Annual 2005 - Hauptman Woodward Institute - University at Buffalo
Annual 2005 - Hauptman Woodward Institute - University at Buffalo
Annual 2005 - Hauptman Woodward Institute - University at Buffalo
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Pioneers of Science High School Educ<strong>at</strong>ional Outreach: Western New York has a long, proud and<br />
rich history of producing leaders in many branches of science. To salute some of these individuals and<br />
to focus local and n<strong>at</strong>ional <strong>at</strong>tention on the high caliber of scientific work th<strong>at</strong> is thriving here,<br />
<strong>Hauptman</strong>-<strong>Woodward</strong> hosts our Pioneers of Science Conference. The event is made up of two separ<strong>at</strong>e<br />
programs: one, a morning lecture and series of workshops for area high school science students and<br />
the second, a dinner and awards present<strong>at</strong>ion gala. The objective of the conference is to inspire high<br />
school students through the achievements and accomplishments of leading Western New York<br />
scientists. They have the opportunity to meet in small workshop sessions led by each of the honored<br />
scientists. The students also will learn of current and future research and career opportunities. The<br />
target audience for this conference is science-inclined high school students currently <strong>at</strong>tending Erie<br />
County public and priv<strong>at</strong>e schools. Approxim<strong>at</strong>ely 200 students particip<strong>at</strong>e in this program.<br />
A New Public Science-M<strong>at</strong>h High School: HWI is particip<strong>at</strong>ing in the development of a new high<br />
school th<strong>at</strong> will be part of the <strong>Buffalo</strong> Public School system where the concentr<strong>at</strong>ion for the students will be<br />
focused primarily on the sciences and m<strong>at</strong>hem<strong>at</strong>ics. The goal is for the school to open in the fall of 2006.<br />
Overall, HWI is str<strong>at</strong>egically moving into the next 50 years. We are in a new home, growing our HWI<br />
family with new scientists, technicians and support staff and we are continually expanding the boundaries<br />
into new arenas of science.<br />
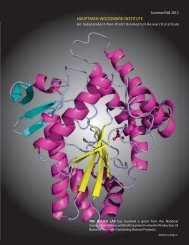
The Science <strong>at</strong> the <strong>Hauptman</strong>-<strong>Woodward</strong> Medical Research <strong>Institute</strong><br />
Structural biology is the study of the shapes of molecules, such as proteins,<br />
th<strong>at</strong> exist in the body. Each protein has a distinct shape and function and<br />
is very often involved in one or more diseases. Knowing the threedimensional<br />
structure of these proteins enables scientists to understand<br />
how they work and can allow them to develop new medic<strong>at</strong>ions to tre<strong>at</strong><br />
disease. At <strong>Hauptman</strong>-<strong>Woodward</strong>, our scientists specialize in structural<br />
biology and the use of x-ray crystallography.<br />
Wh<strong>at</strong> is the Process?<br />
The structural biology process is often complic<strong>at</strong>ed and time consuming,<br />
but each step is critical to understanding disease and to developing<br />
new ways to tre<strong>at</strong>, prevent, and possibly cure them. There are six<br />
distinct steps th<strong>at</strong> take place in the structural biology process <strong>at</strong> HWI.<br />
1.<br />
Protein production and purific<strong>at</strong>ion: In this stage, genes<br />
implic<strong>at</strong>ed in diseases are identified, isol<strong>at</strong>ed, cloned, and<br />
introduced into protein expression systems. Finally, these proteins<br />
are over-expressed in various systems to determine the highest<br />
level of protein production, and purified using high-pressure<br />
liquid chrom<strong>at</strong>ography to achieve gre<strong>at</strong>er than 99 percent purity.<br />
Breast cancer drug target<br />
2.<br />
Crystal growth: Once a purified protein is obtained, crystals are<br />
produced, using high-throughput crystalliz<strong>at</strong>ion, a technique<br />
cre<strong>at</strong>ed by our scientists th<strong>at</strong> performs 1536 experiments <strong>at</strong> a<br />
time using a robotic system, and which records the results in a<br />
d<strong>at</strong>abase for future access.<br />
3.<br />
4.<br />
X-ray diffraction: After crystals of adequ<strong>at</strong>e diffraction quality are<br />
obtained, they are mounted on a diffractometer or brought to a<br />
synchrotron facility where a unique diffraction p<strong>at</strong>tern is produced.<br />
Analysis of diffraction d<strong>at</strong>a: A computer analysis of the diffraction<br />
d<strong>at</strong>a is performed th<strong>at</strong> allows the loc<strong>at</strong>ion of <strong>at</strong>oms in the<br />
molecules to be determined. An electron density map is then<br />
cre<strong>at</strong>ed and viewed, using st<strong>at</strong>e-of-the-art computer graphics.<br />
Methods developed by Dr. <strong>Hauptman</strong> are used <strong>at</strong> this stage.<br />
AIDS-rel<strong>at</strong>ed research<br />
5.<br />
Analysis of molecular structure and function: Scientists are then<br />
able to view the three-dimensional structure th<strong>at</strong> has been<br />
determined to understand how structure guides biological function.<br />
Pioneer of Science honoree Edith Flanigen explains her research to local high school students <strong>at</strong> the<br />
Pioneers of Science student symposium.<br />
Structure-Based Drug Design: By studying the structure and<br />
function of molecules we can design new medic<strong>at</strong>ions th<strong>at</strong> are<br />
more selective and effective in the fight against diseases. The<br />
AIDS drugs th<strong>at</strong> have proved to be so successful in extending<br />
p<strong>at</strong>ients' lives were designed using the three-dimensional<br />
Protein essential for cell prolifer<strong>at</strong>ion<br />
and growth<br />
structure of the AIDS protease.<br />
12 13<br />
6.