Allergic bronchopulmonary aspergillosis - CHEST Publications ...
Allergic bronchopulmonary aspergillosis - CHEST Publications ...
Allergic bronchopulmonary aspergillosis - CHEST Publications ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
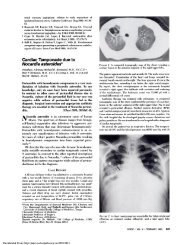
Radiologic Investigations: A wide spectrum of<br />
radiographic appearances can occur in ABPA (Table<br />
4). The chest radiographic findings of ABPA include<br />
transient or fixed pulmonary opacities (Fig 3), tramline<br />
shadows, finger-in-glove opacities, and toothpaste<br />
shadows. 83–87 Findings noted on high-resolution<br />
CT (HRCT) include central bronchiectasis, mucoid<br />
impaction, mosaic attenuation, presence of centrilobular<br />
nodules, and tree-in-bud opacities (Fig<br />
4). 88,89 High-attenuation mucoid impaction (mucus<br />
visually denser than the paraspinal muscle) is a<br />
pathognomonic finding encountered in patients with<br />
ABPA. 23,90–95 Central bronchiectasis with peripheral<br />
tapering of bronchi on HRCT is believed to be a sine<br />
qua non for the diagnosis of ABPA. Bronchiectasis<br />
may not be present in all patients with ABPA, may be<br />
present in patients with CF without ABPA, and<br />
almost 40% of the bronchiectatic segments can also<br />
Table 4—Radiologic Findings Encountered in Patients<br />
With ABPA<br />
1. Chest radiographic findings<br />
Transient changes<br />
Common<br />
Patchy areas of consolidation<br />
Radiologic infiltrates: toothpaste and gloved finger shadows<br />
due to mucoid impaction in dilated bronchi<br />
Collapse: lobar or segmental<br />
Uncommon<br />
Bronchial wall thickening: tramline shadows<br />
Air-fluid levels from dilated central bronchi filled with fluid<br />
Perihilar infiltrates simulating adenopathy<br />
Massive consolidation: unilateral or bilateral<br />
Small nodules<br />
Pleural effusions<br />
Permanent changes<br />
Common<br />
Parallel-line shadows representing bronchial widening<br />
Ring-shadows 1–2 cm in diameter representing dilated<br />
bronchi en face<br />
Pulmonary fibrosis: fibrotic scarred upper lobes with<br />
cavitation<br />
Uncommon<br />
Pleural thickening<br />
Mycetoma formation<br />
Linear scars<br />
2. HRCT findings<br />
Common<br />
Central bronchiectasis<br />
Mucus plugging with bronchoceles<br />
Consolidation<br />
Centrilobular nodules with tree-in-bud opacities<br />
Bronchial wall thickening<br />
Areas of atelectasis<br />
Mosaic perfusion with air trapping on expiration<br />
Uncommon<br />
High-attenuation mucus (finding most helpful in differential<br />
diagnosis)<br />
Pleural involvement<br />
Randomly scattered nodular opacities<br />
have associated peripheral bronchiectasis. 22,96 Minimal<br />
bronchiectasis can also be seen in asthma, 97,98<br />
but the findings of bronchiectasis affecting three or<br />
more lobes, centrilobular nodules, and mucoid impaction<br />
are highly suggestive of ABPA. 99 The uncommon<br />
radiologic manifestations of ABPA include<br />
miliary nodular opacities, 100 perihilar opacities<br />
simulating hilar lymphadenopathy, 84,101,102 pleural<br />
effusions, 103–105 and pulmonary masses. 106–111<br />
Serum Precipitins Against A fumigatus: The precipitating<br />
IgG antibodies are elicited from crude<br />
extracts of A fumigatus and can be demonstrated<br />
using the double gel diffusion technique. 112,113 They<br />
can also be present in other pulmonary disorders and<br />
thus represent supportive not diagnostic evidence for<br />
ABPA. 112–114<br />
Peripheral Eosinophilia: A blood absolute eosinophil<br />
count 1,000 cells/L is also a major criterion<br />
for the diagnosis of ABPA. However, 53% of patients<br />
in our series 22 had an absolute eosinophil count<br />
1,000 cells/L, and thus a low eosinophil count<br />
does not exclude the diagnosis of ABPA.<br />
Sputum Cultures for A fumigatus: Culture of A<br />
fumigatus in the sputum is supportive but not diagnostic<br />
of ABPA. The fungus can also be grown in<br />
patients with other pulmonary diseases due to the<br />
ubiquitous nature of the fungi. We rarely perform<br />
sputum cultures for the diagnosis of ABPA.<br />
Pulmonary Function Tests: These tests help categorize<br />
the severity of the lung disease but have no<br />
diagnostic value in ABPA and need not constitute<br />
the basis for screening. 22 The usual finding is an<br />
obstructive defect of varying severity. 115–117<br />
Role of Specific Aspergillus Antigens: Patients with<br />
ABPA are evaluated with crude extracts from Aspergillus,<br />
which lack reproducibility and consistency,<br />
and they frequently cross-react with other antigens.<br />
118 The advances in molecular techniques have<br />
enabled detection and cloning of specific Aspergillus<br />
antigens. The recombinant allergens Asp f1, Asp f2,<br />
Asp f3, Asp f4, and Asp f6 have been evaluated for<br />
their diagnostic performance in serologic studies in<br />
asthmatic patients 119–122 and in patients with<br />
CF 121,123–125 Preliminary data suggest a promising<br />
role of these antigens in the diagnosis of ABPA.<br />
Further studies are required before they can be<br />
implemented in routine clinical practice.<br />
Diagnosis and Diagnostic Criteria<br />
The Rosenberg-Patterson criteria6,9 are most often<br />
used for the diagnosis (Table 5). There are also a set<br />
810 Global Medicine<br />
Downloaded From: http://jupcvss.chestpubs.org/ on 08/01/2013