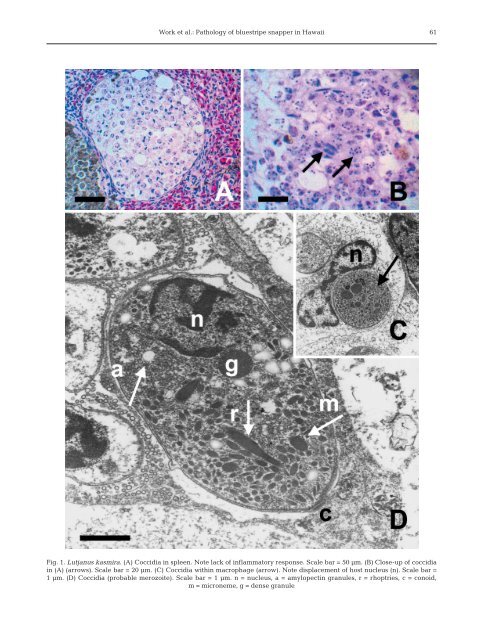
60 Dis Aquat Org 57: 59–66, 2003 that at least one nematode species was <strong>in</strong>troduced <strong>in</strong>to Hawaii with <strong>the</strong> importation of taape. Okihiro (1988) found a high prevalence of sk<strong>in</strong> tumors on native butterfly fish (Chaetodon multic<strong>in</strong>ctus <strong>and</strong> C. miliaris) <strong>in</strong> Maui, <strong>and</strong> suspected contam<strong>in</strong>ants as a possible cause, based on <strong>the</strong> distribution of affected fish. Kent & Heidel (2001) found that pen-raised opakaka Pristipomoides filamentosus suffered ma<strong>in</strong>ly from bacterial <strong><strong>in</strong>fections</strong> of swim bladder <strong>and</strong> hemorrhage <strong>and</strong> emphysema of <strong>the</strong> eyes, although various protozoan <strong>and</strong> metazoan parasites were noted at low <strong>in</strong>fection levels. As part of a larger study, taape caught near Oahu were surveyed for systemic parasites to provide basel<strong>in</strong>e <strong>in</strong>formation on <strong>the</strong>ir health status. MATERIALS AND METHODS Dur<strong>in</strong>g 2001 <strong>and</strong> 2002, fish were collected us<strong>in</strong>g <strong>the</strong> l<strong>in</strong>e <strong>and</strong> hook method at depths rang<strong>in</strong>g from 15 to 70 m throughout Sou<strong>the</strong>rn Oahu. Fish were anes<strong>the</strong>tized with MS-222 <strong>in</strong> seawater, <strong>and</strong> bled from <strong>the</strong> caudal tail ve<strong>in</strong> us<strong>in</strong>g sterile 3 cc syr<strong>in</strong>ges <strong>and</strong> 1 × 38 mm needles. Blood smears were made immediately, air dried, <strong>and</strong> fixed <strong>in</strong> absolute methanol. Fish were <strong>the</strong>n humanely euthanized with an overdose of MS-222 <strong>in</strong> seawater. Necropsies consisted of measur<strong>in</strong>g total <strong>and</strong> fork length (0.5 cm) with a ruler, weight (0.1 g) with an electric scale, <strong>and</strong> a complete external <strong>and</strong> <strong>in</strong>ternal exam. Spleen, liver, cranial <strong>and</strong> caudal kidneys, swim bladder, bra<strong>in</strong>, heart, skeletal muscle, gill, <strong>and</strong> gonad were stored <strong>in</strong> 10% neutral buffered formal<strong>in</strong>. Tissues were processed for histology by paraff<strong>in</strong> embedd<strong>in</strong>g, section<strong>in</strong>g at 5 µm, <strong>and</strong> sta<strong>in</strong><strong>in</strong>g with hematoxyl<strong>in</strong> <strong>and</strong> eos<strong>in</strong>. Tissues were exam<strong>in</strong>ed microscopically for presence of lesions <strong>and</strong> signs of <strong>in</strong>fectious organisms. Giemsa was used to identify protista, <strong>and</strong> Gimenez, <strong>and</strong> Gram sta<strong>in</strong>s were used to identify bacteria (Prophet et al. 1992). Protista <strong>in</strong>cluded microsporidians <strong>and</strong> coccidia, bacteria <strong>in</strong>cluded <strong>epi<strong>the</strong>liocystis</strong>-<strong>like</strong> organisms, <strong>and</strong> metazoans <strong>in</strong>cluded myxozoa, trematodes or <strong>the</strong>ir eggs, <strong>and</strong> migratory tracts associated with helm<strong>in</strong>th. For electron microscopy, tissues were fixed <strong>in</strong> Trump’s fixative (McDowel & Trump 1976), r<strong>in</strong>sed <strong>in</strong> 0.1 M Sorenson’s phosphate buffer, <strong>and</strong> post fixed <strong>in</strong> 2% osmium tetroxide. Epoxy embedded tissues were cut <strong>in</strong>to 1 µm thick toluid<strong>in</strong>e blue-sta<strong>in</strong>ed sections. Ultrath<strong>in</strong> sections were sta<strong>in</strong>ed with uranyl acetate, post sta<strong>in</strong>ed with lead citrate <strong>and</strong> exam<strong>in</strong>ed with a Zeiss EM 109 electron microscope. Blood smears were sta<strong>in</strong>ed with Wright’s Giemsa <strong>and</strong> exam<strong>in</strong>ed for <strong>the</strong> presence of hemoparasites. Data were tested for normality <strong>and</strong> equal variance. We used l<strong>in</strong>ear regression to evaluate <strong>the</strong> relationship between weight <strong>and</strong> fork length, <strong>the</strong> t-test to evaluate <strong>the</strong> difference <strong>in</strong> weight between males <strong>and</strong> females, <strong>and</strong> ANOVA to compare weights of parasitized <strong>and</strong> parasite-free fish (Daniel 1987). For all comparisons, α = 0.05. RESULTS A total of 120 taape (78 males, 42 females) were exam<strong>in</strong>ed. Mean weight (mean ± SD) of males 241.7 ± 59.5 was significantly (t = 6.2, df = 118, p < 0.001) greater than that of females 178.7 ± 40.2. Grossly, no lesions were seen o<strong>the</strong>r than trauma associated with capture. There was a significant relationship between fork length <strong>and</strong> weight (F = 1535, df = 119, R 2 = 0.93, p< 0.0001). With microscopy, protista were <strong>the</strong> most common organisms, which <strong>in</strong>fected 56 (47%) fish. Of <strong>the</strong>se, <strong>in</strong>fection was <strong>in</strong> <strong>the</strong> spleen (42 fish), kidney (4 fish), or both organs (10 fish). Protista were <strong>in</strong> well-def<strong>in</strong>ed multicellular aggregates (Fig. 1A,B), <strong>and</strong> were sometimes heavily <strong>in</strong>filtrated with melanized macrophages. Some appeared banana shaped with an eos<strong>in</strong>ophilic cytoplasm, while o<strong>the</strong>rs appeared to have multiple nuclei. They sta<strong>in</strong>ed weakly positive with Giemsa <strong>and</strong> were not associated with tissue necrosis. On electron microscopy, protista were <strong>in</strong>tracytoplasmic with<strong>in</strong> monocytes <strong>and</strong> appeared to displace <strong>the</strong> host nucleus (Fig. 1C). The organisms were sausage-shaped, <strong>and</strong> sometimes were grouped with<strong>in</strong> a membrane. Individual organisms had a dist<strong>in</strong>ct nucleus, conoid, rhoptries, amylopect<strong>in</strong> granules <strong>and</strong> micronemes (Fig. 1D). No hemoparasites were seen on smears of peripheral blood. Bacteria compatible <strong>in</strong> morphology with <strong>the</strong> <strong>epi<strong>the</strong>liocystis</strong>-<strong>like</strong> organism were seen <strong>in</strong> 26 (22%) fish. Of <strong>the</strong>se, <strong>in</strong>fection was <strong>in</strong> <strong>the</strong> kidney (23 fish), spleen (2 fish), or both organs (1 fish). On light microscopy, <strong>the</strong> <strong>epi<strong>the</strong>liocystis</strong>-<strong>like</strong> organism consisted of well-def<strong>in</strong>ed spherical aggregates of basophilic organisms that sta<strong>in</strong>ed negative with Gram sta<strong>in</strong> <strong>and</strong> positive with Gimenez <strong>and</strong> Giemsa. In putative early <strong><strong>in</strong>fections</strong>, organisms were amorphous <strong>and</strong> homogenous <strong>and</strong> were accompanied by a mild mononuclear response or no <strong>in</strong>flammation (Fig. 2A). In later <strong><strong>in</strong>fections</strong>, particularly <strong>in</strong> <strong>the</strong> kidney, <strong>epi<strong>the</strong>liocystis</strong>-<strong>like</strong> organisms became <strong>in</strong>filtrated with clumps of eos<strong>in</strong>ophilic material <strong>and</strong> were surrounded by a prom<strong>in</strong>ent capsule of collagen mixed with fibroblasts (Fig. 2B). On electron microscopy, <strong>epi<strong>the</strong>liocystis</strong>-<strong>like</strong> organisms had a thick capsule (Fig. 2C) surround<strong>in</strong>g a granular matrix conta<strong>in</strong><strong>in</strong>g mitochondria <strong>in</strong> various stages of degeneration (Fig. 2D). Deeper <strong>in</strong>to <strong>the</strong> organism, a th<strong>in</strong> <strong>in</strong>ner membrane (Fig. 2E) surrounded variably sized aggregates of membrane-bound spherical structures, rang<strong>in</strong>g <strong>in</strong> diameter from 1 to 2 µm. These structures conta<strong>in</strong>ed
Work et al.: Pathology of bluestripe snapper <strong>in</strong> Hawaii 61 Fig. 1. Lutjanus kasmira. (A) Coccidia <strong>in</strong> spleen. Note lack of <strong>in</strong>flammatory response. Scale bar = 50 µm. (B) Close-up of coccidia <strong>in</strong> (A) (arrows). Scale bar = 20 µm. (C) Coccidia with<strong>in</strong> macrophage (arrow). Note displacement of host nucleus (n). Scale bar = 1µm. (D) Coccidia (probable merozoite). Scale bar = 1 µm. n = nucleus, a = amylopect<strong>in</strong> granules, r = rhoptries, c = conoid, m = microneme, g = dense granule