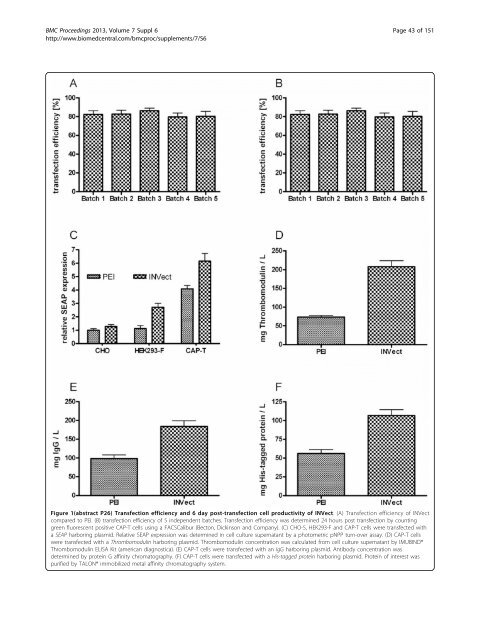
BMC Proceedings 2013, Volume 7 Suppl 6 http://www.biomedcentral.com/bmcproc/supplements/7/S6 Page 42 of 151 Figure 1(abstract P25) Titer and specific productivity comparison of a BAC derived recombinant CHO-S cell line producing gp140 (CN54) and an already existing recombinant plasmid derived CHO-DUKX-B11 cell line. P26 INVect - a novel polycationic reagent for transient transfection of mammalian cells Sebastian Püngel 1 , Miklos Veiczi 2 , Tim Welsink 1 , Daniel Faust 1 , Vanessa Vater 1 , Derek Levison 2 , Uwe Möller 2 , Wolfgang Weglöhner 1* 1 InVivo BioTech Services GmbH, 16761 Hennigsdorf, Germany; 2 emp Biotech GmbH, 13125 Berlin, Germany E-mail: wegloehner@invivo.de BMC Proceedings 2013, 7(Suppl 6):P26 Background: For rapid recombinant protein production in sm<strong>all</strong> to medium size volumes, transient transfection of mammalian cells is still the method of choice in biotechnology [1]. However, due to the high costs of commerci<strong>all</strong>y available lipofectamines or polycationic transfection reagents such as polyethylenimine (PEI), the most widely used transfection reagents available present a substantial economic bottleneck. While these reagents produce seemingly high transient transfection rates [2], there is still a strong desire for transfection reagents providing both secure and easy handling and higher recombinant protein production. As part of our commitment to excellence, InVivo BioTech Services initiated a joint venture with emp Biotech and developed a novel polycationic reagent, named INVect, for transient transfection and recombinant protein production in mammalian cells. Materials and methods: Mammalian cells were cultured in CD-ACF media using shake flasks and standard culture conditions. Cells were transfected with 10 μg per mL of a GOI harboring plasmid at a cell density of 5 × 10 6 cells per mL in FreeStyle Medium (Life Technologies) with INVect to DNA ratio of 6:1 (w/w) and PEI to DNA ration of 2:1 (w/w). Cultures were supplemented with same volume Protein Expression Medium (Life Technologies) 2 hours post transfection. GFP and SEAP expression took place in 8 mL culture volume in 50 mL bioreactor tubes. Expression of other reporter proteins were performed in 150 mL culture volume in 500 mL shake flasks. Transfection efficiency was determined 24 hours post transfection by counting green fluorescent positive cells using a FACSCalibur (Becton, Dickinson and Company). SEAP expression was determined in cell culture supernatant on day 6 post transfection by a photometric pNPP turnover assay. Quantification of IgG was performed by protein G affinity chromatography on day 6 post transfection. Thrombomodulin concentration was calculated from cell culture supernatant on day 6 post transfection by IMUBIND® Thrombomodulin ELISA Kit (american diagnostica). His-tagged recombinant protein was purified on day 6 post transfection by TALON® immobilized metal affinity chromatography system. Results: Cytotoxicity was tested over a broad range of concentrations. Results demonstrate several novel synthetic polymers exhibiting transfection efficiencies even higher than common PEIs after optimized ratios of DNA-topolymer were applied. Transfection efficiency of INVect was compared to PEI, currently the standard transfection reagent for transient gene expression. INVect was found to gener<strong>all</strong>y give better transfection efficiencies of greater 80% in a GFP assay (Figure 1A). Batch-to-batch reproducibility was shown on five independent INVect batches. Transfection results were highly consistent and in the range of 80-90% (Figure 1B). INVect successfully delivers genes to HEK293-F, CHO-S and CAP-T cells as shown in a SEAP expression system (Figure 1C). Post-transfection cell productivity was determined under TGE manufacturing conditions. Thrombomodulin (60 kDa) (Figure 1D), an IgG (144 kDa) (Figure 1E) and a HIS-tagged Protein of Interest (~40 kDa) (Figure 1F) were transiently expressed using INVect as transfection reagent and conventional 25 kDa PEI as control. Cells were transfected with a gene of interest harboring plasmid, with product concentration being measured on day 6 post transfection. The use of INVect provided a minimum 2-fold increase in protein production over PEI (25 kDa) based transfection. Conclusions: INVect is a novel polycationic transfection reagent which demonstrates low cell toxicity for transient transfection of mammalian cells and delivers extremely high transfection efficiencies of up to 90%, 24 h post transfection. The use of INVect for transfection under TGE conditions leads to exception<strong>all</strong>y high levels of protein expression and outperforms 25 kDa linear PEI by 2-fold. INVect can be used effectively with <strong>all</strong> common cell lines and is especi<strong>all</strong>y suited for HEK293-F and CAP-T cells. References 1. Geisse S: Reflections on more than 10 years of TGE approaches. Protein Expr Purif 2009, 64:99-107. 2. Fischer S, Charara N, Gerber A, Wölfel J, Schiedner G, Voedisch B, Geisse S: Transient recombinant protein expression in a human amniocyte cell line: the CAP-T® cell system. Biotechnol Bioeng 2012, 109:2250-2261. P27 Development of a chemic<strong>all</strong>y defined cultivation and transfection medium for HEK cell lines Sebastian Püngel 1 , T Tim Welsink 1 , Penélope Villegas Soto 1 , Wolfgang Weglöhner 1 , Tim F Beckmann 2 , Ina Eickmeier 2 , Stefan Northoff 2 , Christoph Heinrich 2* 1 InVivo BioTech Services GmbH, 16761 Hennigsdorf, Germany; 2 TeutoCell AG, 33615 Bielefeld, Germany E-mail: Christoph.Heinrich@teutocell.de BMC Proceedings 2013, 7(Suppl 6):P27
BMC Proceedings 2013, Volume 7 Suppl 6 http://www.biomedcentral.com/bmcproc/supplements/7/S6 Page 43 of 151 Figure 1(abstract P26) Transfection efficiency and 6 day post-transfection cell productivity of INVect. (A) Transfection efficiency of INVect compared to PEI. (B) transfection efficiency of 5 independent batches. Transfection efficiency was determined 24 hours post transfection by counting green fluorescent positive CAP-T cells using a FACSCalibur (Becton, Dickinson and Company). (C) CHO-S, HEK293-F and CAP-T cells were transfected with a SEAP harboring plasmid. Relative SEAP expression was determined in cell culture supernatant by a photometric pNPP turn-over assay. (D) CAP-T cells were transfected with a Thrombomodulin harboring plasmid. Thrombomodulin concentration was calculated from cell culture supernatant by IMUBIND® Thrombomodulin ELISA Kit (american diagnostica). (E) CAP-T cells were transfected with an IgG harboring plasmid. Antibody concentration was determined by protein G affinity chromatography. (F) CAP-T cells were transfected with a His-tagged protein harboring plasmid. Protein of interest was purified by TALON® immobilized metal affinity chromatography system.
- Page 1 and 2: BMC Proceedings 2013, Volume 7 Supp
- Page 3 and 4: BMC Proceedings 2013, Volume 7 Supp
- Page 5 and 6: BMC Proceedings 2013, Volume 7 Supp
- Page 7 and 8: BMC Proceedings 2013, Volume 7 Supp
- Page 9 and 10: BMC Proceedings 2013, Volume 7 Supp
- Page 11 and 12: BMC Proceedings 2013, Volume 7 Supp
- Page 13 and 14: BMC Proceedings 2013, Volume 7 Supp
- Page 15 and 16: BMC Proceedings 2013, Volume 7 Supp
- Page 17 and 18: BMC Proceedings 2013, Volume 7 Supp
- Page 19 and 20: BMC Proceedings 2013, Volume 7 Supp
- Page 21 and 22: BMC Proceedings 2013, Volume 7 Supp
- Page 23 and 24: BMC Proceedings 2013, Volume 7 Supp
- Page 25 and 26: BMC Proceedings 2013, Volume 7 Supp
- Page 27 and 28: BMC Proceedings 2013, Volume 7 Supp
- Page 29 and 30: BMC Proceedings 2013, Volume 7 Supp
- Page 31 and 32: BMC Proceedings 2013, Volume 7 Supp
- Page 33 and 34: BMC Proceedings 2013, Volume 7 Supp
- Page 35 and 36: BMC Proceedings 2013, Volume 7 Supp
- Page 37 and 38: BMC Proceedings 2013, Volume 7 Supp
- Page 39 and 40: BMC Proceedings 2013, Volume 7 Supp
- Page 41: BMC Proceedings 2013, Volume 7 Supp
- Page 45 and 46: BMC Proceedings 2013, Volume 7 Supp
- Page 47 and 48: BMC Proceedings 2013, Volume 7 Supp
- Page 49 and 50: BMC Proceedings 2013, Volume 7 Supp
- Page 51 and 52: BMC Proceedings 2013, Volume 7 Supp
- Page 53 and 54: BMC Proceedings 2013, Volume 7 Supp
- Page 55 and 56: BMC Proceedings 2013, Volume 7 Supp
- Page 57 and 58: BMC Proceedings 2013, Volume 7 Supp
- Page 59 and 60: BMC Proceedings 2013, Volume 7 Supp
- Page 61 and 62: BMC Proceedings 2013, Volume 7 Supp
- Page 63 and 64: BMC Proceedings 2013, Volume 7 Supp
- Page 65 and 66: BMC Proceedings 2013, Volume 7 Supp
- Page 67 and 68: BMC Proceedings 2013, Volume 7 Supp
- Page 69 and 70: BMC Proceedings 2013, Volume 7 Supp
- Page 71 and 72: BMC Proceedings 2013, Volume 7 Supp
- Page 73 and 74: BMC Proceedings 2013, Volume 7 Supp
- Page 75 and 76: BMC Proceedings 2013, Volume 7 Supp
- Page 77 and 78: BMC Proceedings 2013, Volume 7 Supp
- Page 79 and 80: BMC Proceedings 2013, Volume 7 Supp
- Page 81 and 82: BMC Proceedings 2013, Volume 7 Supp
- Page 83 and 84: BMC Proceedings 2013, Volume 7 Supp
- Page 85 and 86: BMC Proceedings 2013, Volume 7 Supp
- Page 87 and 88: BMC Proceedings 2013, Volume 7 Supp
- Page 89 and 90: BMC Proceedings 2013, Volume 7 Supp
- Page 91 and 92: BMC Proceedings 2013, Volume 7 Supp
- Page 93 and 94:
BMC Proceedings 2013, Volume 7 Supp
- Page 95 and 96:
BMC Proceedings 2013, Volume 7 Supp
- Page 97 and 98:
BMC Proceedings 2013, Volume 7 Supp
- Page 99 and 100:
BMC Proceedings 2013, Volume 7 Supp
- Page 101 and 102:
BMC Proceedings 2013, Volume 7 Supp
- Page 103 and 104:
BMC Proceedings 2013, Volume 7 Supp
- Page 105 and 106:
BMC Proceedings 2013, Volume 7 Supp
- Page 107 and 108:
BMC Proceedings 2013, Volume 7 Supp
- Page 109 and 110:
BMC Proceedings 2013, Volume 7 Supp
- Page 111 and 112:
BMC Proceedings 2013, Volume 7 Supp
- Page 113 and 114:
BMC Proceedings 2013, Volume 7 Supp
- Page 115 and 116:
BMC Proceedings 2013, Volume 7 Supp
- Page 117 and 118:
BMC Proceedings 2013, Volume 7 Supp
- Page 119 and 120:
BMC Proceedings 2013, Volume 7 Supp
- Page 121 and 122:
BMC Proceedings 2013, Volume 7 Supp
- Page 123 and 124:
BMC Proceedings 2013, Volume 7 Supp
- Page 125 and 126:
BMC Proceedings 2013, Volume 7 Supp
- Page 127 and 128:
BMC Proceedings 2013, Volume 7 Supp
- Page 129 and 130:
BMC Proceedings 2013, Volume 7 Supp
- Page 131 and 132:
BMC Proceedings 2013, Volume 7 Supp
- Page 133 and 134:
BMC Proceedings 2013, Volume 7 Supp
- Page 135 and 136:
BMC Proceedings 2013, Volume 7 Supp
- Page 137 and 138:
BMC Proceedings 2013, Volume 7 Supp
- Page 139 and 140:
BMC Proceedings 2013, Volume 7 Supp
- Page 141 and 142:
BMC Proceedings 2013, Volume 7 Supp
- Page 143 and 144:
BMC Proceedings 2013, Volume 7 Supp
- Page 145 and 146:
BMC Proceedings 2013, Volume 7 Supp
- Page 147 and 148:
BMC Proceedings 2013, Volume 7 Supp
- Page 149 and 150:
BMC Proceedings 2013, Volume 7 Supp
- Page 151:
BMC Proceedings 2013, Volume 7 Supp