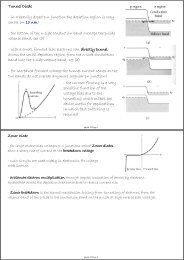
Free Electrons in a Metal - in a typical metal each atom contributes ...
Free Electrons in a Metal - in a typical metal each atom contributes ...
Free Electrons in a Metal - in a typical metal each atom contributes ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
The Solid State<br />
- most of the matter <strong>in</strong> the physical world around us (on earth) is <strong>in</strong> the solid state (which is<br />
not true for the universe as a whole)<br />
- solids consist of <strong>atom</strong>s, ions or molecules closely packed together<br />
- the b<strong>in</strong>d<strong>in</strong>g occurs either by covalent, ionic, van der Waals or <strong>metal</strong>lic bonds all of<br />
which are mediated by electromagnetic forces<br />
structure:<br />
- many solids appear <strong>in</strong> crystall<strong>in</strong>e<br />
structure, i.e. the constituents are arranged<br />
<strong>in</strong> a regular, repeated three dimensional<br />
pattern (a lattice) with long range order<br />
- only few solids appear as s<strong>in</strong>gle crystals<br />
(e.g. Silicon (Si)), most consist of very<br />
many small crystallites<br />
Amorphous Solids<br />
- some solids (e.g. glass, plastics) have an<br />
amorphous structure, i.e. they have no long<br />
range but only short range order<br />
- can be viewed as a very viscous (glassy)<br />
liquid which crystallizes very slowly<br />
phys4.16 Page 7<br />
- amorphous and crystall<strong>in</strong>e B2O3<br />
melt<strong>in</strong>g: - <strong>in</strong> amorphous solids the bond strengths varies throughout the material<br />
- as a result melt<strong>in</strong>g occurs slowly and cont<strong>in</strong>uously<br />
- <strong>in</strong> crystall<strong>in</strong>e solids most of the bonds have equal strengths and thus<br />
melt<strong>in</strong>g occurs at a well def<strong>in</strong>ed temperature<br />
phys4.16 Page 8