Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
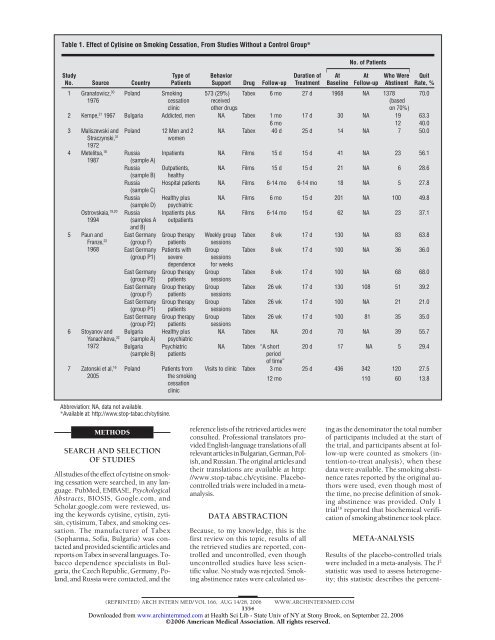
Table 1. Effect of <strong>Cytisine</strong> on <strong>Smoking</strong> <strong>Cessation</strong>, From Studies Without a Control Group*<br />
Study<br />
No. Source Country<br />
1 Granatowicz, 30 Poland<br />
1976<br />
Type of<br />
Patients<br />
<strong>Smoking</strong><br />
cessation<br />
clinic<br />
Behavior<br />
Support Drug Follow-up<br />
573 (29%)<br />
received<br />
other drugs<br />
Duration of<br />
Treatment<br />
At<br />
Baseline<br />
No. of Patients<br />
At<br />
Follow-up<br />
Who Were<br />
Abstinent<br />
Tabex 6 mo 27 d 1968 NA 1378<br />
(based<br />
on 70%)<br />
Quit<br />
Rate, %<br />
70.0<br />
2 Kempe, 21 1967 Bulgaria Addicted, men NA Tabex 1 mo 17 d 30 NA 19 63.3<br />
6 mo 12 40.0<br />
3 Maliszewski and Poland 12 Men and 2 NA Tabex 40 d 25 d 14 NA 7 50.0<br />
Straczynski, 31<br />
1972<br />
women<br />
4 Metelitsa, 18 Russia Inpatients NA Films 15 d 15 d 41 NA 23 56.1<br />
1987<br />
(sample A)<br />
Russia Outpatients, NA Films 15 d 15 d 21 NA 6 28.6<br />
(sample B) healthy<br />
Russia Hospital patients NA Films 6-14 mo 6-14 mo 18 NA 5 27.8<br />
(sample C)<br />
Russia<br />
(sample D)<br />
Healthy plus<br />
psychiatric<br />
NA Films 6 mo 15 d 201 NA 100 49.8<br />
Ostrovskaia, 19,20<br />
1994<br />
5 Paun and<br />
Franze, 22<br />
1968<br />
6 Stoyanov and<br />
Yanachkova, 32<br />
1972<br />
7 Zatonski et al, 16<br />
2005<br />
Russia<br />
(samples A<br />
and B)<br />
East Germany<br />
(group F)<br />
East Germany<br />
(group P1)<br />
East Germany<br />
(group P2)<br />
East Germany<br />
(group F)<br />
East Germany<br />
(group P1)<br />
East Germany<br />
(group P2)<br />
Bulgaria<br />
(sample A)<br />
Bulgaria<br />
(sample B)<br />
Poland<br />
Abbreviation: NA, data not available.<br />
*Available at: http://www.stop-tabac.ch/cytisine.<br />
Inpatients plus<br />
outpatients<br />
Group therapy<br />
patients<br />
Patients with<br />
severe<br />
dependence<br />
Group therapy<br />
patients<br />
Group therapy<br />
patients<br />
Group therapy<br />
patients<br />
Group therapy<br />
patients<br />
Healthy plus<br />
psychiatric<br />
Psychiatric<br />
patients<br />
Patients from<br />
the smoking<br />
cessation<br />
clinic<br />
NA Films 6-14 mo 15 d 62 NA 23 37.1<br />
Weekly group<br />
sessions<br />
Tabex 8 wk 17 d 130 NA 83 63.8<br />
Group Tabex 8 wk 17 d 100 NA 36 36.0<br />
sessions<br />
<strong>for</strong> weeks<br />
Group Tabex 8 wk 17 d 100 NA 68 68.0<br />
sessions<br />
Group Tabex 26 wk 17 d 130 108 51 39.2<br />
sessions<br />
Group Tabex 26 wk 17 d 100 NA 21 21.0<br />
sessions<br />
Group Tabex 26 wk 17 d 100 81 35 35.0<br />
sessions<br />
NA Tabex NA 20 d 70 NA 39 55.7<br />
NA Tabex “A short<br />
period<br />
of time”<br />
20 d 17 NA 5 29.4<br />
Visits to clinic Tabex 3 mo 25 d 436 342 120 27.5<br />
12 mo 110 60 13.8<br />
METHODS<br />
SEARCH AND SELECTION<br />
OF STUDIES<br />
Allstudiesoftheeffectofcytisineonsmoking<br />
cessation were searched, in any language.<br />
PubMed, EMBASE, Psychological<br />
Abstracts, BIOSIS, Google.com, and<br />
Scholar.google.com were reviewed, using<br />
the keywords cytisine, cytisin, zytisin,<br />
cytisinum, Tabex, and smoking cessation.<br />
The manufacturer of Tabex<br />
(Sopharma, Sofia, Bulgaria) was contacted<br />
and provided scientific articles and<br />
reports on Tabex in several languages. Tobacco<br />
dependence specialists in Bulgaria,<br />
the Czech Republic, Germany, Poland,<br />
and Russia were contacted, and the<br />
reference lists of the retrieved articles were<br />
consulted. Professional translators provided<br />
English-language translations of all<br />
relevantarticlesinBulgarian,German,Polish,<br />
and Russian. The original articles and<br />
their translations are available at http:<br />
//www.stop-tabac.ch/cytisine. Placebocontrolled<br />
trials were included in a metaanalysis.<br />
DATA ABSTRACTION<br />
Because, to my knowledge, this is the<br />
first review on this topic, results of all<br />
the retrieved studies are reported, controlled<br />
and uncontrolled, even though<br />
uncontrolled studies have less scientific<br />
value. No study was rejected. <strong>Smoking</strong><br />
abstinence rates were calculated using<br />
as the denominator the total number<br />
of participants included at the start of<br />
the trial, and participants absent at follow-up<br />
were counted as smokers (intention-to-treat<br />
analysis), when these<br />
data were available. The smoking abstinence<br />
rates reported by the original authors<br />
were used, even though most of<br />
the time, no precise definition of smoking<br />
abstinence was provided. Only 1<br />
trial 16 reported that biochemical verification<br />
of smoking abstinence took place.<br />
META-ANALYSIS<br />
Results of the placebo-controlled trials<br />
were included in a meta-analysis. The I 2<br />
statistic was used to assess heterogeneity;<br />
this statistic describes the percent-<br />
(REPRINTED) ARCH INTERN MED/ VOL 166, AUG 14/28, 2006 WWW.ARCHINTERNMED.COM<br />
1554<br />
Downloaded from www.archinternmed.com at Health Sci Lib - State Univ of NY at Stony Brook, on September 22, 2006<br />
©2006 American Medical Association. All rights reserved.