Practical Guide 5: How to report (Q)SARs - ECHA - Europa
Practical Guide 5: How to report (Q)SARs - ECHA - Europa
Practical Guide 5: How to report (Q)SARs - ECHA - Europa
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Select “all fields” in the “Detail level” dropdown menu.<br />
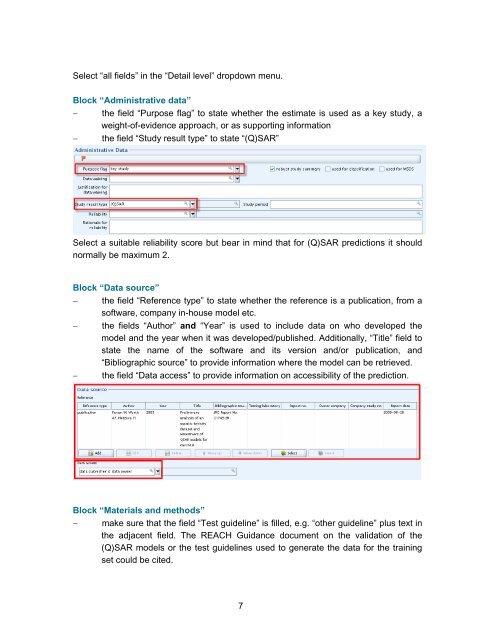
Block “Administrative data”<br />
− the field “Purpose flag” <strong>to</strong> state whether the estimate is used as a key study, a<br />
weight-of-evidence approach, or as supporting information<br />
− the field “Study result type” <strong>to</strong> state “(Q)SAR”<br />
Select a suitable reliability score but bear in mind that for (Q)SAR predictions it should<br />
normally be maximum 2.<br />
Block “Data source”<br />
− the field “Reference type” <strong>to</strong> state whether the reference is a publication, from a<br />
software, company in-house model etc.<br />
− the fields “Author” and “Year” is used <strong>to</strong> include data on who developed the<br />
model and the year when it was developed/published. Additionally, “Title” field <strong>to</strong><br />
state the name of the software and its version and/or publication, and<br />
“Bibliographic source” <strong>to</strong> provide information where the model can be retrieved.<br />
− the field “Data access” <strong>to</strong> provide information on accessibility of the prediction.<br />
Block “Materials and methods”<br />
− make sure that the field “Test guideline” is filled, e.g. “other guideline” plus text in<br />
the adjacent field. The REACH Guidance document on the validation of the<br />
(Q)SAR models or the test guidelines used <strong>to</strong> generate the data for the training<br />
set could be cited.<br />
7