Brazilian Journal of Analytical Chemistry - BRJAC - Brazilian Journal ...
Brazilian Journal of Analytical Chemistry - BRJAC - Brazilian Journal ...
Brazilian Journal of Analytical Chemistry - BRJAC - Brazilian Journal ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
edua r d o sidinei ch av e s, Ju l i a n o ca rva l h o ra m o s , Kl a i a n i bez Fo n ta n a, sa m u e l mo d o l o n, a n d ad i l s o n Jo s é cu rt i u s<br />
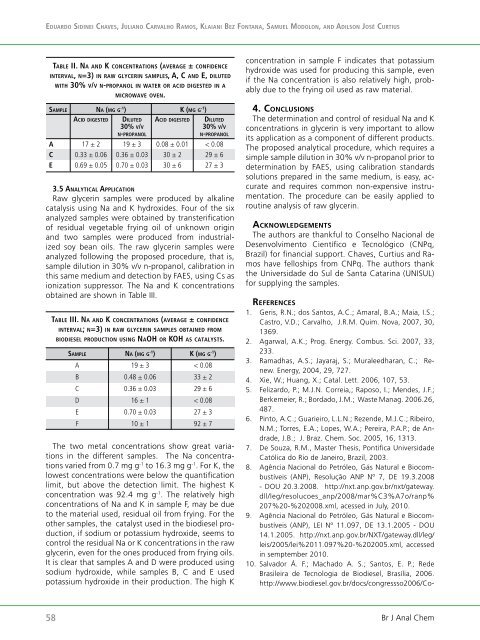
Ta b l e ii. na an d k C o n C e n T r a T i o n s (a v e r a g e ± C o n f i d e n C e<br />
i n T e r v a l, n=3) in r a w gl y C e r i n samples, a, C a n d e, d i l u T e d<br />
w iT h 30% v/v n-pr o p a n o l in wa T e r or aC i d digesTed in a<br />
m iC r o w a v e ov e n.<br />
sa m p l e na (m g g -1 ) k (m g g -1 )<br />
aCid digesTed di l u T e d aCid digesTed di l u T e d<br />
30% v/v 30% v/v<br />
n-p r o p a n o l n-p r o p a n o l<br />
A 17 ± 2 19 ± 3 0.08 ± 0.01 < 0.08<br />
C 0.33 ± 0.06 0.36 ± 0.03 30 ± 2 29 ± 6<br />
E 0.69 ± 0.05 0.70 ± 0.03 30 ± 6 27 ± 3<br />
3.5 an a l y T i C a l ap pl iC a T i on<br />
Raw glycerin samples were produced by alkaline<br />
catalysis using Na and K hydroxides. Four <strong>of</strong> the six<br />
analyzed samples were obtained by transterification<br />
<strong>of</strong> residual vegetable frying oil <strong>of</strong> unknown origin<br />
and two samples were produced from industrialized<br />
soy bean oils. The raw glycerin samples were<br />
analyzed following the proposed procedure, that is,<br />
sample dilution in 30% v/v n-propanol, calibration in<br />
this same medium and detection by FAES, using Cs as<br />
ionization suppressor. The Na and K concentrations<br />
obtained are shown in Table III.<br />
Ta b l e iii. na an d k C o n C e n T r a T i o n s (a v e r a g e ± C o n f i d e n C e<br />
i n T e r v a l; n=3) in r a w gl y C e r i n samples ob T a i n e d fr o m<br />
biodiesel pr o d u C T i o n us i n g naoh o r koh a s Ca T a l y s T s.<br />
sa m p l e na (m g g -1 ) k (m g g -1 )<br />
A 19 ± 3 < 0.08<br />
B 0.48 ± 0.06 33 ± 2<br />
C 0.36 ± 0.03 29 ± 6<br />
D 16 ± 1 < 0.08<br />
E 0.70 ± 0.03 27 ± 3<br />
F 10 ± 1 92 ± 7<br />
The two metal concentrations show great variations<br />
in the different samples. The Na concentrations<br />
varied from 0.7 mg g -1 to 16.3 mg g -1 . For K, the<br />
lowest concentrations were below the quantification<br />
limit, but above the detection limit. The highest K<br />
concentration was 92.4 mg g -1 . The relatively high<br />
concentrations <strong>of</strong> Na and K in sample F, may be due<br />
to the material used, residual oil from frying. For the<br />
other samples, the catalyst used in the biodiesel production,<br />
if sodium or potassium hydroxide, seems to<br />
control the residual Na or K concentrations in the raw<br />
glycerin, even for the ones produced from frying oils.<br />
It is clear that samples A and D were produced using<br />
sodium hydroxide, while samples B, C and E used<br />
potassium hydroxide in their production. The high K<br />
concentration in sample F indicates that potassium<br />
hydroxide was used for producing this sample, even<br />
if the Na concentration is also relatively high, probably<br />
due to the frying oil used as raw material.<br />
4. co n c l u s I o n s<br />
The determination and control <strong>of</strong> residual Na and K<br />
concentrations in glycerin is very important to allow<br />
its application as a component <strong>of</strong> different products.<br />
The proposed analytical procedure, which requires a<br />
simple sample dilution in 30% v/v n-propanol prior to<br />
determination by FAES, using calibration standards<br />
solutions prepared in the same medium, is easy, accurate<br />
and requires common non-expensive instrumentation.<br />
The procedure can be easily applied to<br />
routine analysis <strong>of</strong> raw glycerin.<br />
AcK n o w l e d g e m e n t s<br />
The authors are thankful to Conselho Nacional de<br />
Desenvolvimento Científico e Tecnológico (CNPq,<br />
Brazil) for financial support. Chaves, Curtius and Ramos<br />
have felloships from CNPq. The authors thank<br />
the Universidade do Sul de Santa Catarina (UNISUL)<br />
for supplying the samples.<br />
ref e re n c e s<br />
1. Geris, R.N.; dos Santos, A.C.; Amaral, B.A.; Maia, I.S.;<br />
Castro, V.D.; Carvalho, J.R.M. Quim. Nova, 2007, 30,<br />
1369.<br />
2. Agarwal, A.K.; Prog. Energy. Combus. Sci. 2007, 33,<br />
233.<br />
3. Ramadhas, A.S.; Jayaraj, S.; Muraleedharan, C.; Renew.<br />
Energy, 2004, 29, 727.<br />
4. Xie, W.; Huang, X.; Catal. Lett. 2006, 107, 53.<br />
5. Felizardo, P.; M.J.N. Correia,; Raposo, I.; Mendes, J.F.;<br />
Berkemeier, R.; Bordado, J.M.; Waste Manag. 2006.26,<br />
487.<br />
6. Pinto, A.C.; Guarieiro, L.L.N.; Rezende, M.J.C.; Ribeiro,<br />
N.M.; Torres, E.A.; Lopes, W.A.; Pereira, P.A.P.; de Andrade,<br />
J.B.; J. Braz. Chem. Soc. 2005, 16, 1313.<br />
7. De Souza, R.M., Master Thesis, Pontifica Universidade<br />
Católica do Rio de Janeiro, Brazil, 2003.<br />
8. Agência Nacional do Petróleo, Gás Natural e Biocombustíveis<br />
(ANP), Resolução ANP Nº 7, DE 19.3.2008<br />
- DOU 20.3.2008. http://nxt.anp.gov.br/nxt/gateway.<br />
dll/leg/resolucoes_anp/2008/mar%C3%A7o/ranp%<br />
207%20-%202008.xml, acessed in July, 2010.<br />
9. Agência Nacional do Petróleo, Gás Natural e Biocombustíveis<br />
(ANP), LEI Nº 11.097, DE 13.1.2005 - DOU<br />
14.1.2005. http://nxt.anp.gov.br/NXT/gateway.dll/leg/<br />
leis/2005/lei%2011.097%20-%202005.xml, accessed<br />
in semptember 2010.<br />
10. Salvador Á. F.; Machado A. S.; Santos, E. P.; Rede<br />
Brasileira de Tecnologia de Biodiesel, Brasília, 2006.<br />
http://www.biodiesel.gov.br/docs/congressso2006/Co-<br />
58 Br J Anal Chem