Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
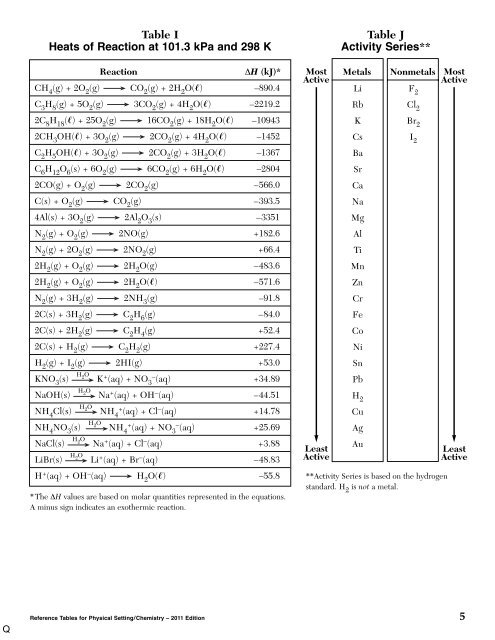
Table I<br />
Heats of Reaction at 101.3 kPa and 298 K<br />
Reaction<br />
ΔH (kJ)*<br />
CH 4<br />
(g) + 2O 2<br />
(g) CO 2<br />
(g) + 2H 2<br />
O() –890.4<br />
C 3<br />
H 8<br />
(g) + 5O 2<br />
(g) 3CO 2<br />
(g) + 4H 2<br />
O() –2219.2<br />
2C 8<br />
H 18<br />
() + 25O 2<br />
(g) 16CO 2<br />
(g) + 18H 2<br />
O() –10943<br />
2CH 3<br />
OH() + 3O 2<br />
(g) 2CO 2<br />
(g) + 4H 2<br />
O() –1452<br />
C 2<br />
H 5<br />
OH() + 3O 2<br />
(g) 2CO 2<br />
(g) + 3H 2<br />
O() –1367<br />
C 6<br />
H 12<br />
O 6<br />
(s) + 6O 2<br />
(g) 6CO 2<br />
(g) + 6H 2<br />
O() –2804<br />
2CO(g) + O 2<br />
(g) 2CO 2<br />
(g) –566.0<br />
C(s) + O 2<br />
(g) CO 2<br />
(g) –393.5<br />
4Al(s) + 3O 2<br />
(g) 2Al 2<br />
O 3<br />
(s) –3351<br />
N 2<br />
(g) + O 2<br />
(g) 2NO(g) +182.6<br />
N 2<br />
(g) + 2O 2<br />
(g) 2NO 2<br />
(g) +66.4<br />
2H 2<br />
(g) + O 2<br />
(g) 2H 2<br />
O(g) –483.6<br />
2H 2<br />
(g) + O 2<br />
(g) 2H 2<br />
O() –571.6<br />
N 2<br />
(g) + 3H 2<br />
(g) 2NH 3<br />
(g) –91.8<br />
2C(s) + 3H 2<br />
(g) C 2<br />
H 6<br />
(g) –84.0<br />
2C(s) + 2H 2<br />
(g) C 2<br />
H 4<br />
(g) +52.4<br />
2C(s) + H 2<br />
(g) C 2<br />
H 2<br />
(g) +227.4<br />
H 2<br />
(g) + I 2<br />
(g) 2HI(g) +53.0<br />
KNO 3<br />
(s) H 2 O K + (aq) + NO 3 – (aq) +34.89<br />
NaOH(s) H 2 O Na + (aq) + OH – (aq) –44.51<br />
NH 4<br />
Cl(s) H 2 O NH 4 + (aq) + Cl – (aq) +14.78<br />
NH 4<br />
NO 3<br />
(s) H 2 O NH 4 + (aq) + NO 3 – (aq) +25.69<br />
NaCl(s) H 2 O Na + (aq) + Cl – (aq) +3.88<br />
LiBr(s) H 2 O Li + (aq) + Br – (aq) –48.83<br />
H + (aq) + OH – (aq) H 2<br />
O() –55.8<br />
*The ΔH values are based on molar quantities represented in the equations.<br />
A minus sign indicates an exothermic reaction.<br />
Most<br />
Active<br />
Least<br />
Active<br />
Table J<br />
Activity Series**<br />
Metals Nonmetals Most<br />
Active<br />
Li<br />
F 2<br />
Rb Cl 2<br />
K Br 2<br />
Cs<br />
I 2<br />
Ba<br />
Sr<br />
Ca<br />
Na<br />
Mg<br />
Al<br />
Ti<br />
Mn<br />
Zn<br />
Cr<br />
Fe<br />
Co<br />
Ni<br />
Sn<br />
Pb<br />
H 2<br />
Cu<br />
Ag<br />
Au<br />
**Activity Series is based on the hydrogen<br />
standard. H 2 is not a metal.<br />
Least<br />
Active<br />
Reference Tables for Physical Setting/Chemistry – 2011 Edition 5