BIOQUELL Clarus L2 (609K) - APEX Laboratory Equipment Company
BIOQUELL Clarus L2 (609K) - APEX Laboratory Equipment Company
BIOQUELL Clarus L2 (609K) - APEX Laboratory Equipment Company
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
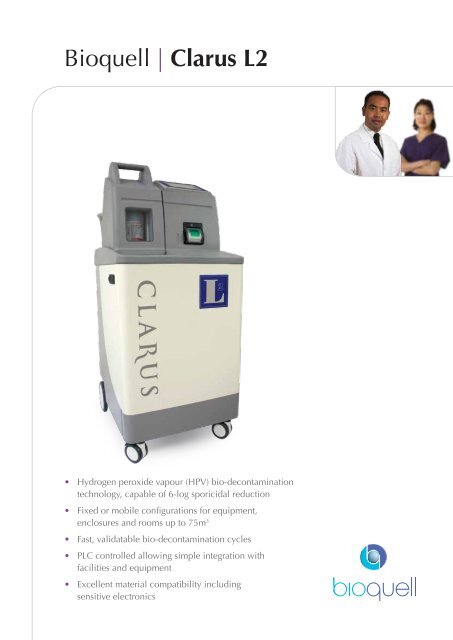
Bioquell | <strong>Clarus</strong> <strong>L2</strong><br />
• Hydrogen peroxide vapour (HPV) bio-decontamination<br />
technology, capable of 6-log sporicidal reduction<br />
• Fixed or mobile configurations for equipment,<br />
enclosures and rooms up to 75m 3<br />
• Fast, validatable bio-decontamination cycles<br />
• PLC controlled allowing simple integration with<br />
facilities and equipment<br />
• Excellent material compatibility including<br />
sensitive electronics
Bioquell <strong>Clarus</strong> <strong>L2</strong> multi-function HPV generator<br />
The Bioquell <strong>Clarus</strong> <strong>L2</strong> is a second generation hydrogen peroxide vapour<br />
(HPV) generator, replacing Bioquell’s widely used <strong>Clarus</strong> L HPV generator.<br />
The <strong>Clarus</strong> <strong>L2</strong> offers enhanced functionality and is able to bio-decontaminate<br />
both enclosures and rooms/laboratories up to 75m 3 . The <strong>Clarus</strong> <strong>L2</strong> has<br />
enhanced HPV delivery and control, as well as an improved user interface<br />
using touch screen controls.<br />
• Accurate measurement of the amount of hydrogen peroxide vaporised<br />
• Simple menu-driven user interface to set cycle parameters and access<br />
to bio-decontamination parameters<br />
• Graphical display of bio-decontamination cycle progress tracking critical<br />
parameters in real time<br />
• Communications port allows integration to building management system<br />
(BMS) or related systems and equipment<br />
• Data logging option allowing data export into Excel ® format<br />
• Multiple language options<br />
• Bioquell dual circuit technology for fast and efficient<br />
bio-decontamination cycles<br />
• No requirement for room dehumidification prior to cycles<br />
Operation<br />
Easy to clean and safe bottle loading mechanism with built-in drip<br />
tray and wipe clean mechanism. Housing accepts Bioquell 0.5 ltr<br />
and 1 ltr hydrogen peroxide solution allowing multiple cycles<br />
without intervention.<br />
A thermal printer allows for hard copy data recording. Each stage of the<br />
cycle is recorded and printed.
Bioquell hydrogen peroxide solution<br />
• Bioquell hydrogen peroxide is specified and<br />
approved for use with Bioquell HPV generators<br />
• Approved for use as a biocide by regulatory<br />
bodies (BPD, EPA)<br />
• Selected for optimal biocidal effectiveness<br />
Optional accessories include:<br />
• Remote control with room gassing distribution facility<br />
• Sensor pack - integrated hydrogen peroxide<br />
vapour sensor<br />
• Data logging and archive with USB interface<br />
• Hand-held HPV detector<br />
• External aeration/catalyst unit to reduce cycle<br />
times further<br />
Validation and service<br />
Fully validatable to comply with requirements of<br />
GMP using GAMP compliant software. Bioquell<br />
provides complete service including:<br />
• Installation and integration<br />
• Preventative maintenance plans<br />
• Extensive field-based service<br />
engineer network<br />
• IQ/OQ/PQ documentation<br />
and execution<br />
• Bio-decontamination cycle<br />
development services<br />
• Ongoing MQ and RQ support*<br />
*Provided as part of a preventative maintenance plan
Materials of external construction<br />
Temperature<br />
Operating 15°C to 35°C<br />
Storage -15°C to 50°C<br />
Relative humidity<br />
Operating<br />
Storage<br />
Top moulding – polypropylene<br />
Side panels – polyurethane coated aluminium<br />
75% RH maximum<br />
95% RH (non-condensing)<br />
Power requirement AC 220 – 240V 50 Hz (Euro) 6 amps<br />
AC 110 – 120V 60 Hz (USA) 12 amps<br />
AC 90 – 110V 50/60 Hz (Japan) 12 amps<br />
Power consumption<br />
Power supply<br />
Operating position<br />
Pollution degree environment 2<br />
Dimensions (WxHxD)<br />
Weight<br />
Maximum H 2<br />
O 2<br />
liquid volume<br />
H 2<br />
O 2<br />
liquid concentration<br />
Hose pipes<br />
Gas hose connections<br />
Control system<br />
Airflow rate<br />
Gassing<br />
Conditioning/aeration<br />
Temperature of supply gas at outlet<br />
H 2<br />
O 2<br />
liquid injection rate<br />
Refrigeration system off coil temperature<br />
Condensate removal<br />
1300W<br />
Installation category II<br />
Upright only<br />
515 x 1140 x 705mm<br />
80 kg<br />
1 litre<br />
30-35% w/w Bioquell hydrogen peroxide (stored at 2-8°C)<br />
1.5 inch nominal bore<br />
male/female<br />
Camlock fittings 1.5 inch<br />
Siemens ® S7 PLC<br />
(GAMP compliant software)<br />
Controlled at 25 m 3 /hr<br />
Controlled at 25 m 3 /hr at 750Pa external pressure<br />
75°C maximum<br />
Inlet air HEPA filter efficacy 99.997%<br />
Gassing & dehum circuit HEPA filter efficacy 99.997%<br />
Pressurisation system flow rate<br />
Pressurisation system range<br />
Noise<br />
1.5 to 6 g/min (gassing); 0 or 1.5 to 6 g/min (dwell)<br />
Approx 2°C (from 20°C return)<br />
Evaporation to atmosphere<br />
100 l/min (max)<br />
+200 to -200 Pa<br />
Less than 60 dBA at 1 metre (free field)<br />
Features in brief<br />
• Dual circuit technology for optimum cycle times<br />
• Closed circuit operation<br />
• Siemens ® colour touch screen<br />
• GAMP compliant software<br />
• Compact and highly portable design<br />
• Easily cleanable external surface<br />
• Volt-free contacts for external signals<br />
• Integral thermal paper printer<br />
• Remote start/stop<br />
• Simple connection, flexible supply/return hoses<br />
and pressure monitoring<br />
• Supply return hoses (2mtrs long)<br />
• Cycle edit and store facility for new applications<br />
• Effective against a wide range of micro-organisms<br />
• Hydrogen peroxide vapour gas sensor<br />
• Utilises 30-35% w/w Bioquell hydrogen peroxide<br />
solution in 0.5 ltr or 1 ltr<br />
• Maximum enclosure bio-decontamination 75m 3
Hydrogen peroxide vapour (HPV) technology<br />
HPV concentration<br />
1<br />
2<br />
3<br />
Inactivation of micro-organisms<br />
t=0*<br />
GASSING DWELL AERATION<br />
TIME<br />
* Conditioning phase not shown (vaporiser reaches temperature)<br />
Note: “features” on graph change e.g. depending on size of<br />
chamber (& related temperature effect)<br />
4<br />
5<br />
Schematic of Bioquell HPV concentration/time graph<br />
1. HPV introduced – initial rapid increase in HPV concentration<br />
2. HPV saturation/“dew point” achieved – onset of rapid bio-decontamination<br />
3. HPV gassing plateau – sustained micro-condensation effecting bio-deactivation<br />
4. Dwell period (contact time)<br />
5. Aeration – removal of HPV from the atmosphere typically by catalytic<br />
conversion to water vapour and oxygen, leaving no residues<br />
Schematic of dew point and Bioquell kill dynamics<br />
1. Injection of HPV into the enclosure starts (t=0)<br />
2. Only slight decline in biological indicator population prior to dew point<br />
3. Rapid micro-biological kill occurs immediately after saturation/“dew point” is achieved –<br />
process optimised to achieve reliable 6-log sporicidal reduction on all exposed surfaces<br />
4. Onset of micro-condensation correlates with rapid bio-deactivation<br />
5. Bio-deactivation achieved via micro-condensation (invisible layer) of hydrogen<br />
peroxide (2-6µm)<br />
* Typical BI: Tyvek ® pouched 6-log<br />
Geobacillus stearothermophilus spores<br />
Key benefits of HPV<br />
• Excellent material compatibility<br />
including sensitive electronics<br />
• Environmentally friendly -<br />
“residue-free” (breaks down to<br />
water vapour and oxygen)<br />
• Effective against a wide range<br />
of micro-organisms<br />
• High level disinfection achieved<br />
(6-log sporicidal reduction)<br />
HPV applications<br />
• GMP cleanrooms<br />
• Aseptic processing<br />
• Biologics<br />
• Biomedical<br />
• Biosafety/security<br />
• Material transfer chambers<br />
• Ambient decontamination of<br />
autoclaves/freeze driers<br />
• Robotic enclosures<br />
• Safety cabinets/isolators<br />
Key features and benefits of the <strong>Clarus</strong> <strong>L2</strong> HPV generator<br />
FEATURES<br />
Optimised cycle performance producing fast and effective<br />
bio-decontamination<br />
Flexible multi-use generator<br />
Critical condition monitoring with alarm set points for<br />
‘Alert’ or ‘Action’<br />
Advanced software and Siemens ® PLC controls for ease of<br />
use and operation<br />
BENEFITS<br />
Minimal process downtime – no need for preconditioning,<br />
process operates with ambient environment<br />
Can bio-decontaminate rooms and enclosures up to 75m 3 .<br />
Small footprint requiring minimal floor space<br />
Compatible with GMP and GLP requirements<br />
Simple integration into a building management system<br />
(BMS) or the <strong>Clarus</strong> PORT rapid decontamination<br />
transfer chamber
Other unique bio-decontamination<br />
solutions from Bioquell<br />
Bioquell RBDS rapid facility bio-decontamination service<br />
A unique bio-decontamination service from Bioquell<br />
for zones, rooms and buildings:<br />
• “Infinitely scalable” – no room or building too large<br />
• Emergency, scheduled, contract or new-build<br />
• Rapid deployment by highly trained Bioquell technicians<br />
Bioquell Material Transfer Solutions<br />
Bio-decontamination chambers:<br />
• Material transfer incorporating HPV bio-decontamination<br />
at ambient temperature<br />
• Rapid bio-decontamination leading to fast material transfer<br />
• Inflatable door seals for reliable, safe operation<br />
Bioquell Z HPV generator<br />
State-of-the-art bio-decontamination equipment offering:<br />
• Large scale room/zone bio-decontamination<br />
• Parametric cycle control<br />
• Simple to operate “plug and play”<br />
Disclaimer: Bioquell UK Ltd or its affiliates, distributors, agents or licensees<br />
(together “Bioquell”) reserves the right to change the specification of<br />
the Bioquell <strong>Clarus</strong> <strong>L2</strong> without notice. In addition, Bioquell makes no<br />
claims as to the guarantee of sterilisation using these products; however,<br />
Bioquell recommends that customers ensure that the requisite level of<br />
bio-decontamination is achieved using standard biological indicators such as<br />
6-log Geobacillus stearothermophilus spores; and the Bioquell <strong>Clarus</strong> <strong>L2</strong>, subject<br />
to appropriate cycle development, is designed to be able to provide such levels of<br />
bio-deactivation.<br />
Your local distributor:<br />
<strong>Clarus</strong> / Bioquell / RBDS are registered trade marks of Bioquell UK Ltd.<br />
© Bioquell UK Ltd (2010). All rights reserved.<br />
For further information on Bioquell’s technology:<br />
E: info@bioquell.com<br />
W: www.bioquell.com<br />
Bioquell UK Ltd<br />
T: +44 (0)1264 835 835<br />
Bioquell Ireland<br />
T: +353 (0)61 603 622<br />
Bioquell Inc<br />
T: +1 (215) 682 0225<br />
Bioquell SAS<br />
T: +33 (0)1 43 78 15 94<br />
Bioquell Asia Pacific Pte Ltd<br />
T: +65 6592 5145<br />
127/10