Effects of heat release in laminar diffusion flames - Mechanical ...
Effects of heat release in laminar diffusion flames - Mechanical ...
Effects of heat release in laminar diffusion flames - Mechanical ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
for the Landau-Squire jet discussed above. This procedure<br />
saves a great deal <strong>of</strong> computational time and<br />
allows for compar<strong>in</strong>g the cold-flow theory <strong>of</strong> lifted<br />
<strong>flames</strong> with simulations under the same framework.<br />
But on the other hand, the predictive capabilities <strong>of</strong><br />
the simulations are restricted. Only the correction to<br />
the lift-<strong>of</strong>f height, h, due to the impact <strong>of</strong> <strong>heat</strong> <strong>release</strong><br />
effects can be computed. In addition, the computations<br />
would not be reliable <strong>in</strong> describ<strong>in</strong>g <strong>flames</strong> lifted<br />
very close to the burner, because, the self-similar<br />
solution is not valid <strong>in</strong> the close field <strong>of</strong> the round jet.<br />
The full simulation would require a careful description<br />
<strong>of</strong> both the lam<strong>in</strong>ar round jet boundary layers<br />
and the far field, a task that is difficult to achieve<br />
together with the accurate fully compressible Direct<br />
Numerical Simulation (DNS) <strong>of</strong> the flame base.<br />
Numerical simulations <strong>of</strong> lam<strong>in</strong>ar lifted <strong>flames</strong> are<br />
performed us<strong>in</strong>g a DNS tool previously developed<br />
for study<strong>in</strong>g turbulent combustion [35–38]. The DNS<br />
procedure follows well established methods [39].<br />
The fully compressible Navier Stokes equations are<br />
solved us<strong>in</strong>g a sixth order PADE scheme [40] comb<strong>in</strong>ed<br />
with third order Runge-Kutta time stepp<strong>in</strong>g and<br />
Navier Stokes characteristic boundary conditions<br />
(NSCBC) [41]. The possibility <strong>of</strong> deal<strong>in</strong>g with axi-<br />
J. Boulanger et al. / Combustion and Flame 134 (355–368)<br />
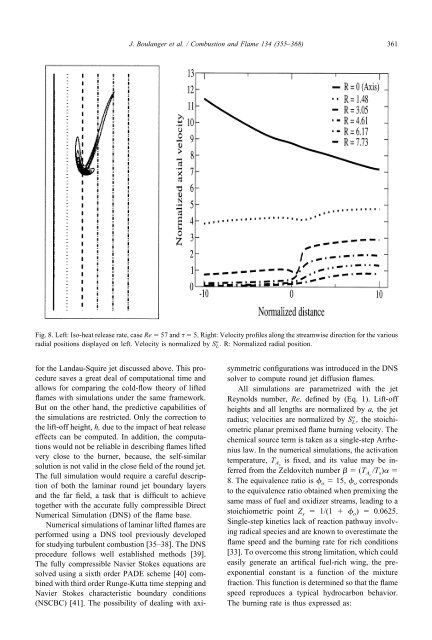
Fig. 8. Left: Iso-<strong>heat</strong> <strong>release</strong> rate, case Re � 57 and � � 5. Right: Velocity pr<strong>of</strong>iles along the streamwise direction for the various<br />
radial positions displayed on left. Velocity is normalized by S L o . R: Normalized radial position.<br />
361<br />
symmetric configurations was <strong>in</strong>troduced <strong>in</strong> the DNS<br />
solver to compute round jet <strong>diffusion</strong> <strong>flames</strong>.<br />
All simulations are parametrized with the jet<br />
Reynolds number, Re, def<strong>in</strong>ed by (Eq. 1). Lift-<strong>of</strong>f<br />
heights and all lengths are normalized by a, the jet<br />
o<br />
radius; velocities are normalized by SL, the stoichiometric<br />
planar premixed flame burn<strong>in</strong>g velocity. The<br />
chemical source term is taken as a s<strong>in</strong>gle-step Arrhenius<br />
law. In the numerical simulations, the activation<br />
temperature, TAc is fixed, and its value may be <strong>in</strong>ferred<br />
from the Zeldovitch number � � (TAc /Ts)� �<br />
8. The equivalence ratio is �o � 15, �o corresponds<br />
to the equivalence ratio obta<strong>in</strong>ed when premix<strong>in</strong>g the<br />
same mass <strong>of</strong> fuel and oxidizer streams, lead<strong>in</strong>g to a<br />
stoichiometric po<strong>in</strong>t Zs � 1/(1 � �o) � 0.0625.<br />
S<strong>in</strong>gle-step k<strong>in</strong>etics lack <strong>of</strong> reaction pathway <strong>in</strong>volv<strong>in</strong>g<br />
radical species and are known to overestimate the<br />
flame speed and the burn<strong>in</strong>g rate for rich conditions<br />
[33]. To overcome this strong limitation, which could<br />
easily generate an artifical fuel-rich w<strong>in</strong>g, the preexponential<br />
constant is a function <strong>of</strong> the mixture<br />
fraction. This function is determ<strong>in</strong>ed so that the flame<br />
speed reproduces a typical hydrocarbon behavior.<br />
The burn<strong>in</strong>g rate is thus expressed as: