impairment<br />
Date:01/30/07ISR Number: 5225876-XReport Type:Direct Company Report #CTU 294564 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Palpitations <strong>Mefloquine</strong> Hcl 250<br />
Panic Attack Mg PS ORAL 1 WEEKLY<br />
Fear<br />
PO<br />
Dyspnoea<br />
Date:01/30/07ISR Number: 5225886-2Report Type:Direct Company Report #CTU 294589 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Lariam-<strong>Mefloquine</strong><br />
Initial or Prolonged Vomiting Hcl- Sandoz Or Roche PS Sandoz Or Roche ORAL 250MG ONCE A<br />
Other Serious Nausea WEEK PO (2<br />
Anxiety<br />
DOSES ONLY)<br />
Feeling Abnormal<br />
Hypophagia<br />
Heart Rate Increased<br />
Malaise<br />
Insomnia<br />
Palpitations<br />
Chills
Date:02/16/07ISR Number: 5239696-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Vertigo<br />
Sleep Disorder<br />
Disturbance In Attention<br />
Cardiovascular Disorder<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 735
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Malaise<br />
Headache<br />
Coordination Abnormal Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Lariam PS Roche UNKNOWN DOSAGE<br />
Anxiety<br />
REGIMEN<br />
Oedema Peripheral<br />
REPORTED AS<br />
Formication<br />
FOUR TABS IN<br />
Asthenia<br />
TOTAL.<br />
Abnormal Dreams<br />
Paraesthesia<br />
Tinnitus<br />
Depression<br />
Tachycardia<br />
Feeling Abnormal<br />
Muscle Twitching<br />
Night Sweats<br />
Date:02/16/07ISR Number: 5239744-0Report Type:Expedited (15-DaCompany Report #ZA-ROCHE-477672 Age:34 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Dry Eye Lariam PS Roche ORAL<br />
Hospitalization - Corneal Scar<br />
Initial or Prolonged Stevens-Johnson Syndrome<br />
Disability<br />
Date:02/20/07ISR Number: 5243672-4Report Type:Direct Company Report #CTU 296345 Age:47 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Middle Insomnia <strong>Mefloquine</strong> Std ? PS ORAL STD 1/ WEEK<br />
Sensitivity Of Teeth<br />
PO<br />
Condition Aggravated<br />
Fatigue<br />
Date:02/21/07ISR Number: 5242741-2Report Type:Expedited (15-DaCompany Report #GB-ROCHE-482808 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Acute Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Lariam SS Roche UNKNOWN<br />
Lariam SS Roche UNKNOWN<br />
Lariam SS Roche UNKNOWN<br />
Date:02/28/07ISR Number: 5252267-8Report Type:Direct Company Report #CTU 296837 Age:23 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Attack Larium <strong>Mefloquine</strong>
Dizziness Roche PS Roche ORAL ONE PILL<br />
Paranoia<br />
-250MG- PER<br />
Abnormal Dreams WEEK PO 10 MON<br />
Hallucination<br />
Anxiety<br />
Heart Rate Increased<br />
Insomnia<br />
Motion Sickness<br />
03-Apr-2012 09:37 AM Page: 736
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/02/07ISR Number: 5254139-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-483909 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Lariam PS Roche ORAL 1 DAY<br />
Initial or Prolonged Delirium<br />
Aggression<br />
Date:03/02/07ISR Number: 5256250-8Report Type:Direct Company Report #CTU 297137 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Barr Laboratories,<br />
Initial or Prolonged Inc. ORAL 1 PILL 1/WEEK<br />
PO<br />
Date:03/06/07ISR Number: 5256605-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Initial or Prolonged Antisocial Behaviour Valium SS Roche INTRAVENOUS 1 DAY<br />
Disability Muscle Rigidity Prozac C ORAL<br />
Speech Disorder<br />
Agitation<br />
Memory Impairment<br />
Depression<br />
Gait Disturbance<br />
Blood Potassium Decreased<br />
Paranoia<br />
Convulsion<br />
Hostility<br />
Headache<br />
Hallucination<br />
Incontinence<br />
Confusional State<br />
Delusion<br />
Disorientation<br />
Fatigue<br />
Loss Of Libido<br />
Drug Ineffective<br />
Aggression<br />
Mania<br />
Pyrexia<br />
Date:03/06/07ISR Number: 5258866-1Report Type:Direct Company Report #CTU 297362 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Lariam PS<br />
Anxiety
Depression<br />
Nightmare<br />
03-Apr-2012 09:37 AM Page: 737
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/08/07ISR Number: 5258961-7Report Type:Expedited (15-DaCompany Report #SE-ROCHE-485061 Age:3 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Increased Upper Airway Lariam PS Roche ORAL 1 DAY<br />
Secretion<br />
Respiratory Arrest<br />
Erythema<br />
Anaphylactic Reaction<br />
Bronchospasm<br />
Date:03/09/07ISR Number: 5260861-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Lariam PS Roche UNKNOWN DOSAGE<br />
Initial or Prolonged Disturbance In Attention REGIMEN<br />
Abnormal Dreams<br />
REPORTED AS<br />
Headache<br />
FOUR TABS IN<br />
Tachycardia TOTAL. 22 DAY<br />
Feeling Abnormal<br />
Agitation<br />
Coordination Abnormal<br />
Fatigue<br />
Oedema Peripheral<br />
Cardiovascular Disorder<br />
Anxiety<br />
Depression<br />
Formication<br />
Asthenia<br />
Sleep Disorder<br />
Muscle Twitching<br />
Malaise<br />
Mental Disorder<br />
Paraesthesia<br />
Night Sweats<br />
Tinnitus<br />
Date:03/16/07ISR Number: 5267373-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-485061 Age:3 MON Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bronchospasm Lariam PS Roche ORAL 1 DAY<br />
Erythema<br />
Respiratory Arrest<br />
Anaphylactic Reaction<br />
Increased Upper Airway<br />
Secretion<br />
Respiratory Disorder
Date:03/19/07ISR Number: 5269055-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Febrile Infection<br />
Disturbance In Attention<br />
Fatigue<br />
Malaise<br />
Formication<br />
03-Apr-2012 09:37 AM Page: 738
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Vertigo<br />
Oedema Peripheral<br />
Dyspepsia Report Source Product Role Manufacturer Route Dose Duration<br />
Headache Lariam PS Roche UNKNOWN DOSAGE<br />
Depression<br />
REGIMEN<br />
Tachycardia<br />
REPORTED AS<br />
Asthenia<br />
FOUR TABS IN<br />
Muscle Twitching TOTAL. WEEKLY 22 DAY<br />
Agitation<br />
Paraesthesia<br />
Feeling Abnormal<br />
Abnormal Dreams<br />
Night Sweats<br />
Cardiovascular Disorder<br />
Anxiety<br />
Tinnitus<br />
Sleep Disorder<br />
Coordination Abnormal<br />
Mental Disorder<br />
Date:03/27/07ISR Number: 5277512-4Report Type:Expedited (15-DaCompany Report #SE-ROCHE-485061 Age:3 MON Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Bronchospasm Lariam PS Roche ORAL TWO DOSES,<br />
Respiratory Depression<br />
THE FIRST<br />
Increased Bronchial<br />
DOSE WAS<br />
Secretion SPITTED OUT. 1 DAY<br />
Increased Upper Airway<br />
Secretion<br />
Respiratory Arrest<br />
Respiratory Disorder<br />
Erythema<br />
Anaphylactic Reaction<br />
Date:03/27/07ISR Number: 5277551-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-488026 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Arrest Lariam PS Roche UNKNOWN<br />
Electrocardiogram Qt Sudafed C<br />
Prolonged<br />
Date:03/29/07ISR Number: 5280375-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-487625 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Homicide <strong>Mefloquine</strong><br />
Confusional State Hydrochloride PS Roche UNKNOWN
Date:03/30/07ISR Number: 5281849-2Report Type:Expedited (15-DaCompany Report #SE-ROCHE-488539 Age:2 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspnoea Lariam PS Roche ORAL REPORTED<br />
Initial or Prolonged Increased Bronchial DOSAGE<br />
Secretion<br />
REGIMEN:<br />
03-Apr-2012 09:37 AM Page: 739
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
62.MG1 MG 1. 1 DAY<br />
Date:04/03/07ISR Number: 5284325-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-439267 Age:14 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Encephalopathy<br />
Disability<br />
Panic Attack<br />
Cognitive Disorder<br />
Speech Disorder<br />
Anxiety Disorder<br />
Depression<br />
Date:04/03/07ISR Number: 5284692-3Report Type:Expedited (15-DaCompany Report #CA-ROCHE-489380 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Toxicity To Various Lariam PS Roche UNKNOWN 18 MON<br />
Agents<br />
Maculopathy<br />
Date:04/05/07ISR Number: 5288333-0Report Type:Expedited (15-DaCompany Report #SE-ROCHE-490181 Age:4 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Pulmonary Fibrosis Lariam PS Roche ORAL DOSAGE<br />
Hospitalization - Respiratory Failure REGIMEN<br />
Initial or Prolonged Cough REPORTED AS<br />
Inflammation 75MGW MG W. 7 WK<br />
Alveolitis Lariam SS Roche ORAL<br />
Infection<br />
Fatigue<br />
Rash<br />
Weight Decreased<br />
Pyrexia<br />
Depressed Level Of<br />
Consciousness<br />
Date:04/05/07ISR Number: 5288700-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-490463 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Road Traffic Accident Lariam PS Roche ORAL<br />
Hyperhidrosis Laroxyl SS Roche ORAL<br />
Malaise Laroxyl SS Roche ORAL<br />
Dry Mouth<br />
Asthenia
Date:04/05/07ISR Number: 5288710-8Report Type:Expedited (15-DaCompany Report #KR-ROCHE-490940 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Myocardial Infarction Lariam PS Roche UNKNOWN 4 WK<br />
03-Apr-2012 09:37 AM Page: 740
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/06/07ISR Number: 5289566-XReport Type:Expedited (15-DaCompany Report #SE-ROCHE-488539 Age:61 DY Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Increased Bronchial Lariam PS Roche ORAL REPORTED<br />
Initial or Prolonged Secretion DOSAGE<br />
Dyspnoea<br />
REGIMEN:<br />
62.MG1 MG 1. 1 DAY<br />
Date:04/11/07ISR Number: 5299140-7Report Type:Direct Company Report #CTU 300388 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam, 250mg,<br />
Initial or Prolonged Impaired Work Ability Hoffman-Laroche PS Hoffman-Laroche 250 MG ONCE<br />
Disability Bipolar I Disorder WEEKLY<br />
Discomfort<br />
Date:04/12/07ISR Number: 5294969-3Report Type:Expedited (15-DaCompany Report #KR-ROCHE-490940 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Myocardial Infarction Lariam PS Roche UNKNOWN 4 WK<br />
Date:04/13/07ISR Number: 5296357-2Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Formication Lariam PS Roche UNKNOWN DOSAGE<br />
Initial or Prolonged Abnormal Dreams REGIMEN<br />
Disturbance In Attention<br />
REPORTED AS<br />
Cardiovascular Disorder<br />
FOUR TABS IN<br />
Fatigue TOTAL. WEEKLY 22 DAY<br />
Agitation<br />
Depression<br />
Sleep Disorder<br />
Headache<br />
Anxiety<br />
Tinnitus<br />
Paraesthesia<br />
Feeling Abnormal<br />
Mental Disorder<br />
Oedema Peripheral<br />
Tachycardia<br />
Muscle Twitching<br />
Night Sweats<br />
Malaise<br />
Febrile Infection<br />
Coordination Abnormal<br />
Dyspepsia<br />
Asthenia
Vertigo<br />
03-Apr-2012 09:37 AM Page: 741
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/13/07ISR Number: 5296514-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-488026 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Cardiac Arrest Lariam PS Roche ORAL DOSE: 250 MG<br />
Hospitalization - Electrocardiogram Qt EVERY WEEK<br />
Initial or Prolonged Prolonged Pseudoephedrine SS ORAL<br />
Date:04/13/07ISR Number: 5300366-4Report Type:Direct Company Report #CTU 300636 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Abnormal Dreams Lariam PS<br />
Adverse Drug Reaction<br />
Suicidal Ideation<br />
Hallucination<br />
Completed Suicide<br />
Depression<br />
Date:04/16/07ISR Number: 5297609-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-477655 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vestibular Disorder Lariam PS Roche UNKNOWN THE PATIENT<br />
Initial or Prolonged Nightmare REPORTED<br />
Convulsion<br />
TAKING THREE<br />
Psychotic Disorder<br />
LARIAM<br />
Aggression<br />
"PILLS" IN<br />
Thinking Abnormal<br />
Insomnia<br />
Suicidal Ideation<br />
Hallucination<br />
Date:04/16/07ISR Number: 5297627-4Report Type:Expedited (15-DaCompany Report #SE-ROCHE-490181 Age:226 WK Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Infection Lariam PS Roche ORAL DOSAGE<br />
Hospitalization - Interstitial Lung Disease REGIMEN<br />
Initial or Prolonged Weight Decreased REPORTED AS<br />
Pulmonary Fibrosis 75MGW MG W. 7 WK<br />
Inflammation Lariam SS Roche ORAL<br />
Respiratory Failure<br />
Rash<br />
Pyrexia<br />
Depressed Level Of<br />
Consciousness<br />
Fatigue<br />
Alveolitis<br />
Cough
Date:04/30/07ISR Number: 5311573-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-490463 Age:46 YR Gender:Male I/FU:F<br />
Outcome<br />
Death<br />
PT<br />
Road Traffic Accident<br />
Hyperhidrosis<br />
Dry Mouth<br />
03-Apr-2012 09:37 AM Page: 742
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Asthenia<br />
Drug Interaction<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Laroxyl I Roche ORAL<br />
Date:04/30/07ISR Number: 5312051-3Report Type:Expedited (15-DaCompany Report #US-ROCHE-439267 Age:14 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Cognitive Disorder<br />
Disability<br />
Encephalopathy<br />
Other Serious<br />
Suicidal Ideation<br />
Depression<br />
Speech Disorder<br />
Psychotic Disorder<br />
Panic Attack<br />
Anxiety Disorder<br />
Date:04/30/07ISR Number: 5312244-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Agitation<br />
Nightmare<br />
Vertigo<br />
Chest Pain<br />
Tinnitus<br />
Nervousness<br />
Photophobia<br />
Phonophobia<br />
Amnesia<br />
Testicular Pain<br />
Disturbance In Attention<br />
Rash<br />
Blood Pressure Increased<br />
Dizziness<br />
Irritable Bowel Syndrome<br />
Nausea<br />
Mood Swings<br />
Fatigue<br />
Psychotic Disorder<br />
Diarrhoea<br />
Memory Impairment<br />
Disorientation<br />
Headache<br />
Abdominal Pain<br />
Eye Pain<br />
Pain Of Skin<br />
Night Sweats
Vomiting<br />
Anxiety Disorder<br />
Nystagmus<br />
Inner Ear Disorder<br />
Depression<br />
Insomnia<br />
Vision Blurred<br />
Vestibular Disorder<br />
03-Apr-2012 09:37 AM Page: 743
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hypoaesthesia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Imodium<br />
C<br />
Compazine C ON 18 APR<br />
2001, THERAPY<br />
REPORTED AS<br />
TWICE DAILY.<br />
Antibiotics Nos C<br />
Ambien<br />
C<br />
Prednisone C BY TAPER.<br />
Valium C ON 18 APR<br />
2001, THERAPY<br />
REPORTED AS:<br />
AS NEEDED.<br />
Phenergan<br />
C<br />
Biaxin<br />
C<br />
Advil<br />
C<br />
Bactrim<br />
C<br />
Antivert<br />
C<br />
Date:04/30/07ISR Number: 5316669-3Report Type:Direct Company Report #CTU 301881 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue<br />
<strong>Mefloquine</strong> 250mg<br />
Photophobia Geneva Pharm PS Geneva Pharm ORAL ONE TAB<br />
Malaise<br />
WEEKLY PRIO,<br />
Dizziness<br />
10 DAYS AFTER<br />
Disorientation<br />
LEA PO<br />
Vision Blurred<br />
Headache<br />
Circadian Rhythm Sleep<br />
Disorder<br />
Nausea<br />
Date:05/02/07ISR Number: 5317809-2Report Type:Direct Company Report #CTU 302172 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Confusional State <strong>Mefloquine</strong> (Lariam) PS Roche ORAL 1 TABLET ONCE<br />
Other Serious Hallucination PO<br />
Required<br />
Amnesia<br />
Intervention to<br />
Anxiety<br />
Prevent Permanent Panic Attack<br />
Impairment/Damage<br />
Date:05/03/07ISR Number: 5316001-5Report Type:Expedited (15-DaCompany Report #KR-ROCHE-490940 Age:40 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Arteriosclerosis Coronary Lariam PS Roche UNKNOWN 4 WK<br />
Artery<br />
Cardiomegaly<br />
Myocardial Infarction<br />
03-Apr-2012 09:37 AM Page: 744
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/07/07ISR Number: 5318027-4Report Type:Expedited (15-DaCompany Report #KR-ROCHE-490940 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Myocardial Infarction Lariam PS Roche UNKNOWN 4 WK<br />
Arteriosclerosis Coronary<br />
Artery<br />
Cardiomegaly<br />
Date:05/09/07ISR Number: 5320400-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abnormal Dreams Lariam PS Roche UNKNOWN DOSAGE<br />
Initial or Prolonged Agitation REGIMEN<br />
Other Serious Dyspepsia REPORTED AS<br />
Headache<br />
FOUR TABS IN<br />
Asthenia<br />
TOTAL. WEEKLY<br />
Mental Disorder<br />
Depression<br />
Formication<br />
Feeling Abnormal<br />
Sleep Disorder<br />
Fatigue<br />
Oedema Peripheral<br />
Coordination Abnormal<br />
Night Sweats<br />
Anxiety<br />
Tachycardia<br />
Tinnitus<br />
Muscle Twitching<br />
Upper Respiratory Tract<br />
Infection<br />
Pyrexia<br />
Paraesthesia<br />
Disturbance In Attention<br />
Vertigo<br />
Cardiovascular Disorder<br />
Malaise<br />
Date:05/11/07ISR Number: 5322571-3Report Type:Expedited (15-DaCompany Report #KR-ROCHE-490940 Age:40 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Myocardial Infarction Lariam PS Roche UNKNOWN 4 WK<br />
Cardiomegaly<br />
Arteriosclerosis Coronary<br />
Artery<br />
Date:05/14/07ISR Number: 5323676-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-490463 Age:46 YR Gender:Male I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hyperhidrosis Lariam PS Roche ORAL<br />
Drug Interaction Laroxyl I Roche ORAL<br />
Road Traffic Accident<br />
Asthenia<br />
Dry Mouth<br />
03-Apr-2012 09:37 AM Page: 745
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/16/07ISR Number: 5326183-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-497219 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiac Disorder Lariam PS Roche ORAL 37 DAY<br />
Hypothyroidism Typhoid Vaccine C 1 DAY<br />
Grand Mal Convulsion Yellow Fever Vaccine C 1 DAY<br />
Hepatitis A Vaccine C 1 DAY<br />
Levothyroxine C ORAL<br />
Date:05/18/07ISR Number: 5328482-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-489574 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Vertigo Lariam PS Roche ORAL<br />
Cognitive Disorder Trinordiol C DOSAGE<br />
Amnesia<br />
REGIMEN<br />
Anxiety REPORTED AS 1<br />
Hearing Impaired<br />
DOSE ONCE A<br />
Muscle Spasms<br />
DAY.<br />
Hallucination<br />
Palpitations<br />
Tremor<br />
Disturbance In Attention<br />
Nausea<br />
Nightmare<br />
Dyspnoea<br />
Date:05/21/07ISR Number: 5330072-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Lariam PS Roche UNKNOWN DOSAGE<br />
Initial or Prolonged Tachycardia REGIMEN<br />
Other Serious Coordination Abnormal REPORTED AS<br />
Mental Disorder<br />
FOUR TABS IN<br />
Night Sweats<br />
TOTAL. WEEKLY<br />
Paraesthesia<br />
Oedema Peripheral<br />
Upper Respiratory Tract<br />
Infection<br />
Agitation<br />
Dyspepsia<br />
Disturbance In Attention<br />
Pyrexia<br />
Headache<br />
Vertigo<br />
Muscle Twitching<br />
Anxiety<br />
Depression<br />
Abnormal Dreams<br />
Formication
Sleep Disorder<br />
Malaise<br />
Asthenia<br />
Tinnitus<br />
Feeling Abnormal<br />
Cardiovascular Disorder<br />
03-Apr-2012 09:37 AM Page: 746
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/29/07ISR Number: 5336930-6Report Type:Expedited (15-DaCompany Report #IT-ROCHE-498140 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Renal Failure Lariam PS Roche ORAL<br />
Hospitalization - Thrombotic<br />
Initial or Prolonged Thrombocytopenic Purpura<br />
Date:05/31/07ISR Number: 5343858-4Report Type:Direct Company Report #CTU 304366 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Lariam 250mg Roche PS Roche ORAL 1 TABLET<br />
Insomnia<br />
WEEKLY PO<br />
Condition Aggravated<br />
Anxiety<br />
Memory Impairment<br />
Impaired Work Ability<br />
Amnesia<br />
Paraesthesia<br />
Vision Blurred<br />
Influenza Like Illness<br />
Mood Altered<br />
Disturbance In Attention<br />
Date:05/31/07ISR Number: 5356549-0Report Type:Periodic Company Report #329978 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Agitation Health Hydrochloride) 250<br />
Dry Mouth Professional Mg PS ORAL 1 DOSE FORM 1<br />
Suicidal Ideation Other PER WEEK ORAL<br />
Depression<br />
Ultran<br />
Dizziness (Phenaglycodol) C<br />
Fatigue<br />
Lorazepam<br />
Dyspnoea (Lorazepam) C<br />
Abnormal Dreams<br />
Unspecified<br />
Headache<br />
Immunizations<br />
Nervousness (Immunisation) C<br />
Hallucination<br />
Effexor (Venlafaxine<br />
Mood Swings Hydrochloride) C<br />
Tremor<br />
Eye Irritation<br />
Nightmare<br />
Heart Rate Increased<br />
Claustrophobia<br />
Memory Impairment<br />
Disturbance In Attention<br />
Balance Disorder<br />
Psychotic Disorder<br />
Insomnia
Date:05/31/07ISR Number: 5356550-7Report Type:Periodic Company Report #444908 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Other Hydrochloride) PS<br />
03-Apr-2012 09:37 AM Page: 747
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/31/07ISR Number: 5356551-9Report Type:Periodic Company Report #452842 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Neurotoxicity Consumer Lariam (<strong>Mefloquine</strong><br />
Fatigue Other Hydrochloride) 250<br />
Simple Partial Seizures Mg PS ORAL 250 MG 1 PER<br />
Abdominal Pain<br />
ONE DOSE ORAL<br />
Asthenia<br />
Date:05/31/07ISR Number: 5356552-0Report Type:Periodic Company Report #466044 Age:10 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Disorder Literature Lariam (<strong>Mefloquine</strong><br />
Health Hydrochloride) PS ORAL 250 MG ORAL<br />
Professional<br />
Other<br />
Date:05/31/07ISR Number: 5356553-2Report Type:Periodic Company Report #466080 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Other Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride) 250<br />
Mg<br />
PS<br />
Prozac (Fluoxetine) C<br />
Date:05/31/07ISR Number: 5356554-4Report Type:Periodic Company Report #466103 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Other Hydrochloride) 250<br />
Mg<br />
PS<br />
Date:05/31/07ISR Number: 5356555-6Report Type:Periodic Company Report #467434 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam (<strong>Mefloquine</strong><br />
Hydrochloride) 250<br />
Mg<br />
PS<br />
Date:05/31/07ISR Number: 5356556-8Report Type:Periodic Company Report #469321 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Ineffective Literature <strong>Mefloquine</strong>
Initial or Prolonged Plasmodium Falciparum Health Hydrochloride<br />
Infection Professional (<strong>Mefloquine</strong><br />
Other Hydrochloride) PS ORAL 250 MG 1 PER<br />
1 WEEK ORAL<br />
03-Apr-2012 09:37 AM Page: 748
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:05/31/07ISR Number: 5356557-XReport Type:Periodic Company Report #484946 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Psychotic Disorder Other Hydrochloride) PS<br />
Date:05/31/07ISR Number: 5356558-1Report Type:Periodic Company Report #487913 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Consumer <strong>Mefloquine</strong><br />
Depression Other Hydrochloride<br />
Headache<br />
(<strong>Mefloquine</strong><br />
Speech Disorder Hydrochloride) 250<br />
Disturbance In Attention Mg PS ORAL 250 MG 1 PER<br />
Hypoaesthesia<br />
Memory Impairment<br />
Zithromax<br />
(Azithromycin)<br />
C<br />
WEEK ORAL<br />
Date:05/31/07ISR Number: 5356559-3Report Type:Periodic Company Report #336177 Age:40 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Rash Papular<br />
Disturbance In Attention<br />
Groin Pain<br />
Panic Attack<br />
Oral Disorder<br />
Agitation<br />
Decreased Activity<br />
Hyperventilation<br />
Nystagmus<br />
Major Depression<br />
Chest Discomfort<br />
Insomnia<br />
Depressed Mood<br />
Tinnitus<br />
Vitreous Floaters<br />
Blood Bilirubin Increased<br />
Dyspnoea<br />
Paraesthesia<br />
Blood Pressure Increased<br />
Blood Pressure<br />
Agoraphobia<br />
Dysuria<br />
Blood Creatinine<br />
Increased<br />
Palpitations<br />
Anxiety<br />
Lichen Planus<br />
Dizziness
Headache<br />
Hyperhidrosis<br />
Nightmare<br />
Fatigue<br />
Balance Disorder<br />
Decreased Interest<br />
Decreased Appetite<br />
Anhedonia<br />
03-Apr-2012 09:37 AM Page: 749
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pain In Extremity<br />
Haematuria<br />
Heart Rate Increased Report Source Product Role Manufacturer Route Dose Duration<br />
Erythema Consumer Lariam (<strong>Mefloquine</strong><br />
Mood Altered Health Hydrochloride) 250<br />
Depression Professional Mg PS ORAL 250 MG ORAL<br />
Epicondylitis<br />
Other<br />
Toxicity To Various<br />
Agents<br />
Vision Blurred<br />
Vertigo<br />
Panic Disorder<br />
Urethritis<br />
Date:05/31/07ISR Number: 5356560-XReport Type:Periodic Company Report #428956 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Consumer Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Health Hydrochloride) PS ORAL ORAL<br />
Professional<br />
Other<br />
Birth Control Pills<br />
Nos (Oral<br />
Contraceptives Nos)<br />
C<br />
Date:06/04/07ISR Number: 5344208-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-499416 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maculopathy Lariam PS Roche UNKNOWN 6 MON<br />
Visual Acuity Reduced<br />
Date:06/05/07ISR Number: 5345982-9Report Type:Expedited (15-DaCompany Report #AU-ROCHE-499414 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hepatitis Fulminant Lariam PS Roche UNKNOWN<br />
Date:06/05/07ISR Number: 5345983-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Other Serious<br />
PT<br />
Upper Respiratory Tract<br />
Infection<br />
Malaise<br />
Agitation<br />
Anxiety<br />
Paraesthesia<br />
Feeling Abnormal<br />
Vertigo
Disturbance In Attention<br />
Pyrexia<br />
Dyspepsia<br />
Asthenia<br />
Depression<br />
Tachycardia<br />
Tinnitus<br />
Formication<br />
03-Apr-2012 09:37 AM Page: 750
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Coordination Abnormal<br />
Night Sweats<br />
Cardiovascular Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Abnormal Dreams Lariam PS Roche UNKNOWN DOSAGE<br />
Headache<br />
REGIMEN<br />
Sleep Disorder<br />
REPORTED AS<br />
Fatigue<br />
FOUR TABS IN<br />
Muscle Twitching<br />
TOTAL. WEEKLY<br />
Mental Disorder<br />
Oedema Peripheral<br />
Date:06/06/07ISR Number: 5347841-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-497864 Age:23 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amenorrhoea Lariam PS Roche ORAL<br />
Date:06/06/07ISR Number: 5347843-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-500150 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amenorrhoea Lariam PS Roche ORAL<br />
Date:06/06/07ISR Number: 5350805-8Report Type:Direct Company Report #CTU 304987 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination <strong>Mefloquine</strong> PS ORAL 1 PILL EVERY<br />
Required Paranoia 7 DAYS PO<br />
Intervention to<br />
Panic Attack<br />
Prevent Permanent<br />
Impairment/Damage<br />
Nightmare<br />
Anxiety<br />
Weight Decreased<br />
Tremor<br />
Crying<br />
Insomnia<br />
Date:06/08/07ISR Number: 5351021-6Report Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Lariam PS Roche ORAL 3 MON<br />
Hallucination, Olfactory Flagyl C ORAL<br />
Hallucination, Auditory Unknown Medication C ORAL<br />
Hallucination Antidepressants Nos C<br />
Homicide<br />
Flashback
Date:06/13/07ISR Number: 5355886-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-497864 Age:23 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amenorrhoea Lariam PS Roche ORAL<br />
Lariam SS Roche ORAL 3 MON<br />
03-Apr-2012 09:37 AM Page: 751
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/13/07ISR Number: 5355888-7Report Type:Expedited (15-DaCompany Report #AU-ROCHE-499414 Age:24 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Vomiting Lariam PS Roche ORAL<br />
Hepatitis Fulminant Rabies Vaccine C<br />
Hepatic Failure Typhoid Vaccine C<br />
Diarrhoea Levlen Ed C<br />
Tetanus Vaccine C<br />
Date:06/18/07ISR Number: 5362132-3Report Type:Direct Company Report #USP 59046 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Drug Dispensing Error Lariam PS TABLET<br />
Medication Error Malarone SS Glaxosmithkline TABLET<br />
Date:06/19/07ISR Number: 5362230-4Report Type:Expedited (15-DaCompany Report #IT-ROCHE-501993 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged<br />
Date:06/20/07ISR Number: 5363677-2Report Type:Expedited (15-DaCompany Report #ES-ROCHE-502414 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Toxic <strong>Mefloquine</strong><br />
Hydrochloride PS Roche UNKNOWN<br />
Hepatitis B Vaccine C<br />
Date:06/22/07ISR Number: 5367205-7Report Type:Expedited (15-DaCompany Report #SE-ROCHE-498551 Age:20 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diplopia Lariam PS Roche ORAL THE PATIENT<br />
Initial or Prolonged Headache TOOK A TOTAL<br />
Neuropathy Peripheral<br />
OF TWO<br />
TABLETS.<br />
REPORTED 1 DAY<br />
Malarone SS ORAL FORM: FILM<br />
COATED (FC)<br />
TABLETS. 5 WK<br />
Unspecified Drug SS UNKNOWN 1 DAY<br />
Lexinor SS ORAL FORM FILM<br />
COATED (FC)<br />
TABLETS. 1 DAY
Date:06/22/07ISR Number: 5367210-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age:36 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Other Serious<br />
PT<br />
Dyspepsia<br />
Muscle Twitching<br />
Paraesthesia<br />
Formication<br />
03-Apr-2012 09:37 AM Page: 752
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Oedema Peripheral<br />
Sleep Disorder<br />
Upper Respiratory Tract Report Source Product Role Manufacturer Route Dose Duration<br />
Infection Lariam PS Roche UNKNOWN DOSAGE<br />
Headache<br />
REGIMEN<br />
Fatigue<br />
REPORTED AS<br />
Night Sweats<br />
FOUR TABS IN<br />
Malaise<br />
TOTAL. WEEKLY<br />
Agitation<br />
Abnormal Dreams<br />
Tinnitus<br />
Asthenia<br />
Feeling Abnormal<br />
Coordination Abnormal<br />
Pyrexia<br />
Tachycardia<br />
Disturbance In Attention<br />
Anxiety<br />
Depression<br />
Vertigo<br />
Mental Disorder<br />
Cardiovascular Disorder<br />
Date:06/25/07ISR Number: 5369184-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-502756 Age:51 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Amyotrophic Lateral Lariam PS Roche ORAL REPORTED AS<br />
Initial or Prolonged Sclerosis TAKEN 5<br />
Paraneoplastic Syndrome<br />
WEEKLY DOSES<br />
Vitamin E<br />
C<br />
Alpha Lipoic Acid C<br />
Vitamins Nos<br />
C<br />
Co-Enzyme<br />
C<br />
Rilutek<br />
C<br />
Vitamin C<br />
C<br />
Date:06/25/07ISR Number: 5370250-9Report Type:Direct Company Report #CTU 306401 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Medication Error Mercaptopurine 50mg<br />
Intervention to Intercepted Drug Tab-Roxane PS Roxane<br />
Prevent Permanent Dispensing Error <strong>Mefloquine</strong> 250mg Tab<br />
Impairment/Damage Roxane SS Roxane<br />
Date:06/29/07ISR Number: 5374239-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-262840 Age:34 YR Gender:Male I/FU:F<br />
Outcome<br />
PT
Disability<br />
Adjustment Disorder With<br />
Anxiety<br />
Photophobia<br />
Phobia<br />
Generalised Anxiety<br />
Disorder<br />
Hypoaesthesia<br />
Amnesia<br />
03-Apr-2012 09:37 AM Page: 753
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Anxiety<br />
Panic Attack<br />
Diarrhoea Report Source Product Role Manufacturer Route Dose Duration<br />
Self Esteem Decreased Lariam PS Roche ORAL THE PATIENT<br />
Headache HAD TAKEN 4-5<br />
Acrophobia<br />
TABLETS OF<br />
Heart Rate Increased<br />
MEFLOQUINE IN<br />
Insomnia TOTAL. 21 DAY<br />
Hypersensitivity Vaccine C OTHER 1 DAY<br />
Musculoskeletal Stiffness<br />
Bronchial Hyperreactivity<br />
Mood Disorder Due To A<br />
General Medical Condition<br />
Dizziness<br />
Paraesthesia<br />
Suicidal Ideation<br />
Abdominal Pain<br />
Heart Rate Irregular<br />
Tinnitus<br />
Disturbance In Attention<br />
Balance Disorder<br />
Vestibular Disorder<br />
Weight Decreased<br />
Lactose Intolerance<br />
Throat Irritation<br />
Nausea<br />
Motion Sickness<br />
Depression<br />
Disorientation<br />
Dyspnoea<br />
Irritable Bowel Syndrome<br />
Phobia Of Flying<br />
Migraine<br />
Nasal Congestion<br />
Sleep Disorder Due To<br />
General Medical<br />
Condition, Hypersomnia<br />
Type<br />
Adjustment Disorder With<br />
Depressed Mood<br />
Sinus Bradycardia<br />
Rash<br />
Sensory Integrative<br />
Dysfunction<br />
Fatigue<br />
Neck Pain<br />
Abdominal Distension<br />
Abdominal Pain Lower<br />
Vertigo<br />
Emotional Distress<br />
Claustrophobia<br />
Sleep Disorder<br />
Back Pain
Date:06/29/07ISR Number: 5374266-8Report Type:Expedited (15-DaCompany Report #IE-ROCHE-337440 Age:27 YR Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Flashback<br />
Hallucination<br />
03-Apr-2012 09:37 AM Page: 754
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucination, Auditory<br />
Homicide<br />
Hallucination, Olfactory Report Source Product Role Manufacturer Route Dose Duration<br />
Paranoia Lariam PS Roche ORAL 3 MON<br />
Flagyl C ORAL<br />
Unknown Medication C ORAL<br />
Antidepressants Nos C<br />
Date:06/29/07ISR Number: 5374267-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Diarrhoea Lariam PS Roche ORAL<br />
Memory Impairment Imodium C<br />
Vestibular Disorder Compazine C ON 18 APR<br />
Nystagmus<br />
2001, THERAPY<br />
Abdominal Pain<br />
REPORTED AS<br />
Phonophobia<br />
TWICE DAILY.<br />
Insomnia Antibiotics Nos C<br />
Vertigo Ambien C<br />
Depression Prednisone C BY TAPER.<br />
Eye Pain Valium C ON 18 APR<br />
Nightmare<br />
2001, THERAPY<br />
Tinnitus<br />
REPORTED AS:<br />
Psychotic Disorder<br />
AS NEEDED.<br />
Agitation Phenergan C<br />
Vomiting Biaxin C<br />
Headache Advil C<br />
Inner Ear Disorder Bactrim C<br />
Photophobia Antivert C<br />
Anxiety Disorder<br />
Dizziness<br />
Irritable Bowel Syndrome<br />
Rash<br />
Pain Of Skin<br />
Blood Pressure Increased<br />
Mood Swings<br />
Disorientation<br />
Amnesia<br />
Nervousness<br />
Night Sweats<br />
Nausea<br />
Fatigue<br />
Chest Pain<br />
Testicular Pain<br />
Vision Blurred<br />
Disturbance In Attention<br />
Hypoaesthesia<br />
Date:06/29/07ISR Number: 5374285-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-314899 Age:40 YR Gender:Female I/FU:F
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Incontinence<br />
Drug Ineffective<br />
Hostility<br />
Headache<br />
Muscle Rigidity<br />
Agitation<br />
03-Apr-2012 09:37 AM Page: 755
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Mania<br />
Psychotic Disorder<br />
Disorientation Report Source Product Role Manufacturer Route Dose Duration<br />
Loss Of Libido Lariam PS Roche ORAL EVERY WEEK. 9 DAY<br />
Confusional State Valium SS Roche INTRAVENOUS 1 DAY<br />
Delusion Prozac C ORAL<br />
Hallucination<br />
Paranoia<br />
Convulsion<br />
Pyrexia<br />
Gait Disturbance<br />
Speech Disorder<br />
Fatigue<br />
Antisocial Behaviour<br />
Aggression<br />
Blood Potassium Decreased<br />
Depression<br />
Memory Impairment<br />
Date:07/03/07ISR Number: 5377491-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-498551 Age:20 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diplopia Lariam PS Roche ORAL THE PATIENT<br />
Initial or Prolonged Neuropathy Peripheral TOOK A TOTAL<br />
Headache<br />
OF TWO<br />
TABLETS.<br />
REPORTED 1 DAY<br />
Malarone SS ORAL FORM: FILM<br />
COATED (FC)<br />
TABLETS. 5 WK<br />
Unspecified Drug SS UNKNOWN 1 DAY<br />
Lexinor SS ORAL FORM FILM<br />
COATED (FC)<br />
TABLETS. 5 DAY<br />
Date:07/09/07ISR Number: 5381827-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-505274 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Torsade De Pointes Lariam PS Roche ORAL<br />
Initial or Prolonged Electrocardiogram Qt Meningococcal<br />
Prolonged Vaccine C<br />
Hepatyrix<br />
C<br />
Revaxis<br />
C<br />
Warfarin<br />
C<br />
Ramipril<br />
C<br />
Date:07/09/07ISR Number: 5382114-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-502756 Age:51 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Amyotrophic Lateral Lariam PS Roche ORAL REPORTED AS<br />
Initial or Prolonged Sclerosis TAKEN 5<br />
Paraneoplastic Syndrome<br />
WEEKLY DOSES<br />
Vitamin E<br />
C<br />
Alpha Lipoic Acid C<br />
Vitamins Nos<br />
C<br />
03-Apr-2012 09:37 AM Page: 756
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Co-Enzyme<br />
Rilutek<br />
Vitamin C<br />
C<br />
C<br />
C<br />
Date:07/09/07ISR Number: 5385345-3Report Type:Direct Company Report #CTU 307608 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare <strong>Mefloquine</strong> 250 Mg<br />
Dizziness Barr Laboratories PS Barr Laboratories ORAL ONCE PER WEEK<br />
Mood Swings<br />
PO<br />
Sleep Disorder<br />
Irritability<br />
Insomnia<br />
Date:07/10/07ISR Number: 5386284-4Report Type:Direct Company Report #CTU 307650 Age:28 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Lariam -<strong>Mefloquine</strong>- PS ORAL 1 TABLET PO<br />
Convulsion<br />
Asthenia<br />
Nervous System Disorder<br />
Paraesthesia<br />
Muscle Spasms<br />
Muscle Twitching<br />
Hypoaesthesia<br />
Date:07/13/07ISR Number: 5390014-XReport Type:Expedited (15-DaCompany Report #GXKR2007TR05543 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Parkinsonism Foreign <strong>Mefloquine</strong> PS<br />
Literature<br />
Health<br />
Professional<br />
Other<br />
Date:07/16/07ISR Number: 5387788-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-503881 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam PS Roche UNKNOWN REPORTED THAT<br />
Tremor<br />
PATIENT TOOK<br />
Ill-Defined Disorder<br />
3 DOSES.<br />
Abnormal Dreams Allegra C ORAL<br />
Pyrexia Wellbutrin C ORAL REPORTED AS<br />
Fatigue<br />
150-450MG.<br />
Sleep Disorder<br />
Unspecified
Mental Disorder Medication C ORAL<br />
Ativan C ORAL<br />
Lexapro C ORAL<br />
03-Apr-2012 09:37 AM Page: 757
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/16/07ISR Number: 5388059-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-54240 Age:52 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam PS Roche ORAL 27 DAY<br />
Initial or Prolonged Visual Impairment Pizotifen I UNKNOWN<br />
Chest Pain<br />
Panic Attack<br />
Fatigue<br />
Head Discomfort<br />
Drug Interaction<br />
Nausea<br />
Date:07/16/07ISR Number: 5388628-6Report Type:Expedited (15-DaCompany Report #CA-ROXANE LABORATORIES, INC-2007-BP-17394RO Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maculopathy <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Retinal Pigmentation Inc. ORAL 1 YR<br />
Date:07/16/07ISR Number: 5388633-XReport Type:Expedited (15-DaCompany Report #TR-ROXANE LABORATORIES, INC-2007-BP-17147RO Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Parkinsonism <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Inc.<br />
ORAL<br />
Date:07/17/07ISR Number: 5389111-4Report Type:Expedited (15-DaCompany Report #NL-ROCHE-505944 Age:4 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion Lariam PS Roche ORAL<br />
Hospitalization -<br />
Initial or Prolonged<br />
Date:07/17/07ISR Number: 5392183-4Report Type:Direct Company Report #CTU 308140 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Myalgia <strong>Mefloquine</strong> 250 Mg PS ORAL 1 ONCE A WEEK<br />
Intervention to Tendonitis PO<br />
Prevent Permanent Swelling<br />
Impairment/Damage Pain In Extremity<br />
Date:07/23/07ISR Number: 5397614-1Report Type:Direct Company Report #CTU 308660 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Infection Larium Hoffman La
Hospitalization - Tongue Disorder Roche PS Hoffman La Roche ORAL 1 TABLET ONCE<br />
Initial or Prolonged Partial Seizures A WEEK PO<br />
Other Serious<br />
Swollen Tongue<br />
Required<br />
Epilepsy<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 758
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/26/07ISR Number: 5397579-2Report Type:Expedited (15-DaCompany Report #NL-ROCHE-507469 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Diarrhoea Lariam PS Roche ORAL<br />
Nightmare Microgynon C ORAL DOSAGE<br />
Psychotic Disorder<br />
REGIMEN<br />
Disturbance In Attention<br />
REPORTED AS<br />
Bone Pain 1X/1<br />
CYCLICAL.<br />
Hepatitis A<br />
Vaccine/Hepatitis B<br />
Vaccine<br />
C<br />
Date:07/27/07ISR Number: 5399351-6Report Type:Expedited (15-DaCompany Report #NL-ROCHE-507408 Age:29 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Depression Lariam PS Roche ORAL<br />
Completed Suicide<br />
Date:07/30/07ISR Number: 5400247-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-477655 Age:33 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cognitive Disorder Lariam PS Roche UNKNOWN THE PATIENT<br />
Initial or Prolonged Memory Impairment REPORTED<br />
Disability Insomnia TAKING THREE<br />
Other Serious Nightmare LARIAM<br />
Suicidal Ideation<br />
"PILLS" IN<br />
Convulsion Ibuprofen C<br />
Flight Of Ideas Birth Control Pill C DRUG NAME<br />
Aggression<br />
REPORTED AS:<br />
Visual Impairment<br />
BCP (TYPE<br />
Vestibular Disorder<br />
UNKNOWN)<br />
Psychotic Disorder Tylenol C<br />
Hallucination<br />
Date:07/30/07ISR Number: 5402419-9Report Type:Direct Company Report #CTU 309170 Age:32 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Mood Altered<br />
Memory Impairment<br />
Headache<br />
Feeling Abnormal<br />
Thinking Abnormal<br />
Confusional State<br />
Dizziness<br />
Disturbance In Attention<br />
Affect Lability
Anxiety<br />
Amnesia<br />
Depression<br />
Motor Dysfunction<br />
Social Avoidant Behaviour<br />
Mood Swings<br />
Anger<br />
Paranoia<br />
03-Apr-2012 09:37 AM Page: 759
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Irritability<br />
Violence-Related Symptom<br />
Crying Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong> (Lariam) PS<br />
1 TABLET<br />
WEEKLY<br />
Date:07/30/07ISR Number: 5402425-4Report Type:Direct Company Report #CTU 309171 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Lariam Roche PS Roche ORAL 1 ONCE A WEEK<br />
Hallucination, Olfactory<br />
PO<br />
Dizziness<br />
Vertigo<br />
Depression<br />
Mania<br />
Mood Altered<br />
Fatigue<br />
Nausea<br />
Hallucination, Auditory<br />
Vomiting<br />
Date:08/03/07ISR Number: 5405080-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-489574 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam PS Roche ORAL FIRST INTAKE<br />
Other Serious Amnesia (ONE TABLET)<br />
Vertigo<br />
ON 19 FEB<br />
Muscle Spasms<br />
2007, SECOND<br />
Asthenia<br />
INTAKE (ONE<br />
Cognitive Disorder Trinordiol C DOSAGE<br />
Disturbance In Attention<br />
REGIMEN<br />
Palpitations REPORTED AS 1<br />
Nightmare<br />
DOSE ONCE A<br />
Hallucination<br />
DAY.<br />
Tremor<br />
Hearing Impaired<br />
Nausea<br />
Dyspnoea<br />
Date:08/03/07ISR Number: 5405240-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-508756 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cerebellar Ataxia Lariam PS Roche ORAL FIRST THREE<br />
Initial or Prolonged<br />
TABLETS, THEN<br />
SIX HOURS<br />
LATER TWO<br />
TABLETS, THEN
Date:08/09/07ISR Number: 5409796-3Report Type:Expedited (15-DaCompany Report #NL-ROCHE-509467 Age:59 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
03-Apr-2012 09:37 AM Page: 760
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Initial or Prolonged<br />
PT Report Source Product Role Manufacturer Route Dose Duration<br />
Orthostatic Hypotension Lariam PS Roche ORAL<br />
Coma Fortzaar SS ORAL<br />
Blood Pressure Decreased Doxazosine SS ORAL DOSAGE<br />
Syncope<br />
REGIMEN: 2 DD<br />
Hyperhidrosis<br />
4 MG.<br />
Tremor<br />
Bradycardia<br />
Date:08/10/07ISR Number: 5411289-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-510074 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depression Lariam PS Roche UNKNOWN TOTAL OF<br />
Initial or Prolonged Headache THREE<br />
Vomiting<br />
TABLETS.<br />
Social Phobia<br />
SECOND TABLET<br />
Tinnitus<br />
TAKEN IN<br />
Paraesthesia<br />
Nausea<br />
Gastrointestinal Disorder<br />
Panic Attack<br />
Anxiety<br />
Hyperhidrosis<br />
Sensation Of Foreign Body<br />
Date:08/21/07ISR Number: 5420894-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-511983 Age:65 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Gynaecomastia Lariam PS Roche ORAL<br />
Date:08/22/07ISR Number: 5424395-5Report Type:Expedited (15-DaCompany Report #6036499 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Coma Health Lariam (250 Mg),<br />
Hospitalization - Blood Pressure Decreased Professional Tablet PS 250 MG (250<br />
Initial or Prolonged Syncope Other MG 1 IN 1 WK) 35 DAY<br />
Loss Of Consciousness<br />
Fortzaar (Tablet)<br />
Bradycardia<br />
Drug Interaction<br />
(Hydrochlorothiazide<br />
, Losartan<br />
Potassium) SS 1 DOSAGE<br />
FORMS, 1 D)<br />
Doxazosin Tablets 4<br />
Mg (4 Mg Tablet)<br />
(Doxazosin) SS ORAL 8 MG (4 MG, 2<br />
IN 1 D), ORAL 97<br />
197 DAY<br />
DAY
Date:08/22/07ISR Number: 5424727-8Report Type:Direct Company Report #CTU 311097 Age: Gender:Female I/FU:I<br />
Outcome<br />
Disability<br />
PT<br />
Dizziness<br />
Memory Impairment<br />
03-Apr-2012 09:37 AM Page: 761
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bruxism<br />
Abdominal Pain Upper<br />
Insomnia Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety<br />
Lariam (<strong>Mefloquine</strong><br />
Hallucination Hcl 250 Mg) PS 250 MG 1 X<br />
Fatigue<br />
WEEK<br />
Depression<br />
Paranoia<br />
Chills<br />
Disturbance In Attention<br />
Vision Blurred<br />
Panic Attack<br />
Diarrhoea<br />
Mood Swings<br />
Suicidal Ideation<br />
Date:08/24/07ISR Number: 5425148-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-511718 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Delinquency Lariam PS Roche UNKNOWN<br />
Schizophrenia<br />
Date:08/24/07ISR Number: 5425388-4Report Type:Expedited (15-DaCompany Report #DE-ROXANE LABORATORIES, INC-2007-DE-04938GD Age:56 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vomiting Morphine PS Roxane Laboratories,<br />
Initial or Prolonged Toxicity To Various Inc.<br />
Agents Codeine SS Roxane Laboratories,<br />
Abdominal Pain<br />
Inc.<br />
Citalopram<br />
SS<br />
Diclofenac<br />
SS<br />
<strong>Mefloquine</strong> SS Roxane Laboratories,<br />
Inc.<br />
Zopiclone<br />
SS<br />
Fluoxetine<br />
SS<br />
Lansoprazole<br />
SS<br />
Penbutolol<br />
SS<br />
Rabeprazole<br />
SS<br />
Senna Preparation SS<br />
Simeticone<br />
SS<br />
Paracetamol<br />
SS<br />
Amitriptyline<br />
SS<br />
Date:08/24/07ISR Number: 5427445-5Report Type:Direct Company Report #CTU 311343 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dizziness <strong>Mefloquine</strong> PS ORAL 250 MG ONCE A
Fear WEEK PO<br />
Impaired Work Ability<br />
Anxiety<br />
Fatigue<br />
Vertigo<br />
03-Apr-2012 09:37 AM Page: 762
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/27/07ISR Number: 5429208-3Report Type:Direct Company Report #CTU 311464 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Chest Pain <strong>Mefloquine</strong> 250 Mg PS ORAL 250MG ONCE A<br />
Initial Insomnia<br />
WEEK PO<br />
Fear<br />
Panic Reaction<br />
Palpitations<br />
Heart Rate Decreased<br />
Date:08/28/07ISR Number: 5435071-7Report Type:Direct Company Report #CTU 311536 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Urticaria<br />
<strong>Mefloquine</strong> 7 Tablets<br />
Rash Pruritic Hoffman-Laroche PS Hoffman-Laroche ORAL 1 TABLET ONCE<br />
A WEEK PO<br />
Date:08/29/07ISR Number: 5429063-1Report Type:Periodic Company Report #A0655132A Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Drug Dispensing Error Malarone PS Glaxosmithkline UNKNOWN 1TAB Per day<br />
Lariam SS UNKNOWN 250MG Unknown<br />
Date:08/30/07ISR Number: 5431010-3Report Type:Expedited (15-DaCompany Report #B0485142A Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paraesthesia Malarone PS Glaxosmithkline 33 DAY<br />
Initial or Prolonged Headache Lexinor SS Glaxosmithkline 5 DAY<br />
Malaria Lariam SS 2TAB Single<br />
Diplopia<br />
dose<br />
Diarrhoea<br />
Abdominal Discomfort<br />
Dehydration<br />
Facial Paresis<br />
Neuropathy Peripheral<br />
Date:08/30/07ISR Number: 5441851-4Report Type:Direct Company Report #CTU 311703 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Accident Laiam Roche PS Roche ORAL 1 WEEKLY PO<br />
Hospitalization -<br />
Initial or Prolonged<br />
Other Serious<br />
Required<br />
Intervention to
Prevent Permanent<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 763
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/04/07ISR Number: 5442028-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-511983 Age:65 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Gynaecomastia Lariam PS Roche ORAL<br />
Esomeprazole<br />
C<br />
Date:09/06/07ISR Number: 5444869-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-509667 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vomiting Lariam PS Roche ORAL<br />
Abdominal Pain Engerix B SS INTRAMUSCULAR DRUG NAME<br />
Myalgia<br />
REPORTED AS<br />
C-Reactive Protein ENGERIX B 10.<br />
Increased<br />
INDICATION<br />
Pyrexia<br />
REPORTED AS<br />
Cytolytic Hepatitis<br />
Date:09/10/07ISR Number: 5447630-6Report Type:Expedited (15-DaCompany Report #BE-ROCHE-514773 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Lariam PS Roche UNKNOWN<br />
Date:09/11/07ISR Number: 5449004-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-515132 Age:75 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Red Blood Cell Lariam PS Roche ORAL 1 DAY<br />
Sedimentation Rate<br />
Increased<br />
Polymyalgia Rheumatica<br />
Date:09/11/07ISR Number: 5449013-1Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vestibular Disorder<br />
Chest Pain<br />
Testicular Pain<br />
Diarrhoea<br />
Anxiety Disorder<br />
Fatigue<br />
Vision Blurred<br />
Hypoaesthesia<br />
Insomnia<br />
Psychotic Disorder<br />
Nystagmus<br />
Inner Ear Disorder
Phonophobia<br />
Nausea<br />
Memory Impairment<br />
Nightmare<br />
Amnesia<br />
Tinnitus<br />
Disturbance In Attention<br />
Eye Pain<br />
03-Apr-2012 09:37 AM Page: 764
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Rash<br />
Nervousness<br />
Deafness Report Source Product Role Manufacturer Route Dose Duration<br />
Night Sweats Lariam PS Roche ORAL<br />
Agitation Imodium C<br />
Vertigo Compazine C ON 18 APR<br />
Headache<br />
2001, THERAPY<br />
Depression<br />
REPORTED AS<br />
Photophobia<br />
TWICE DAILY.<br />
Pain Of Skin Antibiotics Nos C<br />
Mood Swings Ambien C<br />
Disorientation Prednisone C BY TAPER.<br />
Dizziness Valium C ON 18 APR<br />
Vomiting<br />
2001, THERAPY<br />
Abdominal Pain<br />
REPORTED AS:<br />
Irritable Bowel Syndrome<br />
AS NEEDED.<br />
Blood Pressure Increased Phenergan C<br />
Biaxin<br />
C<br />
Advil<br />
C<br />
Bactrim<br />
C<br />
Antivert<br />
C<br />
Date:09/12/07ISR Number: 5452321-1Report Type:Direct Company Report #CTU 312582 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mental Disorder <strong>Mefloquine</strong> 250 Mg<br />
Anxiety Geneva PS Geneva ORAL 250 MG WEEKLY<br />
Panic Attack<br />
PO DOSE TAKEN<br />
Impaired Driving Ability<br />
Date:09/13/07ISR Number: 5451462-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vomiting<br />
Insomnia<br />
Vertigo<br />
Abdominal Pain<br />
Dizziness<br />
Vision Blurred<br />
Hypoaesthesia<br />
Phonophobia<br />
Blood Pressure Increased<br />
Amnesia<br />
Irritable Bowel Syndrome<br />
Depression<br />
Agitation<br />
Vestibular Disorder<br />
Nystagmus<br />
Inner Ear Disorder
Testicular Pain<br />
Rash<br />
Photophobia<br />
Deafness<br />
Diarrhoea<br />
Chest Pain<br />
Memory Impairment<br />
Eye Pain<br />
03-Apr-2012 09:37 AM Page: 765
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Nausea<br />
Mood Swings<br />
Nightmare Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety Disorder Lariam PS Roche ORAL<br />
Disturbance In Attention Imodium C<br />
Nervousness Compazine C ON 18 APR<br />
Pain Of Skin<br />
2001, THERAPY<br />
Fatigue<br />
REPORTED AS<br />
Disorientation<br />
TWICE DAILY.<br />
Headache Antibiotics Nos C<br />
Tinnitus Ambien C<br />
Night Sweats Prednisone C BY TAPER.<br />
Psychotic Disorder Valium C ON 18 APR<br />
2001, THERAPY<br />
REPORTED AS:<br />
AS NEEDED.<br />
Phenergan<br />
C<br />
Biaxin<br />
C<br />
Advil<br />
C<br />
Bactrim<br />
C<br />
Antivert<br />
C<br />
Date:09/14/07ISR Number: 5452778-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-503881 Age:41 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Ill-Defined Disorder Lariam PS Roche UNKNOWN REPORTED THAT<br />
Sleep Disorder<br />
PATIENT TOOK<br />
Pyrexia<br />
3 DOSES.<br />
Fatigue Allegra C ORAL<br />
Tremor Wellbutrin C ORAL REPORTED AS<br />
Abnormal Dreams<br />
150-450MG.<br />
Mental Disorder<br />
Unspecified<br />
Depression Medication C ORAL<br />
Ativan C ORAL<br />
Lexapro C ORAL<br />
Date:09/14/07ISR Number: 5452940-2Report Type:Expedited (15-DaCompany Report #NL-ROCHE-509467 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Blood Pressure Decreased Lariam PS Roche ORAL<br />
Hospitalization - Syncope Fortzaar SS ORAL<br />
Initial or Prolonged Orthostatic Hypotension Doxazosine SS ORAL DOSAGE<br />
Hyperhidrosis<br />
REGIMEN: 2 DD<br />
Tremor<br />
4 MG.<br />
Bradycardia<br />
Date:09/17/07ISR Number: 5453931-8Report Type:Expedited (15-DaCompany Report #AU-ROCHE-499414 Age:24 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hepatitis Fulminant Lariam PS Roche ORAL ONE DOSE<br />
Hepatic Failure<br />
WEEKLY.<br />
Jaundice Rabies Vaccine C<br />
Diarrhoea Typhoid Vaccine C<br />
Vomiting Levlen Ed C ORAL ONE DOSE.<br />
Tetanus Vaccine C<br />
03-Apr-2012 09:37 AM Page: 766
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/18/07ISR Number: 5460867-5Report Type:Expedited (15-DaCompany Report #6036499 Age:59 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Syncope Foreign Doxazosin PS ORAL 8 MG (4 MG, 2<br />
Hospitalization - Blood Pressure Decreased Health IN 1 D) ORAL 97 DAY<br />
Initial or Prolonged Bradycardia Professional Fortzaar<br />
Other<br />
(Hydrochlorothiazide<br />
, Losartan<br />
Potassium) SS 1 DOSAGE<br />
FORMS (1<br />
DOSAGE FORMS,<br />
1 IN 1 D) 197 DAY<br />
Lariam SS 250 MG (250<br />
MG, 1 IN 1<br />
WK) 35 DAY<br />
Date:09/24/07ISR Number: 5465671-XReport Type:Expedited (15-DaCompany Report #JP-ROCHE-518961 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Optic Neuritis <strong>Mefloquine</strong><br />
Hydrochloride PS Roche UNKNOWN<br />
Date:09/24/07ISR Number: 5465673-3Report Type:Expedited (15-DaCompany Report #JP-ROCHE-518962 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Lung Neoplasm Malignant <strong>Mefloquine</strong><br />
Hydrochloride PS Roche UNKNOWN<br />
Date:09/24/07ISR Number: 5465676-9Report Type:Expedited (15-DaCompany Report #JP-ROCHE-518963 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Glomerulonephritis <strong>Mefloquine</strong><br />
Proliferative Hydrochloride PS Roche UNKNOWN<br />
Date:09/28/07ISR Number: 5471134-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Vomiting<br />
Mood Swings<br />
Memory Impairment<br />
Amnesia<br />
Testicular Pain<br />
Irritable Bowel Syndrome<br />
Disturbance In Attention<br />
Eye Pain
Tinnitus<br />
Nervousness<br />
Photophobia<br />
Brain Stem Syndrome<br />
Anxiety Disorder<br />
Nystagmus<br />
Dizziness<br />
Hypoaesthesia<br />
03-Apr-2012 09:37 AM Page: 767
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorientation<br />
Vision Blurred<br />
Nightmare Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue Lariam PS Roche ORAL<br />
Headache Imodium C<br />
Inner Ear Disorder Compazine C ON 18 APR<br />
Night Sweats<br />
2001, THERAPY<br />
Nausea<br />
REPORTED AS<br />
Abdominal Pain<br />
TWICE DAILY.<br />
Depression Antibiotics Nos C<br />
Deafness Ambien C<br />
Agitation Prednisone C BY TAPER.<br />
Insomnia Valium C ON 18 APR<br />
Vertigo<br />
2001, THERAPY<br />
Vestibular Disorder<br />
REPORTED AS:<br />
Phonophobia<br />
AS NEEDED.<br />
Pain Of Skin Phenergan C<br />
Blood Pressure Increased Biaxin C<br />
Diarrhoea Advil C<br />
Chest Pain Bactrim C<br />
Psychotic Disorder Antivert C<br />
Rash<br />
Paraesthesia<br />
Date:09/28/07ISR Number: 5471155-5Report Type:Expedited (15-DaCompany Report #ES-ROCHE-519121 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tumour Marker Increased Lariam PS Roche UNKNOWN<br />
Date:10/02/07ISR Number: 5474876-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-521722 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Frequent Bowel Movements <strong>Mefloquine</strong><br />
Mood Swings Hydrochloride PS Roche ORAL 40 DAY<br />
Abnormal Dreams Unspecified Drug C<br />
Alice In Wonderland Nicotine C TRANSDERMAL DOSING AMOUNT<br />
Syndrome<br />
Clumsiness<br />
AND FREQUENCY<br />
REPORTED AS:<br />
21 MG 24<br />
HOURS 7 DAY<br />
Date:10/02/07ISR Number: 5476961-9Report Type:Direct Company Report #CTU 314199 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Paraesthesia<br />
<strong>Mefloquine</strong><br />
Hydrochloride 250<br />
Mg Sandoz, Inc. PS Sandoz, Inc. ORAL 1 TABLET
WEEKLY PO<br />
03-Apr-2012 09:37 AM Page: 768
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/02/07ISR Number: 5477024-9Report Type:Direct Company Report #CTU 314251 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Initial Insomnia Lariam Unknown Roche<br />
Initial or Prolonged Paralysis Pharmaceuticals PS ORAL ONCE PER WEEK<br />
Other Serious Feeling Abnormal PO<br />
Dyspnoea<br />
Abnormal Dreams<br />
Headache<br />
Paraesthesia<br />
Fear<br />
Immobile<br />
Disturbance In Attention<br />
Date:10/02/07ISR Number: 5477059-6Report Type:Direct Company Report #CTU 314240 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fear <strong>Mefloquine</strong> 250mg Hcl PS ORAL 252MG TWICE<br />
Nightmare<br />
PO<br />
Feeling Abnormal<br />
Date:10/05/07ISR Number: 5478471-1Report Type:Expedited (15-DaCompany Report #DK-ROCHE-521810 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Arrhythmia Lariam PS Roche UNKNOWN<br />
Date:10/05/07ISR Number: 5478472-3Report Type:Expedited (15-DaCompany Report #ES-ROCHE-519121 Age:56 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tumour Marker Test Lariam PS Roche ORAL<br />
Date:10/05/07ISR Number: 5478473-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-521263 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brachial Plexus Injury Lariam PS Roche ORAL<br />
Joint Dislocation Microgynon C ORAL<br />
Grand Mal Convulsion<br />
Date:10/05/07ISR Number: 5478475-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-510074 Age:32 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Nausea<br />
Tachycardia
Depression<br />
Vomiting<br />
Hypertension<br />
Disorientation<br />
Paraesthesia<br />
Hyperhidrosis<br />
Gastrointestinal Disorder<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 769
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tinnitus<br />
Social Phobia<br />
Headache Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL TOTAL OF<br />
THREE<br />
TABLETS.<br />
SECOND TABLET<br />
TAKEN IN<br />
Date:10/18/07ISR Number: 5490518-5Report Type:Direct Company Report #CTU 315637 Age:28 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination Lariam 250 Mg, Roche PS Roche ORAL 250 MG ONE<br />
Anxiety<br />
TAB Q WK PO<br />
Panic Attack Xanax C<br />
Date:10/23/07ISR Number: 5493377-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-525623 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Feeling Abnormal Lariam PS Roche UNKNOWN<br />
Initial or Prolonged<br />
Date:10/23/07ISR Number: 5494663-XReport Type:Direct Company Report #CTU 316116 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation <strong>Mefloquine</strong> PS 1 PER WEEK<br />
Fear<br />
Depression<br />
Date:10/23/07ISR Number: 5496173-2Report Type:Direct Company Report #CTU 316106 Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Sleep Disorder Lariam PS<br />
Panic Attack<br />
Decompression Sickness<br />
Memory Impairment<br />
Depression<br />
Insomnia<br />
Blindness<br />
Disturbance In Attention<br />
Malaise<br />
Unevaluable Event<br />
Vertigo<br />
Dreamy State<br />
Hypoaesthesia
Suicidal Ideation<br />
Thinking Abnormal<br />
Feeling Abnormal<br />
Confusional State<br />
Somnolence<br />
03-Apr-2012 09:37 AM Page: 770
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/24/07ISR Number: 5494720-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Anxiety<br />
Asthenia<br />
Chest Pain<br />
Generalised Anxiety<br />
Disorder<br />
Self-Injurious Ideation<br />
Mitral Valve<br />
Calcification<br />
Humerus Fracture<br />
Oxygen Saturation<br />
Decreased<br />
Rheumatoid Factor<br />
Increased<br />
Pyrexia<br />
Rash Papular<br />
Aggression<br />
Memory Impairment<br />
Vertigo<br />
Dyspnoea<br />
Cranial Nerve Disorder<br />
Asthma<br />
Homicidal Ideation<br />
Dehydration<br />
Convulsion<br />
Abnormal Behaviour<br />
Agitation<br />
Joint Dislocation<br />
Post-Traumatic Stress<br />
Disorder<br />
Tremor<br />
Cognitive Disorder<br />
Toxicity To Various<br />
Agents<br />
Contusion<br />
Antinuclear Antibody<br />
Positive<br />
Ventricular Extrasystoles<br />
Productive Cough<br />
Ill-Defined Disorder<br />
Psychotic Disorder<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Nocturia<br />
Mood Swings<br />
Fatigue<br />
Bronchial Hyperreactivity<br />
Headache<br />
Sleep Apnoea Syndrome<br />
Major Depression
Meniscus Lesion<br />
Violence-Related Symptom<br />
Craniocerebral Injury<br />
Bundle Branch Block Right<br />
Ligament Sprain<br />
Myalgia<br />
Eosinophilia<br />
Visual Impairment<br />
03-Apr-2012 09:37 AM Page: 771
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucination, Visual<br />
Chest Discomfort<br />
Decreased Activity Report Source Product Role Manufacturer Route Dose Duration<br />
Thinking Abnormal Lariam PS Roche ORAL<br />
Dysthymic Disorder Primaquine C<br />
Rhinitis Allergic Cipro C<br />
Insomnia<br />
Disturbance In Attention<br />
Depression<br />
Suicidal Ideation<br />
Selective Iga<br />
Immunodeficiency<br />
Hypertension<br />
Hyperhidrosis<br />
Eye Movement Disorder<br />
Balance Disorder<br />
Panic Disorder<br />
Fall<br />
Musculoskeletal Pain<br />
Mitral Valve Incompetence<br />
Exostosis<br />
Weight Decreased<br />
Respiratory Rate<br />
Increased<br />
Hypoaesthesia<br />
Nystagmus<br />
Date:10/25/07ISR Number: 5497070-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-525623 Age:39 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Feeling Abnormal Lariam PS Roche UNKNOWN<br />
Initial or Prolonged<br />
Date:10/25/07ISR Number: 5498506-XReport Type:Direct Company Report #CTU 316349 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Sleep Terror<br />
<strong>Mefloquine</strong> 250mg<br />
Depression<br />
Tabs Mepha<br />
Syncope Switzerland PS Mepha Switzerland ORAL ONE TABLET<br />
Insomnia<br />
WEEKLY TWO<br />
Vertigo<br />
YEARS PO<br />
Abnormal Dreams<br />
Primaquine 15 Mg<br />
Alopecia Tabs Pharm Chem SS Pharm Chem ORAL 2 TABLETS<br />
Confusional State<br />
DAILY 1X14<br />
DAYS PO<br />
Date:10/30/07ISR Number: 5503946-6Report Type:Direct Company Report #CTU 316659 Age:52 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Artequin -Artesimine<br />
Initial or Prolonged Asthenia 200+Meflouquin 600mg PS ORAL ONE<br />
Other Serious Palpitations ARTESIMINE+<br />
Flushing<br />
ONE<br />
Dizziness<br />
MEFLOUQUIN<br />
Hypoaesthesia<br />
DAILY PO<br />
03-Apr-2012 09:37 AM Page: 772
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/01/07ISR Number: 5504026-6Report Type:Expedited (15-DaCompany Report #FR-ROCHE-499416 Age:46 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maculopathy Lariam PS Roche UNKNOWN 6 MON<br />
Date:11/05/07ISR Number: 5506648-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Aggression<br />
Memory Impairment<br />
Mood Swings<br />
Ill-Defined Disorder<br />
Cranial Nerve Disorder<br />
Hypertension<br />
Cognitive Disorder<br />
Mitral Valve Incompetence<br />
Ligament Sprain<br />
Antinuclear Antibody<br />
Positive<br />
Rheumatoid Factor<br />
Increased<br />
Pyrexia<br />
Brain Stem Syndrome<br />
Anger<br />
Abdominal Distension<br />
Haematochezia<br />
Mood Altered<br />
Depression<br />
Balance Disorder<br />
Generalised Anxiety<br />
Disorder<br />
Meniscus Lesion<br />
Dysthymic Disorder<br />
Craniocerebral Injury<br />
Weight Decreased<br />
Convulsion<br />
Anxiety Disorder<br />
Loose Body In Joint<br />
Diarrhoea<br />
Abdominal Pain Upper<br />
Dyspnoea<br />
Headache<br />
Sleep Apnoea Syndrome<br />
Musculoskeletal Pain<br />
Bundle Branch Block Right<br />
Exostosis<br />
Nystagmus<br />
Contusion<br />
Blood Potassium Decreased<br />
Rash<br />
Merycism
Dyspepsia<br />
Eructation<br />
Abnormal Behaviour<br />
Visual Impairment<br />
Selective Iga<br />
Immunodeficiency<br />
Fatigue<br />
Chest Discomfort<br />
03-Apr-2012 09:37 AM Page: 773
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Chest Pain<br />
Impulsive Behaviour<br />
Acute Coronary Syndrome<br />
Upper Respiratory Tract<br />
Infection<br />
Vertigo<br />
Hallucination, Visual<br />
Asthenia<br />
Panic Disorder<br />
Fall<br />
Tremor<br />
Thinking Abnormal<br />
Toxicity To Various<br />
Agents<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Respiratory Rate<br />
Increased<br />
Eye Movement Disorder<br />
Nocturia<br />
Rash Papular<br />
Coordination Abnormal<br />
Wheezing<br />
Snoring<br />
Vestibular Disorder<br />
Deafness<br />
Hypervigilance<br />
Emotional Disorder<br />
Mucous Membrane Disorder<br />
Constipation<br />
Mucous Stools<br />
Nightmare<br />
Insomnia<br />
Agitation<br />
Anxiety<br />
Post-Traumatic Stress<br />
Disorder<br />
Asthma<br />
Humerus Fracture<br />
Dehydration<br />
Hypoaesthesia<br />
Myalgia<br />
Eosinophilia<br />
Ventricular Extrasystoles<br />
Injury<br />
Visual Field Defect<br />
Priapism<br />
No Therapeutic Response<br />
Tongue Disorder<br />
Dizziness<br />
Family Stress<br />
Nausea
Disturbance In Attention<br />
Bronchial Hyperreactivity<br />
Joint Dislocation<br />
Psychotic Disorder<br />
Self-Injurious Ideation<br />
Hyperhidrosis<br />
Homicidal Ideation<br />
Mitral Valve<br />
03-Apr-2012 09:37 AM Page: 774
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Calcification<br />
Rhinitis Allergic<br />
Palpitations<br />
Tinnitus Report Source Product Role Manufacturer Route Dose Duration<br />
Hydrocephalus Lariam PS Roche ORAL<br />
Suicidal Ideation Primaquine C<br />
Productive Cough Cipro C<br />
Major Depression<br />
Decreased Activity<br />
Oxygen Saturation<br />
Decreased<br />
Joint Range Of Motion<br />
Decreased<br />
Breath Odour<br />
Date:11/08/07ISR Number: 5512218-5Report Type:Direct Company Report #CTU 317413 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Panic Attack <strong>Mefloquine</strong> 250 Mg PS 1 TABLET ONCE<br />
Decreased Appetite A WEEK FOR 5<br />
Pyrexia<br />
ONE PILL PER<br />
Tremor WEEK 5 WK<br />
Feeling Abnormal<br />
Nervousness<br />
Sleep Disorder<br />
Influenza Like Illness<br />
Palpitations<br />
Date:11/14/07ISR Number: 5516163-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Anxiety Disorder<br />
Psychotic Disorder<br />
Insomnia<br />
Amnesia<br />
Vestibular Disorder<br />
Nystagmus<br />
Eye Pain<br />
Rash<br />
Mood Swings<br />
Tinnitus<br />
Disturbance In Attention<br />
Dizziness<br />
Irritable Bowel Syndrome<br />
Hypoaesthesia<br />
Night Sweats<br />
Vision Blurred<br />
Nervousness<br />
Pain Of Skin<br />
Blood Pressure Increased
Brain Stem Syndrome<br />
Vomiting<br />
Memory Impairment<br />
Vertigo<br />
Headache<br />
Phonophobia<br />
Paraesthesia<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 775
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorientation<br />
Chest Pain<br />
Agitation Report Source Product Role Manufacturer Route Dose Duration<br />
Diarrhoea Lariam PS Roche ORAL<br />
Nightmare Imodium C<br />
Fatigue Compazine C ON 18 APR<br />
Inner Ear Disorder<br />
2001, THERAPY<br />
Abdominal Pain<br />
REPORTED AS<br />
Testicular Pain<br />
TWICE DAILY.<br />
Depression Antibiotics Nos C<br />
Photophobia Ambien C<br />
Deafness Prednisone C BY TAPER.<br />
Valium C ON 18 APR<br />
2001, THERAPY<br />
REPORTED AS:<br />
AS NEEDED.<br />
Phenergan<br />
C<br />
Biaxin<br />
C<br />
Advil<br />
C<br />
Bactrim<br />
C<br />
Antivert<br />
C<br />
Date:11/15/07ISR Number: 5518385-1Report Type:Expedited (15-DaCompany Report #CA-ROCHE-472986 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nervousness Lariam PS Roche ORAL PATIENT TOOK<br />
Agitation<br />
ONLY ONE DOSE<br />
Hallucination Wellbutrin SS ORAL FORM: SR<br />
Anxiety<br />
TABLET<br />
Depersonalisation<br />
Date:11/15/07ISR Number: 5518594-1Report Type:Expedited (15-DaCompany Report #CA-ROCHE-529910 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypotonia <strong>Mefloquine</strong><br />
Convulsion Hydrochloride PS Roche UNKNOWN<br />
1 Drug Suspected SS INTRAMUSCULAR FREQUENCY<br />
REPORTED:<br />
FIRST DOSE 1 DAY<br />
Fansidar<br />
C<br />
Date:11/19/07ISR Number: 5521627-XReport Type:Direct Company Report #CTU 318163 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Chills Lariam PS<br />
Intervention to<br />
Diarrhoea<br />
Prevent Permanent Depression
Impairment/Damage<br />
Thinking Abnormal<br />
Dyspnoea<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 776
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/19/07ISR Number: 5525171-5Report Type:Direct Company Report #CTU 318215 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mental Disorder Larium 250 Mg Roche PS Roche ORAL 250 MG ONCE<br />
Adverse Drug Reaction<br />
PER WEEK PO<br />
Dizziness<br />
Paranoia<br />
Anxiety<br />
Date:11/20/07ISR Number: 5522092-9Report Type:Expedited (15-DaCompany Report #CA-ROCHE-529910 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion <strong>Mefloquine</strong><br />
Hypotonia Hydrochloride PS Roche UNKNOWN<br />
1 Drug Suspected SS INTRAMUSCULAR FREQUENCY<br />
REPORTED:<br />
FIRST DOSE 1 DAY<br />
Fansidar<br />
C<br />
Date:11/23/07ISR Number: 5524616-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Agitation<br />
Depression<br />
Bronchial Hyperreactivity<br />
Hypertension<br />
Psychotic Disorder<br />
Decreased Activity<br />
Dysthymic Disorder<br />
Nystagmus<br />
Rheumatoid Factor<br />
Increased<br />
Pyrexia<br />
Insomnia<br />
Fatigue<br />
Dyspnoea<br />
Fall<br />
Cognitive Disorder<br />
Bundle Branch Block Right<br />
Dehydration<br />
Hypoaesthesia<br />
Disturbance In Attention<br />
Aggression<br />
Ill-Defined Disorder<br />
Joint Dislocation<br />
Post-Traumatic Stress<br />
Disorder<br />
Violence-Related Symptom<br />
Eye Movement Disorder
Ligament Sprain<br />
Antinuclear Antibody<br />
Positive<br />
Rash Papular<br />
Abnormal Behaviour<br />
Selective Iga<br />
Immunodeficiency<br />
Asthenia<br />
03-Apr-2012 09:37 AM Page: 777
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Major Depression<br />
Generalised Anxiety<br />
Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Self-Injurious Ideation Lariam PS Roche ORAL<br />
Musculoskeletal Pain Primaquine C<br />
Hyperhidrosis Cipro C<br />
Mitral Valve Incompetence<br />
Oxygen Saturation<br />
Decreased<br />
Convulsion<br />
Balance Disorder<br />
Suicidal Ideation<br />
Productive Cough<br />
Headache<br />
Panic Disorder<br />
Tremor<br />
Thinking Abnormal<br />
Mitral Valve<br />
Calcification<br />
Exostosis<br />
Weight Decreased<br />
Myalgia<br />
Memory Impairment<br />
Visual Impairment<br />
Hallucination, Visual<br />
Asthma<br />
Homicidal Ideation<br />
Toxicity To Various<br />
Agents<br />
Humerus Fracture<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Respiratory Rate<br />
Increased<br />
Contusion<br />
Rhinitis Allergic<br />
Eosinophilia<br />
Mood Swings<br />
Chest Pain<br />
Cranial Nerve Disorder<br />
Nocturia<br />
Anxiety<br />
Vertigo<br />
Chest Discomfort<br />
Sleep Apnoea Syndrome<br />
Meniscus Lesion<br />
Craniocerebral Injury<br />
Ventricular Extrasystoles<br />
Date:11/26/07ISR Number: 5525274-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-525623 Age:39 YR Gender:Female I/FU:F
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Feeling Abnormal<br />
Dizziness<br />
Paraesthesia<br />
Sleep Disorder<br />
Headache<br />
Mental Disorder<br />
03-Apr-2012 09:37 AM Page: 778
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pain In Extremity<br />
Anxiety<br />
Paraesthesia Oral Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche UNKNOWN<br />
Date:11/28/07ISR Number: 5527945-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-529962 Age:40 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche UNKNOWN ON AN<br />
UNSPECIFIED<br />
DATE, THE<br />
PATIENT<br />
RECEIVED 4TH<br />
Date:12/03/07ISR Number: 5531862-2Report Type:Expedited (15-DaCompany Report #ZA-ROCHE-532847 Age:66 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Adverse Drug Reaction Lariam PS Roche ORAL<br />
Hospitalization - Interstitial Lung Disease<br />
Initial or Prolonged Respiratory Failure<br />
C-Reactive Protein<br />
Increased<br />
Overdose<br />
Laboratory Test Abnormal<br />
Date:12/04/07ISR Number: 5533285-9Report Type:Expedited (15-DaCompany Report #ZA-ROCHE-532847 Age:66 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Overdose Lariam PS Roche ORAL<br />
Hospitalization - Adverse Drug Reaction<br />
Initial or Prolonged Respiratory Failure<br />
Interstitial Lung Disease<br />
Date:12/10/07ISR Number: 5541933-2Report Type:Expedited (15-DaCompany Report #NL-ROCHE-505944 Age:4 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Status Epilepticus Lariam PS Roche ORAL<br />
Hospitalization -<br />
Initial or Prolonged<br />
Date:12/10/07ISR Number: 5542551-2Report Type:Expedited (15-DaCompany Report #TW-SANOFI-SYNTHELABO-A01200713585 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Other Serious Hypotension Chloroquine PS UNKNOWN UNK<br />
<strong>Mefloquine</strong> SS UNKNOWN UNK<br />
03-Apr-2012 09:37 AM Page: 779
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/12/07ISR Number: 5551451-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-534849 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia Lariam PS Roche ORAL DOSAGE<br />
Anxiety<br />
REGIMEN<br />
Dyspnoea REPORTED AS 1<br />
Congestive Cardiomyopathy<br />
DOSE PER WEEK<br />
Date:12/12/07ISR Number: 5564269-2Report Type:Expedited (15-DaCompany Report #GXKR2007GR10354 Age:67 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Malaise Foreign <strong>Mefloquine</strong><br />
Initial or Prolonged Eosinophilic Pneumonia Literature (Ngx)(<strong>Mefloquine</strong>)<br />
Blood Lactate Health Unknown PS ORAL 250 MG, QW,<br />
Dehydrogenase Increased Professional ORAL 84 DAY<br />
Pulmonary Toxicity<br />
Ketoconazole<br />
Hypoxia<br />
(Ngx)(Ketoconazole)<br />
Tachycardia Unknown SS TOPICAL TOPICAL<br />
Normochromic Normocytic<br />
Anaemia<br />
Date:12/14/07ISR Number: 5558149-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-535633 Age:67 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea Exertional Lariam PS Roche UNKNOWN<br />
Lung Infiltration<br />
Pyrexia<br />
Eosinophilic Pneumonia<br />
Eosinophilia<br />
Date:12/19/07ISR Number: 5566578-XReport Type:Expedited (15-DaCompany Report #GB-ROCHE-535633 Age:67 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eosinophilic Pneumonia Lariam PS Roche UNKNOWN<br />
Date:12/19/07ISR Number: 5571067-2Report Type:Expedited (15-DaCompany Report #021668 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cognitive Disorder Other <strong>Mefloquine</strong><br />
Mental Disorder<br />
Hydrochloride(Mefloq<br />
Pain<br />
uine Hydrochloride)<br />
Tablet, 250mg<br />
PS
Date:12/21/07ISR Number: 5571533-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-537148 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Eye Pain Lariam PS Roche UNKNOWN<br />
Nausea<br />
Paralysis<br />
Vertigo<br />
Headache<br />
03-Apr-2012 09:37 AM Page: 780
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/27/07ISR Number: 5574447-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-385293 Age:45 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
PT<br />
Disturbance In Attention<br />
Aggression<br />
Psychotic Disorder<br />
Major Depression<br />
Hyperhidrosis<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Ligament Sprain<br />
Depression<br />
Fatigue<br />
Productive Cough<br />
Chest Pain<br />
Sleep Apnoea Syndrome<br />
Joint Dislocation<br />
Mitral Valve Incompetence<br />
Convulsion<br />
Hypoaesthesia<br />
Contusion<br />
Rheumatoid Factor<br />
Increased<br />
Nocturia<br />
Abnormal Behaviour<br />
Mood Swings<br />
Visual Impairment<br />
Suicidal Ideation<br />
Bronchial Hyperreactivity<br />
Panic Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
Meniscus Lesion<br />
Cognitive Disorder<br />
Homicidal Ideation<br />
Dysthymic Disorder<br />
Toxicity To Various<br />
Agents<br />
Humerus Fracture<br />
Dehydration<br />
Respiratory Rate<br />
Increased<br />
Antinuclear Antibody<br />
Positive<br />
Myalgia<br />
Hallucination, Visual<br />
Balance Disorder<br />
Selective Iga<br />
Immunodeficiency<br />
Chest Discomfort<br />
Asthma<br />
Rhinitis Allergic
Pyrexia<br />
Violence-Related Symptom<br />
Fall<br />
Exostosis<br />
Ventricular Extrasystoles<br />
Dyspnoea<br />
Tremor<br />
Thinking Abnormal<br />
03-Apr-2012 09:37 AM Page: 781
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Oxygen Saturation<br />
Decreased<br />
Rash Papular Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia Lariam PS Roche ORAL<br />
Agitation Primaquine C<br />
Anxiety Cipro C<br />
Memory Impairment<br />
Vertigo<br />
Asthenia<br />
Generalised Anxiety<br />
Disorder<br />
Musculoskeletal Pain<br />
Craniocerebral Injury<br />
Mitral Valve<br />
Calcification<br />
Eosinophilia<br />
Ill-Defined Disorder<br />
Headache<br />
Cranial Nerve Disorder<br />
Hypertension<br />
Self-Injurious Ideation<br />
Decreased Activity<br />
Bundle Branch Block Right<br />
Weight Decreased<br />
Nystagmus<br />
Eye Movement Disorder<br />
Date:01/03/08ISR Number: 5581533-1Report Type:Direct Company Report #CTU 321532 Age:39 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hyperventilation Larium ? Hoffman La<br />
Initial or Prolonged Anxiety Roche PS Hoffman La Roche ORAL 1 PER WEEK PO<br />
Other Serious<br />
Thinking Abnormal<br />
Required<br />
Toxicity To Various<br />
Intervention to<br />
Agents<br />
Prevent Permanent<br />
Impairment/Damage<br />
Poor Quality Sleep<br />
Stress<br />
Dehydration<br />
Panic Attack<br />
Initial Insomnia<br />
Chest Discomfort<br />
Heart Rate Increased<br />
Fear<br />
Palpitations<br />
Dyspnoea<br />
Impaired Work Ability<br />
Middle Insomnia<br />
Alcohol Use
Date:01/11/08ISR Number: 5588505-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-415971 Age:61 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Amyotrophy Lariam PS Roche ORAL<br />
Upper Motor Neurone Engerix B SS INTRAMUSCULAR FORM:<br />
Lesion<br />
INHALATION<br />
SOLUTION, THE<br />
PATIENT<br />
03-Apr-2012 09:37 AM Page: 782
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
RECEIVED<br />
Date:01/14/08ISR Number: 5592903-XReport Type:Direct Company Report #CTU 322500 Age:36 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Feeling Abnormal Lariam PS ORAL ONCE PER WEEK<br />
Fear<br />
PO<br />
Psychotic Disorder<br />
Anxiety<br />
Date:01/14/08ISR Number: 5593124-7Report Type:Direct Company Report #CTU 322421 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Road Traffic Accident <strong>Mefloquine</strong> PS ORAL 250MG QWEEK<br />
Vertigo Positional<br />
PO<br />
Date:01/15/08ISR Number: 5593239-3Report Type:Direct Company Report #CTU 322557 Age:38 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mood Swings Lariam PS ORAL 1/WEEK PO<br />
Panic Disorder<br />
Anxiety<br />
Toxicity To Various<br />
Agents<br />
Fear<br />
Date:01/16/08ISR Number: 5593525-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-535633 Age:67 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Eosinophilic Pneumonia <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL 84 DAY<br />
Ketoconazole SS TOPICAL SHAMPOO<br />
Ketoconazole SS TOPICAL SHAMPOO<br />
Date:01/22/08ISR Number: 5601165-6Report Type:Direct Company Report #CTU 322950 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Lariam PS<br />
Required<br />
Mental Disorder<br />
Intervention to<br />
Insomnia<br />
Prevent Permanent Mood Altered<br />
Impairment/Damage Anxiety<br />
Migraine<br />
Visual Impairment
Date:01/29/08ISR Number: 5605775-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-537148 Age: Gender:Male I/FU:F<br />
Outcome<br />
Other Serious<br />
PT<br />
Headache<br />
Vertigo<br />
03-Apr-2012 09:37 AM Page: 783
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Nausea<br />
Eye Pain<br />
Paralysis Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche UNKNOWN<br />
Date:01/30/08ISR Number: 5609148-7Report Type:Direct Company Report #CTU 323900 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Myalgia <strong>Mefloquine</strong> 250mg PS ONCE WEEKLY<br />
Hypoaesthesia<br />
Dizziness<br />
Impaired Work Ability<br />
Arthralgia<br />
Date:02/05/08ISR Number: 5618668-0Report Type:Direct Company Report #CTU 324564 Age:42 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tinnitus <strong>Mefloquine</strong> Hcl 250mg<br />
Initial or Prolonged Insomnia Sandoz PS Sandoz ORAL 1 TABLET ONCE<br />
Anxiety<br />
A WEEK PO<br />
Palpitations<br />
Sinus Tachycardia<br />
Date:02/07/08ISR Number: 5618791-0Report Type:Expedited (15-DaCompany Report #AU-ROCHE-543953 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Depression<br />
Mania<br />
Date:02/07/08ISR Number: 5618980-5Report Type:Expedited (15-DaCompany Report #GR-BOEHRINGER INGELHEIM GMBH, GERMANY-2008-BP-01668RAge:67 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia <strong>Mefloquine</strong> PS Boehringer Ingelheim<br />
Initial or Prolonged Eosinophilic Pneumonia Gmbh, Germany ORAL 12 WK<br />
Normochromic Normocytic Ketoconazole SS TOPICAL<br />
Anaemia<br />
Tachypnoea<br />
Red Blood Cell<br />
Sedimentation Rate<br />
Increased<br />
Blood Lactate<br />
Dehydrogenase Increased<br />
Eosinophilia<br />
Tachycardia
C-Reactive Protein<br />
Increased<br />
Hypoxia<br />
03-Apr-2012 09:37 AM Page: 784
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/11/08ISR Number: 5622046-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Tinnitus Lariam PS Roche ORAL<br />
Other Serious Testicular Pain Imodium C<br />
Photophobia Compazine C ON 18 APR<br />
Night Sweats<br />
2001, THERAPY<br />
Mood Swings<br />
REPORTED AS<br />
Fatigue<br />
TWICE DAILY.<br />
Chest Pain Antibiotics Nos C<br />
Vision Blurred Ambien C<br />
Disturbance In Attention Prednisone C BY TAPER.<br />
Diarrhoea Valium C ON 18 APR<br />
Diabetes Mellitus<br />
2001, THERAPY<br />
Deafness<br />
REPORTED AS:<br />
Disorientation<br />
AS NEEDED.<br />
Vestibular Disorder Phenergan C<br />
Irritable Bowel Syndrome Biaxin C<br />
Eye Pain Advil C<br />
Hypoaesthesia Bactrim C<br />
Vertigo Antivert C<br />
Psychotic Disorder<br />
Nystagmus<br />
Blood Triglycerides<br />
Increased<br />
Agitation<br />
Nausea<br />
Insomnia<br />
Nightmare<br />
Amnesia<br />
Headache<br />
Abdominal Pain<br />
Dizziness<br />
Depression<br />
Blood Pressure Increased<br />
Memory Impairment<br />
Inner Ear Disorder<br />
Rash<br />
Nervousness<br />
Phonophobia<br />
Paraesthesia<br />
Brain Stem Syndrome<br />
Vomiting<br />
Anxiety Disorder<br />
Pain Of Skin<br />
Date:02/19/08ISR Number: 5630886-4Report Type:Expedited (15-DaCompany Report #CH-ROCHE-435016 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Cerebral Ventricle Lariam PS Roche TRANSPLACENTAL INDICATION<br />
Other Serious Dilatation DRUG ACTING
Premature Baby ON OTHER<br />
Foetal Growth Restriction<br />
BLOOD<br />
Maternal Exposure During PROTOZOA 15 DAY<br />
Pregnancy Eltroxin C<br />
Stilnox C REPORTED<br />
FREQUENCY 2<br />
TOTAL. 3 DAY<br />
03-Apr-2012 09:37 AM Page: 785
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/19/08ISR Number: 5636737-6Report Type:Direct Company Report #CTU 326092 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Lariam 250 Mg<br />
Deafness Unilateral Roche PS Roche ORAL 250 MG ONCE<br />
Tinnitus<br />
WEEKLY PO<br />
Crying<br />
(ONE DOSE)<br />
Klonopin<br />
C<br />
Wellbutrin<br />
C<br />
Lamictal<br />
C<br />
Date:02/25/08ISR Number: 5637134-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-548290 Age:30 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Liver Function Test Lariam PS Roche ORAL<br />
Abnormal Imodium SS ORAL<br />
Suicidal Ideation Imodium SS ORAL<br />
Overdose Talvosilen SS ORAL<br />
Somnolence Gelonida SS ORAL<br />
Ibuprofen SS Roche ORAL<br />
Ibuprofen SS Roche ORAL<br />
Talvosilen SS ORAL<br />
Lariam SS Roche ORAL<br />
Gelonida SS ORAL<br />
Date:02/25/08ISR Number: 5637136-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548278 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Homicide Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Completed Suicide<br />
Date:02/25/08ISR Number: 5637320-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548228 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Suicide Attempt<br />
Date:02/25/08ISR Number: 5637331-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548255 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cardiovascular Lariam PS Roche ORAL<br />
Insufficiency Doxam C<br />
Depression<br />
Suicidal Ideation
Date:03/07/08ISR Number: 5655519-2Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548290 Age:30 YR Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Somnolence<br />
Liver Function Test<br />
Abnormal<br />
03-Apr-2012 09:37 AM Page: 786
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Suicidal Ideation<br />
Overdose<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Talvosilen SS ORAL DRUG REPORTED<br />
AS "PANADEINE<br />
CO"<br />
Ibuprofen SS Roche ORAL<br />
Imodium SS ORAL DRUG REPORTED<br />
AS<br />
"LOPERAMIDE<br />
HYDROCHLORIDE<br />
"<br />
Gelonida SS ORAL DRUG REPORTED<br />
AS "PANADEINE<br />
CO"<br />
Date:03/07/08ISR Number: 5655523-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548255 Age:55 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Lariam PS Roche ORAL<br />
Depression Doxam C<br />
Cardiovascular<br />
Insufficiency<br />
Date:03/11/08ISR Number: 5659980-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-550654 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Amyotrophic Lateral Lariam PS Roche ORAL DOSE<br />
Initial or Prolonged Sclerosis REPORTED: 1<br />
COURSE<br />
Date:03/12/08ISR Number: 5661506-0Report Type:Expedited (15-DaCompany Report #IT-ROCHE-550893 Age:73 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Abdominal Distension Lariam PS Roche ORAL<br />
Initial or Prolonged Dyspnoea<br />
Oedema Peripheral<br />
Date:03/12/08ISR Number: 5661510-2Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548228 Age: Gender: I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Suicide Attempt
Date:03/13/08ISR Number: 5662678-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-548278 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Homicide Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Completed Suicide<br />
03-Apr-2012 09:37 AM Page: 787
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/21/08ISR Number: 5676118-2Report Type:Expedited (15-DaCompany Report #CA-ROCHE-940804124001 Age:23 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hallucination Lariam PS Roche ORAL<br />
Life-Threatening Completed Suicide Alcohol C ORAL<br />
Date:03/28/08ISR Number: 5685018-3Report Type:Expedited (15-DaCompany Report #CH-ROCHE-84542 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Nasopharyngitis Lariam Tablets PS Roche ORAL<br />
Death Flunitrazepam C UNKNOWN<br />
Neo-Citran C ORAL<br />
Date:03/28/08ISR Number: 5685028-6Report Type:Expedited (15-DaCompany Report #GB-ROCHE-94161 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Lariam Tablets PS Roche ORAL<br />
Indapamide SS UNKNOWN<br />
Date:03/28/08ISR Number: 5685058-4Report Type:Expedited (15-DaCompany Report #NL-ROCHE-104818 Age:67 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Malaise Lariam PS Roche ORAL<br />
Death<br />
Nausea<br />
Date:03/31/08ISR Number: 5688792-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-550654 Age:62 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Amyotrophic Lateral Lariam PS Roche ORAL DOSE<br />
Hospitalization - Sclerosis REPORTED: 1<br />
Initial or Prolonged<br />
COURSE<br />
(COMMON)<br />
Date:03/31/08ISR Number: 5688827-XReport Type:Expedited (15-DaCompany Report #FR-ROCHE-305523 Age:68 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Lariam PS Roche ORAL<br />
Mental Disorder Lodoz SS ORAL<br />
Date:03/31/08ISR Number: 5689149-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-940400336001 Age:27 YR Gender:Male I/FU:I
Outcome<br />
Death<br />
Other Serious<br />
PT<br />
Drug Interaction<br />
Foot Fracture<br />
Death<br />
Epilepsy<br />
Depression<br />
Accidental Death<br />
03-Apr-2012 09:37 AM Page: 788
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Drowning<br />
Abnormal Behaviour<br />
Psychotic Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination Lariam PS Roche ORAL<br />
Carbamazepine C ORAL<br />
Valproate I ORAL<br />
Date:04/01/08ISR Number: 5690471-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-354441 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Lariam PS Roche ORAL<br />
Other Serious<br />
Arrhythmia<br />
Date:04/01/08ISR Number: 5693443-XReport Type:Direct Company Report #CTU 330870 Age:58 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Larium 250 Mg Tablet PS ORAL 250 MG ONCE<br />
Initial or Prolonged Chest Pain WEEKLY ORAL<br />
Flight Of Ideas<br />
Delusion<br />
Agitation<br />
Psychotic Disorder<br />
Confusional State<br />
Altered State Of<br />
Consciousness<br />
Feeling Abnormal<br />
Claustrophobia<br />
Date:04/03/08ISR Number: 5694409-6Report Type:Direct Company Report #CTU 331148 Age:64 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia <strong>Mefloquine</strong> 250 Mg PS 1 TABLET 1<br />
Dry Mouth<br />
PER WEEK<br />
Asthenia<br />
Blood Cholesterol<br />
Increased<br />
Balance Disorder<br />
Hypotension<br />
Product Quality Issue<br />
Dizziness<br />
Date:04/04/08ISR Number: 5694932-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-531067 Age: Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
PT<br />
Stress At Work
Anxiety<br />
Tachycardia<br />
Sensory Disturbance<br />
Compulsions<br />
Heart Rate Irregular<br />
Depression<br />
Psychosomatic Disease<br />
Irritability<br />
03-Apr-2012 09:37 AM Page: 789
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Muscle Twitching<br />
Panic Attack<br />
Restlessness Report Source Product Role Manufacturer Route Dose Duration<br />
Nervous System Disorder Lariam PS Roche ORAL<br />
Self Esteem Decreased<br />
Affect Lability<br />
Cardiovascular Disorder<br />
Tremor<br />
Sleep Disorder<br />
Personality Change<br />
Tachyarrhythmia<br />
Date:04/07/08ISR Number: 5696150-2Report Type:Expedited (15-DaCompany Report #DE-ROCHE-555182 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Atrial Septal Defect Lariam PS Roche ORAL 3 MON<br />
Date:04/07/08ISR Number: 5699102-1Report Type:Direct Company Report #CTU 331512 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia <strong>Mefloquine</strong> Hcl 250<br />
Other Serious Anxiety Mg Sandoz PS Sandoz ORAL 250 MG 1 A<br />
Balance Disorder<br />
WEEK PO<br />
Hallucination<br />
Paranoia<br />
Nightmare<br />
Dizziness<br />
Suicidal Ideation<br />
Date:04/10/08ISR Number: 5700060-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-550654 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Amyotrophic Lateral Lariam PS Roche ORAL DOSE<br />
Hospitalization - Sclerosis REPORTED: 1<br />
Initial or Prolonged<br />
COURSE<br />
(COMMON)<br />
Date:04/10/08ISR Number: 5702450-XReport Type:Direct Company Report #CTU 331896 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Psychiatric Symptom Lariam 250 Mg PS BUCCAL 8 WEEKLY<br />
Intervention to Neurological Symptom BUCCAL 8 WK<br />
Prevent Permanent<br />
Impairment/Damage
Date:04/18/08ISR Number: 5709774-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age: Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Other Serious<br />
PT<br />
Abnormal Dreams<br />
Vertigo<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 790
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Headache<br />
Fatigue<br />
Tinnitus Report Source Product Role Manufacturer Route Dose Duration<br />
Tachycardia Lariam PS Roche UNKNOWN DOSAGE<br />
Dyspepsia<br />
REGIMEN<br />
Formication<br />
REPORTED AS<br />
Oedema Peripheral<br />
FOUR TABS IN<br />
Unevaluable Event<br />
TOTAL. WEEKLY<br />
Malaise<br />
Paraesthesia<br />
Sleep Disorder<br />
Disturbance In Attention<br />
Anxiety<br />
Pyrexia<br />
Coordination Abnormal<br />
Muscle Twitching<br />
Cardiovascular Disorder<br />
Upper Respiratory Tract<br />
Infection<br />
Agitation<br />
Asthenia<br />
Feeling Abnormal<br />
Mental Disorder<br />
Night Sweats<br />
Date:04/22/08ISR Number: 5712570-1Report Type:Expedited (15-DaCompany Report #GB-AVENTIS-200811608EU Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Indapamide PS Aventis<br />
Pharmaceuticals Inc. ORAL<br />
dose: UNK<br />
Lariam<br />
/00813001/ SS ORAL dose: UNK<br />
Date:04/22/08ISR Number: 5716351-4Report Type:Expedited (15-DaCompany Report #2008S1005710 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Foreign Indapamide<br />
Health (Indapamide) PS<br />
Professional Lariam /00813001/<br />
Other (<strong>Mefloquine</strong>) SS ORAL ORAL<br />
Date:04/25/08ISR Number: 5717904-XReport Type:Expedited (15-DaCompany Report #GB-ABBOTT-98P-167-0078100-00 Age:27 YR Gender:Male I/FU:I<br />
Outcome<br />
Death<br />
PT<br />
Depression<br />
Social Avoidant Behaviour
Foot Fracture<br />
Psychotic Disorder<br />
Fatigue<br />
Abnormal Behaviour<br />
Epilepsy<br />
Drowning<br />
Drug Level Below<br />
Therapeutic<br />
03-Apr-2012 09:37 AM Page: 791
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Drug Interaction<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Valproate Sodium PS ORAL<br />
Valproate Sodium SS ORAL Dose<br />
increased<br />
Valproate Sodium SS ORAL 600 mg daily<br />
Valproate Sodium SS<br />
Carbamazepine C ORAL<br />
Carbamazepine C ORAL Dose tapered<br />
Carbamazepine C ORAL Dose tapered<br />
<strong>Mefloquine</strong> I ORAL 250 mg weekly<br />
Date:04/29/08ISR Number: 5721785-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-560669 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypomania Lariam PS Roche ORAL<br />
Initial or Prolonged Abnormal Behaviour Fortzaar C ORAL<br />
Fortzaar C ORAL<br />
Date:04/30/08ISR Number: 5724928-5Report Type:Direct Company Report #CTU 334288 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Liver Disorder <strong>Mefloquine</strong> PS<br />
Initial or Prolonged<br />
Date:05/05/08ISR Number: 5731287-0Report Type:Expedited (15-DaCompany Report #2008S1005710 Age:60 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Death Health Indapamide<br />
Professional (Indapamide) PS<br />
Other Lariam /00813001/<br />
(<strong>Mefloquine</strong>) SS ORAL ORAL<br />
Date:05/07/08ISR Number: 5730460-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-561917 Age:42 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rhabdomyolysis Lariam PS Roche ORAL<br />
Initial or Prolonged Pancreatitis Acute Xanax C<br />
Areflexia<br />
Pericarditis<br />
Date:05/08/08ISR Number: 5731765-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age: Gender:Male I/FU:F
Outcome<br />
Disability<br />
Other Serious<br />
PT<br />
Nausea<br />
Insomnia<br />
Irritable Bowel Syndrome<br />
Nervousness<br />
Blood Pressure Increased<br />
Disorientation<br />
03-Apr-2012 09:37 AM Page: 792
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Amnesia<br />
Nystagmus<br />
Blood Triglycerides Report Source Product Role Manufacturer Route Dose Duration<br />
Increased Lariam PS Roche ORAL<br />
Toxicity To Various Bactrim C<br />
Agents Imodium C<br />
Agitation Compazine C ON 18 APR<br />
Anxiety Disorder<br />
2001, THERAPY<br />
Eye Pain<br />
REPORTED AS<br />
Rash<br />
TWICE DAILY.<br />
Memory Impairment Antibiotics Nos C<br />
Headache Ambien C<br />
Vestibular Disorder Prednisone C BY TAPER.<br />
Photophobia Valium C ON 18 APR<br />
Paraesthesia<br />
2001, THERAPY<br />
Fatigue<br />
REPORTED AS:<br />
Tinnitus<br />
AS NEEDED.<br />
Inner Ear Disorder Phenergan C<br />
Dizziness Biaxin C<br />
Disturbance In Attention Advil C<br />
Hypoaesthesia Bactrim C<br />
Phonophobia Antivert C<br />
Brain Stem Syndrome<br />
Diarrhoea<br />
Night Sweats<br />
Mood Swings<br />
Vertigo<br />
Chest Pain<br />
Psychotic Disorder<br />
Testicular Pain<br />
Depression<br />
Vomiting<br />
Nightmare<br />
Diabetes Mellitus<br />
Abdominal Pain<br />
Vision Blurred<br />
Pain Of Skin<br />
Deafness<br />
Date:05/12/08ISR Number: 5735434-6Report Type:Expedited (15-DaCompany Report #SE-ROCHE-560756 Age:69 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious International Normalised Lariam PS Roche ORAL THERAPY START<br />
Ratio Increased<br />
AND STOP DATE<br />
REPORTED AS<br />
2008, DOSAGE<br />
REGIMEN: DD<br />
Waran SS ORAL DOSAGE<br />
REGIMEN<br />
REPORTED AS D<br />
D
Date:05/14/08ISR Number: 5737157-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-367621 Age: Gender:Male I/FU:F<br />
Outcome<br />
Disability<br />
Other Serious<br />
PT<br />
Vertigo<br />
Blood Triglycerides<br />
03-Apr-2012 09:37 AM Page: 793
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Increased<br />
Vision Blurred<br />
Rash<br />
Nausea Report Source Product Role Manufacturer Route Dose Duration<br />
Mood Swings Lariam PS Roche ORAL<br />
Insomnia Bactrim C<br />
Amnesia Imodium C<br />
Depression Compazine C ON 18 APR<br />
Blood Pressure Increased<br />
2001, THERAPY<br />
Paraesthesia<br />
REPORTED AS<br />
Diarrhoea<br />
TWICE DAILY.<br />
Vestibular Disorder Antibiotics Nos C<br />
Psychotic Disorder Ambien C<br />
Diabetes Mellitus Prednisone C BY TAPER.<br />
Nervousness Valium C ON 18 APR<br />
Fatigue<br />
2001, THERAPY<br />
Photophobia<br />
REPORTED AS:<br />
Pain Of Skin<br />
AS NEEDED.<br />
Toxicity To Various Phenergan C<br />
Agents Biaxin C<br />
Vomiting Advil C<br />
Nightmare Bactrim C<br />
Testicular Pain Antivert C<br />
Dizziness<br />
Hypoaesthesia<br />
Phonophobia<br />
Anxiety Disorder<br />
Disorientation<br />
Chest Pain<br />
Nystagmus<br />
Inner Ear Disorder<br />
Abdominal Pain<br />
Irritable Bowel Syndrome<br />
Disturbance In Attention<br />
Eye Pain<br />
Night Sweats<br />
Agitation<br />
Headache<br />
Tinnitus<br />
Deafness<br />
Brain Stem Syndrome<br />
Memory Impairment<br />
Date:05/14/08ISR Number: 5742047-9Report Type:Direct Company Report #CTU 336553 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Vertigo Lariam -Mefloquin- PS ORAL 1 PILL ONCE A<br />
Initial or Prolonged Nausea WEEK PO<br />
Required<br />
Hallucination<br />
Intervention to<br />
Headache<br />
Prevent Permanent<br />
Impairment/Damage
Date:05/29/08ISR Number: 5752217-1Report Type:Expedited (15-DaCompany Report #NL-ROCHE-566208 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Respiratory Distress Lariam PS Roche ORAL<br />
Initial or Prolonged Cyanosis<br />
03-Apr-2012 09:37 AM Page: 794
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/02/08ISR Number: 5755389-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-566032 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Poisoning Lariam PS Roche UNKNOWN<br />
Sexual Abuse<br />
Unknown<br />
Benzodiazepines SS UNKNOWN<br />
Date:06/02/08ISR Number: 5758017-0Report Type:Direct Company Report #CTU 338594 Age:55 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Weight Decreased Lariam 250 Mg<br />
Aggression Roche PS Roche ORAL 250 MG<br />
Insomnia<br />
WEEKLY PO<br />
Date:06/03/08ISR Number: 5757295-1Report Type:Expedited (15-DaCompany Report #ZA-ROCHE-566339 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatic Steatosis <strong>Mefloquine</strong><br />
Hydrochloride PS Roche UNKNOWN<br />
Date:06/03/08ISR Number: 5757344-0Report Type:Expedited (15-DaCompany Report #SE-ROCHE-566343 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nightmare Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Hallucination, Visual<br />
Other Serious<br />
Psychotic Disorder<br />
Incoherent<br />
Date:06/04/08ISR Number: 5758918-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-564714 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disturbance In Attention Lariam PS Roche ORAL<br />
Herpes Zoster Lariam C DOSING<br />
Dizziness<br />
FREQUENCY<br />
Memory Impairment REPORTED AS 6<br />
Altered Visual Depth TABLETS IN 2<br />
Perception<br />
DAYS.<br />
Balance Disorder<br />
Colour Blindness<br />
Date:06/04/08ISR Number: 5765655-8Report Type:Periodic Company Report #493511 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Death Suicide Attempt Other Lariam (<strong>Mefloquine</strong><br />
Completed Suicide Hydrochloride) PS<br />
Suicidal Ideation<br />
03-Apr-2012 09:37 AM Page: 795
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/04/08ISR Number: 5765660-1Report Type:Periodic Company Report #512701 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Consumer Lariam (<strong>Mefloquine</strong><br />
Company Hydrochloride) PS<br />
Representative<br />
Other<br />
Date:06/04/08ISR Number: 5765665-0Report Type:Periodic Company Report #534035 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Consumer Lariam (<strong>Mefloquine</strong><br />
Disturbance In Attention Other Hydrochloride) PS<br />
Panic Attack<br />
Anxiety<br />
Abnormal Dreams<br />
Aggression<br />
Depression<br />
Date:06/04/08ISR Number: 5765670-4Report Type:Periodic Company Report #543330 Age:82 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Medication Error Literature Lariam (<strong>Mefloquine</strong><br />
Overdose Health Hydrochloride) PS 250 MG DAILY 61 DAY<br />
Agitation<br />
Professional<br />
Confusional State<br />
Other<br />
Ataxia<br />
Dizziness<br />
Speech Disorder<br />
Deafness<br />
Date:06/04/08ISR Number: 5765673-XReport Type:Periodic Company Report #543771 Age:53 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nausea Literature Lariam (<strong>Mefloquine</strong><br />
Paraesthesia Health Hydrochloride) PS 250 MG DAILY<br />
Overdose<br />
Professional<br />
Asthenia<br />
Other<br />
Depression<br />
Medication Error<br />
Disorientation<br />
Fatigue<br />
Date:06/04/08ISR Number: 5765685-6Report Type:Periodic Company Report #543808 Age: Gender: I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mental Disorder Consumer Lariam (<strong>Mefloquine</strong><br />
Other Hydrochloride) PS<br />
03-Apr-2012 09:37 AM Page: 796
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/11/08ISR Number: 5767227-8Report Type:Direct Company Report #CTU 339638 Age:25 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams <strong>Mefloquine</strong> Malaris E PS<br />
Nightmare<br />
Date:06/12/08ISR Number: 5766736-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-569094 Age:48 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Delusion<br />
Date:06/13/08ISR Number: 5773271-7Report Type:Direct Company Report #CTU 339922 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Confusional State Lariam Hydrochloride<br />
Intervention to Hallucination Hoffman-Laroche<br />
Prevent Permanent Adverse Drug Reaction -Roche- PS Hoffman-Laroche<br />
Impairment/Damage Psychotic Disorder -Roche- ORAL WEEKLY PO<br />
Mania<br />
Depression<br />
Thinking Abnormal<br />
Suicidal Ideation<br />
Date:06/17/08ISR Number: 5775289-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-569005 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rhabdomyolysis Lariam PS Roche ORAL<br />
Initial or Prolonged Xanax C<br />
Date:06/17/08ISR Number: 5776506-XReport Type:Direct Company Report #CTU 340311 Age:46 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicidal Ideation Larium PS ONE TABLET<br />
Hospitalization - Mental Disorder WEEKLY<br />
Initial or Prolonged Panic Attack<br />
Date:06/18/08ISR Number: 5778564-5Report Type:Direct Company Report #CTU 340511 Age:21 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Other Serious<br />
Required<br />
PT<br />
Anxiety<br />
Insomnia<br />
Paranoia
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Attention<br />
Deficit/Hyperactivity<br />
Disorder<br />
Anger<br />
Fatigue<br />
Mood Swings<br />
Suicidal Ideation<br />
Aggression<br />
03-Apr-2012 09:37 AM Page: 797
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Phobia<br />
Nervous System Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Brain Injury Lariam Roche PS Roche ORAL 1 PILL ONCE<br />
PO<br />
Date:06/23/08ISR Number: 5781458-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-569094 Age:48 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Delusion Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Completed Suicide<br />
Date:06/23/08ISR Number: 5786900-9Report Type:Direct Company Report #CTU 340905 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia <strong>Mefloquine</strong> Taken<br />
Suicidal Ideation Once A Wk X 2 Doses PS ORAL 250MG WEEKLY<br />
Abnormal Dreams<br />
PO<br />
Anxiety Albendazole C<br />
Cipro<br />
C<br />
Date:06/30/08ISR Number: 5794845-3Report Type:Expedited (15-DaCompany Report #SE-ROCHE-566343 Age:27 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Incoherent Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Nightmare Malarone C<br />
Other Serious Psychotic Disorder Malarone C<br />
Hallucination, Visual<br />
Date:07/02/08ISR Number: 5798235-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-570689 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Ineffective Lariam PS Roche UNKNOWN<br />
Date:07/03/08ISR Number: 5800412-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-570689 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Ineffective Lariam PS Roche UNKNOWN<br />
Date:07/07/08ISR Number: 5801506-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-572343 Age: Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cognitive Disorder Lariam PS Roche UNKNOWN<br />
03-Apr-2012 09:37 AM Page: 798
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/08/08ISR Number: 5802953-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-573267 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pulmonary Oedema Lariam PS Roche ORAL<br />
Pulmonary Fibrosis Lariam SS Roche ORAL<br />
Pneumonia<br />
Dyspnoea<br />
Date:07/10/08ISR Number: 5806523-2Report Type:Expedited (15-DaCompany Report #US-BOEHRINGER INGELHEIM GMBH, GERMANY-2008-BP-10301RAge:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - White Blood Cell Count <strong>Mefloquine</strong> PS Boehringer Ingelheim<br />
Initial or Prolonged Increased Gmbh, Germany 5 WK<br />
Antipsychotic Drug Level Eskalith Cr SS<br />
Increased Eskalith Cr SS<br />
Mania Lunesta SS<br />
Quetiapine<br />
C<br />
Quetiapine<br />
C<br />
Date:07/14/08ISR Number: 5809717-5Report Type:Direct Company Report #CTU 343343 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety Larium La Roche PS La Roche ORAL 1 TABLET<br />
Muscle Twitching<br />
WEEKLY PO<br />
Malaise<br />
Pain<br />
Depression<br />
Nerve Injury<br />
Date:07/15/08ISR Number: 5810454-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-531067 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Depression Lariam PS Roche ORAL<br />
Tachycardia<br />
Affect Lability<br />
Stress At Work<br />
Anxiety<br />
Restlessness<br />
Nervous System Disorder<br />
Panic Attack<br />
Muscle Twitching<br />
Cardiovascular Disorder<br />
Tremor<br />
Sleep Disorder<br />
Compulsions<br />
Irritability<br />
Heart Rate Irregular
Psychosomatic Disease<br />
Sensory Disturbance<br />
Self Esteem Decreased<br />
Personality Change<br />
Tachyarrhythmia<br />
03-Apr-2012 09:37 AM Page: 799
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/21/08ISR Number: 5816593-3Report Type:Expedited (15-DaCompany Report #NL-ROCHE-575244 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Renal Failure Lariam PS Roche ORAL<br />
Hepatic Failure<br />
Date:07/22/08ISR Number: 5821277-1Report Type:Direct Company Report #CTU 344228 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Activities Of Daily Lariam Roche PS Roche THREE PILLS<br />
Living Impaired<br />
Depression<br />
Anxiety<br />
Insomnia<br />
Disorientation<br />
Crying<br />
Hallucination<br />
Date:07/23/08ISR Number: 5822351-6Report Type:Direct Company Report #CTU 344345 Age:33 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Grand Mal Convulsion <strong>Mefloquine</strong> 250mg PS<br />
Hospitalization - Upper Limb Fracture<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:07/25/08ISR Number: 5823376-7Report Type:Expedited (15-DaCompany Report #DE-ROCHE-314664 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Lariam PS Roche ORAL<br />
Other Serious<br />
Job Dissatisfaction<br />
Depression<br />
Restlessness<br />
Nervous System Disorder<br />
Tremor<br />
Panic Attack<br />
Affect Lability<br />
Palpitations<br />
Sleep Disorder<br />
Compulsions<br />
Irritability<br />
Sensory Disturbance<br />
Self Esteem Decreased<br />
Peripheral Vascular
Disorder<br />
Muscle Twitching<br />
Personality Change<br />
Arrhythmia<br />
Heart Rate Irregular<br />
Tachycardia<br />
Psychosomatic Disease<br />
03-Apr-2012 09:37 AM Page: 800
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/28/08ISR Number: 5824289-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-576460 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cytolytic Hepatitis Lariam PS Roche ORAL DOSE: 15<br />
Initial or Prolonged MG/KG THEN 20<br />
MG/KG EIGHT<br />
HOURS LATER<br />
Date:07/31/08ISR Number: 5832404-4Report Type:Direct Company Report #CTU 345127 Age:46 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Restlessness Lariam/<strong>Mefloquine</strong><br />
Hospitalization - Memory Impairment N/A N/A PS BUCCAL 60 ONE PER<br />
Initial or Prolonged Confusional State DAY BUCCAL<br />
Disability<br />
Visual Impairment<br />
Other Serious<br />
Tinnitus<br />
Required<br />
Anxiety<br />
Intervention to<br />
Palpitations<br />
Prevent Permanent<br />
Impairment/Damage<br />
Dyspepsia<br />
Rash<br />
Asthma<br />
Dyspnoea<br />
Mood Altered<br />
Oedema<br />
Bone Disorder<br />
Hypertension<br />
Tachycardia<br />
Muscular Weakness<br />
Back Disorder<br />
Depression<br />
Myalgia<br />
Muscle Spasms<br />
Arthralgia<br />
Suicidal Ideation<br />
Urticaria<br />
Hypersensitivity<br />
Fatigue<br />
Date:08/07/08ISR Number: 5841157-5Report Type:Direct Company Report #CTU 345741 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anger <strong>Mefloquine</strong> Once A<br />
Hospitalization - Impaired Work Ability Week Dose PS ORAL ONCE A WEEK<br />
Initial or Prolonged Aggression PO<br />
Other Serious<br />
Suicidal Ideation<br />
Required<br />
Bipolar Disorder<br />
Intervention to<br />
Activities Of Daily<br />
Prevent Permanent Living Impaired<br />
Impairment/Damage Mental Disorder
Hallucination<br />
Confusional State<br />
Feeling Abnormal<br />
03-Apr-2012 09:37 AM Page: 801
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/12/08ISR Number: 5842265-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-578606 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Atrial Fibrillation Lariam PS Roche UNKNOWN 3 WK<br />
Ventricular Tachycardia<br />
Sinus Arrhythmia<br />
Date:08/19/08ISR Number: 5849623-3Report Type:Expedited (15-DaCompany Report #GB-ROCHE-580275 Age:4 MON Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Crying Lariam PS Roche ORAL REPORTED AS<br />
Dyspnoea<br />
MEFLOQUINE<br />
GIVEN<br />
'CRUSHED IN<br />
WATER' 1 DAY<br />
Date:08/22/08ISR Number: 5853999-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-581040 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypersensitivity Lariam PS Roche UNKNOWN<br />
Amnesia<br />
General Physical Health<br />
Deterioration<br />
Tachycardia<br />
Date:08/22/08ISR Number: 5854000-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-529962 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS Roche UNKNOWN ON AN<br />
UNSPECIFIED<br />
DATE, THE<br />
PATIENT<br />
RECEIVED 4TH 8 WK<br />
Date:08/29/08ISR Number: 5864202-XReport Type:Expedited (15-DaCompany Report #US-ROCHE-582167 Age:11 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Sepsis <strong>Mefloquine</strong><br />
Hospitalization - Chronic Granulomatous Hydrochloride PS Roche UNKNOWN DRUG NAME<br />
Initial or Prolonged Disease REPORTED AS<br />
Multi-Organ Failure<br />
MEFLOQUINE<br />
Bronchopulmonary<br />
Immunoglobulin<br />
Aspergillosis Injectable SS INTRAMUSCULAR DRUG NAME<br />
Pyrexia<br />
REPORTED AS<br />
IMMUNOGLOBULI
03-Apr-2012 09:37 AM Page: 802<br />
N
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/29/08ISR Number: 5864210-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-582044 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia <strong>Mefloquine</strong><br />
Initial or Prolonged Visual Field Defect Hydrochloride PS Roche UNKNOWN<br />
Restlessness<br />
Diarrhoea<br />
Insomnia<br />
Decreased Appetite<br />
Date:08/29/08ISR Number: 5867094-8Report Type:Direct Company Report #CTU 347996 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Panic Attack Lariam PS 1 PILL 1 PER<br />
Other Serious Ear Pain WEEK<br />
Loss Of Consciousness<br />
Depression<br />
Anxiety<br />
Psychotic Disorder<br />
Hallucination<br />
Headache<br />
Insomnia<br />
Arthralgia<br />
Neck Pain<br />
Mood Swings<br />
Confusional State<br />
Myalgia<br />
Fatigue<br />
Paranoia<br />
Dizziness<br />
Memory Impairment<br />
Date:09/02/08ISR Number: 5868957-XReport Type:Direct Company Report #CTU 348060 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Affect Lability Meflouquine PS ORAL PO 1 WK<br />
Feeling Of Despair<br />
Crying<br />
Cold Sweat<br />
Emotional Disorder<br />
Early Retirement<br />
Insomnia<br />
Marital Problem<br />
Depression<br />
Impaired Work Ability<br />
Suicidal Ideation<br />
Disturbance In Attention<br />
Muscle Disorder<br />
Divorced
Date:09/03/08ISR Number: 5868533-9Report Type:Expedited (15-DaCompany Report #US-ROCHE-582804 Age:24 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Post-Traumatic Stress<br />
Disorder<br />
03-Apr-2012 09:37 AM Page: 803
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Amnesia<br />
Physical Assault<br />
Sexual Abuse Report Source Product Role Manufacturer Route Dose Duration<br />
Theft Lariam PS Roche UNKNOWN<br />
Date:09/08/08ISR Number: 5873719-3Report Type:Expedited (15-DaCompany Report #CH-ROCHE-583994 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Obsessive-Compulsive Lariam PS Roche ORAL<br />
Disorder<br />
Date:09/08/08ISR Number: 5874972-2Report Type:Direct Company Report #CTU 348811 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Disturbance In Attention <strong>Mefloquine</strong> Hci 250<br />
Intervention to Amnesia Mg Tablets Barr<br />
Prevent Permanent Aphasia Laboratories Inc. PS Barr Laboratories<br />
Impairment/Damage Depression Inc. ORAL 1 TABLET ONCE<br />
Thinking Abnormal<br />
A WEEK PO<br />
Date:09/08/08ISR Number: 5876479-5Report Type:Direct Company Report #CTU 348925 Age:18 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder <strong>Mefloquine</strong> 250 Mg PS ORAL 250MG WEEKLY<br />
Initial or Prolonged Abnormal Behaviour PO<br />
Belligerence<br />
Paranoia<br />
Date:09/10/08ISR Number: 5880016-9Report Type:Direct Company Report #CTU 349175 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion <strong>Mefloquine</strong> PS ORAL 1 TABLET<br />
Initial or Prolonged Vomiting WEEKLY PO<br />
Other Serious Nightmare Macrobid C<br />
Required Nausea Dramamine C<br />
Intervention to Feeling Abnormal Seasonique C<br />
Prevent Permanent Suicidal Ideation Ambien Cr C<br />
Impairment/Damage Fear<br />
Frequent Bowel Movements<br />
Impaired Work Ability<br />
Dizziness<br />
Gastroenteritis<br />
Panic Attack<br />
Abnormal Behaviour<br />
Retching
Anxiety<br />
Delusion<br />
Mental Disorder<br />
Pyrexia<br />
03-Apr-2012 09:37 AM Page: 804
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/15/08ISR Number: 5883994-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-585370 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gamma-Glutamyltransferase Lariam PS Roche UNKNOWN DOSE REPORTED<br />
Increased<br />
AS 250 MG/WEE<br />
Date:09/19/08ISR Number: 5889257-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-581040 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Lariam PS Roche UNKNOWN<br />
Incorrect Dose<br />
Administered<br />
Tachycardia<br />
Hypersensitivity<br />
Unresponsive To Stimuli<br />
General Physical Health<br />
Deterioration<br />
Date:09/22/08ISR Number: 5891587-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-586516 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Lariam PS Roche UNKNOWN<br />
Nausea<br />
Euphoric Mood<br />
Vomiting<br />
Persecutory Delusion<br />
Pyrexia<br />
Date:09/29/08ISR Number: 5901813-7Report Type:Expedited (15-DaCompany Report #CA-ROCHE-319480 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Suicide Attempt Lariam PS Roche ORAL<br />
Other Serious<br />
Physical Assault<br />
Homicide<br />
Amnesia<br />
Date:09/30/08ISR Number: 5904319-4Report Type:Direct Company Report #CTU 351238 Age:22 YR Gender:Female I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
PT<br />
Activities Of Daily<br />
Living Impaired<br />
Suicidal Ideation<br />
Abnormal Dreams<br />
Discomfort<br />
Sleep Disorder
Impairment/Damage<br />
Metamorphopsia<br />
Nervousness<br />
Panic Attack<br />
Malaise<br />
Stress<br />
Feeling Hot<br />
Tension<br />
Pyrexia<br />
03-Apr-2012 09:37 AM Page: 805
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Anxiety<br />
Balance Disorder<br />
Malaria Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination<br />
Lariam -<strong>Mefloquine</strong>-<br />
Tremor 250mg Roche/Lariam PS Roche/Lariam ORAL 250MG 1/WEEK<br />
Eating Disorder<br />
PO<br />
Ill-Defined Disorder<br />
Hypochondriasis<br />
Psychotic Disorder<br />
Insomnia<br />
Hypoaesthesia<br />
Paraesthesia<br />
Mental Disorder<br />
Paranoia<br />
Depression<br />
Morbid Thoughts<br />
Asthenia<br />
Feeling Abnormal<br />
Dyskinesia<br />
Date:10/03/08ISR Number: 5907741-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-581040 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious General Physical Health Lariam PS Roche UNKNOWN<br />
Deterioration<br />
Hypersensitivity<br />
Tachycardia<br />
Amnesia<br />
Date:10/09/08ISR Number: 5914065-9Report Type:Expedited (15-DaCompany Report #SE-ROCHE-588873 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrial Fibrillation Lariam PS Roche UNKNOWN<br />
Initial or Prolonged<br />
Date:10/16/08ISR Number: 5920895-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-590200 Age:59 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Shock Lariam PS Roche UNKNOWN<br />
Hospitalization - Acute Respiratory Failure<br />
Initial or Prolonged Pulmonary Toxicity<br />
Disability<br />
Date:10/24/08ISR Number: 5929861-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-587928 Age: Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Polyneuropathy Lariam PS Roche UNKNOWN 'PATIENT TOOK<br />
Initial or Prolonged Spinal Column Stenosis FOUR TABLETS<br />
Other Serious IN TOTAL'. 4 WK<br />
03-Apr-2012 09:37 AM Page: 806
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/30/08ISR Number: 5939669-9Report Type:Direct Company Report #CTU 354413 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Dreams <strong>Mefloquine</strong> Hcl 250mg PS ORAL 250MG 1 PILL<br />
Confusional State<br />
PER WEEK PO<br />
Panic Attack<br />
Paranoia<br />
Nightmare<br />
Depression<br />
Heart Rate Increased<br />
Disturbance In Attention<br />
Dizziness<br />
Flushing<br />
Nonspecific Reaction<br />
Impaired Driving Ability<br />
Date:11/07/08ISR Number: 5945298-3Report Type:Expedited (15-DaCompany Report #NL-ROCHE-594401 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Lariam PS Roche ORAL<br />
Initial or Prolonged Psychotic Disorder<br />
Tongue Disorder<br />
Dizziness<br />
Date:11/12/08ISR Number: 5951184-5Report Type:Direct Company Report #CTU 355843 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Metabolic Disorder Lariam-<strong>Mefloquine</strong>-<br />
Intervention to Bronchitis Hoffman Laroche PS Hoffman Laroche ORAL 250MG WEEKLY<br />
Prevent Permanent Anxiety PO<br />
Impairment/Damage Panic Attack<br />
Hallucination, Auditory<br />
Pneumonitis<br />
Overdose<br />
Vestibular Disorder<br />
Balance Disorder<br />
Date:11/13/08ISR Number: 5955004-4Report Type:Direct Company Report #CTU 356027 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myalgia Lariam 250mg R PS R ORAL 250MG WEEKLY<br />
Initial or Prolonged Dyspnoea PO<br />
Diarrhoea<br />
Anxiety<br />
Vomiting<br />
Dyspepsia<br />
Pulmonary Embolism
Insomnia<br />
Chest Pain<br />
03-Apr-2012 09:37 AM Page: 807
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/19/08ISR Number: 5961252-XReport Type:Expedited (15-DaCompany Report #08-001813 Age:26 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Level Increased Foreign Doryx (Doxycycline)<br />
Vomiting Literature Capsule, 100mg PS ORAL 100 MG, BID,<br />
Cinchonism Health ORAL<br />
Tinnitus Professional <strong>Mefloquine</strong><br />
Headache (<strong>Mefloquine</strong>) SS ORAL 500 MG, ORAL<br />
Pyrexia<br />
Quinine<br />
Nausea<br />
Hydrochloride<br />
Deafness<br />
(Quinine<br />
Parasite Blood Test Hydrochloride) SS INTRAVENOUS 600 MG, QID,<br />
Positive<br />
INTRAVENOUS<br />
Erythema<br />
Flushing<br />
Date:11/20/08ISR Number: 5960089-5Report Type:Expedited (15-DaCompany Report #NL-ROCHE-596597 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Dehydration REPORTED:<br />
Gastrointestinal<br />
1X/WEEK.<br />
Bacterial Infection Lariam SS Roche ORAL<br />
Date:11/24/08ISR Number: 5963991-3Report Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2008-RO-00257RO Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hospitalisation <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Drug Prescribing Error Inc. 47 DAY<br />
Other Serious<br />
Date:11/25/08ISR Number: 5966467-2Report Type:Expedited (15-DaCompany Report #NL-ROCHE-594401 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Lariam PS Roche ORAL<br />
Initial or Prolonged Psychotic Disorder<br />
Dizziness<br />
Tongue Disorder<br />
Date:11/26/08ISR Number: 5972754-4Report Type:Direct Company Report #CTU 357480 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Panic Attack Lariam PS<br />
Intervention to<br />
Feeling Abnormal<br />
Prevent Permanent Claustrophobia
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 808
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/02/08ISR Number: 5976051-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-575286 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged Headache Arthritis Medication<br />
Disability Confusional State Nos C<br />
Other Serious Conversion Disorder Naproxen C ORAL<br />
Weight Decreased Hctz C ORAL<br />
Arthralgia Paxil C<br />
Depression Tramadol C ORAL<br />
Dysphagia Aspirin C ORAL<br />
Dyspnoea Exertional Heparin C<br />
Agitation Ativan C FREQUENCY: 1<br />
Deafness<br />
MG Q.H.S<br />
Suicidal Ideation Seroquel C<br />
Scab Unknown Bp Medicine C<br />
Poor Quality Sleep Enalapril C ORAL<br />
Constipation<br />
Mania<br />
Chest Pain<br />
Date:12/02/08ISR Number: 5976068-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-598858 Age:71 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Psychotic Disorder Fosamax C<br />
Other Serious<br />
Depression<br />
Choking Sensation<br />
Paranoia<br />
Dehydration<br />
Swollen Tongue<br />
Fall<br />
Date:12/08/08ISR Number: 5983945-0Report Type:Expedited (15-DaCompany Report #NL-ROCHE-596597 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Bacterial Infection REPORTED:<br />
Anxiety<br />
1X/WEEK.<br />
Lariam SS Roche ORAL<br />
Date:12/10/08ISR Number: 5993287-5Report Type:Direct Company Report #CTU 358888 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paraesthesia Oral <strong>Mefloquine</strong> Hcl 250<br />
Pain In Extremity<br />
Mg Tablesan Sandoz,<br />
Paraesthesia Inc PS Sandoz, Inc ORAL ONE WEEKLY PO
Mass<br />
Headache<br />
Neck Pain<br />
Burning Sensation<br />
03-Apr-2012 09:37 AM Page: 809
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/11/08ISR Number: 5999479-3Report Type:Direct Company Report #CTU 358964 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Convulsion Lariam PS ORAL WEEKLY MOUTH<br />
Dilantin<br />
C<br />
Date:12/12/08ISR Number: 5995867-XReport Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2008-RO-00369RO Age:4 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Interstitial Lung Disease <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Hospitalization - Pulmonary Fibrosis Inc.<br />
Initial or Prolonged Rash Immunoglobulins C<br />
Pyrexia Corticosteroids C<br />
Fatigue Antibiotics C<br />
Weight Decreased Corticosteroids C<br />
Date:12/18/08ISR Number: 6011449-8Report Type:Expedited (15-DaCompany Report #IE-ROCHE-313825 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Fear Lariam PS Roche UNKNOWN<br />
Other Serious<br />
Completed Suicide<br />
Persecutory Delusion<br />
Mood Altered<br />
Paranoia<br />
Date:12/18/08ISR Number: 6011699-0Report Type:Expedited (15-DaCompany Report #CH-ROCHE-890100204001 Age:70 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Pneumonia Aspiration Lariam PS Roche ORAL<br />
Other Serious Shock Quinine C INTRAVENOUS<br />
Cerebral Malaria<br />
Pulmonary Oedema<br />
Date:12/18/08ISR Number: 6011707-7Report Type:Expedited (15-DaCompany Report #CA-ROCHE-900800054001 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Disease Progression Lariam PS Roche ORAL<br />
Life-Threatening Lariam SS Roche ORAL<br />
Hospitalization - Diclazuril C ORAL<br />
Initial or Prolonged Somatostatin C SUBCUTANEOUS<br />
Disability Spiramycin C INTRAVENOUS<br />
Date:01/15/09ISR Number: 6043585-4Report Type:Direct Company Report #CTU 362740 Age:14 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Tinnitus Lariam-<strong>Mefloquine</strong>-<br />
Initial or Prolonged Vision Blurred 250 Mg Roche<br />
Required Rash Pharmaceuticals PS Roche<br />
Intervention to Psychotic Disorder Pharmaceuticals ORAL 250 MG ONCE A<br />
Prevent Permanent Aggression WEEK PO<br />
Impairment/Damage<br />
03-Apr-2012 09:37 AM Page: 810
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/26/09ISR Number: 6051859-6Report Type:Expedited (15-DaCompany Report #DE-ROCHE-581040 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious General Physical Health Lariam PS Roche ORAL<br />
Deterioration<br />
Tachycardia<br />
Hypersensitivity<br />
Amnesia<br />
Date:01/26/09ISR Number: 6057433-XReport Type:Direct Company Report #CTU 363921 Age:34 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Fear <strong>Mefloquine</strong> 250mg<br />
Anxiety Sandoz, Inc. PS Sandoz, Inc. ORAL 1 TABLET PER<br />
Nausea WEEK PO 7 WK<br />
Tinnitus<br />
Syncope<br />
Headache<br />
Diarrhoea<br />
Dizziness<br />
Date:01/28/09ISR Number: 6055070-4Report Type:Expedited (15-DaCompany Report #NL-ROCHE-596597 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Bacterial Infection REPORTED:<br />
Road Traffic Accident<br />
1X/WEEK.<br />
Anxiety Lariam SS Roche ORAL<br />
Lariam SS Roche ORAL<br />
Date:01/30/09ISR Number: 6059290-4Report Type:Expedited (15-DaCompany Report #GB-ROCHE-609832 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Renal Failure Acute Lariam PS Roche ORAL DRUG<br />
Initial or Prolonged Syncope WITHDRAWN<br />
Rhabdomyolysis<br />
Date:02/02/09ISR Number: 6061512-0Report Type:Expedited (15-DaCompany Report #FR-ROCHE-611253 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Alanine Aminotransferase Lariam PS Roche ORAL<br />
Initial or Prolonged Increased<br />
Gamma-Glutamyltransferase<br />
Increased<br />
Abdominal Pain Upper
Drug Prescribing Error<br />
Nausea<br />
Medication Error<br />
Bradycardia<br />
Vertigo<br />
03-Apr-2012 09:37 AM Page: 811
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/02/09ISR Number: 6065717-4Report Type:Expedited (15-DaCompany Report #A03200803370 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Burkholderia Cepacia Literature Chloroquine -<br />
Hospitalization - Complex Infection Health Unknown - Unit Dose<br />
Initial or Prolonged Lung Infiltration Professional : Unknown PS<br />
Blood Culture Positive<br />
<strong>Mefloquine</strong> - Unknown<br />
Granuloma - Unit Dose :<br />
Pneumonitis Unknown SS<br />
Pyrexia<br />
Aspergillosis<br />
Date:02/09/09ISR Number: 6073845-2Report Type:Direct Company Report #CTU 365767 Age:41 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Panic Attack <strong>Mefloquine</strong> PS ORAL 1 TABLET PER<br />
Initial or Prolonged Amnesia WEEK PO<br />
Required<br />
Suicidal Ideation<br />
Intervention to<br />
Drug Ineffective<br />
Prevent Permanent<br />
Impairment/Damage<br />
Insomnia<br />
Hypomania<br />
Thinking Abnormal<br />
Anxiety<br />
Disturbance In Attention<br />
Depressed Level Of<br />
Consciousness<br />
Impaired Work Ability<br />
Date:02/13/09ISR Number: 6077926-9Report Type:Expedited (15-DaCompany Report #IE-ROCHE-497032 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amnesia Lariam PS Roche ORAL DRUG REPORTED<br />
AS: LARIAM<br />
250 MG<br />
TABLETS<br />
Date:02/17/09ISR Number: 6080007-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-613148 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pre-Eclampsia Lariam PS Roche ORAL<br />
Initial or Prolonged Maternal Exposure During Typhim Vi SS UNKNOWN<br />
Pregnancy Stamaril Unidose SS UNKNOWN DRUG: STAMRIL<br />
Caesarean Section Havrix SS INTRAMUSCULAR<br />
Date:02/25/09ISR Number: 6097639-7Report Type:Direct Company Report #CTU 367568 Age: Gender:Female I/FU:I
Outcome<br />
Other Serious<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
PT<br />
Fear<br />
Paraesthesia<br />
Palpitations<br />
Hypoaesthesia<br />
Tremor<br />
Panic Attack<br />
03-Apr-2012 09:37 AM Page: 812
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Insomnia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam/<strong>Mefloquine</strong> PS ORAL 1 WEEKLY PO 3 WK<br />
Date:03/02/09ISR Number: 6098995-6Report Type:Expedited (15-DaCompany Report #DE-ROCHE-581040 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tachycardia Lariam PS Roche ORAL<br />
General Physical Health<br />
Deterioration<br />
Amnesia<br />
Urticaria<br />
Date:03/03/09ISR Number: 6102926-XReport Type:Direct Company Report #CTU 367987 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia <strong>Mefloquine</strong> PS ORAL 250 MG WEEKLY<br />
Oesophageal Ulcer<br />
PO<br />
Date:03/13/09ISR Number: 6115524-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-619054 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Memory Impairment Lariam PS Roche ORAL<br />
Speech Disorder<br />
Confusional State<br />
Cognitive Disorder<br />
Unresponsive To Stimuli<br />
Date:03/16/09ISR Number: 6119261-6Report Type:Expedited (15-DaCompany Report #US-ROCHE-621179 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aggression Lariam PS Roche UNKNOWN FORM: PILL<br />
Disorientation<br />
<strong>Mefloquine</strong><br />
Vestibular Disorder Hydrochloride SS Roche UNKNOWN FORM: PILL<br />
Anger<br />
Hallucination<br />
Date:03/16/09ISR Number: 6119262-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-621169 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Self Injurious Behaviour Lariam PS Roche UNKNOWN FORM: PILL
Date:03/17/09ISR Number: 6124514-1Report Type:Direct Company Report #CTU 369854 Age:64 YR Gender:Female I/FU:I<br />
Outcome<br />
PT<br />
Dizziness<br />
Insomnia<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 813
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Medication Error<br />
Disorientation<br />
Abnormal Dreams Report Source Product Role Manufacturer Route Dose Duration<br />
Sinusitis<br />
<strong>Mefloquine</strong> Hcl 250mg<br />
Lethargy Sandoz PS Sandoz ORAL ONE TABLET<br />
Wrong Drug Administered<br />
DAILY PO<br />
Date:03/18/09ISR Number: 6123298-0Report Type:Expedited (15-DaCompany Report #SK-ROCHE-619440 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Disseminated <strong>Mefloquine</strong><br />
Intravascular Coagulation Hydrochloride PS Roche INTRAVENOUS<br />
Thrombocytopenia Klimicin SS INTRAVENOUS<br />
Date:03/19/09ISR Number: 6124240-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-620677 Age:38 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lariam PS Roche UNKNOWN<br />
Memory Impairment<br />
Brain Injury<br />
Fear<br />
Convulsion<br />
Date:03/19/09ISR Number: 6124241-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-620680 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Weight Decreased Lariam PS Roche UNKNOWN<br />
Malaise<br />
Tremor<br />
Date:03/27/09ISR Number: 6134700-2Report Type:Expedited (15-DaCompany Report #FR-ROCHE-622237 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Pressure Increased Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Gastritis<br />
Other Serious<br />
Vestibular Neuronitis<br />
Dehydration<br />
Date:03/31/09ISR Number: 6142028-XReport Type:Direct Company Report #CTU 371475 Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Lariam 250mg Sandoz PS Sandoz ORAL 11 WEEKLY PO<br />
Insomnia Lariam C
Anxiety<br />
Activities Of Daily<br />
Living Impaired<br />
Drug Dependence<br />
Palpitations<br />
Asthenia<br />
03-Apr-2012 09:37 AM Page: 814
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/07/09ISR Number: 6145878-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-457571 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dyspepsia Lariam PS Roche UNKNOWN DOSAGE<br />
Initial or Prolonged Paraesthesia REGIMEN<br />
Other Serious Feeling Abnormal REPORTED AS<br />
Sleep Disorder<br />
FOUR TABS IN<br />
Asthenia<br />
TOTAL. WEEKLY<br />
Vertigo<br />
Mental Disorder<br />
Formication<br />
Abnormal Dreams<br />
Fatigue<br />
Oedema Peripheral<br />
Anxiety<br />
Headache<br />
Cardiovascular Disorder<br />
Unevaluable Event<br />
Depression<br />
Pyrexia<br />
Muscle Twitching<br />
Malaise<br />
Upper Respiratory Tract<br />
Infection<br />
Coordination Abnormal<br />
Night Sweats<br />
Agitation<br />
Tachycardia<br />
Tinnitus<br />
Disturbance In Attention<br />
Date:04/10/09ISR Number: 6155267-9Report Type:Direct Company Report #CTU 373056 Age:54 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Skin Burning Sensation Lariam/<strong>Mefloquine</strong><br />
Intervention to Diarrhoea 250 Mg Roche PS Roche ORAL 250 MG ONCE A<br />
Prevent Permanent Tinnitus WEEK PO<br />
Impairment/Damage Pyrexia<br />
Tremor<br />
Dizziness<br />
Anxiety<br />
Date:04/14/09ISR Number: 6154073-9Report Type:Expedited (15-DaCompany Report #FR-ROCHE-622237 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Blood Pressure Increased Lariam PS Roche ORAL<br />
Initial or Prolonged Gastritis<br />
Other Serious<br />
Vestibular Neuronitis<br />
Dehydration
Date:04/14/09ISR Number: 6157667-XReport Type:Direct Company Report #CTU 373296 Age:55 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Other Serious<br />
03-Apr-2012 09:37 AM Page: 815
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Required<br />
Intervention to<br />
Prevent Permanent PT Report Source Product Role Manufacturer Route Dose Duration<br />
Impairment/Damage Tinnitus <strong>Mefloquine</strong> (Lariam) PS Roche Laboratories ORAL 250 MG WEEKLY<br />
Judgement Impaired<br />
PO<br />
Toxicity To Various<br />
Agents<br />
Confusional State<br />
Disorientation<br />
Brain Injury<br />
Stress<br />
Fatigue<br />
Asthenia<br />
Anhedonia<br />
Burning Sensation<br />
Anxiety<br />
Hair Colour Changes<br />
Sexual Dysfunction<br />
Paraesthesia<br />
Dizziness<br />
Balance Disorder<br />
Muscle Twitching<br />
Muscle Contractions<br />
Involuntary<br />
Insomnia<br />
Neuralgia<br />
Sleep Apnoea Syndrome<br />
Nervous System Disorder<br />
Myalgia<br />
Mental Disorder<br />
Emotional Disorder<br />
Date:04/16/09ISR Number: 6156743-5Report Type:Expedited (15-DaCompany Report #US-ROCHE-625513 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Expired Drug Administered Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Malaria<br />
Date:04/24/09ISR Number: 6166557-8Report Type:Expedited (15-DaCompany Report #FI-ROCHE-627019 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Loss Of Consciousness Lariam PS Roche ORAL<br />
Initial or Prolonged Convulsion Primaspan C ORAL<br />
Simvastatin C ORAL<br />
Cozaar Comp C ORAL STRENGTH<br />
REPORTED AS<br />
100MG/25MG.<br />
DRUG<br />
REPORTED:
Date:04/28/09ISR Number: 6171274-4Report Type:Direct Company Report #CTU 374910 Age: Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
03-Apr-2012 09:37 AM Page: 816
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Required<br />
Intervention to<br />
Prevent Permanent PT Report Source Product Role Manufacturer Route Dose Duration<br />
Impairment/Damage Abnormal Dreams Lariam 250 Mg Roche PS Roche ORAL 250 MG WEEKLY<br />
Depression<br />
PO, 3-4 DOSES<br />
Insomnia Risperdal C<br />
Hypophagia Claritin C<br />
Weight Decreased Periactin C<br />
Psychomotor Retardation Wellbutrin C<br />
Dyspnoea Ambien C<br />
Panic Attack Klonopin C<br />
Date:05/07/09ISR Number: 6182450-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-620677 Age:38 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Brain Injury Lariam PS Roche UNKNOWN<br />
Anxiety<br />
Fear<br />
Convulsion<br />
Memory Impairment<br />
Date:05/12/09ISR Number: 6189598-3Report Type:Direct Company Report #CTU 376748 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Crying <strong>Mefloquine</strong> 250 Mg PS ORAL 250 MG WEEKY<br />
Nightmare<br />
PO<br />
Insomnia<br />
Hypervigilance<br />
Agitation<br />
Dyspnoea<br />
Paranoia<br />
Unevaluable Event<br />
Anxiety<br />
Date:05/18/09ISR Number: 6193521-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-631390 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Nightmare Lariam PS Roche ORAL<br />
Initial or Prolonged Insomnia Imovane C<br />
Mania Xanax C<br />
Weight Decreased Laroxyl C<br />
Rivotril<br />
C<br />
Rohypnol<br />
C<br />
Tercian<br />
C<br />
Stilnox<br />
C<br />
Lysanxia<br />
C
Date:06/02/09ISR Number: 6211453-0Report Type:Expedited (15-DaCompany Report #CH-ROCHE-635186 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Optic Atrophy Lariam PS Roche ORAL<br />
03-Apr-2012 09:37 AM Page: 817
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/03/09ISR Number: 6219428-2Report Type:Periodic Company Report #563556 Age:63 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Literature <strong>Mefloquine</strong><br />
Initial or Prolonged Mania Health Hydrochloride<br />
Professional<br />
(<strong>Mefloquine</strong><br />
Other Hydrochloride) PS<br />
Lunesta<br />
(Eszopiclone)<br />
C<br />
Eskalith Cr (Lithium<br />
Nos)<br />
C<br />
Date:06/03/09ISR Number: 6219439-7Report Type:Periodic Company Report #591140 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Completed Suicide Other Lariam (<strong>Mefloquine</strong><br />
Psychogenic Pain Disorder Hydrochloride) PS<br />
Abdominal Pain Upper<br />
Date:06/03/09ISR Number: 6219441-5Report Type:Periodic Company Report #610871 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania Health Lariam (<strong>Mefloquine</strong><br />
Initial or Prolonged Professional Hydrochloride) PS<br />
Other<br />
Date:06/05/09ISR Number: 6219220-9Report Type:Direct Company Report #CTU 379716 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Grand Mal Convulsion <strong>Mefloquine</strong> 250 Mg<br />
Unk PS ORAL 250 MG ONCE<br />
PO<br />
Date:06/12/09ISR Number: 6226710-1Report Type:Expedited (15-DaCompany Report #CA-ROCHE-636293 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Methaemoglobinaemia <strong>Mefloquine</strong><br />
Dyspnoea Hydrochloride PS Roche UNKNOWN<br />
Cyanosis Primaquine SS UNKNOWN<br />
Hypoxia<br />
Date:06/15/09ISR Number: 6233305-2Report Type:Direct Company Report #CTU 380628 Age:20 YR Gender:Male I/FU:I
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Disability<br />
Other Serious<br />
PT<br />
Anxiety<br />
Mood Altered<br />
Aggression<br />
Alcohol Use<br />
Panic Attack<br />
Sinus Arrhythmia<br />
Memory Impairment<br />
03-Apr-2012 09:37 AM Page: 818
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Nightmare<br />
Supraventricular<br />
Extrasystoles Report Source Product Role Manufacturer Route Dose Duration<br />
Depression<br />
<strong>Mefloquine</strong><br />
Tinnitus Hydrochloride PS 023/047<br />
Psychotic Behaviour Larium SS<br />
Paranoia Prolixin C<br />
Agitation Paxil C<br />
Ear Pain Navane C<br />
Date:06/23/09ISR Number: 6241720-6Report Type:Expedited (15-DaCompany Report #JP-ROCHE-493929 Age:54 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Urinary Tract Infection <strong>Mefloquine</strong><br />
Initial or Prolonged Haemolytic Anaemia Hydrochloride PS Roche ORAL<br />
Other Serious Nervous System Disorder Artesunate C INTRAVENOUS<br />
Date:06/23/09ISR Number: 6241739-5Report Type:Expedited (15-DaCompany Report #ES-ROCHE-502414 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatitis Toxic <strong>Mefloquine</strong><br />
Erythema Hydrochloride PS Roche UNKNOWN<br />
Skin Exfoliation Hepatitis B Vaccine C<br />
Date:06/30/09ISR Number: 6250672-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-619054 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Cognitive Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged Dizziness<br />
Other Serious<br />
Fatigue<br />
Aphasia<br />
Dissociation<br />
Upper Respiratory Tract<br />
Infection<br />
Delusion<br />
Headache<br />
Hyperhidrosis<br />
Unresponsive To Stimuli<br />
Skin Disorder<br />
Sleep Disorder<br />
Date:07/22/09ISR Number: 6276324-2Report Type:Expedited (15-DaCompany Report #FI-ROCHE-643178 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Lariam PS Roche ORAL
Throat Irritation<br />
Product Quality Issue<br />
Fatigue<br />
Decreased Appetite<br />
03-Apr-2012 09:37 AM Page: 819
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/06/09ISR Number: 6301233-XReport Type:Direct Company Report #CTU 387916 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Upper <strong>Mefloquine</strong> PS Sandoz 250 MG 1 X<br />
Nausea<br />
DAY<br />
Flatulence<br />
Date:09/03/09ISR Number: 6339884-9Report Type:Expedited (15-DaCompany Report #FI-ROCHE-643178 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Pain Lariam PS Roche ORAL<br />
Fatigue<br />
Throat Irritation<br />
Decreased Appetite<br />
Product Quality Issue<br />
Date:09/04/09ISR Number: 6341111-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-654282 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Constipation Lariam PS Roche UNKNOWN<br />
Initial or Prolonged<br />
Date:09/11/09ISR Number: 6356543-7Report Type:Direct Company Report #CTU 391604 Age:49 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Headache <strong>Mefloquine</strong> PS ORAL 1 WHITE PILL<br />
Intervention to Affective Disorder 50 PULSE PO<br />
Prevent Permanent Insomnia YES<br />
Impairment/Damage Post-Traumatic Stress Lariam SS YES<br />
Disorder<br />
Dizziness<br />
Arthralgia<br />
Tinnitus<br />
Photosensitivity Reaction<br />
Date:09/18/09ISR Number: 6361469-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-653396 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sopor Lariam PS Roche ORAL<br />
Initial or Prolonged<br />
Date:10/02/09ISR Number: 6386657-7Report Type:Expedited (15-DaCompany Report #DE-ROCHE-654282 Age:22 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rash Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Constipation<br />
03-Apr-2012 09:37 AM Page: 820
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/05/09ISR Number: 6387922-XReport Type:Expedited (15-DaCompany Report #DK-ROCHE-658792 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Amenorrhoea Lariam PS Roche ORAL THE FIRST<br />
Acne<br />
PERIOD OF<br />
TREATMENT WAS<br />
TWO YEARS<br />
AGO.<br />
Lariam SS Roche ORAL FREQUENCY:<br />
QWK (PER<br />
WEEK).<br />
Date:10/13/09ISR Number: 6401872-1Report Type:Direct Company Report #CTU 394752 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Insomnia <strong>Mefloquine</strong> PS ORAL 1 PILL 1 TIME<br />
Heart Rate Increased<br />
PER WEEK PO<br />
Activities Of Daily<br />
Living Impaired<br />
Mental Disorder<br />
Fear<br />
Panic Attack<br />
Anxiety<br />
Hypersomnia<br />
Date:10/15/09ISR Number: 6402365-8Report Type:Expedited (15-DaCompany Report #US-ROCHE-448387 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Suicidal Ideation Lariam PS Roche UNKNOWN<br />
Post Concussion Syndrome Cymbalta C<br />
Date:10/26/09ISR Number: 6413276-6Report Type:Expedited (15-DaCompany Report #NL-ROCHE-596597 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Gastrointestinal Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Bacterial Infection REPORTED:<br />
Other Serious Road Traffic Accident 1X/WEEK.<br />
Toxicity To Various Lariam SS Roche ORAL<br />
Agents Lariam SS Roche ORAL<br />
Thermal Burn<br />
Delirium<br />
Psychotic Disorder<br />
Panic Attack<br />
Contusion<br />
Anxiety<br />
Dehydration<br />
Fear
Date:10/28/09ISR Number: 6417569-8Report Type:Expedited (15-DaCompany Report #FR-ROCHE-662936 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancreatitis Lariam PS Roche ORAL<br />
Initial or Prolonged<br />
03-Apr-2012 09:37 AM Page: 821
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/16/09ISR Number: 6441094-1Report Type:Expedited (15-DaCompany Report #KR-ROCHE-667927 Age:57 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Plasmodium Falciparum <strong>Mefloquine</strong><br />
Infection Hydrochloride PS Roche UNKNOWN<br />
Date:11/27/09ISR Number: 6458750-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-669990 Age:16 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Narcolepsy Lariam PS Roche UNKNOWN<br />
Somnolence Vaccines Nos C<br />
Date:12/01/09ISR Number: 6464251-7Report Type:Periodic Company Report #US-ROXANE LABORATORIES, INC.-2009-RO-01199RO Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nausea <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Anxiety<br />
Inc.<br />
Depression<br />
Date:12/03/09ISR Number: 6468605-4Report Type:Expedited (15-DaCompany Report #NL-ROCHE-596597 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Gastrointestinal REPORTED:<br />
Other Serious Bacterial Infection 1X/WEEK.<br />
Road Traffic Accident Lariam SS Roche ORAL<br />
Psychotic Disorder Lariam SS Roche ORAL<br />
Date:12/04/09ISR Number: 6476837-4Report Type:Direct Company Report #CTU 400853 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Epistaxis Foreign <strong>Mefloquine</strong> Mephaquin<br />
8 Lactab Mepha PS Mepha ORAL 250 MG ONCE A<br />
WEEK PO<br />
Lantus Insulin C<br />
Date:12/17/09ISR Number: 6503113-3Report Type:Expedited (15-DaCompany Report #CH-ROCHE-673621 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Immune Reconstitution Lariam PS Roche ORAL<br />
Initial or Prolonged Syndrome Kaletra SS ORAL<br />
Confusional State Remeron SS ORAL<br />
Hallucination Truvada SS ORAL
Keppra C ORAL<br />
Benerva C ORAL<br />
Bactrim C ORAL<br />
03-Apr-2012 09:37 AM Page: 822
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/17/09ISR Number: 6504148-7Report Type:Expedited (15-DaCompany Report #IN-ROXANE LABORATORIES, INC.-2009-RO-01259RO Age:11 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Mania Inc. ORAL<br />
Artesunate SS PARENTERAL<br />
Artesunate SS PARENTERAL<br />
Artesunate SS PARENTERAL<br />
Primaquine<br />
SS<br />
Risperidone<br />
C<br />
Risperidone<br />
C<br />
Date:12/18/09ISR Number: 6506374-XReport Type:Expedited (15-DaCompany Report #CH-ABBOTT-09P-151-0613387-00 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Kaletra Tablets<br />
Initial or Prolonged Aggression 200mg/50mg PS ORAL 2 doses of<br />
Other Serious Cognitive Disorder 200mg/50mg<br />
Frontotemporal Dementia<br />
daily<br />
Abnormal Behaviour <strong>Mefloquine</strong> SS ORAL<br />
Incoherent Truvada SS ORAL 245mg/200mg<br />
Apraxia<br />
daily<br />
Persecutory Delusion <strong>Mefloquine</strong> SS 250mg weekly<br />
Nervous System Disorder Mirtazapine SS ORAL<br />
Grand Mal Convulsion Levetiracetam C ORAL<br />
Confusional State Thiamine Hcl C ORAL<br />
Disturbance In Attention<br />
Sulfamethoxazole And<br />
Immune Reconstitution Trimethoprim C ORAL 1 dose 3<br />
Syndrome<br />
times per<br />
Progressive Multifocal<br />
week<br />
Leukoencephalopathy<br />
Visual Impairment<br />
Date:12/21/09ISR Number: 6509110-6Report Type:Expedited (15-DaCompany Report #CH-ABBOTT-09P-151-0613387-00 Age:64 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Kaletra Tablets<br />
Initial or Prolonged Cognitive Disorder 200mg/50mg PS ORAL 2 doses of<br />
Other Serious Mental Impairment 200mg/50mg<br />
Nervous System Disorder<br />
daily<br />
Neurologic Neglect <strong>Mefloquine</strong> SS ORAL<br />
Syndrome Truvada SS ORAL 245mg/200mg<br />
Progressive Multifocal<br />
daily<br />
Leukoencephalopathy <strong>Mefloquine</strong> SS 250mg weekly<br />
Aggression Mirtazapine SS ORAL<br />
Persecutory Delusion Levetiracetam C ORAL<br />
Immune Reconstitution Thiamine Hcl C ORAL<br />
Syndrome<br />
Sulfamethoxazole And<br />
Incoherent Trimethoprim C ORAL 1 dose 3
Apraxia times per<br />
Disturbance In Attention<br />
week<br />
Confusional State<br />
Mental Disorder<br />
Abnormal Behaviour<br />
Grand Mal Convulsion<br />
03-Apr-2012 09:37 AM Page: 823
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/22/09ISR Number: 6522734-5Report Type:Expedited (15-DaCompany Report #2009SP040553 Age:64 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Grand Mal Convulsion Foreign Remeron<br />
Initial or Prolonged Visual Impairment Health (Mirtazapine)<br />
Other Serious Thinking Abnormal Professional (Mirtazapine) PS ORAL 30 MG;QD;PO<br />
Electroencephalogram<br />
Truvada (Truvada<br />
Abnormal 01778501/) SS ORAL 1 DF;QD;PO<br />
Incoherent<br />
Kaletra (Kaletra<br />
Hallucination 015066501/) SS ORAL 2 DF; BID; PO<br />
Attention<br />
Lariam (<strong>Mefloquine</strong>)<br />
Deficit/Hyperactivity (<strong>Mefloquine</strong>) SS ORAL 250 MG;QD;PO;<br />
Disorder<br />
250 MG;QW;PO<br />
Nervous System Disorder Keppra (Con.) C<br />
Progressive Multifocal Benerva (Con.) C<br />
Leukoencephalopathy Bactrim /00086101/<br />
Abnormal Behaviour (Con.) C<br />
Confusional State<br />
Persecutory Delusion<br />
Psychomotor Hyperactivity<br />
Jc Virus Test Positive<br />
Hallucination, Visual<br />
Aggression<br />
Apraxia<br />
Immune Reconstitution<br />
Syndrome<br />
Date:12/23/09ISR Number: 6518124-1Report Type:Direct Company Report #CTU 402582 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam Unknown PS ORAL 1/WEEK PO<br />
Sleep Disorder<br />
Suicidal Ideation<br />
Date:12/30/09ISR Number: 6521464-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-676934 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pancreatitis Lariam PS Roche UNKNOWN TAKEN FOR 4<br />
Erectile Dysfunction<br />
WEEKS<br />
Date:12/31/09ISR Number: 6524131-5Report Type:Expedited (15-DaCompany Report #CH-ABBOTT-09P-151-0613387-00 Age:64 YR Gender:Male I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
Other Serious<br />
PT<br />
Cognitive Disorder<br />
Nervous System Disorder<br />
Abnormal Behaviour<br />
Incoherent
Persecutory Delusion<br />
Confusional State<br />
Mental Impairment<br />
Immune Reconstitution<br />
Syndrome<br />
Grand Mal Convulsion<br />
Progressive Multifocal<br />
Leukoencephalopathy<br />
03-Apr-2012 09:37 AM Page: 824
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Hallucination<br />
Aggression<br />
Neurologic Neglect Report Source Product Role Manufacturer Route Dose Duration<br />
Syndrome<br />
Kaletra Tablets<br />
Apraxia 200mg/50mg PS ORAL 2 doses of<br />
Mental Disorder<br />
200mg/50mg<br />
Disturbance In Attention<br />
daily<br />
<strong>Mefloquine</strong> SS ORAL<br />
Truvada SS ORAL 245mg/200mg<br />
daily<br />
<strong>Mefloquine</strong> SS 250mg weekly<br />
Mirtazapine SS ORAL<br />
Levetiracetam C ORAL<br />
Thiamine Hcl C ORAL<br />
Sulfamethoxazole And<br />
Trimethoprim C ORAL 1 dose 3<br />
times per<br />
week<br />
Date:01/12/10ISR Number: 6537985-3Report Type:Direct Company Report #CTU 404434 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia <strong>Mefloquine</strong> PS Sandoz ORAL 250 MG 1/WEEK<br />
Emotional Disorder<br />
PO<br />
Psychomotor Hyperactivity<br />
Anxiety<br />
Fear<br />
Feeling Of Despair<br />
Insomnia<br />
Thinking Abnormal<br />
Date:01/13/10ISR Number: 6535336-1Report Type:Expedited (15-DaCompany Report #NZ-ROCHE-678722 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mucosal Ulceration Lariam PS Roche ORAL FREQUENCY<br />
Initial or Prolonged Asthenia REPORTED AS<br />
Agitation<br />
ONCE.<br />
Confusional State<br />
Nausea<br />
Date:01/19/10ISR Number: 6541581-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-679518 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Psychotic Disorder Lariam PS Roche UNKNOWN
Date:01/25/10ISR Number: 6549089-4Report Type:Expedited (15-DaCompany Report #NG-ROCHE-679647 Age:6 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Intravascular Haemolysis <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Artesunate SS ORAL<br />
03-Apr-2012 09:37 AM Page: 825
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/02/10ISR Number: 6567143-8Report Type:Expedited (15-DaCompany Report #CH-SANOFI-AVENTIS-2010SA005048 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Sudden Death Lantus PS SUBCUTANEOUS<br />
Mephaquin SS ORAL<br />
Humalog SS SUBCUTANEOUS<br />
Date:02/03/10ISR Number: 6569558-0Report Type:Direct Company Report #CTU 407019 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Nervous System Disorder Lariam (<strong>Mefloquine</strong>)<br />
Other Serious Suicidal Ideation Roche + Us Army PS Roche ORAL ONE PILL ONCE<br />
Affective Disorder<br />
ORAL<br />
Insomnia<br />
Mental Disorder<br />
Major Depression<br />
Anxiety Disorder<br />
Fatigue<br />
Date:02/17/10ISR Number: 6585163-4Report Type:Expedited (15-DaCompany Report #NL-ROCHE-685113 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mental Disorder Lariam PS Roche ORAL<br />
Anxiety Lariam SS Roche ORAL<br />
Date:02/18/10ISR Number: 6588357-7Report Type:Expedited (15-DaCompany Report #PT-ROCHE-684767 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction <strong>Mefloquine</strong><br />
Initial or Prolonged Atrioventricular Block Hydrochloride PS Roche ORAL<br />
Bisoprolol I UNKNOWN<br />
Date:02/19/10ISR Number: 6590405-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-679518 Age:20 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Roche ORAL REPORTED AS<br />
Initial or Prolonged<br />
LARIAM TABL.<br />
DOSE REPORTED<br />
AS 250 MGW<br />
Date:03/02/10ISR Number: 6610332-4Report Type:Expedited (15-DaCompany Report #GXKR2010DE02231 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration
Hospitalization - Pancytopenia Mirtazapine (Ngx) PS ORAL UNK 70 DAY<br />
Initial or Prolonged<br />
Lariam<br />
/Aut/ SS ORAL 250 mg, QD 65 DAY<br />
Acetylsalicylic Acid<br />
(Ngx) SS ORAL 100 mg, QD<br />
Karvea<br />
C<br />
Actrapid Human C<br />
03-Apr-2012 09:37 AM Page: 826
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Calcilac Kt<br />
C<br />
Digitoxin<br />
C<br />
Prednisolon<br />
C<br />
Spironolactone C<br />
Iscover I ORAL 75 mg, QD<br />
Torem I ORAL 10 mg, QD<br />
Pantozol I ORAL 40 mg, BID<br />
Date:03/02/10ISR Number: 6612109-2Report Type:Direct Company Report #CTU 410025 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Psychotic Disorder Lariam -<strong>Mefloquine</strong>-<br />
Intervention to Insomnia Hoffman-Laroche PS Hoffman-Laroche ORAL WEEKLY PO<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:03/03/10ISR Number: 6611774-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-676934 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Erectile Dysfunction Lariam PS Roche UNKNOWN TAKEN FOR 4<br />
Pancreatitis<br />
WEEKS<br />
Date:03/03/10ISR Number: 6611852-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-687965 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Lariam PS Roche ORAL<br />
Initial or Prolonged Calcilac Kt C ORAL FORM:<br />
CHEWABLE<br />
TABLET<br />
Karvea C ORAL FORM:FILM<br />
COATED TABLET<br />
Digitoxin C ORAL<br />
Prednisolon C ORAL<br />
Spironolactone C ORAL<br />
Actrapid C SUBCUTANEOUS FORM:INJECTIO<br />
N<br />
Acetylsalicylic Acid I<br />
ORAL<br />
Pantozol I ORAL FORM:<br />
GASTRO-RESIST<br />
ANT TABLET<br />
Mirtazapin I ORAL<br />
Torem I Roche ORAL<br />
Iscover I ORAL FORM:FILM<br />
COATED TABLET
Date:03/05/10ISR Number: 6616241-9Report Type:Expedited (15-DaCompany Report #GXKR2010PT02277 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue Bisoprolol (Ngx) PS Novartis Sector:<br />
Initial or Prolonged Drug Interaction Generics UNKNOWN 5 mg/day<br />
Atrioventricular Block <strong>Mefloquine</strong> (Ngx) I ORAL 250 mg, QW<br />
Bradycardia<br />
Presyncope<br />
03-Apr-2012 09:37 AM Page: 827
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/08/10ISR Number: 6627028-5Report Type:Expedited (15-DaCompany Report #2010SP012262 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Foreign Mirtazapine<br />
Initial or Prolonged Rhonchi Health (Mirtazapine<br />
Pyrexia Professional /01293201/) PS ORAL 15 MG;BID;PO<br />
Endocarditis Other Pantozol<br />
C-Reactive Protein<br />
(Pantoprazole<br />
Increased Sodium) SS ORAL 40 MG;BID;PO<br />
Drug Interaction<br />
Acetylsalicylic Acid<br />
(Acetylsalicylic<br />
Acid / 00002701/) SS ORAL 100 MG;QD;PO<br />
Torem<br />
(Torasemide) SS ORAL 10 MG;QD;PO<br />
Lariam<br />
(<strong>Mefloquine</strong>) SS ORAL 250 MG;QW;PO<br />
Iscover<br />
(Clopidogrel<br />
Sulfate) SS ORAL 75 MG;QD;PO<br />
Prednisolon<br />
C<br />
Digitoxin<br />
C<br />
Calcilac Kt<br />
C<br />
Spironolactone C<br />
Karvea<br />
C<br />
Actrapid<br />
C<br />
Date:03/09/10ISR Number: 6621540-0Report Type:Expedited (15-DaCompany Report #DE-WYE-H13829710 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Iscover PS Bristol-Myers Squibb ORAL<br />
Initial or Prolonged Pancytopenia Acetylsalicylic Acid SS Unknown ORAL<br />
Torem SS Boehringer Mannheim<br />
Gmbh<br />
ORAL<br />
Calcilac C Jenapharm Gmbh ORAL<br />
Actrapid Human C Novo Industri A/S SUBCUTANEOUS df-4/12/8<br />
Karvea C Bristol-Myers Squibb ORAL<br />
Spironolactone C Unknown ORAL<br />
Prednisolone C Unknown ORAL<br />
Digitoxin C Unknown ORAL<br />
Pantozol I Wyeth<br />
Pharmaceuticals Inc.<br />
Mirtazapine I Unknown ORAL 70 DAY<br />
Pantozol I Wyeth<br />
Pharmaceuticals Inc. ORAL<br />
Lariam I Hoffman-La Roche,<br />
Inc. ORAL 65 DAY<br />
Date:03/10/10ISR Number: 6624728-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-689118 Age:48 YR Gender:Female I/FU:I
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Supraventricular<br />
Tachycardia<br />
Sleep Disorder<br />
Impaired Driving Ability<br />
Bradycardia<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 828
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Disorientation<br />
Palpitations<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL ACTION TAKEN:<br />
WITHDRAWN.<br />
Date:03/11/10ISR Number: 6631204-5Report Type:Expedited (15-DaCompany Report #DE-MYLANLABS-2010S1003362 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Mirtazapin PS ORAL Dosis je<br />
Initial or Prolonged Drug Interaction Applikation:<br />
15/30 mg<br />
Karvea C ORAL<br />
Spironolactone C ORAL<br />
Calcilac Kt C ORAL 1 Df= 500mg<br />
Calcium<br />
Carbonate +<br />
400IU<br />
Colecalcifero<br />
Digitoxin C ORAL<br />
Actrapid C SUBCUTANEOUS Dosis je<br />
Applikation:<br />
4/12/8 IU<br />
Prednisolon C ORAL<br />
Torem /01036501/ I ORAL<br />
Acetylsalicylic Acid I<br />
ORAL<br />
Pantozol I ORAL<br />
Lariam I ORAL<br />
Iscover I ORAL<br />
Date:03/11/10ISR Number: 6631205-7Report Type:Expedited (15-DaCompany Report #DE-MYLANLABS-2010S1003362 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Drug Interaction Mirtazapin PS Alphapharm ORAL Dosis je<br />
Initial or Prolonged Pancytopenia Applikation:<br />
15/30 mg<br />
Karvea C ORAL<br />
Spironolactone C ORAL<br />
Calcilac Kt C ORAL 1 Df= 500mg<br />
Calcium<br />
Carbonate +<br />
400IU<br />
Colecalcifero<br />
Digitoxin C ORAL<br />
Actrapid C SUBCUTANEOUS Dosis je<br />
Applikation:<br />
4/12/8 IU<br />
Prednisolon C ORAL<br />
Torem /01036501/ I ORAL
Acetylsalicylic Acid I ORAL<br />
Pantozol I ORAL<br />
Lariam I ORAL<br />
Iscover I ORAL<br />
03-Apr-2012 09:37 AM Page: 829
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/15/10ISR Number: 6640138-1Report Type:Expedited (15-DaCompany Report #AUR-APL-2010-00611 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Foreign Mirtazapine<br />
Initial or Prolonged Pancytopenia Health Film-Coated Tablet PS ORAL 15/30MG, BID,<br />
Endocarditis Professional ORAL<br />
Other Pantoprazole SS ORAL 40MG-ORAL<br />
Acetylsalicylic Acid SS ORAL 100MG-ORAL<br />
Torsemide (Torem) SS ORAL 10MG-ORAL<br />
<strong>Mefloquine</strong> (Lariam)<br />
Tablet SS ORAL 250MG,<br />
WEEKLY, ORAL<br />
Clopidogrel Sulfate<br />
(Iscover) SS ORAL 75MG-ORAL<br />
Prednisolone<br />
C<br />
Digitoxin<br />
C<br />
Calcium Carbonate C<br />
Spironolactone C<br />
Karvea (Irbesartan) C<br />
Actrapid (Insulin<br />
Human)<br />
C<br />
Date:03/17/10ISR Number: 6638812-6Report Type:Expedited (15-DaCompany Report #PT-MYLANLABS-2010S1003533 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrioventricular Block Bisoprolol PS UNKNOWN<br />
Initial or Prolonged Second Degree Bisoprolol I UNKNOWN<br />
Other Serious Drug Interaction <strong>Mefloquine</strong> I ORAL<br />
Bisoprolol I UNKNOWN<br />
Date:03/19/10ISR Number: 6642420-0Report Type:Expedited (15-DaCompany Report #US-ROCHE-72710 Age:82 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Deafness Lariam PS Roche ORAL TOTAL DOSE OF<br />
Other Serious Dizziness 15250 MG 61 DAY<br />
Overdose Cardizem C<br />
Abasia Propoxyphene C<br />
Respiratory Disorder Desyrel C<br />
Ataxia Ultram C<br />
Speech Disorder Depakote C ORAL<br />
Bundle Branch Block Apresoline C<br />
Medication Error<br />
Asthenia<br />
Agitation<br />
Confusional State<br />
Alanine Aminotransferase<br />
Increased
Date:03/23/10ISR Number: 6646722-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-676934 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Overdose Lariam PS Roche ORAL<br />
Erectile Dysfunction<br />
Pancreatitis<br />
03-Apr-2012 09:37 AM Page: 830
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/24/10ISR Number: 6648322-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-691204 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mental Disorder Lariam PS Roche UNKNOWN INDICATION<br />
Initial or Prolonged Face Injury REPORTED AS<br />
Ill-Defined Disorder<br />
ANTI MALARIA<br />
Limb Injury<br />
Abdominal Distension<br />
Renal Disorder<br />
Proteinuria<br />
Date:03/26/10ISR Number: 6656270-2Report Type:Direct Company Report #CTU 413026 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Aphasia <strong>Mefloquine</strong> Larium PS ORAL 1 PILL ONCE A<br />
Hospitalization - Educational Problem WEEK PO<br />
Initial or Prolonged<br />
Other Serious<br />
Palpitations<br />
Confusional State<br />
Arrhythmia<br />
Metamorphopsia<br />
Insomnia<br />
Disorientation<br />
Lung Disorder<br />
Thinking Abnormal<br />
Impaired Work Ability<br />
Amnesia<br />
Paranoia<br />
Suicidal Ideation<br />
Dizziness<br />
Paraesthesia<br />
Hypersomnia<br />
Abnormal Dreams<br />
Tremor<br />
Hallucination<br />
Convulsion<br />
Malaise<br />
Dyspnoea<br />
Fatigue<br />
Abasia<br />
Date:03/26/10ISR Number: 6656273-8Report Type:Direct Company Report #CTU 413007 Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dyspnoea <strong>Mefloquine</strong> Hcl 250mg<br />
Hallucination Roxane PS Roxane ORAL 1-250 TABLET<br />
Confusional State WEEKLY PO, 1<br />
Decreased Appetite<br />
TABLET PER<br />
Personality Change<br />
WEEK<br />
Crying
Abnormal Behaviour<br />
Paranoia<br />
Heart Rate Irregular<br />
Speech Disorder<br />
03-Apr-2012 09:37 AM Page: 831
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/30/10ISR Number: 6657893-7Report Type:Expedited (15-DaCompany Report #IT-ROCHE-692416 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Lariam PS Roche ORAL<br />
Initial or Prolonged Malaria<br />
Date:03/31/10ISR Number: 6667768-5Report Type:Expedited (15-DaCompany Report #AUR-APL-2010-00715 Age:57 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Atrioventricular Block Foreign Bisoprolol PS 5 MG, DAILY,<br />
Initial or Prolonged First Degree Literature UNK<br />
Other Serious Fatigue Health <strong>Mefloquine</strong> SS ORAL 250 MG,<br />
Presyncope Professional WEEKLY, ORAL<br />
Sinus Bradycardia<br />
Drug Interaction<br />
Date:04/08/10ISR Number: 6673219-7Report Type:Periodic Company Report #US-ROXANE LABORATORIES, INC.-2010-RO-00351RO Age:19 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hallucination <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Paranoia Inc. ORAL<br />
Dyspnoea Multivitamin C<br />
Depression Nasocort C<br />
Tinnitus Vyvanse C<br />
Speech Disorder Fexofenadine C<br />
Anxiety<br />
Nightmare<br />
Cognitive Disorder<br />
Abdominal Pain Upper<br />
Fatigue<br />
Decreased Appetite<br />
Memory Impairment<br />
Personality Change<br />
Date:04/19/10ISR Number: 6686484-7Report Type:Expedited (15-DaCompany Report #SE-ROCHE-695381 Age:18 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Angioedema Lariam PS Roche ORAL<br />
Dimor<br />
C<br />
Date:04/27/10ISR Number: 6701288-4Report Type:Expedited (15-DaCompany Report #2010040047 Age:74 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Foreign Torem (Torasemide PS ORAL 10 MG (10 MG<br />
Initial or Prolonged Poikilocytosis Health 1 IN 1D),
Haemoglobin Decreased Professional ORAL<br />
Anisocytosis<br />
Mirtazapine<br />
Endocarditis (Mirtazapine) SS ORAL 5 MG (5MG, 1<br />
Pulmonary Congestion<br />
IN 1 D), ORAL<br />
Pantozol<br />
(Pantoprazole) SS 40 MG (40 MG,<br />
1 IN 1 D),<br />
ORAL<br />
03-Apr-2012 09:37 AM Page: 832
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Acetysalicylic Acid<br />
(Acetysalicylic<br />
Acid) SS ORAL 100 MG (100<br />
MG, 1 IN 1<br />
D), ORAL<br />
Lariam (<strong>Mefloquine</strong>) SS ORAL 35.7143 MG<br />
(250 MG, 1 IN<br />
1 WK), ORAL<br />
Iscover<br />
(Clopidogrel) SS ORAL 75 MG (75 MG,<br />
1 IN 1 D),<br />
ORAL<br />
Prednisolon<br />
(Prednisolone) C<br />
Digitoxin<br />
(Digitoxin)<br />
C<br />
Karvea (Irbesartan) C<br />
Calcilac Kt (Calcium<br />
Carbonate)<br />
C<br />
Spironolactone<br />
(Spironolactone) C<br />
Actrapid (Insulin) C<br />
Date:05/03/10ISR Number: 6707157-8Report Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2010-RO-00351RO Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Fatigue <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Contusion Inc. ORAL<br />
Hallucination Allegra C<br />
Paranoia Nasocort C<br />
Tinnitus Multivitamin C<br />
Abdominal Pain Upper Vyvanse C<br />
Depression Fexofenadine C<br />
Personality Change<br />
Dyspnoea<br />
Cognitive Disorder<br />
Decreased Appetite<br />
Speech Disorder<br />
Nightmare<br />
Anxiety<br />
Memory Impairment<br />
Sleep Disorder<br />
Date:05/11/10ISR Number: 6720485-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-569005 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Rhabdomyolysis Lariam PS Roche ORAL<br />
Initial or Prolonged Xanax C
Date:05/12/10ISR Number: 6722843-1Report Type:Direct Company Report #CTU 418008 Age:25 YR Gender:Female I/FU:I<br />
Outcome<br />
PT<br />
Palpitations<br />
Panic Attack<br />
Paranoia<br />
03-Apr-2012 09:37 AM Page: 833
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Nightmare<br />
Suicidal Ideation<br />
Dizziness Report Source Product Role Manufacturer Route Dose Duration<br />
Confusional State<br />
<strong>Mefloquine</strong> 250 Mg<br />
Coordination Abnormal Mepha / Switzerland PS Mepha / Switzerland ORAL 250 MG 1 X<br />
Cold Sweat<br />
WEEK PO<br />
Depression<br />
Disturbance In Attention<br />
Date:05/13/10ISR Number: 6723578-1Report Type:Expedited (15-DaCompany Report #DE-ROCHE-687965 Age:74 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Lariam PS Roche ORAL<br />
Initial or Prolonged Calcilac Kt C ORAL FORM:<br />
CHEWABLE<br />
TABLET<br />
Karvea C ORAL FORM:FILM<br />
COATED TABLET<br />
Digitoxin C ORAL<br />
Prednisolon C ORAL<br />
Spironolactone C ORAL<br />
Actrapid C SUBCUTANEOUS FORM:INJECTIO<br />
N<br />
Acetylsalicylic Acid I<br />
ORAL<br />
Pantozol I ORAL FORM:<br />
GASTRO-RESIST<br />
ANT TABLET<br />
Mirtazapin I ORAL<br />
Torem I Roche ORAL<br />
Iscover I ORAL FORM:FILM<br />
COATED TABLET<br />
Date:05/14/10ISR Number: 6726252-0Report Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2010-RO-00351RO Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Insomnia Inc. ORAL<br />
Sleep Disorder Allegra C<br />
Hallucination Nasocort C<br />
Fatigue Multivitamin C<br />
Speech Disorder Vyvanse C<br />
Abnormal Dreams Fexofenadine C<br />
Sinusitis<br />
Contusion<br />
Paranoia<br />
Cognitive Disorder<br />
Tachycardia<br />
Nightmare<br />
Dyspnoea
Abdominal Pain Upper<br />
Tinnitus<br />
Memory Impairment<br />
Disturbance In Attention<br />
Decreased Appetite<br />
Personality Change<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 834
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/01/10ISR Number: 6750020-7Report Type:Expedited (15-DaCompany Report #GXKR2010DE02231 Age:74 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Leukopenia Prednisolone (Ngx) PS Novartis Sector:<br />
Initial or Prolonged Pyrexia Generics ORAL 5 mg, QD<br />
Mirtazapine (Ngx) SS ORAL UNK 70 DAY<br />
Lariam<br />
/Aut/ SS ORAL 250 mg, QD 69 DAY<br />
Calcilac Kt<br />
C<br />
Digitoxin<br />
C<br />
Spironolactone C<br />
Acetylsalicylic Acid C ORAL 100 mg, QD<br />
Actrapid Human C<br />
Karvea<br />
C<br />
Torem I ORAL 10 mg, QD<br />
Pantozol I ORAL 40 mg, BID<br />
Iscover I ORAL 75 mg, QD<br />
Date:06/03/10ISR Number: 6769667-7Report Type:Expedited (15-DaCompany Report #2010SP012262 Age:74 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Myelodysplastic Syndrome Foreign Mirtazapine<br />
Initial or Prolonged Drug Interaction Health (Mirtazapine<br />
Endocarditis Professional /01293201/) PS ORAL 15 MG; BID;<br />
Leukopenia Other PO<br />
Pyrexia<br />
Pantozol<br />
Rhonchi<br />
(Pantoprazole<br />
Erythema Infectiosum Sodium) SS ORAL 40 MG; BID;<br />
PO<br />
Torem (Torasemide) SS ORAL 10 MG; QD; PO<br />
Iscover (Clopidogrel<br />
Sulfate) (75 Mg) SS ORAL 75 MG; QD; PO<br />
Lariam (<strong>Mefloquine</strong>) SS ORAL 250 MG; QW;<br />
PO<br />
Prednisolon<br />
(Prednisolone) SS ORAL 5 MG; QD; PO<br />
Acetylsalicylic Acid<br />
(Other Mfr)<br />
(Acetylsalicylic<br />
Acid /00002701/) SS ORAL 100 MG; QD;<br />
PO<br />
Digitoxin<br />
C<br />
Calcilac Kt<br />
C<br />
Spironolactone C<br />
Karvea<br />
C<br />
Actrapid<br />
C<br />
Date:06/10/10ISR Number: 6764578-5Report Type:Expedited (15-DaCompany Report #DE-MYLANLABS-2010S1003362 Age:74 YR Gender:Female I/FU:F
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pyrexia Mirtazapin PS Alphapharm ORAL Dosis je<br />
Initial or Prolonged Leukopenia Applikation:<br />
15/30 mg<br />
Torem /01036501/ SS ORAL<br />
Lariam SS ORAL<br />
Prednisolon SS ORAL<br />
03-Apr-2012 09:37 AM Page: 835
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pantozol SS ORAL<br />
Spironolactone C ORAL<br />
Calcilac Kt C ORAL 1 Df= 500mg<br />
Calcium<br />
Carbonate +<br />
400IU<br />
Colecalcifero<br />
Digitoxin C ORAL<br />
Acetylsalicylic Acid C<br />
ORAL<br />
Karvea C ORAL<br />
Actrapid C SUBCUTANEOUS Dosis je<br />
Applikation:<br />
4/12/8 IU<br />
Iscover I ORAL<br />
Date:06/10/10ISR Number: 6764579-7Report Type:Expedited (15-DaCompany Report #DE-MYLANLABS-2010S1003362 Age:74 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Leukopenia Mirtazapin PS ORAL Dosis je<br />
Initial or Prolonged Pyrexia Applikation:<br />
15/30 mg<br />
Torem /01036501/ SS ORAL<br />
Lariam SS ORAL<br />
Prednisolon SS ORAL<br />
Pantozol SS ORAL<br />
Spironolactone C ORAL<br />
Calcilac Kt C ORAL 1 Df= 500mg<br />
Calcium<br />
Carbonate +<br />
400IU<br />
Colecalcifero<br />
Digitoxin C ORAL<br />
Acetylsalicylic Acid C<br />
ORAL<br />
Karvea C ORAL<br />
Actrapid C SUBCUTANEOUS Dosis je<br />
Applikation:<br />
4/12/8 IU<br />
Iscover I ORAL<br />
Date:06/15/10ISR Number: 6769769-5Report Type:Expedited (15-DaCompany Report #IT-ROCHE-708290 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Plasmodium Falciparum Lariam PS Roche ORAL ROUTE: OS<br />
Initial or Prolonged Infection FREQUENCY:<br />
Anaemia<br />
WEEKLY<br />
Date:06/15/10ISR Number: 6775329-2Report Type:Direct Company Report #CTU 421259 Age:42 YR Gender:Female I/FU:I
Outcome<br />
Life-Threatening<br />
Other Serious<br />
PT<br />
Menstrual Disorder<br />
Multiple Allergies<br />
Condition Aggravated<br />
Fatigue<br />
Depression<br />
Memory Impairment<br />
03-Apr-2012 09:37 AM Page: 836
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Balance Disorder<br />
Mood Swings<br />
Feeling Of Body Report Source Product Role Manufacturer Route Dose Duration<br />
Temperature Change <strong>Mefloquine</strong> Tab 250<br />
Vertigo Mg Sandoz Inc PS Sandoz TAB 250 MG 1<br />
Headache<br />
TABLET WEEKLY<br />
Pyrexia<br />
Insomnia<br />
Muscle Spasms<br />
Abnormal Dreams<br />
Panic Attack<br />
Dizziness<br />
Disorientation<br />
Disturbance In Attention<br />
Arthralgia<br />
Coordination Abnormal<br />
Cold Sweat<br />
Confusional State<br />
Date:06/16/10ISR Number: 6771209-7Report Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2010-RO-00351RO Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sleep Disorder <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Memory Impairment Inc. ORAL<br />
Abdominal Pain Upper Allegra C<br />
Depression Nasocort C<br />
Abnormal Dreams Multivitamin C<br />
Cognitive Disorder Vyvanse C<br />
Sinusitis Fexofenadine C<br />
Dyspnoea<br />
Speech Disorder<br />
Hallucination<br />
Nightmare<br />
Anxiety<br />
Decreased Appetite<br />
Personality Change<br />
Fatigue<br />
Paranoia<br />
Insomnia<br />
Tachycardia<br />
Tinnitus<br />
Contusion<br />
Disturbance In Attention<br />
Date:06/16/10ISR Number: 6781696-6Report Type:Expedited (15-DaCompany Report #AUR-APL-2010-00611 Age:74 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Foreign Mirtazapine<br />
Initial or Prolonged Drug Interaction Health Film-Coated Tablet PS ORAL 15/30 MG,
Pancytopenia Professional BID, ORAL<br />
Pyrexia Other Pantoprazole SS ORAL 40MG - ORAL<br />
Prednisolone SS ORAL 5MG - DAILY -<br />
ORAL<br />
Torem SS ORAL 10 MG - ORAL<br />
<strong>Mefloquine</strong> (Lariam)<br />
Tablet SS ORAL 250MG,<br />
WEEKLY, ORAL<br />
03-Apr-2012 09:37 AM Page: 837
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Iscover SS ORAL 75MG - ORAL<br />
Acetylsalicylic Acid C<br />
Digitoxin<br />
C<br />
Calcium Carbonate C<br />
Spironolactone C<br />
Karvea (Irbesartan) C<br />
Actrapid (Insulin<br />
Human)<br />
C<br />
Date:07/06/10ISR Number: 6817110-1Report Type:Expedited (15-DaCompany Report #ES-ROCHE-711590 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Tinnitus Lariam PS Roche UNKNOWN<br />
Visual Impairment<br />
Hangover<br />
Nervousness<br />
Hair Disorder<br />
Insomnia<br />
Date:07/19/10ISR Number: 6856560-4Report Type:Expedited (15-DaCompany Report #NO-ROCHE-714994 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dissociation Lariam PS Roche ORAL<br />
Initial or Prolonged Anxiety<br />
Delusion<br />
Psychotic Disorder<br />
Date:07/22/10ISR Number: 6866191-8Report Type:Expedited (15-DaCompany Report #ES-ROXANE LABORATORIES, INC.-2010-RO-00865RO Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mania <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged<br />
Inc.<br />
Date:08/06/10ISR Number: 6914038-3Report Type:Direct Company Report #CTU 426167 Age:24 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Confusional State <strong>Mefloquine</strong> PS ORAL 250MG 1/WK<br />
Chest Pain<br />
ORAL<br />
Anxiety Primaquine SS ORAL 15MG 2/DAY<br />
Paraesthesia<br />
ORAL<br />
Palpitations<br />
Hyperhidrosis<br />
Dyspnoea<br />
Dizziness
Date:08/20/10ISR Number: 6937750-9Report Type:Direct Company Report #CTU 427536 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression <strong>Mefloquine</strong> Hcl 250<br />
Abnormal Dreams Mg Barr Laboratories PS Barr Laboratories<br />
03-Apr-2012 09:37 AM Page: 838
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:08/31/10ISR Number: 6958591-2Report Type:Expedited (15-DaCompany Report #IT-ROCHE-723784 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Convulsion Lariam PS Roche ORAL<br />
Initial or Prolonged Amnesia<br />
Vomiting<br />
Incontinence<br />
Date:09/09/10ISR Number: 6974608-3Report Type:Direct Company Report #CTU 429157 Age:32 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Paranoia <strong>Mefloquine</strong> (Lariam) PS<br />
Initial or Prolonged Suicidal Ideation<br />
Other Serious<br />
Abnormal Dreams<br />
Delusion<br />
Insomnia<br />
Anxiety<br />
Tachycardia<br />
Date:09/14/10ISR Number: 6980311-6Report Type:Expedited (15-DaCompany Report #IT-ROCHE-723784 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Incontinence Lariam PS Roche ORAL<br />
Initial or Prolonged Amnesia<br />
Convulsion<br />
Vomiting<br />
Date:09/17/10ISR Number: 6992598-4Report Type:Expedited (15-DaCompany Report #CI-ROCHE-726684 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Date:09/17/10ISR Number: 6992643-6Report Type:Expedited (15-DaCompany Report #CI-ROCHE-727703 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Date:09/17/10ISR Number: 6992649-7Report Type:Expedited (15-DaCompany Report #CI-ROCHE-727701 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong>
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
03-Apr-2012 09:37 AM Page: 839
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:09/17/10ISR Number: 6992650-3Report Type:Expedited (15-DaCompany Report #CI-ROCHE-727705 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Date:09/17/10ISR Number: 6992651-5Report Type:Expedited (15-DaCompany Report #CI-ROCHE-727704 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Date:09/17/10ISR Number: 6992653-9Report Type:Expedited (15-DaCompany Report #CI-ROCHE-727706 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Date:09/20/10ISR Number: 7001509-7Report Type:Expedited (15-DaCompany Report #CI-ROCHE-727702 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hepatorenal Failure <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche ORAL<br />
Date:10/07/10ISR Number: 7032717-7Report Type:Expedited (15-DaCompany Report #NL-ROCHE-730731 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria <strong>Mefloquine</strong><br />
Hydrochloride PS Roche ORAL<br />
Date:10/08/10ISR Number: 7035053-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-730882 Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Postural Orthostatic <strong>Mefloquine</strong><br />
Initial or Prolonged Tachycardia Syndrome Hydrochloride PS Roche UNKNOWN<br />
Other Serious<br />
Diarrhoea<br />
Date:10/19/10ISR Number: 7051782-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-732598 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Lariam PS Roche UNKNOWN
Drug Ineffective<br />
03-Apr-2012 09:37 AM Page: 840
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:10/20/10ISR Number: 7053655-XReport Type:Expedited (15-DaCompany Report #IT-ROCHE-734492 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Interstitial Lung Disease Lariam PS Roche ORAL ROUTE: OS,<br />
Initial or Prolonged Hypereosinophilic FREQUENCY:<br />
Syndrome<br />
TOTAL<br />
Date:10/22/10ISR Number: 7056933-3Report Type:Periodic Company Report #US-ROXANE LABORATORIES, INC.-2010-RO-01393RO Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Histamine Intolerance <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Inc.<br />
ORAL<br />
Date:10/25/10ISR Number: 7060713-2Report Type:Expedited (15-DaCompany Report #IT-ROCHE-734492 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Interstitial Lung Disease Lariam PS Roche ORAL ROUTE: OS,<br />
Initial or Prolonged Hypereosinophilic FREQUENCY:<br />
Syndrome<br />
TOTAL<br />
Date:10/27/10ISR Number: 7064702-3Report Type:Expedited (15-DaCompany Report #DE-ROCHE-227783 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Microcephaly Lariam PS Roche TRANSPLACENTAL<br />
Other Serious<br />
Congenital Oesophageal<br />
Anomaly<br />
Developmental Delay<br />
Hypertension<br />
Date:10/27/10ISR Number: 7064703-5Report Type:Expedited (15-DaCompany Report #FR-ROCHE-244148 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Klinefelter'S Syndrome Lariam PS Roche ORAL 4 WK<br />
Talipes<br />
Maternal Drugs Affecting<br />
Foetus<br />
Abortion Induced<br />
Date:10/27/10ISR Number: 7064711-4Report Type:Expedited (15-DaCompany Report #GB-ROCHE-308888 Age:27 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Encephalocele <strong>Mefloquine</strong><br />
Hydrochloride PS Roche ORAL
Date:10/27/10ISR Number: 7064715-1Report Type:Expedited (15-DaCompany Report #FR-ROCHE-49139 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Hypoglycaemia Lariam PS Roche ORAL<br />
Other Serious Death Ranitidine C<br />
03-Apr-2012 09:37 AM Page: 841
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Cotrimoxazole<br />
Ketoconazole<br />
Penicillin<br />
C<br />
C<br />
C<br />
Date:10/27/10ISR Number: 7064718-7Report Type:Expedited (15-DaCompany Report #DE-ROCHE-57420 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Hip Deformity Lariam PS Roche TRANSPLACENTAL<br />
Jaundice Neonatal<br />
Imminent Abortion<br />
Date:10/27/10ISR Number: 7064969-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-950400276001 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Ankyloglossia Congenital Lariam PS Roche<br />
Date:10/27/10ISR Number: 7068288-9Report Type:Direct Company Report #CTU 433646 Age:31 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Restlessness Lariam 250mg PS Roche ORAL 1 PILL ONCE<br />
Nightmare<br />
PER WEEK PO<br />
Hallucination<br />
Product Quality Issue<br />
Hyperacusis<br />
Tremor<br />
Abnormal Dreams<br />
Heart Rate Irregular<br />
Date:10/28/10ISR Number: 7069237-XReport Type:Expedited (15-DaCompany Report #GB-ROXANE LABORATORIES, INC.-2010-RO-01414RO Age:44 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Initial or Prolonged Leukocytosis Inc.<br />
Other Serious<br />
Postural Orthostatic<br />
Tachycardia Syndrome<br />
Blood Urine Present<br />
Nitrite Urine Present<br />
Protein Urine Present<br />
Date:10/29/10ISR Number: 7074206-XReport Type:Expedited (15-DaCompany Report #NL-ROCHE-735888 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Maternal Exposure During Lariam PS Roche ORAL
Pregnancy Lariam SS Roche ORAL<br />
Haemorrhage<br />
Abortion Missed<br />
03-Apr-2012 09:37 AM Page: 842
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:11/05/10ISR Number: 7090977-0Report Type:Direct Company Report #CTU 434769 Age:40 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Completed Suicide <strong>Mefloquine</strong> (Lariam) PS Roche ORAL ONE A WEEK PO 8 MON<br />
Initial or Prolonged Product Quality Issue<br />
Required<br />
Intervention to<br />
Prevent Permanent<br />
Impairment/Damage<br />
Date:11/09/10ISR Number: 7091146-0Report Type:Expedited (15-DaCompany Report #GB-ROCHE-730882 Age:44 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Leukocytosis <strong>Mefloquine</strong><br />
Initial or Prolonged Postural Orthostatic Hydrochloride PS Roche UNKNOWN<br />
Other Serious<br />
Tachycardia Syndrome<br />
Palpitations<br />
Diarrhoea<br />
Chest Discomfort<br />
Dizziness<br />
Sinus Tachycardia<br />
Lethargy<br />
Respiratory Alkalosis<br />
Hyperhidrosis<br />
Fibrin D Dimer Increased<br />
Date:11/11/10ISR Number: 7107348-0Report Type:Direct Company Report #CTU 435243 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Sleep Terror <strong>Mefloquine</strong> PS<br />
Initial or Prolonged Convulsion<br />
Date:11/22/10ISR Number: 7116028-7Report Type:Expedited (15-DaCompany Report #DE-ROCHE-732598 Age:26 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Malaria Lariam PS Roche UNKNOWN<br />
Drug Ineffective<br />
Date:12/03/10ISR Number: 7134773-4Report Type:Expedited (15-DaCompany Report #DE-ROCHE-744914 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Initial Insomnia Lariam PS Roche UNKNOWN<br />
Psychotic Disorder<br />
Depressed Mood<br />
Muscle Contractions
Involuntary<br />
Ill-Defined Disorder<br />
03-Apr-2012 09:37 AM Page: 843
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/23/10ISR Number: 7191336-2Report Type:Expedited (15-DaCompany Report #US-ROCHE-748952 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Lariam PS Roche ORAL<br />
Gait Disturbance<br />
Tremor<br />
Dizziness<br />
Peripheral Paralysis<br />
Balance Disorder<br />
Loss Of Consciousness<br />
Muscular Weakness<br />
Drug Dispensing Error<br />
Memory Impairment<br />
Coordination Abnormal<br />
Insomnia<br />
Date:12/27/10ISR Number: 7197996-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-749160 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delusion Of Grandeur Lariam PS Roche ORAL<br />
Initial or Prolonged Hallucination, Auditory<br />
Hallucinations, Mixed<br />
Date:12/29/10ISR Number: 7201753-XReport Type:Expedited (15-DaCompany Report #IT-ROCHE-750196 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Partial Seizures Lariam PS Roche ORAL FREQUENCY:<br />
Initial or Prolonged Status Epilepticus WEEKLY.<br />
Date:12/29/10ISR Number: 7206059-0Report Type:Direct Company Report #CTU 439781 Age:35 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination <strong>Mefloquine</strong> (Lariam) PS Roche 1 TABLET ONCE<br />
Initial or Prolonged Intentional Self-Injury A WEEK 3 WK<br />
Required<br />
Anxiety<br />
Intervention to<br />
Depression<br />
Prevent Permanent Thermal Burn<br />
Impairment/Damage Paranoia<br />
Date:01/03/11ISR Number: 7207222-5Report Type:Expedited (15-DaCompany Report #IT-ROCHE-750196 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Partial Seizures Lariam PS Roche ORAL FREQUENCY:<br />
Initial or Prolonged Status Epilepticus WEEKLY.
Date:01/13/11ISR Number: 7224924-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-752549 Age:39 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Confusional State<br />
Agitation<br />
Ataxia<br />
03-Apr-2012 09:37 AM Page: 844
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Urinary Incontinence<br />
Amnesia<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS Roche ORAL<br />
Date:01/14/11ISR Number: 7227043-7Report Type:Expedited (15-DaCompany Report #US-ROCHE-102721 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Congenital Hearing Lariam Tablets PS Roche<br />
Disorder Vitamins C<br />
Date:01/14/11ISR Number: 7227050-4Report Type:Expedited (15-DaCompany Report #FR-ROCHE-59906 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Dermatitis Atopic Lariam Tablets PS Roche TRANSPLACENTAL<br />
Premature Baby Paludrine C<br />
Duphaston<br />
C<br />
Yellow Fever Vaccine C<br />
Havrix<br />
C<br />
Hepatitis B Vaccine C<br />
Nivaquine<br />
C<br />
Typhim Vi<br />
C<br />
Date:01/14/11ISR Number: 7227158-3Report Type:Expedited (15-DaCompany Report #FR-ROCHE-753136 Age:26 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Cholestasis Of Pregnancy Lariam PS Roche ORAL FREQUENCY:<br />
WEEKLY<br />
Paludrine<br />
C<br />
Duphaston C ORAL<br />
Yellow Fever Vaccine C<br />
SUBCUTANEOUS<br />
Havrix C SUBCUTANEOUS<br />
Hepatitis B Vaccine C SUBCUTANEOUS<br />
Nivaquine C ORAL<br />
Typhim Vi<br />
C<br />
Date:01/14/11ISR Number: 7235027-8Report Type:Direct Company Report #CTU 441318 Age:42 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Convulsion <strong>Mefloquine</strong> PS ORAL 1 ML ONCE A<br />
WEEK PO 8 WK<br />
Date:01/19/11ISR Number: 7241581-2Report Type:Direct Company Report #CTU 441981 Age:54 YR Gender:Male I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Rash Pruritic <strong>Mefloquine</strong> Hcl 250<br />
Photosensitivity Reaction Mg Tablets PS ORAL 1 TABLET (250<br />
MG) ONCE/WEEK<br />
ORAL (047)<br />
03-Apr-2012 09:37 AM Page: 845
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/31/11ISR Number: 7265091-1Report Type:Expedited (15-DaCompany Report #ZA-SANOFI-AVENTIS-2011SA003745 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pneumonia Ketek PS ORAL<br />
Malaria Mefliam SS UNKNOWN<br />
Malaise Puricos C UNKNOWN<br />
Drug Interaction<br />
Date:02/03/11ISR Number: 7280060-3Report Type:Direct Company Report #CTU 443409 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aggression <strong>Mefloquine</strong> PS Safeway Pharmacy ORAL 250 MG 1 PER<br />
Product Label Issue<br />
WEEK PO<br />
Influenza Like Illness<br />
Date:02/09/11ISR Number: 7292937-3Report Type:Direct Company Report #CTU 443925 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Insomnia Larium PS ORAL 3 LARGE PILLS<br />
Intervention to Panic Attack 3 TIMES A DAY<br />
Prevent Permanent Arthralgia PO 2 WK<br />
Impairment/Damage Myalgia<br />
Self Esteem Decreased<br />
Paranoia<br />
Disturbance In Attention<br />
Thinking Abnormal<br />
Date:02/10/11ISR Number: 7285069-1Report Type:Expedited (15-DaCompany Report #ZA-SANOFI-AVENTIS-2011SA003745 Age:50 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Interaction Ketek PS ORAL<br />
Malaise Puricos C UNKNOWN<br />
Malaria Panafcort C UNKNOWN<br />
Theophen C UNKNOWN<br />
Mefliam I UNKNOWN<br />
Date:02/10/11ISR Number: 7285336-1Report Type:Expedited (15-DaCompany Report #GB-ROCHE-753520 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Weight Decreased Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Depression<br />
Mental Disorder
Date:02/25/11ISR Number: 7327952-4Report Type:Direct Company Report #CTU 445472 Age:61 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Dizziness<br />
Hyperhidrosis<br />
Asthenia<br />
Depression<br />
Nausea<br />
03-Apr-2012 09:37 AM Page: 846
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Paraesthesia<br />
Chest Pain<br />
Flushing Report Source Product Role Manufacturer Route Dose Duration<br />
Bradycardia <strong>Mefloquine</strong> PS West Ward ORAL 250MG ONCE<br />
Headache<br />
WEEKLY ORAL<br />
12/31, 1/7,<br />
1/14, 1/21<br />
Lisinopril<br />
C<br />
Date:02/25/11ISR Number: 7328807-1Report Type:Direct Company Report #CTU 445536 Age:22 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anxiety Lariam PS Hoffman La Roche ORAL 250MG WEEKLY<br />
Initial or Prolonged Insomnia PO<br />
Other Serious<br />
Hallucination, Auditory<br />
Vomiting<br />
Nausea<br />
Weight Decreased<br />
Depression<br />
Date:03/03/11ISR Number: 7328619-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-218344 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Haemangioma Lariam Tablets PS Roche ORAL<br />
Date:03/04/11ISR Number: 7332509-5Report Type:Expedited (15-DaCompany Report #IE-ROCHE-763058 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nerve Injury Lariam PS Roche UNKNOWN FREQUENCY:<br />
Nerve Root Injury<br />
WEEKLY<br />
Cervical<br />
Speech Disorder<br />
Balance Disorder<br />
Depression<br />
Tremor<br />
Gait Disturbance<br />
Date:03/10/11ISR Number: 7343777-8Report Type:Expedited (15-DaCompany Report #GB-ROCHE-107875 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Hypotonia Neonatal Lariam PS Roche<br />
Vision Abnormal Neonatal<br />
Dysmorphism<br />
Mental Retardation<br />
Developmental Delay
Microcephaly<br />
Body Height Above Normal<br />
03-Apr-2012 09:37 AM Page: 847
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/10/11ISR Number: 7344412-5Report Type:Expedited (15-DaCompany Report #DE-ROCHE-940321069001 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Bradycardia Neonatal Lariam PS Roche 3 WK<br />
Neonatal Asphyxia<br />
Hyporeflexia<br />
Hyperbilirubinaemia<br />
Neonatal<br />
Small For Dates Baby<br />
Caesarean Section<br />
Maternal Drugs Affecting<br />
Foetus<br />
Respiratory Disorder<br />
Neonatal<br />
Cyanosis Neonatal<br />
Hypotonia Neonatal<br />
Date:03/14/11ISR Number: 7349953-2Report Type:Expedited (15-DaCompany Report #CA-ROCHE-764134 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Gait Disturbance Lariam PS Roche UNKNOWN DURATION: 23<br />
Diarrhoea<br />
DAYS<br />
Vertigo Advil C<br />
Date:03/23/11ISR Number: 7369129-2Report Type:Expedited (15-DaCompany Report #GB-ROCHE-753520 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Depressed Mood Lariam PS Roche ORAL 4 TABLETS<br />
Initial or Prolonged Weight Decreased DISPENSED<br />
Other Serious<br />
Major Depression<br />
Insomnia<br />
Date:03/28/11ISR Number: 7377882-7Report Type:Expedited (15-DaCompany Report #GB-ROCHE-767499 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaphylactic Reaction <strong>Mefloquine</strong><br />
Initial or Prolonged Hydrochloride PS Roche UNKNOWN DRUG:<br />
MEFLOQUINE<br />
Date:03/30/11ISR Number: 7383944-0Report Type:Expedited (15-DaCompany Report #FR-PFIZER INC-2011065249 Age:58 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Zithromax PS Pfizer, Inc. ORAL UNK 3 DAY<br />
Initial or Prolonged Lariam SS ORAL 250 mg,<br />
weekly
Tilcotil C 20 mg, 1x/day<br />
Inipomp C 20 mg, 1x/day<br />
Actonel C 75 mg twice<br />
monthly<br />
03-Apr-2012 09:37 AM Page: 848
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/11/11ISR Number: 7405879-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-769860 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Pancytopenia Lariam PS Roche ORAL<br />
Initial or Prolonged Zithromax SS ORAL<br />
Tilcotil<br />
C<br />
Inipomp<br />
C<br />
Actonel<br />
C<br />
Date:04/12/11ISR Number: 7409148-0Report Type:Expedited (15-DaCompany Report #IE-ROCHE-769610 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hallucination Lariam PS Roche UNKNOWN<br />
Paranoia<br />
Suicidal Ideation<br />
Psychotic Disorder<br />
Suicide Attempt<br />
Anxiety<br />
Depression<br />
Date:04/13/11ISR Number: 7412305-0Report Type:Expedited (15-DaCompany Report #FI-ROCHE-770160 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychotic Disorder Lariam PS Roche ORAL<br />
Initial or Prolonged<br />
Date:04/18/11ISR Number: 7420609-0Report Type:Expedited (15-DaCompany Report #IE-ROCHE-241671 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Lariam PS Roche ORAL<br />
Other Serious<br />
Suicidal Ideation<br />
Arthralgia<br />
Nightmare<br />
Palpitations<br />
Depression<br />
Vestibular Disorder<br />
Myalgia<br />
Alcohol Intolerance<br />
Insomnia<br />
Fatigue<br />
Visual Impairment<br />
Anxiety Disorder<br />
Dry Throat<br />
Tinnitus<br />
Nausea<br />
Hypotension<br />
Restlessness
Vertigo<br />
03-Apr-2012 09:37 AM Page: 849
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:04/18/11ISR Number: 7420818-0Report Type:Expedited (15-DaCompany Report #IE-ROCHE-769921 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mood Swings Lariam PS Roche UNKNOWN<br />
Skin Disorder<br />
Suicidal Ideation<br />
Abnormal Behaviour<br />
Fatigue<br />
Insomnia<br />
Aggression<br />
Depression<br />
Weight Decreased<br />
Memory Impairment<br />
Acne<br />
Date:04/20/11ISR Number: 7425453-6Report Type:Periodic Company Report #US-ROXANE LABORATORIES, INC.-2011-RO-00514RO Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Decreased Interest Inc. ORAL<br />
Libido Decreased Lexapro C 20 mg<br />
Depressive Symptom<br />
Disturbance In Attention<br />
Asthenia<br />
Chills<br />
Anxiety<br />
Tearfulness<br />
Date:04/25/11ISR Number: 7442461-XReport Type:Direct Company Report #CTU 451598 Age:34 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Confusional State <strong>Mefloquine</strong> PS ORAL WEEKLY PO<br />
Initial or Prolonged Tinnitus<br />
Mood Swings<br />
Toxicity To Various<br />
Agents<br />
Cardiac Disorder<br />
Chest Discomfort<br />
Abnormal Dreams<br />
Anxiety<br />
Depression<br />
Insomnia<br />
Abnormal Behaviour<br />
Date:04/26/11ISR Number: 7444804-XReport Type:Direct Company Report #CTU 451780 Age:20 YR Gender:Female I/FU:I<br />
Outcome<br />
PT
Other Serious<br />
Balance Disorder<br />
Neurotoxicity<br />
Fatigue<br />
Panic Attack<br />
Disturbance In Attention<br />
Anxiety<br />
Vertigo<br />
Cognitive Disorder<br />
03-Apr-2012 09:37 AM Page: 850
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam 250 Mg/Tablet<br />
<strong>Mefloquine</strong> PS ORAL 1 TABLET<br />
WEEKLY PO<br />
Date:05/02/11ISR Number: 7451500-1Report Type:Expedited (15-DaCompany Report #FR-ROXANE LABORATORIES, INC.-2006-BP-10993RO Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Malaria <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Hospitalization - Drug Ineffective Inc. 48 HR<br />
Initial or Prolonged Completed Suicide Proguanil-Chloroquin<br />
e SS 3 MON<br />
Date:05/04/11ISR Number: 7458831-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-774347 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hypoaesthesia Lariam PS Roche ORAL TAKEN FOR<br />
Drug Level Increased<br />
MANY YEARS<br />
Abdominal Discomfort<br />
FOR TIME<br />
PERIODS OF<br />
6-12 WEEKS<br />
Date:05/10/11ISR Number: 7477508-8Report Type:Direct Company Report #CTU 453058 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Pruritus <strong>Mefloquine</strong> 250 Mg<br />
Nausea<br />
Unknown -Given By<br />
Impetigo Peace Corps- PS ORAL 250 MG WEEKLY<br />
Headache<br />
PO<br />
Rash<br />
Insomnia<br />
Bacterial Infection<br />
Stomatitis<br />
Mood Swings<br />
Diarrhoea<br />
Pollakiuria<br />
Malaise<br />
Feeling Jittery<br />
Cough<br />
Lethargy<br />
Dizziness<br />
Depression
Date:06/08/11ISR Number: 7531752-XReport Type:Expedited (15-DaCompany Report #IE-ROCHE-709959 Age: Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Chest Pain<br />
Headache<br />
Fatigue<br />
Hallucination<br />
Diarrhoea<br />
03-Apr-2012 09:37 AM Page: 851
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Renal Pain<br />
Depression<br />
Abdominal Pain Upper Report Source Product Role Manufacturer Route Dose Duration<br />
Joint Swelling Lariam PS Roche UNKNOWN DOSE AND<br />
Muscle Spasms<br />
FREQUENCY NOT<br />
Oedema Peripheral<br />
PROVIDED.<br />
Insomnia<br />
Mental Disorder<br />
Anxiety<br />
Myalgia<br />
Date:06/09/11ISR Number: 7536782-XReport Type:Expedited (15-DaCompany Report #IE-ROCHE-769921 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abnormal Behaviour Lariam PS Roche UNKNOWN<br />
Fatigue<br />
Acne<br />
Arthralgia<br />
Anxiety<br />
Insomnia<br />
Mood Swings<br />
Suicidal Ideation<br />
Depression<br />
Aggression<br />
Memory Impairment<br />
Weight Decreased<br />
Skin Disorder<br />
Date:06/09/11ISR Number: 7537009-5Report Type:Expedited (15-DaCompany Report #PHHY2011BE48648 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety Lamivudine PS 15 mg/day<br />
Drug Interaction Atazanavir SS 300 mg/day<br />
Dyspnoea Tenofovir SS 300 mg/day<br />
Palpitations Ritonavir I 100 mg/day<br />
Asthenia <strong>Mefloquine</strong> I Novartis Sector:<br />
Neurotoxicity Generics 1 DF, QW<br />
Dizziness (starting 2<br />
weeks before<br />
departure)<br />
Date:06/21/11ISR Number: 7560277-0Report Type:Expedited (15-DaCompany Report #BE-ROXANE LABORATORIES, INC.-2011-RO-00817RO Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Phobia Of Flying <strong>Mefloquine</strong> PS Roxane Laboratories,<br />
Inc.<br />
Ritonavir SS 100 mg
Atazanavir SS 300 mg<br />
Tenofovir SS 300 mg<br />
Lamivudine SS 150 mg<br />
03-Apr-2012 09:37 AM Page: 852
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:06/21/11ISR Number: 7563471-8Report Type:Direct Company Report #CTU 457120 Age:33 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare Larium -<strong>Mefloquine</strong><br />
Insomnia<br />
Hydrochloride<br />
Psychotic Disorder<br />
Standard Dose<br />
Fear Generic? PS ORAL WEEKLY PO<br />
Depression<br />
Date:06/22/11ISR Number: 7570018-9Report Type:Direct Company Report #CTU 457416 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Fear Lariam PS 1 TABLET 1<br />
Disability Confusional State PER WEEK<br />
Negative Thoughts Melfloquine SS<br />
Anxiety<br />
Hallucination<br />
Depression<br />
Disturbance In Attention<br />
Completed Suicide<br />
Educational Problem<br />
Date:06/23/11ISR Number: 7568415-0Report Type:Expedited (15-DaCompany Report #CA-ROCHE-782365 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Lariam PS Roche ORAL<br />
Aggression<br />
Psychotic Behaviour<br />
Physical Assault<br />
Paranoia<br />
Suicidal Ideation<br />
Date:06/28/11ISR Number: 7579274-4Report Type:Expedited (15-DaCompany Report #BE-ABBOTT-11P-013-0731972-00 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Asthenia Ritonavir PS<br />
Palpitations Tenofovir C<br />
Anxiety Doxycycline C<br />
Neurotoxicity Lamivudine C<br />
Drug Interaction Atazanavir C<br />
Dizziness <strong>Mefloquine</strong> I<br />
Dyspnoea<br />
Date:07/11/11ISR Number: 7602716-2Report Type:Direct Company Report #CTU 458772 Age:61 YR Gender:Female I/FU:I
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nightmare <strong>Mefloquine</strong> I Don'T<br />
Middle Insomnia Remeber PS ORAL 1 TABLET ONCE<br />
A WEEK PO<br />
03-Apr-2012 09:37 AM Page: 853
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/22/11ISR Number: 7629066-2Report Type:Direct Company Report #US-<strong>FDA</strong>-7629066 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nightmare Lariam PS ORAL 250 mg<br />
Anxiety<br />
Disturbance In Attention<br />
Depression<br />
Suicidal Ideation<br />
Tinnitus<br />
Date:07/22/11ISR Number: 7629067-4Report Type:Direct Company Report #US-<strong>FDA</strong>-7629067 Age:40 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hallucination Lariam PS 1 pill per<br />
Initial or Prolonged Sleep Disorder week<br />
Other Serious<br />
Tachycardia<br />
Acute Stress Disorder<br />
Panic Attack<br />
Bipolar I Disorder<br />
Insomnia<br />
Date:07/22/11ISR Number: 7629077-7Report Type:Direct Company Report #US-<strong>FDA</strong>-7629077 Age:25 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Discomfort <strong>Mefloquine</strong>/Larium PS ORAL unknown<br />
Initial or Prolonged Anxiety<br />
Disability<br />
Somnolence<br />
Other Serious<br />
Sleep Terror<br />
Balance Disorder<br />
Fatigue<br />
Restlessness<br />
Depression<br />
Coordination Abnormal<br />
Nausea<br />
Abnormal Behaviour<br />
Suicidal Ideation<br />
Headache<br />
Sleep Disorder<br />
Memory Impairment<br />
Mood Altered<br />
Confusional State<br />
Decreased Appetite<br />
Diarrhoea<br />
Paranoia<br />
Mental Status Changes<br />
Abdominal Pain Upper<br />
Myalgia<br />
Cognitive Disorder
Date:07/22/11ISR Number: 7629089-3Report Type:Direct Company Report #US-<strong>FDA</strong>-7629089 Age: Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Other Serious<br />
PT<br />
Sleep Disorder<br />
Affective Disorder<br />
Impaired Work Ability<br />
03-Apr-2012 09:37 AM Page: 854
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Neurotoxicity<br />
Paranoia<br />
Major Depression Report Source Product Role Manufacturer Route Dose Duration<br />
Aggression <strong>Mefloquine</strong> -Lariam- PS<br />
Suicidal Ideation<br />
Date:07/22/11ISR Number: 7629092-3Report Type:Direct Company Report #US-<strong>FDA</strong>-7629092 Age:37 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Memory Impairment <strong>Mefloquine</strong> -Lariam- PS<br />
Paranoia<br />
Mental Disorder<br />
Anxiety<br />
Restlessness<br />
Balance Disorder<br />
Tinnitus<br />
Dyspnoea<br />
Depression<br />
Hypoaesthesia<br />
Paraesthesia<br />
Fatigue<br />
Mood Altered<br />
Confusional State<br />
Alopecia<br />
Influenza Like Illness<br />
Coordination Abnormal<br />
Suicidal Ideation<br />
Date:07/25/11ISR Number: 7632099-3Report Type:Direct Company Report #US-<strong>FDA</strong>-7632099 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Insomnia Lariam PS<br />
Mental Disorder<br />
Emotional Disorder<br />
Disturbance In Attention<br />
Anger<br />
Date:07/25/11ISR Number: 7632140-8Report Type:Direct Company Report #US-<strong>FDA</strong>-7632140 Age:37 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anxiety <strong>Mefloquine</strong> PS ORAL 1<br />
Chest Discomfort<br />
Palpitations<br />
Dizziness
Date:07/26/11ISR Number: 7638848-2Report Type:Direct Company Report #CTU 460126 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Required Mental Disorder Mefloquin Lariam PS<br />
Intervention to<br />
Emotional Disorder<br />
Prevent Permanent Insomnia<br />
Impairment/Damage Vision Blurred<br />
Anger<br />
03-Apr-2012 09:37 AM Page: 855
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:07/27/11ISR Number: 7638325-9Report Type:Direct Company Report #US-<strong>FDA</strong>-7638325 Age:50 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression <strong>Mefloquine</strong> PS ORAL 1 pill<br />
Myocardial Infarction<br />
Amnesia<br />
Anxiety<br />
Insomnia<br />
Confusional State<br />
Visual Impairment<br />
Weight Decreased<br />
Date:08/02/11ISR Number: 7652942-1Report Type:Direct Company Report #CTU 460602 Age:44 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Vestibular Disorder Lariam 250 Mg<br />
Hospitalization - (<strong>Mefloquine</strong>) PS ORAL 250 MG WEEKLY<br />
Initial or Prolonged<br />
ORAL<br />
Disability<br />
Date:08/03/11ISR Number: 7653623-0Report Type:Direct Company Report #US-<strong>FDA</strong>-7653623 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Amnesia Mefloquin PS ORAL 250mg 6 WK<br />
Hospitalization - Muscle Spasms<br />
Initial or Prolonged Deafness<br />
Other Serious<br />
Aphasia<br />
Dissociation<br />
Paranoia<br />
Emotional Poverty<br />
Confusional State<br />
Vision Blurred<br />
Feeling Abnormal<br />
Insomnia<br />
Psychotic Disorder<br />
Hallucinations, Mixed<br />
Speech Disorder<br />
Personality Change<br />
Mood Swings<br />
Delirium<br />
Balance Disorder<br />
Depression<br />
Date:08/04/11ISR Number: 7656076-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-792894 Age:62 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Lariam PS Roche ORAL LARIAM TABL
Initial or Prolonged<br />
250 MG<br />
Bricanyl<br />
C<br />
Pulmicort<br />
C<br />
Behepan<br />
C<br />
Entocort C ENTOCORT<br />
PROLONGED-REL<br />
EASE CAPSULE,<br />
HARD<br />
03-Apr-2012 09:37 AM Page: 856
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Kalcipos-D C KALCIPOS-D<br />
CHEWABLE TABL<br />
Date:09/08/11ISR Number: 7737052-7Report Type:Expedited (15-DaCompany Report #FR-ROCHE-787194 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Disease Progression Lariam PS Roche ORAL<br />
Initial or Prolonged Muscular Weakness<br />
Date:09/19/11ISR Number: 7759394-1Report Type:Expedited (15-DaCompany Report #SE-ROCHE-792894 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaemia Lariam PS Roche ORAL DOSE: 2<br />
Initial or Prolonged TABLETS (500<br />
MG) IN 14 DAY<br />
INTERVALS.<br />
Bricanyl<br />
C<br />
Pulmicort<br />
C<br />
Behepan<br />
C<br />
Entocort C ENTOCORT<br />
PROLONGED-REL<br />
EASE CAPSULE,<br />
HARD<br />
Kalcipos-D C KALCIPOS-D<br />
CHEWABLE TABL<br />
Date:09/23/11ISR Number: 7774722-9Report Type:Expedited (15-DaCompany Report #SE-ROCHE-803592 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Diarrhoea Lariam PS Roche UNKNOWN<br />
Initial or Prolonged Cardiac Fibrillation Malarone SS UNKNOWN<br />
Photopsia<br />
Nightmare<br />
Hypoaesthesia<br />
Date:10/06/11ISR Number: 7799537-7Report Type:Direct Company Report #US-<strong>FDA</strong>-7799537 Age:24 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Brain Stem Syndrome <strong>Mefloquine</strong> PS ORAL 250mg<br />
Hospitalization - Mood Altered<br />
Initial or Prolonged Hallucinations, Mixed<br />
Other Serious<br />
Personality Change<br />
Psychotic Disorder
Date:10/14/11ISR Number: 7815089-7Report Type:Direct Company Report #US-<strong>FDA</strong>-7815089 Age:25 YR Gender:Female I/FU:I<br />
Outcome<br />
Life-Threatening<br />
PT<br />
Personality Change<br />
Insomnia<br />
Fatigue<br />
Depression<br />
03-Apr-2012 09:37 AM Page: 857
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Pyrexia<br />
Rash<br />
Hepatitis Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong> PS 25mg<br />
Date:10/19/11ISR Number: 7825961-XReport Type:Expedited (15-DaCompany Report #IE-ROCHE-1005156 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Anger Lariam PS UNKNOWN<br />
Personality Change<br />
Suicidal Ideation<br />
Amnesia<br />
Anxiety<br />
Paranoia<br />
Date:10/31/11ISR Number: 7875822-5Report Type:Expedited (15-DaCompany Report #SE-ROCHE-1007641 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Atrial Fibrillation Lariam PS<br />
Date:10/31/11ISR Number: 7875861-4Report Type:Expedited (15-DaCompany Report #CA-ROCHE-782365 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Aggression Lariam PS ORAL<br />
Other Serious<br />
Paranoia<br />
Depression<br />
Psychotic Behaviour<br />
Suicidal Ideation<br />
Physical Assault<br />
Date:11/04/11ISR Number: 7893372-7Report Type:Expedited (15-DaCompany Report #SE-ROCHE-1007641 Age:35 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Atrial Fibrillation Lariam PS ORAL<br />
Date:11/04/11ISR Number: 7893376-4Report Type:Expedited (15-DaCompany Report #US-ROCHE-1008551 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hip Arthroplasty Lariam PS UNKNOWN<br />
Initial or Prolonged Nervous System Disorder Norco SS UNKNOWN
Date:11/11/11ISR Number: 7910386-9Report Type:Expedited (15-DaCompany Report #NL-ROCHE-596597 Age: Gender:Female I/FU:F<br />
Outcome<br />
Life-Threatening<br />
Hospitalization -<br />
Initial or Prolonged<br />
Other Serious<br />
PT<br />
Rash<br />
Hyperaesthesia<br />
Dizziness<br />
Anxiety<br />
Gastrointestinal<br />
03-Apr-2012 09:37 AM Page: 858
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Bacterial Infection<br />
Road Traffic Accident<br />
Sleep Disorder<br />
Psychotic Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS ORAL<br />
Lariam SS ORAL<br />
Lariam SS ORAL<br />
Date:11/16/11ISR Number: 7917500-XReport Type:Expedited (15-DaCompany Report #MA-ROCHE-1012968 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Mania <strong>Mefloquine</strong> PS<br />
Psychotic Behaviour<br />
Date:11/21/11ISR Number: 7931978-7Report Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2011-RO-01656RO Age:73 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Immune Reconstitution Prednisone PS Roxane Laboratories,<br />
Life-Threatening Syndrome Inc. 10 mg<br />
Other Serious Jc Virus Infection Hydroxychloroquine<br />
Progressive Multifocal Sulfate SS<br />
Leukoencephalopathy Rituximab SS<br />
Leflunomide<br />
SS<br />
Prednisone SS Roxane Laboratories,<br />
Inc.<br />
2.5 mg<br />
<strong>Mefloquine</strong> SS Roxane Laboratories,<br />
Inc.<br />
Date:11/21/11ISR Number: 7932129-5Report Type:Expedited (15-DaCompany Report #US-ROXANE LABORATORIES, INC.-2011-RO-01657RO Age:72 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Death Jc Virus Infection Methotrexate PS Roxane Laboratories,<br />
Other Serious Lymphopenia Inc.<br />
Progressive Multifocal Mirtazapine SS<br />
Leukoencephalopathy Etanercept SS<br />
Adalimumab<br />
SS<br />
<strong>Mefloquine</strong> SS Roxane Laboratories,<br />
Inc.<br />
Rituximab<br />
SS<br />
Date:11/29/11ISR Number: 7945806-7Report Type:Expedited (15-DaCompany Report #BE-ROCHE-1014123 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS<br />
Pregnancy Of Partner
Date:11/29/11ISR Number: 7946293-5Report Type:Expedited (15-DaCompany Report #NO-ROCHE-714994 Age: Gender:Female I/FU:F<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Dissociation<br />
Psychotic Disorder<br />
Delusion<br />
03-Apr-2012 09:37 AM Page: 859
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Anxiety<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
Lariam PS ORAL<br />
Date:11/30/11ISR Number: 7949244-2Report Type:Expedited (15-DaCompany Report #BE-ROCHE-1014127 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abortion Spontaneous Lariam PS<br />
Maternal Exposure During<br />
Pregnancy<br />
Date:12/21/11ISR Number: 7997595-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-1022627 Age:20 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Headache Lariam PS ORAL<br />
Dizziness<br />
White Blood Cell Count<br />
Increased<br />
Pyrexia<br />
Loss Of Consciousness<br />
Date:12/22/11ISR Number: 8003452-3Report Type:Direct Company Report #US-<strong>FDA</strong>-8003452 Age:36 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Sleep Disorder <strong>Mefloquine</strong> PS 250 mg<br />
Anxiety<br />
Balance Disorder<br />
Migraine<br />
Nightmare<br />
Depression<br />
Date:12/22/11ISR Number: 8003453-5Report Type:Direct Company Report #US-<strong>FDA</strong>-8003453 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Suicidal Ideation <strong>Mefloquine</strong> / Lariam PS ORAL 250 mg.<br />
Somnambulism<br />
Nightmare<br />
Confusional State<br />
Dizziness<br />
Anger<br />
Loss Of Consciousness<br />
Fall
Date:12/22/11ISR Number: 8003460-2Report Type:Direct Company Report #US-<strong>FDA</strong>-8003460 Age:44 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Dysarthria<br />
Amnesia<br />
Photophobia<br />
Balance Disorder<br />
03-Apr-2012 09:37 AM Page: 860
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Tremor<br />
Tinnitus<br />
Nausea Report Source Product Role Manufacturer Route Dose Duration<br />
Dizziness Postural Lariam PS ORAL 250mg<br />
Date:12/22/11ISR Number: 8003461-4Report Type:Direct Company Report #US-<strong>FDA</strong>-8003461 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Sleep Disorder Lariam PS<br />
Balance Disorder<br />
Irritability<br />
Mood Swings<br />
Date:12/22/11ISR Number: 8011342-5Report Type:Expedited (15-DaCompany Report #2011P1014269 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delusion Of Grandeur Foreign <strong>Mefloquine</strong> (No Pref.<br />
Initial or Prolonged Mania Literature Name) PS 250 MG;QW<br />
Hallucination<br />
Health<br />
Restlessness<br />
Professional<br />
Hypomania<br />
Delusion<br />
Logorrhoea<br />
Hallucination, Auditory<br />
Neuropsychiatric Syndrome<br />
Flight Of Ideas<br />
Date:12/23/11ISR Number: 8004849-8Report Type:Direct Company Report #US-<strong>FDA</strong>-8004849 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Balance Disorder Lariam PS ORAL 1 pill<br />
Memory Impairment<br />
Tinnitus<br />
Anxiety<br />
Fatigue<br />
Tremor<br />
Disturbance In Attention<br />
Date:12/23/11ISR Number: 8004850-4Report Type:Direct Company Report #US-<strong>FDA</strong>-8004850 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression <strong>Mefloquine</strong> PS ORAL unknown<br />
Paranoia<br />
Anger<br />
Anxiety
Gastrointestinal Disorder<br />
Bipolar Disorder<br />
Post-Traumatic Stress<br />
Disorder<br />
03-Apr-2012 09:37 AM Page: 861
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/23/11ISR Number: 8004866-8Report Type:Direct Company Report #US-<strong>FDA</strong>-8004866 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Unevaluable Event Lariam PS<br />
Date:12/23/11ISR Number: 8004874-7Report Type:Direct Company Report #US-<strong>FDA</strong>-8004874 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Anxiety Larium PS One tablet<br />
Post-Traumatic Stress<br />
Disorder<br />
Depression<br />
Panic Attack<br />
Migraine<br />
Nightmare<br />
Date:12/23/11ISR Number: 8004883-8Report Type:Direct Company Report #US-<strong>FDA</strong>-8004883 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Congenital Anomaly Urticaria <strong>Mefloquine</strong> PS ORAL 250mg<br />
Other Serious<br />
Rash<br />
Arthralgia<br />
Tinnitus<br />
Diarrhoea<br />
Sleep Disorder<br />
Date:12/23/11ISR Number: 8004893-0Report Type:Direct Company Report #US-<strong>FDA</strong>-8004893 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Tinnitus Mefloquin Lariam PS ORAL 1 weekly<br />
Arthralgia<br />
Nightmare<br />
Balance Disorder<br />
Paranoia<br />
Loss Of Consciousness<br />
Anxiety<br />
Date:12/23/11ISR Number: 8005563-5Report Type:Expedited (15-DaCompany Report #GB-ROCHE-1023903 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Ineffective Lariam PS UNKNOWN<br />
Malaria
Date:12/27/11ISR Number: 8011243-2Report Type:Direct Company Report #US-<strong>FDA</strong>-8011243 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Poor Quality Sleep<br />
<strong>Mefloquine</strong><br />
Disturbance In Attention<br />
Hydrochoride<br />
Insomnia -Lariam- PS ORAL 250 mg<br />
03-Apr-2012 09:37 AM Page: 862
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:12/27/11ISR Number: 8011244-4Report Type:Direct Company Report #US-<strong>FDA</strong>-8011244 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Nightmare Lariam PS ORAL 1 YR<br />
Insomnia<br />
Anger<br />
Mood Swings<br />
Bipolar Disorder<br />
Malaise<br />
Date:12/27/11ISR Number: 8011247-XReport Type:Direct Company Report #US-<strong>FDA</strong>-8011247 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Emotional Disorder <strong>Mefloquine</strong> PS ORAL 1 x 250mg<br />
Depression<br />
pill<br />
Abnormal Behaviour<br />
Sleep Disorder<br />
Mental Disorder<br />
Apnoea<br />
Anxiety<br />
Date:12/27/11ISR Number: 8011256-0Report Type:Direct Company Report #US-<strong>FDA</strong>-8011256 Age:19 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Paranoia Lariam PS ORAL 250mg<br />
Hypervigilance<br />
Anxiety<br />
Nightmare<br />
Tinnitus<br />
Date:12/27/11ISR Number: 8011262-6Report Type:Direct Company Report #US-<strong>FDA</strong>-8011262 Age:22 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Hallucination Lariam PS ORAL 20mg<br />
Hospitalization - Insomnia<br />
Initial or Prolonged Mental Disorder<br />
Abnormal Dreams<br />
Bipolar Disorder<br />
Cold Sweat<br />
Major Depression<br />
Date:12/27/11ISR Number: 8011265-1Report Type:Direct Company Report #US-<strong>FDA</strong>-8011265 Age:41 YR Gender:Female I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Anxiety Disorder
Compulsions<br />
Dizziness<br />
Obsessive Thoughts<br />
Asthenia<br />
Tachycardia<br />
Irritability<br />
Panic Attack<br />
Major Depression<br />
03-Apr-2012 09:37 AM Page: 863
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Abnormal Dreams<br />
Nightmare<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong> PS BUCCAL 250MG<br />
Date:12/27/11ISR Number: 8011270-5Report Type:Direct Company Report #US-<strong>FDA</strong>-8011270 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Insomnia Lariam PS ORAL 1pill<br />
Hospitalization - Aphasia<br />
Initial or Prolonged Neuropathy Peripheral<br />
Brain Injury<br />
Convulsion<br />
Vestibular Neuronitis<br />
Hyperacusis<br />
Amnesia<br />
Major Depression<br />
Photophobia<br />
Date:12/27/11ISR Number: 8011277-8Report Type:Direct Company Report #US-<strong>FDA</strong>-8011277 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Mood Altered <strong>Mefloquine</strong> PS ORAL unknown<br />
Initial or Prolonged Depression<br />
Other Serious<br />
Personality Change<br />
Weight Decreased<br />
Psychotic Disorder<br />
Decreased Appetite<br />
Date:12/27/11ISR Number: 8011281-XReport Type:Direct Company Report #US-<strong>FDA</strong>-8011281 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Balance Disorder <strong>Mefloquine</strong> PS ORAL<br />
Mental Disorder<br />
Dizziness<br />
Nervous System Disorder<br />
Date:12/27/11ISR Number: 8011290-0Report Type:Direct Company Report #US-<strong>FDA</strong>-8011290 Age:32 YR Gender:Male I/FU:I<br />
Outcome<br />
Other Serious<br />
PT<br />
Mood Swings<br />
Agoraphobia<br />
Depression<br />
Dizziness<br />
Disturbance In Attention
Memory Impairment<br />
Post-Traumatic Stress<br />
Disorder<br />
Tinnitus<br />
Balance Disorder<br />
Arthralgia<br />
Irritability<br />
Hyperhidrosis<br />
03-Apr-2012 09:37 AM Page: 864
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Neck Pain<br />
Heart Rate Increased<br />
Sleep Disorder Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety Lariam PS ORAL One pill<br />
Nightmare<br />
Multiple Allergies<br />
Migraine<br />
Date:12/28/11ISR Number: 8011094-9Report Type:Expedited (15-DaCompany Report #GB-ROCHE-1023903 Age: Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Drug Dose Omission Lariam PS UNKNOWN<br />
Malaria<br />
Date:12/29/11ISR Number: 8013268-XReport Type:Direct Company Report #US-<strong>FDA</strong>-8013268 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression Larium PS<br />
Anger<br />
Postpartum Stress<br />
Disorder<br />
Date:12/29/11ISR Number: 8014072-9Report Type:Expedited (15-DaCompany Report #DE-ROCHE-1026064 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Abdominal Discomfort Lariam PS TRANSPLACENTAL<br />
Maternal Drugs Affecting<br />
Foetus<br />
Date:12/30/11ISR Number: 8016015-0Report Type:Direct Company Report #US-<strong>FDA</strong>-8016015 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Major Depression Lariam PS ORAL megadose<br />
Suicidal Ideation<br />
Sleep Disorder<br />
Fatigue<br />
Anxiety<br />
Affective Disorder<br />
Date:01/02/12ISR Number: 8017322-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-1022627 Age:20 YR Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Dizziness Lariam PS ORAL
Initial or Prolonged<br />
Other Serious<br />
Nausea<br />
Pyrexia<br />
White Blood Cell Count<br />
Increased<br />
Somnolence<br />
Headache<br />
Loss Of Consciousness<br />
03-Apr-2012 09:37 AM Page: 865
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/03/12ISR Number: 8018678-2Report Type:Direct Company Report #US-<strong>FDA</strong>-8018678 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Vertigo Lariam PS<br />
Gastric Ulcer Haemorrhage<br />
Night Sweats<br />
Mood Swings<br />
Depression<br />
Date:01/06/12ISR Number: 8027763-0Report Type:Direct Company Report #US-<strong>FDA</strong>-8027763 Age:43 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Coordination Abnormal Lariam PS ORAL 250mg<br />
Social Problem<br />
Balance Disorder<br />
Psychotic Disorder<br />
Hallucination, Visual<br />
Nightmare<br />
Abnormal Dreams<br />
Product Quality Issue<br />
Sleep Disorder<br />
Diplopia<br />
Poor Quality Drug<br />
Administered<br />
Product Contamination<br />
Brain Injury<br />
Emotional Poverty<br />
Date:01/09/12ISR Number: 8030390-2Report Type:Direct Company Report #US-<strong>FDA</strong>-8030390 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Anxiety Lariam PS<br />
Depression<br />
Anger<br />
Date:01/09/12ISR Number: 8030431-2Report Type:Direct Company Report #US-<strong>FDA</strong>-8030431 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Tinnitus Lariam PS<br />
Other Serious<br />
Amnesia<br />
Balance Disorder<br />
Anger<br />
Homicidal Ideation<br />
Depression<br />
Somnambulism
Date:01/09/12ISR Number: 8030434-8Report Type:Direct Company Report #US-<strong>FDA</strong>-8030434 Age: Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Other Serious<br />
PT<br />
Somnambulism<br />
Tinnitus<br />
Homicidal Ideation<br />
Balance Disorder<br />
03-Apr-2012 09:37 AM Page: 866
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Anger<br />
Depression<br />
Amnesia Report Source Product Role Manufacturer Route Dose Duration<br />
Mefoquine<br />
PS<br />
Date:01/10/12ISR Number: 8035586-1Report Type:Direct Company Report #CTU 468507 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Tinnitus <strong>Mefloquine</strong> 250 Mg<br />
Disability Skin Infection Generic PS ORAL 250 MG WEEKLY<br />
Loss Of Employment<br />
ORAL<br />
Anxiety Doxycycline C<br />
Neuropsychiatric Syndrome Otc Analgesics C<br />
Depression Lindane C<br />
Headache<br />
Bismuth<br />
Dizziness Subsalicylate C<br />
Visual Impairment Erythromycin C<br />
Paraesthesia Primaquine C<br />
Insomnia<br />
Malaise<br />
Pruritus<br />
Date:01/17/12ISR Number: 8048956-2Report Type:Expedited (15-DaCompany Report #PHHY2012MA001757 Age:31 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Delirium <strong>Mefloquine</strong> PS Novartis Sector:<br />
Initial or Prolonged Agitation Generics 250 mg, QW<br />
Other Serious<br />
Hallucination, Auditory<br />
Mood Altered<br />
Psychomotor Hyperactivity<br />
Logorrhoea<br />
Date:01/17/12ISR Number: 8048957-4Report Type:Expedited (15-DaCompany Report #PHHY2012MA002356 Age:27 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache <strong>Mefloquine</strong> PS Novartis Sector:<br />
Initial or Prolonged Suicidal Ideation Generics 250 mg, QW<br />
Other Serious<br />
Depressive Symptom<br />
Anxiety<br />
Personality Disorder<br />
Vertigo<br />
Psychotic Disorder<br />
Insomnia<br />
Persecutory Delusion<br />
Hallucination, Auditory<br />
Depression<br />
Poisoning
Delirium<br />
Social Avoidant Behaviour<br />
03-Apr-2012 09:37 AM Page: 867
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/17/12ISR Number: 8050687-XReport Type:Direct Company Report #US-<strong>FDA</strong>-8050687 Age:45 MON Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Visual Impairment <strong>Mefloquine</strong> PS ORAL 250mg<br />
Hospitalization - Deafness<br />
Initial or Prolonged Tinnitus<br />
Aggression<br />
Muscle Spasms<br />
Arthralgia<br />
Oedema Peripheral<br />
Pain In Extremity<br />
Insomnia<br />
Suicidal Ideation<br />
Paraesthesia<br />
Abdominal Pain<br />
Diarrhoea<br />
Headache<br />
Musculoskeletal Pain<br />
Paranoia<br />
Depression<br />
Asthenia<br />
Malaise<br />
Nausea<br />
Anxiety<br />
Vision Blurred<br />
Dry Eye<br />
Mood Swings<br />
Pruritus<br />
Date:01/17/12ISR Number: 8050698-4Report Type:Direct Company Report #US-<strong>FDA</strong>-8050698 Age:21 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Hepatic Pain <strong>Mefloquine</strong> PS ORAL 250mg<br />
Ear Discomfort<br />
Panic Attack<br />
Insomnia<br />
Chills<br />
Tinnitus<br />
Amnesia<br />
Glare<br />
Emotional Disorder<br />
Anger<br />
Lentigo<br />
Tremor<br />
Photophobia<br />
Ear Pain<br />
Fatigue<br />
Night Sweats<br />
Deafness<br />
Migraine With Aura<br />
Paraesthesia
Depression<br />
03-Apr-2012 09:37 AM Page: 868
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:01/17/12ISR Number: 8050756-4Report Type:Direct Company Report #US-<strong>FDA</strong>-8050756 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Nervous System Disorder Larium PS ORAL 250<br />
Visual Impairment<br />
Gastrointestinal Disorder<br />
Date:01/17/12ISR Number: 8050758-8Report Type:Direct Company Report #US-<strong>FDA</strong>-8050758 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Depression Lariam PS<br />
Fibromyalgia<br />
Anger<br />
Sensory Disturbance<br />
Nightmare<br />
Myalgia<br />
Irritable Bowel Syndrome<br />
Multiple Allergies<br />
Insomnia<br />
Paranoia<br />
Arthralgia<br />
Date:01/19/12ISR Number: 8058416-0Report Type:Direct Company Report #CTU 469002 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Fear <strong>Mefloquine</strong> - Larian<br />
Disability Insomnia - La Roche PS La Roche ORAL 1X PER WEEK<br />
Other Serious Vertigo ORAL<br />
Required<br />
Pain In Extremity<br />
Intervention to<br />
Panic Reaction<br />
Prevent Permanent<br />
Impairment/Damage<br />
Suicidal Ideation<br />
Muscular Weakness<br />
Feeling Abnormal<br />
Headache<br />
Chest Pain<br />
Dyspnoea<br />
Depression<br />
Anxiety<br />
Nightmare<br />
Euphoric Mood<br />
Palpitations<br />
Date:01/24/12ISR Number: 8066421-3Report Type:Direct Company Report #US-<strong>FDA</strong>-8066421 Age:20 YR Gender:Male I/FU:I<br />
Outcome<br />
Life-Threatening<br />
Other Serious<br />
PT<br />
Hallucination<br />
Anger
Mood Swings<br />
Suicidal Ideation<br />
Aggression<br />
Irritability<br />
Partner Stress<br />
Mental Disorder<br />
Personality Change<br />
Insomnia<br />
03-Apr-2012 09:37 AM Page: 869
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Depression<br />
Balance Disorder<br />
Report Source Product Role Manufacturer Route Dose Duration<br />
<strong>Mefloquine</strong><br />
PS<br />
Date:01/24/12ISR Number: 8067510-XReport Type:Expedited (15-DaCompany Report #AU-ROCHE-1032915 Age: Gender: I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Aplastic Anaemia Lariam PS<br />
Date:01/30/12ISR Number: 8082004-3Report Type:Expedited (15-DaCompany Report #PHHY2012MA001757 Age:31 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Psychomotor Hyperactivity <strong>Mefloquine</strong> PS Novartis Sector:<br />
Initial or Prolonged Hallucination, Auditory Generics 250 mg, QW<br />
Other Serious<br />
Restlessness<br />
Agitation<br />
Delirium<br />
Logorrhoea<br />
Flight Of Ideas<br />
Mood Altered<br />
Date:01/30/12ISR Number: 8082017-1Report Type:Expedited (15-DaCompany Report #PHHY2012MA002356 Age:27 YR Gender:Male I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Social Avoidant Behaviour <strong>Mefloquine</strong> PS Novartis Sector:<br />
Initial or Prolonged Emotional Disorder Generics 250 mg, QW<br />
Other Serious<br />
Persecutory Delusion<br />
Depression<br />
Anxiety<br />
Poisoning<br />
Dizziness<br />
Insomnia<br />
Hallucination, Auditory<br />
Headache<br />
Withdrawal Syndrome<br />
Psychotic Disorder<br />
Depressive Symptom<br />
Delirium<br />
Suicidal Ideation<br />
Date:02/01/12ISR Number: 8183412-2Report Type:Periodic Company Report #286078USA Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Glossodynia Health <strong>Mefloquine</strong>
Swollen Tongue Professional Hydrochloride Tablet<br />
250mg (<strong>Mefloquine</strong>)<br />
PS<br />
03-Apr-2012 09:37 AM Page: 870
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/10/12ISR Number: 8128421-4Report Type:Expedited (15-DaCompany Report #GB-ROCHE-1034260 Age:50 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Skin Disorder Lariam PS ORAL<br />
Asthenia<br />
Migraine<br />
Dyspnoea<br />
Alopecia<br />
Headache<br />
Lethargy<br />
Dizziness<br />
Tearfulness<br />
Date:02/13/12ISR Number: 8133825-XReport Type:Direct Company Report #US-<strong>FDA</strong>-8133825 Age:35 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Vestibular Disorder <strong>Mefloquine</strong> PS ORAL 1 pill<br />
Hospitalization - Dizziness<br />
Initial or Prolonged Aggression<br />
Other Serious<br />
Insomnia<br />
Cognitive Disorder<br />
Brain Injury<br />
Suicidal Ideation<br />
Affective Disorder<br />
Impaired Work Ability<br />
Convulsion<br />
Psychotic Disorder<br />
Date:02/15/12ISR Number: 8138119-4Report Type:Direct Company Report #US-<strong>FDA</strong>-8138119 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious No Adverse Event <strong>Mefloquine</strong> Hcl PS ORAL 250 mg<br />
Doxycycline C ORAL<br />
Date:02/17/12ISR Number: 8143978-5Report Type:Direct Company Report #US-<strong>FDA</strong>-8143978 Age:29 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Abnormal Dreams <strong>Mefloquine</strong> PS 1 pill 8 MON<br />
Panic Attack<br />
Confusional State<br />
Insomnia<br />
Palpitations<br />
Pain<br />
Nightmare<br />
Memory Impairment
Date:02/17/12ISR Number: 8144002-0Report Type:Direct Company Report #US-<strong>FDA</strong>-8144002 Age:29 YR Gender:Male I/FU:I<br />
Outcome<br />
PT<br />
Nightmare<br />
Memory Impairment<br />
Abnormal Dreams<br />
Pain<br />
03-Apr-2012 09:37 AM Page: 871
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Confusional State<br />
Insomnia<br />
Panic Attack Report Source Product Role Manufacturer Route Dose Duration<br />
Palpitations <strong>Mefloquine</strong> PS 1 pill 8 MON<br />
Date:02/17/12ISR Number: 8144507-2Report Type:Expedited (15-DaCompany Report #FI-ROCHE-1037088 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Anxiety Lariam PS ORAL<br />
Hospitalization - Mood Swings<br />
Initial or Prolonged Hostility<br />
Psychotic Disorder<br />
Suicidal Ideation<br />
Restlessness<br />
Depression<br />
Hallucination<br />
Agitation<br />
Fear Of Disease<br />
Confusional State<br />
Panic Disorder<br />
Date:02/21/12ISR Number: 8152664-7Report Type:Expedited (15-DaCompany Report #PHHY2012US011789 Age:48 YR Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Polyneuropathy <strong>Mefloquine</strong> PS Novartis Sector:<br />
Allodynia Generics 250 mg,<br />
Insomnia<br />
ONCE/SINGLE<br />
Depression <strong>Mefloquine</strong> SS Novartis Sector:<br />
Drug Hypersensitivity Generics 250 mg,<br />
Neuropathy Peripheral<br />
ONCE/SINGLE<br />
Pain In Extremity <strong>Mefloquine</strong> SS Novartis Sector:<br />
Pyrexia Generics 250 mg,<br />
Neck Pain<br />
ONCE/SINGLE<br />
Facial Pain <strong>Mefloquine</strong> SS Novartis Sector:<br />
Paraesthesia Generics 250 mg,<br />
Hypoaesthesia<br />
ONCE/SINGLE<br />
Burning Sensation Naproxen Sodium C 550 mg, BID<br />
Gabapentin C 300 mg, UNK<br />
Ibuprofen C 600 mg, TID<br />
Acetaminophen<br />
W/Oxycodone C 2 DF, QHS as<br />
needed<br />
Gabapentin C 600 mg, TID<br />
Zolpidem Tartrate C 10 mg, QHS<br />
Esomeprazole<br />
Magnesium C 40 mg, daily
Date:02/23/12ISR Number: 8160310-1Report Type:Direct Company Report #US-<strong>FDA</strong>-8160310 Age:20 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Quality Of Life Decreased Lariam PS ORAL one pill<br />
03-Apr-2012 09:37 AM Page: 872
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:02/24/12ISR Number: 8162503-6Report Type:Direct Company Report #US-<strong>FDA</strong>-8162503 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Other Serious Depression <strong>Mefloquine</strong> PS<br />
Headache<br />
Insomnia<br />
Hallucination<br />
Suicidal Behaviour<br />
Dizziness<br />
Abnormal Dreams<br />
Abnormal Behaviour<br />
Anxiety<br />
Date:02/28/12ISR Number: 8170623-5Report Type:Expedited (15-DaCompany Report #NO-ROCHE-1042181 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Disability Dizziness Lariam PS ORAL<br />
Deafness<br />
Disability<br />
Tinnitus<br />
Date:03/06/12ISR Number: 8187252-XReport Type:Expedited (15-DaCompany Report #DE-ROCHE-769644 Age:52 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Hypertension Lariam PS UNKNOWN DAILY DOSE 1.<br />
Initial or Prolonged Nausea TOTAL TABLETS<br />
Disability Dizziness ADMINISTERED:<br />
Sinoatrial Block 245<br />
Cardiac Pacemaker<br />
Insertion<br />
Sinus Arrest<br />
Date:03/15/12ISR Number: 8212057-0Report Type:Expedited (15-DaCompany Report #DE-ROCHE-1046709 Age: Gender:Female I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaphylactic Shock Lariam PS ORAL<br />
Initial or Prolonged<br />
Date:03/22/12ISR Number: 8227789-8Report Type:Expedited (15-DaCompany Report #DE-ROCHE-1046709 Age: Gender:Female I/FU:F<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Anaphylactic Shock Lariam PS ORAL<br />
Initial or Prolonged
Date:03/23/12ISR Number: 8231170-5Report Type:Direct Company Report #US-<strong>FDA</strong>-8231170 Age:25 YR Gender:Male I/FU:I<br />
Outcome<br />
Hospitalization -<br />
Initial or Prolonged<br />
PT<br />
Nausea<br />
Depressed Level Of<br />
Consciousness<br />
Fear<br />
03-Apr-2012 09:37 AM Page: 873
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Grand Mal Convulsion<br />
Visual Impairment<br />
Vomiting Report Source Product Role Manufacturer Route Dose Duration<br />
Fatigue <strong>Mefloquine</strong> PS<br />
Amnesia<br />
Asthenia<br />
Abasia<br />
Vision Blurred<br />
Anxiety<br />
Headache<br />
Date:03/23/12ISR Number: 8231209-7Report Type:Direct Company Report #US-<strong>FDA</strong>-8231209 Age:23 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Road Traffic Accident Larium PS ORAL 1 tablet 15 MON<br />
Initial or Prolonged Drug Dependence<br />
Psychotic Disorder<br />
Feeling Abnormal<br />
Date:03/23/12ISR Number: 8231220-6Report Type:Direct Company Report #US-<strong>FDA</strong>-8231220 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Panic Attack Lariam PS<br />
Sleep Disorder<br />
Feeling Abnormal<br />
Date:03/27/12ISR Number: 8237197-1Report Type:Direct Company Report #US-<strong>FDA</strong>-8237197 Age:32 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Amnesia Lariam PS ORAL 250mg<br />
Tinnitus<br />
Irritable Bowel Syndrome<br />
Date:03/27/12ISR Number: 8237261-7Report Type:Direct Company Report #US-<strong>FDA</strong>-8237261 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Life-Threatening Sleep Disorder Larium Or <strong>Mefloquine</strong> PS<br />
Other Serious<br />
Nightmare<br />
Post-Traumatic Stress<br />
Disorder<br />
Suicidal Ideation<br />
Depression
Date:03/27/12ISR Number: 8237273-3Report Type:Direct Company Report #US-<strong>FDA</strong>-8237273 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Hospitalization - Headache Mefloquin PS ORAL 15-25 mg<br />
Initial or Prolonged Hyperhidrosis<br />
Other Serious<br />
Tremor<br />
Memory Impairment<br />
03-Apr-2012 09:37 AM Page: 874
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Date:03/28/12ISR Number: 8240288-2Report Type:Direct Company Report #US-<strong>FDA</strong>-8240288 Age:30 YR Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Amnesia Lariam/<strong>Mefloquine</strong> PS ORAL 250 mg<br />
Vertigo<br />
Tinnitus<br />
Suicidal Ideation<br />
Tremor<br />
Abnormal Dreams<br />
Sleep Disorder<br />
Date:03/28/12ISR Number: 8240324-3Report Type:Direct Company Report #US-<strong>FDA</strong>-8240324 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Unevaluable Event Larium PS ORAL 1 pill<br />
Date:03/28/12ISR Number: 8240330-9Report Type:Direct Company Report #US-<strong>FDA</strong>-8240330 Age: Gender:Male I/FU:I<br />
Outcome PT Report Source Product Role Manufacturer Route Dose Duration<br />
Abnormal Dreams <strong>Mefloquine</strong> PS ORAL Dont remember<br />
Anxiety
03-Apr-2012 09:37 AM Page: 875
<strong>FDA</strong> - Adverse Event Reporting System (<strong>AERS</strong>)<br />
Freedom Of Information (FOI) Report<br />
Summary report for FOI selections:<br />
Selection by inexact search of active ingredient:<br />
MEFLOQUINE%<br />
Selection by inexact search of Tradename/Verbatim:<br />
LARIAM%<br />
Total number of reports: 3,384<br />
From: 01-NOV-1997 To: 03-APR-2012
03-Apr-2012 09:37 AM Page: 876