School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
School of Engineering and Science - Jacobs University
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
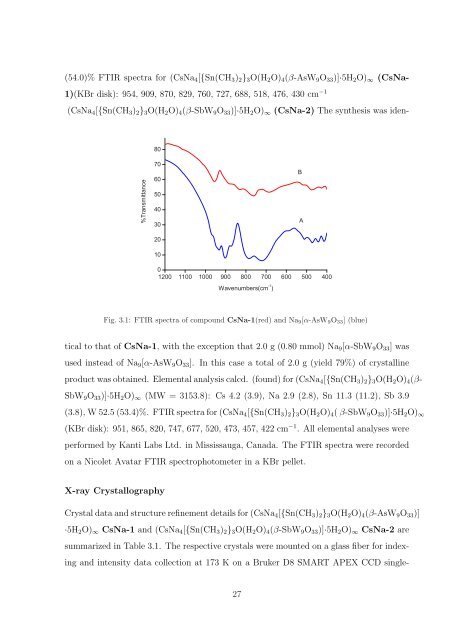
(54.0)% FTIR spectra for (CsNa 4 [{Sn(CH 3 ) 2 } 3 O(H 2 O) 4 (β-AsW 9 O 33 )]·5H 2 O) ∞ (CsNa-<br />
1)(KBr disk): 954, 909, 870, 829, 760, 727, 688, 518, 476, 430 cm −1<br />
(CsNa 4 [{Sn(CH 3 ) 2 } 3 O(H 2 O) 4 (β-SbW 9 O 33 )]·5H 2 O) ∞ (CsNa-2) The synthesis was iden-<br />
Fig. 3.1: FTIR spectra <strong>of</strong> compound CsNa-1(red) <strong>and</strong> Na 9 [α-AsW 9 O 33 ] (blue)<br />
tical to that <strong>of</strong> CsNa-1, with the exception that 2.0 g (0.80 mmol) Na 9 [α-SbW 9 O 33 ] was<br />
used instead <strong>of</strong> Na 9 [α-AsW 9 O 33 ]. In this case a total <strong>of</strong> 2.0 g (yield 79%) <strong>of</strong> crystalline<br />
product was obtained. Elemental analysis calcd. (found) for (CsNa 4 [{Sn(CH 3 ) 2 } 3 O(H 2 O) 4 (β-<br />
SbW 9 O 33 )]·5H 2 O) ∞ (MW = 3153.8): Cs 4.2 (3.9), Na 2.9 (2.8), Sn 11.3 (11.2), Sb 3.9<br />
(3.8), W 52.5 (53.4)%. FTIR spectra for (CsNa 4 [{Sn(CH 3 ) 2 } 3 O(H 2 O) 4 ( β-SbW 9 O 33 )]·5H 2 O) ∞<br />
(KBr disk): 951, 865, 820, 747, 677, 520, 473, 457, 422 cm −1 . All elemental analyses were<br />
performed by Kanti Labs Ltd. in Mississauga, Canada. The FTIR spectra were recorded<br />
on a Nicolet Avatar FTIR spectrophotometer in a KBr pellet.<br />
X-ray Crystallography<br />
Crystal data <strong>and</strong> structure refinement details for (CsNa 4 [{Sn(CH 3 ) 2 } 3 O(H 2 O) 4 (β-AsW 9 O 33 )]<br />
·5H 2 O) ∞ CsNa-1 <strong>and</strong> (CsNa 4 [{Sn(CH 3 ) 2 } 3 O(H 2 O) 4 (β-SbW 9 O 33 )]·5H 2 O) ∞ CsNa-2 are<br />
summarized in Table 3.1. The respective crystals were mounted on a glass fiber for indexing<br />
<strong>and</strong> intensity data collection at 173 K on a Bruker D8 SMART APEX CCD single-<br />
27