Proceedings - Interdisciplinary Center for Nanotoxicity
Proceedings - Interdisciplinary Center for Nanotoxicity
Proceedings - Interdisciplinary Center for Nanotoxicity
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
40<br />
Conference on Current Trends in Computational Chemistry 2009<br />
31+G** basis sets. The minima of the potential surface were found by relaxing the geometric<br />
parameters with the standard optimization methods. Analytical <strong>for</strong>ce constants<br />
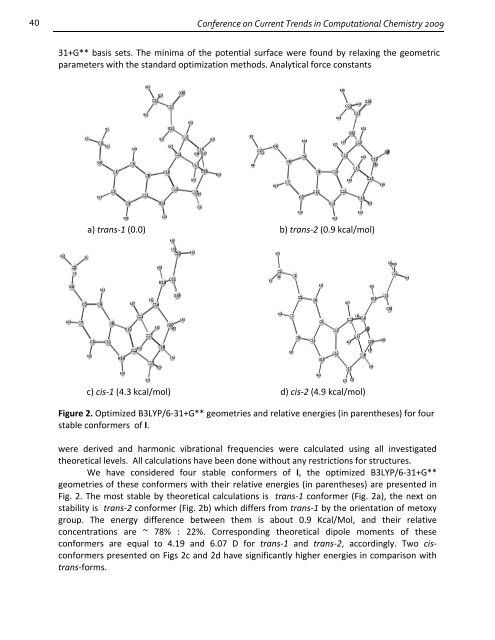
a) trans‐1 (0.0) b) trans‐2 (0.9 kcal/mol)<br />
c) cis‐1 (4.3 kcal/mol) d) cis‐2 (4.9 kcal/mol)<br />
Figure 2. Optimized B3LYP/6‐31+G** geometries and relative energies (in parentheses) <strong>for</strong> four<br />
stable con<strong>for</strong>mers of I.<br />
were derived and harmonic vibrational frequencies were calculated using all investigated<br />
theoretical levels. All calculations have been done without any restrictions <strong>for</strong> structures.<br />
We have considered four stable con<strong>for</strong>mers of I, the optimized B3LYP/6‐31+G**<br />
geometries of these con<strong>for</strong>mers with their relative energies (in parentheses) are presented in<br />
Fig. 2. The most stable by theoretical calculations is trans‐1 con<strong>for</strong>mer (Fig. 2a), the next on<br />
stability is trans‐2 con<strong>for</strong>mer (Fig. 2b) which differs from trans‐1 by the orientation of metoxy<br />
group. The energy difference between them is about 0.9 Kcal/Mol, and their relative<br />
concentrations are ~ 78% : 22%. Corresponding theoretical dipole moments of these<br />
con<strong>for</strong>mers are equal to 4.19 and 6.07 D <strong>for</strong> trans‐1 and trans‐2, accordingly. Two cis‐<br />
con<strong>for</strong>mers presented on Figs 2c and 2d have significantly higher energies in comparison with<br />
trans‐<strong>for</strong>ms.