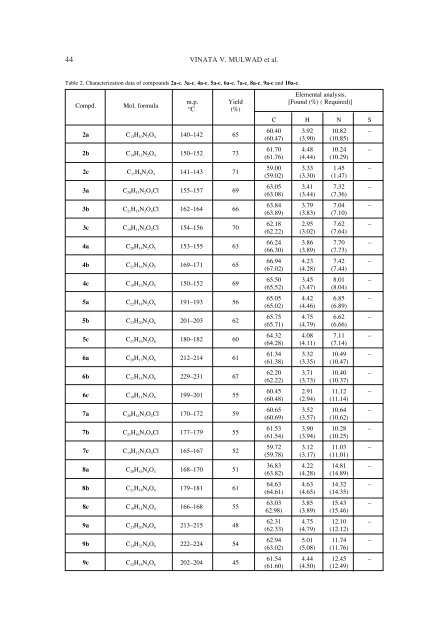
44 VINATA V. MULWAD et al. Table 2. Characterization data of compounds 2a-c, 3a-c, 4a-c, 5a-c, 6a-c, 7a-c, 8a-c, 9a-c and 10a-c. Compd. Mol. formula 2a C 13H 10N 2O 4 140ñ142 65 2b C 14H 12N 2O 4 150ñ152 73 2c C 11H 8N 2O 4 141ñ143 71 3a C 20H 13N 2O 4Cl 155ñ157 69 3b C 21H 15N 2O 4Cl 162ñ164 66 3c C 19H 11N 2O 4Cl 154ñ156 70 4a C 20H 14N 2O 5 153ñ155 63 4b C 21H 16N 2O 5 169ñ171 65 4c C 19H 12N 2O 5 150ñ152 69 5a C 22H 18N 2O 6 191ñ193 56 5b C 23H 20N 2O 6 201ñ203 62 5c C 21H 16N 2O 6 180ñ182 60 6a C 20H 13N 3O 6 212ñ214 61 6b C 21H 15N 3O 6 229ñ231 67 6c C 19H 11N 3O 6 199ñ201 55 7a C 20H 14N 3O 4Cl 170ñ172 59 7b C 21H 16N 3O 4Cl 177ñ179 55 7c C 19H 12N 3O 4Cl 165ñ167 52 8a C 20H 16N 4O 4 168ñ170 51 8b C 21H 18N 4O 4 179ñ181 61 8c C 19H 14N 4O 4 166ñ168 55 9a C 23H 20N 4O 6 213ñ215 48 9b C 24H 22N 4O 6 222ñ224 54 9c C 22H 18N 4O 6 202ñ204 45 m.p. Yield Elemental analysis. [Found (%) ( Required)] OC (%) C H N S 60.40 3.92 10.82 ñ (60.47) (3.90) (10.85) 61.70 4.48 10.24 ñ (61.76) (4.44) (10.29) 59.00 3.33 1.45 ñ (59.02) (3.30) (1.47) 63.05 3.41 7.32 ñ (63.08) (3.44) (7.36) 63.84 3.79 7.04 ñ (63.89) (3.83) (7.10) 62.18 2.95 7.62 ñ (62.22) (3.02) (7.64) 66.24 3.86 7.70 ñ (66.30) (3.89) (7.73) 66.94 4.23 7.42 ñ (67.02) (4.28) (7.44) 65.50 3.45 8.01 ñ (65.52) (3.47) (8.04) 65.05 4.42 6.85 ñ (65.02) (4.46) (6.89) 65.75 4.75 6.62 ñ (65.71) (4.79) (6.66) 64.32 4.08 7.11 ñ (64.28) (4.11) (7.14) 61.34 3.32 10.49 ñ (61.38) (3.35) (10.47) 62.20 3.71 10.40 ñ (62.22) (3.73) (10.37) 60.45 2.91 11.12 ñ (60.48) (2.94) (11.14) 60.65 3.52 10.64 ñ (60.69) (3.57) (10.62) 61.53 3.90 10.28 ñ (61.54) (3.94) (10.25) 59.72 3.12 11.03 ñ (59.78) (3.17) (11.01) 36.83 4.22 14.81 ñ (63.82) (4.28) (14.89) 64.63 4.63 14.32 ñ (64.61) (4.65) (14.35) 63.03 3.85 15.43 ñ 62.98) (3.89) (15.46) 62.31 4.75 12.10 ñ (62.33) (4.79) (12.12) 62.94 5.01 11.74 ñ (63.02) (5.08) (11.76) 61.54 4.44 12.45 ñ (61.60) (4.50) (12.49)
Table 2. cont. Compd. Mol. formula (10), 432 (23), 431 (16), 400 (10), 370 (42), 325 (60), 310 (20), 295 (40), 289 (100), 173 (71), 152 (15), 143 (10), 137 (90), 135 (5), 107 (10), 105 (6) and 32 (8). 9c: m.p. 202ñ204 O C; yield: 45%; IR (KBr, cm -1 ): 3345 (-OH), 3252 (>NH), 2955 (-CH), 1732 (>C=O), 1680 (-CONH), 1589, 1571, 1240, 1040. 1 H-NMR (DMSO-d6, 300 MHz, δ, ppm): 3.81 (s, 6H, -OCH 3), 4.53 (s, 1H, C 5-H), 4.99 (s, 1H, C 6-H), 6.10 (s, 1H, -OH, D 2O exchangeable), 6.35 (d, 1H, J = 9 Hz, C 3í-H), 6.95ñ7.22 (m, 6H, Ar-H), 7.72 (d, 1H, J = 9 Hz, C 4í-H), 9.50 (s, 1H, -NH, D 2O exchangeable).1H-5-(3íí-nitrobenzylidene)-3-(2í-oxo-2íH-benzopyran-6í-yl)imidazolidine-2,4-dione (6a-c) Compound (2a-c) (0.01 mol) was dissolved in glacial acetic acid (10 mL). To this solution, fused sodium acetate (0.015 mol) and 3-nitrobenzaldehyde (0.01 mol) was added and the mixture was refluxed for 8 h. It was then cooled and poured onto crushed ice. The resulting solid was washed with water and recrystallized from ethanol. 6a: m.p. 212ñ214 O C; yield: 61%; IR (KBr, cm -1 ): 3242 (>NH), 2952 (-CH), 1729 (>C=O), 1682 (-CONH), 1602, 1590, 1550, 1352 (-NO 2). 1 H-NMR (CDCl 3, 300 MHz, δ, ppm): 2.33 (s, 3H, C 7í-CH 3), 6.32 (d, 1H, J = 9 Hz, C 3í-H), 6.72ñ7.03 (m, 6H, Ar-H), 7.39 (s, 1H, >CH-), 7.70 (d, 1H, J = 9 Hz, C 4í-H), 8.80 (s, 1H, -NH, D 2O exchangeable). 6b: m.p. 229ñ231 O C; yield: 67%; IR (KBr, cm -1 ): 3245 (>NH), 2950 (-CH), 1732 (>C=O), 1685 (-CONH), 1605, 1595, 1560, 1549, 1350 (-NO 2). 1 H NMR (CDCl 3, 300 MHz, δ, ppm): 2.35 (s, 3H, C 7í-CH 3), 2.46 (s, 3H, C 4í-CH 3), 6.25 (s, 1H, C 3í-H), 6.80ñ7.12 (m, 6H, Ar-H), 7.40 (s, 1H, >CH-), 8.90 (s, 1H, -NH, D 2O exchangeable). MS: m/z (%): 405 (M + , 100), 359 (70), 283 (44), 268 (22), 255 (23), 232 (40), 186 (42), 173 (35), 143 (41), 122 (60), 110 (10), 82 (5), 76 (5), 54 (9) and 46 (10). Synthesis of novel biologically active heterocyclic compounds from... 45 10a C 21H 15N 5O 5S 240ñ242 52 10b C 22H 17N 5O 5S 235ñ237 57 10c C 20H 13N 5O 5S 229ñ231 61 m.p. Yield Elemental analysis. [Found (%) ( Required)] OC (%) C H N S 56.08 3.32 15.55 7.15 (56.12) (3.36) (15.58) (7.13) 57.00 3.66 15.09 6.95 (57.01) (3.70) (15.11) (6.92) 55.13 2.95 16.04 7.39 (55.17) (3.01) (16.08) (7.63) 6c: m.p. 199ñ201 O C; yield: 55%; IR (KBr, cm -1 ): 3240 (>NH), 2948 (-CH), 1730 (>C=O), 1680 (-CONH), 1601, 1585, 1545, 1348 (NO 2). 1 H NMR (CDCl 3, 300 MHz, δ, ppm): 6.33 (d, 1H, J = 9 Hz, C 3í-H), 6.77ñ7.15 (m, 7H, Ar-H), 7.42 (s, 1H, >CH-), 7.71 (d, 1H, J = 9 Hz, C 4í-H), 8.80 (s, 1H, -NH, D 2O exchangeable). 5,7-Dihydro-2-mercapto-6-(3íí-nitrophenyl)-9-(2íoxo-2íH-benzopyran-6í-yl)purin-8-one (10a-c) To a mixture of (6a-c) (0.01 mol) and thiourea (0.01 mol) in ethanol (10 mL), NaOH (10%, 1 mL) was added dropwise and then the mixture was refluxed for 10 h. The reaction was monitored by TLC and at the end was poured onto crushed ice and neutralized with dil. HCl. The resulting solid is purified by column chromatography (60ñ120 mesh silica) using elutent petrolium ether and ethyl acetate (9:1, v/v). 10a: m.p. 240ñ242 O C; yield: 52%; IR (KBr, cm -1 ): 3242 (>NH), 2950 (-CH), 2548 (-SH), 1725 (>C=O), 1685 (-CONH), 1590, 1582, 1560, 1548, 1338 (-NO 2). 1 H-NMR (DMSO-d 6, 300 MHz, δ, ppm): 2.39 (s, 3H, C 7í-CH 3), 3.42 (s, 1H, -SH), 4.90 (s, 1H, C 5-H), 5.23 (s, 1H, C 6-H), 6.30 (d, 1H, J = 9 Hz, C 3í-H), 6.91-7.12 (m, 6H, Ar-H), 7.72 (d, 1H, J = 9 Hz, C 4í-H), 9.52 (s, 1H, -NH, D 2O exchangeable). 10b: m.p. 235ñ237 O C; yield: 57%; IR (KBr, cm -1 ): 3250 (>NH), 2948 (-CH), 2552 (-SH), 1730 (>C=O), 1680 (-CONH), 1585, 1565, 1552, 1340 (-NO 2). 1 H-NMR (DMSO-d 6, 300 MHz, δ, ppm): 2.37 (s, 3H, C 7í-CH 3), 2.49 (s, 3H, C 4í-CH 3), 3.45 (s, 1H, -SH), 4.92 (s, 1H, C 5-H), 5.29 (s, 1H, C 6-H), 6.28 (s, 1H, C 3í-H), 6.99ñ7.19 (m, 6H, Ar-H), 9.50 (s, 1H, -NH, D 2O exchangeable). MS: m/z (%): 463 (M + , 70), 448 (43), 435 (7), 341 (41), 290 (40), 262 (9), 173 (100), 168 (45), 140 (17), 135 (30), 122 (23), 107 (5) and 33 (2).