ETEST APPLICATION SHEET - mediline
ETEST APPLICATION SHEET - mediline
ETEST APPLICATION SHEET - mediline
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
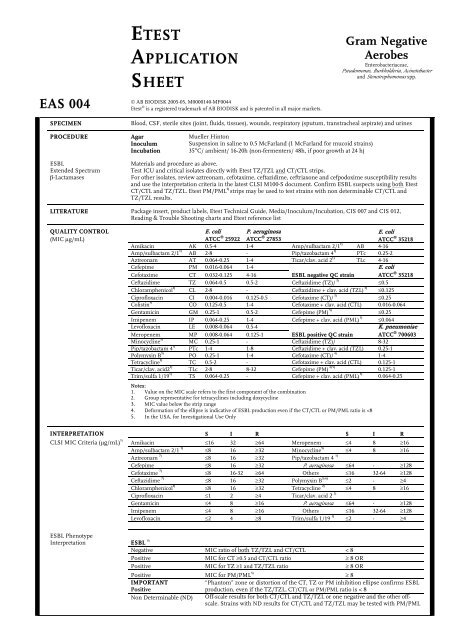
EAS 004<br />
<strong>ETEST</strong><br />
<strong>APPLICATION</strong><br />
<strong>SHEET</strong><br />
© AB BIODISK 2005-05, M0000140-MF0044<br />
Etest ® is a registered trademark of AB BIODISK and is patented in all major markets.<br />
Gram Negative<br />
Aerobes<br />
Enterobacteriaceae,<br />
Pseudomonas, Burkholderia, Acinetobacter<br />
and Stenotrophomonas spp.<br />
SPECIMEN Blood, CSF, sterile sites (joint, fluids, tissues), wounds, respiratory (sputum, transtracheal aspirate) and urines<br />
PROCEDURE<br />
ESBL<br />
Extended Spectrum<br />
β-Lactamases<br />
Agar<br />
Inoculum<br />
Incubation<br />
Mueller Hinton<br />
Suspension in saline to 0.5 McFarland (1 McFarland for mucoid strains)<br />
35°C/ ambient/ 16-20h (non-fermenters/ 48h, if poor growth at 24 h)<br />
Materials and procedure as above.<br />
Test ICU and critical isolates directly with Etest TZ/TZL and CT/CTL strips.<br />
For other isolates, review aztreonam, cefotaxime, ceftazidime, ceftriaxone and cefpodoxime susceptibility results<br />
and use the interpretation criteria in the latest CLSI M100-S document. Confirm ESBL suspects using both Etest<br />
CT/CTL and TZ/TZL. Etest PM/PML 5) strips may be used to test strains with non determinable CT/CTL and<br />
TZ/TZL results.<br />
LITERATURE Package insert, product labels, Etest Technical Guide, Media/Inoculum/Incubation, CIS 007 and CIS 012,<br />
Reading & Trouble Shooting charts and Etest reference list<br />
QUALITY CONTROL<br />
(MIC µg/mL)<br />
E. coli<br />
ATCC ® P. aeruginosa<br />
25922 ATCC ® E. coli<br />
27853<br />
ATCC ® 35218<br />
Amikacin AK 0.5-4 1-4 Amp/sulbactam 2/1 1)<br />
AB 4-16<br />
Amp/sulbactam 2/1 1) AB 2-8 - Pip/tazobactam 4 1) PTc 0.25-2<br />
Aztreonam AT 0.064-0.25 1-4 Ticar/clav. acid 2 1)<br />
TLc 4-16<br />
Cefepime PM 0.016-0.064 1-4 E. coli<br />
Cefotaxime CT 0.032-0.125 4-16 ESBL negative QC strain ATCC ® 35218<br />
Ceftazidime TZ 0.064-0.5 0.5-2 Ceftazidime (TZ)/ 3) ≤0.5<br />
Chloramphenicol 5) CL 2-8 - Ceftazidime + clav. acid (TZL) 3) ≤0.125<br />
Ciprofloxacin CI 0.004-0.016 0.125-0.5 Cefotaxime (CT)/ 3)<br />
≤0.25<br />
Colistin 5) CO 0.125-0.5 1-4 Cefotaxime + clav. acid (CTL) 0.016-0.064<br />
Gentamicin GM 0.25-1 0.5-2 Cefepime (PM) 5) ≤0.25<br />
Imipenem IP 0.064-0.25 1-4 Cefepime + clav. acid (PML) 5) ≤0.064<br />
Levofloxacin LE 0.008-0.064 0.5-4 K. pneumoniae<br />
Meropenem MP 0.008-0.064 0.125-1 ESBL positive QC strain ATCC ® 700603<br />
Minocycline 5) MC 0.25-1 - Ceftazidime (TZ)/ 8-32<br />
Pip/tazobactam 4 1) PTc 1-4 1-8 Ceftazidime + clav. acid (TZL) 0.25-1<br />
Polymyxin B 5)<br />
PO 0.25-1 1-4 Cefotaxime (CT)/ 4)<br />
1-4<br />
Tetracycline 2) TC 0.5-2 - Cefotaxime + clav. acid (CTL) 0.125-1<br />
Ticar/clav. acid2 1) TLc 2-8 8-32 Cefepime (PM) 4) 5) 0.125-1<br />
Trim/sulfa 1/19 1)<br />
TS 0.064-0.25 - Cefepime + clav. acid (PML) 5)<br />
0.064-0.25<br />
Notes:<br />
1. Value on the MIC scale refers to the first component of the combination<br />
2. Group representative for tetracyclines including doxycycline<br />
3. MIC value below the strip range<br />
4. Deformation of the ellipse is indicative of ESBL production even if the CT/CTL or PM/PML ratio is
Notes:<br />
6. Polymyxin B MIC result can predict colistin MIC (CLSI M100-S15, 2005)<br />
7. See CLSI tables for specific ESBL comments<br />
8. See the Etest ESBL package insert and Etest PM/PML 5)<br />
supplement<br />
9. See the latest CLSI M100-S document for further details on species specific and clinical interpretations<br />
READING PRECAUTIONS 1. For bactericidal drugs, read at complete inhibition of growth (microcolonies, hazes and isolated colonies)<br />
2. For bacteriostatic drugs, read at 80% inhibition when trailing of growth is seen<br />
ANTIBIOGRAMS Examples only, please use your own formulary.<br />
REFERENCES<br />
CI or<br />
LE<br />
CI or<br />
LE<br />
PM<br />
or CT )<br />
PTc<br />
GM (256)<br />
or AK<br />
Enterobacteriaceae<br />
GM<br />
or AK<br />
IP<br />
MP or<br />
IP<br />
Pseudomonas spp.<br />
Detection of ESBL<br />
PTc<br />
AT<br />
AT<br />
TZ or<br />
PM<br />
CI or<br />
LE<br />
CI or<br />
LE<br />
AB<br />
or TLc<br />
AK<br />
GM<br />
or AK<br />
Acinetobacter spp. Burkholderia spp.<br />
AB or<br />
TLc<br />
IP<br />
or MP<br />
Stenotrophomonas spp.<br />
Strains with ND results by CT/CTL and TZ/TZL<br />
AK<br />
Mucoid organisms e.g. Klebsiella,<br />
Enterobacter and P. aeruginosa<br />
1. Bolmström et al. (2002) Cefepime • clavulanic acid (CA) in an Etest Configuration for Investigating Non-Determinable (ND) ESBL Results per<br />
NCCLS criteria. ICCAC (Poster D527).<br />
2. Stürenburg et al.(2004). Evaluation of a New Cefepime-clavulanate ESBL Etest to Detect Extended-Spectrum β-lactamases in an<br />
Enterobacteriaceae Strain Collection. JAC. 54: 134- 138.<br />
3. Lincott, A. and Brown, W. (2005). Evaluation of Four Commercially Available Extended-Spectrum Beta-Lactamase Phenotypic Confirmation<br />
Tests. JCM. 43(3): 1081- 1085.<br />
4. Yu, V et al. (2004). Antibiotic therapy for Klebsiella pneumoniae bacteremia: Implications of production of extended-spectrum β-lactamases.<br />
CID. 39: 31- 37.<br />
5. Mohr et al. (2004). Pharmacokinetic/ pharmacodynamic modeling can help guide targeted antimicrobial therapy for nosocomial Gram-negative<br />
infections in critically ill patients. DMID. 48: 125- 130.<br />
6. Shah, P. (2001). Determination of MICs in the routine laboratory. JAC. 48(6): 91.<br />
7. Hanberger, H. et al. (1999). New species-related MIC breakpoints for early detection of development of resistance among Gram-negative<br />
bacteria in Swedish intensive care units. JAC. 44: 611-619.<br />
8. Tsakris et al. (2005). Heteroresistance to carbapenems in Acinetobacter baumannii. JAC. 55(6): 1055- 1056.<br />
9. Walsh et al. (2002). Evaluation of a new Etest for detecting Metallo-β-lactamases in routine clinical testing. JCM. 40(8): 2755-2759.<br />
Dalvägen 10, S-169 56 Solna, Sweden. Tel: +46-(0)8-730 07 60. Fax: +46-(0)8-83 81 58. etest@abbiodisk.se www.abbiodisk.com<br />
TS<br />
MC<br />
MC<br />
TZ or<br />
PM<br />
TZ or<br />
PM<br />
MC<br />
LE<br />
CI or<br />
LE<br />
PM<br />
TS<br />
IP<br />
MP<br />
PTc<br />
TZ<br />
TZ