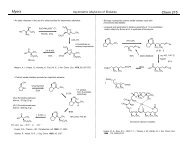
The Olefin Metathesis Reaction
The Olefin Metathesis Reaction
The Olefin Metathesis Reaction
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Myers<br />
Reviews:<br />
Nicolaou, K. C.; Bulger, P. G.; Sarlah, D. Angew. Chem., Int. Ed. Engl. 2005, 44, 4490–4527.<br />
Grubbs, R. H. Tetrahedron 2004, 60, 7117–7140.<br />
Chatterjee, A. K.; Choi, T.-L.; Sanders, D. P.; Grubbs, R. H.<br />
J. Am. Chem. Soc. 2003, 125, 11360–11370.<br />
Connon, S. J.; Blechert, S. Angew. Chem., Int. Ed. Engl. 2003, 42, 1900–1923.<br />
Fürstner, A. Angew. Chem., Int. Ed. Engl. 2000, 39, 3013–3043.<br />
Grubbs, R. H.; Chang, S. Tetrahedron 1998, 54, 4413–4450.<br />
Armstrong, S. K. J. Chem. Soc., Perkin Trans. 1 1998, 371–388.<br />
Ring-Opening <strong>Metathesis</strong> Polymerization (ROMP):<br />
ROMP<br />
• ROMP is thermodynamically favored for strained ring systems, such as 3-, 4-, 8- and<br />
larger-membered compounds.<br />
• When bridging groups are present (bicyclic olefins) the ΔG of polymerization is typically<br />
more negative as a result of increased strain energy in the monomer.<br />
• Block copolymers can be made by sequential addition of different monomers (a<br />
consequence of the "living" nature of the polymerization).<br />
Ring-Closing <strong>Metathesis</strong> (RCM):<br />
RCM<br />
• <strong>The</strong> reaction can be driven to the right by the loss of ethylene.<br />
n<br />
H 2C CH 2<br />
• <strong>The</strong> development of well-defined metathesis catalysts that are tolerant of many functional<br />
groups yet reactive toward a diverse array of olefinic substrates has led to the rapid<br />
acceptance of the RCM reaction as a powerful method for forming carbon-carbon double<br />
bonds and for macrocyclizations.<br />
• Where the thermodynamics of the closure reaction are unfavorable, polymerization of the<br />
substrate can occur. This partitioning is sensitive to substrate, catalyst, and reaction<br />
conditions.<br />
+<br />
<strong>The</strong> <strong>Olefin</strong> <strong>Metathesis</strong> <strong>Reaction</strong> Chem 215<br />
Cross <strong>Metathesis</strong> (CM):<br />
R 1<br />
R 2<br />
R3 R4 R1 R3 CM<br />
+ +<br />
• Self-dimerization reactions of the more valuable alkene may be minimized by the use of<br />
an excess of the more readily available alkene.<br />
Catalysts<br />
i-Pr i-Pr<br />
N<br />
F3C CH3 O Mo Ph<br />
F3C CH O CH3 3<br />
H<br />
F3C F<br />
CH3 3C 1-Mo<br />
Cl<br />
Cl<br />
P(c-Hex) 3<br />
Ru<br />
H<br />
P(c-Hex) 3<br />
2-Ru<br />
Cl<br />
Cl<br />
P(c-Hex) 3<br />
Ru<br />
Ph<br />
H<br />
P(c-Hex) 3<br />
3-Ru<br />
(Grubbs' 1st<br />
Generation Catalyst)<br />
• <strong>The</strong> well-defined catalysts shown above have been used widely for the olefin<br />
metathesis reaction. Titanium- and tungsten-based catalysts have also been developed<br />
but are less used.<br />
• Schrock's alkoxy imidomolybdenum complex 1-Mo is highly reactive toward a broad range<br />
of substrates; however, this Mo-based catalyst has moderate to poor functional group<br />
tolerance, high sensitivity to air, moisture or even to trace impurities present in solvents,<br />
and exhibits thermal instability.<br />
• Grubbs' Ru-based catalysts exhibit high reactivity in a variety of ROMP, RCM, and CM<br />
processes and show remarkable tolerance toward many different organic functional<br />
groups.<br />
Ph<br />
Ph<br />
• <strong>The</strong> electron-rich tricyclohexyl phosphine ligands of the d6 Ru(II) metal center in<br />
alkylidenes 2-Ru and 3-Ru leads to increased metathesis activity. <strong>The</strong> NHC ligand in<br />
4-Ru is a strong -donor and a poor -acceptor and stabilizes a 14 e – σ π<br />
Ru intermediate in<br />
the catalytic cycle, making this catalyst more effective than 2-Ru or 3-Ru.<br />
• Ru-based catalysts show little sensitivity to air, moisture or minor impurities in solvents.<br />
<strong>The</strong>se catalysts can be conveniently stored in the air for several weeks without<br />
decomposition. All of the catalysts above are commerically available, but 1-Mo is<br />
significantly more expensive.<br />
R 2<br />
MesN<br />
Cl<br />
Cl<br />
R 4<br />
NMes<br />
Ph<br />
Ru<br />
H<br />
P(c-Hex) 3<br />
4-Ru<br />
(Grubbs' 2nd<br />
Generation Catalyst)<br />
Scholl, M.; Ding, S.; Lee, C. W.; Grubbs, R. H. Org. Lett. 1999, 1, 953–956.<br />
Schwab, P.; France, M. B.; Ziller, J. W.; Grubbs, R. H. Angew. Chem., Int. Ed Engl. 1995,<br />
34, 2039–2041.<br />
Nguyen, S.-B. T.; Grubbs, R. H. J. Am. Chem. Soc. 1993, 115, 9858–9859.<br />
M. Movassaghi and L. Blasdel
Mechanism:<br />
• <strong>The</strong> olefin metathesis reaction was reported as early as 1955 in a Ti(II)-catalyzed<br />
polymerization of norbornene: Anderson, A. W.; Merckling, M. G. Chem. Abstr. 1955,<br />
50, 3008i.<br />
• 15 years later, Chauvin first proposed that olefin metathesis proceeds via<br />
metallacyclobutanes: Herisson, P. J.-L.; Chauvin, Y. Makromol. Chem. 1970, 141,<br />
161–176.<br />
• It is now generally accepted that both cyclic and acyclic olefin metathesis reactions<br />
proceed via metallacyclobutane and metal-carbene intermediates: Grubbs, R. H.; Burk,<br />
P. L.; Carr, D. D. J. Am. Chem. Soc. 1975, 97, 3265–3266.<br />
EtO 2C<br />
CO 2Et<br />
Cl<br />
P(c-Hex) 3<br />
H<br />
Ru<br />
Cl H<br />
P(c-Hex) 3<br />
5 mol%<br />
CD 2Cl 2, 25 °C<br />
EtO 2C CO 2Et<br />
• A kinetic study of the RCM of diethyl diallylmalonate using a Ru-methylidene describes<br />
two possible mechanisms for olefin metathesis:<br />
• <strong>The</strong> "dissociative" mechanism assumes that upon binding of the olefin a phosphine is<br />
diplaced from the metal center to form a 16-electron olefin complex, which undergoes<br />
metathesis to form the cyclized product, regenerating the catalyst upon recoordination of<br />
the phosphine.<br />
• <strong>The</strong> "associative" mechanism assumes that an 18-electron olefin complex is formed<br />
which undergoes metathesis to form the cyclized product.<br />
• Addition of 1 equivalent of phosphine (with respect to catalyst) decreases the rate of the<br />
reaction by as much 20 times, supporting the dissociative mechanism.<br />
• It was concluded in this study that the "dissociative" pathway is the dominant reaction<br />
manifold (>95%).<br />
Dias, E. L.; Nguyen, S.-B. T.; Grubbs, R. H. J. Am. Chem. Soc. 1997, 119, 3887–3897.<br />
Dissociative:<br />
Cl<br />
Cl<br />
R<br />
Cl<br />
R<br />
Cl<br />
Cl<br />
Cl<br />
P<br />
Ru<br />
P<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
P<br />
Ru<br />
P<br />
H<br />
H<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
EtO 2C CO 2Et<br />
Associative:<br />
H<br />
H<br />
Cl<br />
R<br />
R<br />
c-C 5H 6(CO 2Et) 2<br />
R<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
EtO 2C CO 2Et<br />
–P<br />
+P<br />
Cl<br />
c-C 5H 6(CO 2Et) 2<br />
Cl<br />
P<br />
Cl H<br />
Ru<br />
H<br />
Cl<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
P<br />
Cl H<br />
Ru<br />
H<br />
EtO 2C CO 2Et<br />
Cl<br />
P = P(c-Hex) 3<br />
EtO2C R<br />
Cl<br />
H<br />
R<br />
Cl<br />
H<br />
R<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
EtO 2C CO 2Et<br />
Cl<br />
=<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
Cl H<br />
Ru<br />
H<br />
EtO 2C CO 2Et<br />
P<br />
Cl H<br />
Ru<br />
H<br />
P<br />
Cl<br />
CO 2Et<br />
Cl<br />
H<br />
Cl<br />
R<br />
Cl<br />
H<br />
R<br />
P<br />
Cl<br />
Ru<br />
P<br />
Cl<br />
Ru<br />
EtO 2C CO 2Et<br />
P<br />
Cl<br />
Ru<br />
P<br />
EtO 2C CO 2Et<br />
P<br />
Cl<br />
Ru<br />
P<br />
– C 2H 4<br />
– C 2H 4<br />
M. Movassaghi
Catalytic RCM of Dienes:<br />
substrate product time (h)<br />
O<br />
N X<br />
O Ph<br />
O Ph<br />
O<br />
O<br />
O<br />
R<br />
Ph<br />
Ph<br />
X = CF 3<br />
X = Ot-Bu<br />
R = CO 2H<br />
CH 2OH<br />
CHO<br />
a 2-4 mol% 2-Ru, C6H 6, 20 °C.<br />
O<br />
O Ph<br />
O<br />
O<br />
O<br />
N X<br />
O<br />
Ph<br />
Ph<br />
PhCH2 H<br />
N Cl –<br />
+ 4 mol% 2-Ru<br />
R<br />
Ph<br />
20 °C, 36 h<br />
CH 2Cl 2; NaOH<br />
79%<br />
1<br />
1<br />
CH2Ph N<br />
yield (%) a<br />
93<br />
91<br />
2 84<br />
5 86<br />
8 72<br />
1 87<br />
• Five-, six-, and seven-membered oxygen and nitrogen heterocycles and cycloalkanes are<br />
formed efficiently.<br />
• Catalyst 2-Ru can be used in the air, in reagent-grade solvents (C 6H 6, CH 2Cl 2, THF,<br />
t-BuOH).<br />
• In contrast to the molybdenum catalyst 1-Mo, which is known to react with acids, alcohols,<br />
and aldehydes, the ruthenium catalyst 2-Ru is stable to these functional groups.<br />
• Free amines are not tolerated by the ruthenium catalyst; the corresponding hydrochloride<br />
salts undergo efficient RCM with catalyst 2-Ru.<br />
Fu, G. C.; Nguyen, S.-B. T.; Grubbs, R. H. J. Am. Chem. Soc. 1993, 115, 9856–9857.<br />
1<br />
1<br />
1<br />
87<br />
88<br />
82<br />
Synthesis of Tri- and Tetrasubstituted Cyclic <strong>Olefin</strong>s via RCM<br />
substrate a<br />
E E R<br />
E E<br />
E E<br />
E E<br />
CH 3<br />
E E CH 3<br />
CH 3<br />
CH3E E<br />
E E<br />
R = CH<br />
E E<br />
3<br />
93<br />
CH 3<br />
CH 3<br />
i-Pr<br />
CH 3<br />
t-Bu<br />
Ph<br />
Br<br />
CH 2OH<br />
H 3C<br />
product<br />
H 3C<br />
E<br />
R<br />
E E<br />
CH3 E E<br />
E E<br />
E E<br />
E<br />
CH 3<br />
CH 3<br />
CH 3<br />
yield<br />
with 3-Ru (%) b<br />
98<br />
NR<br />
25<br />
NR<br />
98<br />
No RCM d<br />
NR<br />
NR<br />
yield<br />
with 1-Mo (%) c<br />
100<br />
100<br />
96<br />
97<br />
NR<br />
decomp<br />
97 100<br />
96 100<br />
No RCM d<br />
a E = CO2Et. b 0.01 M, CH 2Cl 2, 5 mol%. c 0.1 M, C 6H 6, 5 mol%. d Only<br />
recovered starting material and an acyclic dimer were observed. e <strong>The</strong><br />
isomeric cyclopentene product is not observed.<br />
93<br />
61<br />
96 e 100 e<br />
• Functional group compatibility permitting, the Mo-alkylidene catalyst is typically more<br />
effective for RCM of substituted olefins.<br />
Kirkland, T. A.; Grubbs, R. H. J. Org. Chem. 1997, 62, 7310–7318.<br />
–<br />
M. Movassaghi
Geminal Substitution<br />
H3C CH3 Si<br />
n<br />
O<br />
R<br />
O<br />
R R<br />
R R<br />
RCM in Methanol and Water<br />
Ph<br />
Cl<br />
Cl<br />
P(c-Hex) 3<br />
Ph<br />
Ru<br />
H<br />
P(c-Hex) 3<br />
3-Ru<br />
RuL n<br />
R<br />
Cl<br />
Cl<br />
R<br />
P<br />
Ru<br />
P<br />
6-Ru<br />
L nRu<br />
N(CH 3) 3 + Cl –<br />
Ph<br />
H<br />
N(CH +<br />
3) 3 Cl –<br />
R<br />
Cl –<br />
H3C CH3 N<br />
+<br />
Cl<br />
Cl<br />
H 3C<br />
P<br />
Ru<br />
P<br />
N Cl –<br />
+<br />
7-Ru<br />
• Alkylidenes 6-Ru and 7-Ru are well-defined, water-soluble Ru-based metathesis catalysts<br />
that are stable for days in methanol or water at 45 °C.<br />
• Although benzylidene 3-Ru is highly active in RCM of dienes in organic solvents, it has no<br />
catalytic acitivity in protic media.<br />
L nRu<br />
Ph<br />
H<br />
EtO 2C<br />
CO 2Et<br />
Ph<br />
solvent:<br />
5 mol% 3-Ru<br />
23 °C<br />
CH 2Cl 2<br />
CH 3OH<br />
EtO 2C<br />
100%<br />
95<br />
methanol<br />
methanol<br />
methanol<br />
water<br />
water<br />
6-Ru<br />
7-Ru<br />
6-Ru<br />
7-Ru<br />
7-Ru<br />
7-Ru<br />
7-Ru<br />
a E = CO2Et. b 5 mol% catalyst (6- or 7-Ru), 0.37 M substrate, 45 °C. c Conversions were<br />
determined by 1 H NMR. d Substrate conc. = 0.1 M. e 30 h. f 2 h. g 10 mol% 7-Ru used.<br />
• Alkylidene 7-Ru is a significantly more active catalyst than alkylidene 6-Ru in these<br />
cyclizations; this higher reactivity is attributed to the more electron-rich phosphines in 7-Ru.<br />
• Cis-olefins are more reactive in RCM than the corresponding trans-olefins.<br />
• Phenyl substitution within the starting material can also greatly increase the yield of RCM in<br />
organic solvents.<br />
5 mol% 3-Ru<br />
CH 2Cl 2<br />
R = H<br />
R = Ph<br />
H H<br />
N +<br />
60%<br />
100%<br />
Kirkland, T. A.; Lynn, D. M.; Grubbs, R. H. J. Org. Chem. 1998, 63, 9904–9909.<br />
M. Movassaghi<br />
Cl –<br />
conversion c<br />
80<br />
95<br />
40<br />
90 e<br />
30<br />
>95 f<br />
90<br />
60<br />
90 g
NHC Ruthenium Catalysts:<br />
Mes N N Mes Mes N N Mes<br />
Cl<br />
Cl<br />
Ru<br />
Ph<br />
H<br />
P(c-Hex) 3<br />
Cl<br />
Cl<br />
Ru<br />
Ph<br />
H<br />
P(c-Hex) 3<br />
Mes N N Mes<br />
Cl<br />
Cl<br />
Ru<br />
8-Ru 4-Ru 9-Ru<br />
substrate a<br />
E E t-Bu<br />
E E CH 3<br />
CH 3<br />
CH3E E<br />
H OH<br />
CH 3<br />
H 3C<br />
H 3C<br />
product<br />
E E<br />
E E<br />
E E<br />
H OH<br />
t-Bu<br />
CH 3<br />
CH 3<br />
time<br />
(h)<br />
1 37<br />
Ph<br />
H<br />
P(c-Hex) 3<br />
a E = CO2Et. b 5 mol% of catalyst, CD 2Cl 2, reflux. c 1.5 h.<br />
yield of product (%) using catalyst: b<br />
1-Mo 3-Ru 8-Ru 4-Ru 9-Ru<br />
0 100 100 100<br />
24 93 0 40 31 55<br />
c<br />
1.5 52 0 95 90 87<br />
0.2 0 0 NA 100 100<br />
• Alkylidenes 4- and 9-Ru are the most reactive Ru-based catalysts.<br />
• In the case of 4- and 9-Ru as little as 0.05 mol% is sufficient for efficient RCM.<br />
Scholl, M.; Ding, S.; Lee, C.-W.; Grubbs, R. H. Org. Lett. 1999,1, 953-956.<br />
Scholl, M.; Trnka, T. M.; Morgan, J. P.; Grubbs, R. H. Tetrahedron Lett. 1999, 40, 2247–2250.<br />
For the first Ru-based metathesis catalyst employing the Arduengo carbene ligand, see:<br />
Weskamp, T.; Schattenmann, W. C.; Spiegler, M.; Herrmann, W. A. Angew. Chem., Int. Ed.<br />
Engl. 1998, 37, 2490–2493.<br />
Mes<br />
Cl<br />
Cl<br />
N N Mes<br />
Ru<br />
P(c-Hex) 3<br />
10-Ru<br />
H<br />
CH 3<br />
CH 3<br />
RCM of functionalized dienes<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
diene product yield (%)<br />
O<br />
O<br />
CH 2<br />
CH 2<br />
CH 2<br />
CH 2<br />
CH 2<br />
CH 2<br />
CH 2<br />
CH 2<br />
CH 3<br />
a <strong>Reaction</strong>s conducted with 5 mol% 10-Ru.<br />
O<br />
O<br />
O<br />
• Substrates containing both allyl and vinyl ethers provide RCM products while no RCM<br />
• α,β-Unsaturated lactones and enones of various ring sizes are produced in good to<br />
Chatterjee, A. K.; Morgan, J. P.; Scholl, M.; Grubbs, R. H. J. Am. Chem. Soc. 2000, 122,<br />
3783–3784.<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
CH 3<br />
products are observed if vinyl ethers alone are present.<br />
excellent yields.<br />
49<br />
0<br />
97<br />
86<br />
93<br />
M. Movassaghi
O<br />
Bn<br />
BnO<br />
RCM Applications in Synthesis:<br />
N<br />
O<br />
O<br />
BnO<br />
H<br />
BnO CO2CH3 O<br />
N<br />
1. n-Bu 2BOTf, Et 3N<br />
CH 2Cl 2, 0 °C<br />
2. CH 2=CHCHO<br />
–78 → 0 °C<br />
82%, >99% de<br />
5 mol% 2-Ru<br />
110 °C, 48 h<br />
70%<br />
BnO<br />
H<br />
BnO<br />
BnO<br />
O<br />
O<br />
Bn<br />
N<br />
Bn<br />
O<br />
O<br />
N<br />
O<br />
O OH<br />
Crimmins, M. T.; King, B. W. J. Org. Chem. 1996, 61, 4192–4193.<br />
Overkleeft, H. S.; Pandit, U. K. Tetrahedron Lett. 1996, 37, 547–550.<br />
O<br />
N<br />
OH<br />
HO<br />
HO<br />
1 mol% 3-Ru<br />
CH 2Cl 2<br />
97%<br />
HO<br />
H OH<br />
N<br />
Castanospermine<br />
• Particularly difficult cyclizations (due to steric congestion or electronic deactivation) can be<br />
achieved by relay ring closing metathesis, which initiates catalysis at an isolated terminal<br />
olefin. <strong>The</strong> reaction is driven by release of cyclopentene.<br />
Hoye, T. R.; Jeffrey, C. S.; Tennakoon, M. A.; Wang, J.; Zhao, H. J. Am. Chem. Soc.<br />
2004, 126, 10210–10211.<br />
TBSO<br />
O<br />
H 3C<br />
O<br />
OPMB<br />
O<br />
3<br />
10 mol% 5-Ru<br />
CH 2Cl 2, 40 °C<br />
TBSO<br />
O<br />
H 3C<br />
O<br />
OPMB<br />
O<br />
RuL n<br />
TBSO<br />
O<br />
O<br />
OPMB<br />
Wang, X.; Bowman, E. J.; Bowman, B. J.; Porco, J. A., Jr. Angew. Chem. Int. Ed. 2004, 43,<br />
3601–3605.<br />
71%<br />
O<br />
trans epoxide<br />
MOMO<br />
MOMO<br />
MOMO<br />
cis epoxide<br />
MOMO<br />
O<br />
O<br />
O CH 3<br />
O<br />
O<br />
H<br />
O CH3 H<br />
O<br />
H<br />
O<br />
HO<br />
H<br />
OH<br />
Cl<br />
O<br />
O CH 3<br />
O<br />
HO<br />
Pochonin C<br />
5 mol% 4-Ru<br />
toluene, 120 °C<br />
10 min<br />
87%<br />
5 mol% 4-Ru<br />
toluene, 120 °C<br />
10 min<br />
21%<br />
Cl<br />
MOMO<br />
MOMO<br />
MOMO<br />
MOMO<br />
O<br />
O CH 3<br />
O<br />
O<br />
H<br />
O CH3 H<br />
O<br />
O<br />
H<br />
• Pre-organization of the substrate can have a dramatic effect upon the reaction efficiency.<br />
• Both epoxide substrates produce macrocycles with good regioselectivity (i.e., the<br />
14-membered ring rather than the 12-membered ring) and E/Z selectivity. However, the<br />
trans epoxide macrocycle is formed in a much higher yield.<br />
Barluenga, S.; Lopez, P.; Moulin, E.; Winssinger, N. Angew. Chem. Int. Ed. 2004, 43,<br />
2367–2370.<br />
O<br />
H<br />
L. Blasdel and M. Movassaghi
Ph<br />
N<br />
O<br />
N<br />
O<br />
CO<br />
H<br />
2CH3 H<br />
N<br />
H CH 2OTDS<br />
N O<br />
O<br />
D<br />
O<br />
N<br />
O<br />
N<br />
H<br />
N<br />
N<br />
H<br />
OH<br />
100 mol% 2-Ru<br />
23 °C, 5 d<br />
C 6D 6<br />
30%<br />
Ph<br />
N<br />
O<br />
N<br />
O<br />
H CH 2OTDS<br />
N O<br />
Borer, B. C.; Deerenberg, S.; Bieraugel, H.; Pandit, U. K. Tetrahedron Lett. 1994, 35,<br />
3191–3194.<br />
5 mol% 1-Mo<br />
50 °C, 4 h<br />
C 6H 6<br />
63%<br />
CO<br />
H<br />
2CH3 N<br />
H<br />
Martin, S. F.; Liao, Y.; Wong, Y.; Rein, T. Tetrahedron Lett. 1994, 35, 691–694.<br />
E<br />
Manzamine A<br />
• <strong>The</strong> use of RCM in construction of both the D and the E rings of Manzamine A<br />
has been reported:<br />
O<br />
O<br />
H 3C<br />
CH3 CH3 20 mol% 1-Mo<br />
OAc<br />
O<br />
H<br />
O<br />
22 °C, 10 h<br />
NHCOCF3 C6H6 H<br />
O<br />
OAc<br />
H3C 91%<br />
N CH3 H<br />
N<br />
H<br />
CH 3<br />
O<br />
H<br />
O<br />
CH 3<br />
CH3 OAc<br />
O<br />
NHCOCF3 H<br />
OAc<br />
• Before the advent of NHC ligands, 1-Mo was used more frequently than the Ru catalysts<br />
for macrocyclization of trisubstituted olefins. <strong>The</strong> latter catalysts are typically less reactive<br />
with sterically hindered substrates.<br />
Zhongmin, X.; Johannes, C. W.; Houri, A. F.; La, D. S.; Cogan, D. A.; Hofilena, G. E.;<br />
Hoveyda, A. H. J. Am. Chem. Soc. 1997, 119, 10302–10316.<br />
Slight changes in substrate structure can control whether the E- or Z-olefin is formed:<br />
H 3C<br />
O<br />
OCH 3 O<br />
O<br />
OP<br />
H 3C<br />
CH 3<br />
CH 2<br />
CH 3<br />
CH 3<br />
H 3C<br />
O<br />
O<br />
O<br />
OCH3 OP<br />
H 3C<br />
CH 3<br />
CH 2<br />
86% 10 mol% 4-Ru<br />
80%<br />
E-olefin only CH2Cl2, 40 °C<br />
Z-olefin only<br />
O<br />
O<br />
PO CH 3O O<br />
O<br />
O<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
P = p-BrBz<br />
O<br />
CH 3<br />
O<br />
PO O OCH3<br />
OHC O<br />
HO<br />
OHC O<br />
HO<br />
Coleophomone B Coleophomone C<br />
Nicolaou, K. C.; Montagnon, T.; Vassilikogiannakis, G.; Mathison, C. J. N. J. Am. Chem. Soc.<br />
2005, 127, 8872–8888.<br />
O<br />
CH 3<br />
O<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
M. Movassaghi and L. Blasdel
Synthesis of Epothilone C:<br />
• Small changes can drastically affect reaction outcome. In the example below, TBS<br />
protective groups changes the E/Z selectivity.<br />
R1O CH3 H3C CH3 H 3C<br />
O<br />
OR 2<br />
O<br />
CH 3<br />
H<br />
O<br />
S<br />
N<br />
CH 3<br />
R1O CH3 H3C CH3 H 3C<br />
O<br />
OR 2<br />
O<br />
CH 3<br />
H<br />
O<br />
R 1 R 2 Catalyst Conditions Yield E/Z<br />
H<br />
H<br />
TBS<br />
TBS<br />
H<br />
TBS<br />
TBS<br />
TBS<br />
1-Mo<br />
3-Ru<br />
3-Ru<br />
1-Mo<br />
50 mol%, PhH, 55 °C<br />
10 mol%, CH 2Cl 2, 25 °C<br />
6 mol%, CH 2Cl 2, 25 °C<br />
50 mol%, PhH, 55 °C<br />
65 %<br />
85%<br />
94%<br />
86%<br />
Nicolaou, K. C.; He, Y.; Vourloumis, D.; Vallberg, H.; Roschangar, F.; Sarabia, F.;<br />
Ninkovic, S.; Yang, Z.; Trujillo, J. I. J. Am. Chem. Soc. 1997, 119, 7960–7973.<br />
Meng, D.; Bertinato, P.; Balog, A.; Su, D.-S.; Kamenecka, T.; Sorensen, E. J.;<br />
Danishefsky, S. J. J. Am. Chem. Soc. 1997, 119, 11073–11092.<br />
Schinzer, D.; Bauer, A.; Bohm, O. M.; Limberg, A.; Cordes, M. Chem. Eur. J. 1999, 5,<br />
2483–2491.<br />
S<br />
N<br />
2 : 1<br />
1 : 1.2<br />
1 : 1.7<br />
1 : 1.7<br />
CH 3<br />
Solid-Phase Synthesis of Epothilone A:<br />
HO<br />
CH3 CH3 H3C O<br />
H3C O<br />
H3C TBSO<br />
O<br />
HO CH3 H3C CH3 H 3C<br />
= Merrifield resin<br />
O<br />
O<br />
OTBS<br />
O<br />
H 3C<br />
CH 3<br />
O<br />
HO CH3 H3C CH3 S<br />
N<br />
S<br />
N<br />
O<br />
CH 3<br />
O<br />
OTBS<br />
CH 3<br />
O<br />
CH 3<br />
S<br />
N<br />
3-Ru (0.75 equiv)<br />
25 °C, 48 h<br />
CH 2Cl 2<br />
CH 3<br />
HO<br />
CH3 CH3 H3C O<br />
H3C O<br />
H3C TBSO<br />
O<br />
15.6% 15.6%<br />
5.2%<br />
HO CH3 H3C CH3 H 3C<br />
O<br />
O<br />
OTBS<br />
15.6%<br />
• <strong>The</strong> amount of alkylidene 3-Ru (75%) used was greater than the total yield of product (52%),<br />
perhaps reflecting the generation of a resin-bound Ru intermediate.<br />
• Addition of n-octene or ethylene has been documented to provide a catalytic cycle; see:<br />
Maarseveen, J. H.; Hartog, J. A. J.; Engelen, V.; Finner, E.; Visser, G.; Kruse, C. G.<br />
Tetrahedron Lett. 1996, 37, 8249.<br />
Nicolaou, K. C.; Winssinger, N.; Pastor, J.; Ninkovic, S.; Sarabia, F.; He, Y.; Vourloumis, D.;<br />
Yang, Z.; Li, T.; Giannakakou, P.; Hamel, E. Nature 1997, 387, 268–272.<br />
O<br />
CH 3<br />
S<br />
N<br />
S<br />
N<br />
CH 3<br />
CH 3<br />
M. Movassaghi and L. Blasdel
Catalytic RCM of <strong>Olefin</strong>ic Enol Ethers:<br />
Ph<br />
CH 3<br />
O<br />
O<br />
O<br />
O Ph<br />
CH 3CHBr 2, TiCl 4<br />
Zn, TMEDA,<br />
cat. PbCl 2,<br />
20 °C, 11 h<br />
THF<br />
55%<br />
CH 3CHBr 2, TiCl 4<br />
Zn, TMEDA,<br />
cat. PbCl 2,<br />
20 °C, 5 h<br />
THF<br />
79%<br />
Tandem <strong>Olefin</strong>ation-<strong>Metathesis</strong><br />
H<br />
BnO<br />
H<br />
O<br />
O<br />
H<br />
O<br />
O<br />
R<br />
CH3 CH 3<br />
Ph<br />
CH 3<br />
CH 3<br />
O<br />
Tebbe reagent<br />
(4.0 equiv)<br />
THF, 25 °C, 0.5 h;<br />
reflux, 4h<br />
O Ph<br />
• Only catalyst 1-Mo is effective for RCM of these substrates.<br />
Fujimura, O.; Fu, G. C.; Grubbs, R. H. J. Org. Chem. 1994, 59, 4029–4031.<br />
Ti CH 2<br />
Cl<br />
H<br />
BnO<br />
H<br />
O<br />
O<br />
H<br />
O<br />
R<br />
R = H<br />
50%<br />
CH3 54%<br />
• Here, a Ti-alkylidene is used in RCM.<br />
12 mol% 1-Mo<br />
20 °C, 3.5 h<br />
n-pentane<br />
Ph<br />
O<br />
O<br />
Al CH 3<br />
CH 3<br />
Nicolaou, K. C.; Postema, M. H. D.; Yue, E. W.; Nadin, A. J. Am. Chem. Soc. 1996, 118,<br />
10335-10336.<br />
88%<br />
12 mol% 1-Mo<br />
20 °C, 7 h<br />
n-pentane<br />
87%<br />
Tebbe reagent<br />
Ph<br />
Tandem Ring Opening-Ring Closing <strong>Metathesis</strong> of Cyclic <strong>Olefin</strong>s<br />
substrate product<br />
H H<br />
O O<br />
H H<br />
O<br />
O<br />
H H<br />
O O<br />
H H<br />
O O<br />
O O<br />
H H O H H O<br />
O<br />
O<br />
H<br />
O<br />
O<br />
H<br />
O<br />
H H<br />
H H<br />
H H<br />
O O<br />
R O O R 6 mol% 3-Ru<br />
H<br />
O<br />
H H<br />
C6H6, 45 °C<br />
6 h<br />
R = H<br />
H<br />
CH3 O<br />
• Without sufficient ring strain in the starting cyclic olefin, competing oligomerization (via CM)<br />
can occur.<br />
• Higher dilution favors intramolecular reaction:<br />
yield<br />
(%)<br />
82<br />
90<br />
70<br />
68<br />
92<br />
0.12 M<br />
0.008 M<br />
0.2 M<br />
catalyst 3-Ru<br />
(mol %)<br />
16%<br />
73%<br />
42%<br />
O<br />
H<br />
• <strong>The</strong> relative rate of intramolecular metathesis versus CM may be further increased<br />
by substitution of the acyclic olefin.<br />
3<br />
5<br />
3<br />
6<br />
5<br />
conc.<br />
(M)<br />
0.1<br />
0.1<br />
0.07<br />
0.04<br />
0.04<br />
time<br />
(h)<br />
1.5<br />
2<br />
6<br />
2<br />
3<br />
temp.<br />
(°C)<br />
45<br />
60<br />
45<br />
45<br />
60<br />
M. Movassaghi
Proposed Mechanism for Ring Opening-Ring Closing <strong>Metathesis</strong>:<br />
H H<br />
O O<br />
H H<br />
O O<br />
H 2C CH 2<br />
L nRu CH 2<br />
L nRu<br />
L nRu CHPh<br />
Ph<br />
H H<br />
O O<br />
H H<br />
O O<br />
RuL n<br />
H H<br />
O O<br />
O<br />
H<br />
RuL n<br />
H<br />
O<br />
• Initial metathesis of the acyclic olefin is supported by the fact that substitution of this olefin<br />
decreases the rate of metathesis and by the beneficial effects of dilution upon the<br />
intramolecular manifold.<br />
• Subtle conformational preferences within the substrate are key to the success of these<br />
transformations; as shown, trans-1,4-dihydronaphthalene diamide undergoes efficient<br />
ring opening-ring closing metathesis while the corresponding diester and diether<br />
derivatives do not.<br />
CH3 CH3 O N<br />
O N<br />
unreactive substrates:<br />
O N<br />
CH3 O O<br />
O O<br />
10 mol% 3-Ru<br />
0.1 M, C 6D 6<br />
40 °C, 8 h<br />
95%<br />
O<br />
O<br />
O<br />
N<br />
CH3 Zuercher, W. J.; Hashimoto, M.; Grubbs, R. H. J. Am. Chem. Soc. 1996, 118, 6634–6640.<br />
H 3C<br />
Examples in Complex Synthesis:<br />
O<br />
CH 3<br />
CH3 2 mol% 3-Ru<br />
H 3C<br />
ethylene<br />
O<br />
98%<br />
H 3C H<br />
HO HO<br />
HO<br />
Ingenol<br />
CH 3<br />
OH<br />
CH 3<br />
H 3C<br />
O<br />
O<br />
O<br />
CH 3<br />
H 3C<br />
CH 3<br />
H<br />
O<br />
O<br />
O<br />
CH 3<br />
H<br />
H 3C<br />
CH 3<br />
OPMB<br />
O<br />
CH 3<br />
CH 3<br />
OPMB<br />
25 mol% 5-Ru<br />
toluene, Δ<br />
Nickel, A.; Maruyama, T.; Tang, H.; Murphy, P. D.; Greene, B.; Yusuff, N.; Wood, J. L. J. Am.<br />
Chem. Soc. 2004, 126, 16300–16301.<br />
O<br />
H 3C<br />
CH 3<br />
O<br />
20 mol% 4-Ru<br />
ethylene, toluene<br />
43% (3 steps)<br />
O<br />
H H<br />
H 3C CH 3<br />
H 3C<br />
O<br />
H3C CH3 H H<br />
CH 3<br />
Cyanthiwigin U<br />
Pfeiffer, M. W. B.; Phillips, A. J. J. Am. Chem. Soc. 2005, 127, 5334–5335.<br />
O<br />
OH<br />
CH3 76%<br />
M. Movassaghi and L. Blasdel
Kinetic Resolution via Asymmetric RCM<br />
Et 3SiO<br />
F3C F3C CH 3<br />
Ar = 2,6-(i-Pr) 2C 6H 3<br />
i-Pr i-Pr<br />
F3C F3C N<br />
Ph<br />
O Mo CH3 O CH3 H<br />
CF3 CF3 CH 3<br />
N<br />
H3C O Mo<br />
O<br />
Ar<br />
CF 3<br />
CF 3<br />
11-Mo<br />
H<br />
DISFAVORED<br />
2 mol% 11-Mo<br />
–20 °C, 660 min<br />
toluene<br />
CH 3<br />
OSiEt 3<br />
H<br />
Et 3SiO<br />
CH 3<br />
F3C F3C CH 3<br />
Proposed Transition State Models for the Observed Selectivity<br />
N<br />
H3C O Mo<br />
O<br />
Ar<br />
+<br />
CF 3<br />
CF 3<br />
H<br />
FAVORED<br />
Fujimura, O.; Grubbs, R. H. J. Org. Chem. 1998, 63, 824–832.<br />
Fujimura, O.; Grubbs, R. H. J. Am. Chem. Soc. 1996, 118, 2499–2500.<br />
Et 3SiO<br />
38%, 48% ee 62%<br />
• <strong>The</strong> first catalytic, asymmetric kinetic resolution via RCM was achieved, with low selectivity,<br />
using the chiral alkylidene 11-Mo.<br />
H 3C<br />
t-Bu<br />
CH3 H3C R 1<br />
O<br />
O<br />
CH 3<br />
CH 3<br />
R 1<br />
N<br />
Mo<br />
H<br />
CH3 R2 CH3 t-Bu<br />
12-Mo: R 1 = i-Pr<br />
13-Mo: R 1 = CH 3<br />
14-Mo: R 1 = Cl<br />
15-Mo: R 1 = Cl<br />
H<br />
R 2 = Ph<br />
R 2 = Ph<br />
R 2 = Ph<br />
R 2 = CH 3<br />
CH 3<br />
OSiEt 3<br />
Catalytic, Enantioselective RCM<br />
CH 3<br />
CH 3<br />
CH 3<br />
H OSiEt 3<br />
H<br />
CH3 OSiEt3 5 mol% 12-Mo<br />
22 °C, 10 min<br />
C 6H 6<br />
5 mol% 12-Mo<br />
22 °C, 2 h<br />
C 6H 6<br />
CH 3<br />
CH 3<br />
CH 3<br />
H OSiEt 3<br />
19%, >99% ee<br />
H<br />
CH3 OSiEt3 50%,
• <strong>The</strong> alkylidene catalysts 12-Mo and 13-Mo are very effective in catalytic, enantioselective<br />
desymmetrization processes, especially in the case of secondary allylic ethers.<br />
H 3C<br />
O<br />
R R<br />
R = H<br />
R = CH 3<br />
O<br />
O<br />
CH 1-2 mol% 13-Mo<br />
3<br />
22 °C, 5 min<br />
neat<br />
H<br />
H 3C<br />
R<br />
H 3C<br />
O<br />
85%, 93% ee<br />
93%, 99% ee<br />
• Remarkably, this catalytic, asymmetric RCM can be carried out in the absence of solvent,<br />
with 20:1 de<br />
Kiely, A. F.; Jernelius, J. A.; Schrock, R. R.; Hoveyda, A. H. J. Am. Chem. Soc. 2002, 124,<br />
2868.<br />
CH 3<br />
catalyst<br />
CH 3<br />
H3C N<br />
Ph<br />
n<br />
PhH, 22 °C<br />
%mol<br />
H3C N<br />
Ph<br />
n<br />
n catalyst catalyst time yield ee<br />
1<br />
2<br />
3<br />
H 3C<br />
O O<br />
CH 3<br />
12-Mo<br />
12-Mo<br />
15-Mo<br />
5 mol% 14-Mo<br />
PhH, 22 °C, 12 h<br />
5<br />
2<br />
5<br />
20 min<br />
7 h<br />
20 min<br />
CH 3<br />
41%, >98% conv.<br />
83% ee O O<br />
• Only 29% ee was observed using 12-Mo. 14-Mo is the catalyst of choice for synthesizing<br />
non-racemic acetals.<br />
Weatherhead, G. S.; Houser, J. H.; Ford, J. G.; Jamieson, J. Y.; Schrock, R. R.;<br />
Hoveyda, A. H. Tetrahedron Lett. 2000, 41, 9553-9559.<br />
78%<br />
90%<br />
93%<br />
98%<br />
95%<br />
>98%<br />
Dolman, S. J.; Sattely, E. S.; Hoveyda, A. H.; Schrock, R. R. J Am. Chem. Soc. 2002, 124,<br />
6991–6997.<br />
M. Movassaghi and L. Blasdel
Catalytic RCM of Dienynes: Construction of Fused Bicyclic Rings<br />
n m n m n m<br />
L n[M]<br />
R R<br />
OSiEt 3<br />
CH 3<br />
OSiEt 3<br />
R<br />
R<br />
H<br />
CH 3<br />
i-Pr<br />
t-Bu<br />
Ph<br />
CO 2CH 3<br />
Si(CH 3) 3<br />
Sn(n-Bu) 3<br />
Cl, Br, I<br />
OSiEt 3<br />
CH3 dienyne<br />
RCM<br />
95%<br />
[M]L n<br />
R R<br />
• Fused [5.6.0], [5.7.0], [6.6.0], and [6.7.0] bicyclic rings have been successfully constructed<br />
by RCM of dienynes.<br />
3 mol% 2-Ru<br />
25 °C, 8 h<br />
0.06 M<br />
CH 2Cl 2<br />
• <strong>The</strong> dienyne RCM is largely favored over the competing diene RCM.<br />
3-5 mol% 2-Ru<br />
0.05-0.1 M<br />
C 6D 6<br />
yield (%) conditions<br />
>98<br />
95<br />
78<br />
NR<br />
96<br />
82<br />
NR<br />
NR<br />
NR<br />
+<br />
23 °C, 15 min<br />
23 °C, 8 h<br />
60 °C, 4 h<br />
60 °C, 3 h<br />
60 °C, 4 h<br />
H 3C<br />
OSiEt 3<br />
R<br />
diene<br />
RCM<br />
Enyne <strong>Metathesis</strong> <strong>Reaction</strong>s Catalyzed by PtCl 2<br />
PhO 2S<br />
H<br />
O<br />
substrate product yield<br />
Ts H<br />
N<br />
H<br />
O<br />
O OCH 3<br />
SO 2Ph<br />
CH 3O<br />
O O H<br />
H<br />
TsN<br />
PhO 2S<br />
H<br />
O<br />
SO 2Ph<br />
70%<br />
54%<br />
80%<br />
a <strong>Reaction</strong>s conducted in toluene at 80 °C using 4-10 mol% of PtCl2<br />
• In most cases commercial PtCl 2 was used as received.<br />
• A cationic reaction pathway, involving the complexation of cationic Pt(II) with the<br />
alkyne, has been proposed.<br />
• Remote alkenes are unaffected.<br />
Fürstner, A.; Szillat, H.; Stelzer, F. J. Am. Chem. Soc. 2000, 122, 6785–6786.<br />
96%<br />
Enyne <strong>Metathesis</strong> in Synthesis<br />
TBSO<br />
H 3C<br />
TBSO<br />
H 3CO<br />
OTBS<br />
TBSO<br />
OCH 3<br />
40 mol% 3-Ru<br />
ethylene, toluene, 45 °C<br />
TBSO<br />
H 3C<br />
CH 3<br />
31%<br />
OTBS<br />
H<br />
O<br />
H<br />
O<br />
H OHC<br />
O<br />
TBSO<br />
CH 3<br />
TBSO<br />
OTBS<br />
CH 3 OTBS<br />
CH 3<br />
O<br />
CH 3<br />
OCH 3<br />
1. 50 mol% 3-Ru<br />
ethylene, CH 2Cl 2, 40 °C<br />
2. TBAF, THF, 0 → 23 °C<br />
42% (two steps)<br />
CH 3<br />
OCH 3<br />
(–)-Longithorone A<br />
Layton, M. E.; Morales, C. A.; Shair, M. D. J. Am. Chem. Soc. 2002, 124, 773–775.<br />
H 3C<br />
CH 3<br />
CH 3<br />
H 3C<br />
CO 2CH 3<br />
CH 3<br />
CH 3<br />
12 mol% 4-Ru<br />
H 3CO 2C<br />
CH2Cl2, reflux, 3 h<br />
CH3 82% H3C CH3 O<br />
OHC<br />
OH<br />
AcO<br />
H3C CH3 CH3 CH3 Guanacastepene A<br />
Boyer, F.-D.; Hanna, I.; Ricard, L. Org. Lett. 2004, 6, 1817–1820.<br />
CH 3<br />
L. Blasdel and M. Movassaghi
Cross <strong>Metathesis</strong><br />
<strong>Olefin</strong> categorization and rules for selectivity<br />
Selective Cross-<strong>Metathesis</strong> <strong>Reaction</strong>s as a Function of Catalyst Structure:<br />
<strong>Olefin</strong> type<br />
Type I<br />
(fast homodimerization)<br />
Type II<br />
(slow homodimerization)<br />
Type III<br />
(no homodimerization)<br />
Type IV<br />
(spectators to CM)<br />
Type I – Rapid homodimerization, homodimers consumable<br />
Type II – Slow homodimerization, homodimers sparingly consumable<br />
Type III – No homodimerization<br />
Type IV – <strong>Olefin</strong>s inert to CM, but do not deactivate catalyst (spectator)<br />
MesN<br />
Cl<br />
Cl<br />
NMes<br />
Ph<br />
Ru<br />
H<br />
P(c-Hex) 3<br />
Cl<br />
Cl<br />
P(c-Hex) 3<br />
Ru<br />
Ph<br />
H<br />
P(c-Hex) 3<br />
4-Ru 3-Ru<br />
terminal olefins, 1° allylic alcohols, esters, allyl<br />
boronate esters, allyl halides, styrenes (no large<br />
ortho substit.), allyl phosphonates, allyl silanes,<br />
allyl phosphine oxides, allyl sulfides, protected<br />
allyl amines<br />
styrenes (large ortho substit.), acrylates,<br />
acrylamides, acrylic acid, acrolein, vinyl keones,<br />
unprotected 3° allylic alcohols, vinyl epoxides, 2°<br />
allylic alcohols, perfluoalkyl substituted olefins<br />
1,1-disubstituted olefins, non-bulky trisub. olefins,<br />
vinyl phosphonates, phenyl vinyl sulfone, 4° allylic<br />
carbons (all alkyl substituents), 3° allylic alcohols<br />
(protected)<br />
vinyl nitro olefins, trisubstituted allyl alcohols<br />
(protected)<br />
terminal olefins, allyl silanes, 1° allylic alcohols,<br />
ethers, esters, allyl boronate esters, allyl halides<br />
styrene, 2° allylic alcohols, vinyl dioxolanes,<br />
vinyl boronates<br />
vinyl siloxanes<br />
<strong>Reaction</strong> between two olefins of Type I................................... Statistical CM<br />
<strong>Reaction</strong> between two olefins of same type (non-Type I)........ Non-selective CM<br />
<strong>Reaction</strong> beween olefins of two different types....................... Selective CM<br />
1,1-disubstituted olefins, disub a,b-unsaturated<br />
carbonyls, 4° allylic carbon-containing olefins,<br />
perfluorinated alkane olefins, 3° allyl amines<br />
(protected)<br />
i-Pr i-Pr<br />
F3C O<br />
F3C CH3 N<br />
Mo<br />
O<br />
CH3 Ph<br />
CH3 H<br />
F3C F3C CH3 1-Mo<br />
terminal olefins, allyl silanes<br />
styrene, allyl stannanes<br />
3° allyl amines, acrylonitrile<br />
1,1-disubstituted olefins<br />
Chatterjee, A. K.; Choi, T.-L.; Sanders, D. P.; Grubbs, R. H. J. Am. Chem. Soc. 2003, 125, 11360–11370. L. Blasdel
Non-selective Cross <strong>Metathesis</strong>: Two Type I <strong>Olefin</strong>s<br />
3<br />
+<br />
AcO<br />
2 equiv<br />
OAc<br />
3 mol% catalyst<br />
CH 2Cl 2, 40 °C, 12 h<br />
80%<br />
catalyst<br />
3-Ru<br />
4-Ru<br />
• <strong>The</strong> difference in E/Z ratios reflects the enhanced activity of 4-Ru relative to 3-Ru.<br />
Because it is more active, 4-Ru can catalyze secondary metathesis of the product,<br />
allowing equilibration of the olefin to the more thermodynamically stable trans isomer.<br />
Chatterjee, A. K.; Choi, T.-L.; Sanders, D. P.; Grubbs, R. H. J. Am. Chem. Soc. 2003,<br />
125, 11360–11370.<br />
E/Z<br />
3.2 : 1<br />
• Selectivity for the trans olefin can also be enhanced using sterically hindered substrates:<br />
PhO<br />
7 : 1<br />
+ SiR3 2 mol% 1-Mo<br />
PhO SiR3 3 DME, 23 °C, 4 h<br />
3<br />
R Yield E/Z<br />
CH 3<br />
Ph<br />
72%<br />
77%<br />
Crowe, W. E.; Goldberg, D. R.; Zhang, Z. J. Tetrahedron Lett. 1996, 37, 2117–2120.<br />
• In addition, steric bulk can assist in favoring the cross metathesis reaction over<br />
homodimerization pathways.<br />
• <strong>The</strong> lower yield obtained with the unprotected alcohol is a result of homodimerization of<br />
the tertiary allylic alcohol. Subjecting this dimer to the reaction conditions results in no<br />
CM product, indicating that the dimer cannot undergo a secondary metathesis reaction.<br />
AcO<br />
+<br />
CH3 OR<br />
CH3 6 mol% 4-Ru<br />
CH 2Cl 2, 40 °C, 12 h<br />
80%<br />
AcO<br />
3<br />
2.6 : 1<br />
7.6 : 1<br />
OAc<br />
CH3 OR<br />
CH3 R = H 58% yield<br />
R = TBS 97% yield<br />
Chatterjee, A. K.; Choi, T.-L.; Sanders, D. P.; Grubbs, R. H. J. Am. Chem. Soc. 2003,<br />
125, 11360–11370.<br />
<strong>Olefin</strong> 1 <strong>Olefin</strong> 2 product a,b<br />
Secondary allylic alcohols (Type I with Type II)<br />
BzO<br />
HO<br />
TBDPSO<br />
CH 3<br />
CH 3<br />
3<br />
3<br />
OAc<br />
OAc<br />
Quaternary allylic olefins (Type I with Type III)<br />
H 3C<br />
HO<br />
CH 3<br />
2.0 equiv<br />
O<br />
H 3C<br />
CH 3<br />
O<br />
1.0 equiv<br />
3<br />
3<br />
3<br />
OAc<br />
OAc<br />
OAc<br />
1,1-Disubstituted olefins (Type I with Type III)<br />
BzO<br />
H 2N<br />
HO<br />
H<br />
O<br />
O<br />
O<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
2.0 equiv<br />
1.0 equiv<br />
2.0 equiv<br />
7<br />
3<br />
8<br />
OAc<br />
2.0 equiv<br />
OTBS<br />
1.2 equiv<br />
CH 3<br />
1.1 equiv<br />
BzO<br />
HO<br />
TBDPSO<br />
CH 3<br />
CH 3<br />
CH 3<br />
H 3C CH 3<br />
HO<br />
O<br />
H 3C<br />
BzO<br />
H 2N<br />
HO<br />
O<br />
O<br />
O<br />
CH 3<br />
CH 3<br />
CH 3<br />
OAc<br />
Isolated<br />
Yield (%) E/Z<br />
82 10 : 1<br />
50 c (62) d 14 : 1<br />
53 6.7 : 1<br />
93 >20 : 1<br />
91 >20 : 1<br />
80 4 : 1<br />
71 > 20 : 1<br />
23 4 : 1<br />
O<br />
H3C OAc<br />
3 H<br />
(OAc) 97 > 20 : 1<br />
CH 3<br />
3<br />
CH3 1.0 equiv<br />
7<br />
3<br />
3<br />
3<br />
3<br />
8<br />
3<br />
OAc<br />
3<br />
OAc<br />
OAc<br />
OAc<br />
OAc<br />
OTBS<br />
CH 3<br />
a 3–5 mol% 4-Ru, CH2Cl 2, 40 °C. b See last reference on left half of this page.<br />
c With 2 equiv <strong>Olefin</strong> 2, the yield was 92%. d <strong>Reaction</strong> was performed at 23 °C.<br />
L. Blasdel
<strong>Olefin</strong> 1 <strong>Olefin</strong> 2 product a<br />
Type II and Type III<br />
HO<br />
t-BuO<br />
HO<br />
EtO<br />
H 3C<br />
H 3C<br />
F<br />
O<br />
O<br />
O<br />
O<br />
F<br />
F<br />
CH 3<br />
R<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
C(CH3) 3 HO<br />
neat<br />
C(CH3) 3 HO<br />
neat<br />
3<br />
4.0 equiv<br />
3<br />
4.0 equiv<br />
CH 3<br />
CH 3<br />
HO<br />
HO<br />
O<br />
O<br />
O<br />
O<br />
AcO OAc F<br />
2.0 equiv<br />
AcO OAc<br />
2.0 equiv<br />
O<br />
OCH3 1.5–2.0 equiv<br />
O<br />
OEt<br />
1.5–2.0 equiv<br />
O<br />
OEt<br />
1.5–2.0 equiv<br />
a 1–5 mol% 4-Ru, CH2Cl 2, 40 °C.<br />
H 3C<br />
F<br />
C(CH 3) 3<br />
C(CH 3) 3<br />
C(CH 3) 3<br />
C(CH 3) 3<br />
OAc<br />
OAc<br />
CO 2CH 3<br />
CH 3<br />
CO 2Et<br />
CO 2Et<br />
Isolated<br />
Yield (%) E/Z<br />
73<br />
73<br />
83<br />
55 R = H<br />
83 R = CH 3<br />
Chatterjee, A. K.; Choi, T.-L.; Sanders, D. P.; Grubbs, R. H. J. Am. Chem. Soc. 2003,<br />
125, 11360–11370.<br />
H 3C<br />
F<br />
CH 3<br />
CH 3<br />
98<br />
50<br />
92<br />
87<br />
5<br />
2 : 1<br />
2 : 1<br />
2 : 1<br />
>20 : 1<br />
>20 : 1<br />
>20 : 1<br />
>20 : 1<br />
>20 : 1<br />
Selective Cross-<strong>Metathesis</strong> <strong>Reaction</strong>s:<br />
Type I<br />
Type I<br />
O<br />
O N H<br />
+<br />
Si(CH 3) 3<br />
1.5 equiv<br />
O<br />
Cl 3C N H<br />
+<br />
OTr<br />
Si(CH 3) 3<br />
1.5 equiv<br />
OTr<br />
Type IV<br />
Type III<br />
10 mol% 4-Ru<br />
CH 2Cl 2, 40 °C, 4 h<br />
10 mol% 1-Mo<br />
CH 2Cl 2, 40 °C, 16 h<br />
(H 3C) 3Si<br />
Brümmer, O; Rückert, A.; Blechert, S. Chem. Eur. J. 1997, 3, 441–446.<br />
H<br />
CbzHN CO 2CH 3<br />
97% ee<br />
+<br />
+<br />
NC R<br />
R<br />
CH 2Si(CH 3) 3<br />
(CH 2) 3OBn<br />
(CH 2) 2CO 2Bn<br />
O<br />
OTr<br />
O<br />
O N H<br />
50% isolated yield<br />
1.5 : 1 E/Z<br />
OTr<br />
Si(CH<br />
Cl3C N<br />
3) 3<br />
H<br />
98% isolated yield<br />
>20 : 1 E/Z<br />
(H 3C) 3Si<br />
H<br />
10 mol% 1-Mo<br />
Si(CH3) 3 CbzHN CO2CH3 CH2Cl2, 8 h<br />
reflux<br />
95%, 92% ee<br />
Brümmer, O; Rückert, A.; Blechert, S. Chem. Eur. J. 1997, 3, 441–446.<br />
5 mol% 1-Mo<br />
23 °C, 3h<br />
CH 2Cl 2<br />
yield (%) E:Z<br />
76<br />
60<br />
44<br />
1:3<br />
1:7.6<br />
1:5.6<br />
• <strong>The</strong> basis for the high cis-selectivity with acrylonitrile as substrate is not known.<br />
Crowe, W. E.; Goldberg, D. R. J. Am. Chem. Soc. 1995, 117, 5162–5163.<br />
NC<br />
R<br />
L. Blasdel and M. Movassaghi
Reagent preparation<br />
A Horner–Wadsworth–Emmons reagent:<br />
O<br />
P<br />
EtO EtO<br />
A Suzuki reagent:<br />
+<br />
O<br />
OEt<br />
H 3C CH3<br />
AcO<br />
3<br />
+<br />
O<br />
B<br />
O<br />
CH3 CH3 CH3 One-pot CM and allylboration reactions:<br />
CH3 H3C H3C O<br />
B<br />
H3C O<br />
2.0 equiv<br />
+<br />
2.0 equiv<br />
4 mol% 4-Ru<br />
CH 2Cl 2 40 °C, 12 h<br />
87%<br />
> 20 : 1 E/Z<br />
5 mol% 4-Ru<br />
CH 2Cl 2 40 °C, 12 h<br />
58%,<br />
>20 : 1 E/Z<br />
1. 3 mol% 3-Ru<br />
CH 2Cl 2 40 °C, 24 h<br />
O<br />
P<br />
EtO EtO<br />
Toste, F. D.; Chatterjee, A. K.; Grubbs, R. H. Pure Appl. Chem. 2002, 74, 7–10.<br />
AcO 3<br />
Morrill, C.; Funk, T. W.; Grubbs, R. H. Tetrahedron Lett. 2004, 45, 7733–7736.<br />
2. PhCHO (2 equiv), 23 °C<br />
Yamamoto, Y.; Takahashi, M.; Miyaura, N. Synlett 2002, 128–130.<br />
O<br />
CH 3<br />
OEt<br />
H 3C CH3<br />
O CH3 B<br />
O CH3 Ph<br />
OH<br />
Ph<br />
88 %<br />
91 : 9 anti/syn<br />
Examples in synthesis<br />
• En route to the ABS ring fragment of thyrsiferol:<br />
H 3C<br />
H 3C<br />
Br<br />
O<br />
H 3C<br />
H 3C<br />
Br<br />
OAc<br />
CH3 O<br />
+<br />
CH3 CH3 O<br />
O<br />
OAc<br />
starting material homodimer<br />
O<br />
O<br />
OBn<br />
OBn<br />
OBn<br />
2<br />
O<br />
3.0 equiv<br />
OTBS<br />
McDonald, F. E.; Wei, X. Org. Lett. 2002, 4, 593–595.<br />
H<br />
O<br />
5 mol% 3-Ru<br />
H 3C<br />
H 3C<br />
Br<br />
CH 2Cl 2, 23 °C, 30 min<br />
OBn<br />
O OBn<br />
H<br />
OBn<br />
95%<br />
10 mol% 4-Ru<br />
CH 2Cl 2, 45 °C<br />
CH3 O<br />
OAc<br />
CH3 O<br />
44% E-isomer<br />
64% after recycling the homodimer<br />
AcO<br />
AcO<br />
H<br />
OAc<br />
5.0 equiv<br />
OAc<br />
H<br />
O<br />
O<br />
OBn<br />
O OBn<br />
H<br />
OBn<br />
H<br />
H<br />
O<br />
OTBS<br />
• CM can be difficult in the presence of strained olefins, as was found in the preparation of the<br />
AB ring fragment of ciguatoxin:<br />
AcO<br />
compound A<br />
OAc<br />
H<br />
19%<br />
via ring opening to compound A<br />
+<br />
40 mol% 3-Ru<br />
CH 2Cl 2, 40 °C<br />
33 h<br />
OBn<br />
O OBn<br />
H<br />
OBn<br />
8%<br />
AB ring fragment of ciguatoxin<br />
Oguri, H.; Sasaki, S.; Oishi, T.; Hirama, M. Tetrahedron Lett. 1999, 40, 5405–5408.<br />
L. Blasdel
Ring Opening Cross-<strong>Metathesis</strong>:<br />
CH 3O 2C<br />
O<br />
O<br />
substrate product alkene a<br />
O<br />
O<br />
CO 2CH 3<br />
NBoc<br />
O<br />
O<br />
CH 3OCH 2<br />
O<br />
Et<br />
Et<br />
CH 3OCH 2<br />
O<br />
CH 3O 2C<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
CO 2CH 3<br />
O<br />
NBoc<br />
CH 2OCH 3<br />
Et<br />
Et<br />
CH 2OCH 3<br />
A<br />
B<br />
C<br />
A 2 89 15 NA<br />
a 25 °C; 1.5 Equivalents of alkene used: A = trans-1,4-dimethoxybut-2-ene;<br />
B = trans-hex-3-ene; C = cis-hex-3-ene. Solvent: C 6H 6 (entries 1 and 2) or<br />
CH 2Cl 2 (entries 3 and 4). b Cat. = 2-Ru. c Cat. = 3-Ru.<br />
O<br />
mol %<br />
cat. b time yield E,E:E,Z<br />
6 96 94 2:1<br />
2 14 85 2:1<br />
8 c<br />
3 73 1.5:1<br />
• In these cases a preference for the E-olefin geometry is observed in ring opening<br />
metathesis.<br />
• Higher yields were achieved by the slow addition of the cyclic alkene to a solution of<br />
the 1,2-disubstituted alkene.<br />
• Faster and more efficient ring opening cross metathesis was observed using<br />
cis-hex-3-ene vs. trans-hex-3-ene.<br />
Schneider, M. F.; Blechert, S. Angew. Chem., Int. Ed. Engl. 1996, 35, 411-412.<br />
Enantioselective ROM–CM reactions have been described: La, D. S.; Ford, J. F.; Sattely,<br />
E. S.; Bonitatebus, P. J.; Schrock, R. R.; Hoveyda, A. H. J. Am. Chem. Soc. 1999, 121,<br />
11603–11604.<br />
Enyne Cross-<strong>Metathesis</strong><br />
• 4-Ru outperforms 3-Ru in both rate and overall conversion in the cross-metathesis of<br />
ethylene and alkynes.<br />
AcO<br />
CH 3<br />
substrate product time (h) yield (%)<br />
BnO<br />
OAc<br />
OR<br />
Ph<br />
CH 3<br />
OAc<br />
NTs<br />
OH<br />
AcO<br />
CH 3<br />
BnO<br />
CH 3<br />
OR<br />
OAc<br />
OAc<br />
NTs<br />
a <strong>Reaction</strong>s conducted in CH2Cl 2 at 23 °C using 5 mol% of 4-Ru at 60 psi of<br />
ethylene pressure.<br />
Smulik, J. A.; Diver, S. T. Org. Lett. 2000, 2, 2271–2274.<br />
R = H<br />
R = Ac<br />
R = TBS<br />
Ph<br />
2.0<br />
2.0<br />
8.5<br />
16 77<br />
4.0 69<br />
4.0 91<br />
OH<br />
73<br />
92<br />
91<br />
6.0 72<br />
• <strong>Reaction</strong>s conducted at 1 atm of ethylene pressure typically gave low conversions even<br />
after extended reaction times.<br />
• <strong>The</strong> more reactive imidazolylidene 4-Ru can tolerate free hydroxyl groups and<br />
coordinatingfunctionality at the propargylic and homopropargylic positions.<br />
• Chiral propargylic alcohols afford chiral diene products without loss of optical purity:<br />
4-Ru (5 mol%)<br />
ethylene (60 psi)<br />
CH 2Cl 2, 23 °C<br />
99% ee 99% ee<br />
M. Movassaghi
<strong>Metathesis</strong> of Alkynes and Diynes<br />
• Inspired by the activation of the triple bond of molecular nitrogen with molybdenum<br />
complexes of the general type Mo[N(t-Bu)Ar] 3 (see: Laplaza, C. E.; Cummins, C. C.<br />
Science, 1995, 268, 861), the reactivity of this class of molybdenum catalysts toward<br />
alkynes was explored.<br />
RO<br />
CH 3<br />
t-Bu<br />
N<br />
CH3 CH3 t-Bu<br />
Mo N<br />
t-Bu<br />
N<br />
16-Mo<br />
CH 3<br />
CH 3<br />
R CH 3<br />
CH 3<br />
CH 3<br />
RX<br />
CH 3<br />
t-Bu<br />
RO<br />
N<br />
CH3 CH3 Cl<br />
t-Bu<br />
Mo<br />
N<br />
N<br />
t-Bu<br />
CH 3<br />
CH 3<br />
X = Cl<br />
X = Br<br />
• Oxidation of the Mo(III)-precatalyst 16-Mo occurs in situ upon addition of ~25 equivalents of<br />
additives such as CH 2Cl 2, CH 2Br 2, CH 2I 2, and BnCl.<br />
• Alkyne metathesis may be achieved with equal efficiency either by in situ oxidation of<br />
precatalyst 16-Mo or by use of pure Mo(IV)-catalysts 17-Mo and 18-Mo.<br />
16-Mo (10 mol%)<br />
CH 2Cl 2, Toluene<br />
17-Mo (10 mol%)<br />
CH 2Cl 2, Toluene<br />
17-Mo,<br />
18-Mo,<br />
CH 3<br />
R R<br />
• Catalyst 17-Mo is sensitive to acidic protons such as those of secondary amides.<br />
• Terminal alkynes are incompatible with the catalysts.<br />
R = H,<br />
R = CN,<br />
R = CH 3,<br />
R = THP,<br />
60%<br />
58%<br />
59%<br />
55%<br />
• Use of CH 2Cl 2 as the reaction solvent or the addition of ~25 equivalents of CH 2Cl 2<br />
per mol of 16-Mo in toluene are equally effective.<br />
• Catalysts 17-Mo and 18-Mo tolerate functional groups such as esters, amides,<br />
thioethers, basic nitrogen atoms, and polyether chains, many of which are<br />
incompatible with the tungsten alkylidyne catalysts previously used.<br />
OR<br />
CH 3<br />
RCM of Diynes<br />
• Efficient synthesis of ≥12-membered rings containing internal alkynes can be<br />
achieved with 17-Mo.<br />
O<br />
Ph<br />
Ph<br />
Si<br />
CH 3<br />
N<br />
O<br />
substrate<br />
O<br />
O<br />
O<br />
O<br />
O(CH 2) 10<br />
O(CH 2) 10<br />
O(CH 2) 10<br />
O(CH 2) 10<br />
O O<br />
O O<br />
O(CH 2) 10<br />
O<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
Ph<br />
Ph<br />
CH 3<br />
O<br />
Si<br />
O<br />
O<br />
O O<br />
O O<br />
O<br />
O<br />
O<br />
product a<br />
Fürstner, A.; Mathes, C.; Lehmann, C. W. J. Am. Chem. Soc. 1999, 121, 9453–9454.<br />
N<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
yield (%)<br />
a <strong>Reaction</strong>s conducted in toluene at 80 °C for 20-48h; 17-Mo was generated in situ<br />
from 16-Mo and CH 2Cl 2 (~25 equiv).<br />
88<br />
82<br />
74<br />
83<br />
91<br />
M. Movassaghi
Synthesis of Cyclic β-Turn Analogs by RCM<br />
H 3C<br />
H<br />
N<br />
Boc<br />
H3C H<br />
H<br />
N<br />
Boc<br />
H<br />
O H3C CH3 N O<br />
N H<br />
O H N O<br />
20 mol% 2-Ru<br />
H<br />
H<br />
O H3C CH3 N O<br />
N H<br />
O H N O<br />
N<br />
H<br />
CH2Cl2, 40 °C N<br />
Bn<br />
Boc<br />
60%<br />
N<br />
H<br />
Bn<br />
• <strong>The</strong> presence of the Pro-Aib sequence in the tetrapeptide induces a β-turn conformation<br />
which was covalently captured by RCM, yielding a 14-membered macrocycle.<br />
Miller, S. J.; Kim, S. H.; Chen, Z. R.; Grubbs, R. H. J. Am. Chem. Soc. 1995, 117, 2108–2109.<br />
Miller, S. J.; Grubbs, R. H. J. Am. Chem. Soc. 1995, 117, 5855-5856.<br />
N<br />
O<br />
CH 3<br />
CH 3<br />
N O<br />
H<br />
O H N O<br />
OBn<br />
30 mol% 3-Ru<br />
0.004 M, 21 h<br />
CH 2Cl 2, 40 °C<br />
60%<br />
H 3C<br />
H3C H<br />
H<br />
N<br />
Boc<br />
N<br />
O<br />
CH 3<br />
CH 3<br />
N O<br />
H<br />
O H N O<br />
• Although interactions that increase the rigidity of the substrate and reduce the entropic<br />
cost of cyclization can be beneficial in RCM, it is not a strict requirement for<br />
macrocyclization byRCM.<br />
Miller, S. J.; Blackwell, H. E.; Grubbs, R. H. J. Am. Chem. Soc. 1996, 118, 9606–9614.<br />
OBn<br />
Template-Directed RCM<br />
O<br />
O O<br />
O<br />
O<br />
O<br />
n<br />
n = 1, 2<br />
O<br />
O<br />
5 mol% 3-Ru<br />
"template"<br />
CH 2Cl 2, THF<br />
45 °C, 1 h<br />
0.02 M<br />
substrate (n) "template" (equiv) yield (%) cis:trans<br />
1<br />
1<br />
1<br />
2<br />
2<br />
none<br />
LiClO 4 (5)<br />
NaClO 4 (5)<br />
none<br />
LiClO 4 (5)<br />
39<br />
>95<br />
42<br />
57<br />
89<br />
O<br />
38:62<br />
100:0<br />
62:38<br />
26:74<br />
61:39<br />
O<br />
O<br />
O O<br />
O<br />
n = 1, 2<br />
• Preorganization of the linear polyether about a complementary metal ion can enhance RCM.<br />
• In general, ions that function best as templates also favor the formation of the cis isomer.<br />
5 mol% 3-Ru<br />
CH 2Cl 2<br />
1.2 M, 23 °C<br />
>95%<br />
5 mol% 3-Ru<br />
LiClO 4<br />
CH 2Cl 2, THF<br />
0.02 M, 50 °C<br />
>95% (cis)<br />
O<br />
M n = 65900<br />
n<br />
cis : trans, 1 : 3.7<br />
• Polymer degradation in the absence of a Li + template produced the corresponding<br />
crown ether as a mixture of cis- and trans-olefins (20% combined yield) along with<br />
other low molecular weight polymers.<br />
Marsella, M. J.; Maynard, H. D.; Grubbs, R. H. Angew. Chem., Int. Ed. Engl. 1997, 36, 1101–<br />
1103.<br />
O<br />
m<br />
M. Movassaghi
RCM-Mediated Covalent Capture<br />
• <strong>The</strong> eight-residue cyclic peptide cyclo[-(L-Phe-D- Me N-Ala-L-HomoallylGly-D- Me N-Ala) 2 - ]<br />
self-assembles to form two slow-exchanging antiparallel β-sheet-like hydrogen bonded<br />
cylinders (K a(CDCl 3) = 99 M –1 , only the reactive isomer is shown).<br />
Ph<br />
O O<br />
O<br />
N<br />
N<br />
O<br />
N<br />
N<br />
N<br />
H<br />
N<br />
N<br />
N<br />
O H O<br />
O H O H<br />
Ph<br />
O H O H<br />
H O H O<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
O<br />
O<br />
O O<br />
Ph Ph<br />
20-25 mol% 2-Ru<br />
CDCl 3, 23 °C, 48 h<br />
65%<br />
Ph<br />
O O<br />
O<br />
N<br />
N<br />
O<br />
N<br />
N<br />
N<br />
H<br />
N<br />
N<br />
N<br />
O H O<br />
O H O H<br />
Ph<br />
O H O H<br />
H O H O<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
O<br />
O<br />
O O<br />
Ph Ph<br />
• <strong>The</strong> hydrogen-bonded ensemble positions the terminal olefins of the four<br />
L-homoallylglycine residues in sufficiently close proximity that each pair undergoes RCM<br />
in the presence of alkylidene 2-Ru to give a tricyclic cylindrical product containing a 38membered<br />
ring as a mixture of three (cis-cis, cis-trans, trans-trans) olefin isomers.<br />
• This covalent capture strategy may be useful in stabilizing kinetically labile α-helical and<br />
β-sheet peptide secondary structures.<br />
Clark, T. D.; Ghadiri, M. R. J. Am. Chem. Soc. 1995, 117, 12364–12365.<br />
Synthesis of Catenanes<br />
2<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
N<br />
N<br />
O O<br />
O<br />
N<br />
N<br />
N<br />
N<br />
O<br />
O O<br />
O<br />
O<br />
32-membered catenane<br />
O<br />
O<br />
O<br />
O<br />
Cu(CH 3CN) 4PF 6<br />
CH 2Cl 2, CH 3CN<br />
100%<br />
KCN, H 2O<br />
CH 3CN<br />
~100%<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
= Cu +<br />
O<br />
O<br />
O<br />
O<br />
O<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
N<br />
O<br />
O<br />
O<br />
O O<br />
trans:cis, 98:2<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
5 mol% 3-Ru<br />
23 °C, 6 h<br />
0.01 M, CH 2Cl 2<br />
92%<br />
• <strong>The</strong> remarkable efficiency of this RCM is proposed to be due to preorganization of the<br />
substrate.<br />
O<br />
O<br />
+ –<br />
PF6 + PF –<br />
6<br />
Mohr, B.; Weck, M.; Sauvage, J.-P.; Grubbs, R. H. Angew. Chem., Int. Ed. Engl. 1997, 36,<br />
1308–1310.<br />
M. Movassaghi