Protozoal and epitheliocystis-like infections in the introduced
Protozoal and epitheliocystis-like infections in the introduced
Protozoal and epitheliocystis-like infections in the introduced
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
62<br />
Dis Aquat Org 57: 59–66, 2003<br />
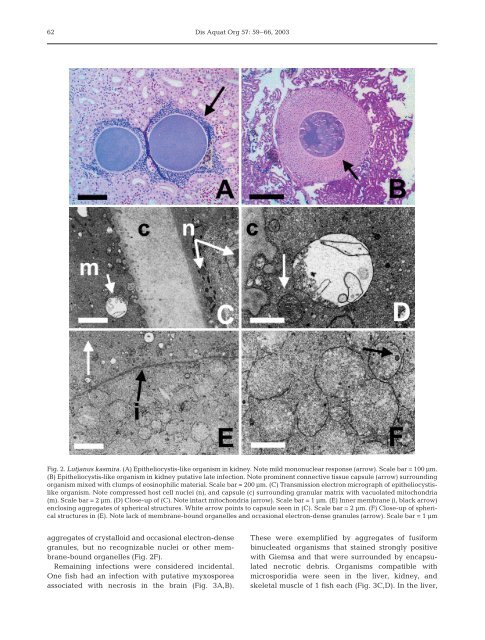
Fig. 2. Lutjanus kasmira. (A) Epi<strong>the</strong>liocystis-<strong>like</strong> organism <strong>in</strong> kidney. Note mild mononuclear response (arrow). Scale bar = 100 µm.<br />
(B) Epi<strong>the</strong>liocystis-<strong>like</strong> organism <strong>in</strong> kidney putative late <strong>in</strong>fection. Note prom<strong>in</strong>ent connective tissue capsule (arrow) surround<strong>in</strong>g<br />
organism mixed with clumps of eos<strong>in</strong>ophilic material. Scale bar = 200 µm. (C) Transmission electron micrograph of <strong>epi<strong>the</strong>liocystis</strong><strong>like</strong><br />
organism. Note compressed host cell nuclei (n), <strong>and</strong> capsule (c) surround<strong>in</strong>g granular matrix with vacuolated mitochondria<br />
(m). Scale bar = 2 µm. (D) Close-up of (C). Note <strong>in</strong>tact mitochondria (arrow). Scale bar = 1 µm. (E) Inner membrane (i, black arrow)<br />
enclos<strong>in</strong>g aggregates of spherical structures. White arrow po<strong>in</strong>ts to capsule seen <strong>in</strong> (C). Scale bar = 2 µm. (F) Close-up of spherical<br />
structures <strong>in</strong> (E). Note lack of membrane-bound organelles <strong>and</strong> occasional electron-dense granules (arrow). Scale bar = 1 µm<br />
aggregates of crystalloid <strong>and</strong> occasional electron-dense<br />
granules, but no recognizable nuclei or o<strong>the</strong>r membrane-bound<br />
organelles (Fig. 2F).<br />
Rema<strong>in</strong><strong>in</strong>g <strong><strong>in</strong>fections</strong> were considered <strong>in</strong>cidental.<br />
One fish had an <strong>in</strong>fection with putative myxosporea<br />
associated with necrosis <strong>in</strong> <strong>the</strong> bra<strong>in</strong> (Fig. 3A,B).<br />
These were exemplified by aggregates of fusiform<br />
b<strong>in</strong>ucleated organisms that sta<strong>in</strong>ed strongly positive<br />
with Giemsa <strong>and</strong> that were surrounded by encapsulated<br />
necrotic debris. Organisms compatible with<br />
microsporidia were seen <strong>in</strong> <strong>the</strong> liver, kidney, <strong>and</strong><br />
skeletal muscle of 1 fish each (Fig. 3C,D). In <strong>the</strong> liver,