Chemistry 2BA3 - Organic Chemistry A F. Capretta phone: ext ...
Chemistry 2BA3 - Organic Chemistry A F. Capretta phone: ext ...
Chemistry 2BA3 - Organic Chemistry A F. Capretta phone: ext ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Chemistry</strong> <strong>2BA3</strong> - <strong>Organic</strong> <strong>Chemistry</strong> A<br />
F. <strong>Capretta</strong> <strong>phone</strong>: <strong>ext</strong>. 27318<br />
ABB-261A e-mail: capretta@mcmaster.ca<br />
Office Hours: Wednesday between 2:30-5:30 pm. (Other times by appointment.)<br />
Course Webpage on McMaster’s WebCT<br />
Lectures: Tuesdays, Wednesdays and Fridays at 9:30<br />
Outline<br />
The course t<strong>ext</strong>book is <strong>Organic</strong> <strong>Chemistry</strong> by Solomons & Fryhle and the lectures will be based on<br />
the following (time permitting):<br />
Week Dates (Week<br />
beginning..)<br />
Chapter Topic<br />
1 September 5, 2005 1 Chemical Bonding<br />
2 September 12, 2005 1 & 2 Functional Groups<br />
3 September 19, 2005 2 & 3 Acids and Bases<br />
4 September 26, 2005 4 Conformational Analysis<br />
5 October 3, 2005 4 & 5 Stereochemistry<br />
6 October 10, 2005 5 Stereochemistry<br />
7 October 17, 2005 5 & 6 Substitution and Elimination<br />
8 October 24, 2005 6 Substitution and Elimination<br />
9 October 31, 2005 7 Alkenes & Alkynes<br />
10 November 7, 2005 8 Alkenes & Alkynes<br />
11 November 14, 2005 11 Alcohols and Ethers<br />
12 November 21, 2005 11& 12 Carbonyls / Oxidation /Reduction<br />
13 November 28, 2005 12 Carbonyls / Oxidation /Reduction<br />
The Thursday lectures will be devoted to discussions of techniques introduced in the labs (distillation,<br />
recrystallization, chromatography) and spectroscopic methods presented in Chapters 2 and 9 (IR,<br />
NMR, MS).<br />
Assessment<br />
Assignments (4) 10%<br />
Term Tests (2) 30%<br />
Laboratory 20%<br />
Final exam 40%<br />
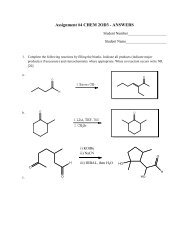
Assignments: A series of questions designed to apply the concepts presented in class. In order to<br />
insure that assignments are graded prior to tests and that answers to assignments can be posted<br />
quickly, LATE ASSIGNMENTS WILL NOT BE ACCEPTED. If you cannot meet a deadline, please<br />
see me in advance (NOT ON THE DATE THE WORK IS DUE).
Handed Out Handed In<br />
Assignment 1 Sept. 16 Sept. 23<br />
Assignment 2 Sept. 30 Oct. 7<br />
Assignment 3 Oct. 28 Nov. 4<br />
Assignment 4 Nov. 18 Nov. 25<br />
Term Tests: NOTE THAT THE TEST DATES ARE TENTATIVE AND WILL BE CONFIRMED IN<br />
CLASS. Ninety minutes each to be held on Wednesday, October 12th and Wednesday, November<br />
9th at 7:00 pm. Students who miss a term test for a valid reason AND who have the Dean's<br />
permission (see below) will have their final exam increased by the value of the term test.<br />
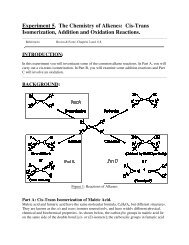
Labs: The Laboratory grade for the organic experiments is composed of two marks, written<br />
laboratory reports and a practical mark which is assigned by the teaching assistant on the basis of<br />
each student’s performance in the laboratory. The TAs are asked to evaluate students on the basis<br />
of preparation, knowledge of material, organization in the laboratory, and technique. The practical<br />
mark will be assigned at the end of term.<br />
Labs begin on the week of September 12th (lab lecture and check-in). Check your timetable for<br />
locations and times.<br />
Laboratory Manual is available on the course web-site.<br />
Note that each lab is a separate .pdf file. Your will need a suitable program (such as Adobe Acrobat)<br />
to view these files. More info on-line.<br />
Although a timetable for handing in labs will be determined by your demonstrator, all labs must be<br />
submitted by the last day of classes. Failure to meet this deadline will result in the award of a mark of<br />
zero for the lab in question. All missed labs must have a Dean's Permission Slip. All laboratory work<br />
must be completed with a passing grade in order to pass the course.<br />
Missed Assignments and Tests: Failure to hand in an assignment, complete a laboratory experiment<br />
or write term test will result in a zero grade, unless a valid reason has been filed with and accepted<br />
by the Associate Dean's office. It is the student's responsibility to ensure that medical slips, etc.<br />
are filed with the Dean. The instructor will not make any exemption decisions under any<br />
circumstances.<br />
Final: Examination to take place in December. A passing grade must be attained in the final<br />
examination to pass the course.<br />
Odds and Ends:<br />
• Additional reference material will be held on reserve for the course.<br />
• Molecular models are recommended<br />
• Lecture notes will be made available on-line<br />
Academic dishonesty:<br />
Academic dishonesty consists of misrepresentation by deception or by other fraudulent means and<br />
can result in serious consequences, e.g. the grade of zero on an assignment, loss of credit with a<br />
notation on the transcript (notation reads: “Grade of F assigned for academic dishonesty”), and/or<br />
suspension or expulsion from the university.
It is your responsibility to understand what constitutes academic dishonesty. For information on the<br />
various kinds of academic dishonesty, please refer to the Academic Integrity Policy, specifically<br />
Appendix 3, located at<br />
http://www.mcmaster.ca/senate/academic/ac_integrity.htm<br />
The following illustrates only three forms of academic dishonesty:<br />
1. Plagiarism, e.g. the submission of work that is not one’s own or for which other credit has been<br />
obtained. For example, submitting someone else’s lab report and results.<br />
2. Improper collaboration in group work. For example, submitting assignments containing<br />
answers generated by a group.<br />
3. Copy or using unauthorized aids in tests and examinations.
<strong>Organic</strong> <strong>Chemistry</strong> is the branch of chemistry dealing with the compounds of carbon. While it is only<br />
the fourteenth most common element on earth, carbon forms by far the greatest number of different<br />
compounds. <strong>Organic</strong> chemistry is of vital importance to the petrochemical, pharmaceutical, polymer<br />
and t<strong>ext</strong>ile industries, where a prime concern is the synthesis of new organic molecules and<br />
polymers. Knowledge of the structure, functionality, and reactivity of organic molecules is critical for<br />
the understanding of numerous and disparate phenomena, from biological and biochemical<br />
processes (enzyme-substate interactions), to medicine (pharmaceuticals), to the properties of<br />
materials (polymers). This course covers the basic and fundamental principles of organic chemistry,<br />
then builds on these concepts in <strong>Chemistry</strong> 2BB3. Topics covered include the utilization of<br />
spectroscopic techniques in elucidating chemical structure, aspects of kinetics and thermodynamics<br />
within organic chemistry, mechanisms of organic transformations, stereochemistry, nucleophilic<br />
substitution and elimination reactions, conformational analysis, functional group manipulation and<br />
carbon-carbon bonding forming reactions. This course provides the necessary fundamental<br />
knowledge to begin understanding the reactivity of organic molecules, mechanisms of important<br />
reactions, and the methods by which numerous organic structures can be synthesized.<br />
Some of the topics covered include (all Images taken from the course t<strong>ext</strong>book <strong>Organic</strong> <strong>Chemistry</strong> by<br />
Solomons & Fryhle (8 th Edition, Wiley, 2005):<br />
Bonding and Hybridization
Understanding Reactivity<br />
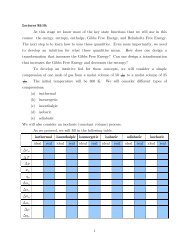
Conformational Analysis
Stereochemistry<br />
Reactions and Synthesis
Mechanisms<br />
Nuclear Magnetic Resonance Spectroscopy