Winter 2013 - Hauptman Woodward Institute
Winter 2013 - Hauptman Woodward Institute
Winter 2013 - Hauptman Woodward Institute
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
The UMLAND and SCHULTZ LABS Receive a DOD Grant<br />
Drs. Timothy C. Umland and L. Wayne Schultz have received a<br />
grant from the Department of Defense to study the transmission of<br />
emergent diseases from animals to humans.<br />
Zoonoses are infectious diseases that are transmitted between<br />
species. Typically, zoonotic pathogens are viruses, and they spread<br />
from other vertebrate species to humans either by direct contact<br />
with the animal or its excretions or via an arthropod (e.g. insect)<br />
vector. The appearance of a pathogen in a new host species is<br />
called “disease emergence”.<br />
Zoonotic viruses are responsible for many of the most serious<br />
infectious diseases confronting modern medicine. Examples include<br />
avian influenza (H5N1), West Nile virus, several forms of encephalitis,<br />
SARS (severe acute respiratory syndrome), Ebola and AIDS, to<br />
name a few. The influenza virus alone accounted for several severe<br />
pandemics in the 20th century including the “Spanish flu” of 1918,<br />
the “Asian flu” of 1957, and the “Hong Kong flu” of 1968.<br />
In animals and humans, the innate immune system provides the<br />
main defense against novel viral attack. Host cells release proteins<br />
called interferons in response to the presence of viruses or other<br />
pathogens. “Interferons” (named after their ability to “interfere”<br />
with viral replication within host cells) facilitate communication<br />
between cells, triggering the immune system to eradicate the<br />
microbial invaders.<br />
In retaliation, viruses have evolved mechanisms for evading the<br />
innate immune responses of their established (donor) host species.<br />
Successful expansion to a new (recipient) host species would require<br />
the ability to control the immune response in that species as well.<br />
It is known that certain viral proteins impair the interferon activation<br />
response through molecular interactions with specific host proteins,<br />
thereby disrupting the intracellular signaling pathways of the host<br />
species. Presumably, the ability of viral proteins to form similar<br />
interactions with proteins in a potential recipient species would lower<br />
the barrier to species jumping. However, little is known about the<br />
actual conservation of virus-host protein interactions in donor and<br />
recipient species or the extent of adaptation necessary for viruses<br />
to form comparable complexes with similar proteins in new hosts.<br />
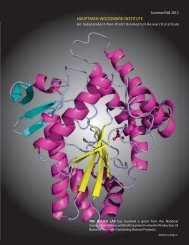
The Umland/Schultz project. HWI Research Scientists Tim Umland<br />
and Wayne Schultz have recently received a 3-year (with an<br />
option to extend to a total of 5 years), $1.1M grant from the<br />
Department of Defense to study the relationship between conserved<br />
protein-protein interactions and successful host-range expansion.<br />
Drs. Umland and Schultz will identify instances of conserved,<br />
cross-species, virus-host protein interactions involving viruses with<br />
histories of host jumps, especially to humans. In particular, they will<br />
examine host proteins involved in the interferon response pathway<br />
for such interactions.<br />
The HWI scientists will define and characterize the virus-host,<br />
protein-protein interfaces by using small-angle x-ray scattering<br />
(SAXS) to create structural models. The amino acid residues<br />
involved in forming the protein interfaces can then be identified.<br />
Mutagenesis studies will be used to verify critical residues that must<br />
be conserved for range expansion to occur.<br />
Potential impact. The public is at risk from emerging infectious<br />
diseases. This danger is particularly high for diseases that infect<br />
animals of economic importance or common wildlife where<br />
human contact and consumption create avenues of exposure. This<br />
HWI-based investigation will provide knowledge of the relative<br />
ease with which a given virus might adapt to a new species,<br />
particularly humans. It will also provide specific molecular details<br />
about common viral-host protein interactions that will be useful in<br />
the design of potential broad-spectrum antiviral therapeutic agents.<br />
New Collaborations for HWI Scientists<br />
The successful pursuit of a scientific research project often<br />
requires the complementary expertise of two or more scientists or<br />
laboratories. At HWI, researchers focus on using x-ray crystallography<br />
to determine the three-dimensional structures of<br />
molecules. Since crystallography requires specialized training,<br />
and the information it reveals is vital to many biologically related<br />
studies, HWI scientists are often asked to collaborate on a variety<br />
of studies. In fact, one HWI scientist recently received funding as<br />
a subcontractor on a project originating in UB’s School of Medicine<br />
and Biomedical Sciences, and two other HWI scientists<br />
recently co-authored research results with a UB collaborator.<br />
Designing Anti-HPV Drugs<br />
Human Papilloma Virus (HPV) is the cause of several types of<br />
cancer. Although currently available vaccines protect against<br />
the most common HPV infections, this virus has more than 180<br />
known variants or subtypes, and only four of these are<br />
covered by the vaccines.<br />
However, Dr. Thomas Melendy, a UB microbiologist, recently<br />
identified a protein (known as E1) that occurs in all HPV subtypes<br />
and might provide a basis for the first<br />
broad-range HPV drug target. He has<br />
been awarded a $1.65 million grant<br />
by the National <strong>Institute</strong>s of Health to<br />
try to develop such a drug.<br />
Drs. Vivian Cody and<br />
Thomas Melendy<br />
The E1 protein is encoded by the viral<br />
genome and interacts with human<br />
proteins involved in DNA synthesis. As<br />
a result, the human DNA synthetic<br />
machinery is “reprogrammed” to<br />
synthesize HPV genomes. Dr. Melendy<br />
is particularly interested in the interaction<br />
between E1 and the human<br />
enzyme, Topoisomerase I (Topol). He<br />
has identified the specific amino acids responsible for this interaction<br />
and has found them to be conserved across all HPV subtypes.<br />
Dr. Melendy’s new NIH grant funds efforts to analyze the E1-Topol<br />
interaction and to design small molecules that will interfere with this<br />
interaction. As part of this effort, Dr. Vivian Cody, a principal<br />
research scientist at HWI, will attempt to co-crystallize E1 and Topol<br />
and to visualize their interactions at the molecular level. Information<br />
derived from her crystallographic studies will be used to fine-tune<br />
the small-molecule inhibitors that show potential as anti-HPV drugs.<br />
continued from page 2<br />
Identifying New Drug Targets for<br />
Drug-Resistant Bacteria<br />
Acinetobacter baumannii is a bacterial species<br />
that is responsible for a growing number of<br />
hospital-acquired infections. It is difficult to<br />
eradicate with common disinfectants, and<br />
many strains are resistant to most, if not all,<br />
anti-microbial drugs. It generally doesn’t affect<br />
healthy people, but it poses an increasing<br />
danger for people who have had surgeries,<br />
soldiers with battlefield injuries, and the elderly.<br />
Recently, Thomas A. Russo, MD, UB’s Departments<br />
of Medicine, Microbiology and Immunology,<br />
and his HWI colleagues, Timothy C.<br />
Umland and L. Wayne Schultz, have<br />
succeeded in identifying several potential drug<br />
targets that may provide new ways for combatting<br />
A. baumannii. Their work suggests that the<br />
usual methods of studying bacteria in rich<br />
laboratory media may not be the best way to<br />
find new antimicrobial drug targets.<br />
The UB/HWI collaborators recognized that<br />
typical laboratory conditions are different<br />
from the environment of the human body<br />
where nutrients the bacteria need may be in<br />
short supply and immune responses are<br />
present. By growing the bacteria in human<br />
ascites, a peritoneal fluid present in a variety<br />
of pathological conditions, they discovered a<br />
novel, previously unrecognized set of genes<br />
essential for the growth of A. baumannii. The<br />
proteins encoded by these genes represent<br />
potential new drug targets since inhibition of<br />
the respective functions of these proteins will<br />
halt bacterial growth and reduce bacterial<br />
survivability within a human host.<br />
The researchers are now pursuing drug<br />
discovery efforts focused on the newly identified<br />
targets. Their research is being supported<br />
by grants from UB, the US Army, and the<br />
Department of Veterans Affairs.<br />
Dr. Eaton Lattman, CEO’s Column<br />
I recently received from one of <strong>Hauptman</strong>-<strong>Woodward</strong>'s<br />
most long-standing friends and supporters a thoughtful<br />
letter that discussed our research programs. It said in<br />
part "... A factor of importance is not just whether the<br />
research is so-called basic, applied or perhaps considered<br />
translational, but whether it is good, excellent, or<br />
outstanding. ... but the fact that the <strong>Institute</strong> personnel<br />
have been able to support their basic research for 50<br />
years suggests that the quality and productivity of investigations<br />
overall have been meritorious. ... In recent<br />
years there has been somewhat more diversity the<br />
sources from which support has been sought."<br />
This letter was implicitly asking us what we are doing to stay outstanding, and<br />
how we are finding new sources of support in the very difficult grant climate<br />
that is causing great distress to many independent institutions like ours. Let me<br />
share with you some of the comments (slightly expanded) that I made in my<br />
response to our friend.<br />
Dear friend,<br />
Thank you both for your generous support, and for your thoughtful commentary on<br />
HWI research. I agree very much with what you said, and I would like to respond<br />
with a brief picture of how I see our future.<br />
To be successful, a small, research <strong>Institute</strong> like <strong>Hauptman</strong>-<strong>Woodward</strong> has to do<br />
something supremely well. We have had Herb's Nobel Prize work in crystallography;<br />
we have had George DeTitta's creation of one of the world's first automated,<br />
high-throughput crystallization laboratories. What do we do for an encore? We<br />
have two directions in which I think we could attain the same kind of unique excellence<br />
that characterized the work of Herb and George. One of these has to do with<br />
the large NSF grant for which we are in the finals. There are 11 applicants left in the<br />
pool, and the NSF will fund five or six. This is a multi-institutional effort to make use<br />
of the new x-ray laser constructed at Stanford for structural biology. It will revolutionize<br />
the field in the same way that synchrotron x-ray sources have done in the past.<br />
This grant would bring many benefits, both scientific and institutional.<br />
• The x-ray laser will allow us to use crystals a thousand times smaller than the ones<br />
we use today. Nanocrystals will bring a whole new universe of proteins into the realm<br />
of crystallography, and will also provide a new technology frontier that will bring our<br />
high-throughput crystallization laboratory to a position of world leadership again.<br />
• The grant is very prestigious, and will re-burnish HWI’s worldwide reputation.<br />
• NSF regulations require that a degree-granting institution submit this type of<br />
grant, and so our proposal was actually sponsored by UB. The grant would<br />
provide for a much closer connection between the our two organizations.<br />
The second of our new directions involves setting up a structure-based drug<br />
design facility that will nucleate a wide variety of drug development projects at<br />
UB and among local biotech companies, and will provide a new academic<br />
paradigm for drug development. The idea is to mate the clinical and genetic<br />
knowledge of a disease developed by more medically oriented investigators<br />
with the deep molecular insights of HWI scientists.<br />
These two directions represent only a part of the new opportunities that we are addressing.<br />
I hope that you will feel excited and encouraged about the adventures that lie before<br />
us, and about the potential for new coming decades of excellence for HWI.<br />
Dr. E. Lattman<br />
continued on page 3<br />
Drs. L. Wayne Schultz, Thomas A. Russo and Timothy C. Umland<br />
2 3